Abstract
During the past decade, considerable evidence has accumulated that non-NMDA glutamate receptors (both AMPA and kainate subtypes) are modulated by the association of the core tetrameric receptor with auxiliary proteins that are integral components of native receptor assemblies. This short review focuses on the effect of two types of auxiliary subunits on the biophysical properties and kinetic behaviour of AMPA and kainate receptors at the level of single receptor molecules. Type I transmembrane AMPA receptor proteins increase the number of AMPA receptor openings that result from a single receptor activation as well as the proportion of openings to conductance levels above 30 pS, resulting in larger peak ensemble currents that decay more slowly and bi–exponentially. Co-expression of Neto1 and 2 with pore-forming kainate receptor subunits also increases the duration of bursts and destabilizes desensitized states, resulting in a rapid component of recovery and clusters of bursts that produce a slow component in desensitization decays. The distinct gating seen in the presence of auxiliary subunits reflects slow switching between gating modes with different single-channel kinetics and open probability. At any given time, the relative proportions of receptors in each gating mode determine both the shape and the amplitude of synaptic currents.
Introduction
By 1990, the cDNAs encoding the four AMPA receptor subunits (GluA1–4) were cloned, touching off a decade of intense investigation of the biophysical properties of recombinant receptors formed by these pore-forming subunits (Dingledine et al. 1999). High-resolution functional studies were further spurred by the steady accumulation of structural data that followed the initial X-ray crystallographic structure of the ligand-binding domain (LBD) of GluA2 (Armstrong et al. 1998). Studies from many groups have contributed to the identification of important structure–function relationships and in total have demonstrated that receptors composed of various combinations of GluA1–4 recapitulate many of the key features of native AMPA receptors (Traynelis et al. 2010). However, in addition to pore-forming subunits that make up the tetrameric core, native AMPA receptors also contain auxiliary subunits that have important effects on receptor trafficking and localization, and which modulate gating behaviour in substantial ways (Jackson & Nicoll, 2011; Yan & Tomita, 2012). In this short review, I focus on one important subfamily of such auxiliary subunits, the type I transmembrane AMPA receptor proteins (TARPs), and their effects on the unitary properties of AMPA receptors. I will also review what is currently known about the modulation of kainate receptor gating by the auxiliary subunits Neto1 and Neto2.
Modulation of AMPA receptor gating by type I TARPs
The first AMPA receptor auxiliary subunit was identified from studies on the spontaneous mutant mouse stargazer and was initially shown to play an essential role in the delivery of AMPA receptors to the cell surface and their targeting to synapses (Hashimoto et al. 1999; Chen et al. 2000; Bredt & Nicoll, 2003). In 2005, however, a flurry of papers demonstrated that this protein, stargazin or γ-2, also had important effects on receptor function, including speeding recovery from desensitization, slowing the decay of currents evoked by short or sustained applications of glutamate (deactivation and desensitization), increasing the apparent efficacy of the AMPA receptor partial agonist kainate, and enhancing steady-state currents and shifting the concentration–response curve for such currents to the left (Priel et al. 2005; Tomita et al. 2005; Turetsky et al. 2005). It was known that γ-2 is one member of a family of related isoforms, which are now referred to as the type I TARPs. Each member of this family, γ-2, γ-3, γ-4 and γ-8, has four predicted transmembrane helices and a large extracellular loop (Ex1) between the first and second transmembrane segments (Tomita et al. 2003). Comparisons of the effect of the four TARPs in patch-clamp experiments demonstrated that they all have qualitatively similar effects on AMPA receptor gating, although γ-4 and γ-8 have larger effects than γ-2 and γ-3, differences that are also evident in experiments on synaptic currents (Cho et al. 2007; Milstein et al. 2007).
Figure 1 illustrates the effects of the four TARP isoforms on receptor responses to a 1 ms pulse of a saturating concentration of glutamate. Each of the TARPs slows the deactivation decay of the population current relative to the current for GluA1 homomeric receptors and also enhances a slow component of deactivation, making the decay of the current clearly bi–exponential. Each TARP also slows the decay of currents during prolonged applications of glutamate, i.e. they all slow receptor desensitization, and desensitization becomes clearly bi–exponential (Cho et al. 2007; Milstein et al. 2007). To compare the slowing of deactivation and desensitization, we calculated weighted time constants (taus) from the bi-exponential fits to the current decays in the two types of fast application experiment (brief vs. sustained glutamate). As shown in Fig. 1F, the four type I TARPs form two subfamilies and they slow deactivation and desensitization to similar extents, a result we have proposed argues that the main effect of TARPs is on activation gating. Some of the effects of type I TARPs on gating are similar to those reported for type II TARPs and the cornichon family of auxiliary subunits (Kato et al. 2007, 2008, 2010; Schwenk et al. 2009; Gill et al. 2011; Coombs et al. 2012; Herring et al. 2013).
Figure 1. Slowing of deactivation by type I TARP isoforms.
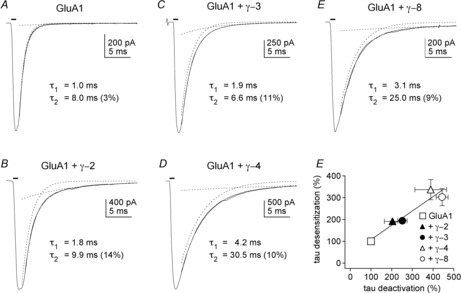
Inward currents elicited by 1 ms applications (bars above current traces) of 10 mm glutamate to outside-out patches transfected with GluA1 (flip splice variant) alone (A) or GluA1 and γ-2, γ-3, γ-4 or γ-8 (B–E). Thin continuous lines show the bi–exponential fits to the decays of the currents. Individual components are shown as dotted lines. The time constants obtained from the fits for each component are indicated, as is the relative amplitude of the slow component of decay (in per cent). All four TARPs slow the deactivation decays and increase the amplitude of the slow component. The time constants of the two components are larger for γ-4 and γ-8 than for γ-2 and γ-3. F, plot of the weighted time constants of deactivation against the corresponding time constants for desensitization. Weighted time constants were calculated from the bi-exponential fits to the decays of 1 ms (deactivation) or 100 ms (desensitization) applications of 10 mm glutamate (n = 4–11 patches per group). The individual values were then expressed as a percentage of the mean value obtained for GluA1 alone. The bars indicate SEM, which in some cases is less than half the symbol size. The linear fit to the data gave a slope of 0.66 and a correlation coefficient of 0.96. Reproduced with permission from Cho et al. (2007, fig. 3).
