Keywords: Inflammatory breast cancer, Cytokines, Proteases, Viral infection
Abstract
Inflammatory breast cancer (IBC) is a highly metastatic and fatal form of breast cancer. In fact, IBC is characterized by specific morphological, phenotypic, and biological properties that distinguish it from non-IBC. The aggressive behavior of IBC being more common among young women and the low survival rate alarmed researchers to explore the disease biology. Despite the basic and translational studies needed to understand IBC disease biology and identify specific biomarkers, studies are limited by few available IBC cell lines, experimental models, and paucity of patient samples. Above all, in the last decade, researchers were able to identify new factors that may play a crucial role in IBC progression. Among identified factors are cytokines, chemokines, growth factors, and proteases. In addition, viral infection was also suggested to participate in the etiology of IBC disease. In this review, we present novel factors suggested by different studies to contribute to the etiology of IBC and the proposed new therapeutic insights.
Introduction
Inflammatory breast cancer (IBC) is the most lethal form of primary breast cancer (TNM classification T4) targeting young women. The term “inflammatory breast cancer” was first suggested in 1924 by Lee and Tannebanm as a type of cancer associated with inflammation of the breast [1]. In 1938, Taylor and Meltzer introduced two clinical varieties of IBC, namely primary IBC and secondary IBC [2], to differentiate between IBC and locally advanced breast cancer. The term “primary IBC” or “de novo IBC” is defined as the new development of IBC in a previously normal breast, whereas the term “secondary IBC” describes the inflammatory recurrence of non-IBC breast cancer [3]. IBC represents about 2.5% of newly diagnosed breast cancers in the United States [4], where incidence of IBC is higher among African–American compared to white women [5]. The frequency of IBC in North African countries such as Tunisia, Morocco, and Egypt represents about 10% to 15% of breast cancer [6,7]. Recent studies conducted by Schairer and colleagues compared percentage diagnosis of IBC at the National Cancer Institute, Egypt, and Institute Salah Azaiz (ISA), Tunisia, and they suggested that the increase in IBC cases in North Africa may be due to misdiagnosis of IBC with other types of locally advanced breast cancer [8]. In addition, the lack of breast cancer national registry programs in developing countries should also be taken into consideration.
There are two well recognized systems for case definition of IBC. The first is the French Poussée Évolutive (PEV) system devised in 1959 which defined IBC as a rapidly growing breast malignancy with PEV2 and PEV3 [9,10]. The second is the American Joint Committee on Cancer (AJCC) staging system that classifies IBC as T4d [11].
IBC diagnosis was shown to be associated with a worse survival rate than other types of breast cancer, which remains a therapeutic challenge despite the advances in treatment. The National Cancer Institute’s Surveillance, Epidemiology, and End Results (SEER) program found that the 3-year disease survival rate increased for IBC patients compared to non-IBC patients between 1975–1979 and 1988–1992. For IBC, survival rate increased from 32% to 42% for IBC patients and from 80% to 85% for non-IBC patients [12]. Improved survival rate of IBC patients may be due to the use of neoadjuvant chemotherapy and combination regimens in the treatment of IBC [13,14]. Clinically, IBC is defined by distinct features, including rapid onset within 6 months, erythema, edema of the breast, and a “peau d’orange” appearance to most areas of breast skin. Moreover, patients presented with positive metastatic lymph node involvement and up to one third of patients have distant metastasis at diagnosis [15]. Pathologically, the presence of dermal and stromal tumor emboli is considered a hallmark of IBC. The subsequent lymphatic obstruction by tumor emboli prevents proper drainage of the lymph fluid causing swelling of the breast tissue and produces the inflammatory nature of the disease [3,16].
Biological markers associated with IBC
Molecular profiling studies suggested that the molecular subtypes of IBC are similar to those described in non-IBC. However, low frequency of luminal A and high expression of HER-2 are enriched among IBC patients as compared with non-IBC [17]. Other studies identified specific biological markers that may be associated with IBC poor prognosis, and disease aggressiveness. For instance, IBC is characterized by amplification/over-expression of growth factor receptor HER2 [17] and down regulation of hormone receptors ER/PR [18–20]. The absence of hormonal receptors expression was shown to be correlated with a high degree of malignancy and breast cancer shorter disease-free survival [21]. IBC patients with ER positive receptors have a better prognosis with a median survival of 4 years compared to 2 years median survival for patients with ER-negative IBC [4]. About 80% of IBC carcinoma tissue samples are characterized by loss of WNT1-inducible-signaling pathway protein 3 (WISP3) and also recognized as loss of inflammatory breast cancer gene [22]. WISP3, also known as CCN6, is a cysteine-rich protein found to inhibit invasive and angiogenic potential of IBC cells in tissue cultures and animal models [23]. In addition, IBC embolus is characterized by over-expression of a number of genes such as ras homolog family member C-guanosine triphosphatase (RhoC-GTPase) and E-cadherin [24]. The epithelial marker E-cadherin is a calcium dependent transmembrane glycoprotein that mediates epithelial cell–cell adhesion [25]. IBC cells are characterized by over-expression of E-cadherin, which is essential for adherence of cells together and formation of tumor emboli. Studies suggested that E-cadherin facilitates the dissemination of IBC within the lymphatic vessels by promoting cell–cell contact and maintaining the integrity of IBC tumor emboli within dermal lymphatics [24,26]. The role of E-cadherin in IBC is opposite to non-IBC. In non-IBC, loss of E-cadherin expression contributes to increased tumor proliferation and to the progression of metastasis and is associated with poor prognosis [27], while increased E-cadherin in IBC contributes to disease aggressiveness and decreased survival rate [25]. Moreover, RhoC-GTPase is over-expressed in 90% of IBC tumors compared with 38% of the stage-matched non-IBC tumors. In IBC cell line SUM149, over-expression of RhoC-GTPaseis associated with loss of WISP3 and restoration of WISP3 in SUM149 cells down-regulates the production of RhoC-GTPase and inhibits invasive potential of SUM140 cells [28]. Indeed, RhoC-GTPase is found to play an essential role in the metastatic behavior of IBC by increasing all aspects of metastatic process such as cellular motility and invasion, cytoskeletal assembly, and cell adhesion. RhoC-GTPase controls the cytoskeletal reorganization by inducing actin stress fiber and focal adhesion contacts formation [29–33]. Studies suggested that RhoC-GTPase is a transforming oncogene for human mammary epithelial (HME) cells leading to increase in motility and invasion [32,34]. Therefore, over-expression of RhoC-GTPase leads to transformation of immortalized HME cells with an invasive phenotype such as IBC [31]. In addition to that, several studies characterized that elevated expression of RhoC-GTPase is linked to high histologic grade, positive lymph node status, negative hormonal receptor status, and over-expression of HER-2 [34,35]. Moreover, RhoC-GTPase is associated with up-regulation of vascular endothelial growth factor (VEGF), basic fibroblast growth factor (bFGF), interleukin-6 (IL-6), and interleukin-8 (IL-8), contributing to a distinct type of angiogenic stroma formation in IBC carcinoma [31,36].
However, all discussed previous markers do not distinguish IBC from non-IBC and fail to explain the specific pathobiology of IBC. This was confirmed by studies showing similar expression levels of LIBC/WISP3, RhoC, and E-cadherin in IBC and non-IBC [15]. DNA microarrays studies showed gene expression differences between IBC and non-IBC, and results detected over-expression of Toll-like receptors (TLR) in IBC tissues versus non-IBC tissues [16]. TLR are highly expressed by myelomonocytic cells, including dendritic cells in response to microbial or viral infections [17]. Over-expression of TLR suggests infiltration of IBC by immune cells and possibility of viral etiology in IBC progression.
Recent studied comparing MicroRNAs (miRNAs) expression profiles in non-IBC, IBC carcinoma tissues, and normal breast tissues found that IBC patients are characterized by five over-expressed miRNAs comprising miR-421, miR-486, miR-503, miR-720, and miR-1303 [37].
Tumor emboli as hallmark of IBC
Within lymphatic and blood vessels, IBC carcinoma cells are characterized by specific phenotype “tumor embolus” where carcinoma cells clump together and retract away from the surrounding endothelial lining of blood and lymphatic vessels [24,26]. Tumor embolism is considered as the main route for dissemination of IBC carcinoma cells in vivo, where IBC spread in the form of clumps of cells within lymphatic and blood vessels leading to distant metastasis and multiorgan failure in IBC patients [38]. The well organized architecture of IBC emboli might be due to over-expression of membranous E-cadherin bounded with α or β-catenin, formation of apical surface microvilli and canalis structures [39].
Although the molecular and cellular structure of IBC tumor emboli was described by different studies, there is an argument about the origin of IBC tumor emboli. Traditionally, tumor emboli were thought to have originated from lymphovascular invasion of carcinoma cells as an action proceeding metastasis [24,26]. Barsky and colleagues studied the formation and the properties of IBC emboli in mice model (MARY-X). Their studies suggested that tumor emboli may be formed due to encircling of endothelial cells to clumps of IBC cells “lymphovasculogenesis” rather than traditional lymphovascular invasion [40]. They added that since IBC tumor emboli morphology resembles “embryonic blastocyst,” they may possess the properties of embryonic stem cells. Their studies showed that IBC emboli express stem cell markers such as Notch 3 and aldehyde dehydrogenase (ALDH) enzyme [41]. In fact, the biology of IBC tumor emboli formation is not well understood. This may be due to the lack of in vitro model that recapitulates the biophysical properties of the lymphatic system. Our studies showed that seeding IBC cell line SUM149 in 3D model, it forms spheroid like structures that resemble patients’ in vivo tumor emboli [42].
