Abstract
Glechoma hederacea (GH), commonly known as ground-ivy or gill-over-the-ground, has been extensively used in folk remedies for relieving symptoms of inflammatory disorders. However, the molecular mechanisms underlying the therapeutic action of GH are poorly understood. Here, we demonstrate that GH constituents inhibit osteoclastogenesis by abrogating receptor activator of nuclear κ-B ligand (RANKL)-induced free cytosolic Ca2+ ([Ca2+]i) oscillations. To evaluate the effect of GH on osteoclastogenesis, we assessed the formation of multi-nucleated cells (MNCs), enzymatic activity of tartrate-resistant acidic phosphatase (TRAP), expression of nuclear factor of activated T-cells cytoplasmic 1 (NFATc1), and [Ca2+]i alterations in response to treatment with GH ethanol extract (GHE) in primarily cultured bone marrow–derived macrophages (BMMs). Treatment of RANKL-stimulated or non-stimulated BMMs with GHE markedly suppressed MNC formation, TRAP activity, and NFATc1 expression in a dose-dependent manner. Additionally, GHE treatment induced a large transient elevation in [Ca2+]i while suppressing RANKL-induced [Ca2+]i oscillations, which are essential for NFATc1 activation. GHE-evoked increase in [Ca2+]i was dependent on extracellular Ca2+ and was inhibited by 1,4-dihydropyridine (DHP), inhibitor of voltage-gated Ca2+ channels (VGCCs), but was independent of store-operated Ca2+ channels. Notably, after transient [Ca2+] elevation, treatment with GHE desensitized the VGCCs, resulting in an abrogation of RANKL-induced [Ca2+]i oscillations and MNC formation. These findings demonstrate that treatment of BMMs with GHE suppresses RANKL-mediated osteoclastogenesis by activating and then desensitizing DHP-sensitive VGCCs, suggesting potential applications of GH in the treatment of bone disorders, such as periodontitis, osteoporosis, and rheumatoid arthritis.
Keywords: natural product, osteoclast, bone remodeling, [Ca2+]i oscillation, NFATc1, periodontitis
Introduction
Chronic inflammation is known to disrupt this balance by eliciting an excessive increase in the osteoclast population, resulting in serious pathologies that affect bone integrity (Walsh et al., 2006; Lorenzo et al., 2008). Especially in periodontitis, excessive and chronic immune responses mediated by an overgrowth of micro-organisms in the periodontium elicit soft-tissue inflammation in the gingiva and the periodontal ligament (Pihlstrom et al., 2005). As inflammation progresses, various inflammatory cytokines elicit bone loss, resulting from rising osteoclast population (Takayanagi et al., 2000; Walsh et al., 2006; Lorenzo et al., 2008).
Recently, RANKL stimulation of BMMs increased free cytosolic Ca2+ ([Ca2+]i) through stimulating [Ca2+]i oscillations (Takayanagi et al., 2002). RANKL-induced [Ca2+]i oscillations transmit signals to the CaMKIV, calcineurin, and NFATc1, which play crucial roles in determining cell fate during late-stage osteoclastogenesis (Takayanagi et al., 2002). Following the recognition of the RANKL-induced [Ca2+]i oscillations in osteoclastogenesis, recent studies examined the effects of exogenous factors present in food on RANKL-induced [Ca2+]i oscillations. RANKL-mediated [Ca2+]i oscillations and osteoclastogenesis were shown to be down-regulated by extracellular Zn+ or by compounds extracted from Chrysanthemum zawadskii herbich var. latilobum kitamura (Gu et al., 2013; Park et al., 2013). These reports strongly suggest that compounds present in food should be investigated for potential effectiveness in the treatment of bone disorders.
