Abstract
A review of national and international publications on paragonimiasis in Ecuador, epidemiological records from the Ministry of Public Health and unpublished research data was conducted to summarise the current status of the parasite/disease. The purpose of the review is to educate physicians, policy-makers and health providers on the status of the disease and to stimulate scientific investigators to conduct further research. Paragonimiasis was first diagnosed in Ecuador 94 years ago and it is endemic to both tropical and subtropical regions in 19 of 24 provinces in the Pacific Coast and Amazon regions. Paragonimus mexicanus is the only known species in the country, with the mollusc Aroapyrgus colombiensis and the crabs Moreirocarcinus emarginatus, Hypolobocera chilensis and Hypolobocera aequatorialis being the primary and secondary intermediate hosts, respectively. Recent studies found P. mexicanus metacercariae in Trichodactylus faxoni crabs of the northern Amazon. Chronic pulmonary paragonimiasis is commonly misdiagnosed and treated as tuberculosis and although studies have demonstrated the efficacy of praziquantel and triclabendazole for the treatment of human infections, neither drug is available in Ecuador. Official data recorded from 1978-2007 indicate an annual incidence of 85.5 cases throughout the 19 provinces, with an estimated 17.2% of the population at risk of infection. There are no current data on the incidence/prevalence of infection, nor is there a national control programme.
Keywords: Paragonimus, paragonimiasis, review, epidemiology, zoonoses, Ecuador
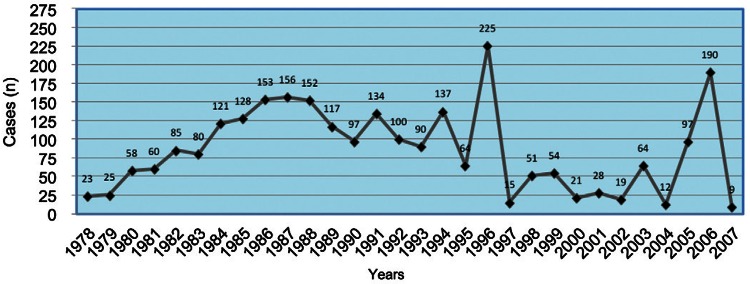
Paragonimiasis, a zoonotic disease caused by several species of Paragonimus, is a food-borne trematodiasis considered by the World Health Organization (WHO) to be one of the most neglected tropical diseases (Savioli & Daumerie 2010). Paragonimus flukes can infect wild and domestic animals in addition to humans following the ingestion of infective metacercariae found in certain species of freshwater crab and crayfish. Human infections are present in Africa, Asia and the Americas, including United States of America, Mexico, Costa Rica, Guatemala, Honduras, Nicaragua, Panama, Colombia, Peru, Venezuela and Brazil (Acha & Szyfres 2003). Ecuador has the highest prevalence of human paragonimiasis in the Americas and the disease is officially considered a public health problem (Díaz et al. 1991). In 1972, following the analysis of weekly epidemiological reports, the Ministry of Public Health (MSP) made it mandatory to report paragonimiasis as a communicable disease. A 1994 WHO report estimated that 21% of the population was at risk for infection based on an expert opinion on the current prevalence of paragonimiasis in Ecuador (Toscano et al. 1994). Estimates in 1998 indicated that approximately 500,000 Ecuadorians could be infected (WHO 2011); however, official reports from the MSP have estimated an average of 85.5 cases per year (Fig. 1) until 2007. This prevalence is lower than expected, which can be attributed to underreporting because human infections occur in rural and remote tropical areas, where infected patients are poor and health services are lacking. Furthermore, the data available at the MSP are from passive records, whereas we have demonstrated in several studies that performing active searches in communities reveals higher rates of diagnosis (Calvopiña et al. 1994, 1995), 1995).
Fig. 1. : the number of human cases registered in 30 years by the Ministry of Public Health. There was a marked increase from 23 cases reported in 1978 to 156 in 1987 and then a gradual decline to nine cases in 2007. The peak counts of 1996 and 2006 are due to active searches in communities by research groups.