Interestingly, a recent study created IBC tumor emboli by seeding IBC cell lines in viscous suspension that resemble physical and biological properties of lymphatic fluid [43], they found that IBC cells form tumor emboli when they were seeded in properties that resemble lymphatic fluid, this was not shown by non-IBC cells. Moreover, the study compared between biological markers of the established in vitro emboli and patient emboli. Results showed that in vitro emboli express epithelial marker E-cadherin and RhoC-GTPase similar to patient emboli [13]. Authors concluded that the newly established model might provide an ideal model “to accurately grow and study inflammatory breast cancer biology” [43]. However, more investigations are warranted to validate the use of the previous model in studying interaction between IBC cells and stromal cells, such as immune cells and fibroblasts, harboring the tumor microenvironment.
IBC and tumor associated macrophages
It is clear that dissemination of carcinoma cells is affected by different factors including cues from the inflammatory cells within the tumor microenvironment. Indeed, macrophages are known to be the major inflammatory cells that infiltrate various types of tumors including breast [44,45], contributing to high levels of growth factors, hormones, and cytokines in the tumor microenvironment [46,47]. Macrophages are phagocytic immune cells, whose main function is to eliminate and kill infected cells and pathogens [48]. Within the tumor microenvironment, tumor associated macrophages (TAM) are differentiated into heterogeneous subpopulations, such as (a) “classically activated macrophages” that secrete pro-inflammatory and inflammatory mediators and recruit T-cells as in an early inflammatory response [49] and (b) “regulatory macrophages” that express anti-inflammatory cytokines and increase tumor growth, invasion, and metastasis [50].
A strong association was found between breast TAM and poor prognosis [51,52]. Macrophages secrete soluble mediators that induce migration, invasion, and metastasis of carcinoma cells [53,54]. For instance, TAM secretes matrix metalloproteinases-2 and 9 (MMP-2 and MMP-9) enzymes that can degrade components of the basement membrane, thereby facilitating tumor cell motility, intravasation, and dissemination [54,55]. Increases in expression of MMPs and their inhibitors in TAM were found to correlate with distant metastasis of invasive ductal carcinomas [56]. Moreover, within tumor microenvironment, TAM stimulate carcinoma cells growth and proliferation by releasing growth factors (e.g., epidermal growth factor (EGF) [57]. Interestingly, analyses by cDNA microarrays showed over-expression of Toll-like receptors (TLR) in IBC tissues [58]. TLR are highly expressed by macrophages in response to microbial or viral infections, such as human cytomegalovirus (HCMV) [59].
Although the role of TAM in breast cancer progression is well established by several studies [44,60], their role in IBC has not yet been investigated. We are interested in studying the interaction between human monocytes/macrophages and IBC cells. Using in vitro 3D models, we co-cultured IBC cell line SUM149 with human monocytes U937 or in media conditioned by human monocytes. We found that human monocytes U937 or media conditioned by human monocytes increase expression and activity of Cathepsin B (CTSB) and also stimulate invasiveness and motility by breast carcinoma cells [61]. Since paracrine interaction between monocytes/macrophages and breast carcinoma cells is modulated by cytokines and chemokines, we profiled secretions of human monocytes to identify key cytokines/chemokines secreted by human monocytes that may induce motility and invasion of IBC cells. Our results showed that human monocytes secrete IL-8 that promotes invasiveness of IBC carcinoma cells via stimulation of PI3K/Akt signaling pathway and increasing the expression of the mesenchymal marker fibronectin [42].
We were interested in studying whether monocytes/macrophages contribute to IBC cancer progression. Using immunohistochemical techniques, we found that monocytes/macrophages highly infiltrate IBC carcinoma tissues and localized around tumor emboli. Moreover, we recorded a cross talk between IBC tumor emboli and surrounding monocytes/macrophages. Within patients, carcinoma tissues tumor emboli are oriented toward monocytes/macrophages (Mohamed et al., unpublished data). We found that influx of macrophages within the IBC tumor microenvironment correlated with increase in the number of positive lymph node metastasis (unpublished data), expression, and activity of proteases such as CTSB [62] and MMP-2 and -9 [63]. Thus, in IBC carcinoma tissues, TAM may secrete cytokines/chemokines that induce invasiveness and expression of proteases by IBC cells. Depending on our results and previously published studies, we suggested that cytokines and proteases may have a role in IBC progression.
Role of proteases in the dissemination of IBC cells
Cancer cells secrete proteases, such as cysteine cathepsins, which enable them to invade and metastasize via degrading extracellular matrix proteins and basement membranes [64]. Protease can act directly or indirectly by activating other proteases through a cascade reaction [65]. Proteases also modulate secretion and activity of cytokines that influence invasive and metastatic behavior of cancer cells [66]. Within breast tumor microenvironment, the cross talk between cell–cell and cell-matrix is modulated by a network of proteases, growth factors, and cytokines [67]. Sloane and colleagues established a 3D co-culture model known as MAME (mammary architecture and microenvironment engineering) to study proteolysis resulting from interaction between breast cancer cells and stromal cells [68]. Using the MAME model, proteolytic pathways that contribute to the transition of breast cancer from pre-invasive ductal carcinoma in situ (DCIS) to invasive ductal carcinomas (IDCs) were identified [69].
Although the role of proteases in non-IBC is well investigated, their role in IBC is poorly studied. The specific invasiveness properties migration to axillary lymph nodes and distant organs of IBC carcinoma cells postulate an important role for the contribution of proteins associated with degradation of extracellular matrix, cell motility, and metastasis [70]. Indeed, cell surface proteins caveolin-1 and -2 the structural proteins of cell surface lipid raft caveolae are linked to IBC disease [70]. In cancer cells, caveolae serve as a home for the inactive proteases [64]. For example, pro-CTSB binds to p11 a light chain of the annexin II heterotetramer. Such binding seems to facilitate conversion of procathepsin B to its active forms. Active CTSB imitates a cascade pericellular proteolytic activity at cancer cell surface [71–73]. CTSB is a member of the cysteine proteases family involved in various steps of cancer invasion, motility and dissemination by digestion of adhesion molecules, degradation of extracellular matrix and regulation of angiogenesis [64,74]. Furthermore, membrane associated CTSB activates receptor-bound single-chain urokinase-type plasminogen activator (pro-uPA). The active receptor-bound urokinase plasminogen activator (uPA) converts plasminogen, a serine protease, to plasmin which is involved in the degradation of ECM and basement membrane invasion [75]. Plasmin initiates a cascade reaction to activate MMPs such as MMP-1, -3, -12, and -13 which are known to be involved in cancer invasion and metastasis [65].
Using life cell imaging proteolysis assay, we showed that IBC cells SUM149 exhibit pericellular proteolytic activity due to the co localization of active CTSB, uPA, and uPAR in the SUM149 cell surface caveolae. The role of CTSB in IBC carcinoma cells motility and invasion was confirmed by the ability of CTSB inhibitor CA074 to significantly inhibit pericellular proteolysis and invasion by SUM149 cells [76]. Besides, we translated our in vitro studies at clinical level by studying the role of CTSB in IBC cancer disease progression. We detected co-expression of caveolin-1 and CTSB in IBC patients’ carcinoma tissues. In addition, there was a significant correlation between the expression of CTSB and positive metastatic lymph nodes in IBC, a correlation that was not observed in non-IBC patients [62]. Thus, our studies were the first to demonstrate CTSB role in IBC carcinoma cells motility, invasion and lymph node metastasis. Furthermore, we introduced CTSB as a potential prognostic marker for lymph node invasion and metastasis in IBC.
Extensive studies linked MMPs to the invasive and metastatic behavior of a wide variety of malignancies. Levels of distinct MMPs in the tumor tissues or serum of patients with advanced cancer and their role as prognostic indicators in cancer were widely examined [77–80]. Certain MMPs such as gelatinases (MMP-2, MMP-9) have special mechanisms associated with poor prognosis of cancer. For instance, MMP-2 and MMP-9 facilitate invasion and metastasis because they degrade type IV, V, VII, and X collagens as well as fibronectin, which are important constituents of ECM [81–83]. In human solid tumors, including colon, breast, and lung carcinoma and melanoma, MMP-2 and MMP-9 are markedly overexpressed during the invasive and metastatic phases, while they are scarcely present or even absent in hyperplastic or normal tissue and in situ tumors [80,83–86]. Moreover, membrane-type MMPs (MT-MMPs) such as MT1-MMP were found to be strongly implicated in oncogenesis [87]. MT1-MMP is localized at invasive edges of the tumors and specialized membrane extensions known as invadopodia, where ECM degradation and cellular invasion can occur [88]. Stages of breast cancer progression are accompanied by an increase in the expression of MT1-MMP, MMP-2, and MMP-9 and suggested to be a predictive biomarker for disease aggressiveness, invasiveness, and poor prognosis [56,79,89,90]. Although MMPs are probably important mediators for the invasiveness, motility, and metastatic potential of non-IBC [82,91], their role in IBC is not well identified.
We compared the expression of MMPs (MT1-MMP, MMP2, and MMP-9) in IBC versus non-IBC patients in an attempt to provide a more validated data on the biological behavior of IBC phenotype. We detected increased expression of MT1-MMP, MMP-2, and MMP-9 in aggressive phenotype IBC compared to non-IBC. Furthermore, MT1-MMP in IBC carcinoma tissues correlates with pro-MMP-2 and pro-MMP-9 expression and the activity of MMP-2, while in non-IBC, expression of MT1-MMP correlates with expression of pro-MMP-9. Our study suggested for the first time that MT1-MMP may play an essential role in IBC progression either directly through promoting cell motility or indirectly by inducing the expression of pro-MMP-2 and pro-MMP-9 and activation of MMP-2 [63].
