Abstract
In this study, we investigated the effects of 8-weeks of swimming exercise on neurogenesis in the subventricular zone (SVZ) and on the levels of nerve growth factor (NGF) and synapsin I protein in the olfactory bulb (OB) of adult rats at a series of relevant time points (2 days, 1 week, 2 weeks, 4 weeks, 3 months, and 6 months). Ninety-six male Sprague Dawley rats were divided into 2 groups: (1) a control group (COG; n = 48, n = 8 for each time point) and (2) a swimming exercise group (SEG; total n = 48; n = 8 for each time point). SEG performed swimming exercise for 5 days per week over a period of 8 weeks. We found that the number of 5-bromo-2’-deoxyuridine-5’-monophosphate (BrdU)- and doublecortin (DCX)-positive cells was significantly higher in SEG than in COG at all time points (Day 2, Week 1, Week 2, Week 4, Month 3, and Month 6; p < 0.001). Furthermore, NGF and synapsin I protein levels were significantly higher in SEG on Day 2, and Weeks 1, 2, and 4 than in COG (p < 0.05 for each time point). Our findings suggest that regular swimming exercise in adult rats increases neurogenesis, neuronal survival, and neuronal maintenance in the SVZ; furthermore, swimming exercise increases the levels of NGF and synapsin I in the OB.
Keywords: physical training, progenitor cells, olfactory bulb, neurotrophic factors, brain, rats
INTRODUCTION
In adults, neurogenesis mostly occurs in the subgranular zone (SGZ) of the dentate gyrus and in the olfactory bulb (OB) [1, 2, 3]. Neuro-genesis in the OB occurs as the result of neuronal precursor cell migration from the subventricular zone (SVZ), which lines the lateral ventricles, and their migration along the rostral migratory stream (RMS) pathway. These immature nerve cells differentiate in the OB into granule cells and periglomerular cells, which are involved in olfactory learning and odour discrimination in the OB [4]. Thus, in adults, newly generated neurons in this area help stabilize the nerve cell population by replacing neurons generated during the previous developing phase [5]. Interestingly, it has been observed in animal models that approximately half of the newly generated neurons in the adult OB die several weeks after differentiation [6], which is further accelerated with increasing age. It has also been demonstrated that loss of olfaction in younger rats due to naris occlusion increases cell death [7, 8] and decreases the rate of neuronal survival. This is observed following long-term olfactory loss [9] as well as when the transcription process is genetically confused [10]. This reduced rate of nerve cell death and neurogenesis in the OB and SVZ leads to decreased brain plasticity and thus a long-term decline in learning abilities. Therefore, these findings highlight the potential role of neurogenesis and neuronal cell death in the pathogenesis of neurodegenerative diseases such as Alzheimer's and Parkinson's disease.
Nerve growth factor (NGF) is a neurotrophic factor that exists in the central and peripheral nervous systems. It binds with high affinity to the tyrosine kinase receptor TrkA and with low affinity to p75. NGF undergoes retrograde axonal transport and exerts specific functions in the perikarya of responsive neurons [11]. NGF stimulates the growth and differentiation of progenitor cells, helping them develop into mature neurons [12]. In addition, NGF and brain-derived neurotrophic factor (BDNF) stimulate free radical scavengers, there-by protecting neurons from oxidative stress [13, 14, 15] and leading to the regeneration of damaged nerve tissues. Therefore, an increased level of NGF in the OB is essential for the continuous generation of neurons in the SVZ of the adult brains and for increase in neuronal plasticity.
It is well known that regular exercise can enhance brain functions, including cognitive abilities [16, 17], and upregulate neurotrophins [18]. Our previous research has indicated that regular exercise promotes neurogenesis in the dentate gyrus. This effect has also been observed following swimming exercise [19] and has been demonstrated to have a positive impact on brain function. However, it is unclear whether a correlation exists between nerve cell generation, survival, and maintenance in the SVZ and changes in NGF and synapsin I protein levels in the OB following a time gap after regular exercise.
In this study, we investigated the effect of regular swimming on nerve cell generation, stabilization, survival, and maintenance as well as changes in NGF and synapsin I protein levels in the OB of adult rats. Neurogenesis in the SVZ and NGF and synapsin I levels in the OB were examined following an 8-week swimming exercise program. The animals were then injected with 5-bromo-2’-deoxyuridine-5’-monophosphate (BrdU), using BrdU and doublecortin (DCX) antibodies at different relevant time points (2 days, 1 week, 2 weeks, 4 weeks, 3 months, and 6 months after the exercise program).
