Abstract
This study examines whether parental report of social-communicative and repetitive behaviors at 12 months can be helpful in identifying autism spectrum disorder (ASD) in younger siblings of children with ASD [high-risk (HR)-siblings]. Parents of HR-siblings and infants without a family history of ASD completed the First Year Inventory at 12 months. Developmental outcomes were based on 24- or 36-month assessments. HR-siblings later diagnosed with ASD showed greater impairments in social communication than those with other developmental outcomes based on parental and clinician ratings. Parental report of decline in play and communication and impaired vocal imitation correctly classified a majority of ASD cases with high specificity. These preliminary findings have important implications for the development of early screening instruments for ASD in HR-siblings.
Keywords: ASD, High-risk siblings, Screening, Imitation
Introduction
Autism spectrum disorder (ASD) is characterized by deficits in social communication and interaction as well as restricted and repetitive behaviors and interests (APA 2013), with a current prevalence of 11.3 per 1,000 children (CDC 2012). Due to the strong genetic basis of ASD, younger siblings of children with ASD (high-risk siblings, HR) are at an increased risk for developing ASD. Depending on the mode of sample ascertainment, recurrence rates of younger siblings of children with ASD have been reported between 6.9 % (Grønborg et al. 2013) and 18.7 % (Ozonoff et al. 2011). HR-siblings are also at risk of developing a range of social-communication and language delays, as well as behavioral rigidities that do not meet criteria for ASD (Georgiades et al. 2012; Landa et al. 2007; Macari et al. 2012; Messinger et al. 2013). The increased risk of ASD and related problems underscores the need for early detection of symptoms among HR-siblings and implementation of targeted interventions to improve their developmental outcomes.
Early detection of ASD among HR-siblings may be complicated for a number of reasons. One of these complexities shared by HR-siblings and the general population is the variable timing and patterns of symptom onset. Although initial observations suggested the presence of social abnormalities in the first year of life (Kanner 1943), later work revealed that in some children with ASD, symptoms may emerge in the second year of life after a period of more or less typical development, either as a result of regression or a failure to progress (Ozonoff et al. 2008a; Siperstein and Volkmar 2004). Partly for that reason, a majority of screening measures have been designed for children 16 months of age or older (Constantino et al. 2003; Robins et al. 2001; Siegel 2004). Screening for ASD in HR-siblings poses additional challenges unique to this cohort, as a fairly large proportion of HR-siblings who do not develop ASD exhibit signs of delayed or atypical social communication skills and unusual interests that may overlap with those seen in infants who later develop ASD and complicate the screening process (Georgiades et al. 2012; Macari et al. 2012; Messinger et al. 2013).
Despite these complexities, empirical studies suggest that as many as 40 % of HR-infants exhibit marked symptoms of ASD as early as 12 months (Macari et al. 2012). On a group level, 12-month-old infants later diagnosed with ASD exhibit reduced eye contact, orienting to name, requesting, joint attention (Ozonoff et al. 2010; Rozga et al. 2011; Zwaigenbaum et al. 2005), atypical speech-like vocalizations (Paul et al. 2011), poor imitation (Young et al. 2011), a limited inventory of gestures (Mitchell et al. 2006), and atypical object exploration (Ozonoff et al. 2008b). These findings suggest that already at 12 months, at least some of the HR-infants who later develop ASD begin to display signs of vulnerabilities in the core diagnostic domains. Moreover, detection of early signs of ASD among HR-siblings may be facilitated by parental experience with regard to early symptoms of ASD. In a recent study, the number of parental concerns about symptoms of ASD at 12 months was significantly associated with the diagnosis of ASD at 36 months (Ozonoff et al. 2009). Considering reports that in some children symptoms intensify in the second year (Landa et al. 2007), it is likely that not all children will be symptomatic at the age of 12 months, necessitating a multi-screen approach that allows for early detection of children who exhibit symptoms at younger ages, with additional screening to detect the children who show symptoms at a later age (Johnson and Myers 2007; Barbaro and Dissanayake 2013; Robins et al. 2014). However, given that a large proportion of infants is likely to be symptomatic at 12 months, it is essential to evaluate whether this group can be reliably identified among those with other difficulties and targeted for intervention. This has become increasingly important as research is emerging on the effectiveness of intervention targeted at infants exhibiting vulnerabilities in social and communicative domains associated with ASD around their first birthday (Koegel et al. 2014; Steiner et al. 2013; Green et al. 2013). At present, there has been little empirical work focused on identifying features in infants with familial risk for ASD that can be collected through parental report at 12 months. This task can be particularly daunting as unlike in general population studies (Barbaro and Dissanayake 2013; Reznick et al. 2007; Turner-Brown et al. 2012), the HR-siblings who later develop ASD need to be differentiated from those HR-siblings who might display some but not all features of ASD due to genetic liability.
To examine whether parental report can be useful for identifying HR-siblings later diagnosed with ASD as early as at 12 months, we have employed the FYI (Baranek et al. 2003). The FYI is a parental questionnaire designed to screen 12-month-old infants for the risk of ASD. It consists of 61 Likert scale or multiple-choice questions representing Social Communication and Sensory-Regulatory domains. The FYI was normed on a sample of 1,496 12-month-old infants from the general population (Reznick et al. 2007). A longitudinal study involving 699 children from the normalization sample (Turner-Brown et al. 2012) found that of the children who exceeded the cut-off score, 85 % exhibited developmental delays or concerns by the age of 3 and 31 % were diagnosed with ASD. In the same study 99 % of the children who scored below the cut-off did not exhibit symptoms of ASD. The predictive properties of the FYI have not been studied in HR-siblings.
The present study aims to (1) investigate whether parental ratings of social-communicative and repetitive behaviors at 12 months differ in HR-siblings later diagnosed with ASD from those with other developmental outcomes; (2) examine associations between parental report and clinician ratings on ASD-related symptoms; and (3) explore what specific behavioral features, reported by parents, may constitute possible warning signs for ASD on an individual level at the age of 12 months.