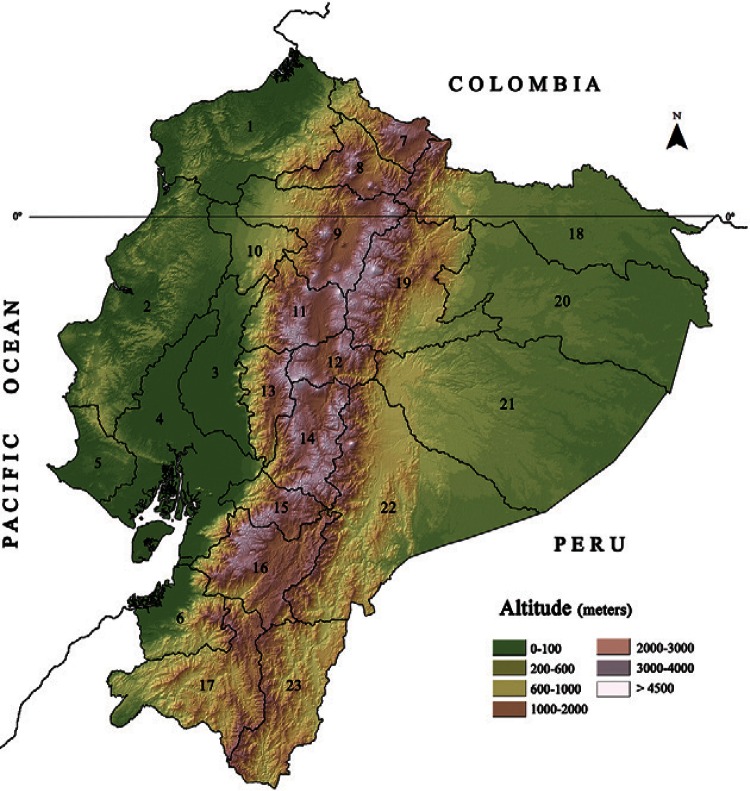
Geographic distribution and prevalence of human infections - Ecuador is located in the northwestern region of South America and is crossed transversely by the equator line and from north to south by the Andes belt, providing three natural regions: the Pacific coastal area, the Amazon to the east and the Andean region in the centre. The first two regions, which comprise 64% of the country, have tropical and subtropical climates, whereas the higher Andean region is cold with warm inter-Andean valleys (Fig. 2).
Fig. 2. : map of Ecuador. The central brown Andes belt marks the Andean region. The Pacific coastal and eastern Amazon regions with tropical climate are in green. The yellow colour corresponds to the foothills of the mountains with subtropical climate. Cases of human paragonimiasis have been reported in all provinces except Galapagos Islands (not showed), Santa Elena (5), Carchi (7), Imbabura (8) and Tungurahua (12). The total country’s area is 283,560 km2. According to 2010 census, there is a total population of 14,483,499 of which 7,236,822 live in the coastal region, 6,449,355 in the Andean, 739,814 in the Amazon and the rest in the Galapagos Islands (INEC 2010). 1: Esmeraldas; 2: Manabí; 3: Los Ríos; 4: Guayas; 6: El Oro; 9: Pichincha; 10: Santo Domingo de los Tsáchilas; 11: Cotopaxi; 13: Bolívar; 14: Chimborazo; 15: Cañar; 16: Azuay; 17: Loja; 18: Sucumbíos; 19: Napo; 20: Orellana; 21: Pastaza; 22: Morona Santiago; 23: Zamora Chinchipe.
Human paragonimiasis in Ecuador was first described in a patient from the coastal region of Chone-Manabí (Heinert 1922). Based on reported clinical cases and epidemiological studies, 511 cases had been documented by 1969 from five coastal and two Amazon provinces (Montalván 1968). From 1972-1976, 316 cases were diagnosed from four coastal provinces (Arzube & Voelker 1978). In 1980, Urrutia reported more than 2,000 cases in the Amazonian provinces (Toscano et al. 1994). Subsequently, foci of infection were identified in the coastal communities of Casacay, Pasaje and Piñas-El Oro, Caluma-Bolívar and Zapallo-Manabí (Palacios et al. 1978, Yokogawa et al. 1983).
Two active field searches of indigenous Chachi communities in the coastal province of Esmeraldas documented the presence of Paragonimus eggs in 7.1% and 30.3% of the sputum samples examined (Paredes et al. 1978, Guevara et al. 1999). From 1988-1991, the MSP recorded 252 cases from various locations in the above province (Vieira et al. 1992). In the Amazon Region, 124 cases were identified in indigenous Quichua and Shuar and in communities of colonists located along the banks of Napo River (Amunárriz 1991a).
Prospective and retrospective studies relying on active and passive searches have identified new foci of transmission. From 1976-1993, 98 cases of pulmonary paragonimiasis were diagnosed in the regional hospital of Latacunga in the province of Cotopaxi. A subsequent active case search between 1992-1993 documented an additional 189 cases from three subtropical cantons: La Maná, Pangua and Sigchos (Calvopiña et al. 1995). In the coastal province of Manabí, 146 patients from the three cantons of El Carmen, Chone and Pedernales were treated in the regional hospital of El Carmen from 1983-1993 (Calvopiña et al. 1994). According to a clinical review (passive search) of 92 cases admitted to the Eugenio Espejo Hospital in Quito, the patients came from communities in both coastal and Amazonia regions (Peñafiel et al. 1981). In subsequent active studies of some of these communities, 216 new cases of pulmonary paragonimiasis were documented (Calvopiña et al. 1998, 2003). According to the presence of human infection, the endemic foci in Ecuador were thus located in rural tropical and subtropical areas in the six Amazonian and five coastal provinces, with extensions to five Andean provinces (Amunárriz 1991a, Calvopiña et al. 1995).
