This section addresses a series of important topics which impact the outcomes of people with CKD. Specifically CVD, interpretation of tests for CVD, infections, vaccinations and hospitalizations, medications, and patient safety are addressed. This section is designed to help the practitioner appreciate the specific nuances related to testing and drug administration in people with CKD and to alert all clinicians to the fact that many common tests may not be as useful in people with CKD to diagnose or evaluate therapies. It was beyond the scope of the guideline and evidence review to thoroughly evaluate all tests with respect to sensitivity and specificity in different groups of people with CKD but future research may consider this as a useful direction.
4.1 CKD and CVD
Population-based studies have demonstrated an increased risk of death and cardiovascular mortality as GFR falls below 60 ml/min/1.73 m2 or when albumin is detected on urinalysis. This is not explained by an increase in traditional risk factors. There are CKD-specific risk factors associated with more advanced CKD which drive the high rates of mortality and morbidity even at young ages. People with CKD are more likely to experience a cardiovascular event than to progress to ESRD, have a worse prognosis with higher mortality after acute myocardial infarction (MI), and have a higher risk of recurrent MI, heart failure and sudden cardiac death. Management of modifiable cardiovascular risk factors, such as improved BP and diabetes control, also reduces CKD progression.
4.1.1: We recommend that all people with CKD be considered at increased risk for cardiovascular disease. (1A)
RATIONALE
This statement is worded in this way to reflect the strong and independent associations between GFR and albuminuria categories and risk of CVD in people with CKD and applies to both adult and pediatric populations.
Evidence Base
Large cohort studies have demonstrated the strong and independent associations between CVD (acute coronary syndrome [ACS], stroke, heart failure and sudden cardiac death) and CKD by category of eGFR, after adjusting for known CVD risk factors, history of CVD events, and proteinuria. In those with an eGFR of 45-59 ml/min/1.73 m2, risk is increased by 43% and in those with eGFR below 15 ml/min/1.73 m2, risk is increased by 343%.58 Although people with GFR category G5 (GFR<15 ml/min/1.73 m2) are at the highest risk of a CVD event, there will be more events in people with GFR categories G3a-G3b (GFR 30–59 ml/min/1.73 m2) because of the much higher prevalence at these categories.420 These events occur at a younger age in people with CKD suggesting that CKD promotes CVD at an accelerated rate.421 The prognosis after an acute event is related to level of GFR with a significant rise in mortality when eGFR falls below 45 ml/min/1.73 m2.422, 423, 424
Albuminuria is associated with duration and severity of hypertension; an adverse lipid profile with higher levels of total cholesterol, triglycerides and lipoprotein(a) and low HDL-C levels;425 and abnormalities of coagulation. The presence of higher levels of proteinuria increases the risk of mortality and MI independently of level of eGFR.426 Many studies have demonstrated low levels of urinary albumin to be associated with the increased risk of CVD in people with diabetes independent of renal function; however population studies of non-diabetic individuals have confirmed that even small amounts of albuminuria are associated with increased CVD risk. In the Third Copenhagen study, in people with microalbuminuria, risk of coronary heart disease was increased independently of age, sex, renal function, diabetes, hypertension, and plasma lipids.427 The Chronic Kidney Disease Prognosis Consortium demonstrated that in general practice cohorts there was an increase in cardiovascular mortality when ACR is higher than 30 mg/g (3 mg/mmol).4 Analysis of data from the Heart Outcomes Prevention Evaluation (HOPE) study demonstrated that any degree of albuminuria is a risk factor for cardiovascular events in individuals with or without diabetes.428 The lack of a threshold of albuminuria for cardiac risk was also confirmed in the HUNT 2 Study429 and the Losartan Intervention For Endpoint Reduction in Hypertension (LIFE) study in patients with LVH.430 Albuminuria and low eGFR were synergistic cardiovascular mortality risk factors in the HUNT 2 study and using both ACR and eGFR improved cardiovascular risk stratification at all age levels, but particularly in persons 70 years and over.429
In the MDRD Study cohort of patients with GFR categories G3a-G4 (GFR 15–59 ml/min/1.73 m2), cystatin C level was strongly associated with all-cause and cardiovascular mortality particularly in the elderly.431 Analysis of data from MESA and the Cardiovascular Health Study (CHS) demonstrated that the worst prognosis of CVD, heart failure, and progressive CKD was found in those subjects with CKD diagnosed using the cystatin C-based equation.432
LVH is common with CKD, is known to reflect target organ damage, and is associated with increased cardiovascular mortality in CKD.360 It is also important to consider the role of CKD-specific risk factors particularly in patients with more severe CKD (GFR <30 ml/min/1.73 m2) and correct them where possible. Anemia has been thought to have a particular role in the early development of CVD in people with CKD. Although treatment of anemia is associated with improved well-being and greater exercise capacity, the results of several RCTs of anemia correction have suggested that complete correction of anemia is not advisable in people with lower GFR (<60 ml/min/1.73 m2). A meta-analysis of 9 RCTs comparing different target Hb levels suggests an increase in mortality and worse BP control with the higher treatment targets, independent of the GFR category.433, 434 Those patients with LVH at baseline, a sign of target organ damage and which was present in 47% of the Cardiovascular Risk Reduction by Early Anemia Treatment with Epoetin Beta Trial (CREATE) cohort, had significantly worse cardiovascular outcomes when treated to the higher targets.435
Abnormalities of mineral metabolism with low 1,25-dihydroxyvitamin D and PTH were found to occur early in CKD in the Study for the Evaluation of Early Kidney disease [SEEK] study, however serum calcium and phosphate were usually normal until eGFR fell below 40 ml/min/1.73 m2.367 An association between elevated serum phosphate and cardiovascular mortality was demonstrated in a prospective study of people with CKD and this was also thought to be associated with low vitamin D levels.436
Pediatric Considerations
While the majority of data for, and evidence of, the high risk of cardiovascular morbidity and mortality in the pediatric renal population stems from the ESRD (dialysis and transplant) populations,437, 438, 439, 440 there is recognition that many if not all of the factors responsible for such outcomes will develop much earlier in this course of CKD. In fact, the current American Heart Association's guidelines for reduction of cardiovascular risk in pediatrics stratified the child with CKD to the highest risk category.441
Based on data from the CKiD trial, Wilson et al.442 demonstrated extremely high prevalence rates for four traditional cardiovascular risk factors (hypertension, dyslipidemia, abnormal glucose metabolism, and obesity) in a subgroup of this cohort of 586 children with iohexol GFR proven mild to moderate CKD. In this cross-sectional analysis of 250 children who had data on all variables of interest, median GFR of 45.2 ml/min/1.73 m2 (IQR 34.6-58.2), the prevalence of hypertension was 46%, dyslipidemia was 44%, abnormal glucose metabolism was present in 21%, and obesity in 15%. Hypertriglyceridemia was common, seen in 33%. In aggregate, only 26% of the cohort had no risk factors present, 39% had at least one and 22% had two, 11% had three and 2% had all four risk factors present. The prevalence of all these factors remained elevated even when the population was restricted to those defined as lean, BMI <85th. In this group, the presence of 2 or 3 cardiovascular risk factors was still seen in 20% and 2%, respectively. Within the lean group 12% demonstrated abnormal glucose metabolism. Finally, multivariate regression analysis of their data demonstrated that glomerular disease as a cause of CKD and the presence of nephrotic range proteinuria were associated with much higher odds of having cardiovascular risk factors, 1.96 (95% CI 1.04-3.72) and 2.04 (95% CI 0.94-4.43), respectively.
It is also well-recognized that carotid intimal media thickness and pulse wave velocity changes, as potential surrogate measures for vascular damage in children, can be increased in in children with CKD.443, 444
For further detailed discussions of the pathophysiology and complexity of CVD in the pediatric CKD population, two recent review papers from Mitsnefes445 and Shroff et al.446 are suggested. Both papers identify and discuss the pathophysiology of both traditional and CKD-specific cardiovascular risk factors as seen in this population.
4.1.2: We recommend that the level of care for ischemic heart disease offered to people with CKD should not be prejudiced by their CKD. (1A)
4.1.3: We suggest that adults with CKD at risk for atherosclerotic events be offered treatment with antiplatelet agents unless there is an increased bleeding risk that needs to be balanced against the possible cardiovascular benefits. (2B)
RATIONALE
These statements are worded in this way to reflect the growing body of evidence that suggests that treatment of traditional risk factors in CKD patients is of benefit, and that attempts to modify or preclude usual investigations and therapies are not necessary in CKD patients. Inasmuch as CKD populations have been understudied, but likely inadvertently included in larger population studies, the rationale for risk factor modification is similar to that in the general population. Modification of risk factors includes:
Smoking cessation
Exercise
Weight reduction to optimal targets
Lipid modification recognizing that the risk reduction associated with statin therapy in adults with CKD is relatively constant across a broad range of baseline low-density lipoprotein cholesterol (LDL-C) levels
Optimal diabetes control HbA1C <7% (53 mmol/mol)
Optimal BP control to <140/90 mm Hg or <130/80 mm Hg in those with CKD and depending on the degree of proteinuria (see Recommendations 3.1.4 and 3.1.5)
Aspirin is indicated for secondary prevention but not primary prevention
Correction of anemia to individualized targets (see KDIGO Clinical Practice Guideline for Anemia in CKD11)
Evidence Base
The maintenance of cardiovascular health ultimately impacts ‘kidney health' given the links between cardiac function, renal perfusion, atherosclerosis, and glomerulosclerois. The details of this complex biology are beyond the scope of these guidelines, but can be found in key references below.
There is evidence in the general population that smoking cessation is associated with a reduction in cardiovascular risk. In CKD there is evidence that smoking is associated with progression of renal disease, though no specific data exist that support cessation of smoking and delay of progression.447
People with CKD have reduced levels of muscle strength and aerobic activity. In those not on dialysis, exercise training is shown to improve functional capacity and BP.448
Weight loss in obese CKD patients can reduce rate of decline, proteinuria, and BP.354
The Study of Heart and Renal Protection (SHARP)449 is the largest RCT in people with CKD to date. The results demonstrate that a lipid-lowering strategy which included fixed dose simvastatin and ezetimibe resulted in a 17% reduction in atherosclerotic events, as compared to placebo. The cohort enrolled included those with eGFR under 60 ml/min/1.73 m2, age greater than 40 years, and was an international study including more than 9000 subjects from around the globe. The lipid-lowering strategy was effective and safe. Lipid targets have not been established specifically for people with CKD. Treatment strategies should be implemented in accordance with current recommendations for high-risk populations.
At the time of this writing, the KDIGO Clinical Practice Guideline for Lipid Management in Chronic Kidney Disease was under preparation for public review. In brief, the key aspects of the draft recommendations include treating those at high risk for atherosclerotic disease with lipid-lowering therapies, regardless of LDL levels, in those 50 years of age and above. Since this Guideline has not yet been finalized, interested readers should refer to the final document when it is formally released in 2013.
The benefits of aspirin in people with CKD and hypertension was demonstrated in a post hoc analysis of the Hypertension Optimal Treatment (HOT) trial.450 Jardine et al. reported that among every 1000 persons with eGFR <45 ml/min/1.73 m2 treated for 3.8 years, 76 major cardiovascular events and 54 all-cause deaths will be prevented while 27 excess major bleeds will occur. They concluded that an increased risk of major bleeding appears to be outweighed by the substantial benefits. Clopidogrel is used as an alternative to aspirin but CKD has been shown to be associated with an increase in platelet reactivity and there is resistance to clopidogrel in people with CKD, diabetes, and CVD.451 The Clopidogrel for Reduction of Events During Observation (CREDO) trial concluded that clopidogrel in mild or moderate CKD may not have the same beneficial effect as it does in people without CKD. Subjects with normal renal function who received 1 year of clopidogrel had a marked reduction in death, MI, or stroke compared with those who received placebo (10.4% versus 4.4%, P <0.001), whereas those with mild and moderate CKD did not have a significant difference in outcomes with clopidogrel therapy versus placebo (mild: 12.8% versus 10.3%, P = 0.30; moderate: 13.1% versus 17.8%, P = 0.24). Clopidogrel use was associated with an increased RR of major or minor bleeding, but this increased risk was not different based on renal function.452
International Relevance
Although it is clear and mentioned in many guidelines that CKD is associated with an increase in CVD risk, it is not included in many assessment tools and there is a deficiency in ethnicity and regional specific tools.
The assessment tool from US Third Report of the National Cholesterol Education Program (NCEP III) does not include CKD. The Fourth Joint Task Force of the European Society of Cardiology suggest that both an eGFR of less than 60 ml/min/1.73 m2 and albuminuria increase risk but do not quantify the risk or include CKD in their Systematic COronary Risk Evaluation (SCORE) assessment tool.453 In the UK, the QRISK®2 online tool includes CKD as ‘yes' or ‘no' and therefore does not allow for level of eGFR or proteinuria.454 The Joint British Societies Guidelines regards proteinuria as a sign of target organ damage, which conveys a risk of at least 20% in 10 years.455
A study of cardiovascular risk estimation in Chinese adults in the USA–People's Republic of China Collaborative Study of Cardiovascular and Cardiopulmonary Epidemiology (USA-PRC Study) did not include CKD as a risk factor at all.456 It is also important to remember that in this population stroke was the predominant CVD. A review of 25 risk assessment tools identified only 2 derived from an Asian population.457 However, links between GFR categories and CVD events are evident in the Asian population, for example in the Japanese Gonryo study.458 A prospective study in a general Japanese population demonstrated links between lower GFR, high creatinine levels, and proteinuria with cardiovascular mortality particularly stroke.459 In Chinese patients who were at least 50 years old and either had existing CVD or were at high risk, 34% had an eGFR <60 ml/min/1.73 m2 and eGFR <45 ml/min/1.73 m2 was found to be an independent predictor of all-cause and cardiovascular death.460 In a population-based study of 2353 people aged over 40 years in Beijing, an eGFR <90 ml/min/1.73 m2 was associated with increased CVD risk, and for each CKD category stroke was more prevalent than MI.461
In India the increasing prevalence of type 2 diabetes is driving an increase in CKD and both are associated with increased cardiovascular risk. In CKD patients attending one clinic in North India, 28% had diabetes, 27% were overweight, and 92% had hypertension. Metabolic syndrome as defined by the International Diabetes Federation (IDF) 2007 guidelines was present in 39% and more frequent in women.462
Implications for Clinical Practice and Public Policy
A full CVD risk assessment in a person with CKD should include an estimate of GFR and a quantitative assessment of albuminuria. Cystatin C may be helpful at risk stratifying those at intermediate risk.
The key aspect of these recommendations is to ensure that people with CKD are not deprived of treatment strategies known to be effective in general populations.
Areas of Controversy, Confusion, or Non-consensus
Many of the traditional cardiovascular risk assessment tools do not adjust for the presence of early CKD. Any tool should also adjust for level of eGFR because of increasing risk associated with lower levels of kidney function. The Framingham risk equation underestimates true events in a CKD population, but no validated different tools exist at present which better quantify cardiovascular or mortality risk in CKD populations.
Proteinuria is considered to be a sign of target organ damage and thus associated with high cardiovascular risk. However, assessment of risk should also include the presence of albuminuria. Few studies exist which demonstrate that targeting lowering of proteinuria results in reduced cardiovascular risk, though a number of studies which have targeted interruption of the RAAS (and in which lowering of urine protein has also been shown) have demonstrated a benefit.
An appropriate risk assessment tool should be available to assess risk in people with CKD. Addition of the presence of CKD to conventional risk factors in the Reykjavik population increased discrimination but did not increase risk to the same degree as smoking or diabetes.463 In the Framingham Heart Study, GFR category 3b (GFR 30–44 ml/min/1.73 m2) was associated with CVD but not equivalent to previous CVD.464
Lifestyle modification has not been studied in large trials in people with CKD but smoking cessation, achievement of optimum weight, regular exercise, and salt restriction should be seen as reasonable aims.
Pediatric Considerations
It is presently unclear whether children with CKD and elevated lipid levels will benefit from lipid-lowering strategies with no prospective trials ongoing in this area. It is hoped that data from both CKiD55 and the 4C trials78 may be able to address these shortcomings.
However, given that these children demonstrably have elevated risk of cardiovascular and atherosclerotic disease (see Recommendation 4.1.1), that there are data supporting the use of a number of statins in the pediatric population,465 and that the adult evidence for benefit of statins in a subset of the SHARP trial449 with a GFR <60 ml/min/1.73 m2 exist, it would seem reasonable to consider the use of such drugs in children with CKD and elevated lipids.
Recommendations from 2006441 for use of dietary modifications, followed by statins in children older than 8 years of age with persistent elevation in LDL-C levels, have been endorsed by the American Academy of Pediatrics. However, at the time of publication of these guidelines, the KDIGO Dyslipidemia Work Group has suggested that statin therapy may not be appropriate.
Age- and renal function-adjusted doses of such drugs should be carefully considered prior to any therapy being initiated (see Recommendation 4.4.1).
Regarding diabetes control, treatment in keeping with national and international diabetes recommendations is prudent. Note is made that there are specific caveats with respect to drugs and side effects that are important (see Recommendations 3.1.15-3.1.18).
There is no literature in the area of antiplatelet agents in atherosclerotic disease in the subset of children with CKD and the suggestion to offer antiplatelet agents does not apply to pediatric practice.
4.1.4: We suggest that the level of care for heart failure offered to people with CKD should be the same as is offered to those without CKD. (2A)
4.1.5: In people with CKD and heart failure, any escalation in therapy and/or clinical deterioration should prompt monitoring of eGFR and serum potassium concentration. (Not Graded)
RATIONALE
This statement is worded this way to make clinicians aware that people with CKD and heart failure should receive at least equal benefit from heart failure therapy as those without CKD, but that heart failure therapies (in particular increase of RAAS blockade and diuretic therapy) may lead to significant changes in GFR and serum potassium concentrations. This does not imply that such therapy should be avoided but only that clinicians are cognizant of this possibility, monitor it, and understand it in the context of individual risks and benefits.
Heart failure is a complex clinical syndrome which can be caused by any structural or functional cardiac disorder that impairs the pump function of the heart and has a high mortality. Within the general population, the commonest causes of heart failure are ischemic heart disease causing left ventricular systolic dysfunction, and hypertensive heart disease with left ventricular hypertrophy and diastolic dysfunction. Thus people with CKD are at increased risk of both. Evidence-based medication for heart failure impacts on GFR and prescribers need to be alert to this. Changes in GFR are often transient and the downward trajectory GFR is not sustained if all other aspects of care and clinical status are stable. Monitoring and close follow-up are required.
Evidence Base
In an outpatient setting, normal GFR is unusual in heart failure. A study from the Alberta Heart Function Clinic found that in people with an ejection fraction of ≤35%, GFR categories G3a and G3b (GFR 30–59 ml/min/1.73 m2) were present in 40% and 16% of people were GFR categories G4 or G5 (GFR<30 ml/min/1.73 m2).466 This study demonstrated a 1% increase in mortality associated with each 1 ml/min fall in CrCl demonstrating the clear link between kidney function and outcome for people with CKD and heart failure. Meta-analysis of 16 studies and over 80,000 people with heart failure by Smith et al. showed that renal impairment was present in 63% and was associated with increased mortality across the range of kidney function, with an adjusted HR of 1.56 (95% CI 1.53-1.60).467 In the Candesartan in Heart Failure-Assessment of Reduction in Mortality and Morbidity (CHARM) Program with candesartan in heart failure, 36% of subjects had eGFR <60 ml/min/1.73 m2 which was associated with increased mortality and hospital admission regardless of ejection fraction.468 The Digitalis Intervention Group (DIG) identified a GFR of 50 ml/min as a risk threshold for increased mortality risk in heart failure.469 Smith et al. demonstrated that 1-year mortality after admission for heart failure was 29% in people with normal kidney function but 52% in those with moderate to severe renal impairment.470 Anand et al. also demonstrated an independent association between the presence of positive reagent strip proteinuria present in 8% of subjects and increased mortality in heart failure.471
In the Alberta Provincial Project for Outcome Assessment in Coronary Heart Disease (APPROACH) study of people with heart failure related to coronary artery disease (CAD), 39% had CrCl <60 ml/min (<1 ml/s). This study also demonstrated that the level of kidney function was a better indicator of poor outcome than cardiac anatomy.472 In people admitted to hospital with heart failure, increasing SCr concentration is associated with a longer hospital stay and worse outcome.473
LVH is present in 40% of people with CKD,474 progressively increasing as kidney function declines.475 Ha et al. demonstrated LVH in 87% of people with predialysis CKD.476 Parfrey et al. in a study of 432 people who were predialysis, found that only 16% had a normal echocardiogram while 41% had concentric LVH, 16% systolic dysfunction, and 28% left ventricular dilatation.477 LVH is related to increased work load caused by arterial stiffness and hypertension, and at lower levels of GFR, volume overload. Alterations in electrolyte balance, anemia, bone metabolism, uremia, oxidative stress, inflammation, and other inflammatory mechanisms all play a role.478
In the majority of people with CKD, the myocardium develops concentric hypertrophy476 with interstitial fibrosis and CKD-associated cardiomyopathy, leading to left ventricular stiffness.474 This results in diastolic heart failure; people with CKD have higher mortality from diastolic heart failure than systolic heart failure.479 Diastolic heart failure is more common in people with CKD who are older females with hypertension and/or diabetes.480
Left ventricular hypertrophy is particularly associated with anemia and a fall in Hb to less than 12.8 g/dl (128 g/l) is associated with left ventricular growth in people with early CKD.481 Lower Hb levels are associated with increased risk of death and hospitalization in heart failure.482 Other cardiovascular conditions, for example MI and atrial fibrillation, are more common in CKD and may exacerbate heart failure.
