Abstract
Consuming plants for their presumed health benefits has occurred since early civilizations. Phytochemicals are found in various plants that are frequently included in the human diet and are generally thought to be safe for consumption because they are produced naturally. However, this is not always the case and in fact many natural compounds found in several commonly consumed plants are potential carcinogens or tumor promoters and should be avoided.
Introduction
Consuming plants for medicinal purposes or to take advantage of their presumed health benefits, including cancer prevention or treatment, has been a common practice since early civilizations. Phytochemicals are present in fruits, vegetables and many other plants and are not essential nutrients, such as vitamins or minerals, but many are commonly consumed or used as herbal remedies or dietary supplements for perceived health benefits. Most phytochemicals in the United States are not subject to regulation by the Food and Drug Administration (FDA) and their potential toxicity is understudied. Over the past few decades, the use of phytochemical dietary supplements and herbal remedies has increased exponentially and this has been particularly apparent in Western countries where supplement use was reported by 49% of the U.S. population (44% of males, 53% of females) (1). In particular, many cancer patients use supplements in conjunction with traditional cancer treatment, but do not necessarily inform their physicians of their use (2–5). In spite of this popularity, only limited data are available regarding the safety and efficacy of most individual phytochemicals in preventing or treating chronic diseases such as cancer (6). Epidemiological studies indicate that many people who use dietary supplements assume that these compounds are safe and might be a more natural alternative to conventional medication (7). Indeed, some reports have suggested beneficial effects of phytochemicals, such as EGCG (epigallocatechin gallate) (8, 9) or resveratrol (10) in the prevention or treatment of cancer. Numerous dietary epidemiologic observations and animal studies for many dietary factors suggest possible protection against various cancers; but in general, these findings have not yet been validated in randomized human clinical trials. In fact, results of most dietary intervention clinical trials have been very disappointing. Dietary-based cancer prevention suffered a major setback in the mid 1990s in a cancer prevention trial in which βeta carotene actually was associated with an increased risk of lung cancer in smokers (11, 12). Results from the Alpha-Tocopherol, Beta-Carotene Cancer Prevention Study (ATBC Study) in Finland indicated that alpha-tocopherol had no effect on lung cancer incidence, but was associated with decreased prostate cancer risk (11). Unexpectedly, a higher incidence of lung cancer was also observed in men receiving βeta carotene compared to those who did not. Concurrently, in the U.S., the Beta Carotene and Retinol Efficacy Trial (CARET) was initiated and involved smokers, former smokers, and workers exposed to asbestos and its purpose was to examine the effect of daily βeta carotene and retinol (vitamin A) supplementation on the incidence of lung cancer (13). Similar to the ATBC, the βeta carotene group had a higher risk of lung cancer compared with the placebo group. A later report indicated that plant foods have an important preventive influence in a population at high risk for lung cancer, but the effect is diminished with βeta carotene supplements (14).
In contrast with earlier research suggesting that dietary supplementation with selenium and vitamin E may lower the risk of prostate cancer (11, 15–18), initial results from the largest-ever prostate cancer prevention study, the Selenium and Vitamin E Cancer Prevention Trial (SELECT), showed that these substances did not help prevent prostate cancer (19). Clinical trials with St. John’s Wort and garlic have generally shown no effect or an anti-cancer effect and these plants are not considered to be human carcinogens. Whether ginkgo is a carcinogen has been somewhat controversial. An NIH Toxicology report published in March of 2013 summarized the results of a 2-year rodent study on the toxicology of ginkgo, but the study was criticized because of the huge doses that were administered.
The Internet is full of ‘pop science’ touting the advantageous properties of many phytochemicals that have not been scientifically proven to be safe or effective. This review article highlights some of the phytochemicals that have been reported to act as carcinogens and appear to be most commonly consumed in food or used in herbal remedies. The review is divided into two parts: potential dietary carcinogens and carcinogens in herbal remedies. For example, capsaicin, phytoestrogens, bracken fern, and safrole are commonly consumed in many parts of the world and are linked to cancer development. Amygdalin, aristolochic acids, and some of the pyrrolizidine alkaloids are not regulated and are commonly used in herbal remedies. The emphasis of this review is to focus on food compounds or herbal remedies that have been shown or suspected to be human carcinogens.
Potential dietary carcinogens
A number of phytochemicals with potential carcinogenic effects are found naturally in many foods and seasonings that have been consumed for centuries by humans as food, supplements to maintain optimal health, or used as treatments for disease (Table 1).
Table 1.
| Dietary Phytochemical/source |
Chemical Name | CAS Number |
Chemical Formula |
Chemical Structure |
|---|---|---|---|---|
| Capsaisin (Chili Pepper) | 8-methyl-N-vanillyl-trans-6-nonenamide | 404-86-4 | C18H27NO3 |
|
| Cycasin | (methyl-ONN-azoxy)methyl b-D-glucopyranoside | 14901-08-7 | C8H16N2O7 |
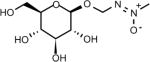
|
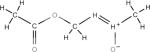
| Methylazoxymethanol –MAM (Cycas Seed) | Methyl-ONN-azoxymethanol | 590-96-5 | C2H6N2O2 |
|
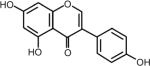
| Phytoestrogens Genistein (soybeans, fava beans, red clover) | 4′,5,7- trihydroxyisoflavone | 446-72-0 | C15H10O5 |
|
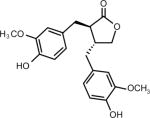
| Phytoestrogens (Lignans matairesinol) (Sesame, flax seed) | (3R,4R)-3,4-bis[(4-hydroxy-3-methoxyphenyl)methyl] oxolan-2-one | 580-72-3 | C20H22O6 |
|
| Ptaquiloside (Bracken Fern) | (2′R)-7′a β-(β-D-glucopyranosyloxy)-1′,3′aβ,4′7′a-tetrahydro-4′α-hydroxy-2′β,4′,6′-trimethylspiro[cyclopropane-1,5′[5H]inden]-3′(2′H)-one | 87625-62-5 | C20H30O8 |
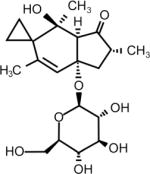
|
| Safrole (Sassafrass Plant) | 5-(2-propenyl)-1,3-benzodioxole | 94-59-7 | C10H10O2 |
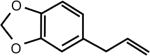
|
Capsaicin
Perhaps one of the most controversial, in spite of being well-studied, phytochemicals is capsaicin. Capsaicin is the principal pungent component in the fruits of plants from the genus Capsicum, which are members of the nightshade family, Solanaceae. These plants are native to the Americas and have been cultivated as part of the diet since at least 7500 B.C. (20). Capsaicin gives chili peppers their intensity or ‘hotness’ when ingested or applied topically to the skin, and is the primary ingredient in pepper spray often used in law enforcement. Even though widely consumed, capsaicin has a long and checkered history as to whether its consumption or topical use is carcinogenic. Conflicting epidemiological and basic research studies suggest that capsaicin could have a role in either preventing cancer or causing cancer. Hundreds of basic research studies show that capsaicin suppresses growth of numerous types of cancer cell, suggesting that it has chemopreventive activities and these studies have been well reviewed and will not be detailed herein (21–23). Animal studies have produced ambiguous results, with some studies showing a carcinogenic effect and some reporting a protective effect. In contrast, epidemiological studies seem to indicate that consumption of hot peppers, which contain variable levels of capsaicin, might be associated with an increased risk of cancer, especially of the gallbladder (24) or stomach (25). However, most of these studies have severe limitations and are descriptive, correlative studies with speculative conclusions. Thus a complete consensus as to whether the primary effect of capsaicin is cancer prevention or promotion has not yet been reached.
