Abstract
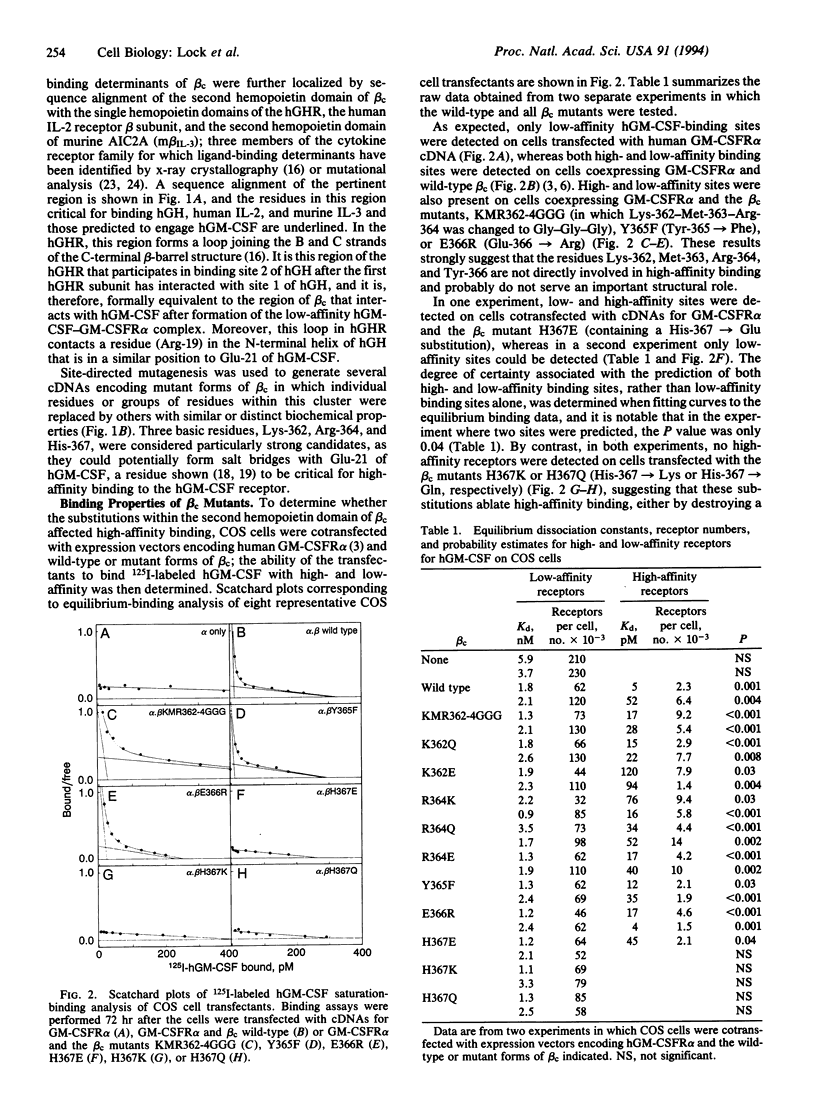
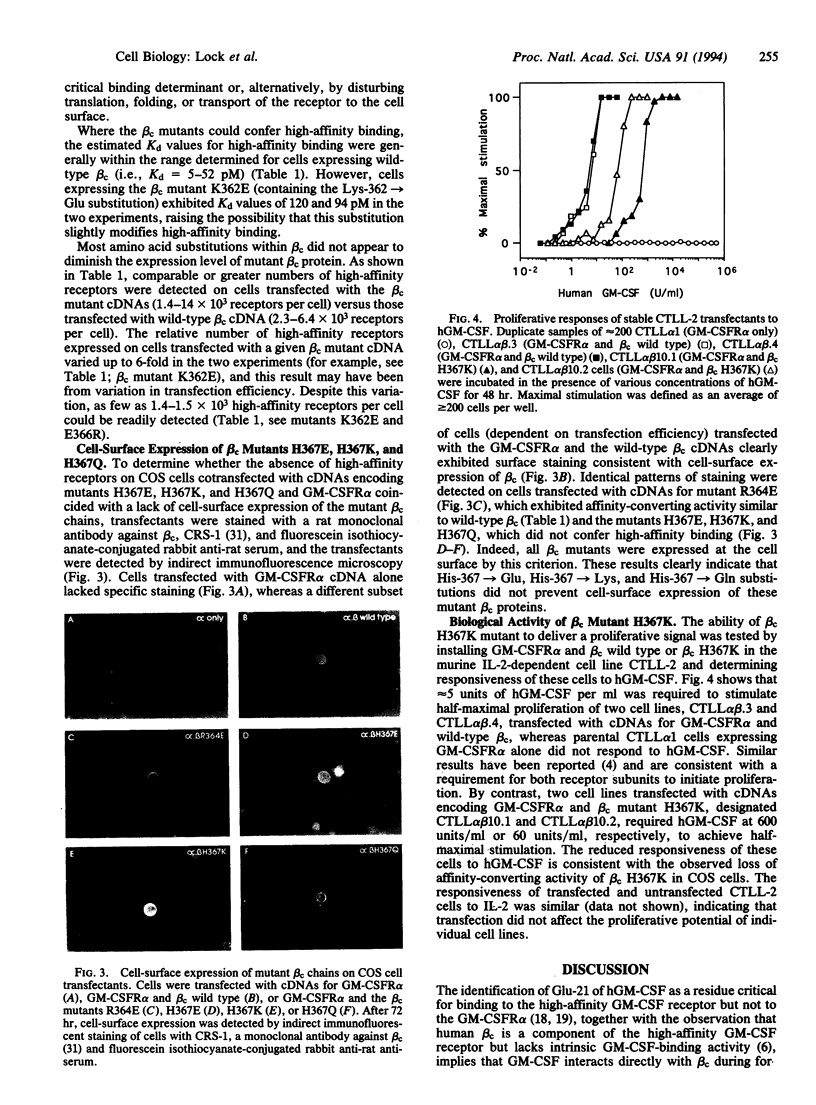
High-affinity receptors for granulocyte-macrophage colony-stimulating factor (GM-CSF), interleukin 3, and interleukin 5 consist of ligand-specific alpha chains (low-affinity subunits) and a common beta chain (beta c) that converts each complex to a high-affinity form. Although beta c alone has no detectable cytokine-binding activity, amino acid substitutions for Glu-21 of human GM-CSF significantly reduce high-affinity but not low-affinity binding, implying that beta c interacts directly with GM-CSF during formation of the high-affinity receptor but only in the presence of the alpha chain. A potential GM-CSF-binding determinant was identified in the second hemopoietin domain of beta c, and the role of individual residues within this region was investigated by determining the ability of mutated beta c chains to confer high-affinity binding when coexpressed with the alpha subunit of the GM-CSF receptor in COS cells. Substitutions involving Met-363, Arg-364, Tyr-365, and Glu-366 did not affect high-affinity binding. However, substitution of His-367 by lysine or glutamine abolished high-affinity binding, suggesting that this residue may form an important part of the high-affinity GM-CSF-binding determinant. Consistent with the loss of high-affinity binding, higher concentrations of human GM-CSF were required to stimulate proliferation of CTLL-2 cell lines transfected with cDNAs for GM-CSF receptor alpha chain and His-367 beta c mutant than those expressing GM-CSF receptor alpha subunit and beta c wild type.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bazan J. F. Haemopoietic receptors and helical cytokines. Immunol Today. 1990 Oct;11(10):350–354. doi: 10.1016/0167-5699(90)90139-z. [DOI] [PubMed] [Google Scholar]
- Cosman D., Lyman S. D., Idzerda R. L., Beckmann M. P., Park L. S., Goodwin R. G., March C. J. A new cytokine receptor superfamily. Trends Biochem Sci. 1990 Jul;15(7):265–270. doi: 10.1016/0968-0004(90)90051-c. [DOI] [PubMed] [Google Scholar]
- Cunningham B. C., Ultsch M., De Vos A. M., Mulkerrin M. G., Clauser K. R., Wells J. A. Dimerization of the extracellular domain of the human growth hormone receptor by a single hormone molecule. Science. 1991 Nov 8;254(5033):821–825. doi: 10.1126/science.1948064. [DOI] [PubMed] [Google Scholar]