MATERIALS AND METHODS
Animals
Male Sprague-Dawley rats (n = 96; age, 24 weeks; weight, 498.6 ± 42.6 g) were adapted to the laboratory environment (temperature, 22°C ± 1°C; relative humidity, 55% ± 3%; 12 h light: 12 h dark photoperiod) for 2 weeks. All rats were housed in pairs, given free access to water, and fed a standard chow diet (protein, 21%; fat, 5%; nitrogen-free extract, 55%; fibre, 4%; adequate mineral and vitamin content; Purina Mills Inc., Korea). Studies were approved by the Ethical Committee of Korea National Sport University, and performed in accordance with the principles of the Declaration of Helsinki (October 2008, Seoul).
Experimental procedure
Rats were allocated to the following groups: (1) control group (COG: total n = 48; n = 8 for each time point); (2) swimming exercise group (SEG: total n = 48; n = 8 for each time point). Following that, the SEG was made to perform swimming exercise. Freestyle swimming was performed, without any weight load, inside a stainless steel container with a water temperature of 30°C ± 2°C. A pair of rats was placed inside the container, and they performed the exercise for 25 min, followed by a 5-min rest. This procedure was repeated twice. This swimming exercise was performed for 50 min per day, and 5 days per week, over a period of 8 weeks. After the swimming exercise was completed, body temperatures of the rats were maintained at 37°C using a hair dryer. At the same time, 50 mg · kg-1 b.w of BrdU (Sigma, St. Louis, MO, USA), a thymidine analogue, was injected into 3 rats of each group at each time point, for 5 days of the last week of exercise, to observe neurogenesis in the SVZ.
Tissue collection
Upon completion of the 8-week exercise program, rats were anaesthetized by an intraperitoneal (i.p.) injection of xylazine (8 mg · kg-1) and ketamine (40 mg · kg-1) at specific time points (2 days, 1 week, 2 weeks, 4 weeks, 3 months, 6 months). For the detection of neurogenesis in the SVZ, 3 rats from each time point were transcardially perfused with 50 mM phosphate-buffered saline (PBS) and fixed with a freshly prepared solution of 4% paraformaldehyde in 100 mM phosphate buffer (PB; pH 7.4). The brains were dissected and postfixed overnight in the same fixative and then stored in 30% sucrose solution. Coronal freezing microtome (Leica, Nussloch, Germany) sections (40 μm) were stored in cryoprotectant (25% ethylene glycol, 25% glycerin, 0.05M phosphate buffer) at -20°C until processing for immunohistochemistry. For analysis of proteins, brains of 5 rats from each time point were quickly excised, then the OBs were dissected, and stored at -70°C.
NGF and synapsin I protein levels
To prepare samples for western blotting, the OB was crushed in a solution containing 150 mM NaCl, 5 mM ethylenediamine tetraacetic acid (EDTA), 50 mM Tris-HCl (pH 8.0), 1% NP 40, 1 mM aprotinin, 0.1 mM leupeptin, and 1 mM pepstatin and then centrifuged at 12,000 g for 15 min at 4°C. The extracted proteins (30 μg) were separated with 10% sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) and transferred to a nitrocellulose membrane (Nitrobind; 0.45 μm; Geneworks, SA, Australia). The membrane was blocked by incubation in a Tris-buffered saline solution containing Tween (TBST) and 5% nonfat milk at 4°C. After washing, the membrane was incubated with anti-NGF and synapsin I antibodies (dilution, 1:1,000) (Cell Signaling, Beverly, MA, USA), washed with TBST (3 × 10 min), and incubated with a goat antirabbit IgG secondary antibody conjugated with alkaline phosphatase (AP) (dilution, 1:2,000) (Santa Cruz Biotech, Santa Cruz, CA, USA) for 1 h. The membrane was washed with TBST (3 × 10 min). The protein bands were imaged using a Kodak Image Station (440CF; PerkinElmer Life Sciences, Boston, MA, USA), and the proteins were quantified using densitometry software (Kodak ID 3.5).