Capsaicin is unique among naturally occurring irritant compounds because the initial neuronal excitation it provokes is followed by a long-lasting refractory period during which time the previously excited neurons are no longer responsive to a broad range of apparently unrelated stimuli (26). This desensitization has been exploited for its therapeutic potential. Capsaicin-containing creams have been in clinical use for many years to relieve painful conditions such as rheumatoid arthritis, osteoarthritis and diabetic neuropathy (27). However, even in this area, evidence for the effectiveness or safety of capsaicin use in pain relief is controversial. We recently reported that capsaicin has a co-carcinogenic effect on 12-O-tetradecanoylphorbol-13-acetate (TPA)-promoted skin carcinogenesis in vivo and is mediated not only through the transient receptor potential vanilloid subfamily member 1 (TRPV1), but also through the tyrosine kinase epidermal growth factor receptor (EGFR) (28) and COX2 (29). These results suggest caution in the prolonged topical application of capsaicin, especially in the presence of a tumor promoter, which would include exposure to solar ultraviolet irradiation.
Cycasin
Cycasin and its metabolite, methylazoxymethanol (MAM), are typically extracted from the seeds and roots of cycad plants (30), which are cone-bearing plants common to the tropics and subtropics. The seeds and roots of cycads were used as food long before their toxicity was recognized and have also been a source of medicine for many people who live in proximity to these plants. However, the plants are highly poisonous and toxicity from seed ingestion is mainly caused by misuse as an edible food, as an agent to improve health, for cancer prevention, cosmetic use, and to treat gastrointestinal discomfort. MAM is a genotoxic metabolite that targets cellular processes involved in neurodegeneration and cancer development (31). This is based on the observation that a single systemic dose of MAM was shown to induce DNA damage in the brains of C57BL6 mice, which was linked with changes in gene expression associated with cancer (31). MAM was shown to induce a variety of tumors, primarily liver and renal cell carcinomas in nonhuman primates in a 32-year study conducted by the National Cancer Institute (NCI) in the U.S. (32) confirming the results of earlier studies (33). When given subcutaneously or intraperitoneally (IP) to newborn rats, tumors were detected in more than 80% of rats of both sexes and kidney tumors were the most common (34). However, whether cycasin acts directly as a carcinogen in humans is not yet clear and the seeds are still consumed in certain areas of the world.
Phytoestrogens
The phytoestrogens are a group of phytochemicals purported to have both beneficial and, under certain conditions, less than desirable effects. Phytoestrogens gained their name because of their ability bind to estrogen receptors and stimulate receptor activity. Genistein is a polyphenol or isoflavone phytoestrogen that is found in soybeans and other plants, including red clover and fava beans. Lignans are another major class of phytoestrogens and matairesinol was one of the first plant lignan phytoestrogens identified in food and is found in numerous foods such as oil seeds, whole grains, vegetables, and fruits (35). Flax seed and sesame seed contain higher levels of lignans than most other foods. Demographic and anthropometric characteristics, a medical history, and 7-day records of diet were collected prospectively from participants (aged 40–79 years) in the European Prospective Investigation into Cancer and Nutrition-Norfolk (EPIC-Norfolk) and results indicated that dietary phytoestrogens might contribute to the risk of colorectal cancer among women and prostate cancer among men (36). On the other hand, both genistein and matairesinol have been shown to exert preventive effects against hormone-associated tumors, such as breast (37–39) and prostate (36) cancers in humans. However, many phytoestrogens seem to be able to negate the effectiveness of aromatase inhibitors, which are designed to reduce the levels of estrogens that can promote tumor growth in some types of breast cancer (40, 41). Local estrogen production is catalyzed by aromatase, which is regulated differentially in healthy and tumorigenic breast tissue. Soy supplements that have been used to alleviate menopausal symptoms can induce MCF-7 breast cancer cell growth by increasing breast cancer-associated aromatase expression and activity (40). Notably, genistein was reported to block the inhibitory activity of the aromatase inhibitors fadrozole (40) and letrozole (41) against MCF-7 breast cancer cell growth in culture and in a xenograft model, respectively. Thus, women who are taking an aromatase inhibitor might need to be cautioned against consuming soy products. Overall, the results supporting a cancer preventive role for phytoestrogens are not yet conclusive and whether lignans, such as matairesinol, can interfere with aromatase inhibitors in humans has not yet been established.
Ptaquiloside (Bracken fern)
A very commonly consumed but extremely toxic plant is the bracken fern. All parts of bracken fern, including rootstocks, fresh or dry leaves, fiddleheads and spores, contain toxic compounds that are poisonous to livestock and humans. Bracken contains a thiaminase, which splits the essential vitamin thiamine (B1) into its two inactive components, pyrimidine and thiazole, causing thiamine deficiency (42). Bracken fronds are still eaten as a vegetable in many parts of the world and particularly in Korea, Japan and parts of China. Evidence linking this plant to human health issues has been strengthened by epidemiological associations (43) and significant advances in understanding the chemical reactions between one of its most notorious components, ptaquiloside, and DNA (44).
Ptaquiloside is the primary carcinogenic component in bracken fern and when activated to a dienone (APT or activated ptaquiloside), forms DNA adducts and eventually leads to tumor development. It is the only known plant carcinogen that causes natural outbreaks of bladder and/or intestinal cancer in livestock (45). Rats given APT developed ischemic tubular necrosis in the kidney (46) and adenocarcinomas of the mammary glands (47). Squamous cell carcinomas of the upper digestive tract of cattle have been associated with chronic bracken fern consumption (48) and milk from cows consuming bracken fern has been suggested as a vehicle for this carcinogen in humans (49). Others have shown that consumption of ptaquiloside-contaminated milk may contribute to human gastric cancer (43). However, the greatest risk to humans appears to be the direct consumption of the fern itself, a practice that continues in various countries throughout the world and is indeed promoted on the Internet with recipes for preparing bracken fiddleheads (http://honest-food.net/2011/06/24/bracken-fern-food-or-poison/). Bracken fern was shown to induce DNA damage and apoptotic death of normal human submandibular gland (HSG) cells and oral epithelium cells (OSCC-3) (50). Other studies reveal that the mere presence of bracken fern and its spores represents a greater risk of dying of gastric adenocarcinoma for people who live more than 20 years in such areas or are exposed in childhood (51).
Safrole
Safrole (also known as shikimol) is found in a variety of spices, including cinnamon, nutmeg, black pepper and herbs such as basil. However, it is typically extracted from the root-bark or the fruit of sassafras plants as sassafras oil and has been widely used as a natural or synthetic flavoring agent. In the U.S, it was frequently used as a food additive in root beer and sassafras tea. Safrole was first shown to be a liver carcinogen in rats (52). In humans, epidemiological evidence indicates that individuals who chew betel nut or areca quid, which contains high levels of safrole, have an increased incidence of oral cancer (53), esophageal cancer (54) and hepatocellular carcinoma (55). This observation appears to be related to the formation of safrole-DNA adducts (53, 54), although some areca nut preparations include tobacco and caustic lime, which could also contribute to increased cancer risk.
Safrole is also associated with oral submucous fibrosis, a precancerous condition (56). However, the actual carcinogenicity of safrole in humans has been questioned because even though its tumorigenic metabolites, 1′-hydroxysafrole and 3′-hydroxyisosafrole, were observed in the urine of rats, they were not found in human urine (57). Notably, not all chemicals that act as carcinogens in rats or mice act as carcinogens in humans or vice versa. In addition, some work has shown that safrole causes marked changes in cellular calcium levels resulting in an inhibition of cancer cell growth. In particular, safrole induced death of human oral squamous cell carcinoma HSC-3 cells in culture and in mice by causing increases in cytosolic calcium levels (58). Similar effects on calcium levels and cell death were induced by safrole in MG63 human osteosarcoma cells (59) and PC3 prostate cancer cells (60). Safrole reportedly caused cell death by activating caspases in human tongue squamous carcinoma SCC-4 cells (61) and in A549 human lung cancer cells (62). Furthermore, safrole also induced apoptosis of human promyelocytic leukemia (HL-60) cells that was associated with increased production of reactive oxygen species (ROS) and Ca2+ levels and enhanced levels of the pro-apoptotic protein BAX and reduced levels of the anti-apoptotic protein BCL-2 (63). Safrole was also reported to alter immune modulation of T cells, B cells and macrophages in leukemic BALB/c mice and inhibited the growth of leukemia WEHI-3 cells in vivo (64). In contrast, increased calcium levels stimulated by safrole appeared to enhance human oral cancer OC2 cell growth (65). Thus, whether safrole is a true carcinogen in humans is still debatable.