It is important to note that in a study of an older population (age >64 years), heart failure was an independent predictor of rapid kidney function decline.483
Standard therapy for the management of systolic heart failure includes an ACE-I or ARB and a beta-blocker licensed for heart failure such as bisoprolol or carvedilol. Meta-analysis of 25 trials of RAAS blockade (ACE-I or ARB) in people with CKD or proteinuria, demonstrated a reduction in the risk for heart failure and reduction in cardiovascular outcomes.484 Subgroup analysis from the Valsartan Heart Failure Trial (Val-HeFT) demonstrated the beneficial effect of valsartan in people with CKD and that valsartan reduced the eGFR by the same amount in people with and without CKD.471 Candesartan was also clinically effective in the CHARM study regardless of underlying kidney function468 while irbesartan was associated with a 23% reduction in hospital admissions related to heart failure in people with nephropathy due to type 2 diabetes.485 Analysis of data on bisoprolol from the Cardiac Insufficiency Bisoprolol Study II (CIBIS II)486 identified 32% of subjects with eGFR <60 ml/min/1.73 m2. These subjects were more likely to die or be admitted to hospital than those with eGFR≥60 ml/min/1.73 m2. However, they showed a similar benefit from bisoprolol treatment. A post hoc analysis of data on carvedilol from the Carvedilol Post-Infarct Survival Control in Left Ventricular Dysfunction (CAPRICORN) and Carvedilol Prospective Randomized Cumulative Survival (COPERNICUS) trials identified 60.8% of participants with eGFR <60 ml/min/1.73 m2.487 Carvedilol was well tolerated by subjects with CKD, and decreased all-cause, cardiovascular, and heart failure mortality. However, doubts were raised over the benefits when eGFR <45 ml/min/1.73 m2.
Aldosterone antagonists are included in standard heart failure management, usually in addition to an ACE-I or ARB, leading to concerns relating to risk of hyperkalemia, particularly in people with lower GFRs. In a study of spironolactone therapy in severe heart failure, Pitt et al.488 included subjects with SCr concentrations up to 2.5 mg/dl (221 μmol/l). There was a 30% reduction in mortality with spironolactone and the incidence of hyperkalemia was low. However, a review of real-world practice demonstrated a significant increase in hyperkalemia with the combination of RAAS blockade and an aldosterone antagonist, highlighting the need for close monitoring with the introduction of combination therapy.489
In the APPROACH study on the specialist management of heart failure, subjects with CAD and CKD, despite being at high risk, were less likely to be taking ACE-Is, beta-blockers, statin, and aspirin. This study demonstrated that those subjects taking beta blockers had lower mortality, however ACE-Is were only of benefit when eGFR was >60 ml/min/1.73 m2.472
International Relevance
The treatment of heart failure is similar around the globe. The ability to closely monitor kidney function or to offer conventional therapies may differ, however. Irrespective, this statement should hold true internationally. The National Heart Care Project of community subjects admitted to hospital with heart failure studied the differences between people of black and white ethnic groups. People with worse renal function were more likely to be black, older and female, and black people had a greater prevalence of hypertension and diabetes but less ischemic heart disease. Black people had a lower risk of mortality at every level of creatinine, for every 0.5 mg/dl (44.2 μmol/l) increase in creatinine, 1-year death risk increased 10% in black people and 15% in white people.470
Implications for Clinical Practice and Public Policy
Cardiorenal syndrome, an impairment of kidney function in the presence of heart disease, is a marker of worse prognosis. In addition to GFR, the presence and severity of albuminuria and anemia aid in prognostication and management.490
Where the link between LVH and heart failure in CKD is so strong and the outcome poor for people with LVH, efforts should be made to optimize care particularly BP. There is a lack of robust evidence for the optimal medical management of heart failure specifically in people with CKD; however, the prevalence of people with early CKD is so high in heart failure trials that we can apply standard treatment to these people. For those people with eGFR <45 ml/min/1.73 m2, the situation is less clear.487
Areas of Controversy, Confusion, or Non-consensus
There are difficulties in making the diagnosis of heart failure in people with CKD, especially diastolic heart failure. Biomarkers such as B-type natriuretic peptide (BNP) may be abnormally elevated in CKD, but may or may not be as responsive to treatment or diagnostically accurate in people with CKD (see Chapter 4.2). There is little evidence to guide the management of left ventricular systolic dysfunction in people with CKD and evidence for the management of diastolic heart failure is generally lacking. However, there is evidence that medications to improve the outcome of people with heart failure are underused in people with CKD.
Appropriate management of the metabolic complications of kidney disease which can exacerbate heart failure needs to be clarified. Clinical studies are required on the role of device therapy in people with CKD (e.g., pacemakers, defibrillators, etc.).
4.2 CAVEATS WHEN INTERPRETING TESTS FOR CVD IN PEOPLE WITH CKD
BNP/N-terminal-proBNP (NT-proBNP)
As GFR diminishes, the prevalence and severity of cardiovascular abnormalities increase and are accepted as major causes of morbidity and mortality in people with CKD. The presence of congestive heart failure (CHF) in people with CKD conveys a worse prognosis than either condition alone. Therefore, early diagnosis and aggressive management of CHF are both highly desirable. A number of cardiac biomarkers (i.e., BNPs and cardiac troponins) appear to have both increasing clinical importance and increasing facility for detection and stratification of CHF. The stimulus for secretion of these biomarkers is the hemodynamic load (i.e., myocardial stretch) and their secretion is associated with the severity of CHF and the degree of left ventricular dysfunction. They are thus useful markers for diagnosis, management, and prognosis in people with normal renal function. However, when the eGFR is less than 60 ml/min/1.73 m2, the accuracy of plasma BNP and NT-proBNP levels for detection and stratification of CHF becomes unreliable and the degree of responsiveness to treatment is not known.
4.2.1: In people with GFR <60 ml/min/1.73 m2 (GFR categories G3a-G5), we recommend that serum concentrations of BNP/NT-proBNP be interpreted with caution and in relation to GFR with respect to diagnosis of heart failure and assessment of volume status. (1B)
RATIONALE
This statement is intended to remind clinicians that although the prevalence of fluid overload and heart failure increases with lower GFR categories, BNP becomes less reliable as a predictor of fluid overload and heart failure at lower GFRs. While it is associated with worse outcomes, cutoff values indicative of heart failure in general populations may or may not be appropriate and changes in values with treatment may or may not have the same meaning.
Evidence Base
Natriuretic peptides belong to a family of circulatory peptide hormones from either myocardial cell origin (atrial natriuretic peptide [ANP] and BNP) or endothelial cell origin (C-type).491, 492 They play an important role in regulating BP and body fluid volume by their natriuretic and diuretic actions, arterial dilatation, and inhibition of the RAAS.493 Natriuretic peptides for the diagnosis of CHF have been a diagnostic breakthrough in cardiology where determination of their concentration in serum can help identify patients with left ventricular systolic dysfunction.494, 495
Concentrations of natriuretic peptides increase in people with CHF and other CVDs as a consequence of pressure and volume overload.496, 497 Among people with progressive LVH, concentrations of NT-proBNP are typically elevated in proportion to the degree of left ventricular mass increase.
Overall, BNP levels provide a better index of left ventricular mass and load than do ANP levels and BNP has emerged as a superior biomarker to ANP for clinical applications involving CHF and left ventricular dysfunction.491, 498 NT-proBNP may have analytical advantages over BNP because of greater stability due to a longer half-life. The cause of an elevated BNP level is multifactorial in origin and may reflect cardiac dysfunction, and/or changes in renal function.499 Since renal dysfunction intrinsically was shown to affect BNP levels in some studies, the diagnostic value of BNP levels in the presence of CKD has been questioned.500, 501
Earlier research hypothesized that measurement of natriuretic peptides could be of use in monitoring excess fluid volume and dry weight in people requiring dialysis,502, 503 but this remains unproven and the significance of plasma natriuretic peptides and their clinical role in dialysis remains unclear.491
Although several studies revealed higher BNP levels as GFR declined, outcomes of other studies suggest that the relationship between renal function and BNP levels may be most strongly dependent on cardiac and volume-related factors.504, 505, 506 To test this hypothesis, Tagore et al. studied BNP levels in a cohort of 143 clinically euvolemic subjects with CKD in whom absence of heart disease was clinically validated and found that plasma BNP levels were independent of GFR.504 Suresh and Farrington507 studied people on dialysis and concluded that BNP levels were predictive of presence of left ventricular dysfunction, cardiac events, and survival in the presence of ESRD, suggesting that BNP levels may be informative across the full range of renal function and even in its absence. As previously established by several studies, the level of NT-proBNP is a strong prognostic marker in both the general population508 and in various disease states, e.g., acute and CHF,509, 510 coronary heart disease,511, 512 and hypertension.513
International Relevance
The issue of how to differentially interpret BNP in the presence or absence of CHF and within the context of declining renal function has received considerable attention but remains incompletely resolved from a clinical perspective. Changes in either renal or cardiac function could potentially impact levels of natriuretic peptides and incomplete knowledge of an individual's clinical status relative to the degree of cardiac pathology or renal dysfunction could confound interpretation of BNP levels. Given the cost of biomarkers assays and the uncertainties related to interpretation of serum levels of BNP/NT-proBNP in people with CKD, competing priorities for health-care resource may dictate reliance on clinical evaluation and local practices of care in some areas of the world. Until better data are available, clinicians are asked to use this laboratory measurement with caution as a diagnostic test for heart failure in those with CKD.
Implications for Clinical Practice and Public Policy
Since people with ESRD with elevated levels of BNP have a higher RR of death, this biomarker could be used as an individual risk marker with the aim of identifying those at high risk. Clinical correlation is of utmost importance to ensure accurate diagnosis and appropriate therapy.
Studies to understand the variability of BNP by categories of eGFR and urine ACR, within and between individuals over time, and the response to therapy should be undertaken so as to better inform clinicians.
Areas of Controversy, Confusion, or Non-consensus
The relationship between renal function and BNP, the role of natriuretic peptides as markers of fluid overload and as predictors of mortality, and the utility of serial monitoring of BNP/NT-proBNP levels in people with CKD, and responsiveness to therapy remains unknown.
Clarification of issues and key points
Natriuretic peptides may be surrogate markers for prediction of mortality in people with CKD reflecting the association of mortality with left ventricular dysfunction and LVH. Before routine measurements of these biomarkers are recommended, their utility in guiding or changing clinical practice should be assessed.
Pediatric Considerations
There is robust literature in both children and neonates regarding both the normative values of BNP514 and its value in predicting outcomes of pediatric heart failure.515 To date no studies in children specifically address the issue of reduced GFR in this population. However, application of similar principles would likely apply.
Troponins
In recent years newer markers of myocardial injury have been introduced into clinical practice.516, 517 Among these, cardiac troponins have proven to be specific markers of myocardial damage.518, 519 Cardiac troponins have been considered the gold standard biochemical test for diagnosis of myocardial damage because they have nearly absolute myocardial tissue specificity, as well as high sensitivity, thereby reflecting even microscopic zones of myocardial necrosis.517, 520 Furthermore, increasing evidence indicates that abnormal troponin measurements identify a subgroup of patients who have an increased risk of major cardiac events519, 521, 522, 523, 524, 525 and that measurement of cardiac troponin T (cTnT) prior to commencing renal replacement is a significant independent predictor of survival.526 However, these markers are renally excreted and are often found to be elevated in people with CKD irrespective of specific symptomatology. Thus, there is uncertainty as to the clinical importance of their different magnitudes of elevation in CKD. There are no data on ‘expected' values of these markers in the context of CKD, or at different categories of GFR and albuminuria.
4.2.2: In people with GFR <60 ml/min/1.73 m2 (GFR categories G3a-G5), we recommend that serum concentrations of troponin be interpreted with caution with respect to diagnosis of acute coronary syndrome. (1B)
RATIONALE
This statement is intended to remind clinicians that isolated elevated serum concentrations of troponin (in the absence of symptoms or electrocardiographic changes) may not necessarily portend a diagnosis of ACS at lower GFR categories. While there are data linking troponin elevations with poor outcomes, the specific implications for an individual in clinical practice are not clear. Hence, this statement simply cautions the clinician with respect to interpretation but does not suggest that the laboratory values are not of significance. In a patient with clinical symptoms suggestive for ischemic heart disease, an elevated cardiac troponin I (cTnI) is suggestive for ACS.
Evidence Base
cTnT and cTnI are low-molecular-weight proteins that form part of the troponin complex and are integral components of the myofibrillar contractile apparatus of the heart.527 Loss of integrity of cardiac myocyte membranes causes release of cardiac troponins into the circulation, which can be detected by highly sensitive assays developed for cTnT and cTnI to diagnose ACS.521 A potent stimulus for the release or production of natriuretic peptides is mechanical stretching,528 whereas plasma cTnT specifically and sensitively reflects myocardial injury and is considered to be an index of irreversible myocardial change.521 In addition, cTnT levels can predict multi-vessel CAD in people requiring dialysis.529 However, the significance of screening people with CKD for CAD using these cardiac biomarkers is questionable530 since increases in serum cardiac troponin concentration can occur in the absence of an ACS.531
Most studies517, 532, 533, 534, 535, 536, 537, 538, 539, 540, 541, 542, 543, 544, 545 have focused on people with ESRD where increases in serum cTnT concentrations have been observed in 20%–90% of subjects540 but generally much lower when cTnI was measured. Information about cardiac troponins and their relationship with comorbidity is sparse in people with CKD who are not receiving dialysis treatment.526, 527, 546, 547 Increased cardiac troponin concentrations may occur early in CKD, including GFR categories 30-59 ml/min/1.73 m2, and more commonly as CKD advances.
Powerful prognostic data emerging from the dialysis population demonstrate that increased concentrations of cardiac troponins reflect either subclinical myocardial damage caused by silent ischemia or myocardial remodeling in the development of LVH.539, 542
Increased levels of cTnT measured with the highly sensitive assay, well below the detection range of standard assays, have been shown to be associated with cardiac structural abnormalities including LVH (both left ventricular wall thickening and dilation) and left ventricular systolic dysfunction,548 and to have a graded association with all-cause and CVD mortality independent of traditional risk factors, renal function, and levels of other biomarkers such as high sensitivity CRP and NT-proBNP.
These findings suggest that chronic elevation of troponin levels may be mediated to a greater extent by indices of heart failure (such as higher left ventricular mass, left ventricular dysfunction, or increased NT-proBNP levels) than indices of atherosclerosis or ischemia.548
International Relevance
The costs of these tests, the differing sensitivity of the assays available, the paucity of data in people with CKD, and the poor correlation of serum troponin levels with ACS in people with CKD should be weighed against competing local health-care priorities and resource before their introduction into routine clinical practice. Emphasis should be placed on the clinical context and local standard practices of care.
Implications for Clinical Practice and Public Policy
Troponin elevation may result from repeated episodes of clinically silent MI,522, 549 although CAD frequently underlies troponin elevation and increased cTnT is also frequently encountered in patients at low risk for CAD. Troponin elevations are as frequent in subjects without CAD as in those with known CAD517, 550, 551, 552 and in people with CKD, serial increasing cTn levels may be of greater utility for diagnosis of ACS. In the context of symptoms and signs of ACS, elevations of troponins should not be ignored nor incorrectly attributed to simply ‘CKD.' Thus the importance of ordering the test for specific indications for those with CKD should be emphasized.
Areas of Controversy, Confusion, or Non-consensus
Information about cardiac troponins and their relationship with comorbidity is sparse in people with CKD who are not receiving dialysis treatment. Although elevated values for TnT are common in people with low GFR in the absence of ACS or CHF, the prevelance of elevated TnI in the absence of ACS or CHF in people with low GFR may be only 0.4-6% depending on the cutoff value chosen.532
Clarification of Issues and Key Points
As pointed out by Roberts et al.,553 although management strategies in people without CKD based on these biomarkers are quite clear, in people with CKD it is not the case despite the strong data on the prognostic implications. These biochemical markers can only be useful if they are interpreted in the context of the clinical history and examination with consideration given as to what management is appropriate for each individual, and if a cardiovascular therapy that is effective in this population is identified or developed.553 The importance of understanding and contextualizing elevations in troponin values in CKD populations cannot be overstated: under-treatment in symptomatic individuals should be avoided, and better understanding of treatment strategies in those asymptomatic individuals is the focus of ongoing study.
Pediatric Considerations
While normative values exist for troponin in children,554 its utility as a general screening marker for myocardial ischemia in pediatrics has recently been called into question,555 and no data exist relating reduced renal function and troponin levels in children.
Non-invasive testing
Most studies of cardiac evaluation in people with CKD have involved candidates for kidney transplantation in order to identify existing cardiac conditions amenable to risk modification and to exclude people with short expected survival due to cardiac morbidity. Among the tests for cardiovascular assessment, coronary angiography is considered invasive, expensive, and associated with risks such as contrast-induced nephropathy and cholesterol embolism. When trying to detect CAD or predict future cardiovascular events in people with CKD, nuclear imaging modalities using thallium tracers have shown conflicting results, especially in the context of small vessel disease. Clinicians should be aware of the limitations of radionuclear testing as a screening tool for significant CAD given the limitations of the test. Exercise-dependent techniques are limited by poor exercise tolerance of many subjects and people with CKD frequently have cardiac evaluation performed with pharmacological agents.556, 557
4.2.3: We recommend that people with CKD presenting with chest pain should be investigated for underlying cardiac disease and other disorders according to the same local practice for people without CKD (and subsequent treatment should be initiated similarly). (1B)
4.2.4: We suggest that clinicians are familiar with the limitations of non-invasive cardiac tests (e.g., exercise electrocardiography [ECG], nuclear imaging, echocardiography, etc.) in adults with CKD and interpret the results accordingly. (2B)
RATIONALE
These statements are worded in this way firstly because although people with CKD are at increased risk from cardiovascular events, observational study has shown that they are frequently discriminated against in terms of investigation and management of such events. Secondly, non-invasive cardiac tests have known additional limitations in people with CKD and such limitations and their consequences need to be clearly understood by health professionals caring for people with CKD.
Evidence Base
Noninvasive stress testing appears to be the most common first approach to cardiac evaluation of asymptomatic patients, prompted by diabetes, age and risk-factor burden. These tests include myocardial perfusion studies, stress echocardiography, and most recently, cardiac computed tomographic angiography. However, people with CKD are underrepresented in studies evaluating the diagnostic sensitivities and specificities of noninvasive tests. Exercise ECG is limited by lack of specificity of the ST-segment response and by inability of many people with CKD to exercise to a diagnostic workload.
These tests have imperfect sensitivity and specificity in people with CKD or, in the case of tomographic angiography, evaluation in this population is as yet unpublished.556 CAD has been detected in a high proportion of people on long–term dialysis therapy. Studies describing associations of angiographic coronary stenosis with subsequent clinical events in people with ESRD, including those undergoing transplant evaluations, have reached inconsistent conclusions.558, 559, 560, 561, 562
Abnormalities on myocardial perfusion studies correlate well with the presence of CAD in the general population, with mean weighted sensitivity of 88% and mean weighted specificity of 74%.563 The performance of myocardial perfusion studies in identifying CAD in people with CKD is more variable, with reported sensitivities and specificities in people with ESRD ranging from 37% to 90% and 40% to 90%, respectively.564, 565, 566, 567 Nonetheless, results from myocardial perfusion studies have prognostic value for cardiac events and mortality.568, 569 In a meta-analysis of 12 studies involving thallium-201 scintigraphy and dobutamine stress echocardiography, people with ESRD with inducible ischemia had approximately 6 times the risk of MI and 4 times the risk of cardiac death as those without inducible defects.570 Moreover, subjects with fixed defects also had nearly 5 times the risk of cardiac death.