BrdU immunohistochemistry
To detect neurogenesis in the SVZ, BrdU immunohistochemistry was performed as previously described [20]. On average, 6 sections encompassing the SVZ (approximately from Bregma 1.0 mm to 1.7 mm) were selected from each brain. Brain sections were permeabilized by incubating in 0.5% Triton X-100 in PBS for 20 min, pretreated in 50% formamide and 2× standard saline citrate (SSC) at 65°C for 2 h, denatured in 2 N HCl at 37°C for 30 min, and rinsed twice in 100 mM sodium borate (pH 8.5). The sections were incubated overnight at 4°C with anti-BrdU mouse monoclonal antibody (1:600; Roche, Mannheim, Germany). The sections were then washed 3 times with PBS and incubated for 1 h with a biotinylated antimouse secondary antibody (1:200; Vector Laboratories). The sections were then incubated for another hour with an avidin– peroxidase complex (1:100; Vector Laboratories). For visualization, the sections were incubated for 5 min in 50 mM Tris–HCl (pH 7.6) containing 0.02% diaminobenzidine (DAB), 40 mg · ml−1 nickel chloride, and 0.03% hydrogen peroxide.
To detect DCX-positive cells in the SVZ, 6 sections on average were selected from the same region as mentioned in the previous section. The brain sections were incubated overnight at 4°C with a mouse monoclonal anti-DCX antibody (1:2000; Santa Cruz Biotech, Santa Cruz, CA, USA). The sections were then washed 3 times with PBS and incubated for 1 h with a biotinylated anti-mouse secondary antibody. For staining, the sections were incubated in a reaction mixture consisting of 0.03% DAB and 0.03% hydrogen peroxide for 5 min. The slides were air dried overnight at room temperature, and the coverslips were mounted using Permount® (Fisher Scientific, New Jersey, NJ, USA).
Six coronal sections from each animal were used to count the number of BrdU- and DCX-positive cells in the SVZ. The numbers of BrdU- and DCX-positive cells in the dorsolateral corner of the lateral ventricle were counted hemilaterally in every sixth section throughout the SVZ at 100× magnification. The image of the SVZ was traced using the Image Pro®Plus image analyzer (Media Cybernetics Inc., Silver Springs, MD, USA) at 40× magnification. Data were expressed as the average total number of BrdU and DCX-positive cells per section.
Statistical analysis
All data were analyzed using the SAS software package (SAS Institute, Cary, NC, USA) and tested against normal distribution. We performed independent t-tests to compare the data between the groups. All values are expressed as means ± standard deviation (SD); a p value of <0.05 was considered as statistically significant.
RESULTS
Swimming exercise increases the number of BrdU- and DCX-positive cells
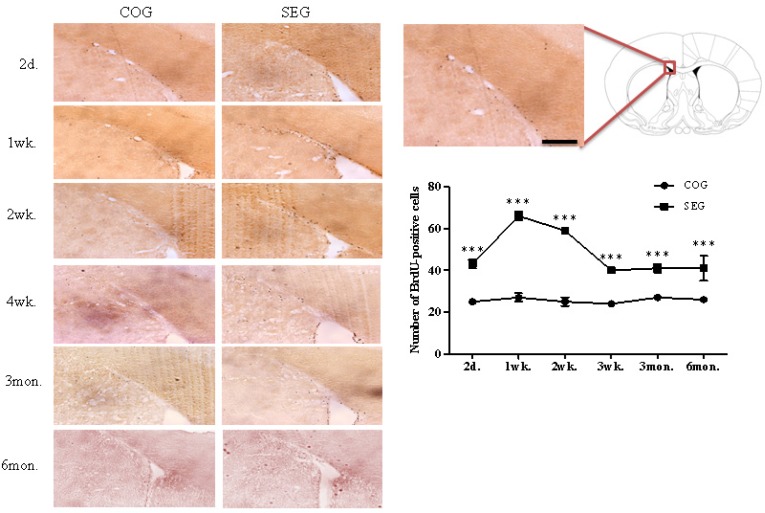
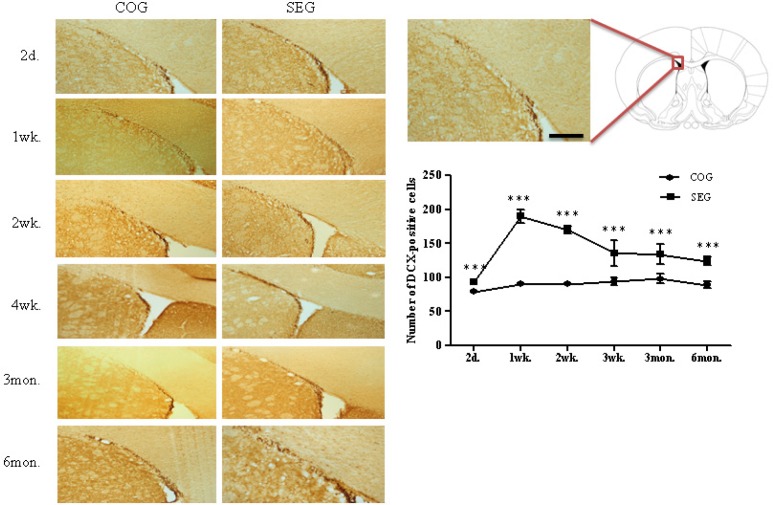
To study the survival time-course of newly generated cells in the adult SVZ, the number of BrdU-labelled cells was assessed at several time points after the swimming exercise. The number of BrdU-positive cells was significantly higher in the swimming exercise group (SEG) than in the control group (COG) at all time points (2 days, 1 week, 2 weeks, 4 weeks, 3 months and 6 months: p < 0.001; Fig. 1). Young immature neurons express a marker, such as DCX. A significant difference was observed between COG and SEG at all time points (2 days, 1 week, 2 weeks, 4 weeks, 3 months and 6 months: p < 0.001; Fig. 2). Specifically, the number of DCX-positive cells was significantly higher in SEG compared to COG.
