Abstract
The aim of the present study was to evaluate the anti-diabetic, anti-inflammatory, antioxidant potential, and total phenolic content (TPC) of green and red kohlrabi cultivars. Anti-diabetic and anti-inflammatory activities were evaluated via protein tyrosine phosphatase (PTP1B) and rat lens aldose reductase inhibitory assays and cell-based lipopolysaccharide (LPS)-induced nitric oxide (NO) inhibitory assays in RAW 264.7 murine macrophages. In addition, scavenging assays using 1,1-diphenyl-2-picrylhydrazyl (DPPH) radical, 2,2′-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) diammonium salt (ABTS) radical, and peroxynitrite (ONOO−) were used to evaluate antioxidant potential and TPC was selected to assess phytochemical characteristics. Between the two kohlrabi cultivars, red kohlrabi (RK) had two times more TPC than green kohlrabi (GK) and showed significant antioxidant effects in DPPH, ABTS, and ONOO− scavenging assays. Likewise, methanol (MeOH) extracts of RK and GK inhibited LPS-induced NO production in a dose dependent manner that was further clarified by suppression of iNOS and COX-2 protein production. The MeOH extracts of RK and GK exhibited potent inhibitory activities against PTP1B with the corresponding IC50 values of 207±3.48 and 287±3.22 μg/mL, respectively. Interestingly, the RK MeOH extract exhibited significantly stronger anti-inflammatory, anti-diabetic, and antioxidant effects than that of GK MeOH extract. As a result, our study establishes that RK extract with a higher TPC might be useful as a potent anti-diabetic, antioxidant, and anti-inflammatory agent.
Keywords: anti-diabetic, anti-inflammation, antioxidant, green kohlrabi, red kohlrabi
INTRODUCTION
Diabetes mellitus (DM) is one of the oldest diseases known to mankind and is a leading health concern throughout the world. In 1936, the distinction between type 1 and type 2 DM was clearly made (1). Type 2 DM accounts for 90% of all diabetes cases, which in the near future is expected to rise to 552 million cases worldwide, stimulating the need for investigation into the mechanism of DM (2). It has been proposed that diabetic complications like retinopathy, cataracts, neuropathy, atherosclerosis, nephropathy, embryopathy, and delayed healing of wounds are initiated or activated by a common mechanism of free radical generation, which results in the generation of a variety of reactive oxygen species, reactive nitrogen species, and heightened oxidative stress (3). Furthermore, the provoked stress causes insulin resistance and altered gene expression. Hyperglycemia induces oxidative stress through various routes. First, the stimulated polyol pathway can divert 30% glucose to sorbitol and fructose, and second, an increase in protein tyrosine phosphatase 1B (PTP1B) activity negatively regulates insulin signaling. These processes are accompanied with alteration in redox status, which triggers type 2 DM and its complications (4).
Aldose reductase (AR) is the first enzyme of the polyol pathway that reduces glucose to sorbitol in the presence of β-nicotinamide adenine dinucleotide phosphate (NADPH) as a cofactor. Sorbitol dehydrogenases oxidize sorbitol to fructose in the polyol pathway, leading to a loss of functional integrity of the lens, subsequent cataract formation, and other severe diabetic complications (5). PTP1B is considered another important therapeutic target for the treatment of type 2 DM via an increase in glucose uptake from vessels into cells, leading to a decrease in postprandial hyperglycemia (6). Thus, inhibiting AR and PTP1B activity has great therapeutic potential for the prevention of DM and its complications.
Inflammation has been implicated as an important etiological factor in the development of both insulin resistance and type 2 DM, which has been predominantly drawn from studies demonstrating associations between elevated (but ‘normal range’) levels of circulating acute phase inflammatory markers and indices of insulin resistance and the development of type 2 DM (7,8). Likewise, inflammation and stress are both responsible for the pathogenesis of DM (9), suggesting the potential importance of antioxidants and anti-inflammatory alternatives. Thus, the search for alternative, effective, and safe anti-diabetic agents is vital in pharmaceutical research as current synthetic anti-diabetic drugs produce numerous side effects including liver disorders, flatulence, abdominal pain, renal tumors, hepatic injury, acute hepatitis, abdominal fullness, and diarrhea (10). Medicinal plants play a vital role in the development of new drugs as they lack toxic side effects. There is much interest in the physiological function of food components relating to the prevention of DM and obesity. Phenolic phytochemicals that function as chemo-preventive agents against oxidative damage have attracted public attention. Consumption of fruits and vegetables rich in phenolic content has been linked to decreased risk of developing chronic diseases by reducing oxidative stress and inhibiting macromolecular oxidation (11). Thus, prevention of oxidative damage with natural antioxidants and inhibition of polyol pathway efflux and PTP1B are important diabetic prevention strategies.
Kohlrabi (Brassica oleracea var. gongylodes) is a member of the Brassicaceae (mustard) family. It is a biennial, herbaceous vegetable grown as an annual crop that produces a swollen bulb-like stem at the base of the plant. It was described by Percival (12) as a form of cabbage with a turnip-like stem that stands quite above the ground that is generally utilized as a cooked vegetable, but the raw grated stem forms an acceptable component of winter salads. Depending on the cultivar, kohlrabi stems may be purple red (RK) or green (GK) with a flesh that is always white (13).
In Korea, kohlrabi is consumed as a curried or pickled vegetable. However, scientific investigations of this plant are limited in the literature. Some research regarding the potent antioxidant activity of its edible sprouts (14) and phytochemical composition (15) and amino acid composition (16) has been reported. Kohlrabi juices from both the RK and GK cultivars have been reported to exhibit potent antioxidant effects conferred by strong total phenolic content (TPC) (17). Eating kohlrabi helps individuals to stay healthy by scavenging free radicals as it provides high amounts of vitamin C (18) and protects against colon and prostate cancer by providing the body with precious phytochemicals including isothiocyanates (19), sulforaphane (20), and indole-3-carbinol (21). Previously, the anti-hyperglycemic and antioxidant activity of the Brassica oleracea var. gongylodes family in vivo has been reported (22), although activities focusing on particular kohlrabi cultivars are limited. Thus, we aimed to broaden the focus on kohlrabi cultivars belonging to this family by comparing the two most used kohlrabi cultivars in relation to their phenolic content, antioxidant, and anti-inflammatory activity.