The prognostic value of myocardial perfusion studies has been shown with other perfusion tracers. For example, in a study of 126 people with ESRD who underwent technetium-99m myocardial perfusion studies as part of their pre-transplant assessment, the presence of a reversible defect was associated with 3 times the risk of post-transplant cardiac events (HR 3.1; 95% CI 1.1-18.2) and nearly twice the risk of death (HR 1.92; 95% CI 1.1-4.4) compared with normal test results.571
De Lima et al.572 prospectively studied 126 renal transplant candidates clinically classified as moderate (age >50 years) or high (diabetes, extra cardiac vascular disease, or known CAD) coronary risk with myocardial perfusion studies, dobutamine stress echocardiography, and coronary angiography. Significant CAD, defined as greater than 70% stenosis in 1 or more major epicardial artery on angiography was found in 42% of the sample. After a median follow-up of 46 months, clinical risk stratification and coronary angiography predicted major cardiac events, but myocardial perfusion studies and dobutamine stress echocardiography did not.
Newer imaging techniques are under current evaluation but a review here is beyond the scope of this guideline; these techniques include: stress myocardial perfusion single photon emission computed tomography (SPECT);557, 573, 574, 575, 576 18F-BMS, a novel fluorine-based myocardial perfusion tracer;557, 577 and gated imaging using radio-labelled ammonia (13NH3).
International Relevance
People with CKD who are being investigated prior to transplantation should be assessed according to the national and international cardiology guidelines and taking into account local practice and availability of resource.
Implications for Clinical Practice and Public Policy
There is still a need for a safe, noninvasive, diagnostic modality that will allow stratification of cardiovascular risk among people with CKD. With the development of new imaging markers, it becomes difficult for clinicians to choose the imaging tests that can best aid clinical decisions for a given patient. Imaging methods should provide accuracy and precision at acceptable cost. Until advanced imaging techniques (e.g., cardiac MRI, positron emission tomography, cardiac computed tomography) are standardized, clinicians should rely on the most reliable and familiar techniques in their environment.578
Areas of Controversy, Confusion, or Non-consensus
Clinical and cost-effectiveness could be improved; cardiac evaluation in people with CKD should be based on data from clinical trials.
Clarification of Issues and Key Points
If cardiac evaluation is at present predominantly used for risk stratification and prognosis, in the future it may guide optimal therapy and monitor clinical progress.557
4.3 CKD AND PERIPHERAL ARTERIAL DISEASE
There is a strong link between CKD and peripheral arterial disease (PAD). Symptoms of PAD may only be present in a minority of people who have clinical evidence of PAD.579 It is therefore important to measure ankle-brachial index and perform regular systematic assessment of the lower limbs of people at high risk of PAD to identify bruits, loss of pulses, cool pale extremities, delay in venous filling, and skin ulceration.580
4.3.1: We recommend that adults with CKD be regularly examined for signs of peripheral arterial disease and be considered for usual approaches to therapy. (1B)
4.3.2: We suggest that adults with CKD and diabetes are offered regular podiatric assessment. (2A)
RATIONALE
These statements are worded in this way in order to highlight the increased risk of PAD in people with CKD, particularly those with CKD and diabetes.
Evidence Base
The prevalence of PAD is common in people with CKD and it increases with lower levels of GFR (Table 31). PAD can be attributed to the greater prevalence of traditional risk factors such as diabetes, hypertension, dyslipidemia, advanced age, and renal-specific factors.581, 582, 583, 584, 585, 586, 587, 588
Table 31. Peripheral arterial disease and CKD.
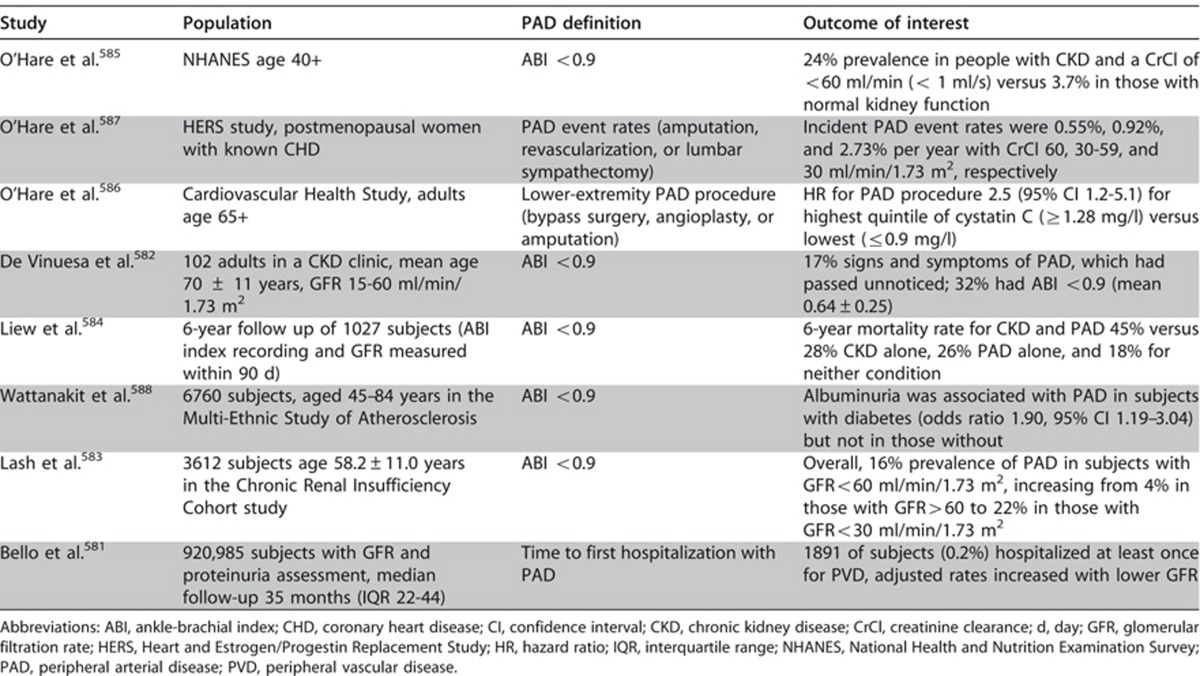
Diabetes is the leading cause of CKD globally and the risk of diabetic foot ulceration and lower extremity amputation in subjects with diabetes increases dramatically with lower GFR. In a cohort of 90,617 individuals with diabetes over a median observation time of 2.4 years, the HR for amputation increased from 2.08 (95% CI 1.68–2.58) for those with GFR 30-59 ml/min/1.73 m2 to 7.71 (95% CI 5.29–11.26) for those with GFR <30 ml/min/1.73 m2 compared to reference subjects with GFR >60 ml/min/1.73 m2.589
Screening for PAD is recommended for adults in the general population based on age and number of risk factors.590 Although an ankle-brachial index of <0.9 is generally considered evidence of PAD, the utility of this measure is unproven in CKD because of the greater prevalence of vessel calcification. Evidence-based medical therapies for PAD in people with CKD are similarly lacking. Smoking cessation is mandatory. Aspirin may be beneficial for prevention of cardiovascular events. Clopidogrel has not been studied in people with CKD and PAD. No RCTs evaluating percutaneous versus surgical revascularization techniques have been conducted in people with CKD and PAD but outcome studies all suggest that CKD confers an increased risk of adverse outcome, regardless of the revascularization technique employed.591, 592 These statements do not apply to pediatric practice.
International Relevance
There is no evidence to suggest that the international approach to identification and management of PAD in people with CKD should differ.
Implications for Clinical Practice and Public Policy
People with CKD are more at risk of PAD and need regular assessment and monitoring. The role of ankle-brachial index versus other diagnostic techniques may have implications for future practice. Prospective data on non-surgical therapies and data regarding percutaneous versus surgical revascularization are required to inform policy and recommended procedure.
Areas of Controversy, Confusion, or Non-consensus
There remains ongoing debate as to the timing and use of specific diagnostic criteria for identification of PAD in people with CKD and no studies have examined the utility of conventional methods in early detection of PAD in CKD. The use of specific medical and surgical management strategies in people with CKD and PAD has not been evaluated. There is no reason to believe that treatment strategies should differ, though risks of diagnostic testing (such as angiography) remain real.
4.4 MEDICATION MANAGEMENT AND PATIENT SAFETY IN CKD
Many drugs and investigative pharmaceuticals are renally excreted and their dosage may need to be reduced in patients with CKD in order to avoid toxicity. As many people with CKD are elderly, there should also be consideration of chronological age.
It is beyond the scope of this guideline to list all the agents that may need dose adjustment in CKD or which should be avoided due to potential nephrotoxicity. This information is widely available in documents which may exist at local, regional, or national bodies (e.g., British National Formulary: www.bnf.org), and other textbooks of pharmacology.593, 594 However key classes of commonly prescribed drugs in people with CKD will be mentioned together with suggestions for dose adjustment. Much of this guidance is based upon our understanding of pharmacology and pharmacokinetics rather than randomized control trial evidence.
The statements presented here are intended to inform clinicians caring for those with CKD or at risk for CKD with respect to common clinical situations or exposures which may put people at risk for AKI or progression of CKD.
4.4.1: We recommend that prescribers should take GFR into account when drug dosing. (1A)
4.4.2: Where precision is required for dosing (due to narrow therapeutic or toxic range) and/or estimates may be unreliable (e.g., due to low muscle mass), we recommend methods based upon cystatin C or direct measurement of GFR. (1C)
4.4.3: We recommend temporary discontinuation of potentially nephrotoxic and renally excreted drugs in people with a GFR <60 ml/min/1.73 m2 (GFR categories G3a-G5) who have serious intercurrent illness that increases the risk of AKI. These agents include, but are not limited to: RAAS blockers (including ACE-Is, ARBs, aldosterone inhibitors, direct renin inhibitors), diuretics, NSAIDs, metformin, lithium, and digoxin. (1C)
4.4.4: We recommend that adults with CKD seek medical or pharmacist advice before using over-the-counter medicines or nutritional protein supplements. (1B)
4.4.5: We recommend not using herbal remedies in people with CKD. (1B)
4.4.6: We recommend that metformin be continued in people with GFR≥45 ml/min/1.73 m2 (GFR categories G1-G3a); its use should be reviewed in those with GFR 30–44 ml/min/1.73 m2 (GFR category G3b); and it should be discontinued in people with GFR <30 ml/min/1.73 m2 (GFR categories G4-G5). (1C)
4.4.7: We recommend that all people taking potentially nephrotoxic agents such as lithium and calcineurin inhibitors should have their GFR, electrolytes and drug levels regularly monitored. (1A)
4.4.8: People with CKD should not be denied therapies for other conditions such as cancer but there should be appropriate dose adjustment of cytotoxic drugs according to knowledge of GFR. (Not Graded)
RATIONALE
These statements are worded in this way in order to inform appropriate prescribing and management of medical conditions in people with CKD. Recommendation 4.4.1 is specifically worded to ensure that clinicians remember that in those circumstances where accurate GFR is required (narrow therapeutic or toxic window), a direct measurement, not an estimate of GFR should be undertaken.
Evidence Base
Potential problems associated with use of medication in people with CKD include:
Reduced ability to excrete drugs and/or their metabolites
Increased sensitivity to medications (e.g., those bound to albumin in hypoalbuminemic states such as nephrotic syndrome)
Diminished tolerance of side effects, particularly in the elderly
Loss of efficacy13
Wherever possible, people with CKD should receive the same treatment as those with normal renal function. However, the dosages may need adjustment according to GFR.
There are medicines whose toxicity is worsened in acute illness particularly in a setting of dehydration such as diarrhea and vomiting. General advice about appropriate dosing and when to restart these agents should be given to people taking these drugs during intercurrent illness, together with a recommendation for consultation with a health-care professional as soon as possible.
Use of herbal and over-the-counter medicines is very common worldwide and some (such as those containing aristolochic acid)595, 596 are known to be nephrotoxic. There is no good quality safety or efficacy data for many of these compounds.
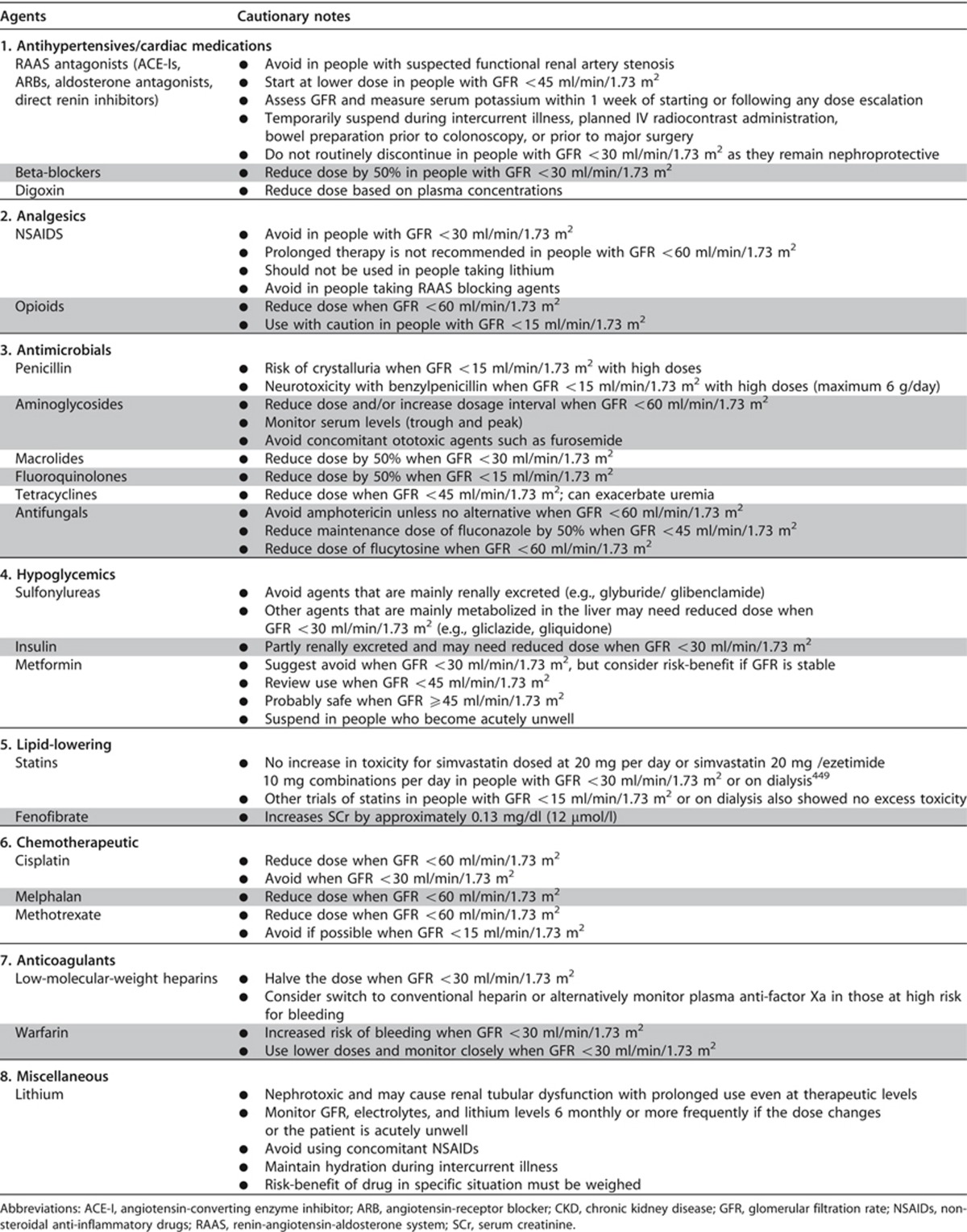
Table 32 details specific advice for various classes of drugs where CKD may be an issue. These include RAAS blocking agents,262 beta-blockers, analgesics, antibiotics, lithium,597 hypoglycemic agents,598, 599, 600 lipid-lowering agents,449, 601, 602, 603, 604 certain chemotherapeutic agents, and anticoagulants.605
Table 32. Cautionary notes for prescribing in people with CKD.
International Relevance
This guidance is based upon knowledge of pharmacology that has universal relevance. The main international implication is centered on costs of some newer therapies compared to the older ones. Somewhat paradoxically, as the weight of evidence resides mainly with agents that have been available for longer, they have the advantage of being less costly, and have the side effects that are well documented; thus many of these older agents are preferred.
Pediatric Considerations
No specific recommendation for or against the use of a BSA adjusted eGFR in relation to drug dosing can be made in children with CKD as the majority of drug studies have been performed in adult males and extrapolated to children without consideration for BSA and/or pediatric renal function or clearance.
The exception to this rule would be that for any drug where pediatric pharmacokinetic or pharmacodynamic studies exist, the method used in calculating the effect of renal function should be used when estimating the need for dose adjustments or modifications for the individual patient.
Numerous resources for dose adjustments for pediatric CKD and ESRD patients exist, including those found in numerous textbooks606, 607 and online resources.608
An additional value of dedicated pediatric renal multidisciplinary teams, in terms of caring for children with CKD and ESRD, is their ability to provide anticipatory guidance for the patients and families. All such teams should at the least provide written or online information to their patients and families directing them to seek advice in situations where they may be prescribed medications from other providers or may be seeking over-the-counter drugs or supplements. The presence of trained pediatric pharmacists as part of such teams can be invaluable in achieving such support and education for both families as well as the CKD health-care providers and community resources such as local pharmacists.
LIMITATIONS
People with CKD are often excluded or not identified from trials of medications for non-renal disease. Recommendations are partly based upon knowledge of pharmacology rather than controlled trials in carefully defined populations.
RESEARCH RECOMMENDATIONS
National and international research groups, and those with CKD-focus organizations (International Society of Nephrology, International Federation of Kidney Foundations, and other national bodies) should ensure adequate representation of people with CKD in clinical trials, leading to an improved understanding of pharmacodynamics of those with CKD.
- 4.5: IMAGING STUDIES
- 4.5.1: Balance the risk of acute impairment in kidney function due to contrast agent use against the diagnostic value and therapeutic implications of the investigation. (Not Graded)
RATIONALE
Use of iodinated radiocontrast media has been associated with AKI with reported rates of 0-11% depending on the population under study, the type of agent that is used, and the definition of nephrotoxicity.609 The following recommendations are concordant with those from the American College of Radiology (ACR),610 the European Society of Urogenital Radiology (ESUR)611 and the KDIGO Clinical Practice Guideine for Acute Kidney Injury.7
Radiocontrast
- 4.5.2: We recommend that all people with GFR <60 ml/min/1.73m2 (GFR categories G3a-G5) undergoing elective investigation involving the intravascular administration of iodinated radiocontrast media should be managed according to the KDIGO Clinical Practice Guideline for AKI including:
- Avoidance of high osmolar agents (1B);
- Use of lowest possible radiocontrast dose (Not Graded);
- Withdrawal of potentially nephrotoxic agents before and after the procedure (1C);
- Adequate hydration with saline before, during, and after the procedure (1A);
- Measurement of GFR 48–96 hours after the procedure (1C).
RATIONALE
These statements have been worded in order to inform safe radiological investigation of people with CKD and to avoid potential nephrotoxcity that can be associated with radiological imaging.