FIG. 1.
CHANGE IN BRDU-POSITIVE CELL NUMBERS IN THE SUBVENTRICULAR ZONE AFTER SWIMMING EXERCISE.
Note: The number of BrdU-positive cells was significantly higher in SEG than in COG at all time points (2 days, 1 week, 2 weeks, 4 weeks, 3 months and 6 months). COG: control group; n=48, SEG: swimming group; n=48 (n=8 for each time point: 3 rats for BrdU immunohistochemistry, 5 rats for western blotting). ***Significantly different between groups at p < 0.001. Scale bar=25μm.
FIG. 2.
CHANGE IN DCX-POSITIVE CELL NUMBERS IN SUBVENTRICULAR ZONE AFTER SWIMMING EXERCISE.
Note: The number of DCX-positive cells was significantly higher in SEG than in COG at all time points (2 days, 1 week, 2 weeks, 4 weeks, 3 months and 6 months). COG: control group; n=48, SEG: swimming group; n=48 (n=8 for each time point: 3 rats for BrdU immunohistochemistry, 5 rats for western blotting). ***Significantly different between groups at p < 0.001. Scale bar=25μm.
Swimming exercise increases NGF and synapsin I levels
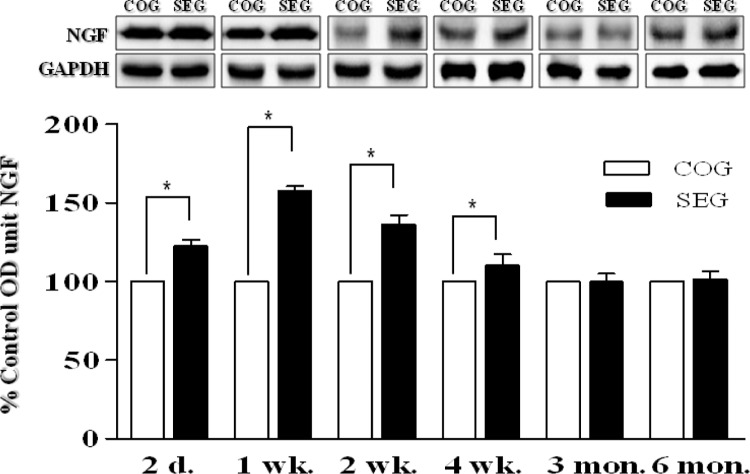
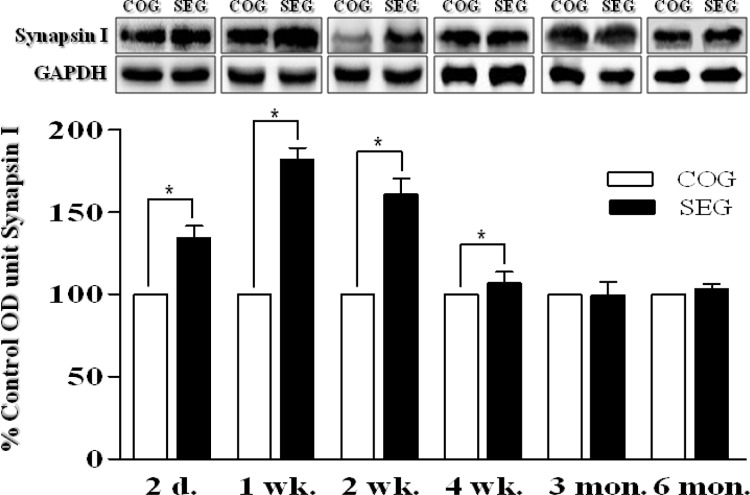
The expression levels of NGF in the adult OB were determined at several time points following the 8-week swimming exercise program. NGF levels in the OB were found to be significantly higher in SEG than in COG at 2 days, 1 week, 2 weeks, and 4 weeks after the exercise (p < 0.05 for each time point; Fig. 3). Similarly, synapsin I levels in the OB were also found to be significantly higher in SEG than in COG 2 at days, 1 week, 2 weeks, and 4 weeks after the exercise program (p < 0.05 for each time point; Fig. 4).
