Abstract
Interleukin (IL)-13 is a T helper 2 (Th2) cytokine that plays important roles in the pathogenesis of asthma. IL-13 induces hypersensitivity of the airways, increased mucous production, elevated serum immunoglobulin (Ig) E levels, and increased numbers of eosinophils. Many patients with IgG4-related disease have allergic backgrounds and show elevated serum IgE levels and an increase in the number of eosinophils. Upregulation of Th2/regulatory T (Treg) cytokines, including IL-13, has been detected in affected tissues of patients with IgG4-related disease. We previously reported that mast cells might be responsible for the production of the Th2/Treg cytokines IL-4, IL-10, and transforming growth factor (TGF)-β1 in IgG4-related disease. In this study, immunohistochemical analysis showed increased numbers of IL-13-positive mast cells in IgG4-related disease, which suggests that mast cells also produce IL-13 and contribute to elevation of serum IgE levels and eosinophil infiltration in IgG4-related disease.
Immunoglobulin (Ig)G4-related disease is a recently established systemic disorder with characteristic clinicopathological features that frequently affects the exocrine organs, including the pancreas, salivary glands, and lacrimal glands, although various systemic organs can also be involved1. The pathogenesis of IgG4-related disease remains unclear and controversial; however, upregulation of T helper (Th) 2 and regulatory T (Treg) cytokines in diseased areas have been reported2,3. To date, these Th2 and Treg reactions have been considered to form characteristic pathological features, including lymphoplasmacytic infiltration, storiform fibrosis, and increased numbers of IgG4-positive plasma cells and eosinophils2,3.
Interleukin (IL)-13 is one of such Th2 cytokines and is closely related to the pathogenesis of asthma4. IL-13 provokes hyperreactivity of the airways, increases in goblet cell numbers and mucous production, activation of fibroblasts, class switching of B-cell antibody from IgM to IgE, and increased numbers of eosinophils in the blood4,5. IL-13 is also considered to be associated with elevated serum IgE levels and increased numbers of eosinophils in IgG4-related disease6.
Upregulation of IL-13 in tissues of patients with IgG4-related disease has been previously demonstrated, and Th2 cells are the most likely candidates for the production of IL-132. However, it has not been confirmed whether Th2/Treg cells directly produce these important cytokines. We recently reported that mast cells can produce Th2 and Treg cytokines, including IL-4, IL-10, and transforming growth factor (TGF)-β1, in IgG4-related disease7. Hence, the potential of mast cells to produce IL-13 was examined in this study.
Methods
Samples
Tissue samples from 9 cases of submandibular gland IgG4-related disease were obtained. Samples from 5 cases of submandibular sialolithiasis and 6 normal submandibular glands were also obtained and used as disease and healthy controls, respectively. These samples were also used in our previous study7. Serum IgG4 levels were elevated in all cases of IgG4-related disease. Samples from formalin-fixed, paraffin-embedded specimens were used for immunohistochemistry and dual immunofluorescence analyses. Informed consent for the use of their samples in research was obtained from all patients.
Methods
The following methods were carried out in accordance with the approved guidelines. All experimental protocols were approved by the Institutional Review Board at Okayama University.
Histological examination and immunohistochemistry
All of the diseased and normal tissue samples used in this study were surgically resected specimens of the submandibular glands. The specimens were fixed in 10% formaldehyde and embedded in paraffin. Serial 4-μm-thick sections were cut from the blocks of paraffin-embedded tissues and stained with hematoxylin and eosin (H&E). The sections were immunohistochemically stained using an automated Bond Max stainer (Leica Biosystems; Wetzlar, Germany). The following primary antibodies were used: IL-13 (2B5; 1:300; Abnova; Taipei City, Taiwan), c-kit/CD117 (YR145; 1:100; EPITOMICS; Burlingame, CA, USA), IgG (polyclonal; 1:20,000; Dako; Glostrup, Denmark), and IgG4 (HP6025; 1:400; The Binding Site; Birmingham, UK).