Evidence Base
Radiocontrast media associated AKI is a largely preventable cause of morbidity and mortality. There is no internationally agreed definition but most studies use an increase in SCr of >0.5 mg/dl (44 μmol/l) and/or a 25% increase from baseline SCr within 3 days of the procedure.7,609, 610, 611 Epidemiological studies and case series have identified the following risk factors for AKI:
GFR <60 ml/min/1.73 m2 (particularly if <30 ml/min/1.73 m2)
Diabetes
Concurrent dehydration
CHF
>70 years of age
Concurrent use of known nephrotoxic agents such as NSAIDs
Use of high osmolality agents (especially in those with GFR <60 ml/min/1.73 m2)
Use of large doses of radiocontrast media
i) Intra-arterial injection
Gout (hyperuricemia)
Numerous studies of preventative strategies have been performed with the following conclusions:
High osmolar agents pose a greater risk of AKI in people with CKD.609
Iso-osmolar agents compared to low-osmolar agents are associated with lower rates of AKI in some but not all studies. Wherever possible iso-osmolar agents should be used in people with CKD at high risk for AKI (although these tend to be more expensive).612
Although risk for AKI increases with GFR <60 ml/min/1.73 m2, the rates are particularly high (7.8% in one study) when GFR is <30 ml/min/1.73 m2. Implementing preventative strategies for all with a GFR <60 ml/min/1.73 m2 may not be practical but a graded risk assessment taking into account all factors may be more realistic. Some guidelines such as American College Radiology610 and ESUR611 provide a checklist as a means of identifying patients at risk of AKI before investigation.
Extracellular volume expansion is widely recommended although there are few good quality trials on which to base an ideal protocol.609 0.9% saline given by continuous infusion appears superior to 0.45% saline or bolus injection and there is no demonstration of consistent superiority of sodium bicarbonate over saline. Current guidance suggests either infusion of 1 ml/kg body weight/hour for 3-12 hours before and after the procedure or 100 ml/hr, beginning 6 to 12 hours before and continuing 4 to 12 hours after intravascular iodinated contrast medium administration.609, 610
Use of N-acetylcysteine or ascorbic acid as preventative measures has not been shown to be a consistent benefit.
International Relevance
This guidance has universal relevance although there are cost implications as the iso-osmolar contrast media are more expensive.
LIMITATIONS
Some of the guidance is based upon limited evidence and there is a need for more research into simple preventative measure such as pre-investigation rehydration (see below). There has not previously been a universal definition for AKI following administration of contrast media. However, recommendations from the KDIGO AKI Guideline suggest that the same general AKI definition and staging be used for changes in kidney function, irrespective of etiology.
RESEARCH RECOMMENDATIONS
Prospective studies using direct measures of GFR before and after administration of radiological contrast media are required to help define the incidence of AKI. Such studies would also be able to validate creatinine or other estimates of GFR in people undergoing radiological investigation.
Prospective controlled trials of rehydration using different fluids (saline, bicarbonate, Hartmann's) and validated estimates of GFR are urgently required.
Definitive studies of N-acetylcysteine and other antioxidants would help determine their usefulness or otherwise.
Gadolinium-containing contrast media
Gadolinium is a rare earth element that is naturally highly toxic. When bound to proprietary chelating agents, it is essentially biologically inert in people with normal renal function and provides excellent contrast during MRI. These chelates are excreted unchanged by the kidneys by glomerular filtration and have much lower direct nephrotoxicity than conventional radioiodine contrast media.613 However over 200 cases of a scleroderma-like condition now termed nephrogenic systemic fibrosis (NSF) following gadolinium use in patients with CKD have been reported. 613, 614
4.5.3: We recommend not using gadolinium-containing contrast media in people with GFR <15 ml/min/1.73 m2 (GFR category G5) unless there is no alternative appropriate test. (1B)
4.5.4: We suggest that people with a GFR <30 ml/min/1.73 m2 (GFR categories G4-G5) who require gadolinium-containing contrast media are preferentially offered a macrocyclic chelate preparation. (2B)
RATIONALE
These statements have been worded in order to enable safe administration of gadolinium to people with CKD. As with all tests requested in the CKD population, clinicians should be aware of the risk-benefit ratio of the use of gadolinium in people with GFR 15-29 ml/min/1.73 m2.
Evidence Base
NSF is an untreatable and sometimes fatal condition that complicates the use of gadolinium-containing contrast media in people with CKD. Prevention is therefore the best approach with avoidance of gadolinium exposure unless clinically indicated and to use the lowest risk agent at the lowest dose.613, 615
Although the risk of AKI with iodinated contrast media is much greater than that for NSF with gadolinium, the former is treatable with dialysis whereas the latter is not. Thus, consideration of more conventional imaging techniques should be undertaken in all with a GFR <30 ml/min/1.73 m2.610, 613, 614, 615
A recent meta-analysis and review have highlighted those patients most at risk and quantified an OR for NSF of between 20-50 for those with a GFR <15 ml/min/1.73 m2.614
While there is general agreement that patients with a GFR >15 ml/min/1.73 m2 are at increased risk, it is not possible to derive a precise estimate. No patients with a GFR >30 ml/min/1.73 m2 have developed NSF without concomitant liver failure.614
There is some suggestion that the type of gadolinium preparation plays a role:614, 615 Linear chelated preparations such as gadodiamide may be more likely to cause NSF and should be avoided when GFR is <30 ml/min/1.73 m2. Gadoteridol, gadobutrol, or gadoterate should be considered if MRI with contrast is clinically essential.610, 613, 615 Moreover, as gadolinium is freely dialysed,616 most guidelines recommend dialysis in patients with a GFR <15 ml/min/1.73 m2 or for those already on dialysis immediately after (and perhaps repeated 24 hours later) completion of the procedure.610, 615 The role of dialysis in people with GFR above 15 ml/min/1.73 m2 is uncertain.
International Relevance
This guidance has universal relevance although there are cost implications as the non-linear chelated preparations are more expensive.
Pediatric Considerations
Regarding both Recommendations 4.5.3 and 4.5.4, a number of considerations specific to the use of gadolinium preparations in young children and neonates must also be considered in addition to the general admonishments against their use in situations of GFR <30 ml/min/1.73 m2. In particular, the FDA currently does not license any gadolinium-based contrast agent (GBCA) product for use in children less than 2 years of age; and likewise the European Medicines Agency cautions against the use of any GBCA in a child less than 1 year of age.
In addition, the ability to accurately estimate the relative value of a neonate or young infant's GFR leads to great difficulty in terms of assigning risk of GBCA exposure if one bases this on renal clearance. In a recent paper by Meng and colleagues,617 they surveyed a worldwide group of both cardiologists and radiologists with respect to their use of gadolinium in their pediatric MRI practice. While 93% of the respondents did evaluate renal function in some or all of their patients, only a slight majority (54%) used an estimating formula, most often the Schwartz equation, with the remainder relying on SCr alone (31%) or urine output (6%). Perhaps of most concern was that renal function was assessed in only 33% and 31%, respectively, of patients classified as ‘all neonates' or ‘all patients <1 week of age.' Equally concerning was the fact that 13% of the respondents would give gadolinium to some of these children in the face of a GFR <30 ml/min/1.73 m2.
In recognition of our inability to accurately measure GFR in the neonate and, by extension, the clearance of compounds such as gadolinium, all nephrologists and radiologists must exercise caution in terms of use of GBCA in this potentially high-risk population, and all other imaging modalities should be considered prior to choosing one requiring gadolinium exposure.
LIMITATIONS
The evidence is largely based upon case series rather than prospective studies or RCTs.
RESEARCH RECOMMENDATIONS
As for iodinated contrast media, a prospective study of people with CKD undergoing nuclear MRI with gadolinium contrast would help define change in GFR and validate estimators. Because NSF is such a serious condition, an RCT of dialysis in people with GFR <30 ml/min/1.73 m2 would help to determine risk-benefit in these patients, though recruitment may be difficult due to potential ethical concerns.
Bowel preparation
The increasing use of colonoscopy as a screening tool for bowel cancer has resulted in many people undergoing bowel preparation with oral sodium phosphate-containing preparations. Case reports of acute and late irreversible renal failure with biopsy-proven phosphate deposition have led to a new disease entity termed acute phosphate nephropathy.618, 619 Less than 40 definite cases have been reported and these have been extensively reviewed.
4.5.5: We recommend not to use oral phosphate-containing bowel preparations in people with a GFR <60 ml/min/1.73 m2 (GFR categories G3a-G5) or in those known to be at risk of phosphate nephropathy. (1A)
RATIONALE
This statement has been worded in order to enable safe bowel preparation in people with CKD who need to undergo investigation of bowel disease.
Evidence Base
Electrolyte disturbances that are sometimes severe and include hyperphosphatemia, hypocalcemia, hypo- and hypernatremia, and hypokalemia have been reported in normal volunteers undergoing oral phosphate bowel preparation.620 Renal injury has been reported in a small number of people although the condition is likely to be under-recorded. A recent study from Iceland estimated the incidence to be around 1 per thousand doses621 but others would suggest incidence rates of between 1% and 4%.618, 619
Two broad patterns of renal injury have been described. An early symptomatic response associated with severe hyperphosphatemia and hypocalcemia and a later (days to months) irreversible kidney injury associated with a specific tubulointerstitial calcium phosphate deposition.618, 619
The following people are said to be at particular risk although the link to kidney injury is associative in many cases and firm evidence is lacking:
GFR <60 ml/min/1.73 m2
>60 years of age
Female
Hypertension
Diabetes
CHF
Dehydration
Active colitis
Concurrent use of RAAS blocking agents, diuretics, lithium, NSAIDs
Large and/or repeat dosing of oral phosphate preparations
Hypoparathyroidism
Although the FDA has banned oral phosphate solutions, there is no qualitative difference with tablet preparations. Both should be avoided in people at risk and this is the current recommendation of the American Society for Gastrointestinal Endoscopy.622
There is some debate as to whether the cause of the kidney injury is entirely due to dehydration rather than phosphate use per se.623 However, it is hard to understand why calcium phosphate deposits occur and cause late renal impairment if dehydration was the only factor.
There is a single RCT of phosphate versus non-phosphate preparations but assessment of renal function was limited to change in SCr and GFR at baseline is not reported.624 Notwithstanding these limitations, there were greater changes in serum potassium, calcium, and phosphate in those given sodium phosphate-containing preparations.
As there are non-phosphate-containing bowel preparations available, these should be used in all the above groups (and arguably in all people given the biochemical abnormalities observed in normal volunteers). As for radiocontrast media, rehydration with saline may be required in the frail and ill irrespective of the bowel preparation that is used.
International Relevance
This guidance has universal relevance but non-phosphate-containing bowel preparations are more expensive, so the use of these agents may differ around the world.
LIMITATIONS
The data are based upon case series with a limited number of affected individuals and retrospective population studies.
RESEARCH RECOMMENDATIONS
Prospective study in people with normal renal function and those with different severities of CKD are urgently required in order to define the acute biochemical and metabolic effects of phosphate-containing bowel preparations. There is also a need for a study of all people undergoing bowel preparation with whatever preparation in order to explore the effects on GFR on the incidence of this complication. More definitive exploration of rehydration therapy (type and volume) in people with CKD undergoing bowel preparation is urgently needed.
4.6 CKD AND RISKS FOR INFECTIONS, AKI, HOSPITALIZATIONS, AND MORTALITY
The following section gives guidance for the care of people with CKD given that they are at increased risk for a number of events such as infections, AKI, CVD, hospitalizations, and mortality. Appreciating that increased risk and implementing some of the recommendations below may result in improved outcomes for people. It will be important to develop policies and robust research agendas to address areas which do not have a substantial evidentiary base.
CKD and risk of infections
CKD is associated with significant major infectious complications, which occur at rates 3 to 4 times the general population. Infection is an important cause of morbidity and mortality among patients with kidney failure and is the second leading cause of death following CVD. CKD may be a risk-multiplier for acute infectious disease-associated mortality, as it is for CVD. Despite a less effective response to vaccination, there are data suggesting benefit from immunization among people with CKD as in the general population.
4.6.1: We recommend that all adults with CKD are offered annual vaccination with influenza vaccine, unless contraindicated. (1B)
4.6.2: We recommend that all adults with eGFR <30 ml/min/1.73 m2 (GFR categories G4-G5) and those at high risk of pneumococcal infection (e.g., nephrotic syndrome, diabetes, or those receiving immunosuppression) receive vaccination with polyvalent pneumococcal vaccine unless contraindicated. (1B)
4.6.3: We recommend that all adults with CKD who have received pneumococcal vaccination are offered revaccination within 5 years. (1B)
4.6.4: We recommend that all adults who are at high risk of progression of CKD and have GFR <30 ml/min/1.73 m2 (GFR categories G4-G5) be immunized against hepatitis B and the response confirmed by appropriate serological testing. (1B)
4.6.5: Consideration of live vaccine should include an appreciation of the patient's immune status and should be in line with recommendations from official or governmental bodies. (Not Graded)
4.6.6: Pediatric immunization schedules should be followed according to official international and regional recommedations for children with CKD. (Not Graded)
RATIONALE
CKD is associated with alterations in primary host defense mechanisms and increases the risk of bacterial infections (Table 33). Epidemiological study suggests that the 3 most commonly seen infectious complications in the CKD population are: urinary tract infection, pneumonia, and sepsis. In the general population, there is strong evidence that preventive measures are effective in adults and there are data suggesting benefit from immunization in people with CKD.
Table 33. Risk factors for infection in people with CKD.
Evidence Base
Previous investigations have firmly established ESRD as a strong risk factor for infectious complications.625, 626, 627, 628 However, few epidemiologic reports have addressed the risk of infections in people with CKD not treated with dialysis.629, 630 Data from the US Renal Data System (USRDS) suggest that higher rates of hospital admission because of septicemia are noted in people with CKD compared with those without CKD.631 Comprehensive studies of the absolute rates, risk factors, clinical course, and outcomes of different types of clinically relevant infections across the spectrum of CKD are not available.
Among people with CKD, very few studies have examined the incidence or prevalence of infections and no published data describe GFR category–specific infection rates. Among Medicare beneficiaries aged more than 66 yr, people with diagnosed CKD seem to have substantially higher rates of hospitalization for diagnoses of pneumonia and sepsis compared with people without diagnosed CKD.628 In addition to an increased incidence of being hospitalized with infections, people with CKD have longer lengths of hospital stay during infection-related admissions compared with people without CKD.632
Among USRDS data admission rates for all causes, CVD and infection, are 38–46% higher for those with CKD than for those without.633 Rates for pneumonia in CKD were nearly three times higher than those for non-CKD. Hospitalizations for bacteremia/septicemia, were nearly four times higher for people with CKD compared to those without. Hospitalization rates for urinary tract infections were three-fold higher for people with CKD.
In people with ESRD, the function of polymorphonuclear white blood cells, lymphocytes, and monocytes is altered, resulting in an impaired host response to infection.636, 637, 638 However, these issues have not been adequately investigated in people with CKD.
There is a growing body of evidence in the general population for reduction in infections, hospitalizations, and mortality as a result of immunizations. Current data suggest that vaccination is an underused prevention strategy in the CKD and ESRD populations.639 Potential barriers to immunization that are specific to the CKD populations have not been systematically examined. Although lower vaccine responsiveness has been widely recognized in the ESRD population, to what extent moderate to advanced CKD modifies vaccine responsiveness remains unclear.640, 641, 642 Studies that have examined vaccination in the setting of CKD or ESRD have been limited by small study size, variable follow-up, and ascertainment of surrogates for vaccine effectiveness such as antibody response and rate of antibody decline after vaccination as opposed to vaccine efficacy for preventing infection.643
Influenza A and B vaccine. Despite potentially impaired antibody responses, a 2-year analysis of US Medicare claims data found that people vaccinated against influenza A and B on dialysis had a substantially lower chance of any-cause hospital admission and any-cause death than those patients not vaccinated on dialysis.644 This finding might indicate clinical effectiveness of vaccinating this population but its observational design might also reflect differences in the underlying clinical status among people vaccinated and not vaccinated. No unique adverse events related to influenza vaccine have been identified in people on dialysis.
Pneumococcal vaccine. People with kidney disease vaccinated with the pneumococcal vaccine seem to develop different serotype-specific titers, develop lower levels of antibody titers, and have a more rapid loss of antibody titers as compared with healthy control subjects.639, 645, 646 Practitioners should be aware of the impact of specific vaccines on responsiveness and duration of responsiveness. Revaccination practices will be dictated by that knowledge.
Hepatitis B vaccine. Widespread hepatitis B virus (HBV) vaccination at the onset of dialysis has led to a marked reduction of HBV infections in people with ESRD, although improved screening of blood products and dissemination of recommendations for reducing the spread of HBV infections in dialysis units have also likely contributed.647 Among people with moderate to advanced CKD, hepatitis B vaccination responsiveness has been shown to range from approximately 60 to 80% depending on the dosage, number of administered vaccines, and study population. Although findings have been inconsistent as to whether the level of GFR affects vaccine responsiveness in people with CKD640, 642 those with higher GFR are more likely to respond with seroconversion, independent of other factors.640
S. aureus vaccine. StaphVAX has not been shown to be efficacious in reducing the risk for Staphylococcus aureus bacteremia in people on hemodialysis.648, 649 No data are published in people with CKD.
Live vaccines. Due to the fact that people with CKD are often immunocompromised, live vaccines should only be used with caution on an individual basis.
In summary, although some vaccines (like influenza) in usual doses provide protection, other vaccines (HBV and pneumococcal) require more frequent dosing or larger doses to achieve and maintain protective antibody titers. Frequency and type of vaccination will vary according to local circumstances and prevalence of disease.
International Relevance
The availability of different vaccinations may vary worldwide, as does the prevalence of specific bacterial, viral, and other infections. It is reasonable to offer individuals appropriate immunization according to local practices.
Implications for Clinical Practice and Public Policy
Vaccines for influenza, hepatitis B, and pneumococcus are currently recommended for people with CKD by the local, regional, or national advisory committees on immunization practices from most countries.
Current recommendations are to:
Provide influenza vaccination annually to people with CKD.
Provide pneumococcal vaccine with a single booster dose 5 years after the initial dose.
Provide HBV vaccine to people with CKD who are likely to require RRT. Although the recommendation is to give the HBV vaccine during more severe CKD (GFR <15 ml/min/1.73 m2), it may be preferable to give this earlier to maximize the chances of achieving immunity; there are data to support this practice.640 This would also ensure that all patients are immunized against HBV before receiving a transplant. As protective antibody levels may fall, this should be checked (possibly annually) with booster doses given if appropriate.
Areas of Controversy, Confusion, or Non-consensus
Much remains to be understood concerning impaired host response to infection in patients with CKD.
Studies should be undertaken to determine the absolute rates, risk factors, and clinical course of different types of clinically relevant infections across the spectrum of CKD, by GFR and albuminuria category, and by cause.
The outcomes across the range of acute infections in CKD population need to be ascertained.
Studies should be undertaken to assess the rate of decline of antibody titers post-vaccination and the efficacy of immunization in people with CKD.
Pediatric Considerations
Current immunization schedules for children are regularly updated by both the US Centers for Disease Control and Prevention650 and American Academy of Pediatrics.651
Current and comprehensive immunization recommendations for children with CKD have been published by Neu in 2012.652 The paper addresses key issues regarding the use of vaccines in CKD pediatric populations who are receiving concomitant immunosuppression and in those awaiting transplantation. The need for, and interpretation of, protective antibody levels for those vaccines where this is indicated is described.
An oversimplified summary of the recommendations would be to provide all recommended childhood vaccines to every child with CKD with the exception of any live viral vaccine in a child receiving immunosuppressive medications. Likewise children on dialysis should not receive the live attenuated influenza vaccine although the inactivated version can and should be given to all children with CKD on an annual basis otherwise. Pneumococcal vaccination is particularly important in children with nephrotic syndrome and those with CKD, and current vaccination schedules and products should be carefully reviewed to ensure proper serotype coverage is being provided. Hepatitis B status and vaccination are of extreme importance in all children who may go onto dialysis – and specific recommendations for ongoing monitoring and interpretation of antibody levels should be carefully reviewed.
CKD and risk of AKI. Due to the epidemiological association between CKD and AKI and the number of observational studies reporting an association between pre-existing CKD and AKI, CKD is considered the most consistent pre-existing condition associated with a high risk of AKI. However the potential linkage between patients with AKI, CKD, and ESRD has been inadequately studied to date and remains ill defined. This section describes AKI as a complication which needs to be managed in those with CKD. Given its association with progression, it is also described in that section.
- 4.6.7: We recommend that all people with CKD are considered to be at increased risk of AKI. (1A)
- 4.6.7.1: In people with CKD, the recommendations detailed in the KDIGO AKI Guideline should be followed for management of those at risk of AKI during intercurrent illness, or when undergoing investigation and procedures that are likely to increase the risk of AKI. (Not Graded)
RATIONALE
Observational data suggest a strong association between pre-existing CKD and AKI. The appreciation that CKD patients may be more susceptible to AKI is the purpose of the above set of statements. However, methodological issues such as how CKD and AKI are defined in clinical studies and the statistical adjustments for non-uniformity of comorbidities among various studies may affect the validity of observed associations.