Nevertheless, safrole has been banned by the FDA in the U.S. and was also banned from use in soap and perfumes by the International Fragrance Association. In spite of these bans, safrole can still be purchased on the Internet as a primary ingredient in sassafras oil or brown camphor oil and instructions as to its synthesis from methylenedioxy compounds are also readily available. In addition, safrole is a precursor in the synthesis of the insecticide synergist piperonyl butoxide, which when added to insecticide mixtures, such as pyrethrin, pyrethroid, and carbamate insecticides, increases their potency (66–68). Many common insecticides and pesticides for home and garden use, mosquito and termite control, veterinary use, and human clothing and bedding use contain piperonyl butoxide, which is classified as a possible human carcinogen by the EPA (69). Safrole is also a primary ingredient in the illegal recreational drug MDMA or ‘Ecstasy’. Thus, safrole still possesses a potential threat to human health.
Carcinogens in herbal remedies
Herbal medicines and remedies are widely available from a variety of sources and are virtually unregulated and many are contaminated with carcinogens (Table 2).
Table 2.
| Herbal Phytochemical/source |
Chemical Name | CAS Number |
Chemical Formula |
Chemical Structure |
|---|---|---|---|---|
| Amygdalin Seed (Cherry seed) | [(6-O-b-D-glucopyranosyl-b-D-glucopyranosyl) oxy] (phenyl) acetonitrile | 29883-15-6 | C20H27NO11 |
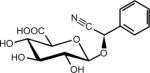
|
| Aristolochic Acids (Wild ginger Mu Tong) | 8-methoxy-3,4-methylenedioxy-10-nitrophenanthrene-1-carboxylicacid | 313-67-7 | C17H11NO7 |
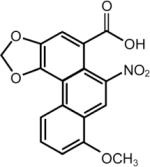
|
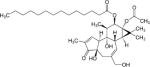
| Phorbol ester (Croton Plant) | Phorbol-12-myristate-13-acetate (12-O-tetradecanoylphorbol-13-acetate) | 16561-29-8 | C36H56O8 |
|
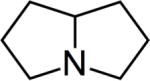
| Pyrrolizidine alkaloids (Coltsfoot Flower Comfrey Leaf Ragwort Riddell) | 2,3,5,6,7,8-Hexahydro-1H-pyrrolizine | 643-20-9 | C7H13 |
|
Amygdalin
Amygdalin is a glycoside initially isolated from the seeds of the tree Prunus dulcis, also known as bitter almonds. Several other related species in the genus of Prunus that contain amygdalin, include apricot and black cherry seeds (70). Amygdalin has been confused with laevomandelonitrile, also referred to as laetrile. Indeed, both amygdalin and laetrile have been promoted as vitamin B17, a cure for cancer, but are not actually a vitamin (71). Even though they are often referred to in the literature as the same chemical, amygdalin and laetrile are not the same chemical compound (72). Neither appear to be carcinogens but are widely touted as alternative cancer treatments that are claimed to be effective by many alternative therapists. However, both are totally ineffective and potentially toxic (73) as a possible cause of cyanide poisoning (74). Systematic reviews by the Cochrane Collaboration concluded that no sound data from any controlled clinical trial supports the claim that laetrile/amygdalin have beneficial effects for cancer patients (75, 76). The FDA prohibited the interstate shipment of amygdalin and laetrile in 1977. However, at least 27 U.S. states legalized the use of amygdalin within those states (77) and it is readily available for purchase on the Internet as an alternative cancer treatment.
Aristolochic acids
Aristolochic acids are a family of compounds that have been used since ancient times in traditional herbal medicines in many parts of the world (78). These compounds have been reported to have antibacterial, antiviral, antifungal and anticancer effects (79). Other traditional uses include treatment for snakebite, scorpion stings, fever, infection, diarrhea and inflammation (80). Several studies in animals and humans have confirmed that exposure to aristolochic acid is associated with a high incidence of cancers associated with the kidney and urinary tract (81). Aristolochic acid has been used as an agent in weight loss plans, but was banned by the FDA in 2001 after a report that the use of the Chinese herb, Aristolochia fangchi, was associated with end-stage kidney failure and urinary system cancer in patients enrolled in a weight loss study in Belgian (82). However, aristolochic acids have been detected recently in some Chinese medicinal herbs and herbal products (83). It is found in wild ginger, which is distinct from the commercial species that is acquired from Zingiber officinale used in cooking today. Even though extensive warnings of the dangers of aristolochic acids have been published (84), this compound is still widely available for purchase on the Internet. In particular, consumption of aristolochic acid-containing Chinese herbal products, like Mu Tong, which is associated with an increased risk of cancer of the urinary tract in humans, can still be purchased on the Internet (85) as an antibiotic and to improve cardiac function.
Phorbol esters
Phorbol esters are the tetracyclic diterpenoids generally known for their potent tumour promoting activity. Phorbol 12-myristate 13-acetate (also known as 12-O-Tetradecanoylphorbol-13-acetate or TPA) is derived from the oil of the seeds of the Croton plant. This shrub, which is native to Southeast Asia, has been used by herbalists and homeopaths for years. The plant causes a skin rash similar to poison ivy, which led scientists to isolate the compounds that were biologically active. The phorbol esters mimic the action of diacylglycerol (DAG), an activator of protein kinase C, which regulates different signal transduction pathways and other cellular metabolic activities (86). The biological activities of the phorbol esters are highly structure specific, with the ester groups of these compounds recognized as being essential for their undesirable activities. Although TPA is used as a tumor promoter in many mouse cancer models, it has also been used to treat cancers. In particular, the oils from the seeds of the plant have been used as an effective cancer therapeutic agent in myelocytic leukemia patients, and have been indicated as a potential colorectal cancer therapeutic (87, 88). TPA has also been shown to increase white blood cell and neutrophil counts in patients with solid tumours (89). TPA also inhibits thyroid cancer cell proliferation and migration (90) and prostate cancer cell growth in combination with paclitaxel (91). However, it is also a potent tumour promoter in skin (92). Thus the evidence indicates that, if applied appropriately, TPA might be utilized effectively for treating leukemia and lymphomas even though it is a well-known tumour promoter in skin.
Pyrrolizidine Alkaloids
Pyrrolizidine alkaloids (PAs), named for the inclusion of a pyrrolizidine nucleus–a pair of linked pyrrole rings, are present in teas and are probably the most commonly used herbs today. PA-containing plants are widespread in the world and are likely the most common poisonous plants affecting livestock, wildlife and humans. PAs are produced by plants as a defense mechanism against insect herbivores and more than 660 PAs have been identified in over 6,000 plants, including 3% of the world’s flowering plants (93), and most of them are toxic (94). As a result, honey (95, 96), milk (97, 98), grains and eggs (99, 100) have been found to contain PAs. However, in contrast to the regulations that exist for herbs and medicines, no international regulation of PAs in food currently exist (101).
PAs reportedly cause liver cancer (102) in rodent and primate animal models. These compounds require metabolic activation to generate pyrrolic metabolites (dehydro-PAs) that bind cellular protein and DNA, leading to genotoxicity and cancer. In particular, dehyro-PAs are the active metabolites responsible for skin cancer formation mediated through the generation of reactive oxygen species to initiate lipid peroxidation (103).
