Abstract
We have engineered a novel fusion cytokine named GIFT4 derived from GM-CSF and IL-4, and displaying robust gain-of-function immunostimulatory effects on B cells. GIFT4-programmed B cells have a unique identity and potent capacity to elicit a tumoricidal - cell response, thus comprising a novel B cell-based cancer immunotherapeutic approach.
Keywords: B cells, cancer immunotherapy, fusion cytokine, GIFT4
B Cells in Tumors
B cells are a heterogeneous lymphocytic population functionally classified into intrinsically distinct subsets, including antibody-producers, antigen-presenting cells and regulatory B cells. B-cell subsets can elicit adaptive humoral immunity and modulate T-cell and innate immune cell responses, thereby contributing to host immune defense against infections, the pathogenesis of autoimmune diseases, and control of the inflammatory response. In the tumoral microenvironment, B cells constitute a proportion of cancer- infiltrating immune cells. The role of B cells in antitumor immunity remains controversial. On one hand, B cells may augment tumorigenesis and promote tumor growth by suppressing antitumor immune responses.1,2 On the other hand, B cells possess antitumor properties capable of producing tumor-suppressive cytokines and enhancing tumoricidal T-cell responses.3-5 Indeed, the presence of B cells in tertiary follicular structures is correlated with long-term survival of cancer patients.6 These observations suggest that there may well exist meaningful immune interactions between endogenous B cells and malignancy, which may be exploited in a manner to improve cancer outcomes. Here, we summarize our recent epiphany that naïve B cells can be augmented in a manner which allows for immune rejection of cancer.
Programming B Cells by GIFT4 Fusokine
We bioengineered a novel fusion cytokine named GIFT4,7 derived from recombining granulocyte macrophage colony-stimulating factor (GM-CSF) and interleukin 4 (IL-4). This technology builds upon our previous successful development of the “fusokine” platform for the generation of GM-CSF and common γ chain Interleukins Fusion Transgenes (GIFT).8 GIFT fusokines display potent gain-of- function which modulate cellular immune responses.7,8 Since GM-CSF and IL-4 are separately utilized to generate dendritic cells from monocytes as antigen-presenting cells for cellular vaccines in clinical trials, we hypothesized that coupling GM-CSF and IL-4 would deploy a synergistic immunostimulatory effect upon dendritic cells. Unexpectedly, GIFT4 fusokine elicits an entirely novel gain-of- function effect on B cells. We have found that GIFT4 treatment induces the proliferation of murine splenocytes as well as human peripheral blood mononuclear cells (PBMC) in the B cell compartment in vitro. The immunostimulatory function of GIFT4 on B cells was confirmed in mice as it led to splenomegaly with a substantial B cell number increase after intravenous GIFT4 treatment for 6 d.7
In comparison with its parental molecules GM-CSF and IL-4, GIFT4 exhibits marked B-cell bioactivity, inducing the clustering of GM-CSF receptor and IL-4 receptor on the B cell surface and the aggregation of downstream Janus kinase signaling adapters JAK1, JAK2, and JAK3, and consequent pan hyperphosphorylation of signal transducers and activators of transcription STAT1, STAT3, STAT5 and STAT6.7 This unique GIFT4 elicited signaling cascade results in robust B-cell activation and expansion. GIFT4-programmed B cells (termed GIFT4-B cells) have a unique identity and express CD40 (cluster of differentiation 40), CD80 and CD86 co-stimulatory molecules, produce substantial amounts of IL-2, IL-6, GM-CSF, CCL3 (chemokine C-C motif ligand 3) and CCL4, and adhesion molecule CD54, but not IL-10 and interferon γ (IFNγ).7 Thus, GIFT4 can reprogram naïve B cells into novel immune effectors akin to antigen-presenting cells with a distinct secretome, set apart from known B cell subtypes described in the literature.9
