Abstract
Background:
Nephrolithiasis is of the most prevalent urinary tract disease. It seems worthwhile to replace the conventional treatments with more beneficial and safer agents, particularly herbal medicines which are receiving an increasing interest nowadays.
Aims:
In this study, we investigated the protective and curative effects of Achillea millefolium L. on ethylene glycol (EG)-induced nephrolithiasis in rats.
Materials and Methods:
The extract of A. millefolium was prepared by soxhlet method. Forty male Wistar rats were randomly divided into five groups (N = 8) as follows. The negative control (group A) received tap drinking water. Rats in sham (positive control group B), curative (group C and D), and preventive (group E) groups all received 1% EG in drinking water according to the experimental protocol for 30 days. In the curative groups, dosages of 200 and 400 mg/kg body weight (BW) of A. millefolium extract were administered orally from day 15 to the end of the experiment, group C and D, respectively. Group E received 200 mg/kg A. millefolium extract from the 1st day throughout the experiment. Urinary oxalate and citrate concentrations were measured by spectrophotometer on the first and 30th days. On day 31, the kidneys were removed and examined histopathologically for counting the calcium oxalate (CaOx) deposits in 50 microscopic fields.
Results:
In the curative and preventive groups, administration of A. millefolium extract showed significant reduction in urinary oxalate concentration (P < 0.05). Also, urinary citrate concentration was significantly increased in group C, D, and E. The CaOx deposits significantly decreased in group C to E compared with the group B.
Conclusions:
According to our results, A. millefolium extract had preventive and curative effects on EG-induced renal calculi.
Keywords: Achillea millefolium, Calcium oxalate, ethylene glycol, nephrolithiasis
Introduction
Renal stones or nephrolithiasis are one of the most common urinary tract problems occurring in 1-15% of the general population.[1] The most important complications of kidney stones are hematuria, abdominal and flank pain, hydronephrosis, urinary tract infections, and even renal failure.[2,3] Despite many investigations, the etiology of renal stone formation remains uncertain. Several factors have been suggested which may predispose individuals to nephrolithiasis, such as genetic predispositions, gender, age, weather conditions, water intake and its solutes, diet, and etc.[4] Approximately 80% of renal stones are composed of calcium which has been combined with oxalate or phosphate.[5] In Iranian traditional medicine, many plants were introduced for disposal, dissolve, and preventing the formation of kidney stones. On the other hand, the present therapies are to some extent effective and there is still no definite treatment for this problem.[6] Therefore, natural and herbal medicines are believed to have many medicinal impacts as well as less adverse effects.
Achillea millefolium L., from Asteraceae family and known as yarrow, is a perennial herb which grows in Europe, Asia, and North America. In Iran, it can be found in the northern area, around the Alborz Mountain and in Azerbaijan, Lorestan, and Isfahan provinces.[7] A. millefolium has been prescribed in traditional medicine as healing the wounds, to heal chapped skin, hair tonic, diaphoretic,[8,9,10,11,12] loss of appetite, dyspepsia,[10] flatulence, anti-dysenteric, astringent, anti-hemorrhoids, anti-inflammatory, anti-pyretic, decrease blood pressure, emmenagogue, cholagogue, anti-hemorrhagic, diuretic, urinary antiseptic, anti-spasmodic[9,10,11,12] anti-bacterial and anti-plasmodium.[12,13]
Currently, besides the present herbal and chemical medications, many invasive procedures such as extracorporeal shock wave lithotripsy, percutaneous lithotomy, and transureteral lithotripsy are widely used to treat nephrolithiasis. However, using these procedures is not cost-effective and may lead to serious complications.[14,15,16] Hence, it seems worthwhile to replace these conventional treatments with more beneficial and safer agents, particularly herbal medicines which are receiving an increasing interest nowadays.
In this study, we aimed to evaluate the protective and curative effects of hydroalcoholic extract of A. millefolium L. on ethylene glycol-induced nephrolithiasis in laboratory rats.
Materials and Methods
Extract preparation
The flowers of Achillea millefolium L. were collected during post-flowering phases (May 2013) from the field of Medicinal Plants Institute-ACECR (Karaj, Iran) and were identified by the Pharmacognosy Department of Tehran University (herbarium code: 83001). The plants were dried in standard condition in dry shade and were powdered. Fifty grams of dried powder were soaked in 300 ml 80% ethanol for 48 hours with intermittent shaking. The plant extracts were filtered through filter paper and the filtrates were dried until a constant dry weight of extract was obtained. The extract was dissolved in distilled water to 200 and 400 mg/ml and administered to rats orally at doses of 200 and 400 mg/kg body weight (BW). The residues were stored at 4°C.