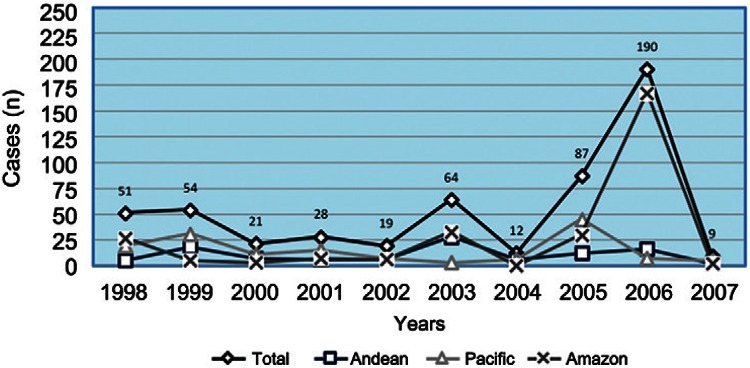
The incidence of paragonimiasis from 1978-2007 according to the records of the MSP is provided in Figs 1, 3, with the incidence from 1998-2007 shown by region. It is important to note that these data represent only patients diagnosed in MSP clinics and hospitals who sought medical assistance (passive search); cases diagnosed in private clinics and hospitals and active cases that were specifically found in remote areas are not considered. Cases are still being diagnosed in hospitals within the endemic areas, with rare migrant cases observed in referral hospitals in non-endemic areas, such as Quito and Guayaquil. Without an official notification, the MSP stopped the obligatory recording of clinical cases of paragonimiasis in 2007.
Fig. 3. : the number of overall cases from 1998-2007 in the three geographical regions as registered by the Ministry of Public Health. There was an annual national average of 53.5 (range 9-190) cases. Based on the size of the population distribution, the highest incidence of cases was in the Amazon Region. In 2006, 166 of the 190 patients came from the province of Sucumbíos.
Life cycle - The parasite - Prior to 1971, it was believed that the infectious Paragonimus species in Ecuador was Paragonimus westermani (Rodríguez 1963). However, a different species was described in 1971 when non-encysted metacercariae were observed in crabs from a coastal subtropical area, Caluma-Bolivar (Yokogawa et al. 1971). These metacercariae were similar to those described in Peru by Miyazaki et al. (1969) and in Mexico by Miyazaki and Ishii (1968). In 1979, Voelker and Arzube (1979) described a new species, Paragonimus ecuadoriensis, after having observed ovary and testis morphologies and egg size and shape that varied from those described for Paragonimus inca, Paragonimus mexicanus and Paragonimus peruvianus. Further comparative studies, however, revealed that P. ecuadoriensis and P. peruvianus were identical to P. mexicanus (Brenes et al. 1980, Miyazaki et al. 1980) and P. ecuadoriensis and P. peruvianus are described as part of P. mexicanus in subsequent publications (Acha & Szyfres 2003). Adult parasites obtained from rats, dogs and cats experimentally infected with metacercariae obtained from Hypolobocera chilensis in Nalpe-Manabí exhibited similar morphological and morphometric characteristics to those of P. mexicanus (Waikagul et al. 2003). A molecular phylogenetic study conducted by sequencing the cytochrome c oxidase subunit 1 and the second internal transcribed spacer genes revealed that isolated metacercariae from Hypolobocera aequatorialis from Concordia-Esmeraldas belonged to P. mexicanus. This finding was later confirmed in adult worms obtained from experimentally infected cats (Iwagami et al. 2003). In the Amazon, studies of metacercariae from Mo- reirocarcinus emarginatus confirmed the presence of P. mexicanus. A recent publication studying Paragonimus species from Mexico using morphological and molecular approaches identified three distinct species following comparisons with the DNA sequences from the species found in Ecuador (López-Caballero et al. 2013). The authors of the publication therefore suggested resurrecting the name P. ecuadoriensis for species in South America. However, further studies using molecular, scanning electron microscopy and morphological techniques are required before accepting this suggestion.
Additionally, a small encysted metacercariae, which was tentatively named Paragonimus napensis, was found in the northern Amazon Region (Amunárriz 1991b). Future molecular taxonomy studies are also needed to clarify the identity of this species.
First intermediate host - A study performed in 1984 on the snail Aroapyrgus colombiensis captured in the Amazon Region revealed it to be the first intermediate host of Paragonimus. A total of 2,350 specimens were microscopically examined using two techniques. The first 1,000 specimens were placed in Petri dishes with water and observed microscopically every 12 h for released rediae and cercariae. The remaining specimens were compressed between two glass plates and then examined microscopically. Rediae and cercariae were observed, indicating an infection rate of 0.042% in the snails (Amunárriz 1991b) and the rediae exhibited characteristic morphological features of P. mexicanus (Malek et al. 1985). In contrast, none of the Hemisinus maculatus molluscs captured in a subtropical region of the province of Bolívar were found to be infected (Yokogawa et al. 1971).
Second intermediate hosts - On the coast, non-encysted metacercariae found in captured H. aequatorialis from Jipijapa in the province of Manabí were initially named P. ecuadoriensis (Arzube & Voelker 1978). In Caluma within the province of Bolívar, non-encysted metacercariae identified as P. mexicanus were isolated from Strengeria eigenmanni (after referred as Pseudotelphusa chilensis) (Yokogawa et al. 1971). Among H. aequatorialis specimens collected from 45 different rivers in four cantons of the province of Esmeraldas, 42.6% of the crabs were found to be infected with P. mexicanus (Vieira et al. 1992). In P. chilensis (at present referred as H. chilensis) captured in Nalpe-Manabí and H. aequatorialis collected in Concordia-Esmeraldas, 16.1% and 68.5% specimens, respectively, were found to be harbour P. mexicanus (Iwagami et al. 2003, Waikagul et al. 2003).