MATERIALS AND METHODS
Chemicals
Lipopolysaccharide (LPS) from sulforaphane, protein tyrosine phosphatase (PTP1B), rat lens aldose reductase (RLAR), 1,1-diphenyl-2-picrylhydrazyl (DPPH), 2,2′-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) diammonium salt (ABTS), peroxynitrite (ONOO−), Griess reagent, 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide (MTT), ethylenediaminetetraacetic acid (EDTA), 2-amino-5,6-dihydro-6-methyl-4H-1,3-thiazine hydrochloride (AMT), p-nitrophenyl phosphate (pNPP), 6-hydroxy-2,5,7,8-tetramethylchroman-2-carboxylic acid (Trolox), fetal bovine serum (FBS), and antibiotics were purchased from Sigma-Aldrich Co. (St. Louis, MO, USA). Dulbecco’s Modified Eagle’s Medium (DMEM) was purchased from HyClone (Logan, UT, USA). Dihydrorhodamine 123 (DHR 123) was purchased from Molecular Probes (Eugene, OR, USA). Various primary antibodies (iNOS, COX-2, and β-actin) were obtained from Cell Signaling Technology, Inc. (Beverly, MA, USA). Polyvinylidene fluoride (PVDF) membrane (Immobilon-P) was obtained from Millipore Co. (Billerica, MA, USA). SuperSignal® West Pico Chemi-luminescent Substrate was obtained from Pierce Biotechnology, Inc. (Rockford, IL, USA). All other chemicals and solvents were purchased from E. Merck Fluka or Sigma-Aldrich unless stated otherwise.
Plant materials and extraction
The green and red kohlrabi cultivars were purchased on April 10, 2013 from a local retailer and authenticated by Professor Jae Sue Choi. A voucher specimen 20131029 was deposited in the authorized laboratory.
Preparation of kohlrabi extracts
Sliced GK (3.0 kg) and RK (5.0 kg) were each refluxed with MeOH for 3 h (5 L×2). The total filtrate was then suspended to dryness in vacuo at 40°C in order to render the MeOH extracts (542.92 g and 754.13 g, respectively).
Determination of total phenolic content
The total phenolic content (TPC) of the extract was determined using Folin-Ciocalteu reagent following a slightly modified method (23). Briefly, 0.5 mL of the 70% MeOH extract (100 μg/mL) was mixed with 2.0 mL Folin-Ciocalteu reagent (diluted 1:10 with de-ionized water) and neutralized with 4.0 mL sodium carbonate solution (7.5%, w/v). The reaction mixture was then incubated at room temperature for 30 min with intermittent shaking for color development. Absorbance of the resulting blue color was measured at 765 nm using an Ultrospec®2100pro UV/Visible spectrophotometer with SWIFT II Applications software (Amersham Biosciences, Piscataway, NJ, USA). The TPC was determined from the linear equation of a gallic acid standard curve.
DPPH radical scavenging activity
The DPPH radical scavenging activity was evaluated using the method of Blois (24), with slight modification. The RK and GK extracts and DPPH were dissolved in MeOH. One hundred sixty microliters of both extracts at various concentrations (100 μM final concentration) were added to 40 μL DPPH (1.5×10−4 M). After mixing gently and standing at room temperature for 30 min, the optical density of the reactant was measured at 520 nm using a VERSAmax microplate spectrophotometer (Molecular Devices). The antioxidant activity of both samples is expressed in terms of IC50 values (μg/mL, required to inhibit DPPH radical formation by 50%), which was calculated from the log-dose inhibition curve. L-Ascorbic acid was used as the positive control.
Trolox equivalent antioxidant capacity (TEAC)
This assay is based on the ability of different substances to scavenge ABTS radical cation (ABTS.+) as compared to the Trolox positive control (25). To oxidize colorless ABTS to blue-green ABTS.+, a 7.0 mM ABTS stock solution was mixed with 2.45 mM potassium per-sulfate (1:1, v/v) and left at room temperature in the dark for 12 to 16 h until the reaction was complete and the absorbance was stable. The blue/green ABTS.+ solution was diluted in ethanol (EtOH) to an absorbance of 0.70±0.02 at 734 nm for measurement. The photometric assay was conducted with 180 μL of the ABTS.+ solution and 20 μL of sample dissolved in EtOH solution (100 μM final concentration) that was stirred for 30 s. The optical density was measured at 734 nm after 2 min using a VERSAmax microplate spectrophotometer (Molecular Devices). The antioxidant activity was calculated by determining the decrease in absorbance at different concentrations using the following equation:
where At and Ac are the absorbance with and without extracts, respectively. Trolox and L-ascorbic acid were used as the positive controls. The TEAC results are expressed as IC50 values (μg/mL).
Assay for ONOO− scavenging activity
The ONOO− scavenging activity was assessed by a modified Kooy’s method, which involves the monitoring of highly fluorescent rhodamine 123, which is rapidly produced from non-fluorescent dihydrorhodamine (DHR) 123 in the presence of ONOO− (26). In brief, the rhodamine buffer (pH 7.4) consisted of 50 mM sodium phosphate dibasic, 50 mM sodium phosphate monobasic, 90 mM sodium chloride, 5.0 mM potassium chloride, and 100 μM diethylenetriamine pentaacetate. The final DHR 123 concentration was 5.0 μM. The assay buffer was prepared prior to use and placed on ice. The extracts were dissolved in 10% DMSO (100 μM final concentration). The background and final fluorescent intensities were measured 5 min after treatment with and without the addition of authentic ONOO− (10 μM), dissolved in 0.3 N sodium hydroxide (NaOH). The fluorescence intensity of the oxidized DHR 123 was evaluated using a fluorescence microplate reader (FL×800, Bio-Tek Instruments Inc., Winooski, VT, USA) at excitation and emission wavelengths of 480 and 530 nm, respectively. The values of ONOO− scavenging activity were calculated as the final fluorescence intensity minus the background fluorescence via the detection of DHR 123 oxidation. L-penicillamine was used as the positive control.
Assay for PTP1B inhibitory activity
Inhibitory activity of the RK and GK extracts against PTP1B was evaluated using pNPP (27). To each well in a 96-well plate (final volume 110 μL), 2.0 mM pNPP and PTP1B were added in a buffer containing 50 mM citrate (pH 6.0), 0.1 M NaCl, 1.0 mM EDTA, and 1.0 mM DTT with or without sample. The plate was pre-incubated at 37°C for 10 min, and then 50 μL pNPP buffer was added. Following incubation at 37°C for 30 min, the reaction was terminated with the addition of 10 M NaOH. The amount of p-nitrophenyl produced after enzymatic de-phosphorylation was estimated by measuring the absorbance at 405 nm using a VERSAmax microplate spectrophotometer (Molecular Devices). The non-enzymatic hydrolysis of 2.0 mM pNPP was corrected by measuring the increase in absorbance at 405 nm obtained in the absence of PTP1B enzyme. The percent inhibition was calculated as:
where Ac was the absorbance of the control, and As was the absorbance of the sample. Ursolic acid was used as the positive control.
