Abstract
Objective:
Autologous chondrocyte transplantation has become an established therapy for full-thickness cartilage defects. Cell-seeded collagen matrix–supported autologous chondrocyte transplantation (ACT-CS) has been introduced as a modification of conventional ACT, which allows easier handling and is intended to combine the advantages of using a cell suspension (i.e., cell viability and mitotic activity) with the stability and self-containment provided by a matrix of biomaterials. Unlike other techniques and products, this seeding step can be easily applied using a porcine collagen type I/III membrane and autologous chondrocytes in an operating room setting. Although some suturing is required, this technique provides the distinct advantage of not requiring a water-tight seal of the bilayer membrane, as is required using the classic cell suspension technique. Comparable to other modifications of ACT, the ACT-CS procedure requires a specific surgical technique that focuses on the following important details: (1) accurate debridement of the cartilage defect; (2) preparation of the cells, and seeding and containment of the cells within the transplantation site; and (3) sealing and suturing around the defect.
Design:
A consensus meeting of leading European orthopedic surgeons specializing in cartilage repair was convened to discuss and standardize the surgical aspects of this technique.
Results & Conclusions:
The present article describes and discusses the adoption of these best surgical practices for implementing the ACT-CS technique, including more detailed descriptions of each phase of the surgery in order to standardize and optimize patient outcomes.
Keywords: autologous chondrocyte implantation, collagen membrane, cartilage defect, cartilage repair, cell transplantation
Introduction
Autologous chondrocyte transplantation (ACT) was first described in 1994 and has become an accepted therapy for symptomatic full-thickness cartilage defects of the knee joint1,2 and osteochondritis dissecans.3,4 Recently, structural and clinical superiority compared with arthroscopic microfracture has been reported,5,6 which supports the hypothesis that using well-characterized cell-based therapies can predict a better structural cartilage repair that can also result in a beneficial clinical outcome. Several studies have also reported midterm and even some long-term results with success rates between 80% and 90%.2,3,6-10
Since its introduction, many adaptations have been made to the original surgical technique using a cell suspension. Originally, an autologous piece of periosteum was harvested and sutured over the debrided cartilage lesion. The cell suspension was then injected beneath the sutured and sealed periosteal flap. This procedure was tedious and required additional surgical time, and the periosteal flap was often fragile, difficult to manipulate, and susceptible to holes, tears, and leaks. It was subsequently observed that the periosteal flap was also associated with graft hypertrophy, frequently requiring additional surgical intervention.11-13 More recently, resorbable biomaterials have been developed, for example, a porcine collagen type I/III membrane (Chondro-Gide®, Fa. Geistlich, Wolhusen, Switzerland)14-16 that can substitute for the periosteal flap. Not only does this biomaterial preclude the tedious harvesting of the periosteum, it is also more robust and less predisposed to tears. In addition, it has reduced the incidence of hypertrophy. This development is today considered as the “second generation” of ACT.17
Following the second generation of ACT, many attempts have been undertaken to combine cultured autologous chondrocyte with 3-dimensional matrix systems prior to implantation (“third generation”).17 Bartlett et al.18 demonstrated that the results from patients randomized to receive cell suspension–based autologous chondrocyte implantation versus matrix-induced autologous chondrocyte implantation were equivalent at the 1-year follow-up. Because the cells are retained and seeded within the matrix, a covering layer (periosteal or biomembrane) is no longer required. Furthermore, some matrices are bioadhesive and allow for direct implantation without suturing.19 At the present time, the majority of the clinical studies using this technology have been large case series observations demonstrating a significant clinical improvement over pre-surgery assessments with observations up to 5 years. In a recent review of 18 studies (total of 731 patients) with an average follow-up of 27.3 months20 Kon et al. reported that only 2 were found to be randomized controlled studies, and the majority (11) were prospective cohort studies or case series. The authors concluded that the limitations in the overall study designs and quality make definitive conclusions regarding the efficacy of this technique difficult. There are also some indications that basal and lateral integration with the surrounding normal cartilage may be lower compared with first-generation cell suspension techniques.13 In contrast to cell suspension, these cells are cultured into the matrix for several weeks prior to implantation, but there is a risk of the accumulation of extracellular matrix and cell maturation hampering integration.