Glechoma hederacea (GH) is an aromatic creeper of the mint family Lamiaceae. It has been widely used in traditional medicine for the treatment of inflammatory diseases (Kumarasamy et al., 2002). Some of the compounds isolated from GH exhibit anti-inflammatory activity by reducing TNFα-induced NF-κB activity, inhibiting iNOS, and reducing COX2 expression in HepG2 cells (Kim et al., 2011). Furthermore, certain compounds present in GH reduce NO production mediated by IFNγ, LPS, and pro-inflammatory cytokines such as IL-12p70 and TNFα in mouse peritoneal macrophages (An et al., 2006). Importantly, these inflammatory cytokines are reported to accelerate bone turnover resulting from the induction of excessive osteoclastogenesis (Walsh et al., 2006; Lorenzo et al., 2008). These reports strongly indicate that GH may have positive effects in inflammation-mediated bone disorders, such as periodontitis. However, the effects of ingested GH on osteoclastogenesis and the mechanism by which it affects cell functions remain uncertain. Here, we describe the molecular mechanisms of the effect of GH ethanol extract (GHE) on RANKL-mediated osteoclastogenesis. GHE transiently inactivates VGCCs following a transient elevation of [Ca2+]i and subsequent channel desensitization, resulting in an abrogation of RANKL-induced [Ca2+]i oscillations and inhibition of osteoclastogenesis.
Materials & Methods
Detailed materials and methods are given in the Appendix. Only key methods are described here.
Experimental Animals and Reagents
All experiments were performed with BMMs isolated from the femur and tibia of C57BL/6J mice. All experiments involving mice were conducted under protocols approved by the Animal Care and Use Committee of Wonkwang University. GHE (95% ethanol) was purchased from the Korea Plant Extract Bank (Daejeon, Korea).
In vitro Osteoclast Formation
Isolated BMMs were cultured in αMEM in the presence of M-CSF (30 ng/mL) and stimulated with RANKL (50 ng/mL) to differentiate into osteoclasts, as previously described (Kim et al., 2010). Cytochemical staining for TRAP expression was performed with the leukocyte acid phosphate assay kit (Sigma-Aldrich, St. Louis, MO, USA), following the manufacturer’s procedures. To measure total TRAP activity, we added p-nitrophenyl phosphate (Sigma-Aldrich) substrate to the culture media containing whole lysates of BMMs. Optical density was measured at an absorbance of 405 nm.
Cell Viability Assay
Following incubation under designated culture conditions, EZ-Cytox reagents were added and cells incubated for an additional 4 hr at 37 °C. Optical density was measured in a microplate reader at 450 nm (Tecan, Männedorf, Switzerland).
Western Blot Analysis
Following incubation under stated conditions, NFATc1 expression levels were analyzed by standard Western blot analysis. Protein was probed with mAb of NFATc1 (1:1,000) and pAb β-actin (1:2,000).
Measurement of [Ca2+]i
[Ca2+]i was determined with the Ca2+-sensitive fluorescence dye Fura2, as described previously (Kim et al., 2010) and in the Appendix.
Statistical Analysis
Results were analyzed by the Student’s two-tailed t test, and data are presented as mean ± SD of the stated number of observations obtained from the indicated number of independent experiments. p values less than .05 were considered statistically significant.
Results
GHE Treatment Suppressed RANKL-mediated Osteoclastogenesis in a Dose-dependent Manner
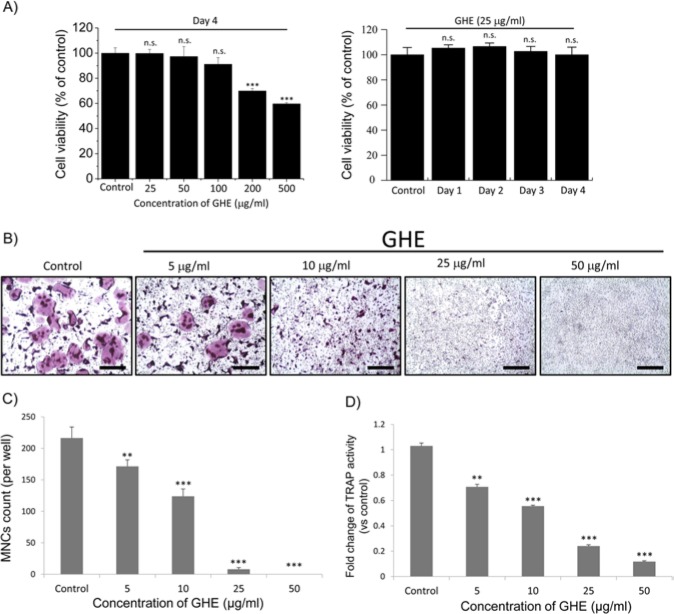