Cytokines/chemokines regulate IBC disease progression
Although cross talks between cells within the tumor microenvironment are modulated by soluble mediators such as cytokines/chemokines, the role of cytokines/chemokines in IBC is not well investigated and more studies are warranted. Using different experimental models, few studies cited the role of cytokines in IBC progression. For instance, the canine inflammatory mammary cancer model, which is a typical form of IBC, characterized by high serum levels of IL-6, IL-8, and IL-10 compared to canine non-inflammatory malignant mammary cancer [92]. Similarly, IBC cell line SUM149 and SUM190 secrete IL-6 and IL-8 cytokines that augment self-renewal of stem cells via Notch signaling pathway [93]. A nearly study which measured the level of cytokines in IBC patients found that IBC carcinoma tissues are characterized by over-expression of IL-6 [94]. In addition, serum IL-6 in IBC patients was significantly high compared to non-IBC patients [95].
IL-6 is a pleiotropic cytokine with multiple biological functions in breast tumor microenvironment. IL-6 promotes tumor growth by stimulating tumor cell proliferation via antiapoptotic response (for review see [96,97]). Furthermore, IL-6 augments breast carcinoma cell invasion and motility [98] and thus may induce dissemination of IBC carcinoma cells. Molecular studies using cDNA microarray identified up-regulation of NF-κB signaling pathways related cytokines, such as IL-8 and its receptors CXCR1/2 in IBC carcinoma tissues [99]. Using in vitro 3D models, we found that human monocytes secrete IL-8 that promotes invasion and motility of IBC carcinoma cells via stimulation of PI3 K/Akt signaling pathway and thus increase the expression of the mesenchymal marker fibronectin [42]. In addition, IL-8 modulates survival of breast cancer stem cells, and IL8/CXCR1 axis is involved in their invasiveness [100]. Recent studies using an in vitro model showed that bone marrow mesenchymal cells secrete inflammatory mediators such as IL-6 and IL-8 that interact with specific receptors stimulating cancer stem cells (CSC) self-renewal of IBC cells SUM149 [101]. Taking in consideration, high metastatic behavior and aggressiveness of IBC disease assumed to be due to stem cell phenotype within tumor emboli [41], cytokines may play a prominent role in inducing stemness of IBC. In summary, only few studies discussed the role of cytokines/chemokines in IBC (Table 1) and more studies are essential to define their role.
Table 1.
The major cytokines and chemokines and their roles in non-IBC and IBC.
| Cytokines | Role | References |
|---|---|---|
| Interleukins (IL) | ||
| IL-1 | IL-1 β involved in breast cancer progression and relapse | [129,130] |
| IL-1 is a potential inducer of IL-8 production by breast cancer cells in vitro | [130] | |
| IL-6 | Contributes to the tumor proliferation via up-regulating antiapoptotic and angiogenic response | [31,94,131] |
| Produced by IBC cell lines (SUM149 and SUM190) stimulate Notch signaling that induces self-renewal pathways of cancer stem cell | [93] | |
| IL-8 | Has been identified as an angiogenic stimulator | [132] |
| Promotes invasion and motility of IBC carcinoma cells by inducing of PI3 k/Akt signaling pathway and increasing the expression of the mesenchymal marker fibronectin | [42] | |
| IBC cell lines (SUM149 and SUM190) secrets IL-8 that promotes cancer stem cell self-renewal pathways through Notch signaling | [93] | |
| IL-10 | Production of IL-10 has been linked to chronicinfection with Mouse Mammary Tumor Virus (MMTV), which related to IBC aggressiveness and etiopathogenesis | [92,109,133] |
| TNF-α | Contributes to epithelial mesenchymal transition (EMT) in breast tumor cells | [134,135] |
| Act as a mediator for IL-6 and IL-8 production | [93] | |
| Induce NF-B signaling pathway activation in stem-like phenotype | [136] | |
| MCP-1 or CCL2 | Promotes breast tumor growth and metastasis | [137] |
| CCL2 and CCL5 are up-regulated by TNF-α and IL-1βin breast cancer cells | [138] | |
A recent study which integrated the results of 3 Affymetrix expression arrays found that TGF-β signaling pathway is suppressed in IBC carcinoma tissues compared to non-IBC [17]. In breast cancer, TGF-β signaling switches breast cancer cells from adhered to single cell motility [102]. Interestingly, adhered carcinoma cells were found to metastasize through lymphatic vessels rather than blood vessels [102,103]. Thus, attenuation of TGF-β signaling pathway may contribute to tumor emboli formation and lymphatic invasion of IBC carcinoma cells [17].
HCMV infection as a factor contribute to IBC disease etiology
The involvement of viruses such as human papillomavirus (HPV) [104], mouse mammary tumor-like viruses (MMTV) [105], a provirus structure with 96% homology with MMTV known as human mammary tumor virus (HMTV) [106], epstein-Barr virus (EBV) [107], and HCMV [108] in breast carcinogenesis was suggested before by different investigations. The involvement of viral infection in IBC was suggested by Pogo and colleagues when they detected HMTV (MMTV-related virus) in 71% of IBC cases compared to 40% of non-IBC cases in American patients [109].
Although studies suggested that pollution, environmental factors, viral infection, and modern lifestyle may have a great impact on the manifestation of different forms of cancer in the Egyptian population including breast cancer [107,110–113] little is known about the underlying cause of IBC, particularly its rapid and wild presentation. The unique phenotype of IBC exhibits properties associated with HCMV infection including secretions of cytokines and proteases that induce cellular migration, angiogenesis [114] and activation of NF-κB signaling pathway, a specific pathway found to be induced by HCMV infection [115].
Investigating the role of HCMV in cancer etiology is recently recommended by different studies after the development of advanced and sensitive laboratory techniques which can detect virus genome, protein, and secretome in cancer tissues [116]. High levels of human cytomegalovirus were detected in newly diagnosed [117] and metastatic breast cancer patients [118]. Furthermore, HCMV proteins and DNA were detected in breast ductal carcinoma in situ and infiltrating ductal carcinoma, suggesting a role of HCMV in breast carcinogenesis [108]. HCMV infection induces production of several cytokines and chemokines such as IL-1, IL-6, IL-8, IL-10, interferon beta (IFN-b), transforming growth factor (TGF)-b, monocyte chemotactic protein (MCP)-1, macrophage inflammatory protein (MIP)-1a, MIP-1b, and RANTES (regulated on activation, normal, T-cell expressed, and secreted) [119]. Furthermore, elevated HCMV IgG levels are linked with mortality, and this association is largely explained by elevated IL-6 and TNF-α [120].
We screened for HCMV infection in non-IBC versus IBC patients [121]. Serological diagnosis indicates that HCMV high antibody titer is higher in IBC versus non-IBC, which agrees with other studies that detected high antibody titer of HCMV in patients newly diagnosed with breast cancer [117]. Furthermore, using nested PCR, we screened for HCMV-DNA in postsurgical cancer and non-cancer breast tissue of non-IBC and IBC patients and healthy volunteers’ tissues obtained from mammoplasty. Our results revealed that HCMV-DNA was detected in cancer tissues of IBC and not in adjacent non-cancer tissues, the results were statistically significant compared to non-IBC patients group. Interestingly, sequence analysis of the detected HCMV-DNA fragment revealed that HCMV infected IBC tissues possess different HCMV strains when compared to infected non-IBC tissues. Polymorphism among HCMV strains may provide important clinical information on the involvement of HCMV in IBC disease etiology [121].
Moreover, we tested whether HCMV infection may modulate the expression and activation of transcriptional factor NF-B/p65 (which controls secretion of different cytokines) in non-IBC versus IBC carcinoma tissues. We found that HCMV infected IBC cancer tissues enhance the expression and activation (phosphorylation) of NF-κB/p65 signaling molecules in IBC patients versus non-IBC patients, this suggests oncomodulatory role for HCMV in IBC and not in non-IBC carcinoma tissues. Thus we demonstrated for the first time that HCMV infection may be associated with the etiology and the progression of IBC versus non-IBC.
Treatment of IBC
Although IBC is the most lethal form of breast cancer affecting young women, there is insufficient evidence from prospective randomized clinical trials for an optimal management for those patients. However, over the past 2 decades, different studies led to the accord that all those patients with primary IBC should receive systemic chemotherapy followed by breast cancer surgery and radiation therapy.
IBC treatment strategies showed that a combination of a taxane and anthracycline increase the response rate to primary systemic chemotherapy, and improves prognosis and efficacy in the neoadjuvant treatment of IBC [122]. As regard targeted therapy, trastuzumab was investigated in 5 prospective clinical trials with systemic chemotherapy for locally advanced breast cancer, including IBC [123].
In these studies of the combination of neo- adjuvant chemotherapies and trastuzumab, they validated the success of trastuzumab in combined systemic chemotherapy regimens for HER2-positive breast cancer, proposing that trastuzumab may be an essential drug in such regimens for patients with HER2-positive IBC. Moreover, in the Neoadjuvant Herceptin (NOAH) trial, patients with high-risk, human epidermal growth factor receptor-2 (HER2+) positive locally advanced or inflammatory breast cancer were randomly allocated to receive preoperative chemotherapy plus trastuzumab followed by completion of a total of 1 year of adjuvant trastuzumab versus the same regimen of preoperative chemotherapy alone. The rate of pCR was doubled in the trastuzumab arm compared with the chemotherapy alone arm [123]. Following the neoadjuvant therapy and for those patients whose disease responded to the systemic treatment, mastectomy with axillary lymph node dissection is shown to be the standard of care and improves the local control rate and survival duration [124].
Negative surgical margins should be the target during surgery as those with negative margins showed better prognosis rather than those with positive margins [125,126]. Although sentinel lymph node biopsy (SLNB) is the standard of care for evaluating axillary lymph node status in patients with early breast cancer, it is not recommended for patients with IBC due to the lymphatic blockage by tumor emboli which is a feature of IBC, and such blockage could prevent the dye or radioactive isotope used from being carried to sentinel lymph nodes. Due to the high rate of dermal lymphatic invasion and the need for post mastectomy radiation therapy, skin-sparing mastectomy or immediate reconstruction is not recommended.