In the Amazon, P. mexicanus metacercariae were found in 96.1% of the M. emarginatus (formerly referred to as Zilchiopsis ecuadoriensis) individuals examined. These crabs were caught in the same streams where infected A. colombiensis molluscs were found. Conversely, none of the Trichodactylus faxoni (formerly referred to as Trichodactylus maytai) crabs captured were parasitised (Amunárriz 1991b).
Metacercariae have been found in various organs of crabs. Among the Hypolobocera species examined, P. mexicanus metacercariae infested the hepatopancreas in 14.4% (Vaca et al. 1989). In H. chilensis from Nalpe-Manabí, metacercariae also showed a preference for the hepatopancreas (Waikagul et al. 2003). However, although H. aequatorialis from Quinindé-Esmeraldas showed a preference for the hepatopancreas, metacercariae were also found in muscle and other organs (Vieira et al. 1992). The metacercariae in crabs from Caluma-Bolívar were found to prefer the hepatopancreas over muscle (Yokogawa et al. 1971). It is unknown whether these locations are associated with the parasite or host intermediate species, as in Asia, where various families of molluscs determine which subspecies of P. westermani is present (Blair et al. 1997). Crabs, both male and female, were found to be infected without any existing predilection, although a higher infection rate was reported in male crabs from Nalpe-Manabí (Waikagul et al. 2003).
In 2013, the authors (HS, MC and DR) studied 35 T. faxoni crabs captured from a small stream in the northern Amazon Region: Nuevo Rocafuerte, in the province of Orellana. Of these 35 crabs, 13 (37.1%) were positive for P. mexicanus. However, in the coastal region, none of the 153 Hypolobocera spp of crabs captured in areas previously considered to be endemic were found to be parasitised by P. mexicanus. Metacercariae that have not yet been identified have also been found (unpublished data). In Ecuador, infected crabs have been found at elevations ranging from 150-2,000 m.
Several patients from both the coast and the Amazon regions reported no clinical history of ingesting raw crab, but did eat crayfish (Fernández 1990). Freshwater crayfish could also be a secondary host in Ecuador, as documented in other countries (Acha & Szyfres 2003). Confirmatory studies on these crustaceans are needed.
Definitive hosts - In captured wild mammals from the coastal region, adult Paragonimus flukes were recovered from the lungs of a Nasua nasua (coati or tejón), whereas no flukes were found in examined Didelphis marsupialis or marmosas (Arzube & Voelker 1978). In the Amazon, pulmonary cysts containing adult Paragonimus were found in captured D. marsupialis, Felis pardalis (ocelot or tigrillo), N. nasua and Tayassu pecari (pécaris) (Vaca et al. 1989, Amunárriz 1991b). Cats experimentally infected with metacercariae from H. aequatorialis captured in the coastal provinces of El Oro and Esmeraldas developed lung nodules containing P. mexicanus flukes (Palacios et al. 1978, Vieira et al. 1992). Infection in cats, dogs and Rattus rattus, but not in hamsters, was achieved with metacercariae obtained from H. chilensis in Nalpe-Manabí (Waikagul et al. 2003).
Human infections - Age and gender - In Ecuador, paragonimiasis is generally diagnosed in adolescents and young adults, but clinical cases have been documented in individuals ranging from four-67 years of age (Calvopiña et al. 1998, 2003). Among patients treated at hospitals in Latacunga and Guayaquil, most affected individuals ranged from 11-30 years of age (Fernández 1990, Calvopiña et al. 1995). In the Chachi indigenous groups from Canandé-Esmeraldas River, most affected individuals were 11-30 years of age (Guevara et al. 1999). During active field searches, infections were found more frequently in children from six-12 years of age (Calvopiña et al. 1998).
Regarding gender, no significant differences have been found. However, variations exist depending on the geographical location of the patients. In the indigenous Quichua and Shuar groups and colonists in the Amazon Region, alongside Napo River, women exhibited infection rates of 51.2% (Amunárriz 1991a) and 52.8% (Calvopiña et al. 2003), respectively. In patients treated in hospitals of Latacunga-Cotopaxi and Quito-Pichincha (non-endemic areas), males exhibited infection rates of 52% and 57.7%, respectively. Of the 45 patients admitted to the hospital in the coastal city of Guayaquil, there were 21 males and 24 females (Fernández 1990). The greatest gender differences were observed in patients treated in a coastal regional hospital in El Carmen-Manabí and those of the indigenous Chachi (province of Esmeraldas), where the infection rates of females reached 66.4% and 90%, respectively (Calvopiña et al. 1994, Guevara et al. 1999). The variation in age and gender ratio of infection depends not only on the specific locality and geographical region, but also on the local customs and habits related to the preparation of crustaceans.
Clinical forms - Based on all publications, reports, clinical records and available studies, 3,822 cases were documented in Ecuador since 1921, of which 99.7% were pulmonary infections. There were 12 cases of cutaneous paragonimiasis in the same family, presenting signs of migratory inflammation, localised pain and the presence of adult worms confirmed using histopathology (Carvajal et al. 1979). A hepatic form of the disease presented with a tumour lesion in the left lobe of the liver (Peñafiel et al. 1981).