Assay for RLAR inhibitory activity
This study adhered to the Guidelines for the Care and Use of Laboratory Animals approved by Pukyong National University. Rat lens homogenate was prepared according to a slightly modified method from Hayman and Kinoshita (28). The lenses were removed from the eyes of Sprague-Dawley rats (Samtako Bio-Korea, Inc., Osan, Korea) weighing 250 to 280 g. The lenses were homogenized in sodium phosphate buffer (pH 6.2), which was prepared from sodium phosphate dibasic (Na2HPO4·H2O, 0.66 g) and sodium phosphate monobasic (NaH2PO4·H2O, 1.27 g) in 100 mL double distilled H2O. The supernatant was obtained by centrifugation of the homogenate at 10,000 rpm at 4°C for 20 min and was frozen until use. A crude AR homogenate with a specific activity of 6.5 U/mg was used in the enzyme inhibition evaluations. The reaction solution consisted of 620 μL 100 mM sodium phosphate buffer (pH 6.2), 90 μL AR homogenate, 90 μL 1.6 mM NADPH, and 91 μL sample. The substrate included 90 μL D L-glyceraldehyde (50 mM). The AR activity was determined by measuring the decrease in NADPH absorption at 340 nm over a 4 min period on an Ultrospec2100 pro UV/Visible spectrophotometer with SWIFT II Applications software (Amersham Biosciences). Quercetin, a well-known AR inhibitor, was used as a reference. The percent inhibition was calculated as:
where ΔA sample/min represents the reduction in absorbance over 4 min for the test sample and substrate, and ΔA control/min represents the same, but with 100% DMSO instead of the test sample. The 50% inhibition concentration is expressed as the mean±SEM.
Cell culture
RAW 264.7 murine macrophages were obtained from the American Type Culture Collection (Rockville, MD, USA). RAW 264.7 cells were cultured in DMEM supplemented with 10% FBS, 100 U/mL penicillin, and 100 μg/mL streptomycin at 37°C with humidified air containing 5% CO2.
Cell viability assay
Cell viability was assessed using the MTT assay. Briefly, RAW 264.7 cells were seeded into 96-well plates at a density of 1.0×104 cells per well and incubated at 37°C for 24 h. The cells were then treated with various sample concentrations (400 to 1,600 μg/mL). After incubation for an additional 24 h at 37°C, 100 μL MTT (0.5 mg/mL in PBS) was added to each well and the incubation continued for another 2 h. The resulting color was assayed at 540 nm using a microplate spectrophotometer (Molecular Devices).
Assay for inhibition of cellular NO production
The nitrite concentration in the medium was measured using Griess reagent as an indicator of NO production. Briefly, RAW 264.7 cells (1.0×105 cells/well in a 24-well plate with 500 μL culture medium) were pre-treated with samples (400 to 1,600 μg/mL) for 2 h and incubated for 18 h with LPS (1.0 μg/mL). After incubation, the nitrite concentration of the supernatants (100 μL/well) was measured by adding 100 μL Griess reagent. To quantify the nitrite concentration, standard nitrite solutions were prepared. The absorbance values of mixtures were determined using a microplate spectrophotometer (Molecular Devices) at 540 nm. The iNOS inhibitor AMT was used as a positive control.
Analysis for inhibition of iNOS and COX-2 protein production
Western blotting was used to measure iNOS and COX-2 protein levels. First, RAW 264.7 cells were cultured in 100 mm culture dishes in the presence or absence of LPS (1.0 μg/mL) with or without test samples (400 to 1,600 μg/mL) for 18 h. Afterwards, the cells were washed twice with ice-cold PBS and lysed with cell lysis buffer (#9803, Cell Signaling Technology, Inc.) on ice for 30 min. Cell extracts were obtained by centrifugation at 14,000 g at 4°C for 20 min. Cytosolic proteins were electrophoretically separated on SDS-PAGE and transferred to PVDF membranes. Membranes were immediately blocked with 5% (w/v) non-fat dry milk in Tris-buffered saline containing 0.1% Tween-20 (pH 7.4) (TBST) buffer at room temperature for 1 h. The membranes were then washed three times (10 min each) in TBST buffer and incubated with primary antibody, diluted 1:1000 in 5% (w/v) non-fat dry milk in TBST buffer at 4°C overnight. After three washings in TBST buffer (10 min each), the membranes were incubated with horseradish peroxidase (HRP)-conjugated secondary antibody diluted 1:2000 in 5% (w/v) non-fat dry milk in TBST buffer at room temperature for 1 h. After three washings in TBST buffer (10 min each), antibody labeling was visualized with SuperSignal West Pico Chemi-luminescent Substrate (Pierce, Rockford, IL, USA) according to the manufacturer’s instructions and was exposed to X-ray film (GE Healthcare Ltd., Amersham, UK). Pre-stained blue protein markers were used for molecular weight determination. Bands were visualized using a LAS3000® Luminescent image analyzer (Fujifilm Life Science, Tokyo, Japan) and quantified by densitometry analysis using PDQuest software (Version 7.0, BIO-RAD, Hercules, CA, USA).
Statistics
Data are presented as the mean±SEM of at least four independent experiments. Statistical comparisons between groups were performed using one-way ANOVA followed by Student’s t-test. A P value less than or equal to 0.05 was considered statistically significant.