Evidence Base
CKD is designated as a risk factor for AKI because of the epidemiological association between the two.263,264 A number of studies in a variety of settings report an association between pre-existing CKD and AKI. 265-271 CKD is a potent predictor of acute decline in kidney function following exposure to radiocontrast,272 major surgery,273 and other medical conditions.274
Hsu et al.14 compared the pre-hospitalization MDRD GFR of 1764 adult members of the Kaiser Permanente Northern California health-care system who developed dialysis-requiring AKI during hospitalization with 600,820 individuals who did not. Compared with a reference baseline GFR of ≥60 ml/min/1.73 m2, a baseline GFR of 45–59 ml/min/1.73 m2 was associated with an adjusted OR of in-hospital AKI of 1.66 (95% CI 1.40–1.97). For GFR values of 15–29 ml/min/1.73 m2, the adjusted OR for in-hospital AKI was 20.42 (95% CI 17.40–23.96). The presence of diabetes, hypertension, and proteinuria increased the likelihood of developing in-hospital AKI, with adjusted ORs of 1.99 (95% CI 1.78–2.23), 1.55 (95% CI 1.37–1.76) and 2.84 (95% CI 2.52–3.19), respectively. The authors concluded that CKD is the main risk factor for AKI during hospitalization. A contrasting approach by Singh et al. defined AKI as dialysis-requiring acute renal failure.275 Because the clinical decision to dialyze a patient is frequently influenced by a higher overall SCr, presence of hemodialysis access, or consideration of inevitable progression to ESRD, this definition of AKI could bias toward capturing more AKI cases in CKD patients. Moreover, in patients with advanced CKD, the progression of CKD to ESRD may sometimes be difficult to separate from acute-on-chronic renal failure. A cohort study by Lafrance et al. followed a referred CKD population in British Columbia for a median of 19.4 months after achieving a GFR of ≤30 ml/min/1.73 m2. Forty-five percent had at least one episode of AKI.276 In another cohort study of 920,985 adults in Alberta, Canada with at least one outpatient measurement of SCr and proteinuria and not requiring chronic dialysis, risk of admission with AKI increased with heavier proteinuria and reduced GFR.16
International Relevance
These guidelines about AKI have relevance around the world. While the causes of AKI may differ by region, country, socio-economic status, and age, the consequences remain the similar. Where there are no facilitities to support AKI or CKD, people will die.
Areas of Controversy, Confusion, or Non-consensus
Interpretation of published data examining the influence of pre-existing CKD on the increased likelihood of AKI is potentially confounded by a number of issues. These include the comorbidities associated with CKD, influenced by repeated exposure to various nephrotoxic insults or in-hospital errors,57,277 or primarily due to the altered physiology in CKD. There are also methodological issues such as how CKD and AKI are defined in clinical studies and the varying statistical adjustments for comorbidities which may affect the validity of observed associations.
A further important issue to clarify is whether pre-existing CKD influences the outcome of AKI. Currently, there is no single biomarker that can differentiate ‘acute' from ‘chronic' kidney disease and help to address this issue. Several large observational and database studies report, surprisingly, lower in-hospital mortality in patients with AKI superimposed on CKD compared with controls.278-283 Data from PICARD reveal lower in-patient mortality and median length of stay in ICU subjects with acute-on-chronic renal injury compared with non-CKD subjects with AKI, though the post-discharge dialysis rates were higher in subjects with pre-existing CKD.284
Pediatric Considerations
The relative paucity of pediatric specific guidelines (due to lack of high-quality studies) in the KDIGO AKI guideline would suggest that the use of pediatric data and review papers as well as relevant pediatric nephrology texts would be of benefit to the practitioner interested in reviewing this topic in greater detail and applying pediatric data to their practice.653, 654, 655, 656, 657, 658
RESEARCH RECOMMENDATIONS
Prospectively designed clinical studies with a clear and uniform definition of CKD and AKI and adjusted for comorbidities are needed to determine:
the frequency of AKI events in a CKD population
the outcome of AKI in patients with CKD condition
the importance of proteinuria in addition to low GFR in the risk of AKI
CKD and risk of hospitalization and mortality
Regardless of the method used to estimate GFR, hospitalization and mortality rates are higher in people with CKD. Exact rates vary with comorbidity and severity of CKD, and are not well-defined. Selection of interventions that could reduce hospitalizations, morbidity, mortality, and costs in people with CKD is not well-studied.
4.6.8: CKD disease management programs should be developed in order to optimize the community management of people with CKD and reduce the risk of hospital admission. (Not Graded)
4.6.9: Interventions to reduce hospitalization and mortality for people with CKD should pay close attention to the management of associated comorbid conditions and cardiovascular disease in particular. (Not Graded)
RATIONALE
There are observational and database studies reporting an association between pre-existing CKD and hospitalizations and mortality. A better understanding of the rates, causes, and risk factors for hospitalization among people with CKD would allow estimates of the economic burden of CKD and identification of those at risk for increased resource utilization. People with CKD are an ideal target for interventions aimed at reduction of morbidity, hospitalization, mortality, and costs. These statements suggest that a coordinated approach to the identification and management would result in better outcomes and are intended as ‘best practices' statements, recognizing the difficulty in developing an evidence base while addressing issues related to resource allocations (Table 34).
Table 34. Components of community CKD management programs.
Evidence Base
Mortality rates remain high (16-22%) with the use of dialysis, with more than half of all deaths related to CVD. Less is known about mortality and CVD rates, and resource use among persons with a reduced GFR who are not yet receiving maintenance dialysis. Few studies have investigated the association between CKD and the risk of hospitalization.
Data from the USRDS reveal that hospitalization rates vary with comorbidity and interact with degrees of CKD. Adjusted rates are 38% higher in people with CKD and 19% greater in people with CKD and GFR under 60 ml/min/ 1.73 m2 than in those with GFR >60 ml/min/1.73 m2, who in turn are 20% higher than in people without CKD-illustrating the graded impact of advancing kidney disease. Not surprisingly, rates of cardiovascular hospitalization are greater for people with CKD, particularly those with increasing severity of CKD.659 In both the CKD and non-CKD populations, adjusted rates of hospitalization increase with greater comorbidity. In 2008, for example, the rate for people with CKD with both diabetes and CHF was 726 per 1000 person-years at risk — 85% greater than the rate of 393 among people with neither diagnosis. Rates of admission for CVD increase even more in the higher categories of GFR. Among Medicare subjects, the rate of 141 admissions per 1000 person-years for those with GFR <60 ml/min/1.73 m2 is 26% higher than the rate of 112 reported for those with CKD and GFR ≥60 ml/min/1.73 m2. The admission rates of 101 and 90 reported for MarketScan and Ingenix i3 subjects with GFR <60 ml/min/1.73 m2 are 48 and 16% greater, respectively, than those occurring in people with higher GFRs.633 Adjusted rates of mortality in USRDS 2008 increased with age, and were highest in people with advanced categories of GFR: 31–72% higher, for example, in people with GFR <60 ml/min/1.73 m2 compared to those with no CKD. By gender, rates in men with CKD were 91.8 per 1000 person-years at risk compared to 85.6 in women. Rates for people with CKD overall were similar in whites and African Americans, but in people with GFR <60 ml/min/1.73 m2, rates for African Americans were 18% higher than those for whites, at 95.0 and 80.5 per 1000 person-years, respectively.633
Khan et al.660 confirmed that hospital utilization among people with CKD is high. During a median follow-up of 11.4 months, 47% of subjects had at least one hospitalization and there were on average 0.96 hospitalizations, 6.6 hospital days, and 4.0 outpatient nephrology visits per person-year at risk. Cardiac disease/hypertension was the most common primary diagnosis of hospitalizations and progression of CKD/acute kidney failure was the most common secondary cause of hospitalization. The authors had previously shown that the dialysis population at their institution had 2.2 hospitalizations and 14.8 hospital days per person-year at risk,659 which was similar to that among US hemodialysis patients between 1996 and 1998, who had 1.9 hospitalizations and 14 hospital days per person-year at risk.661 In the general population, there were 0.31 hospitalizations and 1.9 hospital days per person in 1998.662
Go et al.58 reported an independent, graded association between GFR and the risk of death, cardiovascular events, and hospitalization in 1,120,295 adults within a large, integrated system of health-care delivery in whom SCr had been measured between 1996 and 2000 and who had not undergone dialysis or kidney transplantation. These risks were evident at GFR <60 ml/min/1.73 m2 and substantially increased at GFR <45 ml/min/1.73 m2.
The higher hospital utilization among people with CKD compared to the general population, and the similarity in the comorbid conditions and the causes of hospitalization between people with CKD and ESRD, confirm the hypothesis that the complications and comorbidity observed in ESRD are manifest earlier in the disease process. Previous studies have demonstrated an association between age, gender, race, cardiac disease, peripheral vascular disease, serum albumin and hematocrit levels, and resource utilization among people on dialysis.659, 663, 664 Holland et al.665 identified baseline predictors of first non-elective hospitalization among a retrospective cohort of 362 predialysis subjects. Multivariate analysis, adjusted for baseline creatinine concentration, selected advanced age (RR 1.02; 95% CI 1.01–1.03), angina (RR 1.9; CI 1.37–2.61), peripheral vascular disease (RR 1.55, CI 1.05–2.27), and Hb concentration (RR 0.99, CI 0.94–0.98) as independent predictors of hospitalization. These comorbid conditions progressively worsen with advancing kidney disease and result in a substantial proportion of people having severe complications by the time they come to RRT.
Interventions to reduce hospitalizations and mortality for people with CKD should pay close attention to the management of associated comorbid conditions and CVD.666 Closer attention to the management of heart disease in this population could substantially improve outcomes.667
The influence of correction of anemia on hospitalization is controversial. Drüeke et al.668 randomly assigned 603 subjects with GFR 15-35 ml/min/1.73 m2 and mild-to-moderate anemia (Hb level: 11.0-12.5 g/dl [110-125 g/l]) to a target Hb value in the normal range (13.0-15.0 g/dl [130-150 g/l]) or the subnormal range (10.5-11.5 g/dl [105-115 g/l]). There were no significant differences between the two groups in the incidence of hospital admission (61% and 59%, respectively) or mean duration of hospitalization for cardiovascular reasons (33.0 and 28.2 days, respectively). Singh et al.669 studied 1432 people with CKD, 715 of whom were randomly assigned to receive a dose of epoetin alfa targeted to achieve a Hb level of 13.5 g/dl (135 g/l) and 717 of whom were assigned to receive a dose targeted to achieve a level of 11.3 g/dl (113 g/l). The median study duration was 16 months. The primary end point was a composite of death, MI, hospitalization for CHF (without RRT), and stroke. They observed an increased risk of the primary composite end point in the high Hb group as compared with the low Hb group. Death and hospitalization for CHF accounted for 74.8% of the composite events. The Trial to Reduce Cardiovascular Events with Aranesp Therapy (TREAT)375 study involved 4038 people with diabetes, CKD, and anemia. Subjects were randomly assigned to treatment with darbepoetin alfa to achieve a Hb level of approximately 13 g/dl (130 g/l) or to placebo, with rescue darbepoetin alfa when the Hb level was less than 9.0 g/dl (90 g/l). The primary end points were the composite outcomes of death or a cardiovascular event (nonfatal MI, CHF, stroke, or hospitalization for myocardial ischemia) and of death or ESRD. Again there were no significant between-group differences in the outcomes of interest.
International Relevance
It is of benefit for all jurisdictions to appreciate the increase in resource utilization by CKD populations. Identification of people at risk for increased resource utilization and economic burden of CKD should result in strategies to attenuate that risk or address the resource implications. Selection of interventions that could reduce hospitalizations, morbidity, mortality, and costs in these populations should be evaluated.
DISCLAIMER
While every effort is made by the publishers, editorial board, and ISN to see that no inaccurate or misleading data, opinion or statement appears in this Journal, they wish to make it clear that the data and opinions appearing in the articles and advertisements herein are the responsibility of the contributor, copyright holder, or advertiser concerned. Accordingly, the publishers and the ISN, the editorial board and their respective employers, office and agents accept no liability whatsoever for the consequences of any such inaccurate or misleading data, opinion or statement. While every effort is made to ensure that drug doses and other quantities are presented accurately, readers are advised that new methods and techniques involving drug usage, and described within this Journal, should only be followed in conjunction with the drug manufacturer's own published literature.
References
- Hallan SI, Dahl K, Oien CM, et al. Screening strategies for chronic kidney disease in the general population: follow-up of cross sectional health survey. BMJ. 2006;333:1047. doi: 10.1136/bmj.39001.657755.BE. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McCullough PA, Li S, Jurkovitz CT, et al. Chronic kidney disease, prevalence of premature cardiovascular disease, and relationship to short-term mortality. Am Heart J. 2008;156:277–283. doi: 10.1016/j.ahj.2008.02.024. [DOI] [PubMed] [Google Scholar]
- Anavekar NS, McMurray JJ, Velazquez EJ, et al. Relation between renal dysfunction and cardiovascular outcomes after myocardial infarction. N Engl J Med. 2004;351:1285–1295. doi: 10.1056/NEJMoa041365. [DOI] [PubMed] [Google Scholar]
- Shlipak MG, Heidenreich PA, Noguchi H, et al. Association of renal insufficiency with treatment and outcomes after myocardial infarction in elderly patients. Ann Intern Med. 2002;137:555–562. doi: 10.7326/0003-4819-137-7-200210010-00006. [DOI] [PubMed] [Google Scholar]
- Wright RS, Reeder GS, Herzog CA, et al. Acute myocardial infarction and renal dysfunction: a high-risk combination. Ann Intern Med. 2002;137:563–570. doi: 10.7326/0003-4819-137-7-200210010-00007. [DOI] [PubMed] [Google Scholar]
- Sarnak MJ. Cardiovascular complications in chronic kidney disease. Am J Kidney Dis. 2003;41:11–17. doi: 10.1016/s0272-6386(03)00372-x. [DOI] [PubMed] [Google Scholar]
- Hemmelgarn BR, Manns BJ, Lloyd A, et al. Relation between kidney function, proteinuria, and adverse outcomes. JAMA. 2010;303:423–429. doi: 10.1001/jama.2010.39. [DOI] [PubMed] [Google Scholar]
- Klausen K, Borch-Johnsen K, Feldt-Rasmussen B, et al. Very low levels of microalbuminuria are associated with increased risk of coronary heart disease and death independently of renal function, hypertension, and diabetes. Circulation. 2004;110:32–35. doi: 10.1161/01.CIR.0000133312.96477.48. [DOI] [PubMed] [Google Scholar]
- Gerstein HC, Mann JF, Yi Q, et al. Albuminuria and risk of cardiovascular events, death, and heart failure in diabetic and nondiabetic individuals. JAMA. 2001;286:421–426. doi: 10.1001/jama.286.4.421. [DOI] [PubMed] [Google Scholar]
- Hallan S, Astor B, Romundstad S, et al. Association of kidney function and albuminuria with cardiovascular mortality in older vs younger individuals: The II HUNT Study. Arch Intern Med. 2007;167:2490–2496. doi: 10.1001/archinte.167.22.2490. [DOI] [PubMed] [Google Scholar]
- Ibsen H, Wachtell K, Olsen MH, et al. Albuminuria and cardiovascular risk in hypertensive patients with left ventricular hypertrophy: the LIFE Study. Kidney Int Suppl. 2004. pp. S56–58. [DOI] [PubMed]
- Menon V, Shlipak MG, Wang X, et al. Cystatin C as a risk factor for outcomes in chronic kidney disease. Ann Intern Med. 2007;147:19–27. doi: 10.7326/0003-4819-147-1-200707030-00004. [DOI] [PubMed] [Google Scholar]
- Peralta CA, Katz R, Sarnak MJ, et al. Cystatin C identifies chronic kidney disease patients at higher risk for complications. J Am Soc Nephrol. 2011;22:147–155. doi: 10.1681/ASN.2010050483. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Phrommintikul A, Haas SJ, Elsik M, et al. Mortality and target haemoglobin concentrations in anaemic patients with chronic kidney disease treated with erythropoietin: a meta-analysis. Lancet. 2007;369:381–388. doi: 10.1016/S0140-6736(07)60194-9. [DOI] [PubMed] [Google Scholar]
- Strippoli GF, Tognoni G, Navaneethan SD, et al. Haemoglobin targets: we were wrong, time to move on. Lancet. 2007;369:346–350. doi: 10.1016/S0140-6736(07)60165-2. [DOI] [PubMed] [Google Scholar]
- Eckardt KU, Scherhag A, Macdougall IC, et al. Left ventricular geometry predicts cardiovascular outcomes associated with anemia correction in CKD. J Am Soc Nephrol. 2009;20:2651–2660. doi: 10.1681/ASN.2009060631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kestenbaum B, Sampson JN, Rudser KD, et al. Serum phosphate levels and mortality risk among people with chronic kidney disease. J Am Soc Nephrol. 2005;16:520–528. doi: 10.1681/ASN.2004070602. [DOI] [PubMed] [Google Scholar]
- Groothoff JW, Gruppen MP, Offringa M, et al. Mortality and causes of death of end-stage renal disease in children: a Dutch cohort study. Kidney Int. 2002;61:621–629. doi: 10.1046/j.1523-1755.2002.00156.x. [DOI] [PubMed] [Google Scholar]
- McDonald SP, Craig JC. Long-term survival of children with end-stage renal disease. N Engl J Med. 2004;350:2654–2662. doi: 10.1056/NEJMoa031643. [DOI] [PubMed] [Google Scholar]
- Oh J, Wunsch R, Turzer M, et al. Advanced coronary and carotid arteriopathy in young adults with childhood-onset chronic renal failure. Circulation. 2002;106:100–105. doi: 10.1161/01.cir.0000020222.63035.c0. [DOI] [PubMed] [Google Scholar]
- Parekh RS, Carroll CE, Wolfe RA, et al. Cardiovascular mortality in children and young adults with end-stage kidney disease. J Pediatr. 2002;141:191–197. doi: 10.1067/mpd.2002.125910. [DOI] [PubMed] [Google Scholar]
- Kavey RE, Allada V, Daniels SR, et al. Cardiovascular risk reduction in high-risk pediatric patients: a scientific statement from the American Heart Association Expert Panel on Population and Prevention Science; the Councils on Cardiovascular Disease in the Young, Epidemiology and Prevention, Nutrition, Physical Activity and Metabolism, High Blood Pressure Research, Cardiovascular Nursing, and the Kidney in Heart Disease; and the Interdisciplinary Working Group on Quality of Care and Outcomes Research: endorsed by the American Academy of Pediatrics. Circulation. 2006;114:2710–2738. doi: 10.1161/CIRCULATIONAHA.106.179568. [DOI] [PubMed] [Google Scholar]
- Wilson AC, Schneider MF, Cox C, et al. Prevalence and correlates of multiple cardiovascular risk factors in children with chronic kidney disease. Clin J Am Soc Nephrol. 2011;6:2759–2765. doi: 10.2215/CJN.03010311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Litwin M, Wuhl E, Jourdan C, et al. Altered morphologic properties of large arteries in children with chronic renal failure and after renal transplantation. J Am Soc Nephrol. 16:1494–1500. doi: 10.1681/ASN.2004110932. [DOI] [PubMed] [Google Scholar]