At the level of single-channel currents, co-expression of γ-2 increases the duration of bursts of receptor openings without increasing the mean length of individual openings (Tomita et al. 2005). Bursts of openings arise from single receptor activations, as typically occur during the spike-like rise in glutamate concentration that follows presynaptic release, and burst duration determines the decay of EPSCs. In addition, co-expression of γ-2 was shown to increase the proportion of time the receptor opens to large conductance levels (Tomita et al. 2005). Native AMPA receptors display up to four subconductance states and the frequency of openings to the larger levels increases with receptor occupancy (Smith & Howe, 2000). The most straightforward interpretation of these results is that unitary conductance increases in a step-wise fashion as the LBDs of individual GluA subunits are occupied and the LBDs close (Rosenmund et al. 1998; Armstrong & Gouaux, 2000; Zhang et al. 2008a). With desensitization intact, recombinant receptors open primarily to the two smallest open levels, I believe because closing of the LBDs of individual subunits (the first step along the activation pathway) slows activation and the receptors typically desensitize before the LBDs of three or four subunits are closed at the same time (Robert & Howe, 2003; Zhang et al. 2008a). By favouring activation, TARPs increase the frequency of large-conductance openings, an effect expected to increase the amplitude of synaptic responses.
The effect of TARPs on unitary receptor properties is illustrated in Fig. 2A, where examples of single-channel currents recorded during sustained applications of 10 mm glutamate are shown (Tomita et al. 2005). Outside-out patches were pulled from cells transfected with GluA4 alone or co–transfected with GluA4 and γ-2. Co–expression of γ-2 resulted in long bursts of openings late in the applications that consisted primarily of openings to the largest two open levels. We rarely if ever see such bursts in the absence of TARPs and co-expression of γ-2 resulted in a significant shift toward large conductance levels when individual receptors were open (Fig. 2B). One interpretation of the greater preponderance of large-conductance openings is that γ-2 increases the efficiency of coupling between LBD closure and channel opening (Jin et al. 2003). Such a mechanism has been proposed for the similar effects of CaMKII to increase the frequency of large-conductance openings through GluA1/GluA2 receptors without altering unitary conductance (Kristensen et al. 2011). Shelley et al. (2012) found that TARPs increase the mean conductance of the different conductance levels. We have not observed this effect, but both groups agree that there are fewer small-conductance and more large-conductance openings with TARPs. Type I TARPs also reduce the inward rectification seen for receptors containing the edited (R) version of GluA2 (Turetsky et al. 2005; Cho et al. 2007; Soto et al. 2007). The TARP-mediated reduction in channel block by intracellular polyamines varies with TARP isoforms and is agonist-specific (Jackson et al. 2011), but appears distinct from effects on gating (A. Plested, personal communication).
Figure 2. Type I TARP modulation of gating at the level of single AMPA receptors.
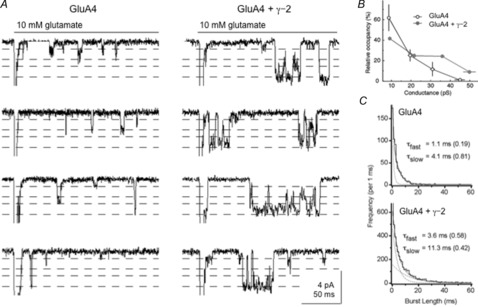
A, unitary currents seen during 200 ms applications of 10 mm glutamate at a holding potential of −100 mV from outside-out patches excised from tsA 201 cells transfected with GluA4 alone (left) or co–transfected with GluA4 and γ-2 (right). The examples shown (low-pass filtered at 2 kHz) were selected from 500 and 1000 applications (left and right, respectively). Analysis of single-channel currents with the SKM algorithm of the QuB software identified four subconductance levels in each patch. The dotted lines show the corresponding current levels for these four open levels. The patches contained several channels and the peak currents (30–40 pA) at the onset of the applications are off the scale. B, mean percentage of time spent at each subconductance level when individual AMPA receptors were open for patches expressing GluA4 alone (open circles, black) and patches co–expressing GluA4 and γ-2 (filled circles, grey). The error bars indicate SEM for the relative occupancies and conductance levels. The data are from three patches analysed for each condition. C, examples of burst-length distributions from a patch expressing GluA4 alone (top, 610 bursts) or a patch expressing GluA4 and γ-2 (bottom, 4464 bursts). Bursts were defined as a series of openings separated by shuttings with durations < 3 ms. The distributions were fitted (continuous line) with two exponential components (dotted lines). The time constants of the two components and their relative areas (in parentheses) are indicated on each panel. The first bin in the GluA4 + γ-2 distribution is off the scale. One burst from the GluA4 patch and 34 bursts from the GluA4 + γ-2 patch had durations greater than 60 ms (off the scale, right). Adapted with permission from Tomita et al. (2005, fig. 5).
Activations of single AMPA receptors during sustained agonist applications consist of bursts of openings that are typically terminated by entry of the receptor into desensitized states. For recombinant receptors, bursts consist largely of rapid transitions between adjacent conductance levels (Zhang et al. 2008a). In addition to increasing the relative proportion of openings to conductance levels above 30 pS, γ-2 and other type I TARPs increase the duration of receptor activation, as indicated by an increase in burst length (Tomita et al. 2005; Shelley et al. 2012). Distributions of burst lengths for GluA4 receptors without and with γ-2 co-expression are shown in Fig. 2C. Co-expression of γ-2 results in a burst-length component that has a time constant similar to the time constants for the slow component of desensitization obtained from bi-exponential fits to the decay of ensemble currents evoked by 100–200 ms applications of 10 mm glutamate. In addition, there is about a three-fold excess of bursts longer than 60 ms, bursts that would be expected to contribute substantially to the larger steady-state current seen with γ-2 co-expression. The longer bursts seen with TARPs are due to more openings per burst, as mean open times were not altered significantly by TARPs (Tomita et al. 2005). In total, our previous single-channel data with γ-2 suggest that an increase in the rate constants for channel opening leads to a greater number of openings per burst before the receptor desensitizes or glutamate dissociates and the consequent increase in burst duration accounts for the slowing of both deactivation and desensitization, as well as the prominent slow component in the clearly bi–exponential decay. Shelley et al. (2012) reported TARP-mediated increases in the duration of open periods, which would also contribute to the slower decays of population currents.