- Gearing D. P., King J. A., Gough N. M., Nicola N. A. Expression cloning of a receptor for human granulocyte-macrophage colony-stimulating factor. EMBO J. 1989 Dec 1;8(12):3667–3676. doi: 10.1002/j.1460-2075.1989.tb08541.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gillis S., Smith K. A. Long term culture of tumour-specific cytotoxic T cells. Nature. 1977 Jul 14;268(5616):154–156. doi: 10.1038/268154a0. [DOI] [PubMed] [Google Scholar]
- Gluzman Y. SV40-transformed simian cells support the replication of early SV40 mutants. Cell. 1981 Jan;23(1):175–182. doi: 10.1016/0092-8674(81)90282-8. [DOI] [PubMed] [Google Scholar]
- Gorman D. M., Itoh N., Kitamura T., Schreurs J., Yonehara S., Yahara I., Arai K., Miyajima A. Cloning and expression of a gene encoding an interleukin 3 receptor-like protein: identification of another member of the cytokine receptor gene family. Proc Natl Acad Sci U S A. 1990 Jul;87(14):5459–5463. doi: 10.1073/pnas.87.14.5459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hara T., Miyajima A. Two distinct functional high affinity receptors for mouse interleukin-3 (IL-3). EMBO J. 1992 May;11(5):1875–1884. doi: 10.1002/j.1460-2075.1992.tb05239.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hayashida K., Kitamura T., Gorman D. M., Arai K., Yokota T., Miyajima A. Molecular cloning of a second subunit of the receptor for human granulocyte-macrophage colony-stimulating factor (GM-CSF): reconstitution of a high-affinity GM-CSF receptor. Proc Natl Acad Sci U S A. 1990 Dec;87(24):9655–9659. doi: 10.1073/pnas.87.24.9655. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Higuchi R., Krummel B., Saiki R. K. A general method of in vitro preparation and specific mutagenesis of DNA fragments: study of protein and DNA interactions. Nucleic Acids Res. 1988 Aug 11;16(15):7351–7367. doi: 10.1093/nar/16.15.7351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Imler J. L., Miyajima A., Zurawski G. Identification of three adjacent amino acids of interleukin-2 receptor beta chain which control the affinity and the specificity of the interaction with interleukin-2. EMBO J. 1992 Jun;11(6):2047–2053. doi: 10.1002/j.1460-2075.1992.tb05262.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Itoh N., Yonehara S., Schreurs J., Gorman D. M., Maruyama K., Ishii A., Yahara I., Arai K., Miyajima A. Cloning of an interleukin-3 receptor gene: a member of a distinct receptor gene family. Science. 1990 Jan 19;247(4940):324–327. doi: 10.1126/science.2404337. [DOI] [PubMed] [Google Scholar]