- Mitsnefes MM, Kimball TR, Kartal J, et al. Cardiac and vascular adaptation in pediatric patients with chronic kidney disease: role of calcium-phosphorus metabolism. J Am Soc Nephrol. 2005;16:2796–2803. doi: 10.1681/ASN.2005030291. [DOI] [PubMed] [Google Scholar]
- Mitsnefes MM. Cardiovascular disease in children with chronic kidney disease. J Am Soc Nephrol. 2012;23:578–585. doi: 10.1681/ASN.2011111115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shroff R, Weaver DJ., Jr Mitsnefes MM. Cardiovascular complications in children with chronic kidney disease. Nat Rev Nephrol. 2011;7:642–649. doi: 10.1038/nrneph.2011.116. [DOI] [PubMed] [Google Scholar]
- Halimi JM, Giraudeau B, Vol S, et al. Effects of current smoking and smoking discontinuation on renal function and proteinuria in the general population. Kidney Int. 2000;58:1285–1292. doi: 10.1046/j.1523-1755.2000.00284.x. [DOI] [PubMed] [Google Scholar]
- Boyce ML, Robergs RA, Avasthi PS, et al. Exercise training by individuals with predialysis renal failure: cardiorespiratory endurance, hypertension, and renal function. Am J Kidney Dis. 1997;30:180–192. doi: 10.1016/s0272-6386(97)90051-2. [DOI] [PubMed] [Google Scholar]
- Baigent C, Landray MJ, Reith C, et al. The effects of lowering LDL cholesterol with simvastatin plus ezetimibe in patients with chronic kidney disease (Study of Heart and Renal Protection): a randomised placebo-controlled trial. Lancet. 2011;377:2181–2192. doi: 10.1016/S0140-6736(11)60739-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jardine MJ, Ninomiya T, Perkovic V, et al. Aspirin is beneficial in hypertensive patients with chronic kidney disease: a post-hoc subgroup analysis of a randomized controlled trial. J Am Coll Cardiol. 2010;56:956–965. doi: 10.1016/j.jacc.2010.02.068. [DOI] [PubMed] [Google Scholar]
- Angiolillo DJ, Bernardo E, Capodanno D, et al. Impact of chronic kidney disease on platelet function profiles in diabetes mellitus patients with coronary artery disease taking dual antiplatelet therapy. J Am Coll Cardiol. 2010;55:1139–1146. doi: 10.1016/j.jacc.2009.10.043. [DOI] [PubMed] [Google Scholar]
- Best PJ, Steinhubl SR, Berger PB, et al. The efficacy and safety of short- and long-term dual antiplatelet therapy in patients with mild or moderate chronic kidney disease: results from the Clopidogrel for the Reduction of Events During Observation (CREDO) trial. Am Heart J. 2008;155:687–693. doi: 10.1016/j.ahj.2007.10.046. [DOI] [PubMed] [Google Scholar]
- Graham I, Atar D, Borch-Johnsen K, et al. European guidelines on cardiovascular disease prevention in clinical practice: executive summary. Fourth Joint Task Force of the European Society of Cardiology and other societies on cardiovascular disease prevention in clinical practice (constituted by representatives of nine societies and by invited experts) Eur J Cardiovasc Prev Rehabil. 2007;14 Suppl 2:E1–40. doi: 10.1097/01.hjr.0000277984.31558.c4. [DOI] [PubMed] [Google Scholar]
- Hippisley-Cox J, Coupland C, Vinogradova Y, et al. Predicting cardiovascular risk in England and Wales: prospective derivation and validation of QRISK2. BMJ. 2008;336:1475–1482. doi: 10.1136/bmj.39609.449676.25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Joint British Societies. JBS 2: Joint British Societies' guidelines on prevention of cardiovascular disease in clinical practice. Heart. 2005;91 Suppl 5:v1–52. doi: 10.1136/hrt.2005.079988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu Y, Liu X, Li X, et al. Estimation of 10-year risk of fatal and nonfatal ischemic cardiovascular diseases in Chinese adults. Circulation. 2006;114:2217–2225. doi: 10.1161/CIRCULATIONAHA.105.607499. [DOI] [PubMed] [Google Scholar]
- Liau SY, Izham MI, Hassali MA, et al. A literature review of the cardiovascular risk-assessment tools: applicability among Asian population. Heart Asia. 2010;2:15–18. doi: 10.1136/ha.2009.001115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakayama M, Sato T, Sato H, et al. Different clinical outcomes for cardiovascular events and mortality in chronic kidney disease according to underlying renal disease: the Gonryo study. Clin Exp Nephrol. 2010;14:333–339. doi: 10.1007/s10157-010-0295-y. [DOI] [PubMed] [Google Scholar]
- Irie F, Iso H, Sairenchi T, et al. The relationships of proteinuria, serum creatinine, glomerular filtration rate with cardiovascular disease mortality in Japanese general population. Kidney Int. 2006;69:1264–1271. doi: 10.1038/sj.ki.5000284. [DOI] [PubMed] [Google Scholar]
- Yang JG, Li J, Lu C, et al. Chronic kidney disease, all-cause mortality and cardiovascular mortality among Chinese patients with established cardiovascular disease. J Atheroscler Thromb. 2010;17:395–401. doi: 10.5551/jat.3061. [DOI] [PubMed] [Google Scholar]
- Zhang L, Zuo L, Wang F, et al. Cardiovascular disease in early stages of chronic kidney disease in a Chinese population. J Am Soc Nephrol. 2006;17:2617–2621. doi: 10.1681/ASN.2006040402. [DOI] [PubMed] [Google Scholar]
- Banerjee D, Chitalia N, Raja R, et al. Metabolic syndrome in chronic kidney disease and renal transplant patients in North India. Int Urol Nephrol. 2011. [DOI] [PubMed]
- Di Angelantonio E, Chowdhury R, Sarwar N, et al. Chronic kidney disease and risk of major cardiovascular disease and non-vascular mortality: prospective population based cohort study. BMJ. 2010;341:c4986. doi: 10.1136/bmj.c4986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parikh NI, Hwang SJ, Larson MG, et al. Chronic kidney disease as a predictor of cardiovascular disease (from the Framingham Heart Study) Am J Cardiol. 2008;102:47–53. doi: 10.1016/j.amjcard.2008.02.095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eiland LS, Luttrell PK. Use of statins for dyslipidemia in the pediatric population. J Pediatr Pharmacol Ther. 2010;15:160–172. [PMC free article] [PubMed] [Google Scholar]
- McAlister FA, Ezekowitz J, Tonelli M, et al. Renal insufficiency and heart failure: prognostic and therapeutic implications from a prospective cohort study. Circulation. 2004;109:1004–1009. doi: 10.1161/01.CIR.0000116764.53225.A9. [DOI] [PubMed] [Google Scholar]
- Smith GL, Lichtman JH, Bracken MB, et al. Renal impairment and outcomes in heart failure: systematic review and meta-analysis. J Am Coll Cardiol. 2006;47:1987–1996. doi: 10.1016/j.jacc.2005.11.084. [DOI] [PubMed] [Google Scholar]
- Hillege HL, Nitsch D, Pfeffer MA, et al. Renal function as a predictor of outcome in a broad spectrum of patients with heart failure. Circulation. 2006;113:671–678. doi: 10.1161/CIRCULATIONAHA.105.580506. [DOI] [PubMed] [Google Scholar]
- Shlipak MG, Smith GL, Rathore SS, et al. Renal function, digoxin therapy, and heart failure outcomes: evidence from the digoxin intervention group trial. J Am Soc Nephrol. 2004;15:2195–2203. doi: 10.1097/01.ASN.0000135121.81744.75. [DOI] [PubMed] [Google Scholar]
- Smith GL, Shlipak MG, Havranek EP, et al. Race and renal impairment in heart failure: mortality in blacks versus whites. Circulation. 2005;111:1270–1277. doi: 10.1161/01.CIR.0000158131.78881.D5. [DOI] [PubMed] [Google Scholar]
- Anand IS, Bishu K, Rector TS, et al. Proteinuria, chronic kidney disease, and the effect of an angiotensin receptor blocker in addition to an angiotensin-converting enzyme inhibitor in patients with moderate to severe heart failure. Circulation. 2009;120:1577–1584. doi: 10.1161/CIRCULATIONAHA.109.853648. [DOI] [PubMed] [Google Scholar]
- Ezekowitz J, McAlister FA, Humphries KH, et al. The association among renal insufficiency, pharmacotherapy, and outcomes in 6,427 patients with heart failure and coronary artery disease. J Am Coll Cardiol. 2004;44:1587–1592. doi: 10.1016/j.jacc.2004.06.072. [DOI] [PubMed] [Google Scholar]
- Gottlieb SS, Abraham W, Butler J, et al. The prognostic importance of different definitions of worsening renal function in congestive heart failure. J Card Fail. 2002;8:136–141. doi: 10.1054/jcaf.2002.125289. [DOI] [PubMed] [Google Scholar]
- Middleton RJ, Parfrey PS, Foley RN. Left ventricular hypertrophy in the renal patient. J Am Soc Nephrol. 2001;12:1079–1084. doi: 10.1681/ASN.V1251079. [DOI] [PubMed] [Google Scholar]
- Levin A, Singer J, Thompson CR, et al. Prevalent left ventricular hypertrophy in the predialysis population: identifying opportunities for intervention. Am J Kidney Dis. 1996;27:347–354. doi: 10.1016/s0272-6386(96)90357-1. [DOI] [PubMed] [Google Scholar]
- Ha SK, Park HS, Kim SJ, et al. Prevalence and patterns of left ventricular hypertrophy in patients with predialysis chronic renal failure. J Korean Med Sci. 1998;13:488–494. doi: 10.3346/jkms.1998.13.5.488. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parfrey PS, Foley RN, Harnett JD, et al. Outcome and risk factors for left ventricular disorders in chronic uraemia. Nephrol Dial Transplant. 1996;11:1277–1285. [PubMed] [Google Scholar]
- Cerasola G, Nardi E, Palermo A, et al. Epidemiology and pathophysiology of left ventricular abnormalities in chronic kidney disease: a review. J Nephrol. 2011;24:1–10. doi: 10.5301/jn.2010.2030. [DOI] [PubMed] [Google Scholar]
- Ahmed A, Rich MW, Sanders PW, et al. Chronic kidney disease associated mortality in diastolic versus systolic heart failure: a propensity matched study. Am J Cardiol. 2007;99:393–398. doi: 10.1016/j.amjcard.2006.08.042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lazzeri C, Valente S, Tarquini R, et al. Cardiorenal syndrome caused by heart failure with preserved ejection fraction. Int J Nephrol. 2011;2011:634903. doi: 10.4061/2011/634903. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Levin A, Thompson CR, Ethier J, et al. Left ventricular mass index increase in early renal disease: impact of decline in hemoglobin. Am J Kidney Dis. 1999;34:125–134. doi: 10.1016/s0272-6386(99)70118-6. [DOI] [PubMed] [Google Scholar]
- Go AS, Yang J, Ackerson LM, et al. Hemoglobin level, chronic kidney disease, and the risks of death and hospitalization in adults with chronic heart failure: the Anemia in Chronic Heart Failure: Outcomes and Resource Utilization (ANCHOR) Study. Circulation. 2006;113:2713–2723. doi: 10.1161/CIRCULATIONAHA.105.577577. [DOI] [PubMed] [Google Scholar]
- Shlipak MG, Katz R, Kestenbaum B, et al. Clinical and subclinical cardiovascular disease and kidney function decline in the elderly. Atherosclerosis. 2009;204:298–303. doi: 10.1016/j.atherosclerosis.2008.08.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Balamuthusamy S, Srinivasan L, Verma M, et al. Renin angiotensin system blockade and cardiovascular outcomes in patients with chronic kidney disease and proteinuria: a meta-analysis. Am Heart J. 2008;155:791–805. doi: 10.1016/j.ahj.2008.01.031. [DOI] [PubMed] [Google Scholar]
- Lewis EJ, Hunsicker LG, Clarke WR, et al. Renoprotective effect of the angiotensin-receptor antagonist irbesartan in patients with nephropathy due to type 2 diabetes. N Engl J Med. 2001;345:851–860. doi: 10.1056/NEJMoa011303. [DOI] [PubMed] [Google Scholar]
- Erdmann E, Lechat P, Verkenne P, et al. Results from post-hoc analyses of the II CIBIS trial: effect of bisoprolol in high-risk patient groups with chronic heart failure. Eur J Heart Fail. 2001;3:469–479. doi: 10.1016/s1388-9842(01)00174-x. [DOI] [PubMed] [Google Scholar]
- Wali RK, Iyengar M, Beck GJ, et al. Efficacy and safety of carvedilol in treatment of heart failure with chronic kidney disease: a meta-analysis of randomized trials. Circ Heart Fail. 2011;4:18–26. doi: 10.1161/CIRCHEARTFAILURE.109.932558. [DOI] [PubMed] [Google Scholar]
- Pitt B, Zannad F, Remme WJ, et al. The effect of spironolactone on morbidity and mortality in patients with severe heart failure. Randomized Aldactone Evaluation Study Investigators. N Engl J Med. 1999;341:709–717. doi: 10.1056/NEJM199909023411001. [DOI] [PubMed] [Google Scholar]
- Juurlink DN, Mamdani MM, Lee DS, et al. Rates of hyperkalemia after publication of the Randomized Aldactone Evaluation Study. N Engl J Med. 2004;351:543–551. doi: 10.1056/NEJMoa040135. [DOI] [PubMed] [Google Scholar]
- Abdel-Qadir HM, Chugh S, Lee DS. Improving prognosis estimation in patients with heart failure and the cardiorenal syndrome. Int J Nephrol. 2011;2011:351672. doi: 10.4061/2011/351672. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Levin ER, Gardner DG, Samson WK. Natriuretic peptides. N Engl J Med. 1998;339:321–328. doi: 10.1056/NEJM199807303390507. [DOI] [PubMed] [Google Scholar]
- Wiley CL, Switzer SP, Berg RL, et al. Association of B-type natriuretic Peptide levels with estimated glomerular filtration rate and congestive heart failure. Clin Med Res. 2010;8:7–12. doi: 10.3121/cmr.2009.867. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Madsen LH, Ladefoged S, Corell P, et al. N-terminal pro brain natriuretic peptide predicts mortality in patients with end-stage renal disease in hemodialysis. Kidney Int. 2007;71:548–554. doi: 10.1038/sj.ki.5002087. [DOI] [PubMed] [Google Scholar]
- Gustafsson F, Steensgaard-Hansen F, Badskjaer J, et al. Diagnostic and prognostic performance of N-terminal ProBNP in primary care patients with suspected heart failure. J Card Fail. 2005;11:S15–S20. doi: 10.1016/j.cardfail.2005.04.022. [DOI] [PubMed] [Google Scholar]
- McCullough PA, Omland T, Maisel AS. B-type natriuretic peptides: a diagnostic breakthrough for clinicians. Rev Cardiovasc Med. 2003;4:72–80. [PubMed] [Google Scholar]
- Maisel A, Mehra MR. Understanding B-type natriuretic peptide and its role in diagnosing and monitoring congestive heart failure. Clin Cornerstone. 2005;7 Suppl 1:S7–17. doi: 10.1016/s1098-3597(05)80009-2. [DOI] [PubMed] [Google Scholar]
- Wright SP, Doughty RN, Pearl A, et al. Plasma amino-terminal pro-brain natriuretic peptide and accuracy of heart-failure diagnosis in primary care: a randomized, controlled trial. J Am Coll Cardiol. 2003;42:1793–1800. doi: 10.1016/j.jacc.2003.05.011. [DOI] [PubMed] [Google Scholar]
- Yamamoto K, Burnett JC., Jr Jougasaki M, et al. Superiority of brain natriuretic peptide as a hormonal marker of ventricular systolic and diastolic dysfunction and ventricular hypertrophy. Hypertension. 1996;28:988–994. doi: 10.1161/01.hyp.28.6.988. [DOI] [PubMed] [Google Scholar]
- Burnett JC., Jr Kao PC, Hu DC, et al. Atrial natriuretic peptide elevation in congestive heart failure in the human. Science. 1986;231:1145–1147. doi: 10.1126/science.2935937. [DOI] [PubMed] [Google Scholar]
- Mueller C, Laule-Kilian K, Scholer A, et al. B-type natriuretic peptide for acute dyspnea in patients with kidney disease: insights from a randomized comparison. Kidney Int. 2005;67:278–284. doi: 10.1111/j.1523-1755.2005.00079.x. [DOI] [PubMed] [Google Scholar]
- Vickery S, Price CP, John RI, et al. B-type natriuretic peptide (BNP) and amino-terminal proBNP in patients with CKD: relationship to renal function and left ventricular hypertrophy. Am J Kidney Dis. 2005;46:610–620. doi: 10.1053/j.ajkd.2005.06.017. [DOI] [PubMed] [Google Scholar]
- Ishibe S, Peixoto AJ. Methods of assessment of volume status and intercompartmental fluid shifts in hemodialysis patients: implications in clinical practice. Semin Dial. 2004;17:37–43. doi: 10.1111/j.1525-139x.2004.17112.x. [DOI] [PubMed] [Google Scholar]
- Takahashi M, Nagake Y, Ichikawa H, et al. Plasma concentrations of natriuretic peptides in patients on hemodialysis. Res Commun Mol Pathol Pharmacol. 1996;92:19–30. [PubMed] [Google Scholar]
- Tagore R, Ling LH, Yang H, et al. Natriuretic peptides in chronic kidney disease. Clin J Am Soc Nephrol. 2008;3:1644–1651. doi: 10.2215/CJN.00850208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takami Y, Horio T, Iwashima Y, et al. Diagnostic and prognostic value of plasma brain natriuretic peptide in non-dialysis-dependent CRF. Am J Kidney Dis. 2004;44:420–428. [PubMed] [Google Scholar]
- Zoccali C, Mallamaci F, Benedetto FA, et al. Cardiac natriuretic peptides are related to left ventricular mass and function and predict mortality in dialysis patients. J Am Soc Nephrol. 2001;12:1508–1515. doi: 10.1681/ASN.V1271508. [DOI] [PubMed] [Google Scholar]
- Suresh M, Farrington K. Natriuretic peptides and the dialysis patient. Semin Dial. 2005;18:409–419. doi: 10.1111/j.1525-139X.2005.17349.x. [DOI] [PubMed] [Google Scholar]
- Kistorp C, Raymond I, Pedersen F, et al. N-terminal pro-brain natriuretic peptide, C-reactive protein, and urinary albumin levels as predictors of mortality and cardiovascular events in older adults. JAMA. 2005;293:1609–1616. doi: 10.1001/jama.293.13.1609. [DOI] [PubMed] [Google Scholar]
- Bettencourt P, Azevedo A, Pimenta J, et al. N-terminal-pro-brain natriuretic peptide predicts outcome after hospital discharge in heart failure patients. Circulation. 2004;110:2168–2174. doi: 10.1161/01.CIR.0000144310.04433.BE. [DOI] [PubMed] [Google Scholar]
- Gardner RS, Ozalp F, Murday AJ, et al. N-terminal pro-brain natriuretic peptide. A new gold standard in predicting mortality in patients with advanced heart failure. Eur Heart J. 2003;24:1735–1743. doi: 10.1016/j.ehj.2003.07.005. [DOI] [PubMed] [Google Scholar]
- Kragelund CB, Gronning BA, Kober L, et al. Prognostic value of N-terminal pro-BNP-type natriuretic peptide in patients with stable coronary heart disease--secondary publication] Ugeskr Laeger. 2006;168:697–700. [PubMed] [Google Scholar]
- Omland T, Persson A, Ng L, et al. N-terminal pro-B-type natriuretic peptide and long-term mortality in acute coronary syndromes. Circulation. 2002;106:2913–2918. doi: 10.1161/01.cir.0000041661.63285.ae. [DOI] [PubMed] [Google Scholar]
- Olsen MH, Wachtell K, Tuxen C, et al. N-terminal pro-brain natriuretic peptide predicts cardiovascular events in patients with hypertension and left ventricular hypertrophy: a LIFE study. J Hypertens. 2004;22:1597–1604. doi: 10.1097/01.hjh.0000125451.28861.2a. [DOI] [PubMed] [Google Scholar]
- Koch A, Singer H. Normal values of B type natriuretic peptide in infants, children, and adolescents. Heart. 2003;89:875–878. doi: 10.1136/heart.89.8.875. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Auerbach SR, Richmond ME, Lamour JM, et al. BNP levels predict outcome in pediatric heart failure patients: post hoc analysis of the Pediatric Carvedilol Trial. Circ Heart Fail. 2010;3:606–611. doi: 10.1161/CIRCHEARTFAILURE.109.906875. [DOI] [PubMed] [Google Scholar]
- Barret BJ, Culleton B. Reducing the burden of cardiovascular disease in patients on dialysis. Dial Transplant. 2002;31:155–163. [Google Scholar]
- Beciani M, Tedesco A, Violante A, et al. Cardiac troponin I (2nd generation assay) in chronic haemodialysis patients: prevalence and prognostic value. Nephrol Dial Transplant. 2003;18:942–946. doi: 10.1093/ndt/gfg057. [DOI] [PubMed] [Google Scholar]
- Apple FS, Wu AH. Myocardial infarction redefined: role of cardiac troponin testing. Clin Chem. 2001;47:377–379. [PubMed] [Google Scholar]
- McLaurin MD, Apple FS, Voss EM, et al. Cardiac troponin I, cardiac troponin T, and creatine kinase MB in dialysis patients without ischemic heart disease: evidence of cardiac troponin T expression in skeletal muscle. Clin Chem. 1997;43:976–982. [PubMed] [Google Scholar]
- Musso P, Cox I, Vidano E, et al. Cardiac troponin elevations in chronic renal failure: prevalence and clinical significance. Clin Biochem. 1999;32:125–130. doi: 10.1016/s0009-9120(98)00089-7. [DOI] [PubMed] [Google Scholar]
- Alpert JS, Thygesen K, Antman E, et al. Myocardial infarction redefined--a consensus document of The Joint European Society of Cardiology/American College of Cardiology Committee for the redefinition of myocardial infarction. J Am Coll Cardiol. 2000;36:959–969. doi: 10.1016/s0735-1097(00)00804-4. [DOI] [PubMed] [Google Scholar]
- Collinson PO, Boa FG, Gaze DC. Measurement of cardiac troponins. Ann Clin Biochem. 2001;38:423–449. doi: 10.1177/000456320103800501. [DOI] [PubMed] [Google Scholar]
- Lindahl B, Venge P, Wallentin L. Troponin T identifies patients with unstable coronary artery disease who benefit from long-term antithrombotic protection. Fragmin in Unstable Coronary Artery Disease (FRISC) Study Group. J Am Coll Cardiol. 1997;29:43–48. doi: 10.1016/s0735-1097(96)00447-0. [DOI] [PubMed] [Google Scholar]
- Rabbani LE. Acute coronary syndromes--beyond myocyte necrosis. N Engl J Med. 2001;345:1057–1059. doi: 10.1056/NEJM200110043451410. [DOI] [PubMed] [Google Scholar]
- Tun A, Khan IA, Win MT, et al. Specificity of cardiac troponin I and creatine kinase-MB isoenzyme in asymptomatic long-term hemodialysis patients and effect of hemodialysis on these cardiac markers. Cardiology. 1998;90:280–285. doi: 10.1159/000006859. [DOI] [PubMed] [Google Scholar]
