Abstract
The use of monoclonal antibodies targeting the CD154 molecule remains one of the most effective means of promoting graft tolerance in animal models, but thromboembolic complications during early clinical trials have precluded their use in humans. Furthermore, the role of Fc-mediated deletion of CD154-expressing cells in the observed efficacy of these reagents remains controversial. Therefore, determining the requirements for anti-CD154-induced tolerance will instruct the development of safer but equally efficacious treatments. To investigate the mechanisms of action of anti-CD154 therapy, two alternative means of targeting the CD40–CD154 pathway were used: a non-agonistic anti-CD40 antibody, and an Fc-silent anti-CD154 domain antibody. We compared these therapies to an Fc-intact anti-CD154 antibody in both a fully allogeneic model and a surrogate minor antigen model in which the fate of alloreactive cells could be tracked. Results indicated that anti-CD40 mAbs as well as Fc-silent anti-CD154 domain antibodies were equivalent to Fc-intact anti-CD154 mAbs in their ability to inhibit alloreactive T cell expansion, attenuate cytokine production of antigen-specific T cells, and promote the conversion of Foxp3+ iTreg. Importantly, iTreg conversion observed with Fc-silent anti-CD154 domain antibodies was preserved in the presence of CTLA4-Ig, suggesting this therapy is a promising candidate for translation to clinical use.
Keywords: Costimulation, Alloreactivity, Regulatory T cells, CD8+ T-lymphocytes
INTRODUCTION
Blockade of CD40–CD154 interactions during T cell priming has shown to be a highly effective means of inducing long-term survival of allografts and transplantation tolerance in both murine and non-human primate models (1, 2). However, the clinical potential of this therapy has yet to be realized due to the thromboembolic complications observed during clinical trials of an anti-CD154 monoclonal antibody (3). Thus, determination of the requirements needed to achieve this salutary effect would guide development of novel therapeutics targeting this pathway with improved safety profiles and potential for translation for clinical use.
The mechanisms by which anti-CD154 antibodies induce profound immunomodulation of donor-reactive T cell responses in animal models, resulting in long-term graft survival and in some cases tolerance, is still controversial. A previous report suggested that the tolerogenic effects of anti-CD154 antibodies were Fc- and complement-dependent (4). However, Daley and colleagues demonstrated that an aglycosylated form of anti-CD154, which exhibits reduced ability to bind Fc receptors and activate complement, was able to prolong graft survival as effectively as the glycosylated form (5). In addition, antibodies that target the CD40 molecule have also been shown to be efficacious in preventing alloreactivity in both mouse and non-human primates (6, 7). Therefore, our study aimed to directly compare the effects of an Fc-intact anti-CD154 antibody to both a non-agonistic anti-CD40 antibody as well as a clinically translatable Fc-silent anti-CD154 domain antibody (dAb). This novel reagent was generated by fusing a human anti-mouse CD154 Vκ domain antibody to a mutated mouse IgG1 Fc (D265A), to abrogate FcγR interactions (8–10).
Accumulating evidence suggests that the presence of CD4+ CD25+ regulatory T cells (Treg) may be critical for the induction of graft tolerance (11, 12). Previous work by our group has shown that perturbation of the CD40–CD154 pathway with an Fc-intact anti-CD154 mAb antibody led to the generation of antigen-specific induced Treg (iTreg) (13). However, whether this conversion requires the deletion of alloreactive effector T cells or simply blockade of the CD40–CD154 interaction remained unknown. Furthermore, previous studies demonstrated that CTLA4-Ig and Fc-intact anti-CD154 synergize to promote graft survival (2, 14), despite the known negative effect of CTLA4-Ig on Treg (15, 16). Therefore, we sought to determine if iTreg generation was preserved when CTLA4-Ig was given in combination with a potentially clinically translatable anti-CD154 dAb.
To address these issues, we employed both a fully allogeneic model of skin transplantation as well as a transgenic system of minor antigen disparity in which the non-self antigen ovalbumin (OVA) is constitutively expressed on donor-derived cells. We demonstrate that, relative to an Fc-intact anti-CD154 antibody, both anti-CD40 as well as Fc-silent anti-CD154 dAbs were able to comparably prolong graft survival, attenuate alloreactive cytokine production and promote robust iTreg conversion. Importantly, the iTreg conversion observed following anti-CD154 dAb treatment was preserved in the presence of CTLA4-Ig treatment, highlighting the potential synergy of these therapies. Taken together, these results suggest that blockade of the CD40–CD154 pathway, rather than Fc-mediated deletion of alloreactive cells, mechanistically underlies the attenuation of donor-reactive CD8+ T cell responses, induction of Foxp3+ iTreg, and prolongation in graft survival observed following CD154 antagonism. These studies provide proof-of-concept that development of Fc silent anti-CD154 domain antibodies may be translatable for use in clinical transplantation.
MATERIALS AND METHODS
Mice
B6-Ly5.2/Cr (H2-Kb, CD45.1), C57BL/6 (H2-Kb, CD45.2), and BALB/c (H-2Kd) mice were obtained from NCI (Frederick, MD). OT-I and OT-II transgenic mice, purchased from Taconic Farms (Germantown, NY), were bred to Thy1.1+ background at Emory University. mOVA mice (17) were a gift from Dr. Marc Jenkins (University of Minnesota, Minneapolis, MN) and were maintained in accordance with Emory University IACUC guidelines (Atlanta, GA). All animals were housed in pathogen-free animal facilities at Emory University.
Donor Specific Transfusion and Adoptive Transfers
For DST administration, 107 BALB/c bone marrow cells or mOVA splenocytes were given prior to transplantation. For adoptive transfers, spleen and mesenteric LNs of OT-I and OT-II mice were processed and 1.5×106 of each CD45.2+ or Thy1.1+ OT-I and OT-II were injected intravenously.
Skin Transplantation and Antibody Treatment
Full thickness tail, ear, or trunk skins were transplanted onto dorsal thorax of recipient mice and secured with adhesive bandages. Where indicated, mice were treated with 250 µg CTLA4-Ig (Bristol Myers Squibb), 250 µg hamster monoclonal anti-mouse CD154 (MR-1, BioExpress, West Lebanon, NJ), rat anti-mouse CD40 (6) (7E1-G2b, Bristol Myers Squibb) or human anti-mouse CD154 Vκ domain antibody fused to a mutated mouse IgG1 Fc (D265A) to abrogate FcγR interactions (Figure S1) (8–10) (Bristol Myers Squibb), intraperitoneally on days 0, 2, 4, and 6 post-transplantation.