The pulmonary symptoms included a productive cough with rust-coloured sputum in 70-100% of the cases, followed by chest pain in 80%. Symptoms were periodic and mild in patients with a short clinical evolution and/or low egg counts in their sputum (Peñafiel et al. 1981, Calvopiña et al. 1994). Most patients showed good general health and nutritional status. However, in the indigenous ethnic group Chachi of Canandé-Esmeraldas River, weight loss and diaphoresis were documented in 60% of cases with a 30% prevalence of fever (Guevara et al. 1999). Complicated cases involving pleural effusion, exudative pleuritis and emphysaema were reported in 19% of patients referred to the Eugenio Espejo Hospital in Quito (Peñafiel et al. 1981). Among 124 patients in the hospital of Nuevo Rocafuerte in the Amazon Region, 12.5% presented with fever and dyspnoea and were in poor health (Amunárriz 1991a). The duration of disease ranged from three months to 12 years one case lasted 14 years before a diagnosis was made (Peñafiel et al. 1981).
Eating habits in relation to paragonimiasis - Indigenous groups in Ecuador and migrants to tropical regions practice hunting and fishing, including freshwater crayfish and crabs, for their basic food supply. Certain beliefs and practices are likely to increase infection rates, such as the idea that eating raw crabs is beneficial for hangovers and freshly prepared juice from raw crabs (made from internal organs) is used as a traditional medicine to treat fevers (Vieira et al. 1992). Certain Amazon communities prepare the “juice” of raw crabs to increase breast milk production in nursing mothers. It is customary for families or groups of neighbours to fish the rivers for crabs and crayfish and then consume them in soup, fried, baked or raw. Children play and fish in the rivers during their free time and eat baked, fried or raw crabs and crayfish (Fernández 1990). In endemic areas, 94% of patients reported eating crabs and 99% reported eating crayfish (Calvopiña et al. 1994). As these eating habits involve the entire family, several members of the same family can become infected, as observed in Chachi (Paredes et al. 1978), in the Amazon (Amunárriz 1991b) and among coastal settlers (Calvopiña et al. 2003).
Diagnosis - In Ecuador, a case of pulmonary paragonimiasis is defined as a person presenting with a productive cough with rusty-brown or blood-stained sputum, accompanied by a history of eating raw or improperly cooked freshwater crabs or crayfish and the presence of the characteristic ova of Paragonimus in sputum and/or stool samples (Díaz et al. 1991). The diagnostic method recommended and utilised is the microscopic observation of eggs in sputum and faeces. No concentration methods are used and no serological and/or molecular methods are available. Eggs of Paragonimus in sputum have been identified following Ziehl Neelsen staining (Calvopiña et al. 2003). The detection of eggs in faecal material can reach up to 13.6%, especially in children because of their habit of swallowing sputum (Fernández 1990). Faecal detection can also occur during intestinal paragonimiasis, a disease that is undiagnosed in Ecuador and rarely occur worldwide (Liu et al. 2012). More than 50% of hospitalised patients showed hypereosinophilia or leucocytosis (11,000-15,000/µL) with normal haemoglobin and haematocrit levels (Peñafiel et al. 1981). Microscopy is specific, but the sensitivity depends on the amount and frequency of expectoration, which is inconsistent in patients with mild and moderate disease. Using soluble antigens obtained from adult Paragonimus, elevated levels of IgG and IgM were found in patients with pulmonary paragonimiasis according to the presence of eggs in sputum, with the subclass IgG4 antibody being the most dominant (Guevara et al. 1995, 1999). In 43 patients, the levels of IgG and IgE determined by ELISA and indirect haemagglutination remained elevated after six months of being treated with praziquantel (PZQ) (Knobloch et al. 1984). These tests were performed only for research purposes.
Crude antigens of P. westermani and Paragonimus heterotremus were used to test for dermal delayed hypersensitivity in residents of endemic areas, with reactive cases found in children and adolescents (Palacios et al. 1978); however, the test was discontinued because of severe sensitivity reactions and low specificity (Díaz et al. 1991). Immunoblot assays were performed using antigens of P. heterotremus, with four of seven children who presented with chronic cough, but were negative for eggs in their sputum exhibiting positive results (Waikagul et al. 2003). Studies using crude antigens from various species carry the risk of false negative results.
Imaging studies - Radiological imaging was performed only on referred hospitalised patients from rural health centres because of the unavailability of diagnostic methods and/or drugs or because of disease severity. In patients referred to the Eugenio Espejo Hospital in Quito, the most common radiological finding was diffuse and nodular infiltrates (54%), with 11% presenting pleurisy, emphysaema and pleural effusion and 11% presenting normal radiographs (Peñafiel et al. 1981). Of the patients treated at hospitals in Nuevo Rocafuerte-Orellana and in Guayaquil, 77% and 85.2% exhibited normal radiological imaging (Fernández 1990, Amunárriz 1991a). In the former hospital, 13% showed exudative pleuritis, hilar lymphadenopathy and caverns (Amunárriz 1991a). In patients identified during active searches, 46% presented with diffuse and nodular infiltrates, whereas 39% did not exhibit any radiographic abnormalities (Calvopiña et al. 1998, 2003). Infiltrative images were found to resolve after treatment, but no radiological changes were noted in patients with nodular lesions, pleural effusion or cavities (Calvopiña et al. 1998). There were no pathognomonic radiological lesions associated with paragonimiasis. No reports are available using computed tomography or magnetic resonance imaging studies.