- Wood GN, Keevil B, Gupta J, et al. Serum troponin T measurement in patients with chronic renal impairment predicts survival and vascular disease: a 2 year prospective study. Nephrol Dial Transplant. 2003;18:1610–1615. doi: 10.1093/ndt/gfg198. [DOI] [PubMed] [Google Scholar]
- Abbas NA, John RI, Webb MC, et al. Cardiac troponins and renal function in nondialysis patients with chronic kidney disease. Clin Chem. 2005;51:2059–2066. doi: 10.1373/clinchem.2005.055665. [DOI] [PubMed] [Google Scholar]
- Kinnunen P, Vuolteenaho O, Ruskoaho H. Mechanisms of atrial and brain natriuretic peptide release from rat ventricular myocardium: effect of stretching. Endocrinology. 1993;132:1961–1970. doi: 10.1210/endo.132.5.8477647. [DOI] [PubMed] [Google Scholar]
- deFilippi C, Wasserman S, Rosanio S, et al. Cardiac troponin T and C-reactive protein for predicting prognosis, coronary atherosclerosis, and cardiomyopathy in patients undergoing long-term hemodialysis. JAMA. 2003;290:353–359. doi: 10.1001/jama.290.3.353. [DOI] [PubMed] [Google Scholar]
- Hayashi T, Obi Y, Kimura T, et al. Cardiac troponin T predicts occult coronary artery stenosis in patients with chronic kidney disease at the start of renal replacement therapy. Nephrol Dial Transplant. 2008;23:2936–2942. doi: 10.1093/ndt/gfn181. [DOI] [PubMed] [Google Scholar]
- Roongsritong C, Warraich I, Bradley C. Common causes of troponin elevations in the absence of acute myocardial infarction: incidence and clinical significance. Chest. 2004;125:1877–1884. doi: 10.1378/chest.125.5.1877. [DOI] [PubMed] [Google Scholar]
- Apple FS, Murakami MM, Pearce LA, et al. Multi-biomarker risk stratification of N-terminal pro-B-type natriuretic peptide, high-sensitivity C-reactive protein, and cardiac troponin T and I in end-stage renal disease for all-cause death. Clin Chem. 2004;50:2279–2285. doi: 10.1373/clinchem.2004.035741. [DOI] [PubMed] [Google Scholar]
- De Zoysa JR. Cardiac troponins and renal disease. Nephrology (Carlton) 2004;9:83–88. doi: 10.1111/j.1440-1797.2003.00235.x. [DOI] [PubMed] [Google Scholar]
- Deegan PB, Lafferty ME, Blumsohn A, et al. Prognostic value of troponin T in hemodialysis patients is independent of comorbidity. Kidney Int. 2001;60:2399–2405. doi: 10.1046/j.1523-1755.2001.00076.x. [DOI] [PubMed] [Google Scholar]
- Dierkes J, Domrose U, Westphal S, et al. Cardiac troponin T predicts mortality in patients with end-stage renal disease. Circulation. 2000;102:1964–1969. doi: 10.1161/01.cir.102.16.1964. [DOI] [PubMed] [Google Scholar]
- Fehr T, Knoflach A, Ammann P, et al. Differential use of cardiac troponin T versus I in hemodialysis patients. Clin Nephrol. 2003;59:35–39. doi: 10.5414/cnp59035. [DOI] [PubMed] [Google Scholar]
- Francis GS, Tang WH. Cardiac troponins in renal insufficiency and other non-ischemic cardiac conditions. Prog Cardiovasc Dis. 2004;47:196–206. doi: 10.1016/j.pcad.2004.07.005. [DOI] [PubMed] [Google Scholar]
- Freda BJ, Tang WH, Van Lente F, et al. Cardiac troponins in renal insufficiency: review and clinical implications. J Am Coll Cardiol. 2002;40:2065–2071. doi: 10.1016/s0735-1097(02)02608-6. [DOI] [PubMed] [Google Scholar]
- Iliou MC, Fumeron C, Benoit MO, et al. Factors associated with increased serum levels of cardiac troponins T and I in chronic haemodialysis patients: Chronic Haemodialysis And New Cardiac Markers Evaluation (CHANCE) study. Nephrol Dial Transplant. 2001;16:1452–1458. doi: 10.1093/ndt/16.7.1452. [DOI] [PubMed] [Google Scholar]
- Lamb EJ, Webb MC, Abbas NA. The significance of serum troponin T in patients with kidney disease: a review of the literature. Ann Clin Biochem. 2004;41:1–9. doi: 10.1258/000456304322664645. [DOI] [PubMed] [Google Scholar]
- Lowbeer C, Ottosson-Seeberger A, Gustafsson SA, et al. Increased cardiac troponin T and endothelin-1 concentrations in dialysis patients may indicate heart disease. Nephrol Dial Transplant. 1999;14:1948–1955. doi: 10.1093/ndt/14.8.1948. [DOI] [PubMed] [Google Scholar]
- Mallamaci F, Zoccali C, Parlongo S, et al. Troponin is related to left ventricular mass and predicts all-cause and cardiovascular mortality in hemodialysis patients. Am J Kidney Dis. 2002;40:68–75. doi: 10.1053/ajkd.2002.33914. [DOI] [PubMed] [Google Scholar]
- Needham DM, Shufelt KA, Tomlinson G, et al. Troponin I and T levels in renal failure patients without acute coronary syndrome: a systematic review of the literature. Can J Cardiol. 2004;20:1212–1218. [PubMed] [Google Scholar]
- Ooi DS, Zimmerman D, Graham J, et al. Cardiac troponin T predicts long-term outcomes in hemodialysis patients. Clin Chem. 2001;47:412–417. [PubMed] [Google Scholar]
- Roberts MA, Fernando D, Macmillan N, et al. Single and serial measurements of cardiac troponin I in asymptomatic patients on chronic hemodialysis. Clin Nephrol. 2004;61:40–46. doi: 10.5414/cnp61040. [DOI] [PubMed] [Google Scholar]
- Collinson PO, Hadcocks L, Foo Y, et al. Cardiac troponins in patients with renal dysfunction. Ann Clin Biochem. 1998;35 (Pt 3:380–386. doi: 10.1177/000456329803500306. [DOI] [PubMed] [Google Scholar]
- Roppolo LP, Fitzgerald R, Dillow J, et al. A comparison of troponin T and troponin I as predictors of cardiac events in patients undergoing chronic dialysis at a Veteran's Hospital: a pilot study. J Am Coll Cardiol. 1999;34:448–454. doi: 10.1016/s0735-1097(99)00207-7. [DOI] [PubMed] [Google Scholar]
- de Lemos JA, Drazner MH, Omland T, et al. Association of troponin T detected with a highly sensitive assay and cardiac structure and mortality risk in the general population. JAMA. 2010;304:2503–2512. doi: 10.1001/jama.2010.1768. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haller C, Zehelein J, Remppis A, et al. Cardiac troponin T in patients with end-stage renal disease: absence of expression in truncal skeletal muscle. Clin Chem. 1998;44:930–938. [PubMed] [Google Scholar]
- Abaci A, Ekici E, Oguzhan A, et al. Cardiac troponins T and I in patients with end-stage renal disease: the relation with left ventricular mass and their prognostic value. Clin Cardiol. 2004;27:704–709. doi: 10.1002/clc.4960271211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ooi DS, House AA. Cardiac troponin T in hemodialyzed patients. Clin Chem. 1998;44:1410–1416. [PubMed] [Google Scholar]
- Parfrey PS, Foley RN. The clinical epidemiology of cardiac disease in chronic renal failure. J Am Soc Nephrol. 1999;10:1606–1615. doi: 10.1681/ASN.V1071606. [DOI] [PubMed] [Google Scholar]
- Roberts MA, Hedley AJ, Ierino FL. Understanding cardiac biomarkers in end-stage kidney disease: Frequently asked questions and the promise of clinical application. Nephrology (Carlton) 2011;16:251–260. doi: 10.1111/j.1440-1797.2010.01413.x. [DOI] [PubMed] [Google Scholar]
- Hirsch R, Landt Y, Porter S, et al. Cardiac troponin I in pediatrics: normal values and potential use in the assessment of cardiac injury. J Pediatr. 1997;130:872–877. doi: 10.1016/s0022-3476(97)70271-3. [DOI] [PubMed] [Google Scholar]
- Liesemer K, Casper TC, Korgenski K, et al. Use and misuse of serum troponin assays in pediatric practice. Am J Cardiol. 2012;110:284–289. doi: 10.1016/j.amjcard.2012.03.020. [DOI] [PubMed] [Google Scholar]
- Lentine KL, Hurst FP, Jindal RM, et al. Cardiovascular risk assessment among potential kidney transplant candidates: approaches and controversies. Am J Kidney Dis. 2010;55:152–167. doi: 10.1053/j.ajkd.2009.06.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McIntyre CW, Odudu A, Eldehni MT. Cardiac assessment in chronic kidney disease. Curr Opin Nephrol Hypertens. 2009;18:501–506. doi: 10.1097/MNH.0b013e328331050b. [DOI] [PubMed] [Google Scholar]
- Bennett WM, Kloster F, Rosch J, et al. Natural history of asymptomatic coronary arteriographic lesions in diabetic patients with end-stage renal disease. Am J Med. 1978;65:779–784. doi: 10.1016/0002-9343(78)90796-9. [DOI] [PubMed] [Google Scholar]
- Braun WE, Phillips DF, Vidt DG, et al. Coronary artery disease in 100 diabetics with end-stage renal failure. Transplant Proc. 1984;16:603–607. [PubMed] [Google Scholar]
- Lorber MI, Van Buren CT, Flechner SM, et al. Pretransplant coronary arteriography for diabetic renal transplant recipients. Transplant Proc. 1987;19:1539–1541. [PubMed] [Google Scholar]
- Manske CL, Wilson RF, Wang Y, et al. Prevalence of, and risk factors for, angiographically determined coronary artery disease in type I-diabetic patients with nephropathy. Arch Intern Med. 1992;152:2450–2455. [PubMed] [Google Scholar]
- Weinrauch L, D'Elia JA, Healy RW, et al. Asymptomatic coronary artery disease: angiographic assessment of diabetics evaluated for renal transplantation. Circulation. 1978;58:1184–1190. doi: 10.1161/01.cir.58.6.1184. [DOI] [PubMed] [Google Scholar]
- Klocke FJ, Baird MG, Lorell BH, et al. ACC/AHA/ASNC guidelines for the clinical use of cardiac radionuclide imaging--executive summary: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (ACC/AHA/ASNC Committee to Revise the 1995 Guidelines for the Clinical Use of Cardiac Radionuclide Imaging) J Am Coll Cardiol. 2003;42:1318–1333. doi: 10.1016/j.jacc.2003.08.011. [DOI] [PubMed] [Google Scholar]
- Dahan M, Viron BM, Faraggi M, et al. Diagnostic accuracy and prognostic value of combined dipyridamole-exercise thallium imaging in hemodialysis patients. Kidney Int. 1998;54:255–262. doi: 10.1046/j.1523-1755.1998.00988.x. [DOI] [PubMed] [Google Scholar]
- Koistinen MJ, Huikuri HV, Pirttiaho H, et al. Evaluation of exercise electrocardiography and thallium tomographic imaging in detecting asymptomatic coronary artery disease in diabetic patients. Br Heart J. 1990;63:7–11. doi: 10.1136/hrt.63.1.7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marwick TH, Steinmuller DR, Underwood DA, et al. Ineffectiveness of dipyridamole SPECT thallium imaging as a screening technique for coronary artery disease in patients with end-stage renal failure. Transplantation. 1990;49:100–103. doi: 10.1097/00007890-199001000-00022. [DOI] [PubMed] [Google Scholar]
- Schmidt A, Stefenelli T, Schuster E, et al. Informational contribution of noninvasive screening tests for coronary artery disease in patients on chronic renal replacement therapy. Am J Kidney Dis. 2001;37:56–63. doi: 10.1053/ajkd.2001.20584. [DOI] [PubMed] [Google Scholar]
- Morrow CE, Schwartz JS, Sutherland DE, et al. Predictive value of thallium stress testing for coronary and cardiovascular events in uremic diabetic patients before renal transplantation. Am J Surg. 1983;146:331–335. doi: 10.1016/0002-9610(83)90409-9. [DOI] [PubMed] [Google Scholar]
- Patel AD, Abo-Auda WS, Davis JM, et al. Prognostic value of myocardial perfusion imaging in predicting outcome after renal transplantation. Am J Cardiol. 2003;92:146–151. doi: 10.1016/s0002-9149(03)00529-0. [DOI] [PubMed] [Google Scholar]
- Rabbat CG, Treleaven DJ, Russell JD, et al. Prognostic value of myocardial perfusion studies in patients with end-stage renal disease assessed for kidney or kidney-pancreas transplantation: a meta-analysis. J Am Soc Nephrol. 2003;14:431–439. doi: 10.1097/01.asn.0000047560.51444.3a. [DOI] [PubMed] [Google Scholar]
- Wong CF, Little MA, Vinjamuri S, et al. Technetium myocardial perfusion scanning in prerenal transplant evaluation in the United kingdom. Transplant Proc. 2008;40:1324–1328. doi: 10.1016/j.transproceed.2008.03.143. [DOI] [PubMed] [Google Scholar]
- De Lima JJ, Sabbaga E, Vieira ML, et al. Coronary angiography is the best predictor of events in renal transplant candidates compared with noninvasive testing. Hypertension. 2003;42:263–268. doi: 10.1161/01.HYP.0000087889.60760.87. [DOI] [PubMed] [Google Scholar]
- Hatta T, Nishimura S, Nishimura T. Prognostic risk stratification of myocardial ischaemia evaluated by gated myocardial perfusion SPECT in patients with chronic kidney disease. Eur J Nucl Med Mol Imaging. 2009;36:1835–1841. doi: 10.1007/s00259-009-1165-2. [DOI] [PubMed] [Google Scholar]
- Kusuoka H, Nishimura S, Yamashina A, et al. Surveillance study for creating the national clinical database related to ECG-gated myocardial perfusion SPECT of ischemic heart disease: J-ACCESS study design. Ann Nucl Med. 2006;20:195–202. doi: 10.1007/BF03027430. [DOI] [PubMed] [Google Scholar]
- Momose M, Babazono T, Kondo C, et al. Prognostic significance of stress myocardial ECG-gated perfusion imaging in asymptomatic patients with diabetic chronic kidney disease on initiation of haemodialysis. Eur J Nucl Med Mol Imaging. 2009;36:1315–1321. doi: 10.1007/s00259-009-1110-4. [DOI] [PubMed] [Google Scholar]
- Nakajima K, Nishimura T. Inter-institution preference-based variability of ejection fraction and volumes using quantitative gated SPECT with 99mTc-tetrofosmin: a multicentre study involving 106 hospitals. Eur J Nucl Med Mol Imaging. 2006;33:127–133. doi: 10.1007/s00259-005-1916-7. [DOI] [PubMed] [Google Scholar]
- Nekolla SG, Reder S, Saraste A, et al. Evaluation of the novel myocardial perfusion positron-emission tomography tracer 18F-BMS-747158-02: comparison to 13N-ammonia and validation with microspheres in a pig model. Circulation. 2009;119:2333–2342. doi: 10.1161/CIRCULATIONAHA.108.797761. [DOI] [PubMed] [Google Scholar]
- Biomarkers Definitions Working Group. Biomarkers and surrogate endpoints: preferred definitions and conceptual framework. Clin Pharmacol Ther. 2001;69:89–95. doi: 10.1067/mcp.2001.113989. [DOI] [PubMed] [Google Scholar]
- Ix JH, Criqui MH. Epidemiology and diagnosis of peripheral arterial disease in patients with chronic kidney disease. Adv Chronic Kidney Dis. 2008;15:378–383. doi: 10.1053/j.ackd.2008.07.007. [DOI] [PubMed] [Google Scholar]
- Lau JF, Weinberg MD, Olin JW. Peripheral artery disease. Part 1: clinical evaluation and noninvasive diagnosis. Nat Rev Cardiol. 2011;8:405–418. doi: 10.1038/nrcardio.2011.66. [DOI] [PubMed] [Google Scholar]
- Bello AK, Hemmelgarn B, Lloyd A, et al. Associations among estimated glomerular filtration rate, proteinuria, and adverse cardiovascular outcomes. Clin J Am Soc Nephrol. 2011;6:1418–1426. doi: 10.2215/CJN.09741110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- de Vinuesa SG, Ortega M, Martinez P, et al. Subclinical peripheral arterial disease in patients with chronic kidney disease: prevalence and related risk factors. Kidney Int Suppl. 2005. pp. S44–47. [DOI] [PubMed]
- Lash JP, Go AS, Appel LJ, et al. Chronic Renal Insufficiency Cohort (CRIC) Study: baseline characteristics and associations with kidney function. Clin J Am Soc Nephrol. 2009;4:1302–1311. doi: 10.2215/CJN.00070109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liew YP, Bartholomew JR, Demirjian S, et al. Combined effect of chronic kidney disease and peripheral arterial disease on all-cause mortality in a high-risk population. Clin J Am Soc Nephrol. 2008;3:1084–1089. doi: 10.2215/CJN.04411007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- O'Hare AM, Glidden DV, Fox CS, et al. High prevalence of peripheral arterial disease in persons with renal insufficiency: results from the National Health and Nutrition Examination Survey 1999-2000. Circulation. 2004;109:320–323. doi: 10.1161/01.CIR.0000114519.75433.DD. [DOI] [PubMed] [Google Scholar]
- O'Hare AM, Newman AB, Katz R, et al. Cystatin C and incident peripheral arterial disease events in the elderly: results from the Cardiovascular Health Study. Arch Intern Med. 2005;165:2666–2670. doi: 10.1001/archinte.165.22.2666. [DOI] [PubMed] [Google Scholar]
- O'Hare AM, Vittinghoff E, Hsia J, et al. Renal insufficiency and the risk of lower extremity peripheral arterial disease: results from the Heart and Estrogen/Progestin Replacement Study (HERS) J Am Soc Nephrol. 2004;15:1046–1051. doi: 10.1097/01.asn.0000119574.27772.fd. [DOI] [PubMed] [Google Scholar]
- Wattanakit K, Folsom AR, Criqui MH, et al. Albuminuria and peripheral arterial disease: results from the multi-ethnic study of atherosclerosis (MESA) Atherosclerosis. 2008;201:212–216. doi: 10.1016/j.atherosclerosis.2007.12.044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Margolis DJ, Hofstad O, Feldman HI. Association between renal failure and foot ulcer or lower-extremity amputation in patients with diabetes. Diabetes Care. 2008;31:1331–1336. doi: 10.2337/dc07-2244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fuster V, Ryden LE, Cannom DS, et al. ACC/AHA/ESC 2006 Guidelines for the Management of Patients with Atrial Fibrillation: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines and the European Society of Cardiology Committee for Practice Guidelines (Writing Committee to Revise the 2001 Guidelines for the Management of Patients With Atrial Fibrillation): developed in collaboration with the European Heart Rhythm Association and the Heart Rhythm Society. Circulation. 2006;114:e257–e354. doi: 10.1161/CIRCULATIONAHA.106.177292. [DOI] [PubMed] [Google Scholar]
- Abbas AE, Goodman LM, Timmis R, et al. Predictors of poor outcome in female patients undergoing endovascular intervention. J Interv Cardiol. 2010;23:401–410. doi: 10.1111/j.1540-8183.2010.00548.x. [DOI] [PubMed] [Google Scholar]
- O'Hare AM, Bertenthal D, Sidawy AN, et al. Renal insufficiency and use of revascularization among a national cohort of men with advanced lower extremity peripheral arterial disease. Clin J Am Soc Nephrol. 2006;1:297–304. doi: 10.2215/CJN.01070905. [DOI] [PubMed] [Google Scholar]
- Goodman and Gilman's Pharmacological Basis of Therapeutics12th Edn. Brunston LL, Chabner BA, Knollman BA (eds).##McGraw-Hill: New York; 2011 [Google Scholar]
- Martindale: The Complete Drug Reference37th Edn. Sweetman SC (ed).##Royal Pharmaceutical Society, London; 2011 [Google Scholar]
- Gokmen MR, Lord GM. Aristolochic acid nephropathy. BMJ. 2012;344:e4000. doi: 10.1136/bmj.e4000. [DOI] [PubMed] [Google Scholar]
- Su T, Zhang L, Li X, et al. Regular use of nephrotoxic medications is an independent risk factor for chronic kidney disease--results from a Chinese population study. Nephrol Dial Transplant. 2011;26:1916–1923. doi: 10.1093/ndt/gfq679. [DOI] [PubMed] [Google Scholar]
- National Institute for Health and Clinical Excellence. NICE Clinical Guideline 38: Bipolar Disorder.2006
- Lipska KJ, Bailey CJ, Inzucchi SE. Use of metformin in the setting of mild-to-moderate renal insufficiency. Diabetes Care. 2011;34:1431–1437. doi: 10.2337/dc10-2361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rachmani R, Slavachevski I, Levi Z, et al. Metformin in patients with type 2 diabetes mellitus: reconsideration of traditional contraindications. Eur J Intern Med. 2002;13:428. doi: 10.1016/s0953-6205(02)00131-0. [DOI] [PubMed] [Google Scholar]
- Salpeter S, Greyber E, Pasternak G, et al. Risk of fatal and nonfatal lactic acidosis with metformin use in type 2 diabetes mellitus. Cochrane Database Syst Rev. 2010. p. CD002967. [DOI] [PMC free article] [PubMed]
- Fellstrom BC, Jardine AG, Schmieder RE, et al. Rosuvastatin and cardiovascular events in patients undergoing hemodialysis. N Engl J Med. 2009;360:1395–1407. doi: 10.1056/NEJMoa0810177. [DOI] [PubMed] [Google Scholar]
- Ginsberg HN, Elam MB, Lovato LC, et al. Effects of combination lipid therapy in type 2 diabetes mellitus. N Engl J Med. 2010;362:1563–1574. doi: 10.1056/NEJMoa1001282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Keech A, Simes RJ, Barter P, et al. Effects of long-term fenofibrate therapy on cardiovascular events in 9795 people with type 2 diabetes mellitus (the FIELD study): randomised controlled trial. Lancet. 2005;366:1849–1861. doi: 10.1016/S0140-6736(05)67667-2. [DOI] [PubMed] [Google Scholar]
- Wanner C, Krane V, Marz W, et al. Atorvastatin in patients with type 2 diabetes mellitus undergoing hemodialysis. N Engl J Med. 2005;353:238–248. doi: 10.1056/NEJMoa043545. [DOI] [PubMed] [Google Scholar]
- Limdi NA, Beasley TM, Baird MF, et al. Kidney function influences warfarin responsiveness and hemorrhagic complications. J Am Soc Nephrol. 2009;20:912–921. doi: 10.1681/ASN.2008070802. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wahba IM, Olyaei AJ, Rozansky D, et al. Handling of drugs in children with abnormal renal function Pediatric Nephrologyeds. Avner ED, Harmon WE, Niaudet P, Yoshikawa, N.Springer-Verlag; 20091693–1711. [Google Scholar]
- Blowey DL.Drug Use and Dosage in Renal Failure Comprehensive Pediatric Nephrologyeds. Geary DF, Schaefer F.Mosby Elsevier: Philadelphia; 2008991–1002. [Google Scholar]
- Drug prescribing in renal failure. http://www.kdp-baptist.louisvi lle.edu/renalbook/ .