Recently, a small randomized clinical trial compared the clinical outcomes of patients with symptomatic cartilage defects treated with matrix-induced autologous chondrocyte implantation versus those treated with microfracture. Matrix-induced autologous chondrocyte implantation was significantly more effective at 24 months versus baseline than the microfracture treatment was, according to 4 different validated outcome measures. Both procedures had a favorable safety profile with no significant differences between the groups.21
Although there is still some debate over the comparative efficacy of the first- and second-/third-generation products, the advantages of each should be considered in optimizing a therapeutic modality for the patient and surgeon. Accordingly, we have adopted a new procedure which combines the potential benefits of the former generations. We modified a second-generation product: the cell suspension is directly seeded onto the collagen membrane prior to implantation into the defect (autologous chondrocyte transplantation–cell seeded [ACT-CS]). This cell-seeded membrane is then sutured into the cartilage defect (cell-side down) and sealed with fibrin glue. This technique has been described by Matthias Steinwachs in 200922 and the first clinical results have been recently reported.23 This procedure allows the patient and surgeon to select their preferred cell expansion provider, retains the robustness and surgical ease of handling of a biomaterial, as opposed to a periosteal flap, enhances the homogeneity of cell distribution on to the membrane surface, requires a less tedious suturing procedure and avoids the increased incidence of hypertrophy associated with the periosteal flap covering for ACT.
Because the technique may be open to various interpretations of methodology of implementation and the possible variations that may be introduced by individual surgeons, a panel of experts convened during an ACT-CS Consensus Meeting. The goal was to incorporate the best clinical practices from the attendees and come to a consensus on standard operational procedures. Therefore, the primary purpose of this article is to provide a detailed description of the surgical procedure to be adopted by the orthopedic community.
The authors of the present article are aware that many of the recommendations provided are expert opinions. Some of them are supported by scientific publications; others are opinions with regard to personal experience (expert opinions, EBM Level IV).24 The authors acknowledge that this should be an evolving document and encourage any scientific work that may be conducted in the future to further test and elaborate on the recommendations given in the present publication. The authors consider it to be important to share their opinion and experience in order to further unify surgical techniques of ACT and initiate a scientific and clinical open forum to further improve this technique.
Principle of ACT-CS and Potential Benefits
Cell-seeded collagen matrix–supported autologous-chondrocyte transplantation (ACT-CS) is a technique that combines a suspension of in vitro expanded articular chondrocytes seeded onto a porcine collagen type I/III membrane (Chondro-Gide®, Fa. Geistlich) before transplantation in the operating room.22 The idea behind this technique is that in using ACT-CS it is possible to combine the benefits of cells delivered as a cell suspension with the benefits of matrix-associated ACT technique. Furthermore, by having isolated adherent cells on the membrane rather than embedded within the matrix,25 it is thought that the cells would not be encumbered by a cell matrix barrier to allow basal and lateral integration with the surrounding native tissue. The membrane in the ACT-CS technique is only used as a cell carrier to deliver chondrocytes to the chondral defect. Figure 1 demonstrates the characteristic distribution of chondrocytes using ACT-CS 5–10 minutes after cell seeding. Notably, the chondrocytes are densely distributed along the surface of the membrane and not embedded within a matrix, in contrast to matrix-associated autologous chondrocyte implantation (MACI).26 In addition, the occlusive side of the Chondro-Gide® membrane also acts as a barrier to an influx of surrounding cells (fibroblasts, synoviocytes, etc.) from being incorporated into the regenerative cartilage. Because the cells are already adherent, the possibility of cell loss due to leaks and seepage may be minimized and accordingly requires less stitching of the membrane to the surrounding tissue. This may be a particular advantage when repairing lesions that are anatomically less accessible, such as posterior parts of the tibia or femoral condyles. A further theory about the ACT-CS technique is that cell seeding could result in a more homogenous distribution of the cells within the defect compared with cell suspension techniques, but this theory still lacks scientific evidence and has not been proven so far.
Figure 1.
