Abstract
Background
We aimed to examine the association between recipient race/ethnicity and sex, donor liver quality, and liver transplant graft survival.
Methods
Adult non-Status 1 liver recipients transplanted between 03/01/2002 and 12/31/2008 were identified using Scientific Registry of Transplant Recipients data. The factors of interest were recipient race/ethnicity and sex. Donor risk index (DRI) was used as a donor quality measure. Logistic regression was used to assess the association between race/ethnicity and sex in relation to the transplantation of low quality (high DRI) or high quality (low DRI) livers. Cox regression was used to assess the association between race/ethnicity and sex and liver graft failure risk, accounting for DRI.
Results
Hispanics were 21% more likely to receive low quality grafts compared to Whites (OR=1.21; p=0.002). Women had greater odds of receiving a low quality graft compared to men (OR=1.24; p<0.0001). Despite adjustment for donor quality, African American recipients still had higher graft failure rates compared to Whites (HR=1.28; p<0.001). Hispanics (HR=0.89; p=0.023) had significantly lower graft failure rates compared to Whites despite higher odds of receiving a higher DRI graft. Using an interaction model of DRI and race/ethnicity, we found that the impact of DRI on graft failure rates was significantly reduced for African-Americans relative to Whites (p=0.02).
Conclusions
This study shows that while liver graft quality differed significantly by recipient race/ethnicity and sex, donor selection practices do not appear to be the dominant factor responsible for worse liver transplant outcomes for minority recipients.
Keywords: racial and ethnic disparities, liver transplantation, liver graft survival, donor risk, clinical outcomes
Introduction
In the U.S., substantial sex-based and racial/ethnic disparities in access to liver transplant from the waiting list have been observed (1–3). However, after patients are registered on a waiting list, the extent to which these disparities pervade the liver transplant process and affect outcomes is unclear (4). Clinical achievements in liver transplant care have improved graft survival to an all-time high, nearly 70% at 5 years (5). This achievement stands at odds with reports of inequities in liver transplant care in the MELD era. Several reports demonstrate inferior outcomes among female and minority recipients (4, 6–10). The Scientific Registry of Transplant Recipients (SRTR) reports that graft survival is lower among racial/ethnic minorities compared to White recipients (11, 12). Ananthakrishnan et al also found that African Americans have higher graft failure and mortality rates after liver transplant compared to Whites in the MELD era (13). Other studies also support the premise that minorities do worse after liver transplant than their White counterparts (14, 15).
These studies underscore an unfortunate reality for female and minority liver transplant recipients, but the mechanism behind this disparity remains unclear. Many factors can contribute to variation in transplant outcomes for minorities, including recipient health status, liver donor graft quality, center practices, and barriers involved in the processes of care (12, 16). A review of the literature reveals that existing studies of race/ethnicity and sex effects on liver graft failure have not included comprehensive covariate adjustment and testing of interactions. It is therefore premature to discuss potential solutions for outcome disparities without a more sophisticated understanding of its details. A more complete evaluation of variation in liver transplant outcomes would better inform clinical strategies and policymaking to ensure equity.
One potentially significant contributor in the liver transplant process is the transplant provider. Obviously, the transplant community and the public at large would find conscious prejudice in transplant clinical decision-making morally repugnant. However, the Institute of Medicine study of U.S. health care disparities suggests that patient race/ethnicity may affect provider decision-making in subtle ways (16). In the liver transplant context, surgeons select donor liver grafts for their patients, and several factors, including donor age, clinical status, and mechanism of death, contribute to this decision. The provider uses expert clinical judgment in a complex clinical situation to either accept or decline an organ for a given recipient. The process is prone to potential bias, and it is unclear how donor selection practices affect disparities in liver transplantation.
Our study had two main objectives. The first was to quantify differences in the donor risk index (DRI) distribution by race and sex among deceased-donor liver transplant recipients in the United States. The second objective was to determine the extent to which differences in DRI account for differences in graft failure rates.
Results
The study cohort was comprised of n=19,249 liver transplant recipients over the study period. Baseline characteristics of these liver transplant recipients are shown in Table 1. Liver transplant recipients in each racial/ethnic group were of similar age, with overall population mean of 52.5 yrs. Approximately one-third of all recipients were female, with the highest proportion in the African American population (38%). African Americans had the highest proportion of recipients with hepatitis C (49%) as an indication for transplant, and Asians had the highest proportion of non-cholestatic liver disease (47%). Recipient height and weight were similar across groups, with Asian recipients being smaller, and having the lowest BMI. ABO blood types varied greatly, and more than half of those classified as Other race/ethnicity were type O. MELD scores at transplant and rates of co-morbidities were similar across the cohort. Hospitalization rates at wait-listing were twofold higher in the Asian population compared to Whites.
Table 1.