Unitary properties of receptors assembled from GluA_TARP tandems
It was clear from inspection of our co-expression results with GluA4 and γ-2 that applications of glutamate that elicited long, large-conductance bursts tended to occur in ‘runs’ where such events would occur in several consecutive trials followed by many applications where such events were absent. To determine whether this apparent non–randomness was statistically significant we performed Runs tests (Horn et al. 1984; Howe & Ritchie, 1992) on data from patches where we were able to collect hundreds of concentration jumps. The results of this analysis were clearly statistically significant (Zhang et al. 2014); however, the patches analysed all contained more than a single active receptor. As yet, there is no consensus about GluA:TARP stoichiometry, and it does not appear to be fixed (Shi et al. 2009; Kim et al. 2010; Hastie et al. 2013; Herring et al. 2013); thus, one simple explanation for the heterogeneity of kinetic behaviour we observed was that it reflected individual receptors with different GluA:TARP stoichiometries. To avoid this potential confounding factor, we repeated and extended these experiments using tandem receptors where the C terminus of the GluA subunit was fused directly to the N terminus of a type I TARP. We reported previously that several properties of the receptors formed from such tandems (rise times of peak currents, deactivation and desensitization decays, peak open probability, EC50 values for peak currents) were indistinguishable from the corresponding properties measured in GluA:TARP co–expression studies, with the exception of the larger proportion of steady-state current during sustained applications of glutamate (Morimoto-Tomita et al. 2009).
Figure 3 shows 30 consecutive trials from a patch containing just one GluA4_γ–2 tandem receptor (Zhang et al. 2014). This portion of the recording contains nine runs where each run consists of consecutive trials of either low- or high-Popen gating, defined respectively by the absence or presence of bursts of openings greater than 10 ms in duration (designations as low- or high-Popen trials are indicated by 0 or 1 to the left of each record). Note that some of the burst activity during high-Popen trials persisted or occurred after the end of the glutamate application. Such events were seen in most tandem-receptor patches and did not reflect imperfect solution exchange (as assessed by tip potential measurements). In this patch, we identified 64 runs in 229 consecutive trials, which was significantly fewer (P < 0.000001) than the 94 runs predicted from the relative frequencies of low- and high-Popen gating (0.71 and 0.29, respectively).
Figure 3. TARP-associated AMPA receptors switch between low- and high-Popen gating modes on a slow time scale.
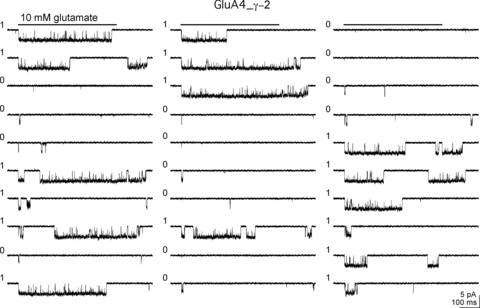
Thirty consecutive records (top to bottom, left to right) from a patch containing one active GluA4_γ-2 tandem receptor. A 500 ms application of 10 mm glutamate (bar above each column of records) was made during each trial and the trials were repeated at 2 s intervals. Each trial was classified as low or high Popen based on the absence or presence of a burst longer than 10 ms. The low and high mode designations are indicated by a 0 or a 1, respectively, to the left of each trace. This portion of the record contained nine runs, where a run was defined as a series of like-designated trials. We identified 64 runs in 229 consecutive trials obtained from this patch, significantly fewer that the 94 runs predicted from the relative frequencies of low- and high-Popen gating (0.712 and 0.288, respectively; P < 0.000001). Reproduced with permission from Zhang et al. (2014).
Our interpretation of these results and similar findings from several patches is that individual receptors switch between distinct gating behaviours or modes on a time scale of seconds or tens of seconds and that the mean dwell times in each mode are longer than the intervals at which the trials were repeated (Zhang et al. 2014). As a result, the probability that a receptor was in the high-Popen gating mode was not constant from trial to trial but was influenced by recent history; and the runs of identical gating modes do not reflect non-stochastic behaviour of the receptors, but rather reflect the slow switching between gating modes and way the data were collected.
The modal behaviour seen at the level of single receptors for the GluA_TARP tandem receptors is reminiscent of behaviour described previously for NMDA receptors composed of GluN1 and GluN2A pore-forming subunits, where slow switching between the two most prevalent modes gives rise to the two components seen in the decay of NMDA receptor ensemble currents and also impacts the amplitude of the population responses (Zhang et al. 2008b). To determine the influence of the gating modes identified for TARP-associated AMPA receptors on the amplitude and kinetics of population responses, we analysed the kinetics of ensemble averages obtained after sorting the single-receptor trials into low- and high-Popen subsets as in Fig. 3.
The low- and high-Popen modes observed at the level of unitary currents correlated well with the fast and slow components in the bi-exponential decay of population currents seen with TARPs (Zhang et al. 2014). Examples of low- and high-Popen trials in another patch containing a single GluA4_γ–2 tandem receptor are shown in Fig. 4A and B. Ensemble currents from low- and high-Popen trials are shown in Fig. 4C and D. The top traces in Fig. 4C and D are the sum of the last five records in A and B (note the smaller low mode peak current) and the bottom traces are the corresponding ensemble averages from all the low- and high-Popen records. Single exponential fits to ensemble averages from low- and high-Popen trials obtained from this patch and other experiments gave mean time constants that agreed closely with values for the fast and slow components in the bi-exponential desensitization decays of large population currents in patches with many receptors (Cho et al. 2007; Milstein et al. 2007). The relative amplitude of the slow component plus the steady-state current from bi-exponential fits to large GluA4_γ–2 population currents was likewise in good agreement with the relative proportion of high-Popen trials for tandem receptors estimated from unitary currents (Zhang et al. 2014). Interestingly, on average, the tandem receptors gated in the low-Popen mode about two-thirds of the time, a mode that was kinetically indistinguishable from behaviour exhibited by receptors formed solely by pore-forming subunits.
Figure 4. TARP-associated modal gating results in ensemble currents with distinct decay kinetics and a unitary current with a high proportion of large-conductance openings.