Animals
Forty male Wistar rats weighing 250-300 g were obtained from the Pasteur Institute, Karaj, Iran. The rats were housed in standard cages at 25 ± 2° C and 12:12-hours light-darkness cycle with free access to water and food ad libitum. All procedures used in the study were approved by the ethics committee for the use of experimental animals at Shiraz University of Medical Sciences, Shiraz, Iran.
Animals were divided randomly into five groups (N = 8) as follows: The control group A (control) received tap drinking water. Rats in sham (group B), curative (group C and D), and preventive (group E) groups all received 1% ethylene glycol (EG) (Mojallali Chemical Labs, Iran) in drinking water according to the experimental protocol for 30 days. In the curative groups dosages of 200 and 400 mg/kg of A. millefolium extract were administered orally, since day 15 to the end of the experiment, group C and D, respectively. Group E received 200 mg/kg of A. millefolium extract from the 1st day through the end of the experiment.
Urine analysis
For 24-hour urine collection, all rats on the first and 30th day were kept individually in metabolic cages. Urinary oxalate and citrate concentrations were measured by diagnostic kits (Darman Kaw, Tehran, Iran) with urine autoanalyzer.
Histology Examination At the end of day 31, all of the rats underwent general anesthesia and were decapitated by guillotine. Then both the kidneys were removed and kept in formalin (10%) for histological studies. Three 5 μm sections were prepared from each kidney and the slides were stained with hematoxylin-eosin (H&E) and examined by light microscope. Aggregations of calcium oxalate (CaOx) deposits in the renal tubules were counted in 10 microscopic fields.
Statistical analysis
Data were presented as the mean± standard deviation (SD). Analysis of variance (ANOVA) test was used to determine difference among all groups. For comparison between each two groups, the Mann Whitney U test was applied. Data were analyzed by statistical software Statistical Package for the Social Sciences (SPSS) version 14.0 (SPSS Inc, Chicago, IL). Significant difference was set at P < 0.05.
Results
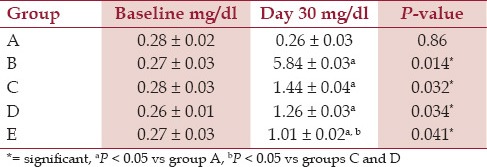
Urinary oxalate concentration in the curative and preventive groups was significantly increased in contrast with the control group [Table 1]. In both curative groups, oxalate excretion was statistically significant at day 30 (P = 0.04). Also, its concentration was decreased significantly in group E in comparison with groups C and D (P = 0.02).
Table 1.
Th e 24-hour urinary oxalate concentration in all groups at the baseline and day 30 of experiment (Mean ± standard deviation, SD)
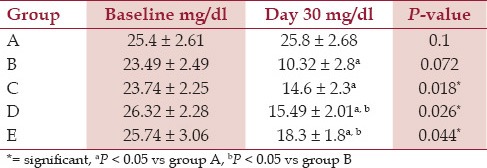
As it is shown in [Table 2], urine citrate concentration of the rats in group B was significantly lower than those of all other groups. Results showed no significant difference between group C and group B regarding urine citrate concentrations (P = 0.06). However, urine citrate levels in groups D and E were significantly different from those in group B (P = 0.03 and P = 0.015, respectively).
Table 2.
The 24-hour urinary citrate concentration in all groups at the baseline and day 30 of experiment (Mean ± standard deviation, SD)
In group A, histopathological findings revealed that there was no CaOx deposit in different segments of the kidney [Figure 1]. The mean number of CaOx deposits in all groups was significantly higher than the group A [Figure 2]. The means of CaOx deposits in group C, D, and E were 20.87 ± 1.98, 16.37 ± 1.99, and 14.25 ± 1.98, respectively. In groups C, D, and E, CaOx deposits were decreased significantly in comparison to the group B (P = 0.001, P = 0.01, and P = 0.004, respectively) [Figure 3]; however, there was no significant difference among A. millefolium-treated groups.
Figure 1.