One of the most popular PAs is comfrey (Symphytum officinale), a well-known medicinal perennial herb, which is characterized as having been “one of the most popular herbal teas in the world (104).” Comfrey has been consumed for over 2000 years and contains PAs that are capable of inducing tumour development (105). Notably, the differentially expressed genes in livers from rats that have been fed comfrey compared to control livers are involved in metabolism, injury of endothelial cells, and liver abnormalities, including liver fibrosis and cancer development (106). Comfrey is readily available for purchase on the Internet and is sold as a topical application that can heal wounds and abraded skin, and for the treatment of broken bones, torn cartilage, tendon damage, lung congestion and ulceration of the gastrointestinal tract (www.takeherb.com/search/search.php?keywords=comfrey). In 2001, the FDA issued a warning against the consumption of herbal products containing comfrey.
Riddelliine is isolated from plants grown in the western United States and is a prototype of genotoxic PAs. Riddelliine can alter the expression of genes involved in cancer, cell death, tissue development, cellular movement, tissue morphology, cell-to-cell signaling and interaction, and cellular growth and proliferation (107). Riddelliine was found to be highly mutagenic in rat liver endothelial cells, which might be partially responsible for the tumourigenic specificity of this agent (108). Notably, the carcinogenesis-related gene expression patterns resulting from treatments with comfrey or riddelliine are very similar (109). However, most reports agree that riddelliine is toxic, but the safety and use of comfrey is still controversial.
Tussilago farfara, commonly known as coltsfoot, is a perennial, herbaceous plant that invades areas throughout much of the eastern United States. It has been used medicinally as a cough suppressant, but also contains tumourigenic PAs (110). Senecionine and senkirkine, present in coltsfoot, have the highest mutagenetic activity of any known PA (111). In spite of the dangers and warning, coltsfoot is also available for purchase on the Internet as a dietary supplement and as an herbal remedy for respiratory ailments (www.takeherb.com/search/search.php?keywords=coltsfoot).
Conclusion
Phytochemicals are found naturally in many plants and their consumption in fruits and vegetables is generally believed to provide beneficial health effects. Evidence from laboratory and epidemiology studies suggest that phytochemicals may reduce the risk of cancer, possibly due to antioxidant and anti-inflammatory effects. On the other hand, consumption of certain phytochemicals may act as carcinogens or tumor promoters. The Internet offers an enormous marketplace for such products. Clinically relevant adverse reactions to unconventional remedies purchased through the Internet have been reported (112, 113). Importantly, even herbal products that pose unacceptable health risks to consumers, including remedies containing the carcinogenic aristolochic acids (82) can be readily found on the Internet. A U.S. study identified 19 products containing aristolochic acids and 95 products suspected to contain these acids for sale on the Internet, 2 years after the FDA had issued warnings and an import alert regarding the dangers of such products (114). Furthermore, the quality and accurateness of herbal information on the Internet is variable and poor (115–117). Consumers need to be aware that dietary supplements containing phytochemicals or other compounds are virtually unregulated and manufacturers do not have to show evidence of safety or health benefits before the product is marketed.
Acknowledgments
This work was supported by The Hormel Foundation and NIH grants CA077646, CA111536, CA120388, ES016548, and R37CA081064.
References cited
- 1.Bailey RL, Gahche JJ, Lentino CV, Dwyer JT, Engel JS, Thomas PR, et al. Dietary supplement use in the United States, 2003–2006. The Journal of nutrition. 2011;141:261–6. doi: 10.3945/jn.110.133025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Correa-Velez I, Clavarino A, Eastwood H. Surviving, relieving, repairing, and boosting up: reasons for using complementary/alternative medicine among patients with advanced cancer: a thematic analysis. J Palliat Med. 2005;8:953–61. doi: 10.1089/jpm.2005.8.953. [DOI] [PubMed] [Google Scholar]
- 3.Fratamico PM, Wasilenko JL, Garman B, Demarco DR, Varkey S, Jensen M, et al. Evaluation of a multiplex real-time PCR method for detecting shiga toxin-producing Escherichia coli in beef and comparison to the U.S. Department of Agriculture Food Safety and Inspection Service Microbiology laboratory guidebook method. J Food Prot. 2014;77:180–8. doi: 10.4315/0362-028X.JFP-13-248. [DOI] [PubMed] [Google Scholar]
- 4.Funk J, Stoeber H, Hauser E, Schmidt H. Correction: molecular analysis of subtilase cytotoxin genes of food-borne Shiga toxin-producing Escherichia coli reveals a new allelic subAB variant. BMC Microbiol. 2014;14:32. doi: 10.1186/1471-2180-13-230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Gaige S, Djelloul M, Tardivel C, Airault C, Felix B, Jean A, et al. Modification of energy balance induced by the food contaminant T-2 toxin: a multimodal gut-to-brain connection. Brain Behav Immun. 2014;37:54–72. doi: 10.1016/j.bbi.2013.12.008. [DOI] [PubMed] [Google Scholar]
- 6.Yeung KS, Gubili J, Cassileth B. Evidence-based botanical research: applications and challenges. Hematol Oncol Clin North Am. 2008;22:661–70. viii. doi: 10.1016/j.hoc.2008.04.007. [DOI] [PubMed] [Google Scholar]
- 7.Shahrokh LE, Lukaszuk JM, Prawitz AD. Elderly herbal supplement users less satisfied with medical care than nonusers. J Am Diet Assoc. 2005;105:1138–40. doi: 10.1016/j.jada.2005.04.002. [DOI] [PubMed] [Google Scholar]
- 8.Stanker LH, Scotcher MC, Cheng L, Ching K, McGarvey J, Hodge D, et al. A monoclonal antibody based capture ELISA for botulinum neurotoxin serotype B: toxin detection in food. Toxins (Basel) 2013;5:2212–26. doi: 10.3390/toxins5112212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Funk J, Stoeber H, Hauser E, Schmidt H. Molecular analysis of subtilase cytotoxin genes of food-borne Shiga toxin-producing Escherichia coli reveals a new allelic subAB variant. BMC Microbiol. 2013;13:230. doi: 10.1186/1471-2180-13-230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Lorenz SC, Son I, Maounounen-Laasri A, Lin A, Fischer M, Kase JA. Prevalence of hemolysin genes and comparison of ehxA subtype patterns in Shiga toxin-producing Escherichia coli (STEC) and non-STEC strains from clinical, food, and animal sources. Appl Environ Microbiol. 2013;79:6301–11. doi: 10.1128/AEM.02200-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.The effect of vitamin E beta carotene on the incidence of lung cancer and other cancers in male smokers. The Alpha-Tocopherol, Beta Carotene Cancer Prevention Study Group. N Engl J Med. 1994;330:1029–35. doi: 10.1056/NEJM199404143301501. [DOI] [PubMed] [Google Scholar]