Confirmation of histological diagnosis of IgG4-related disease
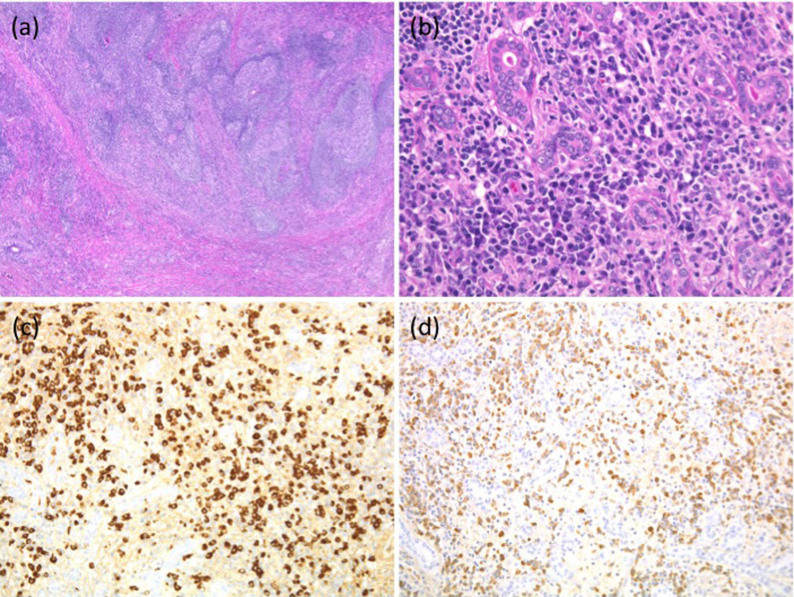
All samples from patients with IgG4-related disease showed typical histological features, including lymphoplasmacytic infiltration and dense fibrosis (Fig 1a, 1b). In accordance with the consensus statement on the pathological features of IgG4-related disease published in 20128, 3 different high-power fields (HPFs) (eyepiece, ×10; lens, ×40) were examined to calculate the average number of IgG4-positive cells per HPF and the IgG4-/IgG-positive cell ratio. In all patients with IgG4-related disease, the average number of IgG4-positive plasma cells was >100 cells/HPF, and the ratio of IgG4-/IgG-positive cells was >40% (Fig. 1c, 1d).
Figure 1. Immunohistochemical analysis of IgG4-related disease.
(a) Tissue samples of patients with IgG4-related submandibular disease showed dense fibrosis with lymphoid follicles (hematoxylin and eosin [H&E] staining; original magnification × 40). (b) Lymphoplasmacytic infiltration was observed in the interfollicular areas (H&E, original magnification × 400). (c) Numerous IgG4-positive cells were detected (IgG4, original magnification × 200). (d) The IgG4/IgG-positive cell ratio was >0.4 (IgG, original magnification × 200).
Calculation of IL-13- and c-kit-positive cells
Cells that were positive for IL-13 and c-kit were counted in 5 and 3 different fields, respectively, that showed the highest density of positive cells (eye piece, ×10; lens, ×20). Two pathologists (M.T. and Y.S.) counted the positive cells, and the numbers were averaged. The average number of positive cells per square millimeter was calculated.
Dual immunofluorescence assays
For indirect dual immunofluorescence assays, paraffin sections were stained with the primary antibodies for c-kit and IL-13. Fluorescein isothiocyanate-conjugated secondary antibodies (Alexa Fluor anti-mouse 555 and Alexa Fluor anti-rabbit 488; both from Life Technologies, Carlsbad, CA, USA) were used at a dilution of 1:400. The stained specimens were examined with a conventional immunofluorescence microscope (IX71; Olympus; Tokyo, Japan).
Statistical analysis
Data are presented as mean ± standard deviation (SD) values. All statistical analyses were performed using the Mann–Whitney U-test with SPSS software (version 14.0; SPSS Inc.; Chicago, IL, USA). A probability value of <0.05 was considered to be statistically significant.