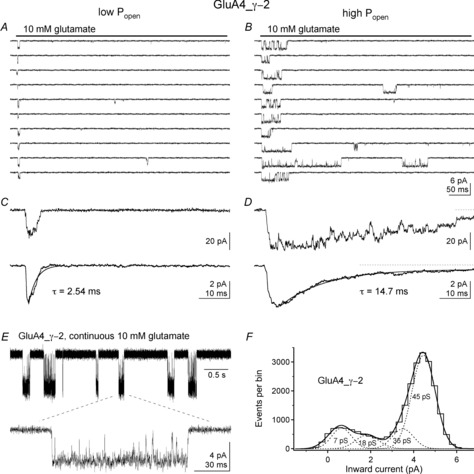
A and B, selected traces (not consecutive) illustrating low-Popen (A) and high-Popen (B) gating during 500 ms applications (bars) of 10 mm glutamate in a patch containing a single GluA4_γ-2 receptor. No double openings were observed in this patch during 300 jumps repeated at 2 s intervals. Runs analysis found 77 runs compared with 126 expected (Z = 6.74, P < 0.000001). Data were low-pass filtered at 3 kHz. C and D, top, the sum of the bottom five traces in A and B on a smaller time scale. Note the larger peak current in D. Bottom, average currents from the complete set of low and high mode traces (210 and 90, respectively). The decay of each ensemble average is adequately fitted by a single exponential component with the indicated time constants. Note the absence of detectable steady-state current in C and the clear presence of steady-state current in D (dotted line indicates the zero current level). E, unitary currents for a GluA4_γ-2 receptor in another patch during part (5 s) of a continuous recording in 10 mm glutamate. Bottom trace shows one long burst on a smaller time scale. Data were low-pass filtered at 3 kHz. F, histogram and Gaussian fits for the amplitude of events obtained from the SKM idealization of the data from 5 min of continuous recording. Four open levels were detected with the indicated conductance levels. Note that the majority of openings are to the 45 pS level. Reproduced with permission from Zhang et al. (2014).
Compared with our previous work on patches from cells co-transfected with GluA4 and γ-2 (Tomita et al. 2005), the GluA4_γ-2 tandem receptors appear to spend more time at large conductance levels (Zhang et al. 2014). Figure 4E shows single-channel currents during continuous glutamate in another patch and a histogram of the amplitude of events from bursts of receptor activity. The four conductance levels seen for GluA4_γ-2 receptors agreed closely with values we reported before for homomeric receptors composed of GluA4 subunits (Tomita et al. 2005), but openings to the two largest levels predominated for the tandem receptors (Zhang et al. 2014). As shown in Fig. 4F, during receptor activations, this GluA4_γ–2 receptor spends most of its time at the largest subconductance level. Shelley et al. (2012) reported that TARPs increase the maximum conductance levels seen for AMPA receptors. We have not observed such effects (Zhang et al. 2014) and the reasons for this apparent disparity remain unresolved.
In summary, type I TARPs modulate AMPA receptors by promoting a distinct gating mode that occurs infrequently but that is long-lived. This behaviour can be accounted for by tiered reaction schemes such as the one in Fig. 5, where the rate constants determining the frequency of transitions between the two qualitatively identical reaction mechanisms (boxed in blue and red) are small relative to the rate constants that determine transitions within each mechanism. The mechanism predicts the slow switching we observe for individual receptors between distinct gating behaviours (TARP uncoupled and TARP coupled). For type 1 TARPs, the boxed mechanisms differ primarily in the rate constants for channel opening and recovery from desensitization, and perhaps also in the stability of the closed LBD/closed pore transition state that precedes both activation and desensitization. These differences account for the effects of type I TARPs on both unitary and ensemble currents listed in Fig. 5. Although the time scale of the switching between modes is slow relative to single EPSCs, the kinetic heterogeneity of the receptor population that results from the different gating modes shapes both the amplitude and the decay kinetics of population responses to rapid applications and may have a significant impact on frequency encoding of information during trains of repetitive stimuli.
Figure 5. Modal gating mechanism.
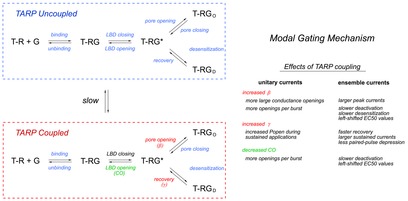
A tiered reaction scheme (left) where, in the presence of TARPs, individual receptors switch on a time scale of seconds between two distinct sets of receptor conformations represented by the simplified kinetic mechanisms boxed in blue and red. The rate constants controlling transitions between discrete states in the top mechanism are indistinguishable from the corresponding rate constants in the absence of TARPs (TARP uncoupled). The TARP-coupled mechanism differs primarily in the rate constants for pore opening and recovery from desensitization. Increases in these rate constants, and perhaps greater stability of the closed LBD/closed pore transition state (T-RG*), result in the changes in unitary and ensemble currents summarized on the right side of the figure. TARP coupling results in larger and more slowly decaying synaptic currents and reduces the blunting of synaptic responses due to desensitization during trains of repetitive stimuli.
Modulation of kainate receptor gating by the auxiliary subunits Neto1 and Neto2
Kainate receptors also have auxiliary subunits (Copits & Swanson, 2012; Tomita & Castillo, 2012; Yan & Tomita, 2012). The proteins neuropilin tolloid-like 1 and 2 (Neto1, Neto2) are single transmembrane subunits that modulate the gating kinetics of recombinant and native kainate receptors (Zhang et al. 2009; Straub et al. 2011a,b2011b; Tang et al. 2011; Fisher & Mott, 2012, 2013).
Figure 6 illustrates the effect of Neto2 co-expression on ensemble and unitary currents through GluK2 receptors (Zhang et al. 2009). Co-expression of Neto2 with GluK2 slowed the fast component of desensitization about two-fold and resulted in a prominent slow component that decayed with a time constant on the order of 100 ms. Deactivation was slowed to a lesser extent (Fig. 6B, C), and this slowing probably indicates that desensitization contributes to the decay of population responses to brief pulses of glutamate (Zhang et al. 2009). Other studies have shown that Neto1 and Neto2 alter desensitization of both homomeric and heteromeric recombinant kainate receptors (Copits et al. 2011; Straub et al. 2011b; Fisher & Mott, 2013). In addition, co-expression of Neto2 resulted in a proportion of the receptors that recover from desensitization much faster than the rate of recovery of GluK2 homomeric receptors (Fig. 6D). A similar speeding of recovery has also been reported for Neto2 co-expression with other GluK subunits (Straub et al. 2011b), as well as for Neto 1 (Fisher & Mott, 2013), and recovery from desensitization of GluK2 receptors is likewise hastened by association with the scaffolding protein PSD-95 (Bowie et al. 2003). The modulation of kainate receptor kinetics by Neto1 has been shown to prolong the decay of synaptic currents (Straub et al. 2011a).
Figure 6. Neto2 alters kainate receptor kinetics and peak open probability.