For patients who do not respond to induction chemotherapy, radiation therapy should be considered and patients should be re-evaluated for operability. All women with IBC who undergo a modified radical mastectomy are recommended to receive postmastectomy radiation therapy. Accelerated Hyper fractionated Radiation Therapy achieves better local control than standard radiation therapy for this aggressive disease. One reason that tumors develop resistance to standard radiation therapy is the rapid repopulation of IBC tumor cells between radiation doses. Since there is a high probability for the involvement of locoregional lymph nodes, which would envisage a high likelihood of locoregional recurrence, it is recommended that radiation therapy also involve these regions including the supraclavicular regions and internal mammary lymph nodes [126]. It is also recommended that the cumulative radiation dose be escalated to 66 Gy in the subset of women who are under age of 45 years, have close or positive surgical margins and have four or more positive lymph nodes following preoperative systemic treatment, or who demonstrated a poor response preoperative systemic treatment [126]. Five years of hormonal treatment with either tamoxifen or an aromatase inhibitor depending on their menopausal status is recommended for all patients showing positive hormone receptors [123].
Targeting inflammatory mediators and associated signaling pathways in IBC is currently being used in pre-clinical phases. For example targeting inflammatory cytokines secreted by IBC carcinoma cells and tumor microenvironment showed that using Notch inhibitor RO4929097 down-regulates expression of the inflammatory cytokines IL-6 and IL-8 and reduces self-renewal properties of IBC stem cells [93]. Drygin and colleagues showed that targeting protein kinase CK2 using the CK2 inhibitor CX-4945 down regulate the expression of IL-6 in tissue culture models and inflammatory breast cancer SUM-149PT xenografts mice models. Translating their findings as clinical trial, their results showed that in phase I clinical trial with CX-4945 delivered as oral tablets to IBC patients reduced IL-6 plasma level of IBC patients [95]. On the other hand, treatment of IBC cell line SUM149 with anti-inflammatory vitamin D calcitriol reduces motility, invasion and tumor spheroid formation of IBC cells [127]. Vitamin D is known to inhibit cytokine secretions in adipocytes [127] ovarian and endometrial cancer cells [128] via inactivating NF-B signaling pathway. Indeed, previous studies suggested the importance of targeting inflammatory cytokines and new treatment strategies for IBC patients.
Conclusions
IBC disease biology is complex; recent investigations introduced new cellular and molecular factors that may contribute to IBC progression. Investigations showed that cytokines, proteases, and viral infection were found to play a crucial role in IBC disease progression (Fig. 1). In fact, preclinical studies targeting new molecules associated with IBC progression are essential for the management of this aggressive type of disease.
Fig. 1.
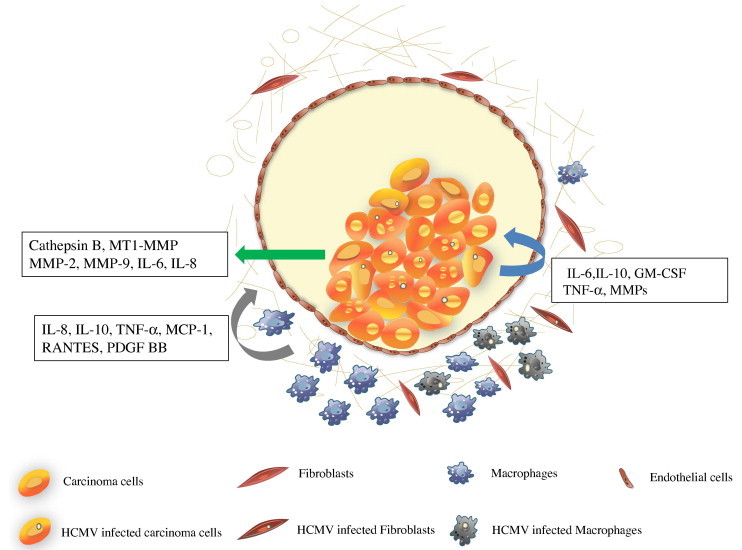
Tumor emboli of IBC, showing carcinoma cells (green arrow) secrete proteases and cytokines that facilitate extracellular matrix degradation, invasion and motility. TAM (gray arrow) secrete cytokines, chemokines, growth factors induce immunosuppression and dissemination of carcinoma cells. HCMV infected cells (blue arrow) secrete cytokines and proteases associated with angiogenesis, immunosuppression, invasion and motility of IBC carcinoma cells.
Conflict of interest
The authors have declared no conflict of interest.
Compliance with Ethics Requirements
This article does not contain any studies with human or animal subjects.
Acknowledgments
We acknowledge the contribution of Ebithal A. Elghonaimy a freelance graphic designer for drawing the cartoon. Author M.M.M. is supported by Avon Foundation, USA and Cairo University Scientific Research Sector.
Biographies

Mona M. Mohamed research interest is studying the interactions in breast cancer among inflammatory macrophages, their associated cytokines and proteolytic enzymes. She is also interesting in determining if a strongly suspected viral infection plays a prominent in the etiology of inflammatory breast cancer. Her ultimate goal is to understand mechanisms molecular mechanisms that may induce breast cancer progression and identifying novel targets for drug development.

Diaa Al-Raawi is assistant lecturer in Sana’a University, Yemen. After finishing her undergraduate studies at University of Sana’a and obtaining her Bachelor degree of Science in 2003, she worked as instructor in Sana’a University. She has been awarded a postgraduate fellowship at Cairo University and spent three years 2008–2011 as postgraduate student at Cancer Biology Laboratory, Faculty of Science, Cairo University – Egypt. Her thesis focused on examine the expression of hormone receptors, human epidermal growth factor receptor-2 (HER-2) and matrix metalloproteinases in an attempt to provide a more validated data on biological features of unique phenotype inflammatory breast cancer (IBC).

Salwa F. Sabet research interest is studying the most prevalent diseases in Egypt as Cancer and Hepatitis C Virus. Regarding cancer, She is interested in studying genetic alterations associated with cancer disease, including epigenetic changes, polymorphism and their contribution in disease progression, which might help in cancer diagnosis and drug development. She is also interested in studying the expression and structure of different proteins of HCV4a in order to determine their active sites for drug development.

Mohamed El-Shinawi M.D., FACS is an Associate Professor of General Surgery – Ain Shams University, Egypt. He is the Lead Trainer of the Sequential Trauma Educational ProgrammS in collaboration with Maryland University, USA. He is the President of AMSRA and a member of a several prestigious associations and societies. He is pursuing a career in breast cancer surgery and research. He also pursues a career in trauma and emergency care and in the area of injury research. His Biography was included in Who’s Who in Medicine and Healthcare. He is an investigator on four projects with different universities in the United States.
Footnotes
Peer review under responsibility of Cairo University.
References
- 1.Lee B.T., Tannenbaum N. Inflammatory carcinoma of the breast: a report of twenty-eight cases from the breast clinic of Memorial Hospital. Surg Gynecol Obste. 1924;39:580–595. [Google Scholar]
- 2.Taylor G., Meltzer A. “Inflammatory carcinoma” of the breast. Am J Cancer. 1938;33:33–49. [Google Scholar]
- 3.Robertson F.M., Bondy M., Yang W., Yamauchi H., Wiggins S., Kamrudin S. Inflammatory breast cancer: the disease, the biology, the treatment. CA Cancer J Clin. 2010;60(6):351–375. doi: 10.3322/caac.20082. [DOI] [PubMed] [Google Scholar]
- 4.Hance K.W., Anderson W.F., Devesa S.S., Young H.A., Levine P.H. Trends in inflammatory breast carcinoma incidence and survival: the surveillance, epidemiology, and end results program at the National Cancer Institute. J Natl Cancer Inst. 2005;97(13):966–975. doi: 10.1093/jnci/dji172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Schairer C., Brown L.M., Mai P.L. Inflammatory breast cancer: high risk of contralateral breast cancer compared to comparably staged non-inflammatory breast cancer. Breast Cancer Res Treat. 2011;129(1):117–124. doi: 10.1007/s10549-010-1324-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Labidi S.I., Mrad K., Mezlini A., Ouarda M.A., Combes J.D., Ben Abdallah M. Inflammatory breast cancer in Tunisia in the era of multimodality therapy. Ann Oncol. 2008;19(3):473–480. doi: 10.1093/annonc/mdm480. [DOI] [PubMed] [Google Scholar]
- 7.Dawood S., Cristofanilli M. Inflammatory breast cancer: what progress have we made? Oncology (Williston Park) 2011;25(3):264–270. 273. [PubMed] [Google Scholar]
- 8.Schairer C., Soliman A.S., Omar S., Khaled H., Eissa S., Ayed F.B. Assessment of diagnosis of inflammatory breast cancer cases at two cancer centers in Egypt and Tunisia. Cancer Med. 2013;2(2):178–184. doi: 10.1002/cam4.48. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Mourali N., Muenz L.R., Tabbane F., Belhassen S., Bahi J., Levine P.H. Epidemiologic features of rapidly progressing breast cancer in Tunisia. Cancer. 1980;46(12):2741–2746. doi: 10.1002/1097-0142(19801215)46:12<2741::aid-cncr2820461234>3.0.co;2-w. [DOI] [PubMed] [Google Scholar]