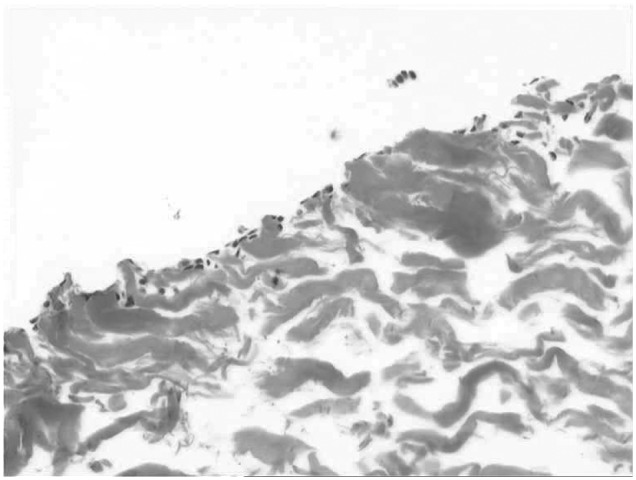
Cell distribution of chondrocytes 10 minutes following seeding on porcine collagen I/III (magnification 10x)
Surgical Technique
Preparation of the Defect
According to accepted recommendations for all ACT procedures, any damaged cartilage should be removed completely and debrided until the defect is surrounded by a well-defined rim of healthy cartilage.1,27,28 The authors recommend a small scalpel (such as No. 15) for the first cut to sharply define the edge of the defect. Afterwards, sharp instruments such as a sharp spoon or a ring curette should be used to carefully remove all damaged cartilage. The subchondral bone plate should not be penetrated and should be left intact. Bleeding from the subchondral bone should be avoided to minimize the risk of infiltration of bone marrow–derived cells into the implant, which may result in fibrocartilage tissue. A negative effect of blood on biochemical and morphological properties of regenerated cartilage has been demonstrated.29,30 In addition, direct contact of the seeded cellular layer of the membrane, unencumbered by a bone marrow–derived cellular infiltrate, may allow for better basal integration.
Subchondral Plate and Bleeding
In case subchondral bone bleeding occurs during debridement, one may address it in one of several ways. For example, one approach is to apply a drop of fibrin glue to the bleeding surface to induce clotting. The glue can be pressed into the opened subchondral bone using a small swab, removing any excess glue after the bleeding is controlled.
Intralesional osteophytes
In case of intralesional osteophytes, often observed following prior bone marrow–stimulating techniques such as microfracture,31 osteophytes should be removed and resected to the level of the surrounding subchondral bone plate. No sclerotic subchondral bone should remain, since it is considered to inhibit basal integration of the implantation with normal tissue. Another way to address small osteophytes is gentle impaction.
Preparation of the Membrane
Following debridement of the lesion, a template of the defect is traced along its edges. Once seeded with the cell suspension, an enlargement of the collagen membrane by approximately 10% occurs. However, the enlargement is not uniform and is influenced by the orientation of the collagen fibers. A space of approximately 1 mm should be left between the template and the surrounding cartilage rim, and the size should be checked again before suturing.
Cell Resuspension
During shipment, the cell suspension commonly settles into a pellet at the bottom of the tube. The cells must first be resuspended. The total volume of suspension fluid can be adapted to the size of the membrane. To do so, excess volume of suspension medium should be discarded prior to cell resuspension. Often, a gentle rocking back and forth of the container will be sufficient to resuspend the cells. A plastic (not metal) 18-gauge (or larger) cannula may also be used. Alternatively, a plastic (not metal) cannula system can be used for resuspension, and a minimum size of 18 gauge is recommended in order to not harm the cells.
Cell Number
The optimal number of chondrocytes per square centimeter may be product-specific and the product insert should be referred to for each preparation. There are no well-controlled clinical trials comparing various doses of cells per square centimeter and clinical outcomes. Most frequently, the dosing studies have been based on large-scale animal data. In general, commercially available sources of chondrocyte suspensions recommend a dose between 0.5 and 2.0 × 106 cells per cm2. In the clinical experience of the authors using ACT-CS, membranes have been seeded at approximately 2 to 3 × 106 for a 4-cm2 collagen membrane.
Cell Seeding Technique
The Chondro-Gide® membrane is delivered in double-packed sterile plastic foil. The authors suggest using the inner packaging for cell application in the operation theater. After resuspension of the delivered chondrocytes in a small amount of fluid (approximately 300–1000 µL), cells are applied on the rough side of the collagen membrane using a plastic cannula (18-gauge) by gently dripping the cell suspension until the membrane is saturated (also see Fig. 2). It is preferable to start cell application on the edges of the membrane and move towards the middle of the membrane. Cell seeding should be performed until the entire membrane is covered with cell suspension and it should be stopped before any fluid runs from the membrane.
Figure 2.