Differential diagnosis and association with tuberculosis (TB) and/or pulmonary mycosis - Overall, 10.8% of the referred patients treated at the Eugenio Espejo Hospital presented with pulmonary TB (Peñafiel et al. 1981), whereas only one (1.5%) patient with TB was found in the hospital of Nuevo Rocafuerte-Orellana (Amunárriz 1991a). No association was found in patients referred to hospitals in El Carmen-Manabí and Francisco de Orellana (Vaca et al. 1989, Calvopiña et al. 1994). During active search studies in La Maná-Cotopaxi communities, 12.9% of patients had or were in treatment for pulmonary TB.
Patients with pulmonary paragonimiasis generally exhibited good health and nutritional status. Episodes of cough were periodic and rust-coloured sputum with haemoptysis, as in TB, rarely occurred (Calvopiña et al. 2003). None of the cases were associated with pulmonary mycoses despite the fact that paracoccidioidomycosis is endemic in these tropical regions (Severo & Fernández 2004).
Treatment - According to the Manual for Paragonimiasis Control in Ecuador, issued in 1991 by the MSP (Díaz et al. 1991) and by the WHO (2011), PZQ is the drug of choice for treating pulmonary paragonimiasis. The recommended dosage for treating paragonimiasis is three administrations totalling 75 mg/kg/day for two days. The treatment of patients from the coast and Amazon regions with PZQ was effective in 95-100% of cases, with minimal and transient adverse effects (Calvopiña et al. 1998). A 100% cure rate was observed in patients treated with PZQ at 50 mg/kg/day for three days (Fernández 1990). In the 1970s and 1980s, treatment with bithionol was effective in 80% of patients, but treatment abandonment was common due to severe adverse effects, especially gastrointestinal. Bithionol was effective in treating 62 of 64 patients at the Amazon regional hospital of Nuevo Rocafuerte, with tolerable/manageable adverse effects under an inpatient regimen (Amunárriz 1991a). A combination of emetine plus chloroquine was effective in only 18.2% of patients treated (Peñafiel et al. 1981). Currently, there are no official guidelines in Ecuador, which has led to a lack of medical knowledge regarding current management and recommended therapy.
In two clinical-therapeutic trials, 216 patients with pulmonary paragonimiasis from the coast and Amazon were treated using triclabendazole (TCB). An efficacy of 100% was obtained, even in patients resistant to PZQ (Calvopiña et al. 1993). Using three distinct dose regimens: 5 mg/kg once per day for three days, a single dose of 10 mg/kg or two doses of 10 mg/kg during a single day, all 62 patients were cured, both clinically and parasitologically. In a second study, 154 patients were treated with either 10 mg/kg twice a day or a single dose of 10 mg/kg, with efficacies of 90.9% and 84.4%, respectively, and all patients were cured following retreatment (Calvopiña et al. 2003). TCB is now recommended as the alternative drug of choice for treating paragonimiasis and the first drug of choice for fascioliasis (WHO 2011). Currently, PZQ and TCB are not available in Ecuador despite their recommendation in the 2013 national therapeutic manual.
Prevention and control measures - At present, there are no strategies for diagnosis, updated patient management or preventive measures in Ecuador. A national control or elimination programme of the disease is non-existent.
Concluding remarks - Paragonimiasis is endemic to tropical and subtropical regions of Ecuador. Although the disease is known to be present in several locations, epidaemiological studies remain incomplete. Furthermore, the lack of recent active field search and nationwide seroepidemiological studies prevent making an estimate of the current infected population. Knowledge of the intermediate hosts and reservoirs is limited and there is no comparative molecular characterisation of the parasites. To identify possible new species in the Amazon, DNA identification methods are needed. In addition, investigations are recommended to determine the impact that ecological and environmental changes, such as farming, chemical application in oil palm and banana plantations, dam construction and deforestation with the disappearance of animal reservoirs and intermediate hosts, may have on the disease. Preventive programmes to educate and implement changes in the eating habits of residents (only eating well-cooked crabs or crayfish) are of the utmost importance. A continued health education programme for professional personnel, together with ensuring the availability of medication for treatment, is necessary to care for those affected by Paragonimus. The lack of drugs recommended for treatment is of great concern because it causes patients to remain infected, with epidaemiological consequences, complications and suffering. The MSP should acquire the drugs and then make them readily available in the health centres of endemic areas.
ACKNOWLEDGEMENTS
To Ronald Guderian, for reviewing this paper, and Alejandro Arteaga, for providing the map layer.