Surface Stains and Flow Cytometry
Spleens or draining axillary and brachial LN were stained for CD4, CD8, Thy1.1, and CD44 (Biolegend). Samples were analyzed using an LSRII FACS machine (BD Biosciences). Data was analyzed using FlowJo software (Treestar, San Carlos, CA).
Intracellular Cytokine Staining
Where indicated, responder splenocytes were stimulated with BALB/c splenocytes or 10nM OVA257–264 (Genscript, Inc.) in the presence of 10 µg/mL Brefeldin A for 4–5 hours. Intracellular staining kit was used to detect TNF, IFN-γ (Biolegend), and IL-2 (BD Biosciences) according to manufacturer’s instructions.
Treg Staining
Splenocytes and draining lymph nodes were processed and stained with antibodies to CD4, CD8, Thy1.1 and CD25 (Biolegend). Intracellular staining with anti-Foxp3 was performed using intranuclear staining kit (eBiosciences), according to manufacturer’s instructions.
Statistical Analysis
Survival data were plotted on Kaplan-Meier curves and log-rank tests were performed. For analysis of T cell accumulation, one-way ANOVA tests were performed, followed by Tukey post-test. Analyses were done using GraphPad Prism software (GraphPad Software Inc).
RESULTS
Anti-CD154 domain antibodies are functionally equivalent to Fc-intact anti-CD154 mAbs
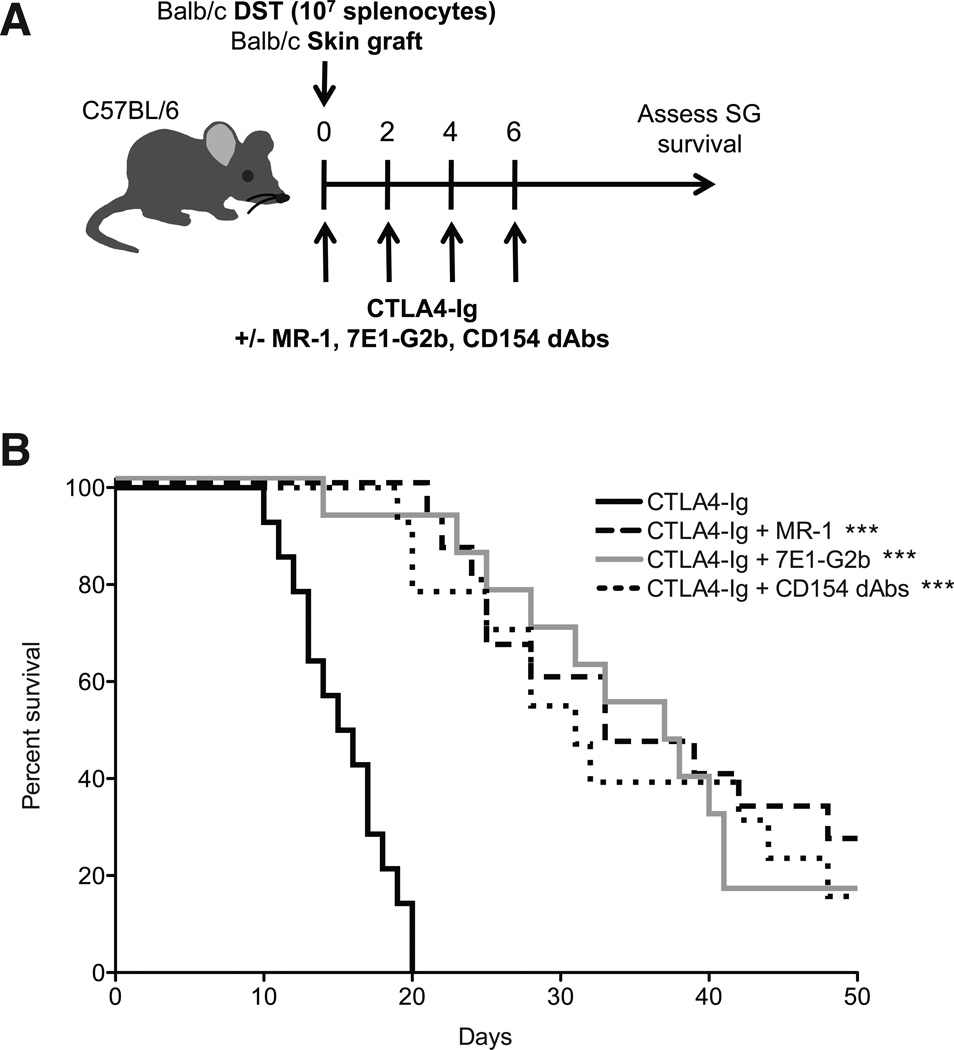
We sought to investigate the requirements for CD40–CD154 immunotherapy-mediated survival by comparing the effects of an anti-CD40 mAb (7E1-G2b) vs. an Fc-modified anti-CD154 dAb that is unable to mediate any effector function (Figure S1) (10) vs. the traditional Fc-intact anti-CD154 mAb. We applied these therapies in a stringent BALB/c B6 model of murine skin transplantation. Control animals treated with CTLA4-Ig alone rapidly rejected their grafts with an MST of 15.5 d. In contrast, the addition of each reagent targeting the CD40–CD154 pathway significantly prolonged graft survival, with anti-CD154, anti-CD40 and anti-CD154 dAb extending MST to 33, 37 and 31 days, respectively (Fig 1A, B). Therefore, blockade of CD40–CD154 interactions with novel anti-CD154 dAbs for only the first 6 days post-transplantation is sufficient to prolong graft survival to a similar extent as Fc-intact anti-CD154 treatment.
Figure 1. Anti-CD154 domain antibodies are functionally equivalent to Fc-intact anti-CD154 mAbs.
(A) On day 0, B6 mice were transplanted with BALB/c skin grafts and were treated with 107 BALB/c DST and/or 250 µg of CTLA4-Ig, MR-1, 7E1-G2b or CD154 dAbs where indicated. (B) Mice treated with CTLA4-Ig alone rejected skin grafts with an MST of 15.5 d. Addition of MR-1 prolonged MST to 33 days, whereas addition of 7E1-G2b or CD154 dAbs led to an MST of 37 and 31 days, respectively. Data are cumulative of three independent experiments with a total of fourteen to fifteen mice per group. *** = p < 0.001 compared to CTLA4-Ig alone. Comparisons between MR-1, 7E1-G2b and CD154 dAbs were not statistically significant.