Aggravated chronic inflammatory conditions, such as periodontitis, are accompanied by local bone loss caused by excessive osteoclastogenesis (Pihlstrom et al., 2005). Considering the clinical uses of GH in folk remedies for reducing the severity of inflammatory responses, compounds present in GH may directly or indirectly suppress osteoclastogenesis. We first evaluated the cytotoxicity of GHE on BMMs. Cells were incubated simultaneously with both GHE and RANKL at a range of concentrations (Fig. 1A, left panel) for 4 days, considerable time for differentiation into osteoclasts. In a second series of experiments, BMMs incubated with GHE at a concentration of 25 μg/mL (previously shown to be non-cytotoxic) were treated with RANKL for a range of time periods (Fig. 1A, right panel). GHE exhibited no significant cytotoxicity on BMMs at the concentrations (25-100 μg/mL) and lengths of incubation (Fig. 1A) used in this study. To evaluate the physiological effects of GHE on osteoclastogenesis, we treated RANKL-stimulated BMMs with GHE at indicated concentrations for 4 days. After incubation, TRAP staining and TRAP activity assay were performed. Results showed that formation of TRAP-positive MNCs and total TRAP activity were markedly reduced by GHE treatment in a dose-dependent manner (Figs. 1B-1D). Along with the cytotoxicity data, we assumed that GHE treatment directly suppresses RANKL-mediated osteoclastogenesis by modulating differentiation-related signaling pathways.
Figure 1.
Effect of Glechoma hederacea ethanol extract (GHE) on cell viability, multi-nucleated cell (MNC) formation, and tartrate-resistant acidic phosphatase (TRAP) activity. (A, left panel) Isolated bone marrow-derived macrophages (BMMs) were simultaneously treated with receptor activator of nuclear factor kappa-B ligand (RANKL) (50 ng/mL) and GHE (0, 25, 50, 100, 200, 500 μg/mL) and then incubated for 4 days. (A, right panel) BMMs were simultaneously treated with RANKL (50 ng/mL) and GHE (25 μg/ml) and then incubated for 1, 2, 3, and 4 days. In each experiment, vehicles of GHE (EtOH, left panel) and RANKL (DW, right panel) were treated, respectively, and regarded as the control group. Data present the percentages in the control group. (B-D) BMMs were treated with 5, 10, 25, and 50 μg/mL GHE in the presence of RANKL (50 ng/mL) and incubated for 4 days. Following incubation, TRAP staining and measurement of TRAP activity were performed. TRAP-positive MNCs, which are identified by the presence of more than 3 nuclei and cell size larger than 100 μm in diameter, present in each well were counted, and total activities of TRAP were subsequently measured in fused and non-fused cells. Data are expressed as the mean ± SD from 3 independent experiments. *p < .05 and ***p < .001 vs. the control group. Scale bar = 200 μm.
Treatment of BMMs with GHE Induces a Large Transient [Ca2+]i Elevation, Abolishing RANKL-induced [Ca2+]i Oscillations and NFATc1 Expression
Based on our previous results showing reduced TRAP expression and MNCs formation, we examined the effects of GHE on NFATc1 expression and [Ca2+]i oscillations. To assess the alterations in NFATc1 expression in response to GHE, we simultaneously treated BMMs with RANKL and GHE and maintained them for the indicated length of time. As a result, GHE treatment completely blocked RANKL-mediated induction of NFATc1 (Fig. 2A). This finding led us to further assessment of the effect of GHE on the generation of [Ca2+]i oscillations, because cytosolic Ca2+ mobilization acts as a key regulator of NFATc1 activity and in auto-amplification in the RANKL-mediated signaling pathway. To estimate the acute effects of GHE, we diluted GHE in HEPES buffer and perfused it directly to the cells. GHE treatment induced a transient and large [Ca2+]i increase in cells presenting [Ca2+]i oscillations (Fig. 2B, right panel) and those without any [Ca2+]i oscillations (Fig. 2B, left panel). Following the [Ca2+]i increase caused by GHE, RANKL-induced [Ca2+]i oscillations disappeared, while [Ca2+]i returned to baseline in the continued presence of GHE (Figs. 2A, 2B). Consistent with the results showing reduced RANKL-mediated osteoclastogenesis and TRAP induction in GHE-treated cells, the current finding indicates that GHE abolishes differentiation-related signaling cascade by interfering with the intracellular Ca2+ signaling responsible for NFATc1 auto-amplification.