- 10.Costa J., Webber B.L., Levine P.H., Muenz L., O’Conor G.T., Tabbane F. Histopathological features of rapidly progressing breast carcinoma in Tunisia: a study of 94 cases. Int J Cancer. 1982;30(1):35–37. doi: 10.1002/ijc.2910300107. [DOI] [PubMed] [Google Scholar]
- 11.Dawood S., Merajver S.D., Viens P., Vermeulen P.B., Swain S.M., Buchholz T.A. International expert panel on inflammatory breast cancer: consensus statement for standardized diagnosis and treatment. Ann Oncol. 2010;22(3):515–523. doi: 10.1093/annonc/mdq345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Chang S., Parker S.L., Pham T., Buzdar A.U., Hursting S.D. Inflammatory breast carcinoma incidence and survival: the surveillance, epidemiology, and end results program of the National Cancer Institute, 1975–1992. Cancer. 1998;82(12):2366–2372. [PubMed] [Google Scholar]
- 13.Cristofanilli M., Buzdar A.U., Hortobagyi G.N. Update on the management of inflammatory breast cancer. Oncologist. 2003;8(2):141–148. doi: 10.1634/theoncologist.8-2-141. [DOI] [PubMed] [Google Scholar]
- 14.Lerebours F., Bieche I., Lidereau R. Update on inflammatory breast cancer. Breast Cancer Res. 2005;7(2):52–58. doi: 10.1186/bcr997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Walshe J.M., Swain S.M. Clinical aspects of inflammatory breast cancer. Breast Dis. 2005;22:35–44. doi: 10.3233/bd-2006-22105. [DOI] [PubMed] [Google Scholar]
- 16.Yamauchi H., Woodward W.A., Valero V., Alvarez R.H., Lucci A., Buchholz T.A. Inflammatory breast cancer: what we know and what we need to learn. Oncologist. 2012;17(7):891–899. doi: 10.1634/theoncologist.2012-0039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Van Laere S.J., Ueno N.T., Finetti P., Vermeulen P.B., Lucci A., Robertson F.M. Uncovering the molecular secrets of Inflammatory Breast Cancer biology: an integrated analysis of three distinct Affymetrix gene expression data sets. Clin Cancer Res. 2013;19(17):4685–4696. doi: 10.1158/1078-0432.CCR-12-2549. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Delarue J.C., May-Levin F., Mouriesse H., Contesso G., Sancho-Garnier H. Oestrogen and progesterone cytosolic receptors in clinically inflammatory tumours of the human breast. Br J Cancer. 1981;44(6):911–916. doi: 10.1038/bjc.1981.291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Jaiyesimi I.A., Buzdar A.U., Hortobagyi G. Inflammatory breast cancer: a review. J Clin Oncol. 1992;10(6):1014–1024. doi: 10.1200/JCO.1992.10.6.1014. [DOI] [PubMed] [Google Scholar]
- 20.Palangie T., Mosseri V., Mihura J., Campana F., Beuzeboc P., Dorval T. Prognostic factors in inflammatory breast cancer and therapeutic implications. Eur J Cancer. 1994;30A(7):921–927. doi: 10.1016/0959-8049(94)90115-5. [DOI] [PubMed] [Google Scholar]
- 21.Zell J.A., Tsang W.Y., Taylor T.H., Mehta R.S., Anton-Culver H. Prognostic impact of human epidermal growth factor-like receptor 2 and hormone receptor status in inflammatory breast cancer (IBC): analysis of 2,014 IBC patient cases from the California Cancer Registry. Breast Cancer Res. 2009;11(1):R9. doi: 10.1186/bcr2225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.van Golen K.L., Davies S., Wu Z.F., Wang Y., Bucana C.D., Root H. A novel putative low-affinity insulin-like growth factor-binding protein, LIBC (lost in inflammatory breast cancer), and RhoC GTPase correlate with the inflammatory breast cancer phenotype. Clin Cancer Res. 1999;5(9):2511–2519. [PubMed] [Google Scholar]
- 23.Kleer C.G., Zhang Y., Pan Q., van Golen K.L., Wu Z.F., Livant D. WISP3 is a novel tumor suppressor gene of inflammatory breast cancer. Oncogene. 2002;21(20):3172–3180. doi: 10.1038/sj.onc.1205462. [DOI] [PubMed] [Google Scholar]
- 24.Gong Y. Pathologic aspects of inflammatory breast cancer: part 2. Biologic insights into its aggressive phenotype. Semin Oncol. 2008;35(1):33–40. doi: 10.1053/j.seminoncol.2007.11.014. [DOI] [PubMed] [Google Scholar]
- 25.Kleer C.G., van Golen K.L., Braun T., Merajver S.D. Persistent E-cadherin expression in inflammatory breast cancer. Mod Pathol. 2001;14(5):458–464. doi: 10.1038/modpathol.3880334. [DOI] [PubMed] [Google Scholar]
- 26.Bonnier P., Charpin C., Lejeune C., Romain S., Tubiana N., Beedassy B. Inflammatory carcinomas of the breast: a clinical, pathological, or a clinical and pathological definition? Int J Cancer. 1995;62(4):382–385. doi: 10.1002/ijc.2910620404. [DOI] [PubMed] [Google Scholar]
- 27.Kowalski P.J., Rubin M.A., Kleer C.G. E-cadherin expression in primary carcinomas of the breast and its distant metastases. Breast Cancer Res. 2003;5(6):R217–R222. doi: 10.1186/bcr651. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Kleer C.G., Zhang Y., Pan Q., Gallagher G., Wu M., Wu Z.F. WISP3 and RhoC guanosine triphosphatase cooperate in the development of inflammatory breast cancer. Breast Cancer Res. 2004;6(2) doi: 10.1186/bcr755. R110-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.van Golen K.L., Bao L., DiVito M.M., Wu Z., Prendergast G.C., Merajver S.D. Reversion of RhoC GTPase-induced inflammatory breast cancer phenotype by treatment with a farnesyl transferase inhibitor. Mol Cancer Ther. 2002;1(8):575–583. [PubMed] [Google Scholar]
- 30.van Golen K.L., Bao L.W., Pan Q., Miller F.R., Wu Z.F., Merajver S.D. Mitogen activated protein kinase pathway is involved in RhoC GTPase induced motility, invasion and angiogenesis in inflammatory breast cancer. Clin Exp Metast. 2002;19(4):301–311. doi: 10.1023/a:1015518114931. [DOI] [PubMed] [Google Scholar]
- 31.van Golen K.L., Wu Z.F., Qiao X.T., Bao L., Merajver S.D. RhoC GTPase overexpression modulates induction of angiogenic factors in breast cells. Neoplasia. 2000;2(5):418–425. doi: 10.1038/sj.neo.7900115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.van Golen K.L., Wu Z.F., Qiao X.T., Bao L.W., Merajver S.D. RhoC GTPase, a novel transforming oncogene for human mammary epithelial cells that partially recapitulates the inflammatory breast cancer phenotype. Cancer Res. 2000;60(20):5832–5838. [PubMed] [Google Scholar]
- 33.Van den Eynden G.G., Van der Auwera I., Van Laere S., Colpaert C.G., van Dam P., Merajver S. Validation of a tissue microarray to study differential protein expression in inflammatory and non-inflammatory breast cancer. Breast Cancer Res Treat. 2004;85(1):13–22. doi: 10.1023/B:BREA.0000021028.33926.a8. [DOI] [PubMed] [Google Scholar]
- 34.Kleer C.G., van Golen K.L., Zhang Y., Wu Z.F., Rubin M.A., Merajver S.D. Characterization of RhoC expression in benign and malignant breast disease: a potential new marker for small breast carcinomas with metastatic ability. Am J Pathol. 2002;160(2):579–584. doi: 10.1016/S0002-9440(10)64877-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Kleer C.G., Griffith K.A., Sabel M.S., Gallagher G., van Golen K.L., Wu Z.F. RhoC-GTPase is a novel tissue biomarker associated with biologically aggressive carcinomas of the breast. Breast Cancer Res Treat. 2005;93(2):101–110. doi: 10.1007/s10549-005-4170-6. [DOI] [PubMed] [Google Scholar]
- 36.Wu M., Wu Z.F., Kumar-Sinha C., Chinnaiyan A., Merajver S.D. RhoC induces differential expression of genes involved in invasion and metastasis in MCF10A breast cells. Breast Cancer Res Treat. 2004;84(1):3–12. doi: 10.1023/B:BREA.0000018426.76893.21. [DOI] [PubMed] [Google Scholar]
- 37.Lerebours F., Cizeron-Clairac G., Susini A., Vacher S., Mouret-Fourme E., Belichard C. miRNA expression profiling of inflammatory breast cancer identifies a 5-miRNA signature predictive of breast tumor aggressiveness. Int J Cancer. 2013;133(7):1614–1623. doi: 10.1002/ijc.28171. [DOI] [PubMed] [Google Scholar]
- 38.Tsoi D.T., Rowsell C., McGregor C., Kelly C.M., Verma S., Pritchard K.I. Disseminated tumor embolism from breast cancer leading to multiorgan failure. J Clin Oncol. 2010;28(12) doi: 10.1200/JCO.2009.25.1009. e180-3. [DOI] [PubMed] [Google Scholar]
- 39.Morales J., Alpaugh M.L. Gain in cellular organization of inflammatory breast cancer: a 3D in vitro model that mimics the in vivo metastasis. BMC Cancer. 2009;9:462. doi: 10.1186/1471-2407-9-462. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Mahooti S., Porter K., Alpaugh M.L., Ye Y., Xiao Y., Jones S. Breast carcinomatous tumoral emboli can result from encircling lymphovasculogenesis rather than lymphovascular invasion. Oncotarget. 2010;1(2):131–147. doi: 10.18632/oncotarget.117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Xiao Y., Ye Y., Yearsley K., Jones S., Barsky S.H. The lymphovascular embolus of inflammatory breast cancer expresses a stem cell-like phenotype. Am J Pathol. 2008;173(2):561–574. doi: 10.2353/ajpath.2008.071214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Mohamed M.M. Monocytes conditioned media stimulate fibronectin expression and spreading of inflammatory breast cancer cells in three-dimensional culture: a mechanism mediated by IL-8 signaling pathway. Cell Commun Signal. 2012;10(1):3. doi: 10.1186/1478-811X-10-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Lehman H.L., Dashner E.J., Lucey M., Vermeulen P., Dirix L., Van Laere S. Modeling and characterization of inflammatory breast cancer emboli grown in vitro. Int J Cancer. 2012 doi: 10.1002/ijc.27928. [DOI] [PubMed] [Google Scholar]