Activation and cytokine production of alloreactive T cells are diminished in the presence of anti-CD154 dAbs
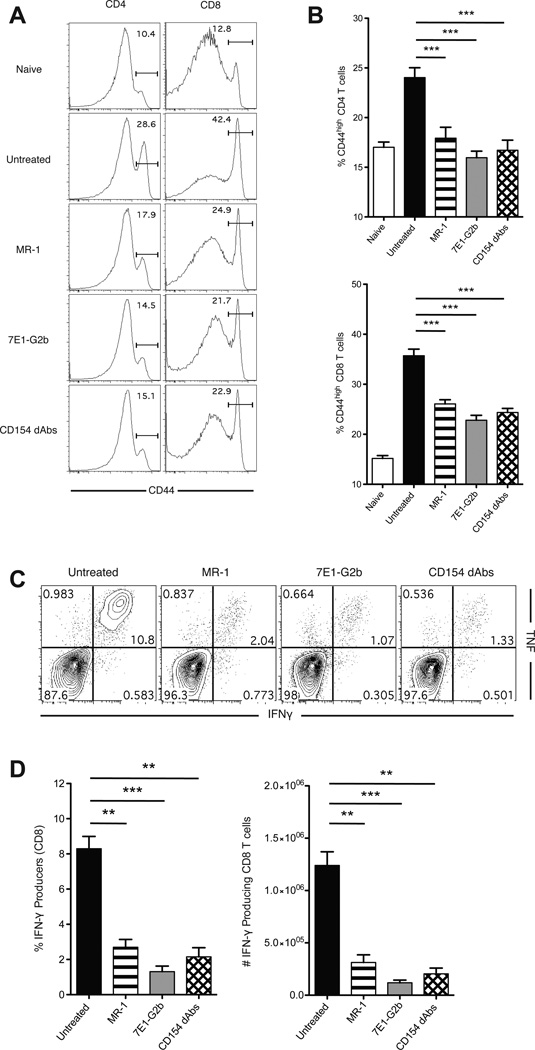
We next sought to obtain a more granular assessment of the impact of each therapeutic strategy on the alloreactive CD4+ and CD8+ T cell response following transplantation. B6 mice received BALB/c skin grafts and were treated with anti-CD154, anti-CD40 or Fc-silent anti-CD154 dAbs. At day 14 post-transplant splenocytes were harvested and analyzed for expression of activation markers. Untreated mice developed a robust allogeneic response, as evidenced by the higher proportion of CD44-expressing CD4+ and CD8+ T cells (26.3±0.8% and 34.7±2.2%), whereas mice treated with anti-CD154, anti-CD40 and Fc-silent anti-CD154 dAbs all showed significant reductions in these populations (Figure 2A, B). A large percentage of CD8+ T cells from untreated animals were able to make both interferon-gamma (IFN-γ) and tumor necrosis factor (TNF) following ex vivo restimulation with BALB/c cells, whereas the cytokine production in animals treated with anti-CD154, anti-CD40 or Fc-silent anti-CD154 dAbs was significantly blunted (Figure 2C, D).
Figure 2. Activation and cytokine production of alloreactive T cells are diminished in the presence of anti-CD154 dAbs.
(A) Representative histograms of CD44 expression on splenic CD4+ and CD8+ T cells isolated from grafted mice at day 14. (B) Frequencies of CD44hi cells among CD4+ and CD8+ T cell populations. Treatment with MR-1, 7E1-G2b or CD154 dAbs results in the reduction of CD44 expression on both CD4+ and CD8+ cells. (C) Representative flow plots of intracellular cytokine staining of recipient splenocytes, gated on CD8+ T cells. 106 recipient splenocytes were incubated with 2×106 BALB/c stimulator splenocytes for 5 hours and then fixed and permeabilized before being stained for FACS analysis. (D) Frequencies and absolute numbers of cytokine-producing CD8+ T cells. Treatment with MR-1, 7E1-G2b or CD154 dAbs results in the reduction of percentage of multicytokine producing CD8+ T cells. Data are cumulative of two separate experiments with a total of 8 to 10 mice per group. ** p < 0.01, *** p < 0.001.
Anti-CD154 dAbs inhibit donor-reactive T cell expansion in response to a minor antigen
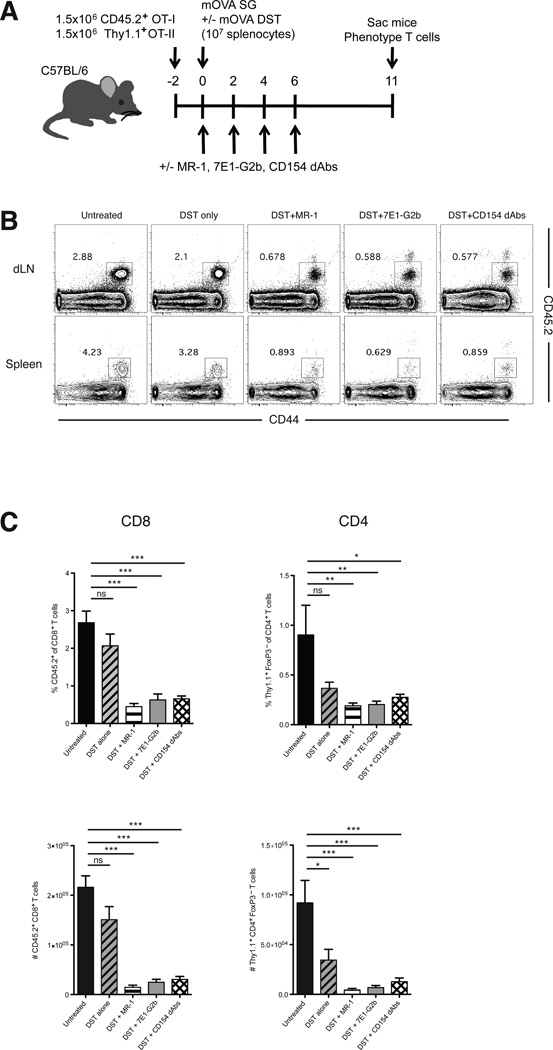
Using this fully allogeneic model, we were unable to determine if alloreactive cells were no longer present in these animals, or if they were present but simply unable to produce cytokines. In order to more fully dissect the mechanism by which these therapies act, we employed a system in which naïve OVA-specific congenically labeled CD45.2+ OT-I and Thy1.1+ OT-II T cells were transferred to naïve mice that then received OVA-expressing skin grafts (Figure 3A). This model allows us to track the effect of CD40–CD154 blockade on the fate of antigen-specific cells following a skin graft (13, 18). We chose to focus on the expansion of donor-reactive CD8+ T cells, as CD8+ cells have been shown to be the primary mediators of costimulation blockade-resistant rejection (19). As expected, untreated mice showed robust expansion of CD45.2+ CD8+ antigen-specific T cells in both the draining lymph nodes and spleen at day 11 post-transplant. DST treatment alone only moderately attenuated this expansion. However, perturbation of the CD40 pathway resulted in a significant reduction in both the percentage and absolute numbers of donor-reactive CD8+ T cells in anti-CD154, anti-CD40, and Fc-silent anti-CD154 dAb-treated animals, and all treatments inhibited donor-reactive CD8+ T cell expansion to equivalent levels. Similar findings were also observed in the donor-reactive effector CD4+ Foxp3− T cell compartment in these animals (Figure 3B, C). These antigen-specific cells express a high level of CD44, indicating that they have undergone activation following encounter with antigen. Longitudinal studies have shown that the majority of these cells eventually undergo apoptosis (13), and that those remaining are subject to peripheral tolerance mechanisms including anergy and regulation (20). These results indicate that blocking CD40–CD154 interactions, rather than selective depletion of activated donor-reactive CD154+ T cells, is sufficient to prevent antigen-specific T cell expansion following transplantation.