Results
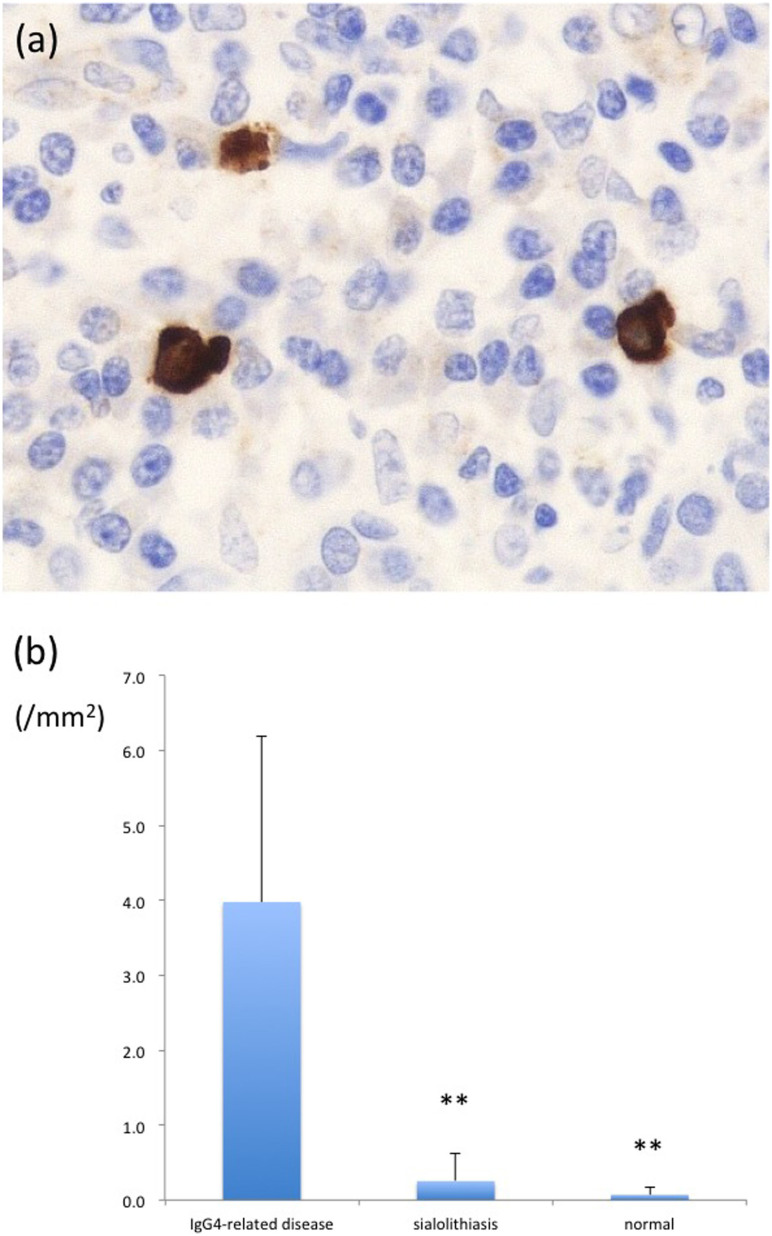
Many IL-13-positive cells were observed in tissues from patients with IgG4-related disease (Fig. 2a). The number of IL-13-positive cells was significantly increased in IgG4-related disease samples (4.00 ± 2.21 cells/mm2) compared to sialolithiasis samples (0.30 ± 0.48 cells/mm2) and normal submandibular glands (0.10 ± 0.11 cells/mm2; Fig. 2b). We used c-kit antibody as a marker of mast cells. The numbers of c-kit-positive cells were significantly higher in IgG4-related disease samples (72.2 ± 24.5 cells/mm2) than in the normal submandibular glands (30.0 ± 11.9 cells/mm2; p < 0.01), whereas no significant difference was observed between the number of mast cells in the IgG4-related disease and submandibular sialolithiasis samples (177 ± 269 cells/mm2; p = 0.73), as previously reported7.
Figure 2. Counts of IL-13-expressing cells by immunostaining.
(a) Immunostaining for IL-13 revealed strong cytoplasmic positivity. Positive cells were morphologically similar to mast cells (IL-13, original magnification × 400). (b) Significantly greater numbers of IL-13-positive cells were observed in the IgG4-related disease group than in the control group (p < 0.01).
The morphological features and distribution of the c-kit-positive mast cells were similar to those of IL-13-positive cells. Dual immunofluorescence assays revealed coexpression of IL-13 and c-kit (Fig. 3).