- 12.Omenn GS, Goodman GE, Thornquist MD, Balmes J, Cullen MR, Glass A, et al. Effects of a combination of beta carotene and vitamin A on lung cancer and cardiovascular disease. N Engl J Med. 1996;334:1150–5. doi: 10.1056/NEJM199605023341802. [DOI] [PubMed] [Google Scholar]
- 13.Omenn GS, Goodman G, Thornquist M, Grizzle J, Rosenstock L, Barnhart S, et al. The beta-carotene and retinol efficacy trial (CARET) for chemoprevention of lung cancer in high risk populations: smokers and asbestos-exposed workers. Cancer research. 1994;54:2038s–43s. [PubMed] [Google Scholar]
- 14.Neuhouser ML, Patterson RE, Thornquist MD, Omenn GS, King IB, Goodman GE. Fruits and vegetables are associated with lower lung cancer risk only in the placebo arm of the beta-carotene and retinol efficacy trial (CARET) Cancer Epidemiol Biomarkers Prev. 2003;12:350–8. [PubMed] [Google Scholar]
- 15.Virtamo J, Pietinen P, Huttunen JK, Korhonen P, Malila N, Virtanen MJ, et al. Incidence of cancer and mortality following alpha-tocopherol and beta-carotene supplementation: a postintervention follow-up. Jama. 2003;290:476–85. doi: 10.1001/jama.290.4.476. [DOI] [PubMed] [Google Scholar]
- 16.Taylor PR, Albanes D. Selenium, vitamin E, and prostate cancer–ready for prime time? Journal of the National Cancer Institute. 1998;90:1184–5. doi: 10.1093/jnci/90.16.1184. [DOI] [PubMed] [Google Scholar]
- 17.Brawley OW, Parnes H. Prostate cancer prevention trials in the USA. Eur J Cancer. 2000;36:1312–5. doi: 10.1016/s0959-8049(00)00105-2. [DOI] [PubMed] [Google Scholar]
- 18.Kumar NB, Besterman-Dahan K. Nutrients in the Chemoprevention of Prostate Cancer: Current and Future Prospects. Cancer Control. 1999;6:580–6. doi: 10.1177/107327489900600604. [DOI] [PubMed] [Google Scholar]
- 19.Lippman SM, Klein EA, Goodman PJ, Lucia MS, Thompson IM, Ford LG, et al. Effect of selenium and vitamin E on risk of prostate cancer and other cancers: the Selenium and Vitamin E Cancer Prevention Trial (SELECT) Jama. 2009;301:39–51. doi: 10.1001/jama.2008.864. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Perry L, Dickau R, Zarrillo S, Holst I, Pearsall DM, Piperno DR, et al. Starch fossils and the domestication and dispersal of chili peppers (Capsicum spp. L.) in the Americas. Science. 2007;315:986–8. doi: 10.1126/science.1136914. [DOI] [PubMed] [Google Scholar]
- 21.Bode AM, Dong Z. The two faces of capsaicin. Cancer Res. 2011;71:2809–14. doi: 10.1158/0008-5472.CAN-10-3756. [DOI] [PubMed] [Google Scholar]
- 22.Ojo OE, Ajuwape AT, Otesile EB, Owoade AA, Oyekunle MA, Adetosoye AI. Potentially zoonotic shiga toxin-producing Escherichia coli serogroups in the faeces and meat of food-producing animals in Ibadan, Nigeria. Int J Food Microbiol. 2010;142:214–21. doi: 10.1016/j.ijfoodmicro.2010.06.030. [DOI] [PubMed] [Google Scholar]
- 23.Immonen E, Kummu M, Petsalo A, Pihlaja T, Mathiesen L, Nielsen JK, et al. Toxicokinetics of the food-toxin IQ in human placental perfusion is not affected by ABCG2 or xenobiotic metabolism. Placenta. 2010;31:641–8. doi: 10.1016/j.placenta.2010.05.002. [DOI] [PubMed] [Google Scholar]
- 24.Serra I, Yamamoto M, Calvo A, Cavada G, Baez S, Endoh K, et al. Association of chili pepper consumption, low socioeconomic status and longstanding gallstones with gallbladder cancer in a Chilean population. Int J Cancer. 2002;102:407–11. doi: 10.1002/ijc.10716. [DOI] [PubMed] [Google Scholar]
- 25.Lopez-Carrillo L, Hernandez Avila M, Dubrow R. Chili pepper consumption and gastric cancer in Mexico: a case-control study. Am J Epidemiol. 1994;139:263–71. doi: 10.1093/oxfordjournals.aje.a116993. [DOI] [PubMed] [Google Scholar]
- 26.Szallasi A, Cortright DN, Blum CA, Eid SR. The vanilloid receptor TRPV1: 10 years from channel cloning to antagonist proof-of-concept. Nat Rev Drug Discov. 2007;6:357–72. doi: 10.1038/nrd2280. [DOI] [PubMed] [Google Scholar]
- 27.Hautkappe M, Roizen MF, Toledano A, Roth S, Jeffries JA, Ostermeier AM. Review of the effectiveness of capsaicin for painful cutaneous disorders and neural dysfunction. Clin J Pain. 1998;14:97–106. doi: 10.1097/00002508-199806000-00003. [DOI] [PubMed] [Google Scholar]
- 28.Bode AM, Cho YY, Zheng D, Zhu F, Ericson ME, Ma WY, et al. Transient receptor potential type vanilloid 1 suppresses skin carcinogenesis. Cancer Res. 2009;69:905–13. doi: 10.1158/0008-5472.CAN-08-3263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Hwang MK, Bode AM, Byun S, Song NR, Lee HJ, Lee KW, et al. Cocarcinogenic effect of capsaicin involves activation of EGFR signaling but not TRPV1. Cancer Res. 2010;70:6859–69. doi: 10.1158/0008-5472.CAN-09-4393. [DOI] [PubMed] [Google Scholar]
- 30.Laqueur GL, Spatz M. Toxicology of cycasin. Cancer Res. 1968;28:2262–7. [PubMed] [Google Scholar]
- 31.Kisby GE, Fry RC, Lasarev MR, Bammler TK, Beyer RP, Churchwell M, et al. The Cycad Genotoxin MAM Modulates Brain Cellular Pathways Involved in Neurodegenerative Disease and Cancer in a DNA Damage-Linked Manner. PLoS One. 2011;6:e20911. doi: 10.1371/journal.pone.0020911. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Thorgeirsson UP, Dalgard DW, Reeves J, Adamson RH. Tumor incidence in a chemical carcinogenesis study of nonhuman primates. Regul Toxicol Pharmacol. 1994;19:130–51. doi: 10.1006/rtph.1994.1013. [DOI] [PubMed] [Google Scholar]
- 33.Sieber SM, Correa P, Dalgard DW, McIntire KR, Adamson RH. Carcinogenicity and hepatotoxicity of cycasin and its aglycone methylazoxymethanol acetate in nonhuman primates. J Natl Cancer Inst. 1980;65:177–89. [PubMed] [Google Scholar]
- 34.Fukunishi R, Kadota A, Yoshida A, Hirota N. Induction of tumors with cycasin in newborn and preweanling rats. J Natl Cancer Inst. 1985;74:1275–81. [PubMed] [Google Scholar]
- 35.Niemeyer HB, Honig DM, Kulling SE, Metzler M. Studies on the metabolism of the plant lignans secoisolariciresinol and matairesinol. Journal of agricultural and food chemistry. 2003;51:6317–25. doi: 10.1021/jf030263n. [DOI] [PubMed] [Google Scholar]
- 36.Ward HA, Kuhnle GG, Mulligan AA, Lentjes MA, Luben RN, Khaw KT. Breast, colorectal, and prostate cancer risk in the European Prospective Investigation into Cancer and Nutrition-Norfolk in relation to phytoestrogen intake derived from an improved database. Am J Clin Nutr. 2010;91:440–8. doi: 10.3945/ajcn.2009.28282. [DOI] [PubMed] [Google Scholar]