Figure 3. Anti-CD154 dAbs inhibit donor-reactive T cell expansion in response to a minor antigen.
(A) Mice were adoptively transferred with 1.5×106 of each CD45.2+ CD8+ OT-I and Thy1.1+ CD4+ OT-II T cells 2 days before transplantation. On day 0, mice were transplanted with mOVA SG and were treated with 107 mOVA DST and/or 250 µg of MR-1, 7E1-G2b or CD154 dAbs where indicated. Mice were sacrificed at day 11. (B-D) Representative flow plots of CD8+ T cells (B) and the frequencies and absolute numbers of antigen-specific CD8+ and effector CD4+ T cells (C) in the draining lymph nodes at day 11. DST treatment alone moderately diminished the frequency of antigen-specific CD8+ T cells, but the addition of MR-1, 7E1-G2b or the CD154 dAbs resulted in a significant reduction in the frequencies of these cells in both organs. Data are representative of four independent experiments with a total of fourteen to sixteen mice per group. * p < 0.05, ** p < 0.01, *** p < 0.001.
Anti-CD154 dAbs reduce numbers of functional graft-specific CD8+ T cells and prevent differentiation into multi-cytokine producers
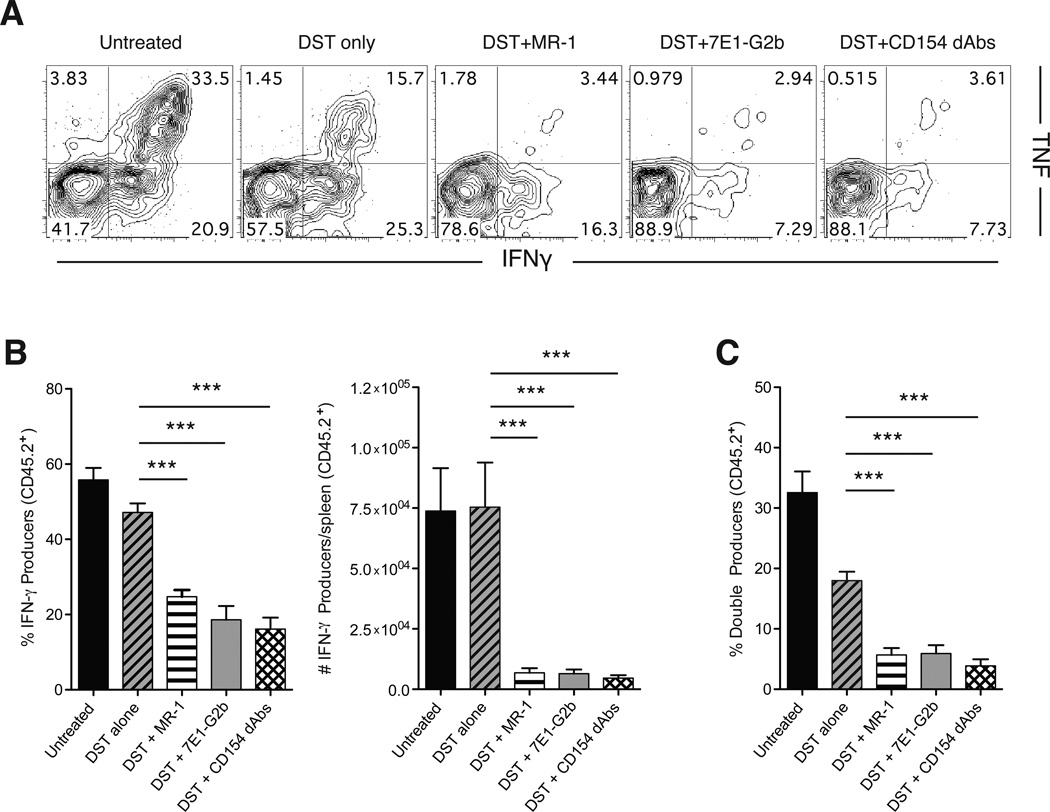
In order to determine whether donor-reactive CD8+ T cell functionality was similarly impacted by the three modalities of CD40 pathway antagonism, we examined donor-reactive CD8+ T cell compartments from treated animals following ex vivo stimulation with cognate antigen. Results showed that greater than 50% of donor-reactive CD8+ T cells in untreated animals produced IFN-γ following restimulation, and more than 30% produced both TNF and IFN-γ (Figure 4A, B). DST treatment alone resulted in a modest reduction in the ability of donor-reactive CD8+ T cells to produce cytokines, resulting in fewer double-cytokine producing cells. However, DST treatment combined with any of the three modalities of CD40 pathway antagonism further blunted this response. Each of the three modalities of CD40 pathway antagonism resulted in a decrease in the frequency of donor-reactive CD8+ IFN-γ+ T cells, and also to a decrease in donor-reactive CD8+ multifunctional IFN-γ+ TNF+ T cells (Figure 4C), which have been shown to be highly potent mediators of graft rejection (21). Thus, in addition to reducing overall expansion of alloreactive cells, an Fc-silent anti-CD154 dAb functionally impairs the remaining cells, further diminishing their potential to mediate graft injury.
Figure 4. Anti-CD154 dAbs reduce numbers of functional antigen-specific cells and prevent differentiation into multiple cytokine producers.