Methods
Participants included 96 infants: 71 HR-infants, defined by the presence of ASD in an older sibling, and 25 typically developing (TD) infants at low risk (LR) for ASD, with no family history of ASD in 1st or 2nd degree relatives. The ASD diagnosis in the older siblings was confirmed by clinical best estimate diagnosis (CBE) as well as the Autism Diagnostic Interview-Revised (Rutter et al. 2003) and/ or Autism Diagnostic Observation Schedule-Generic (ADOS-G) (Lord et al. 2000). All infants participated in an ongoing prospective longitudinal research program, and were enrolled into the study no later than at 6 months of age. Exclusionary criteria included gestational age below 34 weeks, hearing or visual impairment, seizure disorders, or known genetic disorders. Informed consent was obtained from all participants prior to their participation. This study was approved by the University’s Institutional Review Board.
HR-infants were assessed at 12, 18, 24 and 36 months of age. LR infants were assessed at 12, 18, and 24 months and at 30–36 months, underwent screening for social and language delays as well as emotional problems; screen positive cases were evaluated at 36 months. During the 12-month visit, prior to any developmental assessments, parents completed the FYI. At each time-point, the children were assessed with the Mullen Scales of Early Learning (MSEL) (Mullen 1995). Verbal and non-verbal developmental quotients (DQ) were derived by dividing the average age equivalent for the language scales (receptive and expressive) and non-language scales (visual reception and fine motor) by the child’s chronological age and multiplying by 100 (average age equivalent/chronological age × 100 = DQ). Social-communicative behaviors were assessed using the ADOS Toddler Module (ADOS-T) (Lord et al. 2012a, b) and ADOS-G (Lord et al. 2000). The ADOS provides Social Affect and Restrictive and Repetitive Behavior scores as well as a total algorithm score. The ADOS-T was administered at the 12-, 18-, and 24-month visits; at the 36-month visit either Module 1 or Module 2 was administered, depending on the child’s language level. All examiners had extensive experience in the assessment of young children with developmental disorders and had established reliability regarding administration and scoring of the assessment measures.
At the 18-, 24-, and 36-month assessments, a team of expert clinicians, blinded to the child’s risk status and results of the FYI, assigned a CBE diagnosis based on the results of the developmental, social-communicative, and language assessments as well as the child’s developmental and medical history (see Chawarska et al. 2009 for details). HR children were classified as having HR-ASD if they displayed marked abnormalities in social interaction and communication skills as well as atypical sensory or repetitive behaviors during the 18, 24, or 36 month assessments (n = 16). The HR-atypical development (HR-ATYP, n = 36) group included HR children who had a score more than 1.5 standard deviations below the mean on at least one MSEL scale, exhibited behavioral problems, social-communicative delays, atypical language features (e.g., echolalia), or showed unusual sensory and repetitive behaviors at any time between 18 and 36 months, but did not meet criteria for ASD. Children were classified as TD if they did not display clinically relevant clinical symptoms at any time between 18 and 36 months, which resulted in two TD groups according to risk status: HR-TD (n = 19), and LR-TD (n = 25). All infants completed a 24-month assessment. Sixty-seven percent of all HR-siblings, including 94 % of infants from the HR-ASD group, also completed a 36-month assessment.
Of the 96 children included in the study, 68 % were male, with a higher percentage of males in the HR-ATYP group than the HR-TD and LR-TD groups (Table 1); 94 % of parents identified their child’s race as Caucasian, and 13 % identified their child’s ethnicity as Hispanic. Ninety-one percent of mothers and 85 % of fathers completed a 4-year college degree. There were no significant differences in race, ethnicity, or educational status across study groups. As expected, at 24 months the HR-ASD group had significantly higher ADOS-T Social Affect scores than the HR-ATYP, HR-TD and LR-TD groups and a higher Restricted and Repetitive Behaviors score at 24 months than the HR-TD and LR-TD groups. Also, as expected, the verbal developmental quotient (DQ) at 24 months was significantly lower for the HR-ASD group than for the HR-TD and LR-TD groups.
Table 1.
Sample characteristics
| Characteristic | HR-ASD (n = 16) |
HR-ATYP (n = 36) |
HR-TD (n = 19) |
LR-TD (n = 25) |
Group comparison |
|
|---|---|---|---|---|---|---|
| F (3, 92) | p value | |||||
| % Male | 75.0 %a,b | 91.7 %b | 47.4 %a | 44.0 %a | 8.0 | 0.001 |
| Race/ethnicity | ||||||
| % Caucasian | 93.8 % | 94.9 % | 100 % | 88.0 % | 0.9 | 0.45 |
| % Hispanic | 12.5 % | 13.9 % | 15.8 % | 8.0 % | 0.2 | 0.88 |
| Completed college | ||||||
| % Mothers | 93.8 % | 83.3 % | 94.7 % | 96.0 % | 1.2 | 0.31 |
| % Fathers | 81.3 % | 82.4 % | 78.9 % | 95.7 % | 0.96 | 0.41 |
| 24-Month data (SD) | ||||||
| ADOS-T SA | 11.1 (5.8)a | 5.8 (3.4)b | 2.0 (1.7)c | 2.9 (2.5)c | 24.9 | <0.001 |
| ADOS-T RRB | 2.3 (1.5)a | 1.0 (1.0)b | 0.47 (0.61)b | 0.52 (0.59)b | 13.1 | <0.001 |
| Verbal DQ | 97.9 (29.6)a | 96.9 (22.1)a | 122.7 (16.9)b | 125.2 (14.1)b | 13.9 | <0.001 |
| Non verbal DQ | 102.3 (19.6)a,b | 98.2 (11.3)a | 111.6 (11.6)b,c | 117.2 (11.0)c | 13.0 | <0.001 |
SD standard deviation, DQ developmental quotient, SA social affect score, RRB restrictive and repetitive behavior score, N/A not applicable
Different superscripts within each row differ at least at the p < 0.05 level after Bonferroni correction
The First Year Inventory
The FYI (Baranek et al. 2003) consists of 61 questions: 47 formatted on a 4-point Likert scale from “never” to “often,” and 14 are multiple choice questions inquiring about, for instance, ways that the parent may be able to make their baby laugh, with several options from initiation of a social smile to various physical games. The questions are divided into Social Communication and Sensory-Regulatory domains, each of which is subdivided into four constructs. The constructs in the Social Communication domain are: Social Orienting and Receptive Communication, Social Affective Engagement, Imitation, and Expressive Language. The constructs in the Sensory-Regulatory domain are: Sensory Processing, Regulatory Patterns, Reactivity, and Repetitive Behaviors. Risk scores are generated based on assigned risk points that were derived from the normative sample (Reznick et al. 2007). Each construct score ranges from 0 to 50 on a semi-logarithmic scale, with higher scores indicating more symptoms. Each domain score is the average of its four construct scores. In a recently published longitudinal study, Turner-Brown et al. (2012) found that the optimal FYI cut-off scores for the general population were 22.5 for the Social Communication domain and 14.8 for the Sensory Regulatory domain and that its positive predictive ability was enhanced by requiring both domain cut-off scores be reached.