Figure 2.
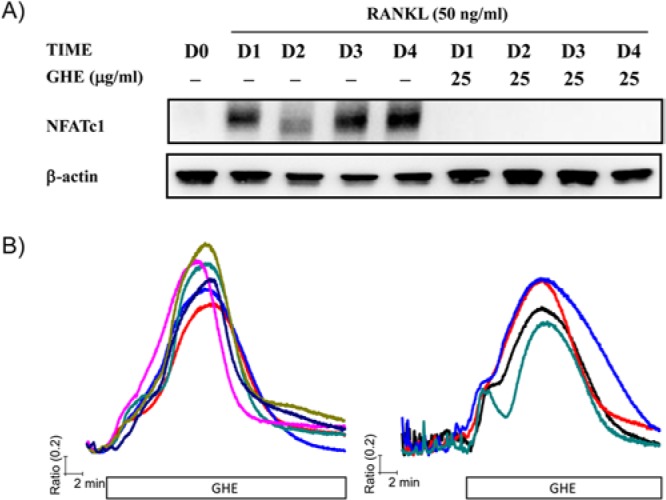
GHE provokes a transient [Ca2+]i elevation, resulting in suppression of RANKL-mediated NFATc1 expression. (A) Isolated BMMs were treated with RANKL (50 ng/mL, D0 presents DW treatment instead of RANKL) in the presence or absence of GHE (25 μg/mL) and cultured for indicated times (0 ~ 4 days). Following incubation, whole-cell lysates were collected and used for the evaluation of total NFATc1 expression. (B) BMMs seeded onto cover glass were maintained for 2 days with (right panel) or without RANKL stimulation (left panel). After incubation, intracellular Ca2+ mobilization in a single cell was measured as described in Materials & Methods. Cells were perfused with HEPES buffer, and then GHE (25 μg/mL) diluted in HEPES buffer was acutely treated until the end of the experiment. Each trace presents cytosolic Ca2+ mobilization in each cell.
GHE-mediated [Ca2+]i Elevation Relies on the Ca2+ Influx Independently of PLC-dependent IP3 Production
Determining the Ca2+ source that contributes to cytosolic Ca2+ elevations is critical for an understanding of the physiological responses associated with intracellular Ca2+ homeostasis (De Smedt et al., 2011). To address this issue, we aimed to confirm the putative contribution of Ca2+ release from the IP3-sensitive Ca2+ stores to GHE-mediated elevation of [Ca2+]i. RANKL-stimulated and non-stimulated BMMs were treated with GHE in the presence of U73122 (PLC inhibitor) and U73343 (an inactive analogue of U73122), respectively. The magnitude of GHE-mediated [Ca2+]i elevation was not affected by the inhibition of PLC (Fig. 3). Next, we tested the involvement of extracellular Ca2+ in GHE-mediated [Ca2+]i elevation. To address this question, we completely removed extracellular Ca2+ by exchanging HEPES buffer with Ca2+-free HEPES buffer containing EGTA prior to GHE treatment. After short incubation with Ca2+-free HEPES buffer, cells underwent acute treatment with GHE. Removal of extracellular Ca2+ completely abolished the GHE-induced [Ca2+]i elevation (Fig. 3). This finding demonstrated that GHE induces a transient and large [Ca2+]i elevation, due to a Ca2+ influx from extracellular medium.
Figure 3.
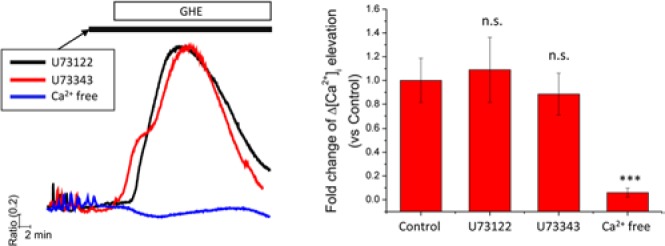
GHE-mediated [Ca2+]i elevation is dependent on an influx of extracellular Ca2+, but not on PLC activation. (Left panel) BMMs seeded on cover glass were incubated in the presence of RANKL (50 ng/mL) for 2 days and then used for intracellular Ca2+ measurement. Prior to GHE, cells were acutely treated with U73122 (10 μM, PLC inhibitor, black line), U73343 (10 μM, inactive control for U73122, red line), and EGTA (1 mM) diluted in HEPES buffer (without CaCl2, blue line). In the presence of U compounds or EGTA, GHE was added and maintained until the end of each experiment. The columns on the right show the maximal [Ca2+]i levels provoked by GHE. Data are presented as mean ± SD of at least 3 independent experiments. ***p < .001 vs. the control group, which was treated with only GHE in the absence of U compounds and EGTA.