RESULTS
Scavenging effect on DPPH radical
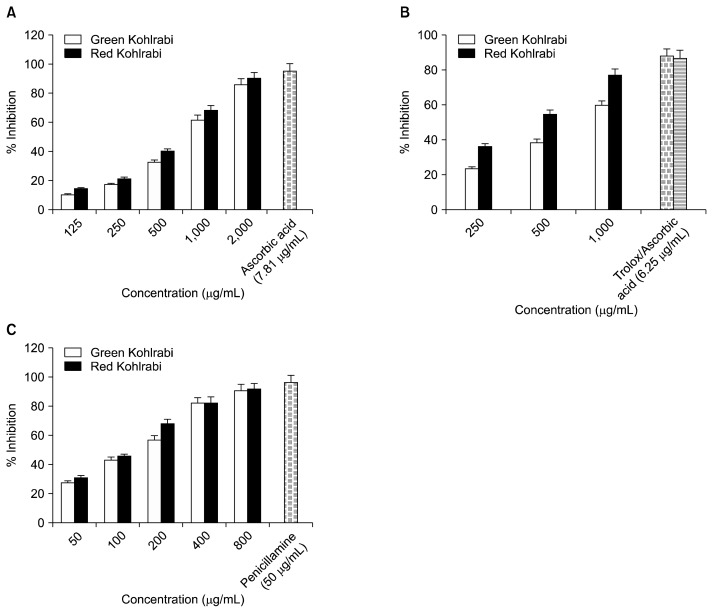
The radical-scavenging activity of MeOH extracts from the green and red kohlrabi cultivars was estimated by comparing the percent inhibition of formation of DPPH radicals by the extracts and L-ascorbic acid (Table 1). RK extract inhibited formation of 45.2% of DPPH radicals. This was greater than the GK extract, which inhibited 34.77% of DPPH radicals at the concentration of 500 μg/mL in a dose dependent manner (Fig. 1A). RK showed potent activity in DPPH scavenging activity with an IC50 value of 696.83±1.16 μg/mL whereas GK exhibited an IC50 value of 801.03±0.80 μg/mL as compared to the positive L-ascorbic acid control (IC50= 2.64±0.05 μg/mL).
Table 1.
Antioxidant activity of the red and green kohlrabi cultivar MeOH extracts
| TPC1) | IC50 | |||
|---|---|---|---|---|
|
| ||||
| ABTS | DPPH | ONOO− | ||
| Red kohlrabi | 8.91 | 472.63±2.322) | 696.83±1.16 | 116.79±0.13 |
| Green kohlrabi | 4.64 | 787.89±5.34 | 801.03±0.80 | 132.20±0.32 |
| Trolox3) | 3.31±0.05 | |||
| L-Ascorbic acid4) | 2.64±0.05 | |||
| L-Penicillamine5) | 1.21±0.26 | |||
Total phenolic content (TPC) is expressed as GAE (mg/g of MeOH extract).
Values are expressed as the mean±SEM of three experiments.
Trolox, L-ascorbic acid, and L-penicillamine were used as positive controls for ABTS, DPPH, and ONOO−, respectively.
Fig. 1.
(A) DPPH, (B) ABTS, and (C) ONOO− scavenging activities of red and green kohlrabi cultivar MeOH extracts.
Scavenging effect on ABTS radical
The ABTS assay is an excellent tool for determining the antioxidant activity of hydrogen-donating and chain-breaking antioxidants. Both extracts efficiently scavenged ABTS radicals generated by the reaction between ABTS and ammonium persulfate (Table 1). The activity increased in a dose-dependent manner from 36% to 76% in RK extracts and from 23% to 59% in GK extracts at a concentration range of 250 to 1,000 μg/mL (Fig. 1B). The RK extract showed potent activity in ABTS scavenging activity with an IC50 value of 472.63±2.32 μg/mL whereas GK exhibited an IC50 value of 787.89±5.34 μg/mL as compared to the positive Trolox control (IC50=3.31±0.05 μg/mL).
Scavenging effect on ONOO− radical
The ONOO− scavenging activities are shown in Table 1. The formation of ONOO− detectable by the oxidation of DHR 123 to rhodamine 123 from decomposition of authentic ONOO− was scavenged by both cultivar extracts to a variable degree. Both the cultivars showed dose dependent inhibition (Fig. 1C) and the ability of these extracts to scavenge authentic ONOO− was similar with respective IC50 values of 116.79±0.13 μg/mL (RK) and 132.20±0.32 μg/mL (GK) as compared to a positive L-penicillamine control (1.21±0.26 μg/mL).
Total phenolic content
The Folin-Ciocalteu assay is a fast and simple method to rapidly determine phenolic group content in samples. Phenolic or polyphenol compounds are secondary plant metabolites that are present in plants and plant products, and many phenolic compounds have been shown to contain high levels of antioxidant activity. The TPC of the two extracts is presented in Table 1. The RK extract contained the highest TPC (8.91 mg/g GAE), which is approximately double the TPC of the GK extract (4.0 mg/g GAE).
PTP1B inhibitory activity
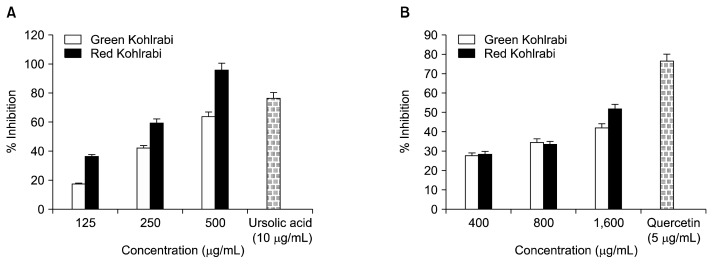
The inhibitory activity of the RK and GK MeOH extracts against PTP1B is presented in Table 2. The MeOH extracts of both cultivars showed potent PTP1B inhibitory activity in a concentration dependent manner. Among the two cultivars, the RK extract showed the highest inhibitory activity with an IC50 value of 207.56±3.48 μg/mL followed by the GK with an IC50 value of 287.13±3.22 μg/mL compared to the positive ursolic acid control (IC50 value of 5.59±0.44 μg/mL). The RK extract (96%) exhibited higher activity as compared to the GK extract (63%) at 500 μg/mL (Fig. 2A).
Table 2.
Anti-diabetic potential of the red and green kohlrabi cultivar MeOH extracts
| IC50 | ||
|---|---|---|
|
|
||
| PTP1B | RLAR | |
| Red kohlrabi | 207.56±3.481) | 1,530.23±2.80 |
| Green kohlrabi | 287.13±3.22 | 1,682.21±3.23 |
| Ursolic acid2) | 5.59±0.44 | |
| Quercetin3) | 0.58±0.02 | |
Values are expressed as the mean±SEM of three experiments.
Ursolic acid and quercetin were used as positive controls for PTP1B and RLAR assays, respectively.
Fig. 2.
(A) PTP1B and (B) RLAR activity of red and green kohlrabi cultivar MeOH extracts.