Statistical Analysis
To compare study groups on parental and clinician ratings of their social and repetitive behaviors as measured by the FYI and ADOS-T, we used a series of between-group analyses of variance (ANOVAs) with significant omnibus effects followed by post hoc Tukey’s HSD tests. Cohen’s d effect sizes were computed for all comparisons. Pearson’s correlation was used to assess for associations between parental report on the FYI and clinician ratings on the ADOS-T in the corresponding domains.
To identify the combination of individual FYI questions that were most predictive of an ASD outcome among HR-infants, we employed classification and regression tree (CART) analysis (Breiman et al. 1984). CART analysis is a decision-tree technique that uses recursive partitions of the data to predict a categorical or continuous response variable. A decision tree is a flow-chart-like structure, where each internal node denotes a test on an attribute (e.g., an FYI item), each branch represents the outcome of a test (e.g., the score of the item), and each leaf represents a class label (e.g., ASD versus non-ASD). At each step, the model selects the best variable (FYI item) and cutoff score among all available FYI items to make a partition. The nested structure of partitions within CART analysis naturally incorporates interactions among variables in the model, and the option to stop the growth of the tree at any partition (i.e., “pruning” the tree) provides a method of variable selection by predictive importance. FYI items and cutoffs are chosen by the CART algorithm for their ability to correctly classify ASD versus non-ASD cases. The selection process stops when additional items yield only marginal improvements in classification accuracy. The resulting sequence of partitions can be displayed graphically in a tree diagram. Each final subgroup, or “leaf”, is dependent upon the interaction of all the FYI items that define the leaf. For example, a leaf specified by three variables is determined by the joint scores of those three variables; failure to meet any one of the three variables’ cutoff criteria would exclude a subject from the leaf. To prevent over-fitting the model to this dataset and to increase the generalizability of the model, we used tenfold cross-validation on the number of leaves and pruned to the size that minimized out-of-sample misclassification. For other examples of recent application of CART analysis with high-risk phenotypes see Macari and colleagues (2012) and Lord and colleagues (2012b). All analyses were conducted in SPSS Version 19 (IBM 2010) and R (2011).
Results
Parent Report: FYI Domains and Constructs
A series of between group ANOVAs on domain and construct scores indicated significant between-group differences in the Social Communication domain [F (3,92) = 5.2, p = 0.002] and two of its four constructs: Social Orienting and Receptive Communication [F (3,92) = 3.0, p = 0.021] and Imitation [F (3,92) = 6.0, p = 0.001] (Table 2). There were no significant group differences for the Sensory-Regulatory domain score or any of its constructs.
Table 2.
FYI mean scores and ANOVA results by diagnostic category
| Domain/construct | Mean scores by diagnosis (SD) |
F (3, 92) F (p value) |
Post-hoc comparisons (p value) |
|||
|---|---|---|---|---|---|---|
| HR-ASD | HR-ATYP | HR-TD | LR-TD | |||
| Social communication domain | 19.0 (14.3) | 10.1 (9.4) | 10.3 (10.8) | 6.0 (7.8) | 5.2 (0.002) | ASD > ATYP (0.025) |
| ASD > LR-TD (0.001) | ||||||
| Social orienting and receptive communication | 17.1 (18.2) | 8.4 (11.7) | 9.6 (15.1) | 4.9 (8.6) | 3.0 (0.021) | ASD > LR-TD (0.021) |
| Social affective engagement | 14.3 (19.6) | 7.6 (12.6) | 9.9 (16.7) | 4.9 (9.8) | 1.6 (0.21) | NS |
| Imitation | 23.7 (18.4) | 8.4 (14.3) | 5.8 (12.2) | 6.2 (13.5) | 6.0 (0.001) | ASD > ATYP (0.004) |
| ASD > HR-TD (0.002) | ||||||
| ASD > LR-TD (0.002) | ||||||
| Expressive communication | 20.9 (19.5) | 16.0 (17.0) | 15.7 (15.1) | 8.2 (11.0) | 2.4 (0.076) | NS |
| Sensory-regulatory domain | 5.0 (7.8) | 4.1 (5.1) | 3.8 (5.3) | 5.5 (6.8) | 0.36 (0.79) | NS |
| Sensory processing | 7.8 (11.9) | 2.4 (4.4) | 2.3 (8.1) | 2.8 (8.5) | 2.0 (0.12) | NS |
| Regulatory patterns | 7.9 (16.9) | 9.2 (17.5) | 6.4 (15.2) | 5.1 (14.2) | 0.34 (0.80) | NS |
| Reactivity | 0.0 (0.0) | 3.2 (10.8) | 5.5 (13.1) | 10.1 (18.5) | 2.3 (0.08) | NS |
| Repetitive play and behavior | 4.2 (10.7) | 1.7 (4.5) | 1.2 (3.5) | 3.8 (6.9) | 1.2 (0.32) | NS |
Domain names are in bold
SD standard deviation, NS not significant
Post-hoc analyses indicated that infants later diagnosed with HR-ASD had significantly higher scores on the Social Communication domain compared to those classified as HR-ATYP (d = 0.75) and LR-TD (d = 1.18). The HR-ASD group also had a significantly higher Social Orienting and Receptive Communication construct score than the LR-TD (d = 0.91) group, but not the HR-ATYP or HR-TYP groups. Finally, the HR-ASD group had higher Imitation construct scores than the HR-ATYP (d = 0.94), HR-TD (d = 1.17), and LR-TD (d = 1.10) groups, with large effect sizes for each comparison. This analysis indicated that the Imitation construct, which taps into early emerging motor, vocal, and social imitation skills, was the most helpful in separating the HR-ASD group from other HR groups including those with a typical developmental patterns.