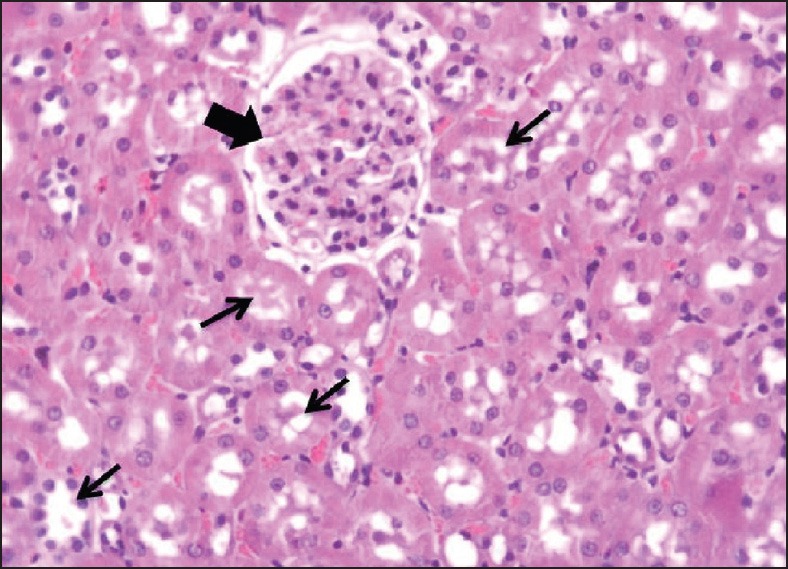
Histopathology (hematoxylin-eosin; original magnification, ×400); Intact renal tubules (thin arrows) and glomeruli (thick arrow) are observed in group A
Figure 2.
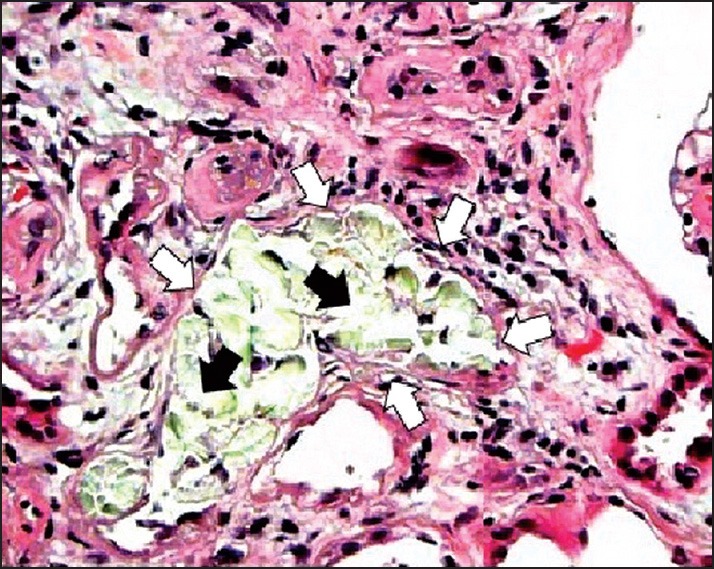
Histopathology (hematoxylin-eosin; original magnification, ×400); Renal tubular dilation (white arrows) with calcium oxalate (CaOx) deposits (black arrows) in group B
Figure 3.
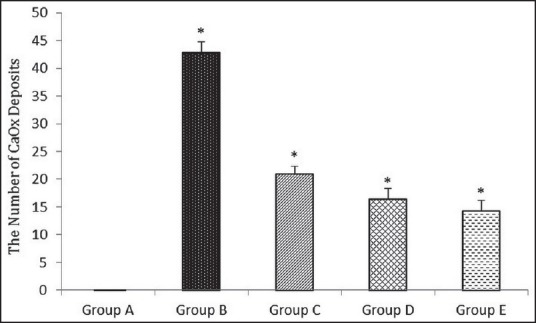
The effect of A. millefolium on the number of calcium oxalate (CaOx) deposits. All the values are expressed as mean ± standard deviation (SD), (n = 8), *P < 0.05
Discussion
In recent years, various herbal and synthetic medicines were produced to reduce nephrolithiasis that may be helpful in the prevention and treatment, but there is no certain medication which brings about complete treatment and prevention of urinary stones; some patients may need surgical interventions at the end due to impassable stones, those which cannot pass the tract due to their large size. Medications can lead to minimizing the stones, breaking them apart, or dilating the lumen for their passage. In this study, we investigated the protective and curative effects of A. millefolium on EG-induced nephrolithiasis in rats.
A high urine saturation of oxalate encourages the CaOx crystal formation in the renal tubules.[17] Previous studies showed hyperoxaluria can damage renal epithelial cells. Free radicals which are produced by injured cell lead to induction of heterogeneous crystal nucleation and can promote calculi formation.[14,18] According to the results of this study, EG intake led to increased urinary oxalate excretion in the rats. In both curative and preventive groups, urinary oxalate was significantly decreased. Also, its concentration was decreased by increased dosage of A. millefolium (200 mg/kg compared with 400 mg/kg). The exact mechanism of yarrow on EG metabolism and degradation is not known. On the other hand, anti-oxidant and anti-inflammatory abilities of A. millefolium have been reported.[11,19] Therefore, one of the possible mechanisms of preventive and curative effects of A. millefolium can be related to these activities which may interfere with renal epithelial cells damage process and inhibit the inflammation.