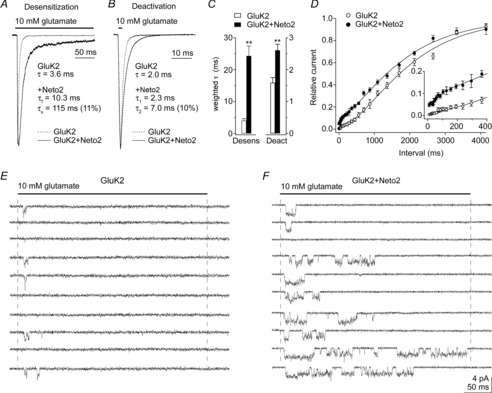
A and B, responses to 10 mm glutamate in outside-out patches from tsA201 cells transfected with GluK2 alone or GluK2 and Neto2. Glutamate was applied for 200 ms (A) or 1 ms (B). The time constants (relative amplitude) are from mono- or bi-exponential fits to the decays of the currents. C, the fits were used to calculate weighted time constants of desensitization and deactivation. Co–expression of Neto2 with GluK2 (30:1 ratio) slowed both desensitization and deactivation significantly (n = 6–9). Data are given as mean ± SEM; **P < 0.01. D, recovery from desensitization for GluK2 alone (n = 5) and GluK2 and Neto2 (n = 6). Results were obtained from conventional two-pulse protocols. The ratio of the amplitude of the peak currents (P2/P1) is plotted as a function of the inter-pulse interval. Neto2 markedly sped recovery at short intervals, but did not alter the slow component of recovery. E and F, examples of single-channel currents (low-pass filtered at 3 kHz) evoked by ten consecutive applications of 10 mm glutamate in patches containing four GluK2 (E) or three GluK2+Neto2 channels (F). The receptors displayed three conductance substates and most of the inward currents reflect the activity of a single receptor. Co-expression of Neto2 increased the probability of channel opening and greatly increased the duration of channel activity in the portion of the record shown from the patch in F.
At the level of unitary currents, we found that co-expression of Neto2 with GluK2 increased the duration of bursts of openings about two-fold, which accounted well for the slowing of the fast component of desensitization (Zhang et al. 2009). What was more striking, however, was that co-expression of Neto2 resulted in trials in concentration-jump experiments where burst activity persisted long into the sustained applications. Figure 6 shows examples of ten consecutive trials in patches containing 3 or 4 GluK2 receptors without (E) and with (F) co-expression of Neto2. The long clusters of bursts seen in the presence of Neto2 appear to reflect the faster component of recovery from desensitization seen in two-pulse protocols with patches that contained hundreds of receptors (Fig. 6D).
Although trials with long clusters of bursts occurred infrequently in experiments where we collected hundreds of concentration jumps, they were often bunched in series of consecutive trials and Runs analysis showed that this deviation from binomial expectations was very statistically significant (Zhang et al. 2014). The results indicated that individual Neto2-associated Gluk2 receptors switch on a slow time scale between gating modes that differ in the rate at which they desensitize and the rate at which they recover from desensitization. Similar to our results for TARP modulation of AMPA receptors, the more frequent of these modes is indistinguishable from the gating behaviour of GluK2 receptors without Neto2, whereas the other gating mode results in substantially higher Popen activity during sustained applications of saturating concentrations of glutamate (Zhang et al. 2014). The decreased rate of entry into desensitized states displayed by the high-Popen mode increases the probability that the receptors open early in the application and prolongs the length of individual activations, both of which lead to a significant (about three-fold) increase in open probability at the peak of ensemble currents (Zhang et al. 2009). The much faster recovery associated with the high-Popen mode leads to clusters of bursts that persist well into long jumps (Fig. 6F), and it is these clusters that give rise to the very slow component of desensitization seen in population responses (Fig. 6A). The effect of Neto1 and Neto2 on kainate receptor kinetics can be accounted for by tiered reaction mechanisms similar to those shown for TARPs in Fig. 5, where the corresponding uncoupled and coupled mechanisms would differ primarily in the rate constants in and out of desensitized states.
Although GluK2 receptors display multiple open levels, Neto2 did not alter the conductance levels or the relative frequency of openings to the three conductance substates (Zhang et al. 2009). However, similar to the effect of TARPs on AMPA receptors (Soto et al. 2007), both Neto1 and Neto2 reduce channel block of GluK2 receptors by intracellular polyamines (Fisher & Mott, 2012). The effect on polyamine block was localized to the intracellular C terminus of the auxiliary subunits, whereas the effects on desensitization depend on sequence elements in the extracellular N-terminal domain (Fisher & Mott, 2012).
Conclusions and future directions
Type I TARPs and Neto1 and Neto2 alter the unitary properties of AMPA and kainate receptors, respectively. Our recent analysis of single-channel activity evoked by repeated rapid applications of saturating concentrations of glutamate indicates that the two families of auxiliary subunits exert their effects by promoting gating behaviours distinct from those seen for receptors composed of pore-forming subunits alone, behaviours that occur relatively infrequently but that persist for seconds or tens of seconds. The slow switching is qualitatively similar to the modal gating described for NMDA receptors in the absence of auxiliary subunits (Maki et al. 2012; Murthy et al. 2012; Popescu, 2012; Vance et al. 2013), although the mechanisms that give rise to the distinct modes differ. For example, whereas modal gating of GluN1/GluN2A receptors arises mostly from differences in the stability of open states (Popescu & Auerbach, 2003; Popescu, 2012), the higher Popen modes seen in the presence of auxiliary subunits primarily reflect alterations in the activation pathway for AMPA receptors and in the stability of the desensitized state for kainate receptors (Zhang et al. 2014). Although open probability is increased for all three subtypes of iGluRs, this mainly reflects longer openings for NMDA receptors, more openings per single receptor activation for AMPA receptors and more frequent escapes from desensitization for kainate receptors, suggesting the mechanisms that give rise to these behaviours are structurally distinct. Similar to NMDA receptors, however, the presence of distinct gating modes for AMPA and kainate receptors impacts the peak amplitude of ensemble currents and accounts for the bi-exponential nature of the decay of such currents (Popescu et al. 2004; Zhang et al. 2008b; Vance et al. 2013; Zhang et al. 2014). Interestingly, the slow component of decay that arises from Neto-promoted modal gating of GluK2 receptors is similar to results reported for GluK2/GluK5 heteromeric receptors (Barberis et al. 2008), where it was also related to destabilization of desensitization, suggesting the underlying mechanisms may be the same in this case.