Moderate effect sizes were present despite the lack of statistical difference between the HR-ASD and HR-ATYP groups on the Social Orienting and Receptive Communication (d = 0.58) and Sensory Processing constructs (d = 0.66) and between the mean HR-ASD and HR-TD scores of the Social Communication domain (d = 0.69) and Sensory Processing construct (d = 0.55), suggesting potentially higher variability among HR-siblings at 12 months with regard to the emergence of these classes of behaviors.
Direct Assessment: ADOS-T Social and Repetitive Behaviors Domains
There was a significant group effect for ADOS-T SA score, F (3, 95) = 14.83, p < 0.001 (Table 3). Post-hoc comparisons indicated that infants later diagnosed with ASD displayed higher SA scores than HR-ATYP (d = 1.19), HR-TYP (d = 1.69), and LR-TYP (d = 2.42) groups at 12 months. There were no significant group differences with regard to ADOS-T RRB scores, F (3, 95) = 2.28, p = 0.066.
Table 3.
Mean ADOS-T scores at 12 months by diagnostic category
| Domain | Mean scores by diagnosis (SD) |
F (3, 95) F (p value) |
Post-hoc comparisons (p value) |
|||
|---|---|---|---|---|---|---|
| HR-ASD | HR-ATYP | HR-TD | LR-TD | |||
| ADOS-T SA | 13.9 (3.5) | 9.3 (4.2) | 6.8 (4.8) | 6.0 (3.1) | 14.8 (0.001) | ASD > HR-ATYP (0.001) |
| ASD > HR-TD (<0.001) | ||||||
| ASD > LR-TD (<0.001) | ||||||
| ADOS-T RRB | 1.7 (1.5) | 0.97 (1.3) | 0.79 (0.86) | 0.72 (1.1) | 2.5 (0.085) | NS |
SD standard deviation, NS not significant
Comparison Between ADOS-T and FYI Scores
There was a significant correlation in the combined samples between parental ratings of social behaviors on the FYI (Social Communication domain) and clinician observation of social behaviors on the ADOS-T (SA score) [r(96) = 0.44, p < 0.001] but not for repetitive and atypical sensory behaviors (FYI Sensory-Regulatory domain; ADOS-T RRB score) [r(96) = 0.04, p = 0.698]. The same comparison in the HR only sample (n = 71) yielded similar results r(71) = 0.43 (p < 0.001) for the social and [r(71) = 0.14 (p = 0.233] and repetitive and atypical sensory behavior domains.
Predicting ASD Outcome in HR-Siblings Based on Individual FYI Items
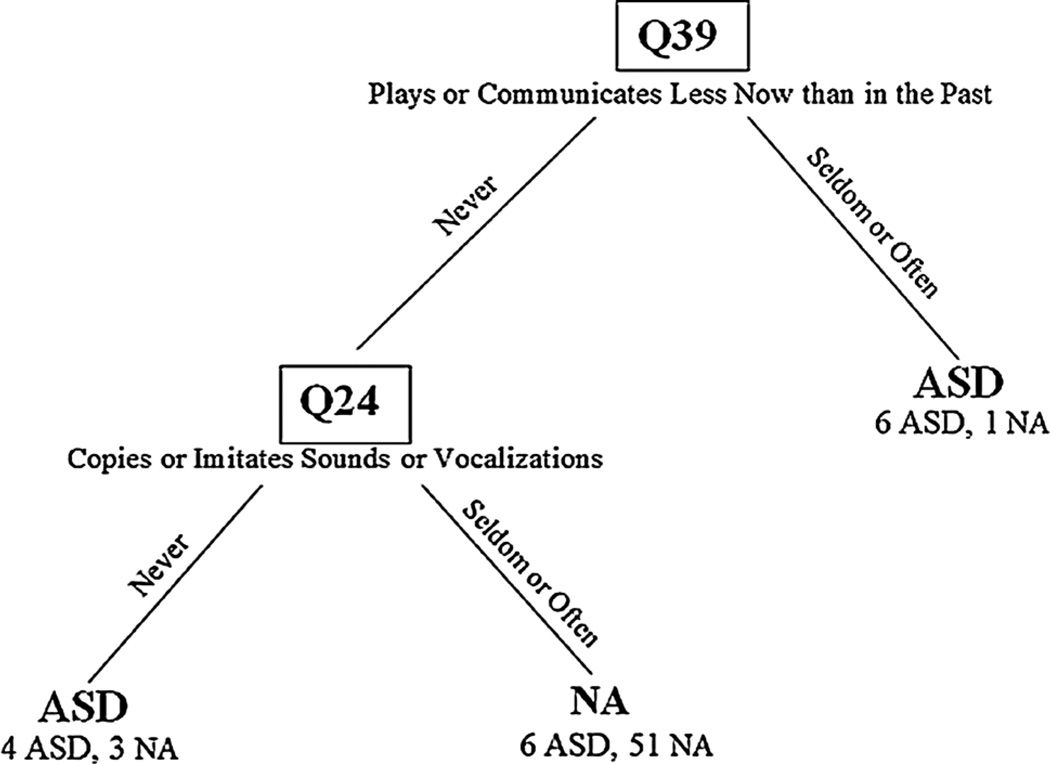
The CART analysis indicated that the optimal out-of-sample prediction rates were obtained with a tree consisting of three leaves (Fig. 1). Using questions, Q39 (“Do you get the feeling that your baby plays or communicates with you less now than in the past?”) and Q24 (“Does your baby copy or imitate you when you make sounds or noises with your mouth?”), the model correctly identified 10 of 16 (63 %) of the ASD group and 51 of 55 (93 %) of the Non-ASD group. Of the properly identified ASD infants, six were seen by their parents as engaging less frequently in play or communication than in the past, and another four were rated as having poor vocal imitation skills. The six remaining infants with ASD were misclassified as non-ASD: parents of these children did not report a loss of play and communication skills and reported typical vocal imitation. On direct assessment at 12 months none of these six infants exhibited delays or atypical behaviors. Infants without ASD who were placed by the CART analysis in the ASD ‘leaves’ (n = 4) were developing atypically as manifested by their diagnoses of expressive language delay (n = 2) and study classification of broader autism phenotype (n = 2).