Figure 3. Dual fluorescent immunostaining for IL-13 and c-kit.
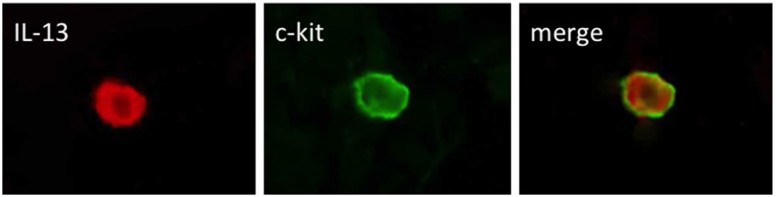
Dual fluorescent immunostaining detected positive cells for IL-13 and c-kit. The merged image demonstrates double positive-stained cells for IL-13 and c-kit.
Discussion
Even though Miklicz disease, now recognized as a member of the IgG4-related disease family, was first reported in the late 19th century, the concept of IgG4-related disease has only become well established in the 21st century9,10. IgG4-related disease was first described in the pancreas. It was initially considered an autoimmune disease, and was therefore termed “autoimmune pancreatitis” because of concomitant extra-pancreatic autoimmune diseases, e.g., Sjögren syndrome, with features including hypergammaglobulinemia, an elevated titer of anti-nuclear antigens, and good response to steroid therapy11. Subsequent research showed that these extra-pancreatic lesions were, in fact, features of multifocal IgG4-related disease; however, the relationship between IgG4-related disease and autoimmunity has not yet been elucidated12.
Although the pathogenesis of IgG4-related disease remains unclear, Th2/Treg immune reactions seem to contribute to disease formation2,3. Enhanced Th2/Treg reaction is important for the establishment of allergic disorders in general, and IgG4-related disease patients often have allergic backgrounds. However, the relationship between allergic backgrounds and pathogenesis of IgG4-related disease is controversial13. A recent study showed that the prevalence of atopy in patients with IgG4-related disease did not differ from that of the general population and the majority of the patients were non-atopic14. According to the study, non-atopic patients with IgG4-related disease also showed elevated IgE and peripheral blood eosinophila, which suggested that an enhanced Th2/Treg response is not related to allergic background but rather to IgG4-related disease itself14.
On the other hand, some reports have suggested the importance of allergic reactions in the pathogenesis of IgG4-related disease. IgG4 is a unique antibody with a poor ability to activate complements and cells because of its low affinity for C1q and Fc receptors15. Unlike other IgG subclasses, IgG4 has anti-inflammatory activity and seems to inhibit IgE-mediated type I allergic responses by competing with IgE15,16. For example, some cases of autoimmune pancreatitis following bronchial asthma have been reported17. Recently, a case of IgG4-related disease was found to have regressed with only treatment of an anti-histamine agent and no systemic steroid therapy18. Based on these findings, IgG4-related disease may be related to an aberrant anti-inflammatory activity against to the allergic reaction.
Mast cells are well known to play important roles in the immediate immune response and release of histamine granules upon binding to IgE. However, previous studies have also shown that mast cells release various cytokines and chemokines and participate in multiple immune reactions19,20. We previously reported the possible role of mast cells in the production of Th2/Treg cytokines (IL-4, IL-10, and TGF-β1) in IgG4-related disease7. In this study, we found that mast cells might also produce IL-13, suggesting that it is a key factor in the elevation of serum IgE levels and number of eosinophils associated with IgG4-related disease. As mast cells are closely related to allergic reaction and IgE stimulation, these results indirectly suggest that a background of an allergic disorder and elevated serum IgE levels can be a trigger for the upregulation of mast cell-derived Th2/Treg cytokines. Further studies on the role of mast cells in IgG4-related disease and their interaction with Th2/Treg cells are required.
Author Contributions
Conceived and designed the experiments: Y.S. Performed the experiments: M.T. and Y.S. Analyzed the data: Y.S., M.T., K.O., K.T. and Y.G. Contributed materials: Y.O. and T.T. Wrote the paper: M.T., Y.S. and T.Y. All authors read and approved the final manuscript.
