Abstract
Osteoradionecrosis (ORN) is a noninfectious, necrotic condition of the bone occurring as a complication of radiotherapy. Most cases occur following trauma or surgical manipulation of the irradiated site. Mandible is the most common bone to be affected following head and neck irradiation. The aim was to develop a successful therapeutic approach for ORN. A spectrum of treatment modalities is practiced for ORN with variable success rate that includes simple irrigation of the affected bone to the partial or complete resection of the jaw bone. In this paper, we present two cases which had successful therapeutic approach for ORN of mandible with autologous bone marrow concentrate stem cells and allogeneic dental pulp stem cells (DPSC) with platelet rich plasma (PRP) following failure of conventional methods. Autologous bone marrow aspirate concentrate (BMAC) was injected around the socket and into the periosteum for one case, and DPSC were mixed with tricalcium phosphate and inserted at the site of the defect in one case. The patient treated with BMAC remained asymptomatic and complete bone remodeling was noticed after 1 year. The extraoral sinus was excised, and healing was uneventful without recurrence in the patient treated with allogeneic DPSC and PRP. Periodic panoramic radiographs revealed an appreciable bone formation from the 2nd month onward. We have successfully treated two cases of ORN with BMAC and DPSC, respectively.
Keywords: Bone marrow aspiration concentrate, dental pulp stem cells, mandible, osteoradionecrosis
INTRODUCTION
Osteoradionecrosis (ORN) is one of the redundant complications of radiotherapy characterized by necrosis of irradiated bone. It develops due to progressive hypoxia, hypo-vascularization, and hypo-cellularity of the affected bone. Radiation causes inflammation of small blood vessels (endarteritis) resulting in the development of thrombi and occlusion of the vascular lumen. It also causes altered collagen synthesis. Compromised tissue perfusion and impaired repair and remodeling capacity make the bone vulnerable to necrosis, even with mild provocation.[1] The mandible is the most commonly affected site following head and neck radiation therapy. Trauma, dental extraction or other surgical manipulations, poor oral hygiene, irritation from ill-fitting denture, usage of tobacco and alcohol are considered to be the precipitating factors.
Clinically, ORN may manifest as mild discomfort when the lesion is small and localized, as the lesion progresses, patient may experience pain, swelling, trismus, sequestrum formation, and pathological fracture. Various treatment modalities are available and are practiced according to the individuality of the lesion. Small and asymptomatic lesions are managed conservatively while reserving more invasive surgical management for progressive and symptomatic lesions.[2] The aim of this article is to present two cases of ORN of mandible that developed following irradiation for squamous cell carcinoma for which adult stem cell therapy alone with surgery was tried following failure of conventional treatment methods. The first case was treated by autologous bone marrow concentrate, and the second case was treated with allogeneic dental pulp stem cells (DPSC).
CASE REPORTS
Case 1
A 48-year-old male reported to our Dental Clinic in March 2010 with the complaint of severe pain and unhealed extraction socket. Past medical history revealed that he had undergone radiotherapy for the management of carcinoma of the soft palate 3 years back. A year after radiotherapy, the tooth on the lower right side was extracted, and the socket had not healed for more than 6 months.
Clinical examination revealed unhealed socket in lower right second molar area with exposure of underlying bone. The extracted socket had apparently developed ORN. The patient was treated by us for nearly 1 year through conventional methods such as wound debridement, warm saline irrigation, socket dressing with zinc oxide eugenol, and antibiotic dressing. Since the complications recurred between periods of conservative treatment and there was no sign of clinical improvement, it was decided to treat the case using stem cell therapy. Detailed and specific informed consent was obtained from the patient and institutional ethical committee approval obtained. Panoramic radiograph taken before current therapy showed an area of bone destruction in right 2nd molar region with irregular margins. Both radiolucency and radio-opacities were observed within the lesion suggestive of chronic osteomyelitis. There was a suspicion of a hairline pathologic fracture (arrow) in the radiograph [Figure 1].
Figure 1.