It has been reported that the main risk factor of recurrent CaOx calculus is hypocitraturia.[20] Urine citrate is able to prevent CaOx crystal formation[20,21] due to inhibition of nucleation and growth of crystals.[21,22] According to our results, A. millefolium extract increased urine citrate concentration in both preventive and curative groups in comparison to group B.
Ahmadi et al. showed that hydroalcoholic extract of Alcearosea had both preventive and curative effects on the formation of CaOx calculi apparently due to its diuretic and anti-inflammatory activities, presence of mucilaginous polysaccharides, and lowering of urinary concentrations of stone-forming constituents.[23] Low volume of urine is one the risk factors of nephrolithiasis due to hypersaturation of all solutes.[23,24] It has been reported that A. millefolium has diuretic effects and increases diuresis by 25% from initial volume without changing of urine pH in the patients with calcium type of urolithiasis.[24] Therefore, the diuretic effect of A. millefolium may be another possible mechanism involved in prevention of new calculi formation and dissolution of preformed stones.
Bacterial infections may play a role in the formation of some renal stones such as struvite calculi, and it has been suggested that it might be involved in calcium-based stones too.[25] Also, Khajavi Rad et al. study indicated that the ethyl acetate fraction of Cynodondactylon had preventive and curative effects on CaOx calculus due to its antioxidant and anti-bacterial effects.[26] Some studies indicated that yarrow has an antimicrobial effects[11] which may be another possible mechanism that can be involved in preventive and curative effects of A. millefolium.
Conclusion
The results of the present study indicated that A. millefolium extract had preventive and curative effects on EG-induced renal stones due to different possible mechanisms such as antioxidant, anti-inflammatory, diuretic, and antibacterial activities. However, we recommend further investigations and clinical trials for determining the effects of this plant on different aspects of nephrolithiasis treatment.
Footnotes
Source of Support: Nil
Conflict of Interest: None declared.
References
- 1.Thornton SN, Scales CD, Jr, Smith AC, Hanley JM, Saigal CS. Urologic Diseases in America Project. Prevalence of kidney stones in the United States. Eur Urol. 2012;62:160–5. doi: 10.1016/j.eururo.2012.03.052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Fink HA, Akornor JW, Garimella PS, MacDonald R, Cutting A, Rutks IR, et al. Diet, fluid, or supplements for secondary prevention of nephrolithiasis: A systematic review and meta-analysis of randomized trials. Eur Urol. 2009;56:72–80. doi: 10.1016/j.eururo.2009.03.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Rule AD, Bergstralh EJ, Melton LJ, Li X, Weaver AL, Lieske JC. Kidney Stones and the risk for chronic kidney disease. Clin J Am Soc Nephrol. 2009;4:804–11. doi: 10.2215/CJN.05811108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Goldfarb DS. Prospects for dietary therapy of recurrent nephrolithiasis. Adv Chronic Kidney Dis. 2009;16:21–9. doi: 10.1053/j.ackd.2008.10.010. [DOI] [PubMed] [Google Scholar]
- 5.Parks JH, Asplin JR, Coe FL. Patient adherence to long-term medical treatment of kidney stones. J Urol. 2001;166:2057–60. [PubMed] [Google Scholar]
- 6.Khalighi-Sigaroodi F, Jarvandi S, Taghizadeh M, editors. Therapeutic Indications of Medicinal Plants. 2nd ed. Vol 2. Tehran: Arjmand Publication; 2010. pp. 186–9. [Google Scholar]
- 7.Akhondzadeh SH. Encyclopedia of Iranian Medicinal Plants. Tehran: Arjmand Publication; 2001. [Google Scholar]
- 8.Zargari A, editor. Medicinal Plants. 6th ed. Vol 5. Tehran: Arjmand Publication; 1996. pp. 146–58. [Google Scholar]
