Abstract
Our previous study has shown berberine prevents damage to the intestinal mucosal barrier during early phase of sepsis in rat through mechanisms independent of the NOD-like receptors signaling pathway. In this study, we explored the regulatory effects of berberine on Toll-like receptors during the intestinal mucosal damaging process in rats. Male Sprague-Dawlay (SD) rats were treated with berberine for 5 d before undergoing cecal ligation and puncture (CLP) to induce polymicrobial sepsis. The expression of Toll-like receptor 2 (TLR 2), TLR 4, TLR 9, the activity of nuclear factor-kappa B (NF-κB), the levels of selected cytokines and chemokines, percentage of cell death in intestinal epithelial cells, and mucosal permeability were investigated at 0, 2, 6, 12 and 24 h after CLP. Results showed that the tumor necrosis factor-α (TNF-α ) and interleukin-6 (IL-6) level were significantly lower in berberine-treated rats compared to the control animals. Conversely, the expression level of tight junction proteins, percentage of cell death in intestinal epithelial cells and the mucosal permeability were significantly higher in berberine-treated rats. The mRNA expression of TLR 2, TLR 4, and TLR 9 were significantly affected by berberine treatment. Our results indicate that pretreatment with berberine attenuates tissue injury and protects the intestinal mucosal barrier in early phase of sepsis and this may possibly have been mediated through the TLRs pathway.
Keywords: Berberine, Cecal ligation and puncture, Intestinal mucosal barrier, Intra-abdominal infections, Toll-like receptors
INTRODUCTION
Sepsis and septic multiple organ dysfunction and injury may lead to a high morbidity and mortality rate in recent years despite of better supportive therapy and care [1,2]. To date the exact mechanisms responsible for sepsis remain unclear, but previous studies have demonstrated that intestinal function plays a critical role in the development of sepsis, and increased intestinal permeability is associated with the development of multiple organ dysfunction syndrome [3,4,5]. Indeed, the intestinal epithelium, which is constantly exposed to bacterial products, is the first line of defense against microorganisms. An intact and functioning intestinal mucosal barrier, maintained by the intestinal epithelial cells, is crucial to prevent intestinal flora, which contains infectious agents like bacteria, from crossing via both transcellular and paracellular pathways [6]. Therefore, it is critical to protect gut barrier function when providing treatment for sepsis.
Previous studies have shown that the tight junction proteins, such as zonula occludens (ZO), occludin and claudins, are critical to the maintenance of the intact intestinal epithelial barrier [7,8]. However, the intestinal barrier function is frequently disrupted in a variety of acute or chronic enteropathies including inflammatory bowel disease, irritable bowel syndrome, and infectious diarrhea. Some pro-inflammatory cytokines, such as TNF-α, IL-6, have been found to contribute to the disruption of intestinal epithelial barrier function [9].
Berberine is a botanical alkaloid isolated from the root and bark of Rhizoma coptidis, an ancient Chinese herb that has been used for thousands of years in China as a traditional herbal medicine to treat gastrointestinal disorders and bacterial infection, and there have been no toxic effects reported to date in clinical studies [10,11]. According to former studies, berberine has been reported to exhibit anti-inflammatory properties in modern medicine [12]. It blocked the NF-κB signaling pathway, suppressed the inflammatory mediator proteins expression, such as TNF-α, IL-1β, cyclooxygenase-2 (COX-2) and inducible nitric oxide synthase (iNOS) in vitro [13,14,15,16,17]. Further evidences demonstrate berberine protects barrier function in both endothelial and epithelial cells [18,19,20]. However, the molecular mechanisms involved in these protective effects of berberine remain to be elucidated.
Toll-like receptors (TLR) and NOD-like receptors play a crucial role in host defense against intestinal infection [21]. Our previous study has shown that berberine prevents damage to the intestinal mucosal barrier during early phase of sepsis in rat through mechanisms independent of the NOD-like receptors signaling pathway. In this study, we further investigated the effects of berberine in preventing the damage on the intestinal mucosa in rats and sought to test the hypothesis that treatment with berberine attenuates tissue injury and protects the intestinal mucosal barrier in early phase of sepsis through Toll-like receptors (TLRs) signaling pathway.