Clinical Characteristics of Deceased Donor Liver Transplant Recipients by Race/Ethnicity in Study Cohort (n=19,249) *
| Variable | White (n=14,427) |
African- American (n=1,727) |
Hispanic (n=2,395) |
Asian (n=521) |
Other (n=179) |
|
|---|---|---|---|---|---|---|
| Age at transplant (yrs) (SD) | 53.0 (9.1) |
49.6 (10.2) |
51.6 (9.3) |
52.3 (10.7) |
50.8 (9.1) |
|
| Sex (Female) (%) | 30% | 38% | 34% | 35% | 37% | |
| Diagnosis | ||||||
| Cholestatic (%) | 10% | 11% | 5% | 6% | 9% | |
| Non-Cholestatic (%) | 40% | 26% | 41% | 47% | 44% | |
| Hepatitis C (%) | 37% | 49% | 43% | 26% | 28% | |
| Acute Hepatic Necrosis (%) | 1% | 2% | 2% | 6% | 5% | |
| Malignant Neoplasm (Non-HCC) (%) | 1% | 2% | 2% | 3% | 4% | |
| Metabolic Disease (%) | 3% | 1% | 1% | 1% | 1% | |
| Other (%) | 8% | 9% | 6% | 11% | 8% | |
| Height (cm)(SD) | 173.7 (10.9) |
172.6 (11.4) |
167.3 (9.7) |
165.3 (9.2) |
168.4 (14.7) |
|
| Weight (kg)(SD) | 87.3 (19.8) |
84.3 (19.1) |
81.6 (18.1) |
69.0 (15.9) |
84.1 (20.4) |
|
| Blood Type | ||||||
| A (%) | 41% | 27% | 31% | 21% | 29% | |
| B (%) | 12% | 25% | 11% | 27% | 15% | |
| O (%) | 42% | 42% | 31% | 38% | 51% | |
| AB (%) | 6% | 6% | 4% | 14% | 6% | |
| Mean Body Mass Index (kg/m2) | 37.1 | 31.3 | 29.2 | 25.1 | 33.6 | |
| MELD Score at Transplant, (median) (25th,75th %ile) |
21 (16, 28) |
24 (19, 30) |
24 (18, 33) |
25 (17, 34) |
23 (18, 32) |
|
| Renal Failure (% dialysis) | 9% | 12% | 15% | 13% | 16% | |
| Diabetes (%) | 7% | 6% | 8% | 5% | 8% | |
| Coronary Artery Disease (%) | 31% | 27% | 29% | 25% | 31% | |
| Chronic Obstructive Pulmonary Disease (%) |
2% | 0.8% | 1% | 0.5% | 2% | |
| Hospitalized at Wait-listing (%) | 17% | 23% | 26% | 34% | 25% | |
| Donor Characteristics | ||||||
| Donor Risk Index Median (25th,75th %ile) |
1.35 (1.09, 1.67) |
1.34 (1.07, 1.64) |
1.36 (1.09, 1.67) |
1.36 (1.10, 1.68) |
1.40 (1.06, 1.70) |
|
| Donor Age (yrs) (SD) | 41.7 (17.4) |
39.9 (16.3) |
41.5 (17.6) |
39.1 (17.8) |
42.5 (17.6) |
|
| Donor Sex (Female) (%) | 39% | 42% | 41% | 46% | 42% | |
| Donor Race (African American)(%) |
15% | 23% | 13% | 16% | 9% | |
| Donor Cause of Death | ||||||
| Cerebrovascular (%) | 43% | 42% | 44% | 42% | 48% | |
| Trauma (%) | 40% | 38% | 38% | 41% | 37% | |
| Anoxia (%) | 14% | 16% | 16% | 15% | 13% | |
| Other (%) | 3% | 4% | 2% | 2% | 1% | |
| Donation after Cardiac Death (%) | 5% | 5% | 4% | 2% | 3% | |
| Donor Height (cm)(SD) | 172.3 (10.8) |
171.8 (10.7) |
170.8 (11.2) |
168.9 (12.2) |
171.2 (9,6) |
|
| Donor Weight (kg) (SD) | 80.1 (20.0) |
78.5 (18.9) |
77.2 (19.2) |
72.8 (18.2) |
80.7 (19.0) |
|
| Organ shared (%) | 30% | 26% | 25% | 23% | 25% | |
| Partial/Split Graft (%) | 1% | 1% | 2% | 5% | 2% | |
| Cold Ischemia Time (hrs)(SD) | 7.6 (3.5) |
7.4 (3.3) |
7.3 (3.7) |
7.5 (2.9) |
7.9 (4.6) |
|
Percentages are column percents.
There was some variation in donor characteristics based on recipient race/ethnicity as well (Table 2). Donor age was similar across all recipient racial/ethnic groups, but there were some statistically significant differences. Among Asian recipients, 46% received liver grafts from female donors versus 39% for White recipients. Donor causes of death did not particularly vary by recipient race/ethnicity, and all groups had 2–5% donation after cardiac death liver transplants. Donor size varied significantly based on racial/ethnic group. Asian recipients received grafts from the shortest and least heavy donors, on average. White recipients had a significantly higher proportion of regional or nationally shared organs, at 30%, versus 23–26% of the other groups. Partial/split graft use was similar across groups as well. Cold ischemia time was greater than seven hours.