Antitumor Property of GIFT4-B Cells
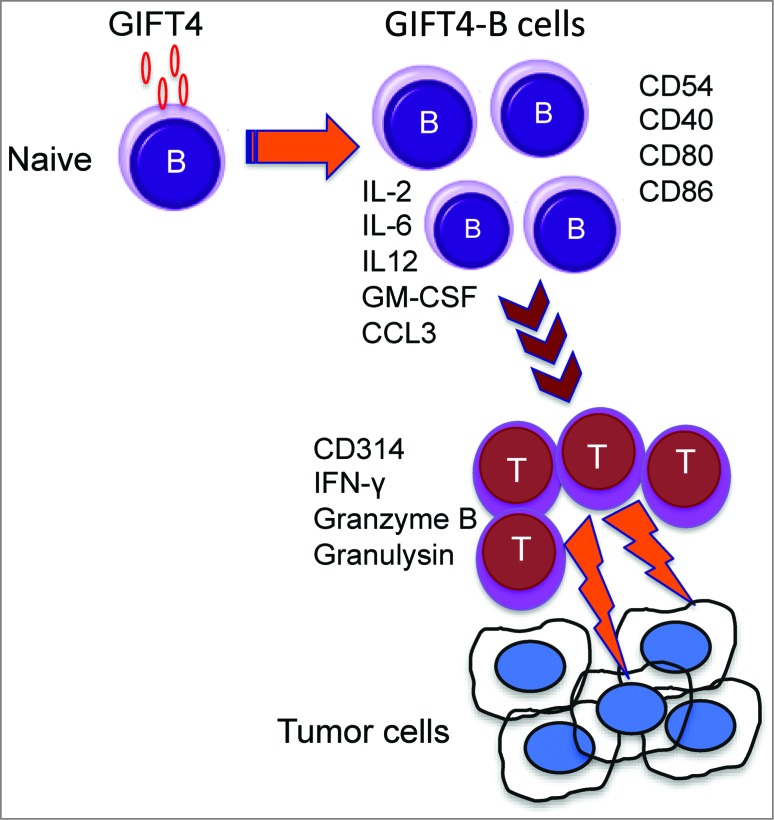
Using immunocompetent syngeneic murine melanoma tumor models, we have shown that B16F0 melanoma cells stably expressing GIFT4 are growth suppressed, an effect absolutely dependent on endogenous B cells. Indeed, we showed that GIFT4 was unable to suppress tumor growth in B-cell deficient μMT mice. However, adoptive transfer of B cells into μMT mice re-established responsiveness to GIFT4 and subsequent antitumor effects.7 In a complementary set of experiments, we found that GIFT4 augmented tumor naïve splenocytes that upon subsequent adoptive transfer displayed direct anti-melanoma activities independently of pre-conditioning with melanoma antigen(s).7 This effect was also dependent on the presence of endogenous T cells suggesting that GIFT4-B cells directly interact with effector T cells as part of their immune augmenting effects. The expression of co-stimulatory molecules and the production of T cell-driving cytokines/chemokines by GIFT4-B cells (Fig. 1) likely contribute to the function GIFT4-B cells, serving as “B helper” effectors in antitumor immunity.
Figure 1.
Anticancer effect of GIFT4-stimulated B cells. Granulocyte macrophage colony stimulating factor (GM-CSF) and common γ chain Interleukins Fusion Transgenes (GIFT) fusokines display immunostimulatory phenotypes. GIFT4 (GM-CSF and IL-4 fusion) stimulation converts naïve B cells into effector cells. GIFT4-programmed B cells (GIFT4-B cells) express co-stimulatory molecules, and secrete T cell-driving cytokines and chemokines, thus priming T cells to become cytotoxic and capable of killing tumor cells.
From a translational perspective, we have successfully generated human GIFT4-B cells from human PBMC isolated from normal subjects or patients with melanoma. We have further defined that human GIFT4-B cells directly prime bystander T cells. T cells primed by GIFT4-B cells express surface tumor-killing molecule CD314 and produce soluble tumor-toxic factors, such as IFNγ, granzyme B and granulysin, permitting specific cytolysis of human melanoma cells in vitro, but not vascular endothelial cells.7 In immune deficient NOD-scidIL2Rγnull mice pre-established with human melanoma tumors, adoptive transfer of human GIFT4-B cells primed T cells significantly inhibited human melanoma growth, further supporting the antitumor property of GIFT4-B cells via their tumor-killing T cells priming capability.7 These data strongly suggest that GIFT4 deploys a B helper function that augments T cells to express effector and innate killer pathways such as CD314.
Clinical Application
With the advent of checkpoint blockade, cancer immunotherapy writ large is undergoing a renaissance. Taking a forward-looking perspective, coupling immune augmentation to checkpoint blockade may allow for improved antitumor efficacy. Adoptive cell therapy as an immune augmentation strategy has also undergone a revolution as exemplified by the use of autologous genetically modified chimeric antigen receptor T cells (CAR-T).10 One can foresee that the coupling of checkpoint blockade and adoptive cell therapy as a strategy for improving cancer outcomes. We have discovered that an autologous B cell-based immunotherapy might serve as an entirely novel means of augmenting the cellular immune response by acting upstream of effector T cells in a B helper-like function. The key discovery has been developing a means to augment B cells in a manner that fully deploys this B cell functionality. We propose that GIFT4-B cells may serve as a means to generate immune augmentation as part of a personalized B cell therapy strategy for cancer therapy in a manner heretofore unrealized.