METHODS
All experimental procedures were approved by the Animal Study Ethics Committee of Nanjing General Hospital of Nanjing Military Command and were performed in accordance with the institutional criteria for the care and use of laboratory animals in research.
Animals
Adult male Sprague-Dawlay (SD) rats, weighing 200~230 g, were obtained from the Nanjing General Hospital of Nanjing Military Command, PLA, Nanjing, China. Rats were housed individual cages in a restricted-access room with controlled conditions (22±1℃ and 65~70% humidity). Food and water were provided ad libitum.
Berberine treatment establishment of polymicrobial sepsis model
A total of 60 male SD rats were randomly divided into two groups (30 rats/group): normal saline group (NS group) and berberine group (Ber group) according to the reagent by intragastric administration; Berberine was dissolved in saline solution before injection and volumes of the injections were adjusted so that each rat received a final dosage of 50 mg/kg bodyweight. Berberine was administered for 5 d before the polymicrobial sepsis model establishment. In the NS group, rats only received same volume of saline solution daily for 5 d. On the fifth day of the Berberine treatment, polymicrobiol sepsis was induced in the rats by cecal ligation and puncture (CLP) one hour after the last injection. CLP is a reliable model for mimicking clinical polymicrobial sepsis [22,23]. Briefly, after rats were anesthetized by intraperitoneal injection of ketamine, a midline incision about 2cm was made on the anterior abdomen. The cecum was carefully isolated and the distal 30% was ligated. Then the ligated portion was punctured through twice with a sterile 18-gauge needle. Small amount of fecal material was released from the wounds by gentle squeezing. The cecum was then placed back and the incision was closed.
Tissue collection
Tissue samples were collected from the rats at 0 h, 2 h, 6 h, 12 h and 24 h after CPL described by our previous report [24]. Each time six rats were removed from both the NS group and the Ber group. The 0 h sampling was done immediately after the NS procedure. And for the other time spots, rats were anesthetized before the tissue collections. Up to 4 ml of blood was drawn from the inferior vena cava. Serum was isolated by centrifugation at 4,000 g for 10 min after clotting and stored at -80℃ until analysis. After blood collection, the rats were decapitated under anesthesia; a section of the small intestine (~10 cm of the ileum) was excised from the rats. After cleaning of the fecal material, the mucosa was carefully removed and stored at -80℃ for later analysis.
TLR2, TLR4 and TLR9 mRNA levels in the mucosa of the small intestine
Expressions of TLR2, TLR4 and TLR9 mRNA in rat intestine were assessed by reverse transcription quantitative polymerase chain reaction (RT-qPCR) as described previously [24]. Briefly, the frozen mucosa tissue was ground into fine powder with mortar and pestle in liquid nitrogen. Total RNA was extracted with TriZol reagent (Life Technologies, Invitrogen, Carlsbad, CA) following the manufacturer's instructions. All RNA samples were treated with DNase I before RT-PCR to eliminate DNA residues and final concentrations of the RNA samples were determined by spectrophotometric optical density measurement at 260 nm. 1 µg total RNA from each sample was reverse transcribed into cDNA using a Reverse Transcription System Kit (TOYOBO, Shanghai, China).
PCR reactions were carried out using a Rotorgene 3000A qPCR machine (Corbett Reseach Inc.). Each reaction contains 2 µl of cDNA sample, 12.5 µl qPCR master mix, 0.2 µl of forward and reserve primers each, and ddH2O. The total reaction volume is 25 µl. Glyceraldehyde-3-phosphate dehydrogenase (GAPDH) was used as a normalization control. The primer sequences were:
TLR2 (sense): 5'-TGCTATGATGCCTTTGTTTCC-3',
TLR2 (antisense): 5'-CATGAGGTTCTCCACCCAAT-3';
TLR4 (sense): 5'-GGATGATGCCTCTCTTGCAT-3',
TLR4 (antisense): 5'-TGATCCATGCATTGGTAGGTAA-3';
TLR9 (sense): 5'-GCTGGGACGTCTGGTACTGT-3',
TLR9 (antisense): 5'-CACGGGTCAGCAAAGGTAG-3';
GAPDH (sense): 5'-AACGACCCCTTCATTGACCTC-3',
GAPDH (antisense): 5'-CCTTGACTGTGCCGTTGAACT-3'.