RLAR activity
It is evident that the RK extract showed a higher RLAR inhibitory activity (IC50=1,530.23±2.80 μg/mL) compared to the positive quercetin control (IC50 value of 0.58 μg/mL) (Table 2). As shown in Fig. 2B, both cultivars exhibited dose dependent inhibition with RK (53%) and GK (45%) inhibition at 1,600 μg/mL. In addition, the GK extract exhibited weak RLAR inhibitory activity with an IC50 value of 1,682.21±3.23 μg/mL.
Effect of RK and GK extracts on cell viability and NO production in LPS-induced RAW 264.7 cells
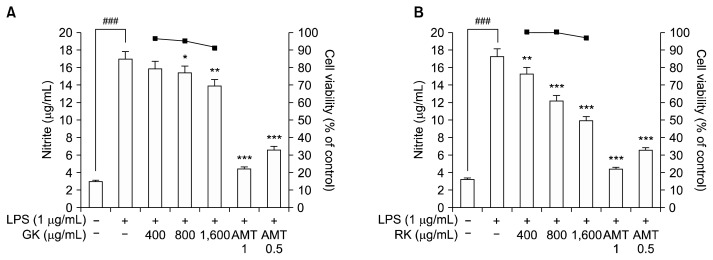
The cytotoxicity of the two cultivars was assessed via MTT assay. As shown in Fig. 3, both RK and GK MeOH extracts did not exhibit significant toxicity even at a concentration of 1,600 μg/mL. Thus, we further evaluated NO scavenging using this non-toxic concentration. Increased NO production is a typical phenomenon that occurs in LPS-stimulated macrophages and serves as an indicator of a typical inflammatory response. Nitrite, a stable metabolite of NO, was used as an indicator of NO production in the medium. During the 18 h incubation with LPS (1.0 μg/mL), NO production in RAW 264.7 cells increased dramatically. Pretreatment with the GK MeOH extract resulted in a relatively moderate dose dependent inhibition of cellular NO production; however, the RK extract showed potent activity to inhibit NO production in a dose-dependent manner (Fig. 3). With respect to the cellular NO production assay, GK at 1,600 μg/mL resulted in a 19% inhibition while RK at the same concentration resulted in a 47.23% inhibition compared with the positive AMT control (90% at 0.5 μg/mL). Thus, the NO inhibitory potency of RK was greater than that of GK (IC50=1621.34 μg/mL).
Fig. 3.
Effect of green kohlrabi (GK) (A) and red kohlrabi (RK) (B) extracts on cell viability and LPS-induced NO production in RAW 264.7 cells. Cell viability was determined by MTT assay. Cells were seeded in 96-well plates at a density of 1.0×104 cells/well and incubated at 37°C for 24 h. The cells were then treated with various concentrations of sample (400 to 1,600 μg/mL). After an additional 24 h of incubation at 37°C, 100 μL MTT (0.5 mg/mL in PBS) was added to the wells and mixed. Cells were pretreated with the indicated concentrations for 2 h, after which LPS (1.0 μg/mL) was added. After 18 h of incubation, the NO in the culture supernatants was measured using Griess reagent. Values are expressed as the mean±SEM of triplicate experiments. ###P<0.001 indicates a significant difference from the unstimulated control group, *P<0.05, **P<0.01, and ***P<0.001 indicate significant differences from the LPS-stimulated control group.
Effect of RK extract on iNOS and COX-2 production in RAW 264.7 cells
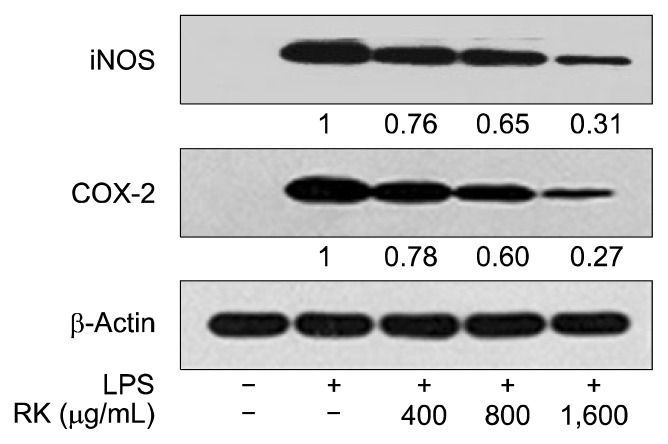
Western blot analysis was conducted to assess the activity of RK extract on the production of iNOS and COX-2, which are well-characterized markers of NF-κB-responsive inflammation. As shown in Fig. 4, iNOS and COX-2 protein production in unstimulated RAW 264.7 cells was almost undetectable, but it was significantly stimulated upon treatment with 1.0 μg/mL of LPS. Pretreatment with the RK MeOH extract at concentrations of 400, 800, and 1,600 μg/mL significantly down-regulated the production of iNOS and COX-2 protein in a dose dependent manner. RK had a greater effect on the inhibition of COX-2 protein production than on iNOS production, which was further elucidated by densitometry analysis showing the potent inhibition of COX-2 expression in a dose-dependent manner.
Fig. 4.
Effect of red kohlrabi (RK) on the production of iNOS and COX-2 protein in RAW 264.7 cells. Cytosolic lysates from RAW 264.7 cells stimulated for 18 h were separated by SDS-PAGE and the protein levels of iNOS, COX-2, and β-actin were detected by Western blot.
DISCUSSION
Although significant improvement has been made in intensive glycemic control over the last decade, a number of problems remain unsolved as indicated by the large proportion of patients unable to reach recommended therapeutic targets. There have been several recent attempts devoted to the discovery of new and effective anti-diabetic agents for the treatment of DM, including sulfonylurea derivatives, biguanides, thiazolidinedione, and alpha-glucosidase inhibitors (29). However, the use of these anti-diabetics is limited due to toxic side effects. Recently, plant secondary metabolites have been investigated as a natural source of alternative drugs for the treatment of various ailments such as DM because of their reduced toxicity (30). Thus, we aimed our efforts to investigate the possible development of natural drug alternatives derived from kohlrabi extracts.
The presence of anthocyanins, isothiocyanates, and over 120 glucosinolates from kohlrabi has been reported (19,31,32). Thus, we conducted phytochemical investigations in order to determine the phenolic compounds present in two different kohlrabi cultivars (red and green). Both the RK and GK cultivars showed the presence of phenolic compounds as compared to gallic acid, which is consistent with a previous report on the green cultivar published by Stratil et al. (33). However, our study presents a report for the presence of phenolic compounds in both red and green cultivars as it is well known that phenolic compounds exert antioxidant activity in biological systems due to the presence of available hydroxyl groups present in the chemical structure (34,35).