Disclosure of Potential Conflicts of Interest
No potential conflicts of interest were disclosed
Funding
This work was supported by NIH grant 5R01AI093881 (to J Galipeau) and Emory Winship Robbins Scholar Award and Winship Melanoma Research Fund (to J Deng).
References
- 1.Bodogai M, Lee Chang C, Wejksza K, Lai J, Merino M, Wersto RP, Gress RE, Chan AC, Hesdorffer C, Biragyn A. Anti-CD20 antibody promotes cancer escape via enrichment of tumor-evoked regulatory B cells expressing low levels of CD20 and CD137L. Cancer Res 2013; 73:2127-38; PMID:; http://dx.doi.org/ 10.1158/0008-5472.CAN-12-4184 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Zhang Y, Morgan R, Podack ER, Rosenblatt J. B cell regulation of anti-tumor immune response. Immunol Res 2013; 57:115-24; PMID:; http://dx.doi.org/ 10.1007/s12026-013-8472-1 [DOI] [PubMed] [Google Scholar]
- 3.Penafuerte C, Ng S, Bautista-Lopez N, Birman E, Forner K, Galipeau J. B effector cells activated by a chimeric protein consisting of IL-2 and the ectodomain of TGF-β receptor II induce potent antitumor immunity. Cancer Res 2012; 72:1210-20; PMID:; http://dx.doi.org/ 10.1158/0008-5472.CAN-11-1659 [DOI] [PubMed] [Google Scholar]
- 4.Tomihara K, Shin T, Hurez VJ, Yagita H, Pardoll DM, Zhang B, Curiel TJ, Shin T. Aging- associated B7-DC+ B cells enhance anti-tumor immunity via Th1 and Th17 induction. Aging Cell 2012; 11:128-38; PMID:; http://dx.doi.org/ 10.1111/j.1474-9726.2011.00764.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.DiLillo DJ, Yanaba K, Tedder TF. B cells are required for optimal CD4+ and CD8 +T cell tumor immunity: therapeutic B cell depletion enhances B16 melanoma growth in mice. J Immunol 2010; 184:4006-16; PMID:; http://dx.doi.org/ 10.4049/jimmunol.0903009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Germain C, Gnjatic S, Tamzalit F, Knockaert S, Remark R, Goc J, Lepelley A, Becht E, Katsahian S, Bizouard G, et al. . Presence of B cells in tertiary lymphoid structures is associated with a protective immunity in patients with lung cancer. Am J Respir Crit Care Med 2014; 189:832-44; PMID:; http://dx.doi.org/ 10.1164/rccm.201309-1611OC [DOI] [PubMed] [Google Scholar]
- 7.Deng J, Yuan S, Pennati A, Murphy J, Wu JH, Lawson D, Galipeau J. Engineered fusokine GIFT4 licenses the ability of B cells to trigger a tumoricidal T-cell response. Cancer Res 2014; 74:4133-44; PMID: 24938765; doi: 10.1158/0008-5472.CAN-14-0708 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Rafei M, Hsieh J, Zehntner S, Li M, Forner K, Birman E, Boivin MN, Young YK, Perreault C, Galipeau J. A granulocyte-macrophage colony-stimulating factor and interleukin-15 fusokine induces a regulatory B cell population with immune suppressive properties. Nat Med 2009; 15:1038-45; PMID:; http://dx.doi.org/ 10.1038/nm.2003 [DOI] [PubMed] [Google Scholar]
- 9.Kaminski DA, Wei C, Qian Y, Rosenberg AF, Sanz I. Advances in human B cell phenotypic profiling. Front Immunol 2012; 3:302; PMID:; http://dx.doi.org/ 10.3389/fimmu.2012.00302 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Cheadle EJ, Gornall H, Baldan V, Hanson V, Hawkins RE, Gilham DE. CART cells: driving the road from the laboratory to the clinic. Immunol Rev 2014; 257:91-106; PMID:; http://dx.doi.org/ 10.1111/imr.12126 [DOI] [PubMed] [Google Scholar]