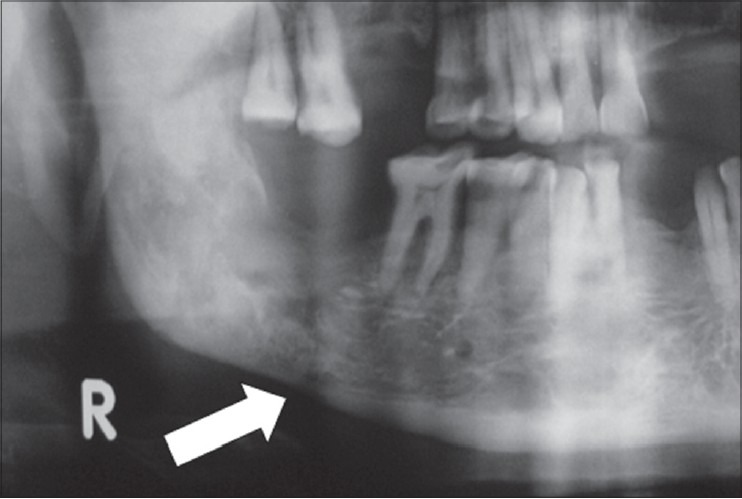
Preoperative radiograph showing necrosed bone with suspected hairline fracture
Bone marrow aspiration and stem cell delivery
The patient was thoroughly assessed for spinal anesthesia and physician's opinion taken for bone marrow aspiration. Under strict aseptic conditions, an Orthopedic surgeon aspirated bone marrow from left superior posterior iliac crest as published previously.[3] About 60 ml of blood was aspirated using three 20 ml syringes each containing 0.3 ml of heparin. From this 60 ml, 6 ml of concentrated bone marrow cells (BMCs) was prepared using Harvest smart prep machine (R) (Harvest Technologies Corporation, 40 Grissom Road, Suite 100 Plymouth, MA 02360 USA).
Under local anesthesia, the socket was curetted, necrotic bone was trimmed to reduce the height of the socket and to induce fresh bleeding. Of 6 ml concentrate, 3 ml of BMCs was injected around the socket and into the periosteum. The remaining 3 ml was mixed with commercially available β-tricalcium phosphate/hydroxyapatite forming a bioactive matrix. This stem cell matrix was spread inside the socket, and gingivovestibular flap was sutured. The material is seen in the X-ray of the patient [Figure 2].
Figure 2.
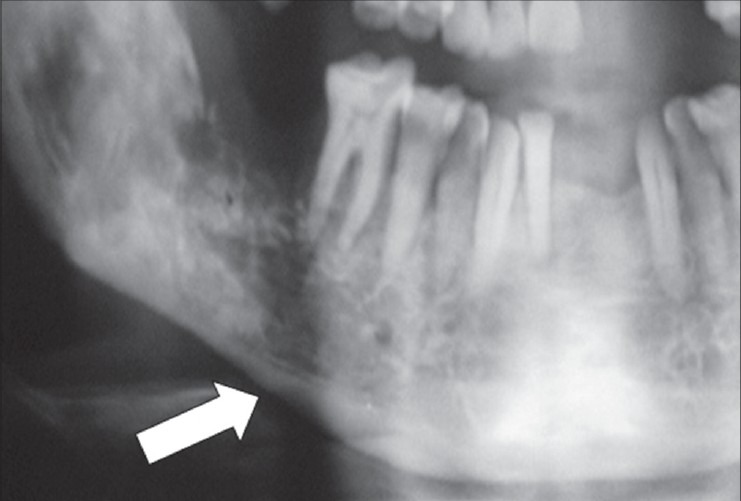
Stem cell matrix in place
Clinical outcome
The patient responded well with a gradual reduction in pain, and there was no discharge from intraoral lesion after 2 months. The radiograph showed signs of early bone formation at 2nd month [Figure 3] and evidence of consolidated bone formation after 6 months with resolution of suspected fracture line [Figure 4]. The patient remained asymptomatic and complete bone remodeling was noticed after 1 year. The patient was followed for more than 2 years and was symptom-free. There were no untoward effects noted related to this treatment.