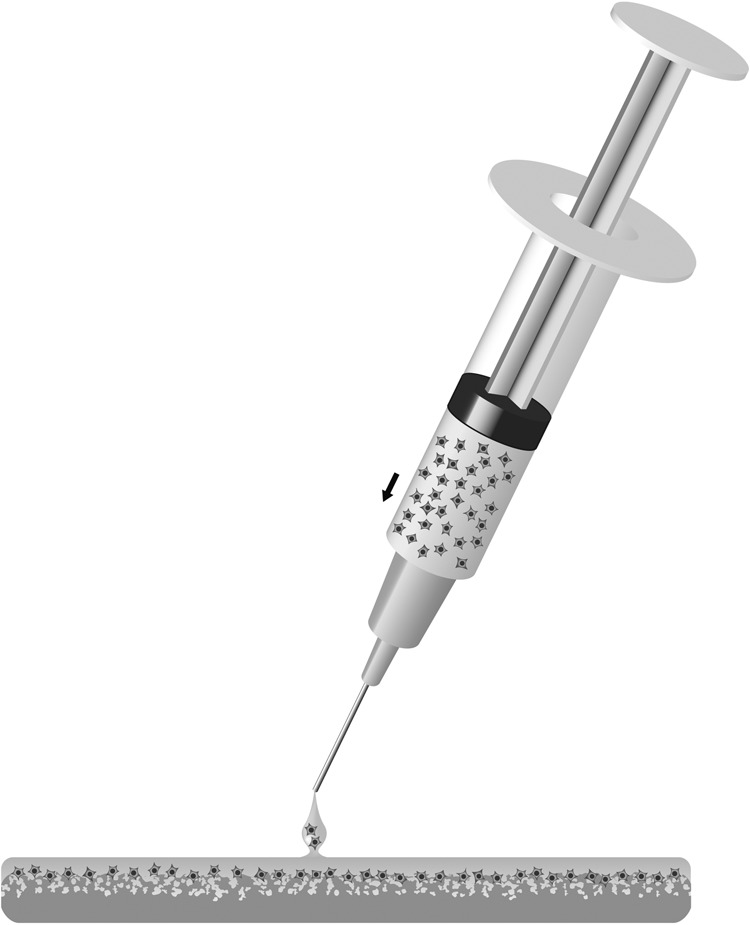
Using the ACT-CS technique, chondrocytes are directly applied to the membrane immediately before implantation into the prepared cartilage defect
Adherence Time
Specific time for cell adherence to the membrane has not been rigorously tested, but 5 to 10 minutes has been found as sufficient to allow cells to adhere to the membrane. In ACT-CS, total cell number per ml is determined by the suspension volume absorbed by the membrane. If a volume is used that exceeds the capacity of the membrane to absorb the suspension, then the solution with cells will run off the membrane. Unequal distribution and excessive drying may occur if too low a volume is used. The authors recommend applying sufficient fluid to the membrane, such that a liquid film is visible on the membrane but not all of the fluid is absorbed completely. Once the cell suspension has been applied and allowed to adhere, small amounts of additional fluid (such as NaCl 0.9%) or a moist compress saturated with media may be added to keep the membrane moist. It is important that any materials or liquids coming into contact with the cells be isotonic and buffered.
Cell distribution on the membrane after 10 minutes is demonstrated in Figure 1. After the 10-minute adhesion time, the membrane may then be implanted into the debrided lesion and sutured. The authors restrict the adhesion/seeding time to 10–15 minutes as they believe this optimizes the ability to more effectively integrate with the subchondral bone plate layer.
Transfer of the Cell-Matrix Construct into the Defect
The seeded membrane must be handled with care as it is implanted into the defect. The authors strongly recommend using a small pair of tweezers or forceps. On implantation, the rough surface of the membrane should face the subchondral bone plate. The membrane edges should not overhang on top of the normal cartilage. If it does, it is important to trim the membrane to size. There should be a direct contact between the cell-seeded surface of the membrane and the subchondral bone.
Fixation of the Membrane
Sutures to fix the membrane to the adjacent cartilage is the classic technique that has previously been described in the original first-generation method.1 In the original technique, a distance of 4–6 mm between suture knots was recommended to ensure stable fixation allowing the injection of the cell suspension beneath the membrane. The number of sutures (hence time of surgery) can be reduced using the ACT-CS technique. The spacing and number of sutures should be sufficient to just allow mechanical stability of the cell-seeded membrane to the defect edges and onto the bottom of the defect. Gaps between the seeded membrane and adjacent cartilage should be avoided, depending on the lesion size, and often 6–12 sutures per defect are sufficient to achieve adequate positioning and stability of the implant.
The authors recommend a monofilament suture material (i.e., PDS 6-0, Fa. Ethicon, Nordersted, Germany) as monofilament sutures are considered more compatible and less of an irritant to normal cartilage and membrane. Although the degradation time for the sutures could play a critical role in clinical outcome, the authors are unaware of any detailed studies that have examined this in order to find an optimal material. Hunziker et al. demonstrated that suturing was associated with some local degeneration based on a histological evaluation,32 but it remains unclear if these observations are of clinical relevance.