FIG. 3.
CHANGE IN NGF PROTEIN LEVELS IN OLFACTORY BULB AFTER SWIMMING EXERCISE.
Note: NGF levels in the olfactory bulb were found to be significantly higher in SEG than in COG 2 days, 1 week, 2 weeks and 4 weeks after the exercise. COG: control group; n=48, SEG: swimming group; n=48 (n=8 for each time point: 3 rats for BrdU immunohistochemistry, 5 rats for western blotting). *Significantly different between groups at p < 0.05.
FIG. 4.
CHANGE IN SYNAPSIN I PROTEIN LEVELS IN OLFACTORY BULB AFTER SWIMMING EXERCISE.
Note: Synapsin I levels in the olfactory bulb were found to be significantly higher in SEG than in COG 2 days, 1 week, 2 weeks and 4 weeks after the exercise. COG: control group; n=48, SEG: swimming group; n=48 (n=8 for each time point: 3 rats for BrdU immunohistochemistry, 5 rats for western blotting). *Significantly different between groups at p < 0.05.
DISCUSSION
This study was performed on adult rats following an 8-week swimming exercise program to examine whether there is any change in the number of BrdU and DCX-positive cells in the SVZ at various post-exercise time points (2 days, 1 week, 2 weeks, 4 weeks, 3 months, and 6 months). Furthermore, we examined the changes in NGF and synapsin I levels in the OB. The findings of this study suggest that regular swimming exercise increases neurogenesis, induces an increase in the number of immature neurons in the SVZ, and upregulates the levels of NGF and synapsin I protein in the OB.
In addition to recruiting new neurons in the OB through the RMS pathway [21], neural precursor cells also migrate to areas of brain damage under pathophysiological conditions, thereby replacing damaged cells with newly differentiated neurons. For example, when brain damage occurs, cell proliferation in the SVZ increases significantly; these cells then move to the damaged areas of the striatum and differentiate into nerve cells [22]. It has previously been demonstrated that the generation of neural precursor cells in the SVZ increases in an animal model of Huntington's disease (HD) caused by striatal neurodegeneration [23]. Furthermore, endogenous factors and receptors that regulate the cell cycle and differentiation of neural precursor cells have been discovered in the SVZ of patients with HD [22]. This suggests that increased neurogenesis in the SVZ may stabilize nerve cells generated in the previous developing phase and play an important role in the replacement or regeneration of nerve cells in damaged brain areas. The present study was undertaken to examine neurogenesis in the SVZ of adult rats over a period of time after exercise; neurogenesis was assessed by counting the number of BrdU- and DCX-positive cells, which led to our finding that swimming exercise significantly increases the number of BrdU-positive cells (nerve cell proliferation) for at least 6 months. In addition, we found that the number of DCX-positive cells increases following exercise and is maintained for a similar period as mentioned above. Increased nerve cell proliferation and differentiation were highest at the 1-week time point, after which they gradually reduced; by Week 4, the levels had stabilized. This suggests that regular swimming is a potential strategy for stimulating neurogenesis in the SVZ, which in turn can have a positive impact on areas of brain damage and play a role in the discrimination function of the OB. These results are supported by the findings of van Praag et al. [24], who showed that exercise increased the long-term survival of nerve cells, and the study of Yasuhara et al. [25], which demonstrated that a lack of exercise reduced neurogenesis in the SVZ, indicating that exercise may be involved in sustaining neurogenesis in the SVZ. Especially, enriched accommodation and voluntary running exercise increased the survival and migration of stem cells harvested from the SVZ [26]. This also confirms the study of Jizi et al. [27], who reported that voluntary running for a long time induces an increase in nerve cell precursors in the SVZ, which in turn increases migration of nerve cells to a lesion area. Furthermore, Enwere et al. [28] reported that decline of SVZ neurogenesis appears to be associated with deficits in minute odour discrimination in the OB. This observation is similar to that of our previous study [19], in which regular exercise, especially swimming exercise, promoted neurogenesis through proliferation, development, and maintenance of nerve cells in the dentate gyrus. On the other hand, the study of Brown et al. [29] revealed that voluntary exercise for 12 days did not give a positive effect on neural progenitor cell proliferation of the SVZ, showing a result different from that of this study. These results suggested that the experiment model used for the study is a brain-injured animal model and the experimental design (exercise types) is different from that of this study.