Figure 3.
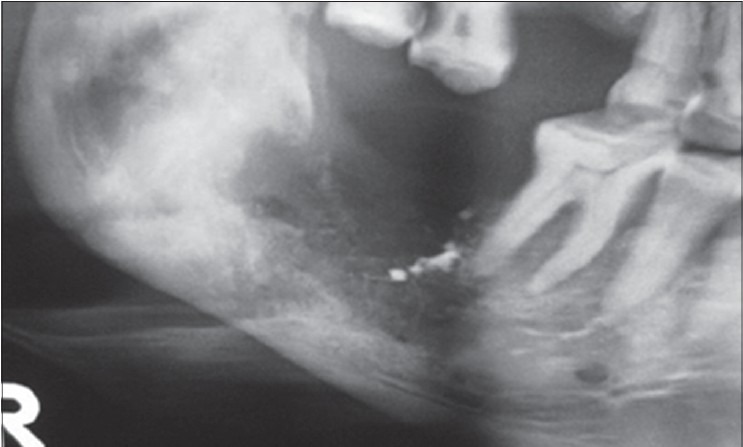
Bone formation 2 months postoperative
Figure 4.
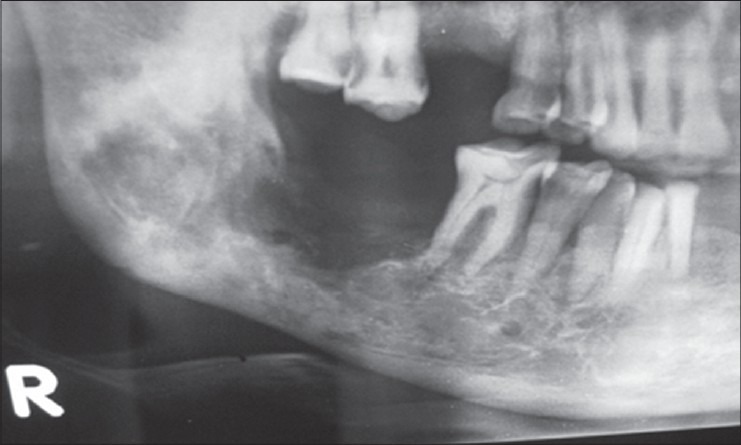
Six months postoperative showing bone formation
Case 2
A 47-year-old male reported to our department with the complaints of pus discharge on the left side cheek for 1 year [Figure 5].
Figure 5.
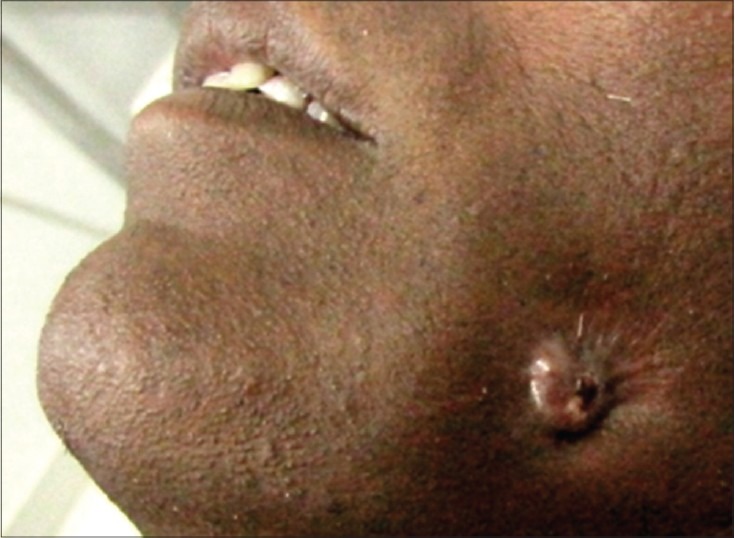
Extraoral sinus
The history revealed post-surgical radiotherapy and chemotherapy for squamous cell carcinoma in left tonsillar region. Clinical examination showed nonhealing wound in the 36, 37 region and multiple grossly decayed teeth in the left mandibular quadrant with pus discharge communicating extraorally through a sinus. Orthopantomogram revealed mixed radiolucency and multiple dental caries in left side body of the mandible [Figure 6]. Eventually, the condition was diagnosed as ORN of mandible.