- 9.Benedek B, Kopp B. Achillea millefolium L. sl revisited: Recent findings confirm the traditional use. Wien Med Wochenschr. 2007;157:312–4. doi: 10.1007/s10354-007-0431-9. [DOI] [PubMed] [Google Scholar]
- 10.Fleming T, editor. PDF for Herbal Medicine. 1st ed. Vol 1. New Jersey: Medical Economics Company; 1998. pp. 604–1217. [Google Scholar]
- 11.Khare CP, editor. Indian Medicinal Plants: An Illustrated Dictionary. 2nd ed. Vol 1. Berlin: Springer; 2007. pp. 91–3. [Google Scholar]
- 12.Yaeesh S, Jamal Q, Khan A, Gilani A. Studies on hepatoprotective, antispasmodic and calcium antagonist activities of the aqueous-methanol extract of Achillea millefolium. Phytother Res. 2006;20:546–51. doi: 10.1002/ptr.1897. [DOI] [PubMed] [Google Scholar]
- 13.Vitalini S, Beretta G, Iriti M, Orsenigo S, Basilico N, Dall'Acqua S, et al. Phenolic compounds from Achillea millefolium L. and their bioactivity. Acta Biochim Pol. 2011;58:203–9. [PubMed] [Google Scholar]
- 14.Hadjzadeh M, Khoei A, Hadjzadeh Z, Parizady M. Ethanolic extract of nigella sativa L seeds on ethylene glycol-induced kidney calculi in rats. Urol J. 2007;4:86–90. [PubMed] [Google Scholar]
- 15.Atmani F, Slimani Y, Mimouni M, Aziz M, Hacht B, Ziyyat A. Effect of aqueous extract from Herniaria hirsuta L. on experimentally nephrolithiasic rats. J Ethnopharmacol. 2004;95:87–93. doi: 10.1016/j.jep.2004.06.028. [DOI] [PubMed] [Google Scholar]
- 16.Laroubi A, Touhami M, Farouk L, Zrara I, Aboufatima R, Benharref A, et al. Prophylaxis effect of Trigonella foenum graecum L. seeds on renal stone formation in rats. Phytother Res. 2007;21:921–5. doi: 10.1002/ptr.2190. [DOI] [PubMed] [Google Scholar]
- 17.Robertson W, Peacock M. The cause of idiopathic calcium stone disease: Hypercalciuria or hyperoxaluria? Nephron. 1980;26:105–10. doi: 10.1159/000181963. [DOI] [PubMed] [Google Scholar]
- 18.Hadjzadeh MA, Rad AK, Rajaei Z, Tehranipour M, Monavar N. The preventive effect of N-butanol fraction of Nigella sativa on ethylene glycol-induced kidney calculi in rats. Pharmacogn Mag. 2011;7:338–43. doi: 10.4103/0973-1296.90416. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Trumbeckaite S, Benetis R, Bumblauskiene L, Burdulis D, Janulis V, Toleikis A, et al. Achillea millefolium L. s.l. herb extract: Antioxidant activity and effect on the rat heart mitochondrial functions. Food Chem. 2011;127:1540–8. [Google Scholar]
- 20.Menon M, Mahle C. Urinary citrate excretion in patients with renal calculi. J Urol. 1983;129:1158–60. doi: 10.1016/s0022-5347(17)52618-x. [DOI] [PubMed] [Google Scholar]
- 21.Xu H, Zisman AL, Coe FL, Worcester EM. Kidney stones: An update on current pharmacological management and future directions. Expert Opin Pharmacother. 2013;14:435–47. doi: 10.1517/14656566.2013.775250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Goldberg H, Grass L, Vogl R, Rapoport A, Oreopoulos D. Urine citrate and renal stone disease. CMAJ. 1989;141:217–21. [PMC free article] [PubMed] [Google Scholar]
- 23.Ahmadi M, Rad AK, Rajaei Z, Hadjzadeh MAR, Mohammadian N, Tabasi NS. Alcea rosea root extract as a preventive and curative agent in ethylene glycol-induced urolithiasis in rats. Indian J Pharmacol. 2012;44:304–7. doi: 10.4103/0253-7613.96298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Gaybullaev A, Kariev S. Phytotherapy of calcium urolithiasis with extracts of medicinal plants: Changes of diuresis, urine pH and crystalluria. Appl Technol Innov. 2012;7:59–66. [Google Scholar]
- 25.Kramer G, Klingler HC, Steiner GE. Role of bacteria in the development of kidney stones. Curr Opin Urol. 2000;10:35–8. doi: 10.1097/00042307-200001000-00009. [DOI] [PubMed] [Google Scholar]
- 26.Khajavi Rad A, Hadjzadeh MAR, Rajaei Z, Mohammadian N, Valiollahi S, Sonei M. The beneficial effect of Cynodondactylon fractions on ethylene glycol-induced kidney calculi in rats. Urol J. 2011;8:179–84. [PubMed] [Google Scholar]