REFERENCES
- Acha P, Szyfres B. Parasitosis. 3. III. PAHO; Washington DC: 2003. Zoonosis y enfermedades transmisibles comunes al hombre y a los animales; pp. 158–164. [Google Scholar]
- Amunárriz M. Estudios sobre patologías tropicales en la Amazonia Ecuatoriana. 1. CICAME; Quito: 1991a. Paragonimiasis. Clínica y patología; pp. 77–90. [Google Scholar]
- Amunárriz M. Intermediate hosts of Paragonimus in the eastern Amazonic region of Ecuador. Trop Med Parasitol. 1991b;42:160–164. [PubMed] [Google Scholar]
- Arzube ME, Voelker J. Sobre la incidencia de la paragonimiasis en el Ecuador 1972-1976. Rev Ecuat Hig Med Trop. 1978;31:73–76. [Google Scholar]
- Blair D, Agatsuma T, Watanobe T, Okamoto A, Ito A. Geographical genetic structure within the human lung fluke, Paragonimus wes- termani, detected from DNA sequences. Parasitology. 1997;15:411–417. doi: 10.1017/s0031182097001534. [DOI] [PubMed] [Google Scholar]
- Brenes RR, Zeledon R, Rojas G. Biological cycle and taxonomic position of a Costa Rica Paragonimus and the present status of paragonimiasis from the world. Brenesia. 1980;18:353–366. [Google Scholar]
- Calvopiña M, Aguirre FL, Falcones MC, García VW, Guderián RH. Paragonimiasis pulmonar en el Hospital de el Carmen, Manabí, Ecuador. Educ Méd Contin. 1994;44:17–21. [Google Scholar]
- Calvopiña M, Guderian RH, Paredes W, Chico M, Cooper PJ. Treatment of human pulmonary paragonimiasis with triclabendazole: clinical tolerance and drug efficacy. Trans R Soc Trop Med Hyg. 1998;92:566–569. doi: 10.1016/s0035-9203(98)90919-1. [DOI] [PubMed] [Google Scholar]
- Calvopiña M, Guderian RH, Paredes WY, Cooper P. Comparison of two single-day regimens of triclabendazole for the treatment of human pulmonary paragonimiasis. Trans R Soc Trop Med Hyg. 2003;97:451–454. doi: 10.1016/s0035-9203(03)90088-5. [DOI] [PubMed] [Google Scholar]
- Calvopiña M, Paredes W, Guderian RH, Poltera AA. Efficacy of triclabendazole in human pulmonary paragonimiasis refractary to emetine, bithionol and praziquantel. Bol Chil Parasitol. 1993;17:44–46. [Google Scholar]
- Calvopiña M, Paredes W, Guerrero G, Guevara A, Sanchez M, Guderian RH. Paragonimiasis en la provincia de Cotopaxi, Ecuador. Educ Méd Contin. 1995;49:18–20. [Google Scholar]
- Carvajal L, Zerega F, Loaiza M, Borja A, Rumbea J. Paragonimiasis cutánea, clínica e histología, eosinofilia tropical o síndrome de helmintiasis parenteral. Rev Ecuat Hig Med Trop. 1979;32:69–82. [Google Scholar]
- Díaz G, Calvopiña M, Guderian R, Amunárriz M. Boletín Epidemiológico, Programa Enfermedades Tropicales. Ministerio de Salud Pública; Quito: 1991. Control de la paragonimiasis en el Ecuador.18 [Google Scholar]
- Fernández T. Paragonimiasis pulmonar. Aspectos clínicos y tratamiento con praziquantel. Rev Facultad Cien Med Guayaquil. 1990;2:17–24. [Google Scholar]
- Guevara A, Vieira JC, Araujo E, Calvopiña M, Guderian RH, Carlier Y. Antibody isotypes, including IgG subclasses, in Ecuadorian patients with pulmonary paragonimiasis. Mem Inst Oswaldo Cruz. 1995;90:497–502. doi: 10.1590/s0074-02761995000400012. [DOI] [PubMed] [Google Scholar]
- Guevara GA, Vieira JC, Guachamín P, Villegas V, Murnam T, Lovato R, Mancero T. Paragonimiasis pulmonar en el Río Canandé, provincia de Esmeraldas. VozAndes. 1999;12:46–49. [Google Scholar]
- Heinert JF. Paragonimiasis pulmonar. Anales de la Sociedad Medico Quirúrgica del Guayas. 1922;2:43–51. [Google Scholar]
- INEC - Instituto Nacional de Estadísticas y Censos/Ecuador VII Censo de Población y VI de Vivienda. 2010 redatam.inec.gob.ec/cgibin/RpWebEngine.exe/PortalAction?&MODE=MAIN&BASE=CPV2010&MAIN=WebServerMain.inl
- Iwagami M, Monroy C, Rosas MA, Pinto MR, Guevara AG, Vieira JC, Agatsuma Y, Agatsuma T. A molecular phylogeographic study based on DNA sequences from individual metacercariae of Paragonimus mexicanus from Guatemala and Ecuador. J Helminthol. 2003;77:33–38. doi: 10.1079/JOH2002147. [DOI] [PubMed] [Google Scholar]
- Knobloch J, Paz G, Feldmeier H, Wegner D, Voelker J. Serum antibody levels in human paragonimiasis before and after therapy with praziquantel. Trans R Soc Trop Med Hyg. 1984;78:835–836. doi: 10.1016/0035-9203(84)90038-5. [DOI] [PubMed] [Google Scholar]