(A) Representative flow plots of intracellular cytokine staining of splenic antigen-specific CD8+ T cells harvested at day 11 and stimulated for 4 hours in vitro with SIINFEKL peptide. (B) Percentages and absolute numbers of IFN-γ producing antigen-specific CD8+ T cells. Compared to DST alone, treatment with MR-1, 7E1-G2b or CD154 dAbs significantly reduced the frequency and absolute numbers of IFN-γ-producing antigen-specific CD8+ T cells in the spleen. (C) Frequencies of double cytokine producing antigen-specific CD8+ T cells within each treatment group. Treatment with MR-1, 7E1-G2b or CD154 dAbs resulted in the reduction of percentage of multicytokine producing antigen-specific CD8+ T cells. Data are representative of three independent experiments with a total of eleven to twelve mice per group. *** p < 0.001.
Administration of Fc-silent anti-CD154 dAbs results in antigen-specific iTreg generation
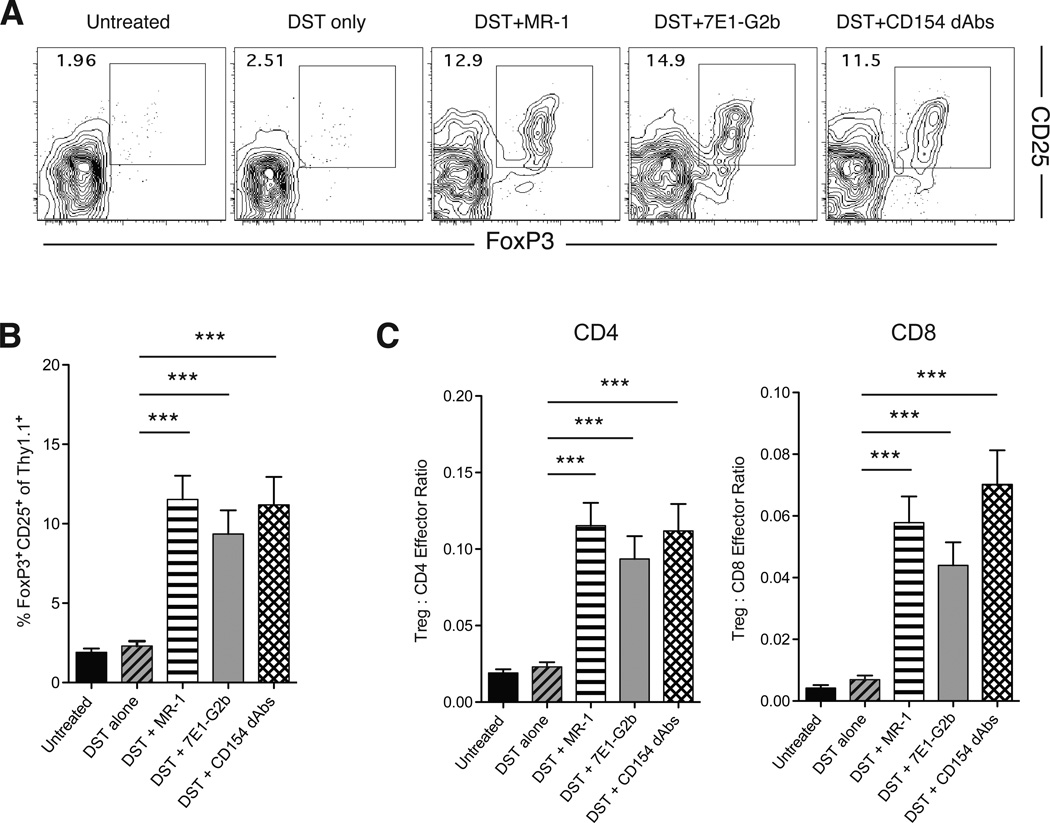
Previous results from our group and others have shown that treatment with DST and anti-CD154 leads to the peripheral conversion of donor-reactive CD4+ T cells into Foxp3+ iTreg, which may be important for the establishment of stable graft tolerance (11, 13). Therefore, we interrogated the ability of alternative CD40–CD154 blocking therapies to mediate this effect. We determined the frequency of Thy1.1+ CD4+ donor-reactive cells that converted from naïve CD4+ cells into CD4+ CD25+ Foxp3+ Treg. Untreated animals showed a low frequency of donor-reactive Thy1.1+ CD4+ Foxp3+ iTreg in the draining lymph nodes by day 11, and DST treatment alone had no effect on the number of Thy1.1+ CD4+ Foxp3+ iTreg. As we have shown previously, treatment with Fc-intact anti-CD154 resulted in a significant peripheral conversion of donor-reactive Thy1.1+ CD4+ T cell precursors into Foxp3+CD25+ iTreg. Notably, similar frequencies of Thy1.1+ CD4+ Foxp3+ iTreg were observed in animals treated with anti-CD40 antibodies and Fc-silent anti-CD154 dAbs (Figure 5A, B). In addition, the ratio of Treg cells to effectors has been shown to be an important parameter in determining the overall suppressive capacity of Treg populations (22). Treatment with all three CD40 pathway antagonists increased the Treg:effector cell ratios for both CD4+ and CD8+ cells (Figure 5D). Taken together, these results indicate that alternative CD40–CD154 blockade in the presence of antigen can result in donor-reactive Foxp3+ iTreg conversion and establishment of dominant regulatory mechanisms, which may be critical for the establishment of transplant tolerance.
Figure 5. Administration of Fc-silent anti-CD154 dAbs results in antigen-specific iTreg conversion.
(A) Representative flow plots of CD25+ Foxp3+ iTreg among antigen-specific Thy1.1+ CD4+ T cells in draining lymph nodes at day 11. (B) Summary data of percentages of CD25+ Foxp3+ cells within the antigen-specific CD4+ T cell compartment. Treatment with MR-1, 7E1-G2b or CD154 dAbs resulted in a significant increase in the percentage of antigen-specific iTregs. (C) Treg:Effector cell ratio for antigen-specific CD4+ and CD8+ T cells in each treatment group. Treatment with MR-1, 7E1-G2b or CD154 dAbs results in a significant increases in the ratio of Tregs to both CD4+ and CD8+ antigen-specific effector cells. Data are cumulative of two separate experiments with a total of eight to nine mice per group. *** p < 0.001.