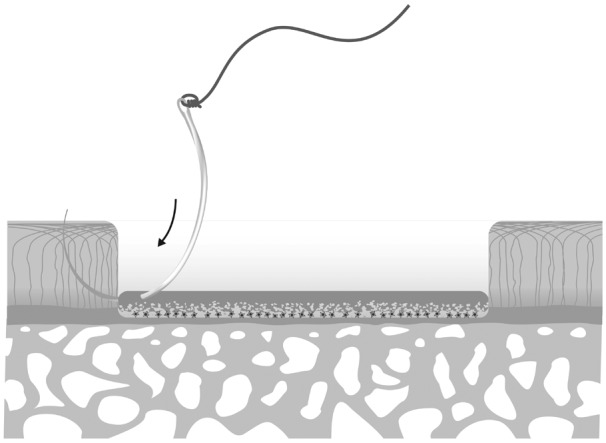
The authors recommend positioning the needle insertion close to the bottom of the defect, in close proximity to the subchondral bone. Positioning the membrane on top of the cartilage should be avoided since this could result in delamination or disintegration to the borders and lifting of the membrane from the defect with any shear force. An example of the preferred technique is shown in Figures 3 and 4.
Figure 3.

Cell-seeded membrane is placed on the bottom of the prepared defect, cell-loaded side of the membrane directed toward and in direct contact with the subchondral bone plate
Figure 4.
PDS 6-0 is recommended as a suture material to fix the cell/membrane construct into the adjacent cartilage. A strict position of the needle close to the subchondral bone needs to be regarded in order to provide a close contact of cells and subchondral bone
There is general agreement that knots can potentially cause problems, especially in a mechanically active joint. Placing the knots on the cartilage surface increases local shear forces and should be avoided. The authors agree that the best position for knots is beneath the surface of adjacent cartilage, directly on the transplanted membrane. This position also forces the membrane to the bottom of the defect directly opposed to the subchondral bone plate. An example of proper positioning of knots is displayed in Figure 5.
Figure 5.

Knots are typically placed under the surface of the adjacent cartilage in order to avoid any irritation of the adjacent cartilage
Sealing and Use of Fibrin Glue
After fixation of the cell-seeded membrane by sutures, the authors recommend an additional sealing of the border using fibrin glue (see Fig. 6). The cell compatibility of fibrin glue and the ability of fibrin to support chondrogenic phenotype has been reported in various studies.33-37 This has also been demonstrated for the combination of fibrin glue and the Chondro-Gide® membrane.38 Any fibrin glue used in ACT-CS should have demonstrated compatibility with chondrocytes and the collagen membrane used. TissuCol (Baxter, Unterschleißheim, Germany) has been shown to have good chondrocyte compatibility,37 and this type of fibrin glue has also been used in the ACT-CS study.23 The amount of fibrin glue should be limited. There is also consensus on the fact that the entire defect should not be filled or covered with fibrin glue.
Figure 6.
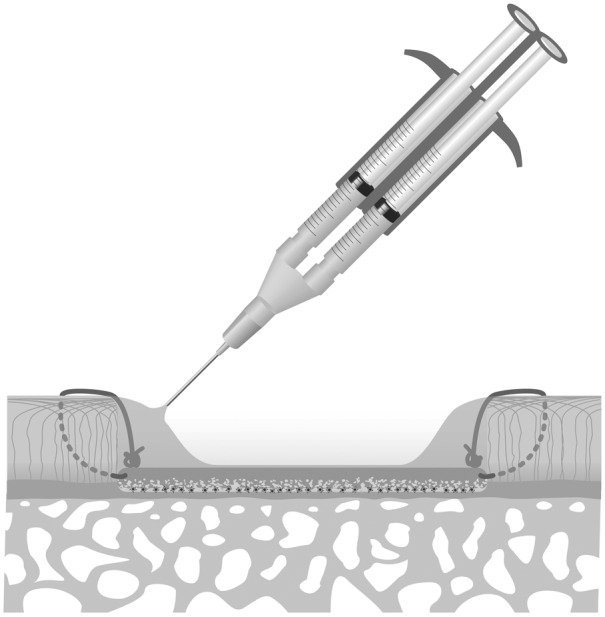
Fibrin glue is carefully placed at the interface of membrane and adjacent cartilage in order to seal this intersection. The amount of fibrin glue should be limited in order to reach an appropriate sealing
Clinical Experience with ACT-CS
ACT-CS as described in the present paper uses a porcine collagen type I/III membrane (Chondro-Gide®, Fa. Geistlich) and represents an adoption of the initially described technique using a cell suspension injected beneath the identical membrane.14,15 This technique has been introduced as the “second-generation” ACT.17 For this technique and for the MACI technique, which also represents an adoption using the identical biomaterial,25 various studies report safety, and midterm clinical outcome has been reported in several case series in the treatment of cartilage defects and osteochondritis dissecans.4,11,14-16,25 All these studies do not report any specific side effects and adverse events in context with the application of the collagen membrane. In addition clinical outcome seems promising and success rates vary between 82% and 95%. Using the collagen membrane seems to further reduce the incidence of graft hypertrophy as demonstrated in a prospective randomized trial versus periosteum-covered ACT11 as well as in large retrospective studies including more than 400 patients with ACT.13 Compared with conventional periosteum-covered ACT, as a possible disadvantage a higher rate of malfusion of the regenerative tissue into the adjacent cartilage has been reported, but this observation does not seem to be specific for the collagen membrane, and it has also been observed in other artificial biomaterials.13