PCR conditions were 5 min at 95℃ followed by 40 cycles of 20s at 95℃, 20s at 52℃, and 20s at 72℃. The last cycle was followed by a final incubation from 72℃ to 99℃ for 3 min (℃/5s) to establish a PCR melting curve. Relative quantities of NOD2 and NLRP3 mRNA were analyzed using the 2-ΔΔCT method [25].
Serum TNF-α and IL-6 concentrations
The TNF-α and IL-6 concentrations in the blood serum were measured by commercial ELISA kits (R&D Systems, Inc.) according to the manufacturers' instructions.
ZO-1 protein levels in intestinal mucosa tissue
The expression of ZO-1 in intestinal mucosa was measured by western blot as described previously [24]. A small piece of intestinal mucosa tissue (50 mg) was placed in 1ml of RIPA protein lysate and incubated on ice for 30 min. The reaction mix was then transferred to a 1.5 ml microcentrifuge tube and centrifuged at 4℃ for 10 min at 12,000 g. The protein concentrations in the supernatant were determined by Coomassie brilliant blue taining method. Prior to use, concentrations in all samples were normalized to 2 µg/µl.
The extracted protein mixture was separated by electrophoresis on a 10% sodium dodecyl sulfate-polyacrylamide minigel (SDS-PAGE) and transferred onto membranes at 0.8 mA/cm2 for 2 h. Bands were probed with enzyme-conjugated anti- ZO-1 antibodies. The protein bands were detected by enhanced chemiluminescence and quantified by Image J software (National Institutes of Health, Bethesda, Maryland).
Histology studies
Laser scanning confocal microscope (LSCM)
After fixed by embedding medium, trimmed terminal ileum of rats were fixed in liquid nitrogen and series of slices were prepared by a freezing microtome (Leica, Germany) at -20℃ with the thickness of 5 µm. Slides were then stored in sealed box at -80℃ for LSCM.
The prepared frozen slides were incubated at room temperature after the addition of 3% H2O2, blocked with 10% normal goat serum for 20 min, and incubated with mouse monoclonal anti- ZO-1 antibody (Invitrogen) overnight at 4℃. The sections were incubated at 37℃ for another 2 h after the addition of rabbit anti-mouse IgG antibody conjugated with fluorescein isothiocyanate (FITC) (Invitrogen, CA, USA). The slides were then sealed with 10% phosphate buffered saline (PBS) containing 90% glycerin and observed by a Laser scanning confocal microscope (LSCM-510, Carl Zeiss, Germany) with an excitation wavelength of 552 nm and an emission wavelength of 570 nm.
Cell death
Apoptotic cells in intestinal epithelial cells was detected using the terminal deoxynucleotidyl transferase-mediated deoxyuridine triphosphate nick-end labeling (TUNEL) assay (In Situ Cell Death Detection Kit, Roche, Switzerland), following the manufacturer's instruction. Briefly, frozen sections of intestinal mucosa were fixed with 4% paraformaldehyde for 20 min at 20℃. After washed with PBS for 30 min, sections were blocked with 3% H2O2 in methanol for 10 min at 20℃, PBS washed and then incubated with 1% Triton X-100 and 0.1% NaN3 at 4℃ for 8 min for permeabilization. Sections were washed with PBS and reacted with terminal deoxynucleotidyl transferase enzyme for 30 min at 37℃. Slides were rinsed with PBS and incubated with 50 µl DAB substrate for 10 min at 20℃. Slides were washed before haematoxylin was used for nuclear staining and observed with the light microscopy.