In the neurogenic areas of the brain, the SVZ and hippocampus, creation and destruction of nerve cells is continuous. However, to maintain steady differentiation and growth of nerve cells, regulation by neurotrophic factors is necessary [30]. Of these, NGF levels increase with exercise. Indeed, in this study, we observed that NGF level in the OB significantly increased following an 8-week exercise program. As with our previous findings that exercise increases NGF levels in the hippocampus [31], the findings of this study demonstrated that swimming effectively increases NGF levels in the OB. Moreover, increased NGF levels were maintained for 4 weeks following the swimming exercise. NGF is expected not only to induce differentiation, growth, and development of nerve cells in the OB, but also to contribute to the enhancement of synaptic plasticity; this is supported by our finding that increased synapsin I levels were also maintained for 4 weeks following the swimming exercise. Synapsin I is present in axon terminals and plays a role in regulating the secretion of neurotransmitters; thus, it influences synaptic plasticity by regulating pre- and postsynaptic vesicular release. We speculate that in this study, the increased synapsin I levels occurred as the result of migration of SVZ-derived new nerve cells to the OB following swimming. Additionally, we speculate that synaptic intensity and neural networks may have been reinforced by the increased NGF level in the OB. Li et al. [32] and Takei et al. [33] reported that synaptic density was significantly decreased in SYNI mice lacking synapsin I. This is further supported by the finding that regular exercise increased synapsin I level and induced enhancement of BDNF function, which is physiologically similar to NGF [34]. However, increased NGF and synapsin I levels, increased through the swimming exercise, are thought to contribute to the induction of synaptic plasticity and nerve cell growth in the OB, even though it has increased by swimming exercise itself or been influenced by NGF increased through swimming exercise.
CONCLUSIONS
We found that regular swimming exercise for a period of 8 weeks significantly increases the number of newly generated nerve cells in the SVZ of adult rats and extends their survival and maintenance period. Furthermore, we found that swimming exercise increases and maintains NGF and synapsin I protein levels in the OB.
Conflict of interest
The authors declare that there is no conflict of interest.
REFERENCES
- 1.Alvarez–Buylla A, Garcia–Verdugo J. Neurogenesis in adult subventricular zone. J Neurosci. 2002;22:629–634. doi: 10.1523/JNEUROSCI.22-03-00629.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Carleton A, Petreanu LT, Lansford R, Alvarez–Buylla A, Lledo PM. Becoming a new neuron in the adult olfactory bulb. Nat Neurosci. 2003;6:507–518. doi: 10.1038/nn1048. [DOI] [PubMed] [Google Scholar]
- 3.Song H, Stevens C, Gage F. Neural stem cells from adult hippocampus develop essential properties of functional CNS neurons. Nat Neurosci. 2002;5:438–445. doi: 10.1038/nn844. [DOI] [PubMed] [Google Scholar]
- 4.Gheusi G, Cremer H, McLean H, Chazal G, Vincent JD, Lledo PM. Importance of newly generated neurons in the adult olfactory bulb for odor discrimination. Proc Natl Acad Sci USA. 2000;97:1823–1828. doi: 10.1073/pnas.97.4.1823. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Dayer AG, Ford AA, Cleaver KM, Yassaee M, Cameron HA. Short-term and long-term survival of new neurons in the rat dentate gyrus. J Comp Neurol. 2003;460:563–572. doi: 10.1002/cne.10675. [DOI] [PubMed] [Google Scholar]
- 6.Winner B, Cooper–Kuhn CM, Aigner R, Winkler J, Kuhn HG. Long-term survival and cell death of newly generated neurons in the adult rat olfactory bulb. Eur J Neurosci. 2002;16:1681–1689. doi: 10.1046/j.1460-9568.2002.02238.x. [DOI] [PubMed] [Google Scholar]