Anti-CD154 dAbs results in iTreg generation even in the presence of CTLA4-Ig
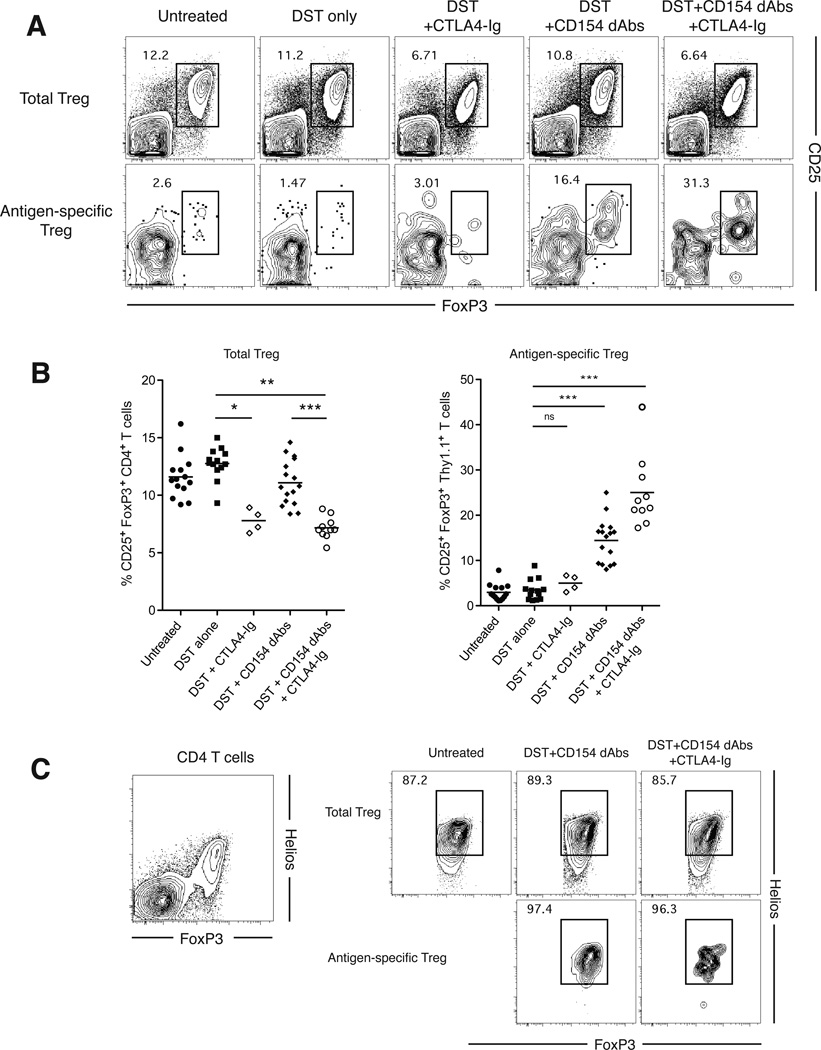
Given the potential for clinical translation of Fc-silent anti-CD154 therapy and long-standing interest in combination therapy with CD28 costimulation blockade, we sought to determine the effect of CD28 blockade on CD154 antagonism-induced iTreg generation. This question was especially pertinent due to the known negative impact of CTLA4-Ig costimulation blockade on the frequency of Treg (15, 16). We found that the frequencies of endogenous CD4+ CD25+ Foxp3+ nTreg were decreased when CTLA4-Ig was administered either alone or in combination with anti-CD154 dAbs (Figure 6A, B). Importantly, however, while CTLA4-Ig treatment alone showed no effect on iTreg conversion, anti-CD154 dAb-induced donor-reactive Thy1.1+ Foxp3+ iTreg conversion was preserved in the presence of CTLA4-Ig (Figure 6A, B). This preserved induction of antigen-specific Foxp3+ iTreg in the presence of CTLA4-Ig was similar to that observed when animals were treated with Fc-intact CD154 in the presence of CTLA4-Ig (data not shown).
Figure 6. Fc-Silent Anti-CD154 dAbs results in iTreg generation even in the presence of CTLA4-Ig.
(A) Representative flow plots of total CD4+ T cells (top panels) and antigen-specific CD4+ (Thy1.1+) T cells (bottom panels) in draining lymph nodes at day 11, with gates on CD25+ Foxp3+ cells. (B) Percentages of CD25+ Foxp3+ cells within the total CD4+ T cell compartment and the antigen-specific CD4+ T cell compartment. (C) Representative flow plots of Helios expression on total CD4+ T cells (left panel), as well as on total endogenous CD4+ CD25+ Foxp3+ Treg (upper right panels) and antigen-specific Thy1.1+ iTreg (lower right panels). Data are cumulative of four separate experiments with a total of ten to fourteen mice per group. * p < 0.05, ** p < 0.01, *** p < 0.001.
We further investigated the relative expression levels of the transcription factor Helios within donor-reactive Thy1.1+ Foxp3+ iTreg generated following CD154 antagonism with anti-CD154 dAb. Early reports suggested that Helios was a marker of thymic-derived natural Treg (nTreg) (23), but subsequent studies have provided evidence to the contrary (24). Here, we observed high levels of Helios expression on both endogenous nTreg (Figure 6C, left panel) and donor-reactive Thy1.1+ Foxp3+ iTreg compartments (Figure 6C, right panels), supporting recent work suggesting that Helios is a marker of recent antigen encounter rather than a marker of nTregs (25). Helios expression was not affected by the addition of CTLA-4-Ig in the presence of anti-CD154 dAbs (Figure 6C).
DISCUSSION
In this study, we demonstrated that blockade of CD40–CD154 interactions, as opposed to FcR-dependent deletion of activated CD154-expressing alloreactive effectors, is sufficient to prevent the expansion of alloreactive CD8+ T cells and promote the conversion of antigen-specific iTreg. This study therefore provides proof-of-concept that an Fc-silent anti-CD154 domain antibody may provide a clinically translatable treatment with comparable efficacy to an Fc-intact therapy. In addition, the results presented here are consistent with previous data showing that aglycosylated anti-CD154 antibodies recapitulated the effects of Fc-intact anti-CD154 reagents. However, generation of these reagents required a complicated “sewing PCR” technique, and large scale production of these reagents for clinical use may prove challenging. (5).
While our data suggest that it is blockade of CD40–CD154 interactions that is responsible for the attenuation of alloreactive T cell responses, the precise cellular interactions most critically impacted by CD40–CD154 blockade remain to be elucidated. While the most likely interaction inhibited by anti-CD154 antibody treatment is that between CD154 expressed on activated CD4+ T cells and CD40 expressed on dendritic cells or other APC, emerging data suggests that CD40–CD154 interactions between other cell types may be affected as well. Bhadra and colleagues have shown that CD40 expression on CD8+ T cells is potentially important in preventing their exhaustion (26). In addition, work from our group has indicated that CD40 expression on CD8+ T cells can prevent iTreg induction (Liu and Ford, manuscript in preparation). A recent report also revealed that CD154 can be expressed on dendritic cells and is important for CD8+ T cell responses to certain viral infections (27). However, it is unknown if these interactions are relevant during alloreactive responses, and further studies will help clarify the individual cellular and molecular interactions inhibited by this antibody therapy.