Both the RK and GK cultivars showed potent inhibitory activity against DPPH, ABTS, and ONOO−, which can be attributed to the presence of potent phenolic compounds such as luteolin, myricetin, apigenin, and kaempferol (36). Previously, edible kohlrabi sprouts have also been reported to show potent antioxidant activity (14) conferred by their phytochemical constituents (15). Free radicals ultimately have been reported to induce inflammation that further contributes to the onset and progression of inflammation in distant organs (37). Our results show that both the RK and GK cultivars possess dose-dependent anti-inflammatory activity, particularly the RK extract, in a manner attributed to phenolic content conferring antioxidant potential (38). Phenolics and flavonoids act as excellent anti-inflammatory agents (39). Thus, the phenolic compounds with potent anti-oxidant activity are capable of exhibiting anti-inflammatory activity, with the potential to prevent DM and its complications. Further, the RK extract inhibited the production of iNOS and COX-2 protein, which further supports it as a candidate for an anti-inflammatory agent. Enhanced production of iNOS and COX-2 proteins is associated with the inflammatory response (40). In our study, we demonstrated the suppression of both iNOS and COX-2 protein production by RK for the first time. This result demonstrates that RK inhibited NO production through the suppression of both iNOS and COX-2 production in LPS-stimulated RAW 264.7 cells. Previously, the anti-inflammatory activity of kohlrabi was attributed to the presence of isothiocyanates (21). However, our study adds to this information by showing that phenolics are also responsible for the anti-inflammatory activity.
PTP1B, which is localized to the cytoplasmic face of the endoplasmic reticulum and acts as a key regulator of both the insulin and leptin signaling pathways, is ubiquitously produced in classical insulin-targeted tissues including the liver, muscle, and fat (41). PTP1B can associate with and dephosphorylate both activated insulin receptor or insulin receptor substrates. Clinical studies have found a correlation between insulin resistance states and levels of PTP1B protein in muscle and adipose tissues, suggesting that PTP1B plays a major role in the insulin resistance associated with obesity and non-insulin dependent DM (42). Thus, inhibition of PTP1B is an effective therapeutic approach for insulin sensitive drugs targeted for the treatment and prevention of metabolic disorders (43). It is known that PTP1B overexpression in multiple tissues in obesity is regulated by inflammation (44). Our study investigated potent PTP1B inhibitors present in the MeOH extracts of RK and GK cultivars. The MeOH extracts and their different fractions showed potent PTP1B inhibitory activity. The RK extract in particular showed potent PTP1B inhibitory activity and is therefore a promising candidate for DM treatment.
DM associated complications can also arise from increased flux of glucose through the polyol pathway. AR, a cytosolic monomeric oxido-reductase is the first enzyme in the polyol pathway. Under normoglycemic conditions, AR has low affinity for glucose and only a small percentage of glucose is metabolized by this pathway. However, under hyperglycemic conditions, increased intracellular glucose results in increased enzymatic conversion to sorbitol, with a concomitant decrease in NADPH (45,46). The increased polyol pathway flux leads to accumulation of sorbitol in the lens fiber, which causes an influx of water, the generation of osmotic stress, and subsequent cataract formation (47). Due to poor penetration across membranes and inefficient metabolism, sorbitol and its metabolites accumulate in the nerves, retina, and kidneys, and cause the development of diabetic complications, including retinopathy, neuropathy, and nephropathy (48). Therefore, inhibition of AR has emerged as an important therapeutic approach for preventing and reducing long term diabetic complications. In our investigation, the RK extract showed reasonable RLAR inhibitory activity as compared to GK, which highlights its potential anti-diabetic activity. Usually, the activity of extracts cannot be correlated with individual compounds due to the presence of a diverse range of compounds, with each of the compounds differing in the range of activity. Thus, although RLAR activity was reasonable in our study, the individual compounds present in the kohlrabi cultivars may be more potent.
Increasing evidence in both experimental and clinical studies suggests that oxidative stress plays a major role in the pathogenesis of both types of DM. Abnormally high levels of free radicals and the simultaneous decline of antioxidant defense mechanisms can lead to the damage of cellular organelles that can promote the development of complications of DM (49,50). Consistently, our results show that potent anti-diabetic activity can be correlated with potent antioxidant properties conferred by phenolic compounds. Our study reveals the potent activity of two kohlrabi cultivars, establishing them as an alternate source of anti-diabetic agents.
New therapeutic innovations that are dedicated to prevent DM and DM complications are of interest. Our study distinguished the effectiveness of two kohlrabi cultivars and found that the RK cultivar shows potent anti-diabetic (PTP1B and RLAR), antioxidant (DPPH, ABTS, and ONOO−), and anti-inflammatory (NO) activity. In addition to the presence of anthocyanins, isothiocyanates, and glucosinolates, our study revealed the presence of phenolic compounds responsible for anti-oxidant activity with the potential to prevent inflammation and DM. Thus, the multifactorial activity of kohlrabi, and in particular the RK cultivar, shows promising anti-diabetic activity as a new alternative therapeutic agent for the treatment of DM as well as its complications.
Footnotes
AUTHOR DISCLOSURE STATEMENT
The authors declare no conflict of interest.