- 7.Brunjes PC. Unilateral naris closure and olfactory system development. Brain Res Brain Rev. 1994;19:146–160. doi: 10.1016/0165-0173(94)90007-8. [DOI] [PubMed] [Google Scholar]
- 8.Cummings DM, Brunjes PC. The effects of variable periods of functional deprivation on olfactory bulb development in rats. Exp Neurol. 1997;148:360–366. doi: 10.1006/exnr.1997.6660. [DOI] [PubMed] [Google Scholar]
- 9.Corotto FS, Henegar JR, Maruniak JA. Odor deprivation leads to reduced neurogenesis and reduced neuronal survival in the olfactory bulb of the adult mouse. Neurosci. 1994;61:739–744. doi: 10.1016/0306-4522(94)90397-2. [DOI] [PubMed] [Google Scholar]
- 10.Petreanu L, Alvarez-Buylla A. Maturation and death of adult-born olfactory bulb granule neurons:role of olfaction. J Neurosci. 2002;22:6106–6113. doi: 10.1523/JNEUROSCI.22-14-06106.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Yamashita T, Tucker KL, Barde YA. Neurotrophin binding to the p 75 receptor modulates Rho activity and axonal outgrowth. Neuron. 1999;24:585–593. doi: 10.1016/s0896-6273(00)81114-9. [DOI] [PubMed] [Google Scholar]
- 12.Mobley WC, Rutkowski JL, Tennekoon GI, Buchanan K, Johnston MV. Choline acetyltransferase activity in stratum of neonatal rats increased by nerve growth factor. Science. 1985;229:284–287. doi: 10.1126/science.2861660. [DOI] [PubMed] [Google Scholar]
- 13.Cheng B, Mattson MP. NGF and bFGF protect rat hippocampal and human cortical neurons against hypoglycemic damage by stabilizing calcium homeostasis. Neuron. 1991;7:1031–1041. doi: 10.1016/0896-6273(91)90347-3. [DOI] [PubMed] [Google Scholar]
- 14.Cheng B, Mattson MP. Glucose deprivation elicits neurofibrillary tangle-like antigenic change in hippocampal neurons:prevention by NGF and bFGF. Exp Neurol. 1992;117:114–123. doi: 10.1016/0014-4886(92)90120-f. [DOI] [PubMed] [Google Scholar]
- 15.Nistico G, Ciriolo MR, Fiskin K, Iannone M, De MA, Rotilio G. NGF restores decrease in catalase activity and increases superoxide dismutase and glutathione peroxidase activity in the brain of aged rats. Free Radic Biol Med. 1992;12:1771–1781. doi: 10.1016/0891-5849(92)90024-b. [DOI] [PubMed] [Google Scholar]
- 16.Radak Z, Kanebo T, Tahara S, Nakamoto H, Pucsok J, Sasvari M, Nyakas C, Goto S. Regular exercise improves cognitive function and decreases oxidative damage in rat brain. Neurochem Int. 2001;38:17–23. doi: 10.1016/s0197-0186(00)00063-2. [DOI] [PubMed] [Google Scholar]
- 17.Uysal N, Tugyan K, Kayatekin BM, Acikgoz O, Bagriyanik HA, Gonenc S, Ozdemir D, Aksu I, Topcu A, Semin I. The effects of regular aerobic exercise in adolescent period on hippocampal neuron density, apoptosis and spatial memory. Neurosci Letter. 2005;383:241–245. doi: 10.1016/j.neulet.2005.04.054. [DOI] [PubMed] [Google Scholar]
- 18.Radak Z, Toldy A, Szabo Z, Siamilis S, Nyakas C, Silye G, Jakus J, Goto S. The effects of training and detraining on memory, neurotrophins and oxidative stress markers in rat brain. Neurochem Int. 2006;49:387–392. doi: 10.1016/j.neuint.2006.02.004. [DOI] [PubMed] [Google Scholar]
- 19.Lee HC, Hong YP, Kim HT. Effects of exercise on nerve growth factor levels and neurogenesis, and cognitive function in the hippocampal dentate gyrus of adult rats. Kor J Sport. 2013;11:287–299. [Google Scholar]
- 20.Kuhn HG, Dickinson-Anson H, Gage FH. Neurogenesis in the dentate gyrus of adult rat:age-related decrease of neuronal progenitor proliferation. J Neurosci. 1996;16:2027–2033. doi: 10.1523/JNEUROSCI.16-06-02027.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Ventura RE, Goldman JE. Dorsal radial glia generate olfactory bulb interneurons in the postnatal murine brain. J Neurosci. 2007;27:4297–4302. doi: 10.1523/JNEUROSCI.0399-07.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Arvidsson A, Collin T, Kirik D, Kokaia Z, Lindvall O. Neuronal replacement from endogenous precursors in the adult brain after stroke. Nature Med. 2002;8:963–970. doi: 10.1038/nm747. [DOI] [PubMed] [Google Scholar]