Our data also revealed an Fc-independent ability of CD154 antagonism to result in the conversion of antigen-specific CD4+ T cells into Foxp3+ iTreg. The phenomenon of antigen-specific iTreg generation following CD154 antagonism has been noted in previous studies (13, 28), but the mechanisms underlying this effect remain to be fully elucidated. Our results demonstrate for the first time that Fc-independent blockade of the CD40–CD154 pathway is sufficient to generate high frequencies of antigen-specific CD4+ iTreg in the context of an alloreactive response. Further research is needed to compare the relative suppressive and migratory capacities of Foxp3+ Treg generated under these conditions.
Because we observed that one of the major effects of CD154 antagonism is to induce a population of Foxp3+ iTreg, we investigated the effect of CD28 blockade using CTLA4-Ig on these cells. We found that CD28 blockade had no effect on the induction of antigen-specific iTreg, which was somewhat surprising given the well-appreciated role of CD28 in the generation and maintenance of Foxp3+ nTreg, and the fact that CTLA4-Ig administration profoundly diminishes nTreg populations (15, 16). These data are potentially clinically important in light of the recent FDA approval of belatacept, a second-generation CTLA4-Ig fusion protein (29). While this drug provides a significant benefit over standard calcineurin-inhibitor based regimens due to its reduced cardiovascular and nephrotoxicity profile, patients treated with belatacept experienced higher rates of acute rejection (30). As such, there may be an opportunity to reduce the frequency of early rejection events in belatacept-treated patients by coupling it with other low toxicity T cell-directed immunotherapies. Indeed, numerous published reports over the last two decades have demonstrated in both mouse and non-human primate trials that anti-CD154 antibodies can synergize with CTLA4-Ig in attenuating alloreactivity and promoting graft survival (2, 14), Our data demonstrating antigen-specific CD4+ Foxp3+ iTreg generation in animals treated with both Fc-silent anti-CD154 dAbs and CTLA4-Ig provide further evidence that combinatorial use of these costimulation blockers may be beneficial in attenuating the alloreactive T cell response, and highlight the potential for these therapies to be used together to prolong graft survival following clinical transplantation.
Supplementary Material
ACKNOWLEDGEMENTS
This work was supported by National Institutes of Health R01 AI073707 to MLF.
James Tamura, Aaron Yamniuk, and Steven G. Nadler are employees of Bristol-Myers Squibb. Anish Suri is a former employee of Bristol-Myers Squibb. Steven Grant is an employee of Domantis and GlaxoSmithKline.
Abbreviations
- Treg
regulatory T cell
- nTreg
natural Treg
- iTreg
induced Treg
- Fc
crystallizable fragment
- FcγR
Fc receptor gamma
- CTLA4-Ig
cytotoxic lymphocyte antigen 4
- IFN-γ
interferon-gamma
- TNF
tumor necrosis factor
- MST
median survival time
- OVA
ovalbumin
- dLN
draining lymph node
Footnotes
DISCLOSURES
The authors of this manuscript have conflicts of interest to disclose as described by the American Journal of Transplantation.
SUPPORTING INFORMATION
Additional supporting information may be found in the online version of this article:
Figure S1
Supplemental Materials and Methods
References
- 1.Kirk AD, Burkly LC, Batty DS, Baumgartner RE, Berning JD, Buchanan K, et al. Treatment with humanized monoclonal antibody against CD154 prevents acute renal allograft rejection in nonhuman primates. Nat Med. 1999;5(6):686–693. doi: 10.1038/9536. [DOI] [PubMed] [Google Scholar]
- 2.Larsen CP, Elwood ET, Alexander DZ, Ritchie SC, Hendrix R, Tucker-Burden C, et al. Long-term acceptance of skin and cardiac allografts after blocking CD40 and CD28 pathways. Nature. 1996;30:434–438. doi: 10.1038/381434a0. [DOI] [PubMed] [Google Scholar]
- 3.Kawai T, Andrews D, Colvin RB, Sachs DH, Cosimi AB. Thromboembolic complications after treatment with monoclonal antibody against CD40 ligand. Nat Med. 2000;6:114. doi: 10.1038/72162. [DOI] [PubMed] [Google Scholar]
- 4.Monk NJ, Hargreaves RE, Marsh JE, Farrar CA, Sacks SH, Millrain M, et al. Fc-dependent depletion of activated T cells occurs through CD40L-specific antibody rather than costimulation blockade. Nat Med. 2003;9(10):1275–1280. doi: 10.1038/nm931. [DOI] [PubMed] [Google Scholar]
- 5.Daley SR, Cobbold SP, Waldmann H. Fc-disabled anti-mouse CD40L antibodies retain efficacy in promoting transplantation tolerance. Am J Transplant. 2008;8(11):2265–2271. doi: 10.1111/j.1600-6143.2008.02382.x. [DOI] [PubMed] [Google Scholar]
- 6.Gilson CR, Milas Z, Gangappa S, Hollenbaugh D, Pearson TC, Ford ML, et al. Anti-CD40 monoclonal antibody synergizes with CTLA4-Ig in promoting long-term graft survival in murine models of transplantation. J Immunol. 2009;183(3):1625–1635. doi: 10.4049/jimmunol.0900339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Haanstra KG, Ringers J, Sick EA, Ramdien-Murli S, Kuhn EM, Boon L, et al. Prevention of kidney allograft rejection using anti-CD40 and anti-CD86 in primates. Transplantation. 2003;75(5):637–643. doi: 10.1097/01.TP.0000054835.58014.C2. [DOI] [PubMed] [Google Scholar]
- 8.Clynes RA, Towers TL, Presta LG, Ravetch JV. Inhibitory Fc receptors modulate in vivo cytotoxicity against tumor targets. Nat Med. 2000;6(4):443–446. doi: 10.1038/74704. [DOI] [PubMed] [Google Scholar]
- 9.Nimmerjahn F, Bruhns P, Horiuchi K, Ravetch JV. FcgammaRIV: a novel FcR with distinct IgG subclass specificity. Immunity. 2005;23(1):41–51. doi: 10.1016/j.immuni.2005.05.010. [DOI] [PubMed] [Google Scholar]