- Liu CT, Chen YC, Chen TH, Barghouth U, Fan CK. Intestinal paragonimiasis with colonic ulcer and hematochezia in an elderly Taiwanese woman. Korean J Parasitol. 2012;50:349–352. doi: 10.3347/kjp.2012.50.4.349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- López-Caballero J, Oceguera-Figueroa A, León-Règagnon V. Detection of multiple species of human Paragonimus from Mexico using morphological data and molecular barcodes. Mol Ecol Resour. 2013;13:1125–1136. doi: 10.1111/1755-0998.12093. [DOI] [PubMed] [Google Scholar]
- Malek E, Ibañez M, Guerra A. Description of redia and cercaria of Paragonimus peruvianus from experimental infected Aroapyrgus colombiensis of Condebamba Velley, Peru. J Parasitol. 1985;71:253–256. [PubMed] [Google Scholar]
- Miyazaki I, Ibañez N, Miranda H. On a new fluke found in Peru, Paragonimus peruvianus sp. n. (Trematoda-Troglotrematidae). Jpn J Parasitol. 1969;18:23–30. [Google Scholar]
- Miyazaki I, Ishii Y. Studies on the Mexican lung flukes with special reference to a description of Paragonimus mexicanus sp. nov. (Trematoda: Troglotrematidae). Jpn J Parasitol. 1968;17:445–453. [Google Scholar]
- Miyazaki I, Kifune T, Lamothe-Argumedo R. Taxonomical and biological studies on the lung flukes of Central America. 2. Department of Parasitology/School of Medicine/Fukuoka University,; Fukuoka: 1980. 28 Occasional Publication nº. [Google Scholar]
- Montalván JA. Paragonimus en el Ecuador. Estudio clínico-epidemiológico. Rev Facultad Cien Med Guayaquil. 1968;3:1–48. [Google Scholar]
- Palacios M, Serrano L, Barragán B, Bravo D. Epidemiología de la paragonimiasis en la Cuenca del Río Jubones. Rev Facultad Cien Med Guayaquil. 1978;2:57–68. [Google Scholar]
- Paredes L, Paulson G, Lazo R, Célleri W, Rumbea J, Borrero E, Plaza L, Fernandez T. Investigación médico-ecológico y socio-económico en una tribu Cayapa de la provincia de Esmeraldas. Rev Ecuat Hig Med Trop. 1978;31:63–71. [Google Scholar]
- Peñafiel W, Dávalos R, Coloma M. Paragonimiasis pulmonar. Revisión clínica de 92 casos. Rev Fac Ciencias Médicas. 1981;6:253–261. [Google Scholar]
- Rodríguez J. Contribución al estudio del ciclo evolutivo del Pa- ragonimus westermani. Rev Ecuat Med Cienc Biol. 1963;1:20–34. [Google Scholar]
- Savioli L, Daumerie D. Working to overcome the global impact of neglected tropical diseases: first WHO report on neglected tropical diseases. WHO; Geneve: 2010. 172 [Google Scholar]
- Severo LC, Fernández T. Paracoccidiodomicosis. In: Fernández T, editor. Tratado de medicina tropical. 3. Imprenta Mariscal; Guayaquil: 2004. pp. 299–312. [Google Scholar]
- Toscano C, Hai YS, Nunn P, Mott KE. Paragonimiasis and tuberculosis - Diagnostic confusion: a review of the literature. 1994 apps.who.int/iris/handle/10665/59147.
- Vaca O, Guderian RH, Blankespoor H. Estudio de un foco de paragonimiasis pulmonar en el Oriente Ecuatoriano. Actualidad. 1989;14:19–23. [Google Scholar]
- Vieira JC, Blankespoor HD, Cooper PJ, Guderian RH. Paragonimiasis in Ecuador: prevalence and geographical distribution of parasitisation of second intermediate hosts with Paragonimus me- xicanus in Esmeraldas province. Trop Med Parasitol. 1992;43:249–252. [PubMed] [Google Scholar]
- Voelker J, Arzube ME. Ein neuer Lungenegel aus der Kustenkordillere von Ecuador: Paragonimus ecuadoriensis n. sp. (Paragonimidae: Trematoda). Tropenmed Parasitol. 1979;30:249–263. (Ger). [PubMed] [Google Scholar]
- Waikagul J, Lazo R, Cornejo E. Paragonimus infection in Pedernales, Ecuador. Bulletin of the Central Research Institute Fukuoka University Series E Interdisciplinary Sciences. 2003;1:259–273. [Google Scholar]
- WHO - World Health Organization Report of the WHO expert consultation on foodborne trematode infections and taeniasis/cysticercosis. WHO; Geneve: 2011. 59 [Google Scholar]
- Yokogawa M, Inatomi S, Tsuji M, Kojima M, Hata H, Miranda H, Ibáñez N, Rumbea J. Pathobiological studies on paragonimiasis in Peru and Ecuador. Report of the results of the research supported by Grant-in-Aid for Scientific Research (Grant-in-Aid for Overseas Scientific Survey) in 1982. Chiba University; Chiba: 1983. 20 [Google Scholar]
- Yokogawa M, Montalván J, Rumbea J, Drouet W. Unas metacercarias de Paragonimus recientemente encontradas en la República del Ecuador. Rev Ecuat Hig Med Trop. 1971;28:75–82. [Google Scholar]