ACT-CS represents an adoption of the initial technique using the collagen membrane for ACT. The first patients were treated using the ACT-CS technique in October 2005, and the principles of the technique were described in 2009.22 Until preparation of the present manuscript, the authors (M.S., P.V., and P.N.) have conducted approximately 250 autologous chondrocyte transplantations using the ACT-CS technique. Clinical 2-year results of the first 59 patients treated with ACT-CS have been reported recently,23 demonstrating a success rate (ICRS “A” and “B” at 24 months) of 89% and a rate of 94 % improved knee function in the subgroup of patients with single defects. Three-year results are upcoming. No technique-related complications were observed during the application of ACT-CS so far. ACT-CS appears to be safe and reproducible. Nevertheless, long-term follow-ups are not yet available and results of ACT-CS used for the treatment for larger defects are still elusive.
Conclusion
In conclusion, although the clinical evidence is limited, the present paper provides concrete guidelines to surgeons on a standardized methodology for using the ACT-CS technique for the treatment of symptomatic full-thickness cartilage defects. All recommendations were based on a consensus meeting of the authors of the present article. The recommended procedures for ACT-CS are based on the authors’ clinical experience in treating more than 200 patients with ACT-CS over the past 5 years. A standardized methodology provides a framework for further comparative studies between various techniques to identify optimal treatment modalities, especially with evolving innovative regenerative medicine products.
Footnotes
References
- 1. Brittberg M, Lindahl A, Nilsson A, Ohlsson C, Isaksson O, Peterson L. Treatment of deep cartilage defects in the knee with autologous chondrocyte transplantation. N Engl J Med. 1994;331(14):889-95. [DOI] [PubMed] [Google Scholar]
- 2. Peterson L, Brittberg M, Kiviranta I, Akerlund EL, Lindahl A. Autologous chondrocyte transplantation. Biomechanics and long-term durability. Am J Sports Med. 2002;30(1):2-12. [DOI] [PubMed] [Google Scholar]
- 3. Peterson L, Minas T, Brittberg M, Lindahl A. Treatment of osteochondritis dissecans of the knee with autologous chondrocyte transplantation: results at two to ten years. J Bone Joint Surg Am. 2003;85-A(Suppl 2):17-24. [DOI] [PubMed] [Google Scholar]
- 4. Krishnan SP, Skinner JA, Carrington RW, Flanagan AM, Briggs TW, Bentley G. Collagen-covered autologous chondrocyte implantation for osteochondritis dissecans of the knee: two- to seven-year results. J Bone Joint Surg Br 2006; 88(2):203-5. [DOI] [PubMed] [Google Scholar]
- 5. Saris DB, Vanlauwe J, Victor J, Haspl M, Bohnsack M, Fortems Y, et al. Characterized chondrocyte implantation results in better structural repair when treating symptomatic cartilage defects of the knee in a randomized controlled trial versus microfracture. Am J Sports Med. 2008;36(2):235-46. [DOI] [PubMed] [Google Scholar]
- 6. Saris DB, Vanlauwe J, Victor J, Almqvist KF, Verdonk R, Bellemans J, et al. Treatment of symptomatic cartilage defects of the knee: characterized chondrocyte implantation results in better clinical outcome at 36 months in a randomized trial compared to microfracture. Am J Sports Med. 2009;37(Suppl 1):10S-19S. [DOI] [PubMed] [Google Scholar]
- 7. Peterson L, Vasiliadis HS, Brittberg M, Lindahl A. Autologous chondrocyte implantation: a long-term follow-up. Am J Sports Med. 2010;38(6):1117-24. [DOI] [PubMed] [Google Scholar]