REFERENCES
- 1.Olokoba AB, Obateru OA, Olokoba LB. Type 2 diabetes mellitus: a review of current trends. Oman Med J. 2012;27:269–273. doi: 10.5001/omj.2012.68. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Khazrai YM, Defeudis G, Pozzilli P. Effect of diet on type 2 diabetes mellitus: a review. Diabetes Metab Res Rev. 2014;30:24–33. doi: 10.1002/dmrr.2515. [DOI] [PubMed] [Google Scholar]
- 3.Devasagayam TP, Tilak JC, Boloor KK, Sane KS, Ghaskadbi SS, Lele RD. Free radicals and antioxidants in human health: current status and future prospects. J Assoc Physicians India. 2004;52:794–804. [PubMed] [Google Scholar]
- 4.Singh PP, Mahadi F, Roy A, Sharma P. Reactive oxygen species, reactive nitrogen species and antioxidants in etiopathogenesis of diabetes mellitus type-2. Indian J Clin Biochem. 2009;24:324–342. doi: 10.1007/s12291-009-0062-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Lee HS. Rat lens aldose reductase inhibitory activities of Coptis japonica root-derived isoquinoline alkaloids. J Agric Food Chem. 2002;50:7013–7016. doi: 10.1021/jf020674o. [DOI] [PubMed] [Google Scholar]
- 6.Jung HA, Islam MN, Lee CM, Oh SH, Lee S, Jung JH, Choi JS. Kinetics and molecular docking studies of an anti-diabetic complication inhibitor fucosterol from edible brown algae Eisenia bicyclis and Ecklonia stolonifera. Chem Biol Interact. 2013;206:55–62. doi: 10.1016/j.cbi.2013.08.013. [DOI] [PubMed] [Google Scholar]
- 7.Greenfield JR, Campbell LV. Relationship between inflammation, insulin resistance and type 2 diabetes: cause or effect? Curr Diabetes Rev. 2006;2:195–211. doi: 10.2174/157339906776818532. [DOI] [PubMed] [Google Scholar]
- 8.Luft VC, Schmidt MI, Pankow JS, Couper D, Ballantyne CM, Young JH, Duncan BB. Chronic inflammation role in the obesity-diabetes association: a case-cohort study. Diabetol Metab Syndr. 2013;5:31. doi: 10.1186/1758-5996-5-31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Montane J, Cadavez L, Novials A. Stress and the inflammatory process: a major cause of pancreatic cell death in type 2 diabetes. Diabetes Metab Syndr Obes. 2014;7:25–34. doi: 10.2147/DMSO.S37649. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Narmadha R, Devaki K. In vitro antioxidant activity and in vitro α-glucosidase and α-amylase inhibitory activity of Barleria cristata L. RJPBCS. 2012;3:780. [Google Scholar]
- 11.Wang Y, Xiang L, Wang C, Tang C, He X. Antidiabetic and antioxidant effects and phytochemicals of mulberry fruit (Morus alba L.) polyphenol enhanced extract. PLoS ONE. 2013;8:e71144. doi: 10.1371/journal.pone.0071144. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Percival J. Agricultural botany: Theoretical and practical. 8th ed. Duckworth; London, UK: 1936. p. 839. [Google Scholar]
- 13.Kaloo G, Bergh BO. Genetic improvement of vegetable crops. 1st ed. Pergamon press Ltd; Oxford, UK: 1993. p. 191. [DOI] [Google Scholar]
- 14.Lim JH, Park KJ, Jeong JW, Park JJ, Kim BK, Kim JC, Jeong SW. Antioxidant activity and antioxidant compounds in edible sprouts. FASEB J. 2013;27:lb260. [Google Scholar]
- 15.Baenas N, Moreno DA, García-Viguera C. Selecting sprouts of Brassicaceae for optimum phytochemical composition. J Agric Food Chem. 2012;60:11409–11420. doi: 10.1021/jf302863c. [DOI] [PubMed] [Google Scholar]
- 16.Choi SH, Ryu DK, Park SY, Ann KG, Lim YP, An GH. Composition analysis between kohlrabi (Brassica oleracea var. gongylodes) and radish (Raphanus sativus) Kor J Hort Sci Technol. 2010;28:469–475. [Google Scholar]
- 17.Kim DB, Oh JW, Shin GH, Kim YH, Lee JS, Park IJ, Cho JH, Lee OH. Inhibitory effect of kohlrabi juices with antioxidant activity on oxidative stress in human dermal fibroblasts (LB394) FASEB J. 2014;28:LB394. [Google Scholar]
- 18.Warne LGG. Kohlrabi as a source of vitamin C. Br Med J. 1942;1:387. doi: 10.1136/bmj.1.4237.387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Gerendás J, Breuning S, Stahl T, Mersch-Sundermann V, Mühling KH. Isothiocyanate concentration in Kohlrabi (Brassica oleracea L. var. gongylodes) plants as influenced by sulfur and nitrogen supply. J Agric Food Chem. 2008;56:8334–8342. doi: 10.1021/jf800399x. [DOI] [PubMed] [Google Scholar]
- 20.You Y, Wu Y, Mao J, Zou L, Liu S. Screening of Chinese brassica species for anti-cancer sulforaphane and erucin. Afr J Biotechnol. 2008;7:147–152. [Google Scholar]
- 21.Higdon JV, Delage B, Williams DE, Dashwood RH. Cruciferous vegetables and human cancer risk: epidemiologic evidence and mechanistic basis. Pharmacol Res. 2007;55:224–236. doi: 10.1016/j.phrs.2007.01.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Rasal V, Shetty B, Sinnathambi A, Yeshmaina S, Ashok P. Antihyperglycaemic and antioxidant activity of Brassica oleracea in streptozotocin diabetic rats. The Internet Journal of Pharmacology. 2005 IJPHARM/4/2/7659. [Google Scholar]
- 23.Ainsworth EA, Gillespie KM. Estimation of total phenolic content and other oxidation substrates in plant tissues using Folin-Ciocalteu reagent. Nat Protoc. 2007;2:875–877. doi: 10.1038/nprot.2007.102. [DOI] [PubMed] [Google Scholar]
- 24.Blois MS. Antioxidant determination by the use of a stable free radical. Nature. 1958;181:1199–1200. doi: 10.1038/1811199a0. [DOI] [Google Scholar]
- 25.Re R, Pellegrini N, Proteggente A, Pannala A, Yang M, Rice-Evans C. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic Biol Med. 1999;26:1231–1237. doi: 10.1016/S0891-5849(98)00315-3. [DOI] [PubMed] [Google Scholar]
- 26.Kooy NW, Royall JA, Ischiropoulos H, Beckman JS. Peroxynitrite-mediated oxidation of dihydrorhodamine 123. Free Radic Biol Med. 1994;16:149–156. doi: 10.1016/0891-5849(94)90138-4. [DOI] [PubMed] [Google Scholar]