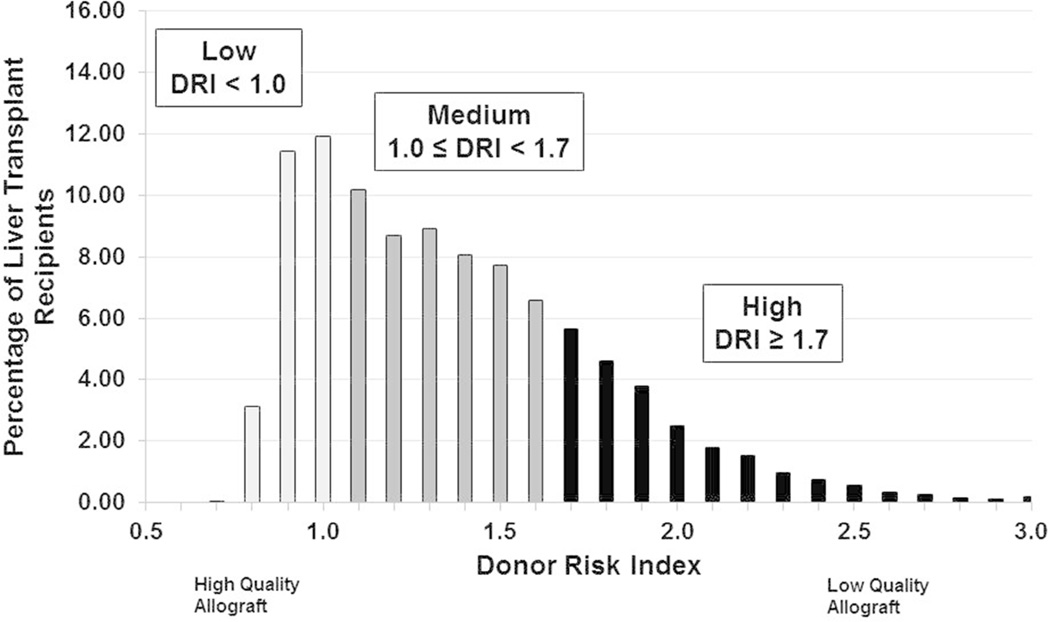
DRI did not vary significantly by race/ethnicity or sex on univariate analysis. The distribution of DRI across the study cohort is shown in Figure 1. The distribution was right skewed. The lowest quartile of DRI was defined by a DRI ≤ 1.0, which corresponds historically with 87.6% adjusted 1-year graft survival (17). The highest quartile of DRI was defined by DRI ≥ 1.7, which corresponds to 76.9% adjusted 1-year graft survival (17). Notably, the median DRI for African Americans, Hispanics, and Whites was nearly identical (median DRI range = 1.33 – 1.40). By race/ethnicity, the DRI of transplanted liver grafts was widely distributed within each group, but there were no significant differences in DRI between groups. By sex, the DRI distribution was similar between females and males, but with a significantly higher trend toward higher DRIs for female recipients (female median DRI = 1.46 vs. male median DRI = 1.40, p<0.0001). In modeling the effect of recipient race/ethnicity and sex on donor selection, we observed significant effects by DRI quartiles. The effect of recipient demographics on the receipt of high and low quality organs is shown in Figure 2. Female liver transplant recipients had 24% greater adjusted odds of receiving a low quality liver allograft compared to their male counterparts. Compared to White liver transplant recipients, recipients of Hispanic ethnicity had significantly greater adjusted odds of receiving a low quality liver allograft. African Americans, Asians, and those of Other race/ethnicity had similar adjusted odds of receiving a low quality liver allograft as Whites, respectively. However, recipient sex or race/ethnicity was not predictive of receiving a high quality liver allograft, after adjusting for other recipient factors. We also modeled the center-adjusted effect of race/ethnicity and sex on donor selection on a continuous DRI scale, and we did not observe an incremental covariate effect.
Figure 1. Histogram of Donor Risk Index of Deceased Donor Liver Transplant Recipients.
The DRI distribution of liver grafts transplanted in the study period was left-shifted, with a long right-sided tail. This tail was related to the likely few providers that select such high risk organs for the recipient phenotype in our cohort. Liver grafts with a DRI ≤ 1.0 were in the lowest DRI quartile and represented the highest quality liver grafts, and grafts with a DRI ≥ 1.7 were in the highest DRI quartile, and of the lowest quality of transplanted grafts.
Figure 2. Recipient Demographics and Variation in Liver Graft Quality.