The slow and infrequent switching between gating modes makes it unlikely that individual receptors switch modes during a single synaptic event. However, at any given moment, modal gating gives rise to populations of native glutamate receptors that are kinetically heterogeneous and the proportion of receptors in each gating mode at the moment of glutamate release shapes the amplitude and kinetics of synaptic currents. In addition, the effects of both families of auxiliary subunits to speed recovery from desensitization (Priel et al. 2005; Zhang et al. 2009; Straub et al. 2011b) would be expected to impact postsynaptic responses to repeated stimuli applied at brief intervals and influence the frequency dependence of synaptic transmission (Cho et al. 2007; Fisher & Mott, 2013). Recent work also suggests that repetitive stimulation results in the accumulation of TARP-associated AMPA receptors in ‘superactive’ states that resemble the high-Popen mode described in single channel records (Andrew Plested, personal communication), suggesting TARP-modulation of modal gating is dynamic and may provide a receptor-mediated mechanism for short-term synaptic plasticity.
Modal gating in the absence of auxiliary subunits has been reported for AMPA receptors during depolarizing jumps (Prieto & Wollmuth, 2010) or under steady-state conditions in the presence of cyclothiazide (Poon et al. 2010, 2011). How the distinct gating behaviours seen in the presence of TARPs (Tomita et al. 2005; Shelley et al. 2012; Zhang et al. 2014) relate to these other results is at present unclear. The effects of type I TARPs on activation gating appear to be primarily mediated by the Ex1 domain (Tomita et al. 2005; Cho et al. 2007; Milstein et al. 2007), but other regions have also been implicated (Milstein & Nicoll, 2009), and the sequence elements responsible for TARP-mediated speeding of recovery remain unclear. While it appears that the extracellular LDLa domain of Neto1 and Neto2 is important for their effect to modulate receptor gating (Fisher & Mott, 2012), the regions of GluK subunits important for interactions with the auxiliary subunits are unidentified, which is also true for the GluA regions involved in modulation by type I TARPs. Should the protein–protein interfaces important for functional modulation be identified, these interfaces may be an interesting target for drugs. Structural data would certainly be welcome. The slow switching between gating modes we have found (Zhang et al. 2014) implies not only that the distinct gating modes arise from different sets of protein conformations but that the transition energy barriers between the different conformational sets are substantial. This may make structural readouts of these conformations easier to capture and raises the possibility that the different kinetics associated with each mode may reflect differences in multiple rate constants (Howe, 2013; MacLean, 2013).
Acknowledgments
This work was supported by USPHS grants GM-058926, NS-047712 and NS-057725.
Glossary
- LBD
ligand-binding domain
- TARP
transmembrane AMPA receptor protein
Biography
James R. Howe received a B.A. in Physiology (1980) and a Ph.D. (1983) in Pharmacology from the University of Minnesota. After postdoctoral work in Munich and London, he moved to the Department of Pharmacology at Yale University School of Medicine in 1987, where he currently holds the rank of Professor. He has worked on ionotropic glutamate receptors and their role in synaptic transmission since 1986.
Additional information
Competing interests
None declared.
Funding
None declared.
References
- Armstrong N. Gouaux E. Mechanisms for activation and antagonism of an AMPA-sensitive glutamate receptor: crystal structures of the GluR2 ligand binding core. Neuron. 2000;28:165–181. doi: 10.1016/s0896-6273(00)00094-5. [DOI] [PubMed] [Google Scholar]
- Armstrong N, Sun Y, Chen GQ. Gouaux E. Structure of a glutamate-receptor ligand-binding core in complex with kainate. Nature. 1998;395:913–917. doi: 10.1038/27692. [DOI] [PubMed] [Google Scholar]
- Barberis A, Sachidhanandam S. Mulle C. GluR6/KA2 kainate receptors mediate slow-deactivating currents. J Neurosci. 2008;28:6402–6406. doi: 10.1523/JNEUROSCI.1204-08.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bowie D, Garcia EP, Marshall J, Traynelis SF. Lange GD. Allosteric regulation and spatial distribution of kainate receptors bound to ancillary proteins. J Physiol. 2003;547:373–385. doi: 10.1113/jphysiol.2002.033076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bredt DS. Nicoll RA. AMPA receptor trafficking at excitatory synapses. Neuron. 2003;40:361–379. doi: 10.1016/s0896-6273(03)00640-8. [DOI] [PubMed] [Google Scholar]
- Chen L, Chetkovich DM, Petralia RS, Sweeney NT, Kawasaki Y, Wenthold RJ, Bredt DS. Nicoll RA. Stargazin regulates synaptic targeting of AMPA receptors by two distinct mechanisms. Nature. 2000;408:936–943. doi: 10.1038/35050030. [DOI] [PubMed] [Google Scholar]
- Cho CH, St-Gelais F, Zhang W, Tomita S. Howe JR. Two families of TARP isoforms that have distinct effects on the kinetic properties of AMPA receptors and synaptic currents. Neuron. 2007;55:890–904. doi: 10.1016/j.neuron.2007.08.024. [DOI] [PubMed] [Google Scholar]
- Coombs ID, Soto D, Zonouzi M, Renzi M, Shelley C, Farrant M. Cull-Candy SG. Cornichons modify channel properties of recombinant and glial AMPA receptors. J Neurosci. 2012;32:9796–9804. doi: 10.1523/JNEUROSCI.0345-12.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Copits BA, Robbins JS, Frausto S. Swanson GT. Synaptic targeting and functional modulation of GluK1 kainate receptors by the auxiliary neuropilin and tolloid-like (NETO) proteins. J Neurosci. 2011;31:7334–7340. doi: 10.1523/JNEUROSCI.0100-11.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Copits BA. Swanson GT. Dancing partners at the synapse: auxiliary subunits that shape kainate receptor function. Nat Rev Neurosci. 2012;13:675–686. doi: 10.1038/nrn3335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dingledine R, Borges K, Bowie D. Traynelis SF. The glutamate receptor ion channels. Pharmacol Rev. 1999;51:7–61. [PubMed] [Google Scholar]