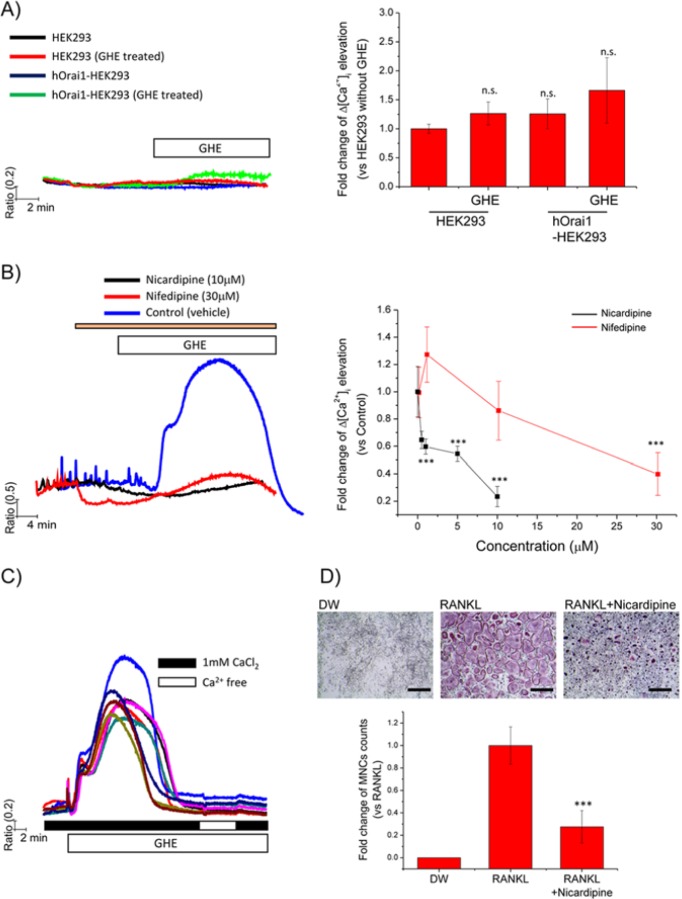
GHE Mediates [Ca2+]i Elevation via 1,4-dihydropyridine (DHP)-sensitive VGCCs
To identify the mechanism by which extracellular Ca2+ moves into the cell following GHE treatment, we tested the involvement of plasma membrane Ca2+ channels. There are 2 representative Ca2+ channel types. One is the SOCCs, such as Orai1, that are activated by STIM1 in response to ER Ca2+ depletion. The other type refers to VGCCs, which are activated by membrane depolarization (Dolphin, 2006). We hypothesized that GHE may directly or indirectly activate SOCCs, VGCCs, or both. To address this question, we first applied GHE treatment to HEK293 cells, since HEK293 cells are non-excitable cells that do not express VGCC, and the characteristics of HEK293 cells are useful to roughly define the involvement of SOCCs. We measured [Ca2+]i elevation in response to GHE in control cells and cells overexpressing hOrai1. GHE did not significantly increase [Ca2+]i in either control HEK293 cells or HEK293 cells overexpressing hOrai1 (Fig. 4A). Next, we examined the effects of VGCC inhibitors on GHE-mediated [Ca2+]i elevation. Several groups have reported that DHP effectively inhibits the function of VGCCs and prevents [Ca2+]i mobilization in both immature and mature osteoclasts (Miyauchi et al., 1990; Kajiya et al., 2003). Based on these findings, RANKL-stimulated BMMs were treated with GHE in the presence of the DHP derivatives nicardipine (0, 0.5, 1, 5, 10 μM) and nifedipine (0, 1, 10, 30 μM), respectively. Fig. 4B shows that blockade of VGCC by DHP abolished GHE-mediated [Ca2+]i elevation in a dose-dependent manner. Notably, we confirmed that treatment with DHP derivatives alone also abrogates RANKL-induced [Ca2+]i oscillations (Fig. 4B). Taken together, these results indicate that GHE-mediated [Ca2+]i elevation is solely dependent on the influx of extracellular Ca2+ via VGCCs. Additionally, channel function of VGCCs is essential for the generation of RANKL-induced [Ca2+]i oscillations.