In a multivariable logistic model to determine how recipient demographics affect the receipt of low quality (high DRI) liver grafts, we observed that female recipients had 24% greater adjusted odds of receiving a low quality graft compared to male recipients. Hispanic recipients had 21% greater adjusted odds of receiving a low quality liver graft compared to White recipients. African Americans, Asians, and Other recipients had similar odds, respectively, to White recipients. Neither recipient sex nor race/ethnicity were associated with the receipt of a high quality (low DRI) liver graft. Of note, these multivariable models were adjusted for several recipient co-morbidities, age, diagnosis, MELD score at transplant, and donation service area.
In order to determine why Hispanics and females received lower quality grafts, we performed a sub- analysis by fitting a sequence of separate models with the individual DRI components as the response variable and the adjustment covariates as above. Based on this analysis, it appears that the Hispanic effect is driven by the selection of older donors, shorter height donors, and a higher selection rate of donation after cardiac death (DCD) donors. The female effect is related to the selection of shorter donors and relatively higher risk of receiving grafts from older donors. No other DRI elements were significantly associated with the recipient being Hispanic or female.
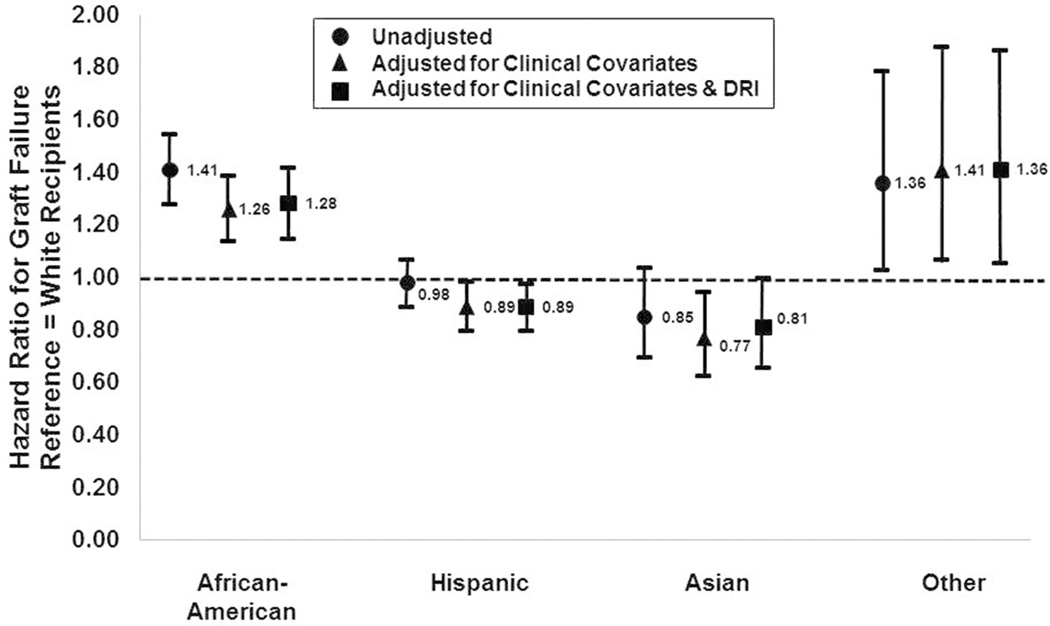
We observed significant racial/ethnic associations with risk of graft failure (Figure 3). African Americans had a 41% higher unadjusted risk of liver graft failure compared to White recipients. Recipients of Other race/ethnicity also had a significant 36% increase in the graft failure risk compared to Whites. Hispanics and Asians had similar unadjusted graft failure risk as White recipients. After adjustment for other clinical covariates, the graft failure risk was attenuated for the major racial/ethnic subdivisions. Asian recipients displayed significantly lower graft failure risk compared to Whites, by 23%. Adjusted directly for DRI and clinical covariates, the magnitude of the excess graft failure risk for African Americans vs. Whites did not change. However, the adjusted graft failure risk for Hispanics declined, resulting in an 11% lower risk compared to Whites. At this level of adjustment, the differences in graft failure risk between Asians and Whites was not statistically significant. Males and females had similar graft failure rates, even after adjustment for clinical covariates and DRI (data not shown). Of note, adjustments for DRI in these models produce the average effect of DRI within a group.
Figure 3. The Effect of Recipient Race/Ethnicity on Liver Graft Failure Risk Based On Various Levels of Covariate-Adjustment.
In the unadjusted models, compared to White recipients, African Americans had a 41% higher risk of graft failure and Other race/ethnicity patients had 36% higher risk of graft failure. African Americans had significantly higher graft failure risk after adjusting for recipient clinical covariates, as did Other race/ethnicity patients. Hispanic recipients had similar graft failure risk, but this risk-adjustment contributed to Asian patients having significantly lower graft failure risk (23% lower) than similar White recipients. Further adjustment for DRI led to lower estimates of graft failure risk for all groups. Hispanic recipients had a significantly lower graft failure versus White recipients, by 11%. The graft survival differences between Asians and Whites were minimal when adjusting for DRI, but African Americans had persistently elevated graft failure risk.