- Fisher JL. Mott DD. The auxiliary subunits Neto1 and Neto2 reduce voltage-dependent inhibition of recombinant kainate receptors. J Neurosci. 2012;32:12928–12933. doi: 10.1523/JNEUROSCI.2211-12.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fisher JL. Mott DD. Modulation of homomeric and heteromeric kainate receptors by the auxiliary subunit Neto1. J Physiol. 2013;591:4711–4724. doi: 10.1113/jphysiol.2013.256776. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gill MB, Kato AS, Roberts MF, Yu H, Wang H, Tomita S. Bredt DS. Cornichon-2 modulates AMPA receptor-transmembrane AMPA receptor regulatory protein assembly to dictate gating and pharmacology. J Neurosci. 2011;31:6928–6938. doi: 10.1523/JNEUROSCI.6271-10.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hashimoto K, Fukaya M, Qiao X, Sakimura K, Watanabe M. Kano M. Impairment of AMPA receptor function in cerebellar granule cells of ataxic mutant mouse stargazer. J Neurosci. 1999;19:6027–6036. doi: 10.1523/JNEUROSCI.19-14-06027.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hastie P, Ulbrich MH, Wang HL, Arant RJ, Lau AG, Zhang Z, Isacoff EY. Chen L. AMPA receptor/TARP stoichiometry visualized by single-molecule subunit counting. Proc Natl Acad Sci U S A. 2013;110:5163–5168. doi: 10.1073/pnas.1218765110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Herring BE, Shi Y, Suh YH, Zheng CY, Blankenship SM, Roche KW. Nicoll RA. Cornichon proteins determine the subunit composition of synaptic AMPA receptors. Neuron. 2013;77:1083–1096. doi: 10.1016/j.neuron.2013.01.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Horn R, Vandenberg CA. Lange K. Statistical analysis of single sodium channels. Effects of N-bromoacetamide. Biophys J. 1984;45:323–335. doi: 10.1016/S0006-3495(84)84158-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Howe JR. CrossTalk proposal: TARPs modulate AMPA receptor gating transitions. J Physiol. 2013;591:1581–1583. doi: 10.1113/jphysiol.2012.247486. discussion 1589. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Howe JR. Ritchie JM. Multiple kinetic components of sodium channel inactivation in rabbit Schwann cells. J Physiol. 1992;455:529–566. doi: 10.1113/jphysiol.1992.sp019315. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jackson AC, Milstein AD, Soto D, Farrant M, Cull-Candy SG. Nicoll RA. Probing TARP modulation of AMPA receptor conductance with polyamine toxins. J Neurosci. 2011;31:7511–7520. doi: 10.1523/JNEUROSCI.6688-10.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jackson AC. Nicoll RA. The expanding social network of ionotropic glutamate receptors: TARPs and other transmembrane auxiliary subunits. Neuron. 2011;70:178–199. doi: 10.1016/j.neuron.2011.04.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jin R, Banke TG, Mayer ML, Traynelis SF. Gouaux E. Structural basis for partial agonist action at ionotropic glutamate receptors. Nat Neurosci. 2003;6:803–810. doi: 10.1038/nn1091. [DOI] [PubMed] [Google Scholar]
- Kato AS, Gill MB, Ho MT, Yu H, Tu Y, Siuda ER, Wang H, Qian YW, Nisenbaum ES, Tomita S. Bredt DS. Hippocampal AMPA receptor gating controlled by both TARP and cornichon proteins. Neuron. 2010;68:1082–1096. doi: 10.1016/j.neuron.2010.11.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kato AS, Siuda ER, Nisenbaum ES. Bredt DS. AMPA receptor subunit-specific regulation by a distinct family of type II TARPs. Neuron. 2008;59:986–996. doi: 10.1016/j.neuron.2008.07.034. [DOI] [PubMed] [Google Scholar]
- Kato AS, Zhou W, Milstein AD, Knierman MD, Siuda ER, Dotzlaf JE, Yu H, Hale JE, Nisenbaum ES, Nicoll RA. Bredt DS. New transmembrane AMPA receptor regulatory protein isoform, γ-7, differentially regulates AMPA receptors. J Neurosci. 2007;27:4969–4977. doi: 10.1523/JNEUROSCI.5561-06.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim KS, Yan D. Tomita S. Assembly and stoichiometry of the AMPA receptor and transmembrane AMPA receptor regulatory protein complex. J Neurosci. 2010;30:1064–1072. doi: 10.1523/JNEUROSCI.3909-09.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kristensen AS, Jenkins MA, Banke TG, Schousboe A, Makino Y, Johnson RC, Huganir R. Traynelis SF. Mechanism of Ca2+/calmodulin-dependent kinase II regulation of AMPA receptor gating. Nat Neurosci. 2011;14:727–735. doi: 10.1038/nn.2804. [DOI] [PMC free article] [PubMed] [Google Scholar]
- MacLean DM. CrossTalk opposing view: TARPs modulate AMPA receptor conformations before the gating transitions. J Physiol. 2013;591:1585–1586. doi: 10.1113/jphysiol.2012.247726. discussion 1587. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maki BA, Aman TK, Amico-Ruvio SA, Kussius CL. Popescu GK. C-terminal domains of N-methyl-d-aspartic acid receptor modulate unitary channel conductance and gating. J Biol Chem. 2012;287:36071–36080. doi: 10.1074/jbc.M112.390013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Milstein AD. Nicoll RA. TARP modulation of synaptic AMPA receptor trafficking and gating depends on multiple intracellular domains. Proc Natl Acad Sci U S A. 2009;106:11348–11351. doi: 10.1073/pnas.0905570106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Milstein AD, Zhou W, Karimzadegan S, Bredt DS. Nicoll RA. TARP subtypes differentially and dose-dependently control synaptic AMPA receptor gating. Neuron. 2007;55:905–918. doi: 10.1016/j.neuron.2007.08.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morimoto-Tomita M, Zhang W, Straub C, Cho CH, Kim KS, Howe JR. Tomita S. Autoinactivation of neuronal AMPA receptors via glutamate-regulated TARP interaction. Neuron. 2009;61:101–112. doi: 10.1016/j.neuron.2008.11.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Murthy SE, Shogan T, Page JC, Kasperek EM. Popescu GK. Probing the activation sequence of NMDA receptors with lurcher mutations. J Gen Physiol. 2012;140:267–277. doi: 10.1085/jgp.201210786. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Poon K, Ahmed AH, Nowak LM. Oswald RE. Mechanisms of modal activation of GluA3 receptors. Mol Pharmacol. 2011;80:49–59. doi: 10.1124/mol.111.071688. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Poon K, Nowak LM. Oswald RE. Characterizing single-channel behavior of GluA3 receptors. Biophys J. 2010;99:1437–1446. doi: 10.1016/j.bpj.2010.06.058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Popescu G. Auerbach A. Modal gating of NMDA receptors and the shape of their synaptic response. Nat Neurosci. 2003;6:476–483. doi: 10.1038/nn1044. [DOI] [PubMed] [Google Scholar]