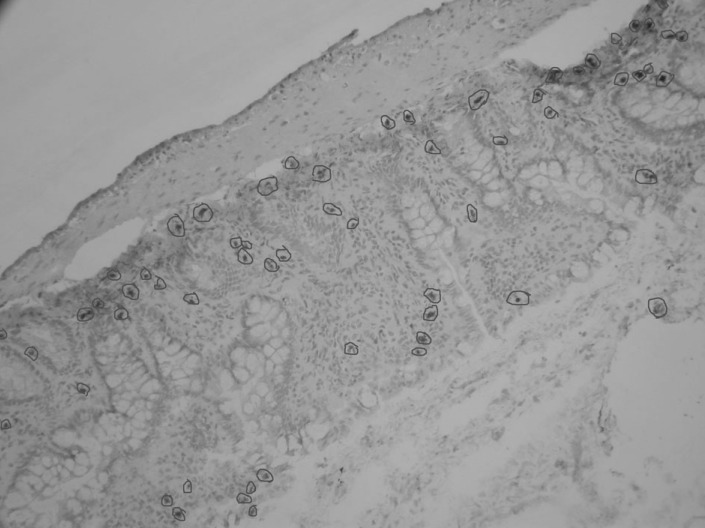
The following three types of cell morphology were defined as positive cell death: (1) cytoplasm and chromatin condensation; (2) apoptotic-body formation in cytoplasm; (3) chromatin fragmentation. The percentage of cell death in intestinal epithelial cells was calculated according to the following formula: (number of dead cells)/(number of dead cells+number of live cells)×100%. The percentage of cell death was counted in randomly selected the magnification view (Fig. 1); the counted cell number was not less than 1000.
Fig. 1.
The percentage of cell death counting in a randomly selected the magnification view (200×), death cells were marked with red circles.
Intestinal permeability test
Flux of fluorescein isothiocyanate (FITC)-conjugated dextran (FITC-dextran, 4 kDa, Sigma) was used to represent the paracellular permeability of intestinal mucosal barrier as described previously. Briefly, after anesthesia, ligation were performed at both sides of a section of ileum (about 20 cm) from the rats at each sampling points after CLP, the blood supply of this section of ileum was not interrupted. 1 ml of 25 mg/ml FITC-dextran was gently injected into the ileum. Sixty min later, 4 mL plasma was drawn from the inferior vena cava and the fluorescence was determined using a fluorescent plate reader (Fluorescence spectrophotometer-F-7000, Hitachi Limited, Japan) with an excitation wavelength of 480 nm and an emission wavelength of 520 nm. The paracellular flux was calculated from a standard curve and expressed as mg/L.
Statistical analysis
Differences between the Ber group and the NS group were analyzed using ANOVA for all parameters investigated. All calculations were performed with SPSS13.0 (SPSS incorporated, Chicago, USA). Differences with p<0.05 were considered to be significant. Data were expressed as mean±S.D. in results.
RESULTS
TLR2, TLR4 and TLR9 mRNA expressions in rat intestinal mucosa
Expression levels of TLR2, TLR4 and TLR9 mRNA are shown in Table 1. mRNA level of intestinal TLR2 increased continuously at 2 h and peaked at 12 h after CLP in both Ber group and the NS group. TLR4 mRNA level remained low during the entire experimental period. TLR9 mRNA level in the Ber group remained higher than that in the NS group during the entire experimental period. For TLR2 and TLR4, mRNA levels was significantly lower in the Ber group than the NS group at 6h (p<0.01, Table 1); whereas, for TLR9, mRNA levels was significantly higher in the Ber group than the NS group at 6 h.
Table 1.
In our study, NS group referred to normal saline group
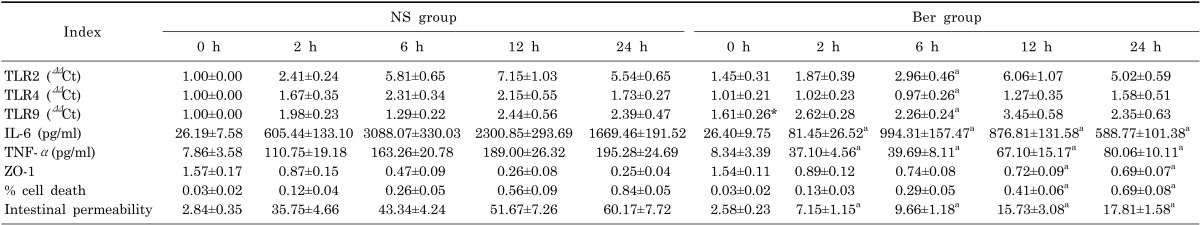
ap<0.01, vs NS group.