Figure 6.
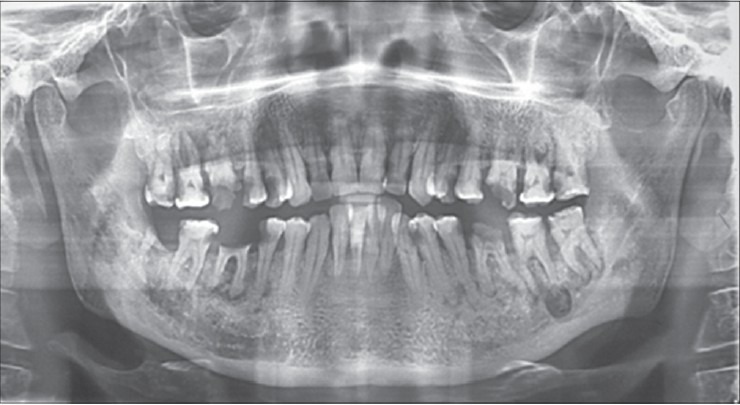
Preoperative OPG
Initially, the patient was treated with a broad spectrum antibiotics and clindamycin. Patient was also prescribed pentoxyphylline (trentol) 500 mg and Vitamin E (evion) twice daily for 6 weeks to improve circulation. It was then decided to complete the treatment with stem cell therapy.
Management and outcome
The allogeneic DPSC were obtained from teeth extracted for orthodontic management with prior consent.
Surgical curettage was done followed by placement of stem cell graft. The graft consisted of platelet-rich plasma (PRP) as a medium and alloplastic graft (tricalcium phosphate) as a scaffold. DPSC were mixed with these, and the matrix was inserted into the defect. Primary closure was given. Stem cells with PRP were also injected into the surrounding soft tissues. The extraoral sinus was excised, and healing was uneventful without recurrence [Figure 7]. Periodic panoramic radiographs revealed an appreciable bone formation from the 2nd month onward and is clearly noted at 6th month after procedure [Figure 8].
Figure 7.
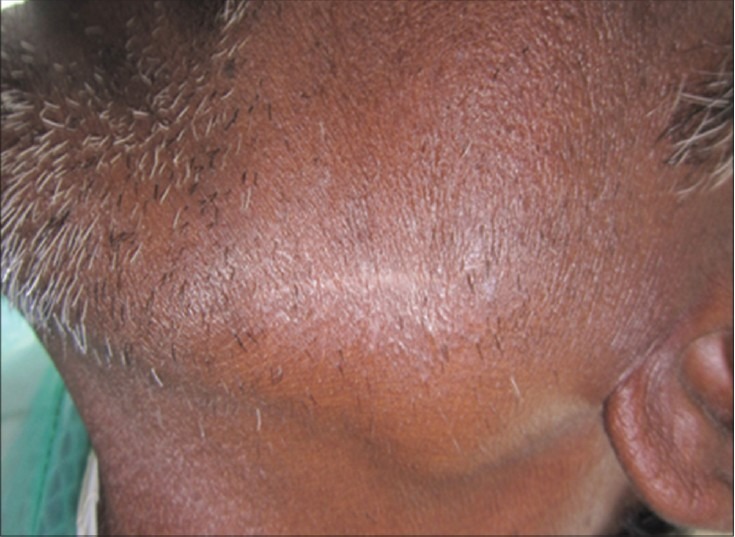
Six months postoperative healed sinus area
Figure 8.