Next, we fitted a model that included product terms (statistical interactions) for each of the race subgroups except for the reference (White) and for DRI (centered at 1.35). With this model, we evaluated the interaction between race/ethnicity and DRI, i.e., the degree to which the effect of race/ethnicity depended on DRI and, correspondingly, the degree to which the DRI effect differed by race/ethnicity. Figure 4 demonstrates the racial/ethnic-specific DRI effects on graft survival. The reference (HR=1.00) was a White recipient of a sample median DRI (DRI=1.35) liver. The differences in DRI effect by race/ethnicity were notable. The DRI had a significantly different effect on graft failure (p=0.02) for African-American vs. White recipients, and the effect was most prominent for the highest DRI livers. In particular, the effect of DRI on graft failure was significantly less pronounced among African-American (relative to White) transplant patients, as evidenced by the flatter slope for the African American group compared to Whites. The slope for Hispanics was similar to that for Whites, suggesting similar DRI effects on the respective risks of graft loss.
Figure 4. The Effect of Recipient Race/Ethnicity on Liver Graft Failure by Donor Risk Index.
Using a race/ethnicity-DRI interaction model, we observed a unique effect of DRI within racial/groups on the risk of graft failure, demonstrated here by the differing slopes for each curve. Each point of each curve represents a hazard ratio for a particular group compared to White recipients at median DRI. The effect of DRI on graft failure risk is different across racial/ethnic groups. Asian recipients had a distinct DRI slope. At the highest DRIs, Asian recipients approached the graft failure risk of White recipients at median DRI (HR=1.00), but graft loss in the Asian population is less affected by changes in DRI compared to White recipients. The curves for White and Hispanic recipients are relatively parallel, suggesting a similar effect of DRI on graft failure risk for both populations. While African Americans have higher graft failure risk compared to Whites, graft loss in this population is significantly less sensitive to changes in DRI (p=0.02), as depicted by the flatter slope. This suggests that the higher graft loss is attributable to issues beyond donor selection.
Discussion
Our investigation has demonstrated that current liver donor allograft selection practices result in deceased-donor livers of lower quality for female and Hispanic liver transplant recipients in the United States. However, differences in graft survival by sex and race/ethnicity are not attributable to corresponding differences in donor quality.
Disparities in liver transplant outcomes are driven by several factors including recipient characteristics, donor factors, conduct of the transplant procedure, and post-transplant medical and surgical management (4). Most studies on disparities in these outcomes have only focused on recipient differences without properly accounting for donor factors. We used the liver DRI as a donor quality measure, and found, as in previous studies, that outcomes for African American recipients are inequitable. Hispanics and Asian recipients had similar, if not lower, graft failure risk compared to their White counterparts. Interestingly, we have shown that female sex and Hispanic ethnicity were significantly associated with the receipt of low quality (high DRI) liver grafts, which may be related to graft selection based on other characteristics that fit unfavorably in the calculation of liver DRI, such as donor age, donor height, and DCD status. Despite this association, however, we did not observe any tangibly increased risk on the eventual transplant outcome across the population. This suggests that disparities in liver transplant outcomes are not uniform across all minority groups, and African American recipients carry a disproportionately higher burden of inequity.
For female recipients, the increased odds of receiving higher DRI livers may be related to smaller graft and donor size, which is a natural clinical consequence of the need to transplant smaller individuals. Despite the utilization of higher DRI organs in this group, outcomes are comparable to those with better quality liver allografts across the sexes. The populations at risk for receiving higher risk livers had equivalent graft outcomes to their “lower risk” counterparts, which highlights the appropriateness of use by the providers selecting these organs and conducting post-transplant care.
An interesting finding in our analysis is that DRI affected graft failure risk differently across racial/ethnic groups. Notably, the greatest variation in DRI effect occurred with the highest risk organs, even after accounting for center effects. Compared to Whites, African American liver transplant recipient outcomes were significantly less sensitive to the effects of DRI, despite being at a significantly increased overall risk of graft loss. Provider bias does not seem to drive the inferior outcomes in the minority population, at least through its potential role in allograft selection. Our data suggest that factors beyond donor selection practices are responsible for lower graft survival in the African American population. These factors are likely unmeasured by the SRTR, and may include specific recipient clinical characteristics, non-medical social factors, patient compliance, provider practices related to the management of co-morbidities, or other considerations. These factors clearly merit further study, as they are likely driving the nearly 30% higher risk of graft loss in the African American population compared to White recipients.