- 37.Torres-Sanchez L, Galvan-Portillo M, Wolff MS, Lopez-Carrillo L. Dietary consumption of phytochemicals and breast cancer risk in Mexican women. Public Health Nutr. 2009;12:825–31. doi: 10.1017/S136898000800325X. [DOI] [PubMed] [Google Scholar]
- 38.Linseisen J, Piller R, Hermann S, Chang-Claude J. Dietary phytoestrogen intake and premenopausal breast cancer risk in a German case-control study. International journal of cancer Journal international du cancer. 2004;110:284–90. doi: 10.1002/ijc.20119. [DOI] [PubMed] [Google Scholar]
- 39.Touillaud MS, Thiebaut AC, Fournier A, Niravong M, Boutron-Ruault MC, Clavel-Chapelon F. Dietary lignan intake and postmenopausal breast cancer risk by estrogen and progesterone receptor status. Journal of the National Cancer Institute. 2007;99:475–86. doi: 10.1093/jnci/djk096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.van Duursen MB, Nijmeijer SM, de Morree ES, de Jong PC, van den Berg M. Genistein induces breast cancer-associated aromatase and stimulates estrogen-dependent tumor cell growth in in vitro breast cancer model. Toxicology. 2011 doi: 10.1016/j.tox.2011.07.005. [DOI] [PubMed] [Google Scholar]
- 41.Ju YH, Doerge DR, Woodling KA, Hartman JA, Kwak J, Helferich WG. Dietary genistein negates the inhibitory effect of letrozole on the growth of aromatase-expressing estrogen-dependent human breast cancer cells (MCF-7Ca) in vivo. Carcinogenesis. 2008;29:2162–8. doi: 10.1093/carcin/bgn161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Li J, Chen J, Vidal JE, McClane BA. The Agr-like quorum-sensing system regulates sporulation and production of enterotoxin and beta2 toxin by Clostridium perfringens type A non-food-borne human gastrointestinal disease strain F5603. Infect Immun. 2011;79:2451–9. doi: 10.1128/IAI.00169-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Alonso-Amelot ME, Avendano M. Possible association between gastric cancer and bracken fern in Venezuela: an epidemiologic study. Int J Cancer. 2001;91:252–9. doi: 10.1002/1097-0215(20010115)91:2<252::aid-ijc1028>3.0.co;2-o. [DOI] [PubMed] [Google Scholar]
- 44.Potter DM, Baird MS. Carcinogenic effects of ptaquiloside in bracken fern and related compounds. Br J Cancer. 2000;83:914–20. doi: 10.1054/bjoc.2000.1368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Seawright AA. Directly toxic effects of plant chemicals which may occur in human and animal foods. Nat Toxins. 1995;3:227–32. doi: 10.1002/nt.2620030411. discussion 42. [DOI] [PubMed] [Google Scholar]
- 46.Shahin M, Moore MR, Worrall S, Smith BL, Seawright AA, Prakash AS. H-ras activation is an early event in the ptaquiloside-induced carcinogenesis: comparison of acute and chronic toxicity in rats. Biochem Biophys Res Commun. 1998;250:491–7. doi: 10.1006/bbrc.1998.9341. [DOI] [PubMed] [Google Scholar]
- 47.Shahin M, Smith BL, Worral S, Moore MR, Seawright AA, Prakash AS. Bracken fern carcinogenesis: multiple intravenous doses of activated ptaquiloside induce DNA adducts, monocytosis, increased TNF alpha levels, and mammary gland carcinoma in rats. Biochem Biophys Res Commun. 1998;244:192–7. doi: 10.1006/bbrc.1998.8240. [DOI] [PubMed] [Google Scholar]
- 48.Masuda EK, Kommers GD, Martins TB, Barros CS, Piazer JV. Morphological factors as indicators of malignancy of squamous cell carcinomas in cattle exposed naturally to bracken fern (Pteridium aquilinum) J Comp Pathol. 2010;144:48–54. doi: 10.1016/j.jcpa.2010.04.009. [DOI] [PubMed] [Google Scholar]
- 49.Evans IA, Jones RS, Mainwaring-Burton R. Passage of bracken fern toxicity into milk. Nature. 1972;237:107–8. doi: 10.1038/237107a0. [DOI] [PubMed] [Google Scholar]
- 50.Campos-da-Paz M, Pereira LO, Bicalho LS, Dorea JG, Pocas-Fonseca MJ, Santos Mde F. Interaction of bracken-fern extract with vitamin C in human submandibular gland and oral epithelium cell lines. Mutat Res. 2008;652:158–63. doi: 10.1016/j.mrgentox.2008.01.009. [DOI] [PubMed] [Google Scholar]
- 51.Alonso-Amelot ME, Avendano M. Human carcinogenesis and bracken fern: a review of the evidence. Curr Med Chem. 2002;9:675–86. doi: 10.2174/0929867023370743. [DOI] [PubMed] [Google Scholar]
- 52.Liu TY, Chen CC, Chen CL, Chi CW. Safrole-induced oxidative damage in the liver of Sprague-Dawley rats. Food Chem Toxicol. 1999;37:697–702. doi: 10.1016/s0278-6915(99)00055-1. [DOI] [PubMed] [Google Scholar]
- 53.Liu TY, Chung YT, Wang PF, Chi CW, Hsieh LL. Safrole-DNA adducts in human peripheral blood–an association with areca quid chewing and CYP2E1 polymorphisms. Mutat Res. 2004;559:59–66. doi: 10.1016/j.mrgentox.2003.12.013. [DOI] [PubMed] [Google Scholar]
- 54.Lee JM, Liu TY, Wu DC, Tang HC, Leh J, Wu MT, et al. Safrole-DNA adducts in tissues from esophageal cancer patients: clues to areca-related esophageal carcinogenesis. Mutat Res. 2005;565:121–8. doi: 10.1016/j.mrgentox.2004.10.007. [DOI] [PubMed] [Google Scholar]
- 55.Chung YT, Chen CL, Wu CC, Chan SA, Chi CW, Liu TY. Safrole-DNA adduct in hepatocellular carcinoma associated with betel quid chewing. Toxicol Lett. 2008;183:21–7. doi: 10.1016/j.toxlet.2008.09.013. [DOI] [PubMed] [Google Scholar]
- 56.Shieh DH, Chiang LC, Shieh TY. Augmented mRNA expression of tissue inhibitor of metalloproteinase-1 in buccal mucosal fibroblasts by arecoline and safrole as a possible pathogenesis for oral submucous fibrosis. Oral Oncol. 2003;39:728–35. doi: 10.1016/s1368-8375(03)00101-5. [DOI] [PubMed] [Google Scholar]
- 57.Benedetti MS, Malnoe A, Broillet AL. Absorption, metabolism and excretion of safrole in the rat and man. Toxicology. 1977;7:69–83. doi: 10.1016/0300-483x(77)90039-7. [DOI] [PubMed] [Google Scholar]
- 58.Sun QZ, Li ZJ, Li J, Wang YT, Jin D, Zheng X, et al. [Secretion expression of cholera toxin B subunit in food-grading Lactococcus lactis expression system] Zhonghua Liu Xing Bing Xue Za Zhi. 2009;30:1288–91. [PubMed] [Google Scholar]
- 59.Lin HC, Cheng HH, Huang CJ, Chen WC, Chen IS, Liu SI, et al. Safrole-induced cellular Ca2+ increases and death in human osteosarcoma cells. Pharmacol Res. 2006;54:103–10. doi: 10.1016/j.phrs.2006.03.002. [DOI] [PubMed] [Google Scholar]
- 60.Chang HC, Cheng HH, Huang CJ, Chen WC, Chen IS, Liu SI, et al. Safrole-induced Ca2+ mobilization and cytotoxicity in human PC3 prostate cancer cells. J Recept Signal Transduct Res. 2006;26:199–212. doi: 10.1080/10799890600662595. [DOI] [PubMed] [Google Scholar]
- 61.Meneely JP, Sulyok M, Baumgartner S, Krska R, Elliott CT. A rapid optical immunoassay for the screening of T-2 and HT-2 toxin in cereals and maize-based baby food. Talanta. 2010;81:630–6. doi: 10.1016/j.talanta.2009.12.055. [DOI] [PubMed] [Google Scholar]
- 62.Du A, Zhao B, Yin D, Zhang S, Miao J. Safrole oxide induces apoptosis by activating caspase-3, -8, and -9 in A549 human lung cancer cells. Bioorg Med Chem Lett. 2006;16:81–3. doi: 10.1016/j.bmcl.2005.09.050. [DOI] [PubMed] [Google Scholar]