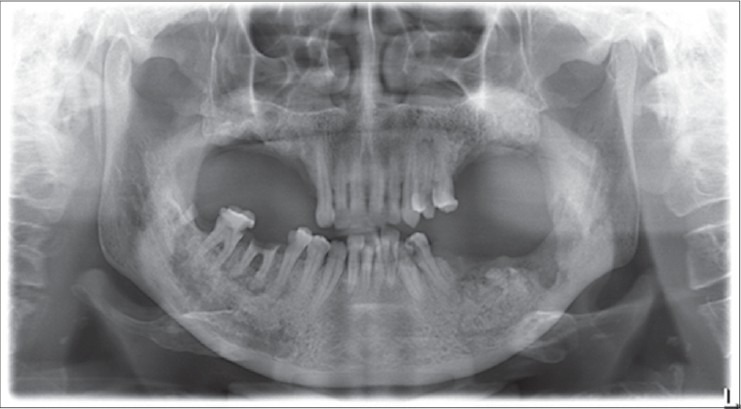
Six months postoperative OPG
DISCUSSION
Mesenchymal stem cells derived from dental pulp differentiate into osteoblasts capable of producing bone. Evidences exist showing that mesenchymal stem cells can be seeded on scaffolds and used to repair human mandibular defects.[4,5,6] According to Ito et al.[7] a gel made with DPSC and PRP showed excellent osteogenic characteristics and significant new bone formation. The DPSC used in this case were cultured in a GMP lab and obtained from the 2nd passage.
The therapeutic application of bone marrow-derived cells for human bone lesions is not new. Adult stem cells have been the subject of many in vitro and animal experiments, demonstrating significant potential in the management of bone lesions. Stimulation with granulocyte colony-stimulating factor, blood aphaeresis, and expanded bone marrow in autologous and homologous procedures have been attempted.[8] There is substantial evidence that human bone marrow supplies the patient with not only hematopoietic cells but also a significant number of mesenchymal stem cells.[9] Bone Marrow harvested stem cells and progenitor cells (BMSC) may be capable of solid organ repair and it has been demonstrated that adult human mesenchymal stem cells from bone marrow can represent a promising source of skeletal regeneration.[10]
The authors use of bone marrow aspirate concentrate (BMAC) for the treatment of ORN of mandible is first of its kind and the authors are not aware of any earlier reports published on the above said procedure for ORN of mandible. There are significant data supporting the clinical efficacy of BMAC stem cells. An immediate autologous transplantation of BMAC can prevent complications of transplanted cells such as pre-aging (telomere shortening), reduced viability, or dedifferentiation/reprogramming that is associated with in-vitro cultivation.[11]
Recently, images of neo-angiogenesis following stem cell therapy in ORN of mandible have been demonstrated by gadolinium-enhanced magnetic resonance scan. Vascular growth is the basis for regeneration in all tissues, delivering oxygen, nutrients, chemical signaling, migrating cells, and immunity. Good blood supply with sufficient viable cells are required for osteogenesis.[8]
Earlier Ficoll-hypaque centrifugation procedures were used for isolation and cellular expansion in-vitro.[10] The cellular composition of concentrate prepared by BMAC system differs from Ficoll method with respect to the number of colony forming unit cells (mesenchymal cells).[11]
The volume reduction protocol of the closed centrifuge allows for the highest concentrate of the mesenchymal stem cells and therefore is a promising candidate for instant stem cell therapy.[12] Sauerbier et al. have shown hard tissue regeneration in vitro by using both Ficoll method and BMAC method.[11] Another major concern in stem cell therapy is safety. To our knowledge, there have been no reports of adverse reactions or complications.
CONCLUSION
Authors have successfully treated two cases of ORN with BMAC and DPSC and PRP, respectively. More experience is required to arrive at a definitive conclusion regarding the benefits of the stem cell therapy for mandibular ORN of the bone due to various etiological factors.
Stem cell-based reconstruction may be attempted in unresponsive ORN cases before other major deformative procedures are planned.
Footnotes
Source of Support: Nil
Conflict of Interest: None declared.