Our study has several limitations. We did not directly adjust our models for socioeconomic and education factors, which clearly affect disparities in a broader health care context. In the liver transplant process, racial/ethnic disparities have been reported at earlier steps in the continuum of liver disease care, particularly in referral to a transplant center, and in the likelihood of receipt of a liver transplant (4, 6, 16, 18–21). Patient socioeconomic status and education are likely to affect a patient’s ability to navigate these early steps. After patients have gone through the entire transplant process, some degree of parity in socioeconomic status and education has to be present – all of the recipients were deemed reasonable candidates for receipt of a transplant, and would have demonstrated the resources necessary for a successful outcome at the time of listing. This was a major consideration in choosing not to include socio-economic factors in our analysis. Moreover, these are not well tracked in the existing SRTR data. We applied DRI in our models as a composite measure. It was derived from several components, whose individual effects may have changed over time. However, the DRI score has gained clinical relevance and common usage as a composite; adjusting for its individual components would potentially be difficult to interpret. Our models were based on data submitted by transplant centers to the OPTN, and did not include other elements that might modify the associations observed in our study. In order to limit bias, we focused on a homogeneous cohort of the adult liver transplant population, and did not address disparities for pediatric recipients, those with acute liver failure, or those who had hepatocellular carcinoma.
Our results may have implications from an organ selection perspective. However, an informed decision regarding the quality of a donor liver that a patient is willing to accept should also depend on the patient’s pre-transplant prognosis. Comparisons among demographic subgroups with respect to the survival benefit of liver transplantation would require a much different analytic approach than that used in the current report. Such analysis is beyond the scope of this report, but would be a valuable future undertaking.
This study provides one of the most comprehensive assessments of disparities in liver transplantation, and improves upon previous analyses by eliminating donor quality as a potential mediator of variation. However, solutions to this multifactorial problem will not arrive from secondary data analyses alone, and qualitative and mixed-methods studies are needed to better understand the mechanisms behind disparities in all steps of the transplant process. Future patient-centered studies on liver transplant outcomes will also help elucidate the medical and non-medical factors that affect variation in graft survival.
Methods
We obtained data from the Scientific Registry of Transplant Recipients (SRTR) based on patient-level data submitted by transplant centers to the Organ Procurement and Transplantation Network (OPTN). We excluded patients with acute liver failure (Status 1); those listed at age less than 18 years; and patients who received MELD exceptions. Collectively, this resulted in excluding 8,492 transplant recipients. The final study cohort consisted of adult deceased donor liver transplant recipients (n=19,249) who had received transplants for chronic end-stage liver disease between March 1, 2002 and December 31, 2008.
As in previous studies using SRTR data, recipient race and ethnicity were considered jointly, based on race and ethnicity definitions provided by the transplant centers using the OPTN data collection infrastructure (18). Race and ethnicity were defined as non-Hispanic White (White), African-American, Hispanic, Asian, or Other; with the latter including Native Americans, Native Alaskans, Native Hawaiians, and those of undefined or mixed race/ethnicity (e.g., Black Hispanics).
Model Development
Logistic regression was used to estimate the effect of recipient demographics, namely sex and race/ethnicity, on the receipt of a high or poor quality liver allograft. Liver allograft quality was quantified through the donor risk index (DRI), as previously described by Feng et al (17). Donor characteristics included in the DRI were donor age, donor race/ethnicity (African American vs. White), donor height, cause of death, donation after cardiac death, the use of a partial/split graft, cold ischemia time, and sharing status (local, regional share, national share). We subsequently determined the distribution of DRI in the study population and defined high quality as the lowest DRI quartile (DRI ≤1.0), and low quality as the highest DRI quartile (DRI >1.7). The middle two quartiles (1.0 < DRI ≤ 1.7) served as the reference group. Specifically, to evaluate the probability that a transplanted patient received a low DRI liver, a logistic model was fitted to low and medium DRI recipients. Through this model, we assessed the association between recipient race/ethnicity and sex on the odds of receiving a low (as opposed to medium) DRI organ. Similarly, we estimated the effect of race and gender on the odds of receiving a high DRI liver by fitting a logistic model to high and medium DRI liver recipients. We selected this model approach to avoid bias. The distribution of DRI is highly skewed, as shown in Figure 1, and the lack of a normal distribution invalidates its use as a response variable in a linear regression approach. Log transformation of DRI would render all results of the model uninterpretable as there is no valid way to back-transform the slopes attributed to the log of the exposure and adjustment covariates. Recipient and other factors included in the risk-adjustment were age, diagnosis, height, weight, blood type, hospitalization status, diabetes, ascites, encephalopathy, albumin, utilization of renal replacement therapy, MELD score at transplant, time until transplant, and donor service area. The effects of these models were expressed as odds ratios (OR), and differences between groups were expressed in percentage difference of odds. Note that our fitting of two separate logistic models is numerically equivalent to fitting a generalized logistic model based on three outcome levels (high, medium and low DRI) with the reference set to medium DRI (22). We chose logistic regression since it is used much more frequently in biomedical studies than the generalized logit model.