- 63.Oses SM, Rantsiou K, Cocolin L, Jaime I, Rovira J. Prevalence and quantification of Shiga-toxin producing Escherichia coli along the lamb food chain by quantitative PCR. Int J Food Microbiol. 2010;141(Suppl 1):S163–9. doi: 10.1016/j.ijfoodmicro.2010.05.010. [DOI] [PubMed] [Google Scholar]
- 64.Kanki M, Seto K, Sakata J, Harada T, Kumeda Y. Simultaneous enrichment of shiga toxin-producing Escherichia coli O157 and O26 and Salmonella in food samples using universal preenrichment broth. J Food Prot. 2009;72:2065–70. doi: 10.4315/0362-028x-72.10.2065. [DOI] [PubMed] [Google Scholar]
- 65.Huang JK, Huang CJ, Chen WC, Liu SI, Hsu SS, Chang HT, et al. Independent [Ca2+]i increases and cell proliferation induced by the carcinogen safrole in human oral cancer cells. Naunyn Schmiedebergs Arch Pharmacol. 2005;372:88–94. doi: 10.1007/s00210-005-1086-y. [DOI] [PubMed] [Google Scholar]
- 66.Jones RG, Marks JD. Use of a new functional dual coating (FDC) assay to measure low toxin levels in serum and food samples following an outbreak of human botulism. J Med Microbiol. 2013;62:828–35. doi: 10.1099/jmm.0.053124-0. [DOI] [PubMed] [Google Scholar]
- 67.Diercke M, Kirchner M, Claussen K, Mayr E, Strotmann I, Frangenberg J, et al. Transmission of shiga toxin-producing Escherichia coli O104:H4 at a family party possibly due to contamination by a food handler, Germany 2011. Epidemiol Infect. 2014;142:99–106. doi: 10.1017/S0950268813000769. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Wang F, Yang Q, Kase JA, Meng J, Clotilde LM, Lin A, et al. Current trends in detecting non-O157 Shiga toxin-producing Escherichia coli in food. Foodborne Pathog Dis. 2013;10:665–77. doi: 10.1089/fpd.2012.1448. [DOI] [PubMed] [Google Scholar]
- 69.Srivastava S, Kumar V, Ali MA, Solanki PR, Srivastava A, Sumana G, et al. Electrophoretically deposited reduced graphene oxide platform for food toxin detection. Nanoscale. 2013;5:3043–51. doi: 10.1039/c3nr32242d. [DOI] [PubMed] [Google Scholar]
- 70.Swain E, Poulton JE. Utilization of Amygdalin during Seedling Development of Prunus serotina. Plant Physiol. 1994;106:437–45. doi: 10.1104/pp.106.2.437. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Lerner IJ. Laetrile: a lesson in cancer quackery. CA Cancer J Clin. 1981;31:91–5. doi: 10.3322/canjclin.31.2.91. [DOI] [PubMed] [Google Scholar]
- 72.Fenselau C, Pallante S, Batzinger RP, Benson WR, Barron RP, Sheinin EB, et al. Mandelonitrile beta-glucuronide: synthesis and characterization. Science. 1977;198:625–7. doi: 10.1126/science.335509. [DOI] [PubMed] [Google Scholar]
- 73.Moertel CG, Ames MM, Kovach JS, Moyer TP, Rubin JR, Tinker JH. A pharmacologic and toxicological study of amygdalin. Jama. 1981;245:591–4. [PubMed] [Google Scholar]
- 74.O’Brien B, Quigg C, Leong T. Severe cyanide toxicity from ’vitamin supplements’. Eur J Emerg Med. 2005;12:257–8. doi: 10.1097/00063110-200510000-00014. [DOI] [PubMed] [Google Scholar]
- 75.Milazzo S, Lejeune S, Ernst E. Laetrile for cancer: a systematic review of the clinical evidence. Support Care Cancer. 2007;15:583–95. doi: 10.1007/s00520-006-0168-9. [DOI] [PubMed] [Google Scholar]
- 76.Milazzo S, Ernst E, Lejeune S, Schmidt K. Laetrile treatment for cancer. Cochrane Database Syst Rev. 2006:CD005476. doi: 10.1002/14651858.CD005476.pub2. [DOI] [PubMed] [Google Scholar]
- 77.Unproven methods of cancer management. Laetrile. CA Cancer J Clin. 1991;41:187–92. doi: 10.3322/canjclin.41.3.187. [DOI] [PubMed] [Google Scholar]
- 78.Nolin TD, Himmelfarb J. Mechanisms of drug-induced nephrotoxicity. Handb Exp Pharmacol. 2010:111–30. doi: 10.1007/978-3-642-00663-0_5. [DOI] [PubMed] [Google Scholar]
- 79.Zhang H, Cifone MA, Murli H, Erexson GL, Mecchi MS, Lawlor TE. Application of simplified in vitro screening tests to detect genotoxicity of aristolochic acid. Food Chem Toxicol. 2004;42:2021–8. doi: 10.1016/j.fct.2004.07.016. [DOI] [PubMed] [Google Scholar]
- 80.Jimenez-Ferrer JE, Perez-Teran YY, Roman-Ramos R, Tortoriello J. Antitoxin activity of plants used in Mexican traditional medicine against scorpion poisoning. Phytomedicine : international journal of phytotherapy and phytopharmacology. 2005;12:116–22. doi: 10.1016/j.phymed.2003.10.001. [DOI] [PubMed] [Google Scholar]
- 81.Arlt VM, Stiborova M, Schmeiser HH. Aristolochic acid as a probable human cancer hazard in herbal remedies: a review. Mutagenesis. 2002;17:265–77. doi: 10.1093/mutage/17.4.265. [DOI] [PubMed] [Google Scholar]
- 82.Nortier JL, Martinez MC, Schmeiser HH, Arlt VM, Bieler CA, Petein M, et al. Urothelial carcinoma associated with the use of a Chinese herb (Aristolochia fangchi) N Engl J Med. 2000;342:1686–92. doi: 10.1056/NEJM200006083422301. [DOI] [PubMed] [Google Scholar]
- 83.Cheung TP, Xue C, Leung K, Chan K, Li CG. Aristolochic acids detected in some raw Chinese medicinal herbs and manufactured herbal products–a consequence of inappropriate nomenclature and imprecise labelling? Clin Toxicol (Phila) 2006;44:371–8. doi: 10.1080/15563650600671712. [DOI] [PubMed] [Google Scholar]
- 84.Final report on carcinogens background document for aristolochic acids. Rep Carcinog Backgr Doc. 2008:i–xxv. 1–246. [PubMed] [Google Scholar]
- 85.Lai MN, Wang SM, Chen PC, Chen YY, Wang JD. Population-based case-control study of Chinese herbal products containing aristolochic acid and urinary tract cancer risk. J Natl Cancer Inst. 2010;102:179–86. doi: 10.1093/jnci/djp467. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Blumberg PM. Protein kinase C as the receptor for the phorbol ester tumor promoters: sixth Rhoads memorial award lecture. Cancer research. 1988;48:1–8. [PubMed] [Google Scholar]
- 87.Han ZT, Zhu XX, Yang RY, Sun JZ, Tian GF, Liu XJ, et al. Effect of intravenous infusions of 12-O-tetradecanoylphorbol-13-acetate (TPA) in patients with myelocytic leukemia: preliminary studies on therapeutic efficacy and toxicity. Proceedings of the National Academy of Sciences of the United States of America. 1998;95:5357–61. doi: 10.1073/pnas.95.9.5357. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Lee HW, Ahn DH, Crawley SC, Li JD, Gum JR, Jr, Basbaum CB, et al. Phorbol 12-myristate 13-acetate up-regulates the transcription of MUC2 intestinal mucin via Ras, ERK, and NF-kappa B. J Biol Chem. 2002;277:32624–31. doi: 10.1074/jbc.M200353200. [DOI] [PubMed] [Google Scholar]