Fig. 1.
Classification tree results. The FYI items used in the model are specified at each branch of the tree, with the values they take for each side of the split. The number of children from each diagnostic group for each leaf is listed below the predicted classification. For example, of the seven children who scored “seldom” or “often” on Q39, 6 are from the ASD group and are classified correctly, and one is from the Non-ASD group and is misclassified. The 64 children who scored “never” on Q39 were further classified based on the results of Q24. Q question, ASD autism spectrum disorder, NA non-ASD
Discussion
The primary objective of this preliminary study was to investigate whether parents of HR-siblings who later develop ASD rate their children’s behavior in key diagnostic areas differently than in those with other developmental outcomes. We found that both clinicians and parents of HR-siblings later diagnosed with ASD rated their social functioning as more abnormal compare to those with other developmental outcomes and that there was a significant correlation between parent and clinician ratings of social functioning on both instruments. Among the Social-Communication Domain constructs, the construct of Imitation provided the greatest separation between ASD cases and groups with other outcomes as seen by effect sizes exceeding 1. This construct consisted of items probing for parental perception of imitation of motor actions, vocalizations, facial gestures and actions on objects exhibited by their children in a context of everyday activities. In contrast, as a group, the ASD cases did not differ with regard to the Sensory-Regulatory Domain or any of its constructs. These results parallel those based on clinicians’ ratings encapsulated in the Repetitive and Restricted Behaviors domain scores of the ADOS-T. The correlations between clinician ratings and parent report in this domain were negligible. Taken together, these findings suggest that at 12 months, both clinicians and parents rate HR-siblings who are later diagnosed with ASD as more atypical with regard to their social and communicative skills but not unusual sensory and repetitive behaviors.
Interestingly, the scale that achieved the greatest separation between ASD cases, particularly those HR-siblings who had other atypical outcomes was the Imitation scale. Does this suggest that at 12 months ASD-related vulnerability in HR-siblings is expressed chiefly through deficits in imitation? Experimental and observational studies suggest that ASD-specific deficits in high-risk siblings are present in other domains as well including limited eye contact, social smiling, and orienting, (Barbaro and Dissanayake 2013; Mitchell et al. 2006; Ozonoff et al. 2010; Rozga et al. 2011; Zwaigenbaum et al. 2005) and increased rates of atypical object exploration (Ozonoff et al. 2008a). Indeed, in the present study, ASD cases differed from those with other outcomes in ADOS-T Social-Communication algorithms even though items probing for imitation skills are not included into the calculation of the algorithm scores. Thus, the reasons for such a pattern of results might lie elsewhere. We would like to argue that some of the most diagnostic behaviors at this age might be difficult for parents to notice or judge as atypical in their infants. A study comparing concurrent parental report on the ADI-R (Rutter et al. 2003) and clinician ratings of social communicative skills and repetitive behaviors on the ADOS-G (Lord et al. 2000) in toddlers with ASD found that although parents and clinicians provided similar ratings of communicative behaviors such as gestures, parents systematically underreported abnormalities in eye contact, social smiling, and facial expressions (Chawarska et al. 2007). The discrepancy might be driven by parents having difficulties in making fine-grained discriminations, such as between a smile elicited by touching or anticipatory routine versus a purely affiliative behavior. In turn, imitation of sounds or actions can be reliably elicited in TD 12-month-old infants, and motor and vocal ‘imitation games’ constitute integral components of the playful routines between parents and their infants and are often structured in a trial-like manner (e.g., mom says ‘bah,’ baby follows, or dad waves, baby waves). Thus, missed opportunities for imitation are readily noticeable and reportable, and it is not surprising that the clearly worded questions regarding discrete aspects of social imitation on the FYI proved to be sensitive to deficits in social responsivity.
Imitation has been considered as one of the key areas of early impairments in ASD (McDuffie et al. 2007; Williams et al. 2004), with deficits in imitation distinguishing toddlers with ASD, developmental delay, and typical development as early as 20 months (Charman et al. 1997). A recent study, however, suggested that at 12 months such deficits might not differentiate between HR-infants who later develop ASD and those with other delays (Young et al. 2011). This study examined imitative skills in HR-infants using a structured assessment battery in a laboratory setting. At 12 months, infants later diagnosed with ASD showed imitation deficits compared to TD low-risk controls, but not compared to HR-infants with delays but without ASD. Our findings differ from these results, possibly due to differences in approach to the measurement of target behaviors. Parent report measures, such as the FYI, take advantage of parents’ ability to observe their children over longer time intervals in highly familiar contexts and during well-practiced routines (e.g., waving bye or blowing raspberries). It is plausible that the HR-ATYP infants have a greater capacity to take advantage of such scaffolding than the HR-ASD infants and thus present as more capable in these contexts, but display vulnerabilities when these supports are removed in a laboratory setting.