- 23.Curtis MA, Penney EB, Pearson AG, van Roon-Mon WMC, Butterworth NJ, Dragunow M, Connor B, Faull RLM. Increased cell proliferation and neurogenesis in the adult human Huntington's disease brain. Proc Natl Acad Sci USA. 2003;100:9023–9027. doi: 10.1073/pnas.1532244100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.van Praag H, Kempermann G, Gage FH. Running increase cell proliferation and neurogenesis in the adult mouse dentate gyrus. Nat Neurosci. 1999;2:203–205. doi: 10.1038/6368. [DOI] [PubMed] [Google Scholar]
- 25.Yasuhara T, Hara K, Maki M, Matsukawa N, Fujino H, Date I, Borlongan CV. Lack of exercise, via hindlimb suspension, impedes endogenous neurogenesis. Neurosci. 2007;149:182–191. doi: 10.1016/j.neuroscience.2007.07.045. [DOI] [PubMed] [Google Scholar]
- 26.Hicks AU, Hewlett K, Windle V, Chernenko G, Ploughman M, Jolkkonen J, Weiss S, Corbett D. Enriched environment enhances transplanted subventricular zone stem cell migration and functional recovery after stroke. Neurosci. 2007;146:31–40. doi: 10.1016/j.neuroscience.2007.01.020. [DOI] [PubMed] [Google Scholar]
- 27.Jizi J, Kang HM, Park C. Voluntary exercise enhances survival and migration of neural progenitor cells after intracerebral haemorrhage in mice. Brain Injury. 2010;8:533–540. doi: 10.3109/02699051003610458. [DOI] [PubMed] [Google Scholar]
- 28.Enwere E, Shingo T, Gregg C, Fujikawa H, Ohta S, Weiss S. Aging results in reduced epidermal growth factor receptor signaling, diminished olfactory neurogenesis, and deficits in fine olfactory discrimination. J Neurosci. 2004;24:8354–8365. doi: 10.1523/JNEUROSCI.2751-04.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Brown J, Cooper-Kuhn CM, Kempermann G, van Praag H, Winkler J, Gage FH, Kuhn HG. Enriched environment and physical activity stimulate hippocampal but not olfactory bulb neurogenesis. Eur J Neurosci. 2003;17:2042–2046. doi: 10.1046/j.1460-9568.2003.02647.x. [DOI] [PubMed] [Google Scholar]
- 30.Jin K, Sun Y, Xie L, Batteur S, Mao XO, Smelick C, Logvinova A, Greenberg DA. Neurogenesis and aging: FGF-2 and HB-EGF restore neurogenesis in hippocampus and subventricular zone of aged mice. Aging Cell. 2003;2:175–183. doi: 10.1046/j.1474-9728.2003.00046.x. [DOI] [PubMed] [Google Scholar]
- 31.Chae CH, Kim HT. Forced, moderate-intensity treadmill exercise suppresses apoptosis by increasing the level of NGF and stimulating phosphatidylinositol 3-kinase signaling in the hippocampus of aging rats. Neurochem Int. 2009;55:208–213. doi: 10.1016/j.neuint.2009.02.024. [DOI] [PubMed] [Google Scholar]
- 32.Li L, Chin LS, Shupliakov O, Brodin L, Sihra TS, Hvalby O, Jensen V, Zheng D, McNamara JO, Greengard P, Anderson P. Impairment of synaptic vesicle clustering and of synaptic transmission, and increased seizure propensity in synapsin I-deficient mice. Proc Natl Acad Sci USA. 1995;92:9235–9239. doi: 10.1073/pnas.92.20.9235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Takei Y, Harada A, Takeda S, Kobayashi K, Terada S, Noda T, Taka Hirokawa N. Synapsin I deficiency results in the structural change in the presynaptic terminals in the murine nervous system. J Cell Biol. 1995;131:1789–1800. doi: 10.1083/jcb.131.6.1789. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Vaynman S, Ying Z, Gomez-Pinilla F. Exercise induces BDNF and synapsin I to specific hippocampal subfields. J Neurosci Res. 2004;76:356–362. doi: 10.1002/jnr.20077. [DOI] [PubMed] [Google Scholar]