Serum concentration of TNF-α and IL-6
CLP resulted in continuous increase in serum IL-6 and TNF-α concentration in both the NS group and the Ber group. However, at all sampling points from 2 h to 24 h, serum IL-6 and TNF-α concentrations in the Ber group were significantly lower than those in the NS group (p<0.01, Table 1).
Zonula occludens-1 (ZO-1) in rat intestinal mucosa
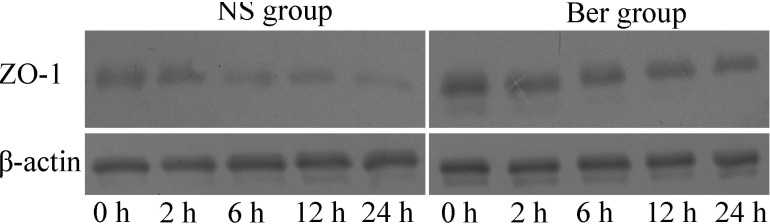
The expression of tight junction (TJ) component proteins ZO-1 decreased continuously after CLP in both NS and Ber groups. However, the decrease appeared to be slower, due to the ZO-1 level in the Ber group was significantly higher than that in NS group at 12 h or 24 h after CLP (p<0.05, Fig. 2).
Fig. 2.
Levels of zonula occludens -1 (ZO-1) in protein extract from rat intestinal mucosa. β-actin was used as the reference for comparison of ZO-1 levels from different samples. The expression of tight junction (TJ) component proteins ZO-1 decreased continuously after CLP in both NS and Ber groups. However, the decrease appeared to be slower, due to the ZO-1 level in the Ber group was significantly higher than that in NS group at 12 h or 24 h after CLP (p<0.05).
Detection of ZO-1 in fixed rat intestinal mucosa tissue
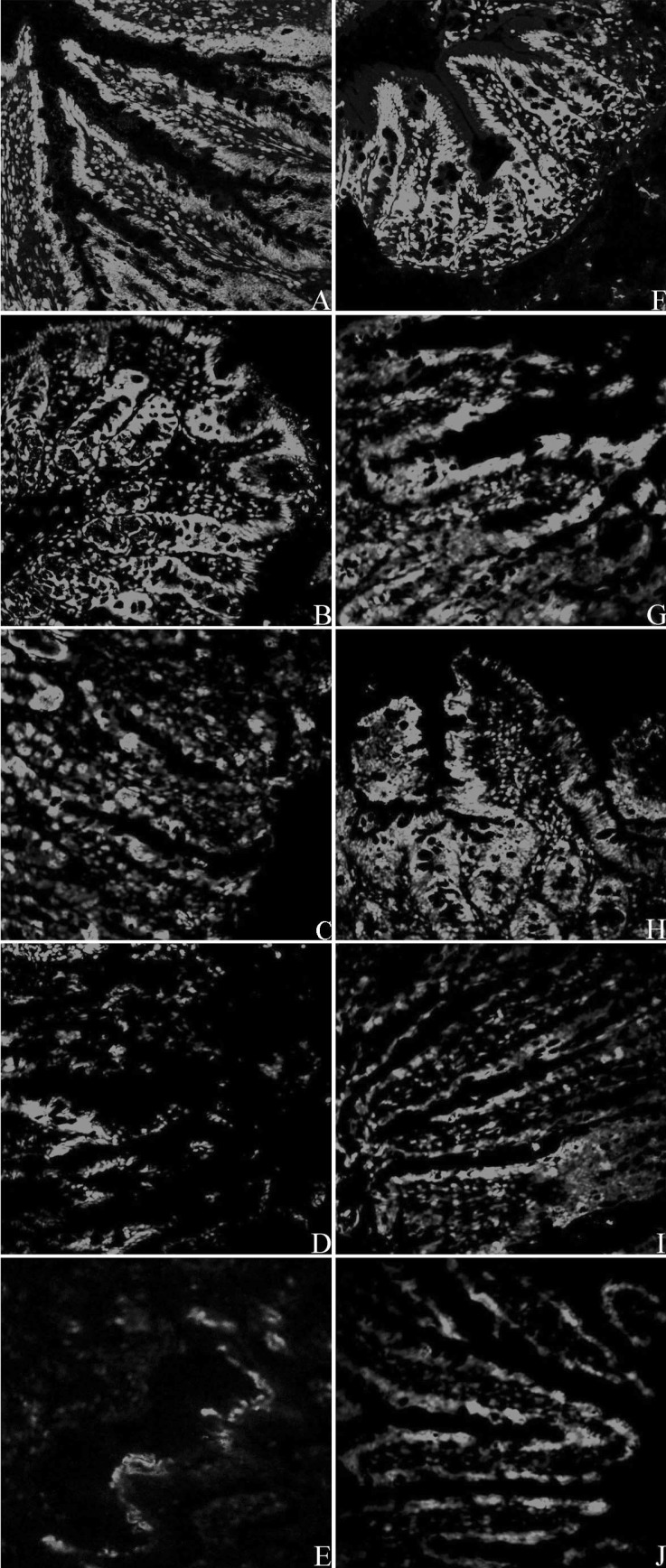