Analysis of the individual items of the FYI suggests that a combination of two items may be useful for distinguishing infants with ASD from the remaining HR-infants. While these results are preliminary and require replication on a larger, more diverse sample; they reveal specific features that may assist in screening at least a subset of HR-siblings. By parental report, approximately 38 % of the infants with ASD exhibited a decrease in communication or social play by 12 months; an additional 25 % of infants showed limited vocal imitation skills even though their parents did not report a developmental slow-down. Ninety-three percent of HR non-ASD cases were correctly classified as not affected. Important implications of this analysis include: (1) accuracy of identifying non-ASD cases at 12 months was high, with false positive rates not exceeding 7 %; (2) ASD outcome was predicted by different combinations of features at 12 months; and (3) the analysis failed to identify 37 % of ASD cases. Based on direct assessment, the missed cases did not exhibit clinical symptoms at 12 months. Although preliminary, taken together, these findings are promising as they point to the set of skills that parents can identify as deficient at 12 months and that may be useful for screening purposes.
Limitations
This study is limited by its relatively small sample size, high education level of the participating families, and lack of ethnic diversity. The results require replication in a larger and more diverse group of high-risk siblings and their parents. Given the potential influence that raising a child with ASD may have on reporting on behavioral features of a subsequent offspring, as well as the presence of atypical features in non-affected infants, results of this study may not generalize to screening in families with no familial history of ASD. Not all of the children completed 36 month evaluations; though this was true of only 1 child with ASD. This could potentially have resulted in some of the children with ASD not being identified as such in the study, though the rates of ASD in this sample are largely consistent with other reports (Ozonoff et al. 2011; Zwaigenbaum et al. 2012). The results of the CART analysis serve as an illustration of the utility of the analytic approach to identifying siblings at greatest risk of ASD; replication of these results in larger and more diverse samples will be necessary.
Conclusion
Results of this preliminary study show that parent report of the early emerging social communicative skills provides a promising avenue for identifying HR-siblings who are at the greatest risk of developing ASD as early as 12 months. Decrease in the level of social engagement and play as well as limited motor, vocal, and action imitation skills constitute important red flags among HR-siblings later diagnosed with ASD from their non-ASD counterparts. Even though not all siblings that eventually develop ASD may exhibit red flags at this age, identification of those who do is imperative given the importance of early intervention for ameliorating the effects of social disability on subsequent neural and behavioral development. Effective screeners for ASD in infancy need to accommodate heterogeneous developmental trajectories and need to capitalize on readily observable and quantifiable behaviors, within the context of complex home environments and interactions. These findings have important implications for designing developmentally sensitive screening instruments for both high and low-risk infants at 12 months.
Acknowledgments
We would also like to thank Celine Saulnier, Amanda Steiner, Karen Bearss, Amy Carney, Liz Simmons, and Megan Lyons for their contribution to the sample characterization as well as Kerry O’Loughlin and Jessica Garczarek for assistance in data collection. We would like to thank Grace Baranek, Steve Reznick, Lauren Brown, Linda Watson, and Elizabeth Crais at the University of North Carolina for their comments on the manuscript. We express our appreciation to the families and their children for their time and participation. This study was supported by the National Institute of Child Health and Human Development P01 HD003008, Project 1 (PI: K Chawarska), the National Institutes of Mental Health R01MH087554-01 (PI: K Chawarska), and Autism Speaks #7614 (PI: Campbell).
Footnotes
Conflict of interest Daniel Cambell, PhD, receives salary and stock options from Amgen Inc. All other authors declare no conflict of interest. The content of this study is solely the responsibility of the authors and does not necessarily represent the views of the National Institute of Child Health and Human Development, National Institutes of Mental Health, Yale Child Study Center, United States Air Force Medical Service or Department of Defense.
Ethical standards Informed consent was obtained from all participants prior to their participation. This study was approved by the University’s Institutional Review Board and was conducted in accordance with the ethical standards established in the Declaration of Helsinki and its later amendments.
Contributor Information
Justin Rowberry, Developmental and Behavioral Pediatrics, Mike O’Callaghan Federal Medical Center, 4700 Las Vegas Boulevard North, Building 1300, Nellis Air Force Base, NV 89191, USA justin.rowberry@us.af.mil.
Suzanne Macari, Child Study Center, Yale School of Medicine, New Haven, CT, USA.
Grace Chen, School of Education, Stanford University, Stanford, CA, USA.
Daniel Campbell, Amgen Inc., Thousand Oaks, CA, USA.
John M. Leventhal, Developmental and Behavioral Pediatrics, Yale School of Medicine, New Haven, CT, USA
Carol Weitzman, Developmental and Behavioral Pediatrics, Yale School of Medicine, New Haven, CT, USA.
Katarzyna Chawarska, Child Study Center, Yale School of Medicine, New Haven, CT, USA.