- 8. Peterson L, Minas T, Brittberg M, Nilsson A, Sjogren-Jansson E, Lindahl A. Two- to 9-year outcome after autologous chondrocyte transplantation of the knee. Clin Orthop Relat Res. 2000;(374):212-34. [DOI] [PubMed] [Google Scholar]
- 9. Kreuz PC, Muller S, Ossendorf C, Kaps C, Erggelet C. Treatment of focal degenerative cartilage defects with polymer-based autologous chondrocyte grafts: four-year clinical results. Arthritis Res Ther. 2009;11(2):R33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Knutsen G, Drogset JO, Engebretsen L, Grontvedt T, Isaksen V, Ludvigsen TC, et al. A randomized trial comparing autologous chondrocyte implantation with microfracture. Findings at five years. J Bone Joint Surg Am. 2007;89(10):2105-12. [DOI] [PubMed] [Google Scholar]
- 11. Gooding CR, Bartlett W, Bentley G, Skinner JA, Carrington R, Flanagan A. A prospective, randomised study comparing two techniques of autologous chondrocyte implantation for osteochondral defects in the knee: periosteum covered versus type I/III collagen covered. Knee. 2006;13(3):203-10. [DOI] [PubMed] [Google Scholar]
- 12. Gomoll AH, Probst C, Farr J, Cole BJ, Minas T. Use of a type I/III bilayer collagen membrane decreases reoperation rates for symptomatic hypertrophy after autologous chondrocyte implantation. Am J Sports Med. 2009;37(Suppl 1):20S-23S. [DOI] [PubMed] [Google Scholar]
- 13. Niemeyer P, Pestka JM, Kreuz PC, Erggelet C, Schmal H, Suedkamp NP, et al. Characteristic complications after autologous chondrocyte implantation for cartilage defects of the knee joint. Am J Sports Med. 2008;36(11):2091-99. [DOI] [PubMed] [Google Scholar]
- 14. Haddo O, Mahroof S, Higgs D, David L, Pringle J, Bayliss M, et al. The use of Chondrogide membrane in autologous chondrocyte implantation. Knee. 2004;11(1):51-5. [DOI] [PubMed] [Google Scholar]
- 15. Steinwachs MR, Kreuz PC. Clinical results of autologous chondrocyte transplantation (ACT) using a collagen membrane. In: Henrich C, Nöth U, Eulert J, editors. Cartilage surgery and future perspectives. Berlin: Springer; 2003. p. 37-48. [Google Scholar]
- 16. Steinwachs M, Kreuz PC. Autologous chondrocyte implantation in chondral defects of the knee with a type I/III collagen membrane: a prospective study with a 3-year follow-up. Arthroscopy. 2007;23(4):381-7. [DOI] [PubMed] [Google Scholar]
- 17. Marlovits S, Zeller P, Singer P, Resinger C, Vecsei V. Cartilage repair: generations of autologous chondrocyte transplantation. Eur J Radiol. 2006;57(1):24-31. [DOI] [PubMed] [Google Scholar]
- 18. Bartlett W, Skinner JA, Gooding CR, Carrington RW, Flanagan AM, Briggs TW, et al. Autologous chondrocyte implantation versus matrix-induced autologous chondrocyte implantation for osteochondral defects of the knee: a prospective, randomised study. J Bone Joint Surg Br. 2005;87(5):640-5. [DOI] [PubMed] [Google Scholar]
- 19. Kon E, Gobbi A, Filardo G, Delcogliano M, Zaffagnini S, Marcacci M. Arthroscopic second-generation autologous chondrocyte implantation compared with microfracture for chondral lesions of the knee: prospective nonrandomized study at 5 years. Am J Sports Med. 2009;37(1):33-41. [DOI] [PubMed] [Google Scholar]
- 20. Kon E, Verdonk P, Condello V, Delcogliano M, Dhollander A, Filardo G, et al. Matrix-assisted autologous chondrocyte transplantation for the repair of cartilage defects of the knee: systematic clinical data review and study quality analysis. Am J Sports Med. 2009;37(Suppl 1):156S-66S. [DOI] [PubMed] [Google Scholar]
- 21. Basad E, Ishaque B, Bachmann G, Sturz H, Steinmeyer J. Matrix-induced autologous chondrocyte implantation versus microfracture in the treatment of cartilage defects of the knee: a 2-year randomised study. Knee Surg Sports Traumatol Arthrosc. 2010;18(4):519-27. [DOI] [PubMed] [Google Scholar]