As shown in Fig. 3, the green fluorescence around the intestinal villus, which shows the presence of ZO-1 protein, can be seen clearly at 0 h after CLP in both NS group and Ber group, indicating normal expression of the tight junction component protein, ZO-1, in intestinal mucosa. The intensity of the green fluorescence weakened gradually, and was only barely visible in the NS group at 24 h after CLP. Although a decrease in the fluorescence intensity was also observed in the Ber group, they appear to be still brighter than the NS group at 6 h, 12 h and 24 h. These results were consistent with those from the Western blot.
Fig. 3.
Abundance and distribution of ZO-1 protein in the intestinal villi of rats after CLP. A: NS group, 0 h; B: NS group, 2 h; C: NS group, 6 h; D: NS group, 12 h; E: NS group, 24 h; F: Ber group, 0 h; G: Ber group, 2 h; H: Ber group, 6 h; I: Ber group, 12 h; J: Ber group, 24 h (400×).
Percentage of cell death in intestinal epithelial cells
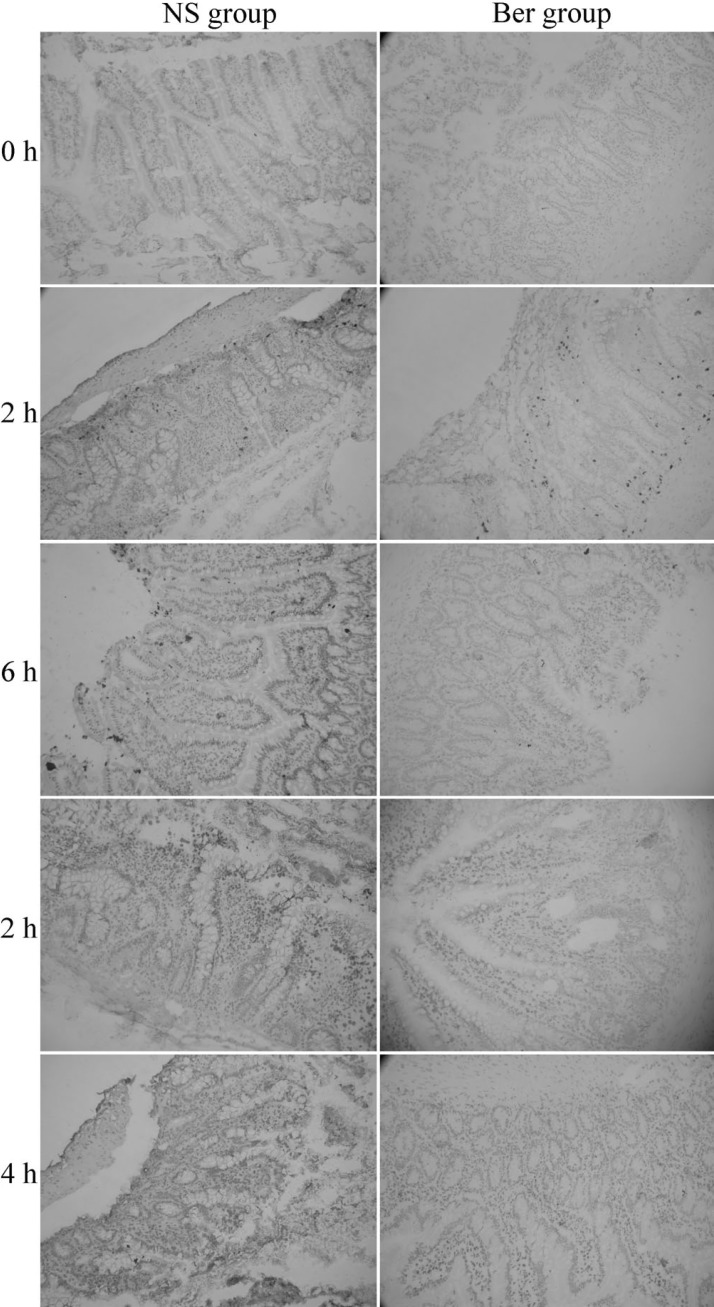
The percentage of cell death in Situ intestinal epithelial cells increased continuously after CLP in both NS group and Ber group (Fig. 4). Intestinal epithelial cell mortality was significantly lower in the Ber group than the NS group at 12 h and 24 h (p<0.01 for both, Table 1).