- 89.Han ZT, Tong YK, He LM, Zhang Y, Sun JZ, Wang TY, et al. 12-O-Tetradecanoylphorbol-13-acetate (TPA)-induced increase in depressed white blood cell counts in patients treated with cytotoxic cancer chemotherapeutic drugs. Proceedings of the National Academy of Sciences of the United States of America. 1998;95:5362–5. doi: 10.1073/pnas.95.9.5362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Afrasiabi E, Ahlgren J, Bergelin N, Tornquist K. Phorbol 12-myristate 13-acetate inhibits FRO anaplastic human thyroid cancer cell proliferation by inducing cell cycle arrest in G1/S phase: evidence for an effect mediated by PKCdelta. Molecular and cellular endocrinology. 2008;292:26–35. doi: 10.1016/j.mce.2008.04.018. [DOI] [PubMed] [Google Scholar]
- 91.Zheng X, Chang RL, Cui XX, Avila GE, Hebbar V, Garzotto M, et al. Effects of 12-O-tetradecanoylphorbol-13-acetate (TPA) in combination with paclitaxel (Taxol) on prostate Cancer LNCaP cells cultured in vitro or grown as xenograft tumors in immunodeficient mice. Clin Cancer Res. 2006;12:3444–51. doi: 10.1158/1078-0432.CCR-05-2823. [DOI] [PubMed] [Google Scholar]
- 92.Furstenberger G, Berry DL, Sorg B, Marks F. Skin tumor promotion by phorbol esters is a two-stage process. Proceedings of the National Academy of Sciences of the United States of America. 1981;78:7722–6. doi: 10.1073/pnas.78.12.7722. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Smith LW, Culvenor CC. Plant sources of hepatotoxic pyrrolizidine alkaloids. J Nat Prod. 1981;44:129–52. doi: 10.1021/np50014a001. [DOI] [PubMed] [Google Scholar]
- 94.Radominska-Pandya A. Preface. Drug Metab Rev. 2010;42:1–2. doi: 10.3109/03602530903205450. [DOI] [PubMed] [Google Scholar]
- 95.Edgar JA, Roeder E, Molyneux RJ. Honey from plants containing pyrrolizidine alkaloids: a potential threat to health. J Agric Food Chem. 2002;50:2719–30. doi: 10.1021/jf0114482. [DOI] [PubMed] [Google Scholar]
- 96.Kempf M, Reinhard A, Beuerle T. Pyrrolizidine alkaloids (PAs) in honey and pollen-legal regulation of PA levels in food and animal feed required. Mol Nutr Food Res. 2010;54:158–68. doi: 10.1002/mnfr.200900529. [DOI] [PubMed] [Google Scholar]
- 97.Ceuppens S, Rajkovic A, Heyndrickx M, Tsilia V, Van De Wiele T, Boon N, et al. Regulation of toxin production by Bacillus cereus and its food safety implications. Crit Rev Microbiol. 2011;37:188–213. doi: 10.3109/1040841X.2011.558832. [DOI] [PubMed] [Google Scholar]
- 98.Martin A, Beutin L. Characteristics of Shiga toxin-producing Escherichia coli from meat and milk products of different origins and association with food producing animals as main contamination sources. Int J Food Microbiol. 2011;146:99–104. doi: 10.1016/j.ijfoodmicro.2011.01.041. [DOI] [PubMed] [Google Scholar]
- 99.Edgar JA, Colegate SM, Boppre M, Molyneux RJ. Pyrrolizidine alkaloids in food: a spectrum of potential health consequences. Food Addit Contam Part A Chem Anal Control Expo Risk Assess. 2011;28:308–24. doi: 10.1080/19440049.2010.547520. [DOI] [PubMed] [Google Scholar]
- 100.Biesta-Peters EG, Reij MW, Blaauw RH, In ’t Veld PH, Rajkovic A, Ehling-Schulz M, et al. Quantification of the emetic toxin cereulide in food products by liquid chromatography-mass spectrometry using synthetic cereulide as a standard. Appl Environ Microbiol. 2010;76:7466–72. doi: 10.1128/AEM.01659-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Coulombe RA., Jr Pyrrolizidine alkaloids in foods. Adv Food Nutr Res. 2003;45:61–99. doi: 10.1016/s1043-4526(03)45003-1. [DOI] [PubMed] [Google Scholar]
- 102.Schoental R. Toxicology and carcinogenic action of pyrrolizidine alkaloids. Cancer Res. 1968;28:2237–46. [PubMed] [Google Scholar]
- 103.Zhao Y, Xia Q, Yin JJ, Lin G, Fu PP. Photoirradiation of dehydropyrrolizidine alkaloids-Formation of reactive oxygen species and induction of lipid peroxidation. Toxicol Lett. 2011;205:302–9. doi: 10.1016/j.toxlet.2011.06.020. [DOI] [PubMed] [Google Scholar]
- 104.Betz JM, Eppley RM, Taylor WC, Andrzejewski D. Determination of pyrrolizidine alkaloids in commercial comfrey products (Symphytum sp.) J Pharm Sci. 1994;83:649–53. doi: 10.1002/jps.2600830511. [DOI] [PubMed] [Google Scholar]
- 105.Mei N, Guo L, Fu PP, Fuscoe JC, Luan Y, Chen T. Metabolism, genotoxicity, and carcinogenicity of comfrey. J Toxicol Environ Health B Crit Rev. 2010;13:509–26. doi: 10.1080/10937404.2010.509013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Mei N, Guo L, Zhang L, Shi L, Sun YA, Fung C, et al. Analysis of gene expression changes in relation to toxicity and tumorigenesis in the livers of Big Blue transgenic rats fed comfrey (Symphytum officinale) BMC Bioinformatics. 2006;7(Suppl 2):S16. doi: 10.1186/1471-2105-7-S2-S16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Mei N, Guo L, Liu R, Fuscoe JC, Chen T. Gene expression changes induced by the tumorigenic pyrrolizidine alkaloid riddelliine in liver of Big Blue rats. BMC Bioinformatics. 2007;8(Suppl 7):S4. doi: 10.1186/1471-2105-8-S7-S4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Mei N, Chou MW, Fu PP, Heflich RH, Chen T. Differential mutagenicity of riddelliine in liver endothelial and parenchymal cells of transgenic big blue rats. Cancer Lett. 2004;215:151–8. doi: 10.1016/j.canlet.2004.06.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Guo L, Mei N, Dial S, Fuscoe J, Chen T. Comparison of gene expression profiles altered by comfrey and riddelliine in rat liver. BMC Bioinformatics. 2007;8(Suppl 7):S22. doi: 10.1186/1471-2105-8-S7-S22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Fu PP, Yang YC, Xia Q, Chou MC, Cui YY, Lin G. Pyrrolizidine alkaloids-tumorigenic components in Chinese herbal medicines and dietary supplements. J Food Drug Anal. 2002;10:198–211. [Google Scholar]
- 111.Roder E. Medicinal plants in Europe containing pyrrolizidine alkaloids. Pharmazie. 1995;50:83–98. [PubMed] [Google Scholar]
- 112.Weisbord SD, Soule JB, Kimmel PL. Poison on line–acute renal failure caused by oil of wormwood purchased through the Internet. N Engl J Med. 1997;337:825–7. doi: 10.1056/NEJM199709183371205. [DOI] [PubMed] [Google Scholar]
- 113.Winickoff JP, Houck CS, Rothman EL, Bauchner H. Verve and Jolt: deadly new Internet drugs. Pediatrics. 2000;106:829–30. doi: 10.1542/peds.106.4.829. [DOI] [PubMed] [Google Scholar]
- 114.Gold LS, Slone TH. Aristolochic acid, an herbal carcinogen, sold on the Web after FDA alert. N Engl J Med. 2003;349:1576–7. doi: 10.1056/NEJM200310163491619. [DOI] [PubMed] [Google Scholar]
- 115.Bessell TL, Anderson JN, Silagy CA, Sansom LN, Hiller JE. Surfing, self-medicating and safety: buying non-prescription and complementary medicines via the internet. Qual Saf Health Care. 2003;12:88–92. doi: 10.1136/qhc.12.2.88. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Bonakdar RA. Herbal cancer cures on the Web: noncompliance with The Dietary Supplement Health and Education Act. Fam Med. 2002;34:522–7. [PubMed] [Google Scholar]
- 117.Morris CA, Avorn J. Internet marketing of herbal products. Jama. 2003;290:1505–9. doi: 10.1001/jama.290.11.1505. [DOI] [PubMed] [Google Scholar]