- 22. Steinwachs M. New technique for cell-seeded collagen-matrix-supported autologous chondrocyte transplantation. Arthroscopy. 2009;25(2):208-11. [DOI] [PubMed] [Google Scholar]
- 23. Niemeyer P, Lenz P, Kreuz PC, Salzmann GM, Sudkamp NP, Schmal H, et al. Chondrocyte-seeded type I/III collagen membrane for autologous chondrocyte transplantation: prospective 2-year results in patients with cartilage defects of the knee joint. Arthroscopy. 2010;26(8):1074-82. [DOI] [PubMed] [Google Scholar]
- 24. Obremskey WT, Pappas N, Attallah-Wasif E, Tornetta P, 3rd, Bhandari M. Level of evidence in orthopaedic journals. J Bone Joint Surg Am. 2005;87(12):2632-8. [DOI] [PubMed] [Google Scholar]
- 25. Behrens P, Bitter T, Kurz B, Russlies M. Matrix-associated autologous chondrocyte transplantation/implantation (MACT/MACI)—5-year follow-up. Knee. 2006;13(3):194-202. [DOI] [PubMed] [Google Scholar]
- 26. Gigante A, Bevilacqua C, Ricevuto A, Mattioli-Belmonte M, Greco F. Membrane-seeded autologous chondrocytes: cell viability and characterization at surgery. Knee Surg Sports Traumatol Arthrosc. 2007;15(1):88-92. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Minas T, Nehrer S. Current concepts in the treatment of articular cartilage defects. Orthopedics. 1997;20(6):525-38. [DOI] [PubMed] [Google Scholar]
- 28. Peterson L. Articular cartilage injuries treated with autologous chondrocyte transplantation in the human knee. Acta Orthop Belg. 1996;62(Suppl 1):196-200. [PubMed] [Google Scholar]
- 29. Sosio C, Boschetti F, Mangiavini L, Scotti C, Manzotti S, Buragas MS, et al. Blood exposure has a negative effect on engineered cartilage. Knee Surg Sports Traumatol Arthrosc. 2011;19(6):1035-42. [DOI] [PubMed] [Google Scholar]
- 30. Sosio C, Boschetti F, Bevilacqua C, Mangiavini L, Scotti C, Buragas MS, et al. Effect of blood on the morphological, biochemical and biomechanical properties of engineered cartilage. Knee Surg Sports Traumatol Arthrosc. 2007;15(10):1251-7. [DOI] [PubMed] [Google Scholar]
- 31. Minas T, Gomoll AH, Rosenberger R, Royce RO, Bryant T. Increased failure rate of autologous chondrocyte implantation after previous treatment with marrow stimulation techniques. Am J Sports Med. 2009;37(5):902-8. [DOI] [PubMed] [Google Scholar]
- 32. Hunziker EB, Stahli A. Surgical suturing of articular cartilage induces osteoarthritis-like changes. Osteoarthritis Cartilage. 2008;16(9):1067-73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Chang J, Rasamny JJ, Park SS. Injectable tissue-engineered cartilage using a fibrin sealant. Arch Facial Plast Surg. 2007;9(3):161-6. [DOI] [PubMed] [Google Scholar]
- 34. Dare EV, Griffith M, Poitras P, Wang T, Dervin GF, Giulivi A, et al. Fibrin sealants from fresh or fresh/frozen plasma as scaffolds for in vitro articular cartilage regeneration. Tissue Eng Part A. 2009;15(8):2285-97. [DOI] [PubMed] [Google Scholar]
- 35. Dickhut A, Gottwald E, Steck E, Heisel C, Richter W. Chondrogenesis of mesenchymal stem cells in gel-like biomaterials in vitro and in vivo. Front Biosci. 2008;13:4517-28. [DOI] [PubMed] [Google Scholar]
- 36. Dare EV, Vascotto SG, Carlsson D, Hincke MT, Griffith M. Differentiation of a fibrin gel encapsulated chondrogenic cell line. Int J Artif Organs. 2007;30(7):619-27. [DOI] [PubMed] [Google Scholar]
- 37. Silverman RP, Passaretti D, Huang W, Randolph MA, Yaremchuk MJ. Injectable tissue-engineered cartilage using a fibrin glue polymer. Plast Reconstr Surg. 1999;103(7):1809-18. [DOI] [PubMed] [Google Scholar]
- 38. Gille J, Meisner U, Ehlers EM, Muller A, Russlies M, Behrens P. Migration pattern, morphology and viability of cells suspended in or sealed with fibrin glue: a histomorphologic study. Tissue Cell. 2005;37(5):339-48. [DOI] [PubMed] [Google Scholar]