References
- American Psychiatric Association. Diagnostic and statistical manual of mental disorders. 5th ed. Arlington, VA: Author; 2013. [Google Scholar]
- Baranek GT, Watson LR, Crais E, Reznick S. First year inventory (FYI) 2.0. Chapel Hill: University of North Carolina at Chapel Hill; 2003. [Google Scholar]
- Barbaro J, Dissanayake C. Early markers of autism spectrum disorders in infants and toddlers prospectively identified in the social attention and communication study. Autism. 2013;17(1):64–86. doi: 10.1177/1362361312442597. [DOI] [PubMed] [Google Scholar]
- Breiman L, Friedman J, Olshen R, Stone C. Classification and regression trees. New York, NY: Wadsworth; 1984. [Google Scholar]
- Centers for Disease Control and Prevention. Prevalence of autism spectrum disorders–autism and developmental disabilities monitoring network, 14 sites, United States, 2008. Morbidity and Mortality Weekly Report. 2012;61(3):1–19. [PubMed] [Google Scholar]
- Charman T, Swettenham J, Baron-Cohen S, Cox A, Baird G, Drew A. Infants with autism: An investigation of empathy, pretend play, joint attention, and imitation. Developmental Psychology. 1997;33(5):781–789. doi: 10.1037//0012-1649.33.5.781. [DOI] [PubMed] [Google Scholar]
- Chawarska K, Klin A, Paul R, Macari S, Volkmar F. A prospective study of toddlers with ASD: Short-term diagnostic and cognitive outcomes. Journal of Child Psycholology and Psychiatry. 2009;50(10):1235–1245. doi: 10.1111/j.1469-7610.2009.02101.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chawarska K, Paul R, Klin A, Hannigen S, Dichtel LE, Volkmar F. Parental recognition of developmental problems in toddlers with autism spectrum disorders. Journal of Autism and Developmental Disorders. 2007;37(1):62–72. doi: 10.1007/s10803-006-0330-8. [DOI] [PubMed] [Google Scholar]
- Constantino JN, Davis SA, Todd RD, Schindler MK, Gross MM, Brophy SL, et al. Validation of a brief quantitative measure of autistic traits: Comparison of the social responsiveness scale with the autism diagnostic interview-revised. Journal of Autism and Developmental Disorders. 2003;33(4):427–433. doi: 10.1023/a:1025014929212. [DOI] [PubMed] [Google Scholar]
- Georgiades S, Szatmari P, Zwaigenbaum L, Bryson S, Brian J, Roberts W, et al. A prospective study of autistic-like traits in unaffected siblings of probands with autism spectrum disorder. Archives of General Psychiatry. 2012;3:1–7. doi: 10.1001/2013.jamapsychiatry.1. [DOI] [PubMed] [Google Scholar]
- Green J, Wan MW, Guiraud J, Holsgrove S, McNally J, Slonims V, et al. Intervention for infants at risk of developing autism: A case series. Journal of Autism and Developmental Disorders. 2013;43(11):2502–2514. doi: 10.1007/s10803-013-1797-8. [DOI] [PubMed] [Google Scholar]
- Grønborg TK, Schendel DE, Parner ET. Recurrence of autism spectrum disorders in full-and half-siblings and trends over time: A population-based cohort study. JAMA pediatrics. 2013;167(10):947–953. doi: 10.1001/jamapediatrics.2013.2259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- IBM SPSS Statistics Version 19. Release 19.0.0. 2010 [Google Scholar]
- Johnson CP, Myers SM. Identification and evaluation of children with autism spectrum disorders. Pediatrics. 2007;120(5):1183–1215. doi: 10.1542/peds.2007-2361. [DOI] [PubMed] [Google Scholar]
- Kanner L. Autistic disturbances of affective contact. Nervous child. 1943;2(3):217–250. [PubMed] [Google Scholar]
- Koegel LK, Singh AK, Koegel RL, Hollingsworth JR, Bradshaw J. Assessing and improving early social engagement in infants. Journal of Positive Behavior Interventions. 2014;16(2):69–80. doi: 10.1177/1098300713482977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Landa RJ, Holman KC, Garrett-Mayer E. Social and communication development in toddlers with early and later diagnosis of autism spectrum disorders. Archives of General Psychiatry. 2007;64(7):853–864. doi: 10.1001/archpsyc.64.7.853. [DOI] [PubMed] [Google Scholar]
- Lord C, Luyster R, Gotham K, Guthrie W, Risi S, Rutter M. Autism diagnostic observation schedule–toddler module manual. Los Angeles, CA: Western Psychological Services; 2012a. [Google Scholar]
- Lord C, Petkova E, Hus V, Gan W, Lu F, Martin DM, et al. A multisite study of the clinical diagnosis of different autism spectrum disorders. Archives of General Psychiatry. 2012b;69(3):306–313. doi: 10.1001/archgenpsychiatry.2011.148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lord C, Risi S, Lambrecht L, Cook EH, Jr, Leventhal BL, et al. The autism diagnostic observation schedule-generic: A standard measure of social and communication deficits associated with the spectrum of autism. Journal of Autism and Developmental Disorders. 2000;30(3):205–223. [PubMed] [Google Scholar]
- Macari SL, Campbell D, Gengoux GW, Saulnier CA, Klin AJ, Chawarska K. Predicting developmental status from 12 to 24 months in infants at risk for autism spectrum disorder: A preliminary report. Journal of Autism and Developmental Disorders. 2012;42(12):2636–2647. doi: 10.1007/s10803-012-1521-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McDuffie A, Turner L, Stone W, Yoder P, Wolery M, Ulman T. Developmental correlates of different types of motor imitation in young children with autism spectrum disorders. Journal of Autism and Developmental Disorders. 2007;37(3):401–412. doi: 10.1007/s10803-006-0175-1. [DOI] [PubMed] [Google Scholar]
- Messinger D, Young GS, Ozonoff S, Dobkins K, Carter A, Zwaigenbaum L, et al. Beyond autism: A baby siblings research consortium study of high-risk children at three years of age. Journal of the American Academy of Child and Adolescent Psychiatry. 2013;52(3):300–308. doi: 10.1016/j.jaac.2012.12.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mitchell S, Brian J, Zwaigenbaum L, Roberts W, Szatmari P, Smith I, et al. Early language and communication development of infants later diagnosed with autism spectrum disorder. Journal of Developmental and Behavioral Pediatrics. 2006;27(2):S69–S78. doi: 10.1097/00004703-200604002-00004. [DOI] [PubMed] [Google Scholar]