Figure 4.
GHE treatment desensitizes VGCCs following Ca2+ influx via DHP-sensitive VGCCs, reducing the formation of TRAP-positive MNCs. Fura2-loaded cells were used to measure [Ca2+]i in each experiment. (A) HEK293 (vehicle vector only) and hOrai1-overexpressed HEK293 cells were acutely treated with GHE diluted in HEPES buffer. Only HEPES buffer-perfused cells without GHE were used as controls. The columns on the right show the maximal [Ca2+]i levels responding to GHE. (B) Isolated BMMs were stimulated with RANKL (50 ng/mL) and incubated for 2 days, after which cells were used for measuring [Ca2+]i responding to GHE. DHP derivatives, such as nicardipine (0, 0.5, 1, 5, 10 μM; black trace) and nifedipine (0, 1, 10, 30 μM, red trace), respectively, were treated to inhibit VGCCs prior to GHE treatment. GHE was then acutely added to the cells in the presence of DHP derivatives. The left panel presents representative GHE-mediated Ca2+ traces from cells pre-treated with the maximal concentration of each DHP derivative. The maximal [Ca2+]i levels reached following GHE are presented as relative values compared with those in the control group treated with GHE in the presence of vehicle of DHP (blue trace). (C) GHE was acutely treated to provoke transient [Ca2+]i elevation. Once [Ca2+]i returned to the basal level following GHE treatment; extracellular Ca2+ was temporarily removed by the addition of EGTA and then added back in the presence of GHE. (D) RANKL-stimulated BMMs were cultured with nicardipine (10 μM) for 4 days. Following incubation, TRAP staining was performed as described previously. TRAP-positive MNCs in each well were counted. Relative values compared with those in control (RANKL-treated) were presented. All data are presented as mean ± SD of at least 3 independent experiments. ***p < .001 vs. the control group. Scale bar = 200 μm.
GHE Desensitizes VGCCs after Their Transient Activation, Resulting in Suppression of Osteoclastogenesis
For further study of the effects of GHE on [Ca2+]i, we aimed to identify the mechanism by which GHE abrogates RANKL-induced [Ca2+]i oscillations. As shown in previous results, GHE elicits an acute increase in [Ca2+]i, with [Ca2+]i subsequently returning to basal levels. We decided to evaluate whether GHE consistently inactivates VGCCs following transient [Ca2+]i elevation. To investigate this question, we temporarily removed extracellular Ca2+ in the presence of GHE, and then added it back. If VGCCs were in an activated state in the continued presence of GHE, [Ca2+]i elevation should be observed upon the return of extracellular Ca2+. Minimal increase in [Ca2+]i was observed when the cells were perfused with Ca2+-free HEPES buffer followed by the HEPES buffers containing 1 mM Ca2+ (Fig. 4C). This result clearly indicates that VGCCs of GHE-treated BMMs desensitize after transient [Ca2+]i elevation. Notably, this finding is consistent with results shown in Fig. 4B and suggests that VGCCs play important roles in the RANKL-induced [Ca2+]i oscillations necessary for osteoclastogenesis. To confirm whether VGCCs are involved in osteoclastogenesis, we treated BMMs with the DHP derivative nicardipine, and RANKL-induced osteoclastogenesis was evaluated. Inhibition of DHP-sensitive VGCCs suppressed MNC formation (Fig. 4D). Taken together, our findings suggest that GHE abrogates RANKL-induced [Ca2+]i oscillations by inactivating the VGCCs after mediating a transient [Ca2+]i elevation, providing evidence for the involvement of DHP-sensitive VGCCs in osteoclastogenesis.