- 10.Baudino L, Shinohara Y, Nimmerjahn F, Furukawa J, Nakata M, Martinez-Soria E, et al. Crucial role of aspartic acid at position 265 in the CH2 domain for murine IgG2a and IgG2b Fc-associated effector functions. J Immunol. 2008;181(9):6664–6669. doi: 10.4049/jimmunol.181.9.6664. [DOI] [PubMed] [Google Scholar]
- 11.Taylor PA, Noelle RJ, Blazar BR. CD4(+)CD25(+) immune regulatory cells are required for induction of tolerance to alloantigen via costimulatory blockade. J Exp Med. 2001;193(11):1311–1318. doi: 10.1084/jem.193.11.1311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Quezada SA, Bennett K, Blazar BR, Rudensky AY, Sakaguchi S, Noelle RJ. Analysis of the underlying cellular mechanisms of anti-CD154-induced graft tolerance: the interplay of clonal anergy and immune regulation. J Immunol. 2005;175(2):771–779. doi: 10.4049/jimmunol.175.2.771. [DOI] [PubMed] [Google Scholar]
- 13.Ferrer IR, Wagener ME, Song M, Kirk AD, Larsen CP, Ford ML. Antigen-specific induced Foxp3+ regulatory T cells are generated following CD40/CD154 blockade. Proc Natl Acad Sci U S A. 2011;108(51):20701–20706. doi: 10.1073/pnas.1105500108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Kirk AD, Harlan DM, Armstrong NN, Davis TA, Dong Y, Gray GS, et al. CTLA4-Ig and anti-CD40 ligand prevent renal allograft rejection in primates. Proc Natl Acad Sci U S A. 1997;94(16):8789–8794. doi: 10.1073/pnas.94.16.8789. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Tang Q, Henriksen KJ, Boden EK, Tooley AJ, Ye J, Subudhi SK, et al. Cutting edge: CD28 controls peripheral homeostasis of CD4+CD25+ regulatory T cells. J Immunol. 2003;171(7):3348–3352. doi: 10.4049/jimmunol.171.7.3348. [DOI] [PubMed] [Google Scholar]
- 16.Riella LV, Liu T, Yang J, Chock S, Shimizu T, Mfarrej B, et al. Deleterious effect of CTLA4-Ig on a Treg-dependent transplant model. Am J Transplant. 2012;12(4):846–855. doi: 10.1111/j.1600-6143.2011.03929.x. [DOI] [PubMed] [Google Scholar]
- 17.Ehst BD, Ingulli E, Jenkins MK. Development of a novel transgenic mouse for the study of interactions between CD4 and CD8 T cells during graft rejection. Am J Transplant. 2003;3(11):1355–1362. doi: 10.1046/j.1600-6135.2003.00246.x. [DOI] [PubMed] [Google Scholar]
- 18.Ford ML, Koehn BH, Wagener ME, Jiang W, Gangappa S, Pearson TC, et al. Antigen-specific precursor frequency impacts T cell proliferation, differentiation, and requirement for costimulation. J Exp Med. 2007;204(2):299–309. doi: 10.1084/jem.20062319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Trambley J, Bingaman AW, Lin A, Elwood ET, Waitze SY, Ha J, et al. Asialo GM1(+) CD8(+) T cells play a critical role in costimulation blockade-resistant allograft rejection. J Clin Invest. 1999;104(12):1715–1722. doi: 10.1172/JCI8082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Wang T, Chen L, Ahmed E, Ma L, Yin D, Zhou P, et al. Prevention of allograft tolerance by bacterial infection with Listeria monocytogenes. J Immunol. 2008;180(9):5991–5999. doi: 10.4049/jimmunol.180.9.5991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Seder RA, Darrah PA, Roederer M. T-cell quality in memory and protection: implications for vaccine design. Nat Rev Immunol. 2008;8(4):247–258. doi: 10.1038/nri2274. [DOI] [PubMed] [Google Scholar]
- 22.Wood KJ, Bushell A, Hester J. Regulatory immune cells in transplantation. Nat Rev Immunol. 2012;12(6):417–430. doi: 10.1038/nri3227. [DOI] [PubMed] [Google Scholar]
- 23.Thornton AM, Korty PE, Tran DQ, Wohlfert EA, Murray PE, Belkaid Y, et al. Expression of Helios, an Ikaros transcription factor family member, differentiates thymic-derived from peripherally induced Foxp3+ T regulatory cells. J Immunol. 2010;184(7):3433–3441. doi: 10.4049/jimmunol.0904028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Verhagen J, Wraith DC. Comment on "Expression of Helios, an Ikaros transcription factor family member, differentiates thymic-derived from peripherally induced Foxp3+ T regulatory cells". J Immunol. 2010;185(12):7129. doi: 10.4049/jimmunol.1090105. author reply 30. [DOI] [PubMed] [Google Scholar]
- 25.Gottschalk RA, Corse E, Allison JP. Expression of Helios in peripherally induced Foxp3+ regulatory T cells. J Immunol. 2012;188(3):976–980. doi: 10.4049/jimmunol.1102964. [DOI] [PubMed] [Google Scholar]
- 26.Bhadra R, Gigley JP, Khan IA. Cutting edge: CD40-CD40 ligand pathway plays a critical CD8-intrinsic and -extrinsic role during rescue of exhausted CD8 T cells. J Immunol. 2011;187(9):4421–4425. doi: 10.4049/jimmunol.1102319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Johnson S, Zhan Y, Sutherland RM, Mount AM, Bedoui S, Brady JL, et al. Selected Toll-like receptor ligands and viruses promote helper-independent cytotoxic T cell priming by upregulating CD40L on dendritic cells. Immunity. 2009;30(2):218–227. doi: 10.1016/j.immuni.2008.11.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Ochando JC, Homma C, Yang Y, Hidalgo A, Garin A, Tacke F, et al. Alloantigen-presenting plasmacytoid dendritic cells mediate tolerance to vascularized grafts. Nat Immunol. 2006;7(6):652–662. doi: 10.1038/ni1333. [DOI] [PubMed] [Google Scholar]
- 29.Vincenti F, Larsen C, Durrbach A, Wekerle T, Nashan B, Blancho G, et al. Costimulation blockade with belatacept in renal transplantation. N Engl J Med. 2005;353(8):770–781. doi: 10.1056/NEJMoa050085. [DOI] [PubMed] [Google Scholar]
- 30.Vincenti F, Charpentier B, Vanrenterghem Y, Rostaing L, Bresnahan B, Darji P, et al. A phase III study of belatacept-based immunosuppression regimens versus cyclosporine in renal transplant recipients (BENEFIT study) Am J Transplant. 2010;10(3):535–546. doi: 10.1111/j.1600-6143.2009.03005.x. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.