- 27.Cui L, Na MK, Oh HC, Bae EY, Jeong DG, Ryu SE, Kim SH, Kim BY, Oh WK, Ahn JS. Protein tyrosine phosphatase 1B inhibitors from morus root bark. Bioorg Med Chem Lett. 2006;16:1426–1429. doi: 10.1016/j.bmcl.2005.11.071. [DOI] [PubMed] [Google Scholar]
- 28.Hayman S, Kinoshita JH. Isolation and properties of lens aldose reductase. J Biol Chem. 1965;240:877–882. [PubMed] [Google Scholar]
- 29.Krentz AJ, Bailey CJ. Oral antidiabetic agents: current role in type 2 diabetes mellitus. Drugs. 2005;65:385–411. doi: 10.2165/00003495-200565030-00005. [DOI] [PubMed] [Google Scholar]
- 30.Nabi SA, Kasetti RB, Sirasanagandla S, Tilak TK, Kumar MV, Rao CA. Antidiabetic and antihyperlipidemic activity of Piper longum root aqueous extract in STZ induced diabetic rats. BMC Complement Altern Med. 2013;13:37. doi: 10.1186/1472-6882-13-37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Park WT, Kim JK, Park S, Lee SW, Li X, Kim YB, Uddin MR, Park NI, Kim SJ, Park SU. Metabolic profiling of glucosinolates, anthocyanins, carotenoids, and other secondary metabolites in kohlrabi (Brassica oleracea var. gongylodes) J Agric Food Chem. 2012;60:8111–8116. doi: 10.1021/jf301667j. [DOI] [PubMed] [Google Scholar]
- 32.Fahey JW, Zalcmann AT, Talalay P. The chemical diversity and distribution of glucosinolates and isothiocyanates among plants. Phytochemistry. 2001;56:5–51. doi: 10.1016/S0031-9422(00)00316-2. [DOI] [PubMed] [Google Scholar]
- 33.Stratil P, Klejdus B, Kubán V. Determination of total content of phenolic compounds and their antioxidant activity in vegetables–evaluation of spectrophotometric methods. J Agric Food Chem. 2006;54:607–616. doi: 10.1021/jf052334j. [DOI] [PubMed] [Google Scholar]
- 34.Jacobo-Velázquez DA, Cisneros-Zevallos L. Correlations of antioxidant activity against phenolic content revisited: a new approach in data analysis for food and medicinal plants. J Food Sci. 2009;74:R107–R113. doi: 10.1111/j.1750-3841.2009.01352.x. [DOI] [PubMed] [Google Scholar]
- 35.Kähkönen MP, Hopia AI, Vuorela HJ, Rauha JP, Pihlaja K, Kujala TS, Heinonen M. Antioxidant activity of plant extracts containing phenolic compounds. J Agric Food Chem. 1999;47:3954–3962. doi: 10.1021/jf990146l. [DOI] [PubMed] [Google Scholar]
- 36.Bhagwat S, Haytowitz DB, Holden JM. USDA database for the flavonoid content of selected foods release 2.1. Agricultural Research Service, U.S. Department of Agriculture; Beltsville, MD, USA: 2007. [Google Scholar]
- 37.Closa D, Folch-Puy E. Oxygen free radicals and the systemic inflammatory response. IUBMB Life. 2004;56:185–191. doi: 10.1080/15216540410001701642. [DOI] [PubMed] [Google Scholar]
- 38.Alhakmani F, Kumar S, Khan SA. Estimation of total phenolic content, in-vitro antioxidant and anti-inflammatory activity of flowers of Moringa oleifera. Asian Pac J Trop Biomed. 2013;3:623–627. doi: 10.1016/S2221-1691(13)60126-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Zhang L, Ravipati AS, Koyyalamudi SR, Jeong SC, Reddy N, Smith PT, Bartlett J, Shanmugam K, Münch G, Wu MJ. Antioxidant and anti-inflammatory activities of selected medicinal plants containing phenolic and flavonoid compounds. J Agric Food Chem. 2011;59:12361–12367. doi: 10.1021/jf203146e. [DOI] [PubMed] [Google Scholar]
- 40.Yun HY, Dawson VL, Dawson TM. Neurobiology of nitric oxide. Crit Rev Neurobiol. 1996;10:291–316. doi: 10.1615/CritRevNeurobiol.v10.i3-4.20. [DOI] [PubMed] [Google Scholar]
- 41.Feldhammer M, Uetani N, Miranda-Saavedra D, Tremblay ML. PTP1B: a simple enzyme for a complex world. Crit Rev Biochem Mol Biol. 2013;48:430–445. doi: 10.3109/10409238.2013.819830. [DOI] [PubMed] [Google Scholar]
- 42.Kusari J, Kenner KA, Suh KI, Hill DE, Henry RR. Skeletal muscle protein tyrosine phosphatase activity and tyrosine phosphatase 1B protein content are associated with insulin action and resistance. J Clin Invest. 1994;93:1156–1162. doi: 10.1172/JCI117068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Harley EA, Levens N. Protein tyrosine phosphatase 1B inhibitors for the treatment of type 2 diabetes and obesity: recent advances. Curr Opin Investig Drugs. 2003;4:1179–1189. [PubMed] [Google Scholar]
- 44.Zabolotny JM, Kim YB, Welsh LA, Kershaw EE, Neel BG, Kahn BB. Protein-tyrosine phosphatase 1B expression is induced by inflammation in vivo. J Biol Chem. 2008;283:14230–14241. doi: 10.1074/jbc.M800061200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Tarr JM, Kaul K, Chopra M, Kohner EM, Chibber R. Pathophysiology of diabetic retinopathy. ISRN Ophthalmology. 2013;2013:343560. doi: 10.1155/2013/343560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Chung SS, Ho EC, Lam KS, Chung SK. Contribution of polyol pathway to diabetes-induced oxidative stress. J Am Soc Nephrol. 2003;14:S233–S236. doi: 10.1097/01.ASN.0000077408.15865.06. [DOI] [PubMed] [Google Scholar]
- 47.Hashim Z, Zarina S. Osmotic stress induced oxidative damage: possible mechanism of cataract formation in diabetes. J Diabetes Complications. 2012;26:275–279. doi: 10.1016/j.jdiacomp.2012.04.005. [DOI] [PubMed] [Google Scholar]
- 48.Greene DA, Lattimer SA, Sima AA. Sorbitol, phosphoinositides, and sodium-potassium-ATPase in the pathogenesis of diabetic complications. N Engl J Med. 1987;316:599–606. doi: 10.1056/NEJM198703053161007. [DOI] [PubMed] [Google Scholar]
- 49.Maritim AC, Sanders RA, Watkins JB., 3rd Diabetes, oxidative stress, and antioxidants: a review. J Biochem Mol Toxicol. 2003;17:24–38. doi: 10.1002/jbt.10058. [DOI] [PubMed] [Google Scholar]
- 50.Oberley LW. Free radicals and diabetes. Free Radic Biol Med. 1988;5:113–124. doi: 10.1016/0891-5849(88)90036-6. [DOI] [PubMed] [Google Scholar]