Discussion
RANKL-mediated intracellular Ca2+ mobilization and subsequent activation of CaMKIV, calcineurin, and NFATc1 have been evaluated in the study of RANKL-mediated osteoclastogenesis. These Ca2+ responses are generated in the form of oscillations which typically regulate physiological functions in diverse cell types. Recent studies have demonstrated that RANKL-induced [Ca2+]i oscillations play crucial roles in RANKL-mediated osteoclastogenesis. Manipulation of [Ca2+]i oscillations by means of specific inhibitors of Ca2+ channels or by modifying the expression of genes encoding SERCA, PMCA, and PLCγ has been found to affect the expression of genes involved in the late stage of osteoclastogenesis. Specifically, the activity of NFATc1 is primarily dependent on the duration and frequency of [Ca2+]i oscillations (Kajiya, 2012; Kim et al., 2013). In terms of cellular decision-making, [Ca2+]i oscillations regulate the translocation of NFATc1 into the nucleus, with the accumulation of NFATc1 in the nucleus determining the fate of the cell by modulating gene expression. Based on this role of NFATc1, the interaction between [Ca2+]i and NFATc1 has now been generally accepted to provide a nodal point for the regulation of osteoclastogenesis and bone remodeling. Besides gene modification, multiple compounds obtained from foods such as medicinal herbs also appear to play important roles in balancing bone metabolism, especially in alveolar bone. However, the molecular mechanisms underlying the effects of ingested medicinal herbs on bone metabolism remain unclear. In this study, we determined that GHE strongly negatively regulated RANKL-mediated osteoclastogenesis by inducing Ca2+ influx through VGCCs and subsequent desensitization of the channels. Conversely, GHE-mediated [Ca2+]i elevation was independent of Ca2+ release from IP3-sensitive internal Ca2+ stores.
One of the interesting findings in this study is that GHE provokes a transient, excessive, and bell-shaped [Ca2+]i mobilization via DHP-sensitive VGCCs, which eventually results in an abrogation of RANKL-induced [Ca2+]i oscillations (Figs. 2, 3). Previously, it was unclear whether Ca2+ influx via VGCC contributes to the generation of RANKL-induced [Ca2+]i oscillations. To our knowledge, our results are the first to show that DHP-sensitive VGCCs are involved in the generation of RANKL-induced [Ca2+]i oscillations and that inactivation of VGCCs by DHP derivatives results in a suppression of osteoclastogenesis (Figs. 3, 4). Although not studied here in detail, defining the exact VGCC subunit modulated by GHE appears to be important. So far, Ca2+ influx via VGCC has been implicated in the control of the assembly of podosomes, responsible for cell motility and fusion (Miyauchi et al., 1990). Membrane depolarization by a high extracellular K+ concentration ([K+]e) has been reported to elicit conflicting effects on [Ca2+]i mobilization during osteoclastogenesis. High levels of [K+]e depolarize the cells and induce [Ca2+]i elevation only in fully mature osteoclasts (resorbing state). Conversely, high levels of [K+]e reduced basal [Ca2+]i in immature osteoclasts (non-resorbing state) (Miyauchi et al., 1990). In the present study, we report that the inhibition of VGCCs by DHP derivatives reduces basal [Ca2+]i and abrogates RANKL-induced [Ca2+]i oscillations (Fig. 4B), suggesting that DHP-sensitive VGCCs are involved in generating [Ca2+]i oscillations. This cell-state-dependent effect of Ca2+ influx via VGCCs may provide a novel approach and allow for the development of conditional drugs. Taken together, our results suggest that GHE and GHE-interacting subunits (or isoforms) of VGCCs should be considered as potential therapeutic agents and targets for the treatment of bone disorders caused by excessive osteoclastogenesis.
This study clearly demonstrates that multiple compounds found in GH directly suppress RANKL-mediated osteoclastogenesis regardless of local inflammatory responses or interactions with osteoblasts. Specifically, GHE treatment of BMMs mediated a large transient influx of extracellular Ca2+ via VGCCs, subsequently resulting in desensitization of the channels. These effects of GHE cause an abrogation of RANKL-induced [Ca2+]i oscillations, inhibition of NFATc1 expression, and reduction of MNC formation. Taken together, our findings strongly suggest that GH should be considered as a potential therapeutic agent for the treatment of bone disorders caused by excessive osteoclastogenesis.
Supplementary Material
Footnotes
A supplemental appendix to this article is published electronically only at http://jdr.sagepub.com/supplemental.
This research was supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education, Science and Technology (NRF-2012R1A1A1038381, 2011-0030130).
The authors declare no potential conflicts of interest with respect to the authorship and/or publication of this article.