We used Cox regression to determine the effect of recipient demographics on post-transplant graft failure, with and without adjustment for donor quality. Patients began follow-up on the date they were transplanted. They were followed until the earliest of graft failure (i.e., death or repeat liver transplantation) or the end of the observation period (12/31/2008). These models were also adjusted for all recipient characteristics used in the previously described logistic models, for DRI, and were further stratified by transplant center. We also aimed to determine whether DRI had a variable effect on liver allograft failure depending on recipient race/ethnicity, and subsequently fitted a graft failure model including interaction terms between DRI and recipient race/ethnicity. Differences in graft failure rates between race subgroups were expressed as covariate-adjusted hazard ratios (HR), and described as percent changes in relative rates.
With respect to model checking, the proportional hazards assumption was verified by fitting interactions with various functions of post-transplant follow up time, such as time, log time, and indicator of time less than one year. Most covariates were categorical and were treated as such. The linearity assumption of several covariates (recipient height, weight, albumin, etc.) had been previously determined (23). Functional form was tested by modeling the respective covariates through a set of categories and plotting the resulting log hazard ratios against their corresponding category medians. Missing data were infrequent. One variable (cold ischemia time) was missing in 10%, and four variables (recipient height, weight, and albumin; and donor height) were each missing in 0.5% of cases. We employed median imputation for these variables to retain valid risk adjustment in the models.
This study was approved by the Institutional Review Board and was performed in accordance with U.S. Health Resources and Services Administration (HRSA) SRTR data use agreement. All statistical analysis was performed using SAS v9.2 (SAS Institute; Cary, NC). Statistical significance was defined by p < 0.05.
Acknowledgements
Funding support was obtained from the University of Michigan Center for Integrated Approaches in Health Disparities (AKM, HZ), and in part by National Institutes of Health grants 5R01DK070869 (DES, HZ), T32 CA009672-18 (AKM), and L60 MD002968-01 (AKM).
List of Abbreviations
- DRI
Donor Risk Index
- SRTR
Scientific Registry of Transplant Recipients
- OPTN
Organ Procurement and Transplant Network
- MELD
Model for End Stage Liver Disease
- HR
Hazard Ratio
Footnotes
-
Amit K. Mathur MD MS
-
-Participated in research design
-
-Participated in the performance of the research
-
-Participated in data analysis
-
-Participated in the writing of the paper
-
-
-
Douglas E. Schaubel PhD
-
-Participated in research design
-
-Participated in the performance of the research
-
-Participated in data analysis
-
-Participated in the writing of the paper
-
-
-
Hui Zhang MS
-
-Participated in the performance of the research
-
-Participated in data analysis
-
-
-
Mary K. Guidinger, MS
-
-Participated in research design
-
-Participated in data analysis
-
-
-
Robert M. Merion MD
-
-Participated in research design
-
-Participated in the performance of the research
-
-Participated in data analysis
-
-Participated in the writing of the paper
-
-
The authors have no conflicts of interest to disclose, and this project was designed and completed without any contributions from commercial entities.
References
- 1.Mathur AK, Schaubel DE, Gong Q, Guidinger MK, Merion RM. Sex-based disparities in liver transplant rates in the United States. Am J Transplant. 11(7):1435–1443. doi: 10.1111/j.1600-6143.2011.03498.x. PMCID:3132137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Mathur AK, Osborne NH, Lynch RJ, Ghaferi AA, Dimick JB, Sonnenday CJ. Racial/ethnic disparities in access to care and survival for patients with early-stage hepatocellular carcinoma. Arch Surg. 2010;145(12):1158–1163. doi: 10.1001/archsurg.2010.272. [DOI] [PubMed] [Google Scholar]
- 3.Mathur AK, Schaubel DE, Gong Q, Guidinger MK, Merion RM. Racial and ethnic disparities in access to liver transplantation. Liver Transpl. 2010;16(9):1033–1040. doi: 10.1002/lt.22108. PMCID: 2936696. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Mathur AK, Sonnenday CJ, Merion RM. Race and Ethnicity in Access to and Outcomes of Liver Transplantation: A Critical Literature Review. American Journal of Transplantation. 2009;9:12, 2662–2668. doi: 10.1111/j.1600-6143.2009.02857.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Thuluvath PJ, Guidinger MK, Fung JJ, Johnson LB, Rayhill SC, Pelletier SJ. Liver transplantation in the United States, 1999–2008. Am J Transplant. 2010;10(4 Pt 2):1003–1019. doi: 10.1111/j.1600-6143.2010.03037.x. [DOI] [PubMed] [Google Scholar]
- 6.Nguyen GC, Thuluvath PJ. Racial disparity in liver disease: Biological, cultural, or socioeconomic factors. Hepatology. 2008;47(3):1058–1066. doi: 10.1002/hep.22223. [DOI] [PubMed] [Google Scholar]
- 7.Kemmer N, Neff GW. Liver transplantation in the ethnic minority population: challenges and prospects. Dig Dis Sci. 2009;55(4):883–889. doi: 10.1007/s10620-009-0803-7. [DOI] [PubMed] [Google Scholar]
- 8.Nair S, Eustace J, Thuluvath PJ. Effect of race on outcome of orthotopic liver transplantation: a cohort study. Lancet. 2002;359(9303):287–293. doi: 10.1016/S0140-6736(02)07494-9. [DOI] [PubMed] [Google Scholar]
- 9.Nair S, Thuluvath PJ. Does race-matched liver transplantation offer any graft survival benefit? Transplant Proc. 2001;33(1–2):1523–1524. doi: 10.1016/s0041-1345(00)02581-1. [DOI] [PubMed] [Google Scholar]
- 10.Hong JC, Yersiz H, Farmer DG, Duffy JP, Ghobrial RM, Nonthasoot B, et al. Longterm outcomes for whole and segmental liver grafts in adult and pediatric liver transplant recipients: a 10-year comparative analysis of 2,988 cases. J Am Coll Surg. 2009;208(5):682–689. doi: 10.1016/j.jamcollsurg.2009.01.023. discusion 9–91. [DOI] [PubMed] [Google Scholar]
- 11.U.S. Scientific Registry of Transplant Recipients. 2010 [updated 2010 cited]; Available from: http://www.ustransplant.org.