- 44.Pollard J.W. Macrophages define the invasive microenvironment in breast cancer. J Leukoc Biol. 2008;84(3):623–630. doi: 10.1189/jlb.1107762. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Mukhtar R.A., Nseyo O., Campbell M.J., Esserman L.J. Tumor-associated macrophages in breast cancer as potential biomarkers for new treatments and diagnostics. Expert Rev Mol Diagn. 2011;11(1):91–100. doi: 10.1586/erm.10.97. [DOI] [PubMed] [Google Scholar]
- 46.Aaltomaa S., Lipponen P., Eskelinen M., Kosma V.M., Marin S., Alhava E. Lymphocyte infiltrates as a prognostic variable in female breast cancer. Eur J Cancer. 1992;28A(4–5):859–864. doi: 10.1016/0959-8049(92)90134-n. [DOI] [PubMed] [Google Scholar]
- 47.Georgiannos S.N., Renaut A., Goode A.W., Sheaff M. The immunophenotype and activation status of the lymphocytic infiltrate in human breast cancers, the role of the major histocompatibility complex in cell-mediated immune mechanisms, and their association with prognostic indicators. Surgery. 2003;134(5):827–834. doi: 10.1016/s0039-6060(03)00292-7. [DOI] [PubMed] [Google Scholar]
- 48.Pollard J.W. Trophic macrophages in development and disease. Nat Rev Immunol. 2009;9(4):259–270. doi: 10.1038/nri2528. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Ojalvo L.S., King W., Cox D., Pollard J.W. High-density gene expression analysis of tumor-associated macrophages from mouse mammary tumors. Am J Pathol. 2009;174(3):1048–1064. doi: 10.2353/ajpath.2009.080676. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Mosser D.M., Edwards J.P. Exploring the full spectrum of macrophage activation. Nat Rev Immunol. 2008;8(12):958–969. doi: 10.1038/nri2448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Lewis C.E., Leek R., Harris A., McGee J.O. Cytokine regulation of angiogenesis in breast cancer: the role of tumor-associated macrophages. J Leukoc Biol. 1995;57(5):747–751. doi: 10.1002/jlb.57.5.747. [DOI] [PubMed] [Google Scholar]
- 52.Leek R.D., Lewis C.E., Whitehouse R., Greenall M., Clarke J., Harris A.L. Association of macrophage infiltration with angiogenesis and prognosis in invasive breast carcinoma. Cancer Res. 1996;56(20):4625–4629. [PubMed] [Google Scholar]
- 53.Condeelis J., Pollard J.W. Macrophages: obligate partners for tumor cell migration, invasion, and metastasis. Cell. 2006;124(2):263–266. doi: 10.1016/j.cell.2006.01.007. [DOI] [PubMed] [Google Scholar]
- 54.Mantovani A., Schioppa T., Porta C., Allavena P., Sica A. Role of tumor-associated macrophages in tumor progression and invasion. Cancer Metast Rev. 2006;25(3):315–322. doi: 10.1007/s10555-006-9001-7. [DOI] [PubMed] [Google Scholar]
- 55.Hagemann T., Wilson J., Kulbe H., Li N.F., Leinster D.A., Charles K. Macrophages induce invasiveness of epithelial cancer cells via NF-kappa B and JNK. J Immunol. 2005;175(2):1197–1205. doi: 10.4049/jimmunol.175.2.1197. [DOI] [PubMed] [Google Scholar]
- 56.Gonzalez L.O., Pidal I., Junquera S., Corte M.D., Vazquez J., Rodriguez J.C. Overexpression of matrix metalloproteinases and their inhibitors in mononuclear inflammatory cells in breast cancer correlates with metastasis-relapse. Br J Cancer. 2007;97(7):957–963. doi: 10.1038/sj.bjc.6603963. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.O’Sullivan C., Lewis C.E., Harris A.L., McGee J.O. Secretion of epidermal growth factor by macrophages associated with breast carcinoma. Lancet. 1993;342(8864):148–149. doi: 10.1016/0140-6736(93)91348-p. [DOI] [PubMed] [Google Scholar]
- 58.Van Laere S., Van der Auwera I., Van den Eynden G.G., Fox S.B., Bianchi F., Harris A.L. Distinct molecular signature of inflammatory breast cancer by cDNA microarray analysis. Breast Cancer Res Treat. 2005;93(3):237–246. doi: 10.1007/s10549-005-5157-z. [DOI] [PubMed] [Google Scholar]
- 59.Compton T., Kurt-Jones E.A., Boehme K.W., Belko J., Latz E., Golenbock D.T. Human cytomegalovirus activates inflammatory cytokine responses via CD14 and Toll-like receptor 2. J Virol. 2003;77(8):4588–4596. doi: 10.1128/JVI.77.8.4588-4596.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Lewis C.E., Pollard J.W. Distinct role of macrophages in different tumor microenvironments. Cancer Res. 2006;66(2):605–612. doi: 10.1158/0008-5472.CAN-05-4005. [DOI] [PubMed] [Google Scholar]
- 61.Mohamed M.M., Cavallo-Medved D., Sloane B.F. Human monocytes augment invasiveness and proteolytic activity of inflammatory breast cancer. Biol Chem. 2008;389(8):1117–1121. doi: 10.1515/BC.2008.117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Nouh M.A., Mohamed M.M., El-Shinawi M., Shaalan M.A., Cavallo-Medved D., Khaled H.M. Cathepsin B: a potential prognostic marker for inflammatory breast cancer. J Transl Med. 2011;9:1. doi: 10.1186/1479-5876-9-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Al-Raawi D., Abu-El-Zahab H., El-Shinawi M., Mohamed M.M. Membrane type-1 matrix metalloproteinase (MT1-MMP) correlates with the expression and activation of matrix metalloproteinase-2 (MMP-2) in inflammatory breast cancer. Int J Clin Exp Med. 2011;4(4):265–275. [PMC free article] [PubMed] [Google Scholar]
- 64.Mohamed M.M., Sloane B.F. Cysteine cathepsins: multifunctional enzymes in cancer. Nat Rev Cancer. 2006;6(10):764–775. doi: 10.1038/nrc1949. [DOI] [PubMed] [Google Scholar]
- 65.Lee M., Fridman R., Mobashery S. Extracellular proteases as targets for treatment of cancer metastases. Chem Soc Rev. 2004;33(7):401–409. doi: 10.1039/b209224g. [DOI] [PubMed] [Google Scholar]
- 66.Opdenakker G., Van Damme J. Cytokines and proteases in invasive processes: molecular similarities between inflammation and cancer. Cytokine. 1992;4(4):251–258. doi: 10.1016/1043-4666(92)90064-x. [DOI] [PubMed] [Google Scholar]
- 67.Place A.E., Jin Huh S., Polyak K. The microenvironment in breast cancer progression: biology and implications for treatment. Breast Cancer Res. 2011;13(6):227. doi: 10.1186/bcr2912. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Sameni M., Anbalagan A., Olive M.B., Moin K., Mattingly R.R., Sloane B.F. MAME models for 4D live-cell imaging of tumor: microenvironment interactions that impact malignant progression. J Vis Exp. 2012;(60) doi: 10.3791/3661. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Rothberg J.M., Sameni M., Moin K., Sloane B.F. Live-cell imaging of tumor proteolysis: impact of cellular and non-cellular microenvironment. Biochim Biophys Acta. 2012;1824(1):123–132. doi: 10.1016/j.bbapap.2011.07.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Van den Eynden G.G., Van Laere S.J., Van der Auwera I., Merajver S.D., Van Marck E.A., van Dam P. Overexpression of caveolin-1 and -2 in cell lines and in human samples of inflammatory breast cancer. Breast Cancer Res Treat. 2006;95(3):219–228. doi: 10.1007/s10549-005-9002-1. [DOI] [PubMed] [Google Scholar]
- 71.Cavallo-Medved D., Mai J., Dosescu J., Sameni M., Sloane B.F. Caveolin-1 mediates the expression and localization of cathepsin B, pro-urokinase plasminogen activator and their cell-surface receptors in human colorectal carcinoma cells. J Cell Sci. 2005;118(Pt 7):1493–1503. doi: 10.1242/jcs.02278. [DOI] [PubMed] [Google Scholar]
- 72.Cavallo-Medved D., Rudy D., Blum G., Bogyo M., Caglic D., Sloane B.F. Live-cell imaging demonstrates extracellular matrix degradation in association with active cathepsin B in caveolae of endothelial cells during tube formation. Exp Cell Res. 2009;315(7):1234–1246. doi: 10.1016/j.yexcr.2009.01.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Cavallo-Medved D., Sloane B.F. Cell-surface cathepsin B: understanding its functional significance. Curr Top Dev Biol. 2003;54:313–341. doi: 10.1016/s0070-2153(03)54013-3. [DOI] [PubMed] [Google Scholar]
- 74.Ren W.P., Sloane B.F. Cathepsins D and B in breast cancer. Cancer Treat Res. 1996;83:325–352. doi: 10.1007/978-1-4613-1259-8_16. [DOI] [PubMed] [Google Scholar]
- 75.Kobayashi H., Moniwa N., Sugimura M., Shinohara H., Ohi H., Terao T. Effects of membrane-associated cathepsin B on the activation of receptor-bound prourokinase and subsequent invasion of reconstituted basement membranes. Biochim Biophys Acta. 1993;1178(1):55–62. doi: 10.1016/0167-4889(93)90109-3. [DOI] [PubMed] [Google Scholar]