References
- An HJ, Jeong HJ, Um JY, Kim HM, Hong SH. (2006). Glechoma hederacea inhibits inflammatory mediator release in IFN-gamma and LPS-stimulated mouse peritoneal macrophages. J Ethnopharmacol 106:418-424. [DOI] [PubMed] [Google Scholar]
- De Smedt H, Verkhratsky A, Muallem S. (2011). Ca(2+) signaling mechanisms of cell survival and cell death: an introduction. Cell Calcium 50:207-210. [DOI] [PubMed] [Google Scholar]
- Dolphin AC. (2006). A short history of voltage-gated calcium channels. Br J Pharmacol 147 (Suppl 1):S56-62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gu DR, Hwang JK, Erkhembaatar M, Kwon KB, Kim MS, Lee YR, et al. (2013). Inhibitory effect of Chrysanthemum zawadskii Herbich var. latilobum Kitamura extract on RANKL-induced osteoclast differentiation. Evid Based Complement Alternat Med 2013:509482. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kajiya H. (2012). Calcium signaling in osteoclast differentiation and bone resorption. Adv Exp Med Biol 740:917-932. [DOI] [PubMed] [Google Scholar]
- Kajiya H, Okamoto F, Fukushima H, Takada K, Okabe K. (2003). Mechanism and role of high-potassium-induced reduction of intracellular Ca2+ concentration in rat osteoclasts. Am J Physiol Cell Physiol 285:C457-C466. [DOI] [PubMed] [Google Scholar]
- Kim H, Kim T, Jeong BC, Cho IT, Han D, Takegahara N, et al. (2013). Tmem64 modulates calcium signaling during RANKL-mediated osteoclast differentiation. Cell Metab 17:249-260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim J, Song S, Lee I, Kim Y, Yoo I, Ryoo I, et al. (2011). Anti-inflammatory activity of constituents from Glechoma hederacea var. longituba . Bioorg Med Chem Lett 21:3483-3487. [DOI] [PubMed] [Google Scholar]
- Kim MS, Yang YM, Son A, Tian YS, Lee SI, Kang SW, et al. (2010). RANKL-mediated reactive oxygen species pathway that induces long lasting Ca2+ oscillations essential for osteoclastogenesis. J Biol Chem 285:6913-6921. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kumarasamy Y, Cox PJ, Jaspars M, Nahar L, Sarker SD. (2002). Biological activity of Glechoma hederacea . Fitoterapia 73:721-723. [DOI] [PubMed] [Google Scholar]
- Lorenzo J, Horowitz M, Choi Y. (2008). Osteoimmunology: interactions of the bone and immune system. Endocrine Rev 29:403-440. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miyauchi A, Hruska KA, Greenfield EM, Duncan R, Alvarez J, Barattolo R, et al. (1990). Osteoclast cytosolic calcium, regulated by voltage-gated calcium channels and extracellular calcium, controls podosome assembly and bone resorption. J Cell Biol 111(6 Pt 1):2543-2552. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Park KH, Park B, Yoon DS, Kwon SH, Shin DM, Lee JW, et al. (2013). Zinc inhibits osteoclast differentiation by suppression of Ca2+-Calcineurin-NFATc1 signaling pathway. Cell Commun Signal 11:74. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pihlstrom BL, Michalowicz BS, Johnson NW. (2005). Periodontal diseases. Lancet 366:1809-1820. [DOI] [PubMed] [Google Scholar]
- Takayanagi H, Ogasawara K, Hida S, Chiba T, Murata S, Sato K, et al. (2000). T-cell-mediated regulation of osteoclastogenesis by signalling cross-talk between RANKL and IFN-gamma. Nature 408:600-605. [DOI] [PubMed] [Google Scholar]
- Takayanagi H, Kim S, Koga T, Nishina H, Isshiki M, Yoshida H, et al. (2002). Induction and activation of the transcription factor NFATc1 (NFAT2) integrate RANKL signaling in terminal differentiation of osteoclasts. Dev Cell 3:889-901. [DOI] [PubMed] [Google Scholar]
- Walsh MC, Kim N, Kadono Y, Rho J, Lee SY, Lorenzo J, et al. (2006). Osteoimmunology: interplay between the immune system and bone metabolism. Annu Rev Immunol 24:33-63. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.