- Goldfarb S, McCullough PA, McDermott J, et al. Contrast-induced acute kidney injury: specialty-specific protocols for interventional radiology, diagnostic computed tomography radiology, and interventional cardiology. Mayo Clin Proc. 2009;84:170–179. doi: 10.4065/84.2.170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- American College Radiology Guidelines. Manual on Contrast Media Version 8. http://www.acr.org/∼/media/ACR /Documents/PDF/QualitySafety/R esources/Contrast%20Manual/Con trast%20Nephrotoxicity.pdf . Accessed October 12,2012
- European Society for Urological Radiology (ESUR). http://www.esur.org/Contrast-m edia.51.0.html Accessed October 9,2012
- Heinrich MC, Haberle L, Muller V, et al. Nephrotoxicity of iso-osmolar iodixanol compared with nonionic low-osmolar contrast media: meta-analysis of randomized controlled trials. Radiology. 2009;250:68–86. doi: 10.1148/radiol.2501080833. [DOI] [PubMed] [Google Scholar]
- Perazella MA. Current status of gadolinium toxicity in patients with kidney disease. Clin J Am Soc Nephrol. 2009;4:461–469. doi: 10.2215/CJN.06011108. [DOI] [PubMed] [Google Scholar]
- Agarwal R, Brunelli SM, Williams K, et al. Gadolinium-based contrast agents and nephrogenic systemic fibrosis: a systematic review and meta-analysis. Nephrol Dial Transplant. 2009;24:856–863. doi: 10.1093/ndt/gfn593. [DOI] [PubMed] [Google Scholar]
- American College Radiology Guidelines. Manual on Contrast Media Version 8. http://www.acr.org/∼/media/ACR /Documents/PDF/QualitySafety/R esources/Contrast%20Manual/Nep hrogenic%20Systemic%20Fibrosis .pdf . Accessed October 12,2012
- Saitoh T, Hayasaka K, Tanaka Y, et al. Dialyzability of gadodiamide in hemodialysis patients. Radiat Med. 2006;24:445–451. doi: 10.1007/s11604-006-0055-9. [DOI] [PubMed] [Google Scholar]
- Meng H, Grosse-Wortmann L. Gadolinium in pediatric cardiovascular magnetic resonance: what we know and how we practice. J Cardiovasc Magn Reson. 2012;14:56. doi: 10.1186/1532-429X-14-56. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lien YH. Is bowel preparation before colonoscopy a risky business for the kidney. Nat Clin Pract Nephrol. 2008;4:606–614. doi: 10.1038/ncpneph0939. [DOI] [PubMed] [Google Scholar]
- Markowitz GS, Perazella MA. Acute phosphate nephropathy. Kidney Int. 2009;76:1027–1034. doi: 10.1038/ki.2009.308. [DOI] [PubMed] [Google Scholar]
- Heher EC, Thier SO, Rennke H, et al. Adverse renal and metabolic effects associated with oral sodium phosphate bowel preparation. Clin J Am Soc Nephrol. 2008;3:1494–1503. doi: 10.2215/CJN.02040408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Palmadottir VK, Gudmundsson H, Hardarson S, et al. Incidence and outcome of acute phosphate nephropathy in Iceland. PLoS One. 2010;5:e13484. doi: 10.1371/journal.pone.0013484. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wexner SD, Beck DE, Baron TH, et al. A consensus document on bowel preparation before colonoscopy: prepared by a task force from the American Society of Colon and Rectal Surgeons (ASCRS), the American Society for Gastrointestinal Endoscopy (ASGE), and the Society of American Gastrointestinal and Endoscopic Surgeons (SAGES) Gastrointest Endosc. 2006;63:894–909. doi: 10.1016/j.gie.2006.03.918. [DOI] [PubMed] [Google Scholar]
- Brunelli SM. Association between oral sodium phosphate bowel preparations and kidney injury: a systematic review and meta-analysis. Am J Kidney Dis. 2009;53:448–456. doi: 10.1053/j.ajkd.2008.09.022. [DOI] [PubMed] [Google Scholar]
- Johanson JF, Popp JW., Jr Cohen LB, et al. A randomized, multicenter study comparing the safety and efficacy of sodium phosphate tablets with 2 L polyethylene glycol solution plus bisacodyl tablets for colon cleansing. Am J Gastroenterol. 2007;102:2238–2246. doi: 10.1111/j.1572-0241.2007.01363.x. [DOI] [PubMed] [Google Scholar]
- Foley RN, Guo H, Snyder JJ, et al. Septicemia in the United States dialysis population, 1991 to 1999. J Am Soc Nephrol. 2004;15:1038–1045. doi: 10.1097/01.asn.0000119144.95922.c4. [DOI] [PubMed] [Google Scholar]
- Hoen B, Paul-Dauphin A, Hestin D, et al. EPIBACDIAL: a multicenter prospective study of risk factors for bacteremia in chronic hemodialysis patients. J Am Soc Nephrol. 1998;9:869–876. doi: 10.1681/ASN.V95869. [DOI] [PubMed] [Google Scholar]
- Ishani A, Collins AJ, Herzog CA, et al. Septicemia, access and cardiovascular disease in dialysis patients: the USRDS Wave 2 study. Kidney Int. 2005;68:311–318. doi: 10.1111/j.1523-1755.2005.00414.x. [DOI] [PubMed] [Google Scholar]
- Sarnak MJ, Jaber BL. Mortality caused by sepsis in patients with end-stage renal disease compared with the general population. Kidney Int. 2000;58:1758–1764. doi: 10.1111/j.1523-1755.2000.00337.x. [DOI] [PubMed] [Google Scholar]
- Fried LF, Katz R, Sarnak MJ, et al. Kidney function as a predictor of noncardiovascular mortality. J Am Soc Nephrol. 2005;16:3728–3735. doi: 10.1681/ASN.2005040384. [DOI] [PubMed] [Google Scholar]
- Nissenson AR, Collins AJ, Hurley J, et al. Opportunities for improving the care of patients with chronic renal insufficiency: current practice patterns. J Am Soc Nephrol. 2001;12:1713–1720. doi: 10.1681/ASN.V1281713. [DOI] [PubMed] [Google Scholar]
- US Renal Data System. Atlas of Chronic Kidney Disease in the United States. Bethesda, MD: National Institutes of Diabetes and Digestive and Kidney Diseases, National Institutes of Health; 2007. [Google Scholar]
- Naqvi SB, Collins AJ. Infectious complications in chronic kidney disease. Adv Chronic Kidney Dis. 2006;13:199–204. doi: 10.1053/j.ackd.2006.04.004. [DOI] [PubMed] [Google Scholar]
- US Renal Data System. Atlas of Chronic Kidney Disease in the United States. Bethesda, MD: National Institutes of Diabetes and Digestive and Kidney Diseases, National Institutes of Health; 2010. [Google Scholar]
- McIntyre P, Craig JC. Prevention of serious bacterial infection in children with nephrotic syndrome. J Paediatr Child Health. 1998;34:314–317. doi: 10.1046/j.1440-1754.1998.00232.x. [DOI] [PubMed] [Google Scholar]
- Vanholder R, Ringoir S. Infectious morbidity and defects of phagocytic function in end-stage renal disease: a review. J Am Soc Nephrol. 1993;3:1541–1554. doi: 10.1681/ASN.V391541. [DOI] [PubMed] [Google Scholar]
- Cohen G, Haag-Weber M, Horl WH. Immune dysfunction in uremia. Kidney Int Suppl. 1997;62:S79–S82. [PubMed] [Google Scholar]
- Minnaganti VR, Cunha BA.Infections associated with uremia and dialysis Infect Dis Clin North Am 200115385–406.viii. [DOI] [PubMed] [Google Scholar]
- Pesanti EL. Immunologic defects and vaccination in patients with chronic renal failure. Infect Dis Clin North Am. 2001;15:813–832. doi: 10.1016/s0891-5520(05)70174-4. [DOI] [PubMed] [Google Scholar]
- Kausz AT, Guo H, Pereira BJ, et al. General medical care among patients with chronic kidney disease: opportunities for improving outcomes. J Am Soc Nephrol. 2005;16:3092–3101. doi: 10.1681/ASN.2004110910. [DOI] [PubMed] [Google Scholar]
- DaRoza G, Loewen A, Djurdjev O, et al. Stage of chronic kidney disease predicts seroconversion after hepatitis B immunization: earlier is better. Am J Kidney Dis. 2003;42:1184–1192. doi: 10.1053/j.ajkd.2003.08.019. [DOI] [PubMed] [Google Scholar]
- Kausz AT, Gilbertson DT. Overview of vaccination in chronic kidney disease. Adv Chronic Kidney Dis. 2006;13:209–214. doi: 10.1053/j.ackd.2006.04.007. [DOI] [PubMed] [Google Scholar]
- McNulty CA, Bowen JK, Williams AJ. Hepatitis B vaccination in predialysis chronic renal failure patients a comparison of two vaccination schedules. Vaccine. 2005;23:4142–4147. doi: 10.1016/j.vaccine.2005.03.020. [DOI] [PubMed] [Google Scholar]
- Robinson J. Efficacy of pneumococcal immunization in patients with renal disease--what is the data. Am J Nephrol. 2004;24:402–409. doi: 10.1159/000079883. [DOI] [PubMed] [Google Scholar]
- Gilbertson DT, Unruh M, McBean AM, et al. Influenza vaccine delivery and effectiveness in end-stage renal disease. Kidney Int. 2003;63:738–743. doi: 10.1046/j.1523-1755.2003.00787.x. [DOI] [PubMed] [Google Scholar]
- Dinits-Pensy M, Forrest GN, Cross AS, et al. The use of vaccines in adult patients with renal disease. Am J Kidney Dis. 2005;46:997–1011. doi: 10.1053/j.ajkd.2005.08.032. [DOI] [PubMed] [Google Scholar]
- Nikoskelainen J, Koskela M, Forsstrom J, et al. Persistence of antibodies to pneumococcal vaccine in patients with chronic renal failure. Kidney Int. 1985;28:672–677. doi: 10.1038/ki.1985.182. [DOI] [PubMed] [Google Scholar]
- Miller ER, Alter MJ, Tokars JI. Protective effect of hepatitis B vaccine in chronic hemodialysis patients. Am J Kidney Dis. 1999;33:356–360. doi: 10.1016/s0272-6386(99)70312-4. [DOI] [PubMed] [Google Scholar]
- Fattom A, Fuller S, Propst M, et al. Safety and immunogenicity of a booster dose of Staphylococcus aureus types 5 and 8 capsular polysaccharide conjugate vaccine (StaphVAX) in hemodialysis patients. Vaccine. 2004;23:656–663. doi: 10.1016/j.vaccine.2004.06.043. [DOI] [PubMed] [Google Scholar]
- Shinefield H, Black S, Fattom A, et al. Use of a Staphylococcus aureus conjugate vaccine in patients receiving hemodialysis. N Engl J Med. 2002;346:491–496. doi: 10.1056/NEJMoa011297. [DOI] [PubMed] [Google Scholar]
- CDC immunization schedules. http://www.cdc.gov/vaccines/sc hedules/index.html .
- American Academy of Pediatrics. Immunization: http://www2.aap.org/immunizati on/ .
- Neu AM. Immunizations in children with chronic kidney disease. Pediatr Nephrol. 2012;27:1257–1263. doi: 10.1007/s00467-011-2042-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goldstein SL. Acute kidney injury in children: prevention, treatment and rehabilitation. Contrib Nephrol. 2011;174:163–172. doi: 10.1159/000329394. [DOI] [PubMed] [Google Scholar]
- Goldstein SL. Acute kidney injury in children and its potential consequences in adulthood. Blood Purif. 2012;33:131–137. doi: 10.1159/000334143. [DOI] [PubMed] [Google Scholar]
- Zappitelli M, Goldstein SL.Acute kidney failure in children Management of Acute Kidney Problemseds. Jorres A, Ronco C, Kellum JA.Springer; 2010459–467. [Google Scholar]
- Andreoli SP.Clinical evaluation of acute kidney injury in children Pediatric Nephrology(6th edn), eds. Avner ED, Harmon WE, Niaudet P, Yoshikawa N.Springer; 20091603–1618. [Google Scholar]
- Zappitelli M, Goldstein SL.Management of acute kidney failure Pediatric Nephrology(6th edn), eds. Avner ED, Harmon WE, Niaudet P, Yoshikawa N.Springer; 20091619–1628. [Google Scholar]
- Zappitelli M, Goldstein SL.Acute kidney injury: General aspects Pediatric Nephrology in the ICUeds. Kiessling SG, Goebel J, Somers MJG.Springer; 200985–97. [Google Scholar]
- Arora P, Kausz AT, Obrador GT, et al. Hospital utilization among chronic dialysis patients. J Am Soc Nephrol. 2000;11:740–746. doi: 10.1681/ASN.V114740. [DOI] [PubMed] [Google Scholar]
- Khan SS, Kazmi WH, Abichandani R, et al. Health care utilization among patients with chronic kidney disease. Kidney Int. 2002;62:229–236. doi: 10.1046/j.1523-1755.2002.00432.x. [DOI] [PubMed] [Google Scholar]
- Becker BN, Coomer RW, Fotiadis C, et al. Risk factors for hospitalization in well-dialyzed chronic hemodialysis patients. Am J Nephrol. 1999;19:565–570. doi: 10.1159/000013521. [DOI] [PubMed] [Google Scholar]
- Popovic JR, Kozak LJ. National hospital discharge survey: annual summary, 1998. Vital Health Stat 13. 2000. pp. 1–194. [PubMed]
- Rocco MV, Soucie JM, Reboussin DM, et al. Risk factors for hospital utilization in chronic dialysis patients. Southeastern Kidney Council (Network 6) J Am Soc Nephrol. 1996;7:889–896. doi: 10.1681/ASN.V76889. [DOI] [PubMed] [Google Scholar]
- Thamer M, Ray NF, Fehrenbach SN, et al. Relative risk and economic consequences of inpatient care among patients with renal failure. J Am Soc Nephrol. 1996;7:751–762. doi: 10.1681/ASN.V75751. [DOI] [PubMed] [Google Scholar]
- Holland DC, Lam M. Predictors of hospitalization and death among pre-dialysis patients: a retrospective cohort study. Nephrol Dial Transplant. 2000;15:650–658. doi: 10.1093/ndt/15.5.650. [DOI] [PubMed] [Google Scholar]
- Culleton BF, Larson MG, Wilson PW, et al. Cardiovascular disease and mortality in a community-based cohort with mild renal insufficiency. Kidney Int. 1999;56:2214–2219. doi: 10.1046/j.1523-1755.1999.00773.x. [DOI] [PubMed] [Google Scholar]
- Meyer KB, Levey AS. Controlling the epidemic of cardiovascular disease in chronic renal disease: report from the National Kidney Foundation Task Force on cardiovascular disease. J Am Soc Nephrol. 1998;9:S31–S42. [PubMed] [Google Scholar]
- Drueke TB, Locatelli F, Clyne N, et al. Normalization of hemoglobin level in patients with chronic kidney disease and anemia. N Engl J Med. 2006;355:2071–2084. doi: 10.1056/NEJMoa062276. [DOI] [PubMed] [Google Scholar]
- Singh AK, Szczech L, Tang KL, et al. Correction of anemia with epoetin alfa in chronic kidney disease. N Engl J Med. 2006;355:2085–2098. doi: 10.1056/NEJMoa065485. [DOI] [PubMed] [Google Scholar]