- Popescu G, Robert A, Howe JR. Auerbach A. Reaction mechanism determines NMDA receptor response to repetitive stimulation. Nature. 2004;430:790–793. doi: 10.1038/nature02775. [DOI] [PubMed] [Google Scholar]
- Popescu GK. Modes of glutamate receptor gating. J Physiol. 2012;590:73–91. doi: 10.1113/jphysiol.2011.223750. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Priel A, Kolleker A, Ayalon G, Gillor M, Osten P. Stern-Bach Y. Stargazin reduces desensitization and slows deactivation of the AMPA-type glutamate receptors. J Neurosci. 2005;25:2682–2686. doi: 10.1523/JNEUROSCI.4834-04.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Prieto ML. Wollmuth LP. Gating modes in AMPA receptors. J Neurosci. 2010;30:4449–4459. doi: 10.1523/JNEUROSCI.5613-09.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Robert A. Howe JR. How AMPA receptor desensitization depends on receptor occupancy. J Neurosci. 2003;23:847–858. doi: 10.1523/JNEUROSCI.23-03-00847.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rosenmund C, Stern-Bach Y. Stevens CF. The tetrameric structure of a glutamate receptor channel. Science. 1998;280:1596–1599. doi: 10.1126/science.280.5369.1596. [DOI] [PubMed] [Google Scholar]
- Schwenk J, Harmel N, Zolles G, Bildl W, Kulik A, Heimrich B, Chisaka O, Jonas P, Schulte U, Fakler B. Klocker N. Functional proteomics identify cornichon proteins as auxiliary subunits of AMPA receptors. Science. 2009;323:1313–1319. doi: 10.1126/science.1167852. [DOI] [PubMed] [Google Scholar]
- Shelley C, Farrant M. Cull-Candy SG. TARP-associated AMPA receptors display an increased maximum channel conductance and multiple kinetically distinct open states. J Physiol. 2012;590:5723–5738. doi: 10.1113/jphysiol.2012.238006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shi Y, Lu W, Milstein AD. Nicoll RA. The stoichiometry of AMPA receptors and TARPs varies by neuronal cell type. Neuron. 2009;62:633–640. doi: 10.1016/j.neuron.2009.05.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith TC. Howe JR. Concentration-dependent substate behavior of native AMPA receptors. Nat Neurosci. 2000;3:992–997. doi: 10.1038/79931. [DOI] [PubMed] [Google Scholar]
- Soto D, Coombs ID, Kelly L, Farrant M. Cull-Candy SG. Stargazin attenuates intracellular polyamine block of calcium-permeable AMPA receptors. Nat Neurosci. 2007;10:1260–1267. doi: 10.1038/nn1966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Straub C, Hunt DL, Yamasaki M, Kim KS, Watanabe M, Castillo PE. Tomita S. Distinct functions of kainate receptors in the brain are determined by the auxiliary subunit Neto1. Nat Neurosci. 2011a;14:866–873. doi: 10.1038/nn.2837. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Straub C, Zhang W. Howe JR. Neto2 modulation of kainate receptors with different subunit compositions. J Neurosci. 2011b;31:8078–8082. doi: 10.1523/JNEUROSCI.0024-11.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tang M, Pelkey KA, Ng D, Ivakine E, McBain CJ, Salter MW. McInnes RR. Neto1 is an auxiliary subunit of native synaptic kainate receptors. J Neurosci. 2011;31:10009–10018. doi: 10.1523/JNEUROSCI.6617-10.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tomita S, Adesnik H, Sekiguchi M, Zhang W, Wada K, Howe JR, Nicoll RA. Bredt DS. Stargazin modulates AMPA receptor gating and trafficking by distinct domains. Nature. 2005;435:1052–1058. doi: 10.1038/nature03624. [DOI] [PubMed] [Google Scholar]
- Tomita S. Castillo PE. Neto1 and Neto2: auxiliary subunits that determine key properties of native kainate receptors. J Physiol. 2012;590:2217–2223. doi: 10.1113/jphysiol.2011.221101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tomita S, Chen L, Kawasaki Y, Petralia RS, Wenthold RJ, Nicoll RA. Bredt DS. Functional studies and distribution define a family of transmembrane AMPA receptor regulatory proteins. J Cell Biol. 2003;161:805–816. doi: 10.1083/jcb.200212116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Traynelis SF, Wollmuth LP, McBain CJ, Menniti FS, Vance KM, Ogden KK, Hansen KB, Yuan H, Myers SJ. Dingledine R. Glutamate receptor ion channels: structure, regulation, and function. Pharmacol Rev. 2010;62:405–496. doi: 10.1124/pr.109.002451. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Turetsky D, Garringer E. Patneau DK. Stargazin modulates native AMPA receptor functional properties by two distinct mechanisms. J Neurosci. 2005;25:7438–7448. doi: 10.1523/JNEUROSCI.1108-05.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vance KM, Hansen KB. Traynelis SF. Modal gating of GluN1/GluN2D NMDA receptors. Neuropharmacology. 2013;71:184–190. doi: 10.1016/j.neuropharm.2013.03.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yan D. Tomita S. Defined criteria for auxiliary subunits of glutamate receptors. J Physiol. 2012;590:21–31. doi: 10.1113/jphysiol.2011.213868. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang W, Cho Y, Lolis E. Howe JR. Structural and single-channel results indicate that the rates of ligand binding domain closing and opening directly impact AMPA receptor gating. J Neurosci. 2008a;28:932–943. doi: 10.1523/JNEUROSCI.3309-07.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang W, Howe JR. Popescu GK. Distinct gating modes determine the biphasic relaxation of NMDA receptor currents. Nat Neurosci. 2008b;11:1373–1375. doi: 10.1038/nn.2214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang W, Devi SP, Tomita S. Howe JR. Auxiliary proteins promote modal gating of AMPA- and kainate-type glutamate receptors. Eur J Neurosci. 2014;39:1138–1147. doi: 10.1111/ejn.12519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang W, St-Gelais F, Grabner CP, Trinidad JC, Sumioka A, Morimoto-Tomita M, Kim KS, Straub C, Burlingame AL, Howe JR. Tomita S. A transmembrane accessory subunit that modulates kainate-type glutamate receptors. Neuron. 2009;61:385–396. doi: 10.1016/j.neuron.2008.12.014. [DOI] [PMC free article] [PubMed] [Google Scholar]