- 12.Fan PY, Ashby VB, Fuller DS, Boulware LE, Kao A, Norman SP, et al. Access and outcomes among minority transplant patients, 1999–2008, with a focus on determinants of kidney graft survival. Am J Transplant. 2010;10(4 Pt 2):1090–1107. doi: 10.1111/j.1600-6143.2009.03009.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Ananthakrishnan AN, Saeian K. Racial differences in liver transplantation outcomes in the MELD era. Am J Gastroenterol. 2008;103(4):901–910. doi: 10.1111/j.1572-0241.2008.01809.x. [DOI] [PubMed] [Google Scholar]
- 14.Kemmer N, Neff GW. Liver Transplantation in the Ethnic Minority Population: Challenges and Prospects. Dig Dis Sci. 2009 doi: 10.1007/s10620-009-0803-7. [DOI] [PubMed] [Google Scholar]
- 15.Neff GW, Kemmer N, Kaiser T, Zacharias V, Majoras N, Safdar K. Outcomes in adult and pediatric liver transplantation among various ethnic groups. Transplant Proc. 2007;39(10):3204–3206. doi: 10.1016/j.transproceed.2007.09.031. [DOI] [PubMed] [Google Scholar]
- 16.AYS Brian D Smedley, Nelson Alan R., editors. Institute of Medicine. Unequal Treatment: Confronting Racial and Ethnic Disparities in Health Care. Washington DC: National Academic Press; 2002. [PMC free article] [PubMed] [Google Scholar]
- 17.Feng S, Goodrich NP, Bragg-Gresham JL, Dykstra DM, Punch JD, DebRoy MA, et al. Characteristics associated with liver graft failure: the concept of a donor risk index. Am J Transplant. 2006;6(4):783–790. doi: 10.1111/j.1600-6143.2006.01242.x. [DOI] [PubMed] [Google Scholar]
- 18.Mathur AK, Schaubel DE, Gong Q, Guidinger MK, Merion RM. Racial and Ethnic Disparities in Access to Liver Transplantation in the United States. Liver Transplantation. 2010;16(9):1033–1040. doi: 10.1002/lt.22108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Mathur A, Schaubel DE, Gong Q, Guidinger MK, Merion RM. Sex-Based Disparities in Liver Transplant Rates in the United States. American Journal of Transplantation. 2011 doi: 10.1111/j.1600-6143.2011.03498.x. In Press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Moylan CA, Brady CW, Johnson JL, Smith AD, Tuttle-Newhall JE, Muir AJ. Disparities in liver transplantation before and after introduction of the MELD score. JAMA. 2008;300(20):2371–2378. doi: 10.1001/jama.2008.720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Volk ML, Choi H, Warren GJ, Sonnenday CJ, Marrero JA, Heisler M. Geographic variation in organ availability is responsible for disparities in liver transplantation between Hispanics and Caucasians. Am J Transplant. 2009;9(9):2113–2118. doi: 10.1111/j.1600-6143.2009.02744.x. [DOI] [PubMed] [Google Scholar]
- 22.Stokes ME, Davis CS, Koch GG. Categorical Data Analysis Using the SAS System. New York, NY: John Wiley & Sons; 2001. [Google Scholar]
- 23.Schaubel DE, Guidinger MK, Biggins SW, Kalbfleisch JD, Pomfret EA, Sharma P, et al. Survival benefit-based deceased-donor liver allocation. Am J Transplant. 2009;9(4 Pt 2):970–981. doi: 10.1111/j.1600-6143.2009.02571.x. [DOI] [PMC free article] [PubMed] [Google Scholar]