- Kitamura T., Hayashida K., Sakamaki K., Yokota T., Arai K., Miyajima A. Reconstitution of functional receptors for human granulocyte/macrophage colony-stimulating factor (GM-CSF): evidence that the protein encoded by the AIC2B cDNA is a subunit of the murine GM-CSF receptor. Proc Natl Acad Sci U S A. 1991 Jun 15;88(12):5082–5086. doi: 10.1073/pnas.88.12.5082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kitamura T., Miyajima A. Functional reconstitution of the human interleukin-3 receptor. Blood. 1992 Jul 1;80(1):84–90. [PubMed] [Google Scholar]
- Lopez A. F., Shannon M. F., Hercus T., Nicola N. A., Cambareri B., Dottore M., Layton M. J., Eglinton L., Vadas M. A. Residue 21 of human granulocyte-macrophage colony-stimulating factor is critical for biological activity and for high but not low affinity binding. EMBO J. 1992 Mar;11(3):909–916. doi: 10.1002/j.1460-2075.1992.tb05129.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Metcalf D., Nicola N. A., Gearing D. P., Gough N. M. Low-affinity placenta-derived receptors for human granulocyte-macrophage colony-stimulating factor can deliver a proliferative signal to murine hemopoietic cells. Proc Natl Acad Sci U S A. 1990 Jun;87(12):4670–4674. doi: 10.1073/pnas.87.12.4670. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mizushima S., Nagata S. pEF-BOS, a powerful mammalian expression vector. Nucleic Acids Res. 1990 Sep 11;18(17):5322–5322. doi: 10.1093/nar/18.17.5322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Munson P. J., Rodbard D. Ligand: a versatile computerized approach for characterization of ligand-binding systems. Anal Biochem. 1980 Sep 1;107(1):220–239. doi: 10.1016/0003-2697(80)90515-1. [DOI] [PubMed] [Google Scholar]
- Nicola N. A., Metcalf D. Binding, internalization, and degradation of 125I-multipotential colony-stimulating factor (interleukin-3) by FDCP-1 cells. Growth Factors. 1988;1(1):29–39. doi: 10.3109/08977198809000244. [DOI] [PubMed] [Google Scholar]
- Nicola N. A., Wycherley K., Boyd A. W., Layton J. E., Cary D., Metcalf D. Neutralizing and nonneutralizing monoclonal antibodies to the human granulocyte-macrophage colony-stimulating factor receptor alpha-chain. Blood. 1993 Sep 15;82(6):1724–1731. [PubMed] [Google Scholar]