REFERENCES
- 1.Silvestre-Rangil J, Silvestre FJ. Clinico-therapeutic management of osteoradionecrosis: A literature review and update. Med Oral Patol Oral Cir Bucal. 2011;16:e900–4. doi: 10.4317/medoral.17257. [DOI] [PubMed] [Google Scholar]
- 2.Vanderpuye V, Goldson A. Osteoradionecrosis of the mandible. J Natl Med Assoc. 2000;92:579–84. [PMC free article] [PubMed] [Google Scholar]
- 3.Turan RG, Bozdag-Turan I, Ortak J, Akin I, Kische S, Schneider H, et al. Improvement of cardiac function by intracoronary freshly isolated bone marrow cells transplantation in patients with acute myocardial infarction. Circ J. 2011;75:683–91. doi: 10.1253/circj.cj-10-0817. [DOI] [PubMed] [Google Scholar]
- 4.Giuliani A, Manescu A, Langer M, Rustichelli F, Desiderio V, Paino F, et al. Three years after transplants in human mandibles, histological and in-line holotomography revealed that stem cells regenerated a compact rather than a spongy bone: Biological and clinical implications. Stem Cells Transl Med. 2013;2:316–24. doi: 10.5966/sctm.2012-0136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Wolff J, Sándor GK, Miettinen A, Tuovinen VJ, Mannerström B, Patrikoski M, et al. GMP-level adipose stem cells combined with computer-aided manufacturing to reconstruct mandibular ameloblastoma resection defects: Experience with three cases. Ann Maxillofac Surg. 2013;3:114–25. doi: 10.4103/2231-0746.119216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Sándor GK, Numminen J, Wolff J, Thesleff T, Miettinen A, Tuovinen VJ, et al. Adipose stem cells used to reconstruct 13 cases with cranio-maxillofacial hard-tissue defects. Stem Cells Transl Med. 2014;3:530–40. doi: 10.5966/sctm.2013-0173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Ito K, Yamada Y, Nakamura S, Ueda M. Osteogenic potential of effective bone engineering using dental pulp stem cells, bone marrow stem cells, and periosteal cells for osseointegration of dental implants. Int J Oral Maxillofac Implants. 2011;26:947–54. [PubMed] [Google Scholar]
- 8.Mendonça JJ, Juiz-Lopez P. Regenerative facial reconstruction of terminal stage osteoradionecrosis and other advanced craniofacial diseases with adult cultured stem and progenitor cells. Plast Reconstr Surg. 2010;126:1699–709. doi: 10.1097/PRS.0b013e3181f24164. [DOI] [PubMed] [Google Scholar]
- 9.Smiler D, Soltan M, Albitar M. Toward the identification of mesenchymal stem cells in bone marrow and peripheral blood for bone regeneration. Implant Dent. 2008;17:236–47. doi: 10.1097/ID.0b013e3181835b13. [DOI] [PubMed] [Google Scholar]
- 10.Cella L, Oppici A, Arbasi M, Moretto M, Piepoli M, Vallisa D, et al. Autologous bone marrow stem cell intralesional transplantation repairing bisphosphonate related osteonecrosis of the jaw. Head Face Med. 2011;7:16. doi: 10.1186/1746-160X-7-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Sauerbier S, Stricker A, Kuschnierz J, Bühler F, Oshima T, Xavier SP, et al. In vivo comparison of hard tissue regeneration with human mesenchymal stem cells processed with either the FICOLL method or the BMAC method. Tissue Eng Part C Methods. 2010;16:215–23. doi: 10.1089/ten.TEC.2009.0269. [DOI] [PubMed] [Google Scholar]
- 12.Kasten P, Beyen I, Egermann M, Suda AJ, Moghaddam AA, Zimmermann G, et al. Instant stem cell therapy: Characterization and concentration of human mesenchymal stem cells in vitro. Eur Cell Mater. 2008;16:47–55. doi: 10.22203/ecm.v016a06. [DOI] [PubMed] [Google Scholar]