- 76.Victor B.C., Anbalagan A., Mohamed M.M., Sloane B.F., Cavallo-Medved D. Inhibition of cathepsin B activity attenuates extracellular matrix degradation and inflammatory breast cancer invasion. Breast Cancer Res. 2011;13(6):R115. doi: 10.1186/bcr3058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Sternlicht M.D., Werb Z. How matrix metalloproteinases regulate cell behavior. Annu Rev Cell Dev Biol. 2001;17:463–516. doi: 10.1146/annurev.cellbio.17.1.463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Vihinen P., Ala-aho R., Kahari V.M. Matrix metalloproteinases as therapeutic targets in cancer. Curr Cancer Drug Targets. 2005;5(3):203–220. doi: 10.2174/1568009053765799. [DOI] [PubMed] [Google Scholar]
- 79.Kohrmann A., Kammerer U., Kapp M., Dietl J., Anacker J. Expression of matrix metalloproteinases (MMPs) in primary human breast cancer and breast cancer cell lines: new findings and review of the literature. BMC Cancer. 2009;9:188. doi: 10.1186/1471-2407-9-188. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Patel S., Sumitra G., Koner B.C., Saxena A. Role of serum matrix metalloproteinase-2 and -9 to predict breast cancer progression. Clin Biochem. 2011;44(10–11):869–872. doi: 10.1016/j.clinbiochem.2011.04.019. [DOI] [PubMed] [Google Scholar]
- 81.Ray J.M., Stetler-Stevenson W.G. The role of matrix metalloproteases and their inhibitors in tumour invasion, metastasis and angiogenesis. Eur Respir J. 1994;7(11):2062–2072. [PubMed] [Google Scholar]
- 82.Duffy M.J., Maguire T.M., Hill A., McDermott E., O’Higgins N. Metalloproteinases: role in breast carcinogenesis, invasion and metastasis. Breast Cancer Res. 2000;2(4):252–257. doi: 10.1186/bcr65. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Noel A., Gutierrez-Fernandez A., Sounni N.E., Behrendt N., Maquoi E., Lund I.K. New and paradoxical roles of matrix metalloproteinases in the tumor microenvironment. Front Pharmacol. 2012;3:140. doi: 10.3389/fphar.2012.00140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Noh S., Jung J.J., Jung M., Kim T.S., Park C.H., Lim S.J. MMP-2 as a putative biomarker for carcinomatosis in gastric cancer. Hepatogastroenterology. 2011;58(112):2015–2019. doi: 10.5754/hge11209. [DOI] [PubMed] [Google Scholar]
- 85.Damodharan U., Ganesan R., Radhakrishnan U.C. Expression of MMP2 and MMP9 (gelatinases A and B) in human colon cancer cells. Appl Biochem Biotechnol. 2011;165(5–6):1245–1252. doi: 10.1007/s12010-011-9342-8. [DOI] [PubMed] [Google Scholar]
- 86.Gialeli C., Theocharis A.D., Karamanos N.K. Roles of matrix metalloproteinases in cancer progression and their pharmacological targeting. FEBS J. 2011;278(1):16–27. doi: 10.1111/j.1742-4658.2010.07919.x. [DOI] [PubMed] [Google Scholar]
- 87.Sounni N.E., Noel A. Membrane type-matrix metalloproteinases and tumor progression. Biochimie. 2005;87(3–4):329–342. doi: 10.1016/j.biochi.2004.07.012. [DOI] [PubMed] [Google Scholar]
- 88.Noel A., Maillard C., Rocks N., Jost M., Chabottaux V., Sounni N.E. Membrane associated proteases and their inhibitors in tumour angiogenesis. J Clin Pathol. 2004;57(6):577–584. doi: 10.1136/jcp.2003.014472. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Figueira R.C., Gomes L.R., Neto J.S., Silva F.C., Silva I.D., Sogayar M.C. Correlation between MMPs and their inhibitors in breast cancer tumor tissue specimens and in cell lines with different metastatic potential. BMC Cancer. 2009;9:20. doi: 10.1186/1471-2407-9-20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Jiang W.G., Davies G., Martin T.A., Parr C., Watkins G., Mason M.D. Expression of membrane type-1 matrix metalloproteinase, MT1-MMP in human breast cancer and its impact on invasiveness of breast cancer cells. Int J Mol Med. 2006;17(4):583–590. [PubMed] [Google Scholar]
- 91.Kousidou O.C., Roussidis A.E., Theocharis A.D., Karamanos N.K. Expression of MMPs and TIMPs genes in human breast cancer epithelial cells depends on cell culture conditions and is associated with their invasive potential. Anticancer Res. 2004;24(6):4025–4030. [PubMed] [Google Scholar]
- 92.de Andres P.J., Illera J.C., Caceres S., Diez L., Perez-Alenza M.D., Pena L. Increased levels of interleukins 8 and 10 as findings of canine inflammatory mammary cancer. Vet Immunol Immunopathol. 2012 doi: 10.1016/j.vetimm.2012.12.010. [DOI] [PubMed] [Google Scholar]
- 93.Debeb B.G., Cohen E.N., Boley K., Freiter E.M., Li L., Robertson F.M. Pre-clinical studies of Notch signaling inhibitor RO4929097 in inflammatory breast cancer cells. Breast Cancer Res Treat. 2012;134(2):495–510. doi: 10.1007/s10549-012-2075-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Bieche I., Lerebours F., Tozlu S., Espie M., Marty M., Lidereau R. Molecular profiling of inflammatory breast cancer: identification of a poor-prognosis gene expression signature. Clin Cancer Res. 2004;10(20):6789–6795. doi: 10.1158/1078-0432.CCR-04-0306. [DOI] [PubMed] [Google Scholar]
- 95.Drygin D., Ho C.B., Omori M., Bliesath J., Proffitt C., Rice R. Protein kinase CK2 modulates IL-6 expression in inflammatory breast cancer. Biochem Biophys Res Commun. 2011;415(1):163–167. doi: 10.1016/j.bbrc.2011.10.046. [DOI] [PubMed] [Google Scholar]
- 96.Kurebayashi J. Regulation of interleukin-6 secretion from breast cancer cells and its clinical implications. Breast Cancer. 2000;7(2):124–129. doi: 10.1007/BF02967443. [DOI] [PubMed] [Google Scholar]
- 97.Knupfer H., Preiss R. Significance of interleukin-6 (IL-6) in breast cancer (review) Breast Cancer Res Treat. 2007;102(2):129–135. doi: 10.1007/s10549-006-9328-3. [DOI] [PubMed] [Google Scholar]
- 98.Arihiro K., Oda H., Kaneko M., Inai K. Cytokines facilitate chemotactic motility of breast carcinoma cells. Breast Cancer. 2000;7(3):221–230. doi: 10.1007/BF02967464. [DOI] [PubMed] [Google Scholar]
- 99.Lerebours F., Vacher S., Andrieu C., Espie M., Marty M., Lidereau R. NF-kappa B genes have a major role in inflammatory breast cancer. BMC Cancer. 2008;8:41. doi: 10.1186/1471-2407-8-41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Charafe-Jauffret E., Ginestier C., Iovino F., Wicinski J., Cervera N., Finetti P. Breast cancer cell lines contain functional cancer stem cells with metastatic capacity and a distinct molecular signature. Cancer Res. 2009;69(4):1302–1313. doi: 10.1158/0008-5472.CAN-08-2741. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Liu S., Ginestier C., Ou S.J., Clouthier S.G., Patel S.H., Monville F. Breast cancer stem cells are regulated by mesenchymal stem cells through cytokine networks. Cancer Res. 2011;71(2):614–624. doi: 10.1158/0008-5472.CAN-10-0538. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Giampieri S., Manning C., Hooper S., Jones L., Hill C.S., Sahai E. Localized and reversible TGFbeta signalling switches breast cancer cells from cohesive to single cell motility. Nat Cell Biol. 2009;11(11):1287–1296. doi: 10.1038/ncb1973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Giampieri S., Pinner S., Sahai E. Intravital imaging illuminates transforming growth factor beta signaling switches during metastasis. Cancer Res. 2010;70(9):3435–3439. doi: 10.1158/0008-5472.CAN-10-0466. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Glenn W.K., Heng B., Delprado W., Iacopetta B., Whitaker N.J., Lawson J.S. Epstein-Barr virus, human papillomavirus and mouse mammary tumour virus as multiple viruses in breast cancer. PLoS ONE. 2012;7(11):e48788. doi: 10.1371/journal.pone.0048788. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Wang Y., Holland J.F., Bleiweiss I.J., Melana S., Liu X., Pelisson I. Detection of mammary tumor virus env gene-like sequences in human breast cancer. Cancer Res. 1995;55(22):5173–5179. [PubMed] [Google Scholar]
- 106.Liu B., Wang Y., Melana S.M., Pelisson I., Najfeld V., Holland J.F. Identification of a proviral structure in human breast cancer. Cancer Res. 2001;61(4):1754–1759. [PubMed] [Google Scholar]
- 107.Fawzy S., Sallam M., Awad N.M. Detection of Epstein-Barr virus in breast carcinoma in Egyptian women. Clin Biochem. 2008;41(7–8):486–492. doi: 10.1016/j.clinbiochem.2007.12.017. [DOI] [PubMed] [Google Scholar]
- 108.Harkins L.E., Matlaf L.A., Soroceanu L., Klemm K., Britt W.J., Wang W. Detection of human cytomegalovirus in normal and neoplastic breast epithelium. Herpesviridae. 2010;1(1):8. doi: 10.1186/2042-4280-1-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Pogo B.G., Holland JF.., Levine P.H. Human mammary tumor virus in inflammatory breast cancer. Cancer. 2010;116(11 Suppl):2741–2744. doi: 10.1002/cncr.25179. [DOI] [PubMed] [Google Scholar]