- Park L. S., Martin U., Sorensen R., Luhr S., Morrissey P. J., Cosman D., Larsen A. Cloning of the low-affinity murine granulocyte-macrophage colony-stimulating factor receptor and reconstitution of a high-affinity receptor complex. Proc Natl Acad Sci U S A. 1992 May 15;89(10):4295–4299. doi: 10.1073/pnas.89.10.4295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sanderson C. J., Campbell H. D., Young I. G. Molecular and cellular biology of eosinophil differentiation factor (interleukin-5) and its effects on human and mouse B cells. Immunol Rev. 1988 Feb;102:29–50. doi: 10.1111/j.1600-065x.1988.tb00740.x. [DOI] [PubMed] [Google Scholar]
- Seed B. An LFA-3 cDNA encodes a phospholipid-linked membrane protein homologous to its receptor CD2. 1987 Oct 29-Nov 4Nature. 329(6142):840–842. doi: 10.1038/329840a0. [DOI] [PubMed] [Google Scholar]
- Shanafelt A. B., Kastelein R. A. High affinity ligand binding is not essential for granulocyte-macrophage colony-stimulating factor receptor activation. J Biol Chem. 1992 Dec 15;267(35):25466–25472. [PubMed] [Google Scholar]
- Takaki S., Murata Y., Kitamura T., Miyajima A., Tominaga A., Takatsu K. Reconstitution of the functional receptors for murine and human interleukin 5. J Exp Med. 1993 Jun 1;177(6):1523–1529. doi: 10.1084/jem.177.6.1523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tavernier J., Devos R., Cornelis S., Tuypens T., Van der Heyden J., Fiers W., Plaetinck G. A human high affinity interleukin-5 receptor (IL5R) is composed of an IL5-specific alpha chain and a beta chain shared with the receptor for GM-CSF. Cell. 1991 Sep 20;66(6):1175–1184. doi: 10.1016/0092-8674(91)90040-6. [DOI] [PubMed] [Google Scholar]
- Wang H. M., Ogorochi T., Arai K., Miyajima A. Structure of mouse interleukin 3 (IL-3) binding protein (AIC2A). Amino acid residues critical for IL-3 binding. J Biol Chem. 1992 Jan 15;267(2):979–983. [PubMed] [Google Scholar]
- Watanabe Y., Kitamura T., Hayashida K., Miyajima A. Monoclonal antibody against the common beta subunit (beta c) of the human interleukin-3 (IL-3), IL-5, and granulocyte-macrophage colony-stimulating factor receptors shows upregulation of beta c by IL-1 and tumor necrosis factor-alpha. Blood. 1992 Nov 1;80(9):2215–2220. [PubMed] [Google Scholar]
- de Vos A. M., Ultsch M., Kossiakoff A. A. Human growth hormone and extracellular domain of its receptor: crystal structure of the complex. Science. 1992 Jan 17;255(5042):306–312. doi: 10.1126/science.1549776. [DOI] [PubMed] [Google Scholar]