- Mullen E. Mullen scales of early learning. Circle Pines, MN: American Guidance Services; 1995. [Google Scholar]
- Ozonoff S, Heung K, Byrd R, Hansen R, Hertz-Picciotto I. The onset of autism: Patterns of symptom emergence in the first years of life. Autism Research. 2008a;1:320–328. doi: 10.1002/aur.53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ozonoff S, Iosif AM, Baguio F, Cook IC, Hill MM, Hutman T, et al. A prospective study of the emergence of early behavioral signs of autism. Journal of the American Academy of Child & Adolescent Psychiatry. 2010;49(3):256–266. [PMC free article] [PubMed] [Google Scholar]
- Ozonoff S, Macari S, Young GS, Goldring S, Thompson M, Rogers SJ. Atypical object exploration at 12 months of age is associated with autism in a prospective sample. Autism. 2008b;12(5):457–472. doi: 10.1177/1362361308096402. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ozonoff S, Young GS, Carter A, Messinger D, Yirmiya N, Zwaigenbaum L, et al. Recurrence risk for autism spectrum disorders: A baby siblings research consortium study. Pediatrics. 2011;128(3):e488–e495. doi: 10.1542/peds.2010-2825. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ozonoff S, Young GS, Steinfeld MB, Hill MM, Cook I, Hutman T, et al. How early do parent concerns predict later autism diagnosis? Journal of Developmental and Behavioral Pediatrics. 2009;30(5):367–375. doi: 10.1097/dbp.0b013e3181ba0fcf. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Paul R, Fuerst Y, Ramsay G, Chawarska K, Klin A. Out of the mouths of babes: Vocal production in infant siblings of children with ASD. Journal of Child Psychology and Psychiatry. 2011;52(5):588–598. doi: 10.1111/j.1469-7610.2010.02332.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- R Development Core Team. R: A language and environment for statistical computing. Vienna, Austria: R Foundation for Statistical Computing; 2011. [Google Scholar]
- Reznick JS, Baranek GT, Reavis S, Watson LR, Crais ER. A parent-report instrument for identifying one-year-olds at risk for an eventual diagnosis of autism: The first year inventory. Journal of Autism and Developmental Disorders. 2007;37(9):1691–1710. doi: 10.1007/s10803-006-0303-y. [DOI] [PubMed] [Google Scholar]
- Robins DL, Casagrande K, Barton M, Chen CMA, Dumont-Mathieu T, Fein D. Validation of the modified checklist for autism in toddlers, revised with follow-up (M-CHAT-R/F) Pediatrics. 2014;133(1):37–45. doi: 10.1542/peds.2013-1813. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Robins DL, Fein D, Barton ML, Green JA. The modified checklist for autism in toddlers: An initial study investigating the early detection of autism and pervasive developmental disorders. Journal of Autism and Developmental Disorders. 2001;31(2):131–144. doi: 10.1023/a:1010738829569. [DOI] [PubMed] [Google Scholar]
- Rozga A, Hutman T, Young G, Rogers S, Ozonoff S, Dapretto M, et al. Behavioral profiles of affected and unaffected siblings of children with autism: Contribution of measures of mother-infant interaction and nonverbal communication. Journal of Autism and Developmental Disorders. 2011;41(3):287–301. doi: 10.1007/s10803-010-1051-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rutter M, Le Couteur A, Lord C. Autism diagnostic interview-revised. Los Angeles, CA: Western Psychological Services; 2003. [Google Scholar]
- Siegel B. The pervasive developmental disorders screening test II (PDDST-II) San Antonio TX: Psychological Corporation; 2004. [Google Scholar]
- Siperstein R, Volkmar F. Brief report: Parental reporting of regression in children with pervasive developmental disorders. Journal of Autism and Developmental Disorders. 2004;34(6):731–734. doi: 10.1007/s10803-004-5294-y. [DOI] [PubMed] [Google Scholar]
- Steiner AM, Gengoux GW, Klin A, Chawarska K. Pivotal response treatment for infants at-risk for autism spectrum disorders: A pilot study. Journal of Autism and Developmental Disorders. 2013;43(1):91–102. doi: 10.1007/s10803-012-1542-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Turner-Brown LM, Baranek GT, Reznick JS, Watson LR, Crais ER, Watson LR, et al. The first year inventory: A longitudinal follow-up of 12-month-old to 3-year-old children. Autism. 2012;37(1):49–61. doi: 10.1177/1362361312439633. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Williams JH, Massaro DW, Peel NJ, Bosseler A, Suddendorf T. Visual-auditory integration during speech imitation in autism. Research in Developmental Disabilities. 2004;25(6):559–575. doi: 10.1016/j.ridd.2004.01.008. [DOI] [PubMed] [Google Scholar]
- Young GS, Rogers SJ, Hutman T, Rozga A, Sigman M, Ozonoff S. Imitation from 12 to 24 months in autism and typical development: A longitudinal Rasch analysis. Developmental Psychology. 2011;47(6):1565–1578. doi: 10.1037/a0025418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zwaigenbaum L, Bryson S, Rogers T, Roberts W, Brian J, Szatmari P. Behavioral manifestations of autism in the first year of life. International Journal of Developmental Neuroscience. 2005;23(2):143–152. doi: 10.1016/j.ijdevneu.2004.05.001. [DOI] [PubMed] [Google Scholar]
- Zwaigenbaum L, Bryson SE, Szatmari P, Brian J, Smith IM, Roberts W, et al. Sex differences in children with autism spectrum disorder identified within a high-risk infant cohort. Journal of Autism and Developmental Disorders. 2012;42(12):2585–2596. doi: 10.1007/s10803-012-1515-y. [DOI] [PubMed] [Google Scholar]