Acknowledgments
This work was supported by a Grant-in-Aid for Scientific Research (C) (no. 24591447) from the Japan Society for the Promotion of Science and ‘Research on Measures for Intractable Disease' Project: matching fund subsidy from the Ministry of Health Labor and Welfare, Japan.
References
- Sato Y. et al. IgG4-related disease: Histological overview and pathology of hematological disorders. Pathol Int. 60, 247–258 (2010). [DOI] [PubMed] [Google Scholar]
- Zen Y. et al. Th2 and regulatory immune reactions are increased in immunoglobin G4-related sclerosing pancreatitis and cholangitis. Hepatology. 45, 1538–1546 (2007). [DOI] [PubMed] [Google Scholar]
- Tanaka A. et al. Th2 and regulatory immune reactions contribute to IgG4 production and the initiation of Mikulicz disease. Arthritis Rheum. 64, 254–263 (2012). [DOI] [PubMed] [Google Scholar]
- Corren J. Role of Interleukin-13 in Asthma. Curr Allergy Asthma Rep. 13, 415–420 (2013). [DOI] [PubMed] [Google Scholar]
- Klein Wolterink R. G. & Hendriks R. W. Type 2 innate lymphocytes in allergic airway inflammation. Curr Allergy Asthma Rep. 13, 271–280 (2013). [DOI] [PubMed] [Google Scholar]
- Moriyama M. et al. T helper subsets in Sjögren's syndrome and IgG4-related dacryoadenitis and sialoadenitis: A critical review. J Autoimmun. 51, 81–88 (2014). [DOI] [PubMed] [Google Scholar]
- Takeuchi M. et al. T helper 2 and regulatory T-cell cytokine production by mast cells: a key factor in the pathogenesis of IgG4-related disease. Mod Pathol. 27, 1126–1136 (2014). [DOI] [PubMed] [Google Scholar]
- Deshpande V. et al. Consensus statement on the pathology of IgG4-related disease. Mod Pathol. 25, 1181–1192 (2012). [DOI] [PubMed] [Google Scholar]
- Himi T. et al. A novel concept of Mikulicz's disease as IgG4-related disease. Auris Nasus Larynx. 39, 9–17 (2012). [DOI] [PubMed] [Google Scholar]
- Umehara H. et al. A novel clinical entity, IgG4-related disease (IgG4RD): general concept and details. Mod Rheumatol. 22, 1–14 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yoshida K. et al. Chronic pancreatitis caused by an autoimmune abnormality. Proposal of the concept of autoimmune pancreatitis. Dig Dis Sci. 40, 1561–1568 (1995). [DOI] [PubMed] [Google Scholar]
- Kamisawa T. et al. Close relationship between autoimmune pancreatitis and multifocal fibrosclerosis. Gut. 52, 683–687 (2003). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smarr C. B., Bryce P. J. & Miller S. D. Antigen-specific tolerance in immunotherapy of Th2-associated allergic diseases. Crit Rev Immunol. 33, 389–414 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Della Torre E. et al. Prevalence of atopy, eosinophila, and IgE elevation in IgG4-related disease. Allergy. 69, 269–272 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- van der Neut Kolfschoten M. et al. Anti-inflamatory activity of human IgG4 antibodies by dynamic Fab arm exchange. Science. 317, 1554–1557 (2007). [DOI] [PubMed] [Google Scholar]
- Aalberse R. C. & Schuurman J. IgG4 breaking the rules. Immunology. 105, 9–19 (2002). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ito S. et al. Three cases of bronchial asthma preceding Igg4-related autoimmune pancreatitis. Allergol Int. 61, 171–174 (2012). [DOI] [PubMed] [Google Scholar]
- Oshima K., Sato Y. & Yoshino T. A case of IgG4-related dacryoadenitis that regressed without systemic steroid administration. J Clin Exp Hematop. 53, 53–56 (2013). [DOI] [PubMed] [Google Scholar]
- Gri G. et al. Mast cell: an emerging partner in immune interaction. Front Immunol. 3, 120 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Amin K. The role of mast cells in allergic inflammation. Respir Med. 106, 9–14 (2012). [DOI] [PubMed] [Google Scholar]