- 110.Soliman A.S., Banerjee M., Lo A.C., Ismail K., Hablas A., Seifeldin I.A. High proportion of inflammatory breast cancer in the Population-based Cancer Registry of Gharbiah, Egypt. Egypt. Breast J. 2009;15(4):432–434. doi: 10.1111/j.1524-4741.2009.00755.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Soliman A.S., Lo A.C., Banerjee M., El-Ghawalby N., Khaled H.M., Bayoumi S. Differences in K-ras and p53 gene mutations among pancreatic adenocarcinomas associated with regional environmental pollution. Carcinogenesis. 2007;28(8):1794–1799. doi: 10.1093/carcin/bgm138. [DOI] [PubMed] [Google Scholar]
- 112.Soliman A.S., Vulimiri S.V., Kleiner H.E., Shen J., Eissa S., Morad M. High levels of oxidative DNA damage in lymphocyte DNA of premenopausal breast cancer patients from Egypt. Int J Environ Health Res. 2004;14(2):121–134. doi: 10.1080/0960312042000209534. [DOI] [PubMed] [Google Scholar]
- 113.Soliman A.S., Wang X., Stanley J.D., El-Ghawalby N., Bondy M.L., Ezzat F. Geographical clustering of pancreatic cancers in the Northeast Nile Delta region of Egypt. Arch Environ Contam Toxicol. 2006;51(1):142–148. doi: 10.1007/s00244-005-0154-0. [DOI] [PubMed] [Google Scholar]
- 114.Fiorentini S., Luganini A., Dell’Oste V., Lorusso B., Cervi E., Caccuri F. Human cytomegalovirus productively infects lymphatic endothelial cells and induces a secretome that promotes angiogenesis and lymphangiogenesis through interleukin-6 and granulocyte-macrophage colony-stimulating factor. J Gen Virol. 2011;92(Pt 3):650–660. doi: 10.1099/vir.0.025395-0. [DOI] [PubMed] [Google Scholar]
- 115.Juckem L.K., Boehme K.W., Feire A.L., Compton T. Differential initiation of innate immune responses induced by human cytomegalovirus entry into fibroblast cells. J Immunol. 2008;180(7):4965–4977. doi: 10.4049/jimmunol.180.7.4965. [DOI] [PubMed] [Google Scholar]
- 116.Michaelis M., Doerr H.W., Cinatl J. The story of human cytomegalovirus and cancer: increasing evidence and open questions. Neoplasia. 2009;11(1):1–9. doi: 10.1593/neo.81178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Fagundes C.P., Glaser R., Alfano C.M., Bennett J.M., Povoski S.P., Lipari A.M. Fatigue and herpesvirus latency in women newly diagnosed with breast cancer. Brain Behav Immun. 2011 doi: 10.1016/j.bbi.2011.09.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Breathnach O., Donnellan P., Collins D., McNicholas W., Crown J. Cytomegalovirus pneumonia in a patient with breast cancer on chemotherapy: case report and review of the literature. Ann Oncol. 1999;10(4):461–465. doi: 10.1023/a:1008360927507. [DOI] [PubMed] [Google Scholar]
- 119.Cheeran M.C., Hu S., Yager S.L., Gekker G., Peterson P.K., Lokensgard J.R. Cytomegalovirus induces cytokine and chemokine production differentially in microglia and astrocytes: antiviral implications. J Neurovirol. 2001;7(2):135–147. doi: 10.1080/13550280152058799. [DOI] [PubMed] [Google Scholar]
- 120.Roberts E.T., Haan M.N., Dowd J.B., Aiello A.E. Cytomegalovirus antibody levels, inflammation, and mortality among elderly Latinos over 9 years of follow-up. Am J Epidemiol. 2010;172(4):363–371. doi: 10.1093/aje/kwq177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.El-Shinawi M., Mohamed H.T., El-Ghonaimy E.A., Tantawy M., Younis A., Schneider R.J. Human cytomegalovirus infection enhances NF-kappaB/p65 signaling in inflammatory breast cancer patients. PLoS ONE. 2013;8(2):e55755. doi: 10.1371/journal.pone.0055755. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Cristofanilli M., Gonzalez-Angulo A.M., Buzdar A.U., Kau S.-W., Frye D.K., Hortobagyi G.N. Paclitaxel improves the prognosis in estrogen receptor negative inflammatory breast cancer: the M.D. Anderson Cancer Center experience. Clin. Breast Cancer. 2004;4(6):415–419. doi: 10.3816/cbc.2004.n.004. [DOI] [PubMed] [Google Scholar]
- 123.Gianni L., Eiermann W., Semiglazov V., Manikhas A., Lluch A., Tjulandin S. Neoadjuvant chemotherapy with trastuzumab followed by adjuvant trastuzumab versus neoadjuvant chemotherapy alone, in patients with HER2-positive locally advanced breast cancer (the NOAH trial): a randomised controlled superiority trial with a parallel HER2-negative cohort. Lancet. 2010;375(9712):377–384. doi: 10.1016/S0140-6736(09)61964-4. [DOI] [PubMed] [Google Scholar]
- 124.Hurley J., Doliny P., Reis I., Silva O., Gomez-Fernandez C., Velez P. Docetaxel, cisplatin, and trastuzumab as primary systemic therapy for human epidermal growth factor receptor 2-positive locally advanced breast cancer. J Clin Oncol: Off J Am Soc Clin Oncol. 2006;24(12):1831–1838. doi: 10.1200/JCO.2005.02.8886. [DOI] [PubMed] [Google Scholar]
- 125.Curcio L.D., Rupp E., Williams W.L., Chu D.Z., Clarke K., Odom-Maryon T. Beyond palliative mastectomy in inflammatory breast cancer – a reassessment of margin status. Ann Surg Oncol. 1999;6(3):249–254. doi: 10.1007/s10434-999-0249-3. [DOI] [PubMed] [Google Scholar]
- 126.Bristol I.J., Woodward W.A., Strom E.A., Cristofanilli M., Domain D., Singletary S.E. Locoregional treatment outcomes after multimodality management of inflammatory breast cancer. Int J Radiat Oncol Biol Phys. 2008;72(2):474–484. doi: 10.1016/j.ijrobp.2008.01.039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Hillyer R.L., Sirinvasin P., Joglekar M., Sikes R.A., van Golen K.L., Nohe A. Differential effects of vitamin D treatment on inflammatory and non-inflammatory breast cancer cell lines. Clin Exp Metast. 2012;29(8):971–979. doi: 10.1007/s10585-012-9486-0. [DOI] [PubMed] [Google Scholar]
- 128.Kavandi L., Collier M.A., Nguyen H., Syed V. Progesterone and calcitriol attenuate inflammatory cytokines CXCL1 and CXCL2 in ovarian and endometrial cancer cells. J Cell Biochem. 2012;113(10):3143–3152. doi: 10.1002/jcb.24191. [DOI] [PubMed] [Google Scholar]
- 129.Miller L.J., Kurtzman S.H., Anderson K., Wang Y., Stankus M., Renna M. Interleukin-1 family expression in human breast cancer: interleukin-1 receptor antagonist. Cancer Invest. 2000;18(4):293–302. doi: 10.3109/07357900009012171. [DOI] [PubMed] [Google Scholar]
- 130.Pantschenko A.G., Pushkar I., Miller L.J., Wang Y.P., Anderson K., Peled Z. In vitro demonstration of breast cancer tumor cell sub-populations based on interleukin-1/tumor necrosis factor induction of interleukin-8 expression. Oncol Rep. 2003;10(4):1011–1017. [PubMed] [Google Scholar]
- 131.Angelo L.S., Kurzrock R. Vascular endothelial growth factor and its relationship to inflammatory mediators. Clin Cancer Res. 2007;13(10):2825–2830. doi: 10.1158/1078-0432.CCR-06-2416. [DOI] [PubMed] [Google Scholar]
- 132.Lin Y., Huang R., Chen L., Li S., Shi Q., Jordan C. Identification of interleukin-8 as estrogen receptor-regulated factor involved in breast cancer invasion and angiogenesis by protein arrays. Int J Cancer. 2004;109(4):507–515. doi: 10.1002/ijc.11724. [DOI] [PubMed] [Google Scholar]
- 133.Papiernik M., Do Carmo Leite-de-Moraes M., Pontoux C., Joret A.M., Rocha B., Penit C. T cell deletion induced by chronic infection with mouse mammary tumor virus spares a CD25-positive, IL-10-producing T cell population with infectious capacity. J Immunol. 1997;158(10):4642–4653. [PubMed] [Google Scholar]
- 134.Li C.-W., Xia W., Huo L., Lim S.-O., Wu Y., Hsu J.L. Epithelial–mesenchymal transition induced by TNF-α requires NF-κB-mediated transcriptional upregulation of Twist1. Cancer Res. 2012;72(5):1290–1300. doi: 10.1158/0008-5472.CAN-11-3123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Wu S.-T., Sun G.-H., Hsu C.-Y., Huang C.-S., Wu Y.-H., Wang H.-H. Tumor necrosis factor-α induces epithelial–mesenchymal transition of renal cell carcinoma cells via a nuclear factor kappa B-independent mechanism. Exp Biol Med. 2011;236(9):1022–1029. doi: 10.1258/ebm.2011.011058. [DOI] [PubMed] [Google Scholar]
- 136.Storci G., Sansone P., Mari S., D’Uva G., Tavolari S., Guarnieri T. TNFalpha up-regulates SLUG via the NF-kappaB/HIF1alpha axis, which imparts breast cancer cells with a stem cell-like phenotype. J Cell Physiol. 2010;225(3):682–691. doi: 10.1002/jcp.22264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Qian B.-Z., Li J., Zhang H., Kitamura T., Zhang J., Campion L.R. CCL2 recruits inflammatory monocytes to facilitate breast-tumour metastasis. Nature. 2011;475(7355):222–225. doi: 10.1038/nature10138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Raman D., Baugher P.J., Thu Y.M., Richmond A. Role of chemokines in tumor growth. Cancer Lett. 2007;256(2):137–165. doi: 10.1016/j.canlet.2007.05.013. [DOI] [PMC free article] [PubMed] [Google Scholar]