Fig. 4.
Comparison of cell death in Situ intestinal epithelial cells in NS group and Ber group. Intestinal epithelial cell death was significantly lower in the Ber group than the NS group and Ber group at 12 h and 24 h (200×).
Intestinal permeability
Intestinal permeability increased in a time-related manner in both Ber group and NS group. Statistical analysis showed that intestinal permeability in Ber group remained significantly lower than that in NS group from 2 h to 24 h (p<0.01, Table 1).
DISCUSSION
In the present study, we successfully replicate the model of septic shock in rats by cecal ligation and puncture, and demonstrated the hypothesis that pretreatment with Ber attenuates tissue injury and protects the intestinal mucosal barrier in early phase of sepsis through Toll-like receptors (TLRs) signaling pathway.
Intra-abdominal infections are one of the most common causes of the intestinal barrier dysfunction, which can lead to death of the patients, however, the underlying mechanisms remains unclear. Recent data have suggested an im portant role of the Toll-like receptors (TLRs) for the intestinal epithelial barrier and confers commensal tolerance [21], which suggests that TLRs signaling may involve the process.
TLRs are capable of recognizing and discriminating diverse pathogen-associated molecular patterns (PAMPs) [21]. TLR2, recognizes bacterial lipopeptides and lipoteichoic acid which are found abundantly in cell walls of Gram positive bacteria [26]. TLR2 may cooperate with TLR6 and TLR1, suggesting an essential mechanism for diversifying the repertoire of TLRs mediated responses [27]. TLR4 is the major receptor for lipopolysaccharide (LPS) activation [28] which may require the presence of accessory proteins, such as MD-2, CD14 and LPS binding protein. TLR9 may recognize the bacterial DNA which may enhance proinflammatory cytokine release [29].
In this present study, we observed that mRNA level of intestinal TLR2 and TLR4 increased after CLP in all rats, and significantly lower in the Ber group than the NS group at 6 h which indicate that berberine after CLP could suppress TLR2 and TLR4 expressions in rat intestine, which is consistent with the previous report [30]. In addition to TLR2 and TLR4, our investigation demonstrated that mRNA level of TLR9 in the Ber group remained higher than that in the NS group during the entire experimental period, especially at 6 h after CLP. Besides, we observed a reciprocal pattern of expression of TLR4 and TLR9, suggesting that reciprocal TLR4 and TLR9 signaling may occur in the pathogenesis of sepsis, which is consistent with a previous study [31]. These results above may imply: (1) TLR2, TLR 4, and TLR9 may be involved in the pathophysiology of sepsis following CLP; (2) the mechanisms of the protective effect of berberine for intestinal mucosal barrier may correlate with the decreased expression of TLR2 and TLR4 and increased TLR9 expression within the intestine.
Current studies suggest that NF-κB regulates the expressions of a wide variety of genes which are essential in innate immune responses, including those encoding cytokines such as TNF-α and IL-6 [32,33]. Persistent activation of NF-κB may cause excessive inflammatory cytokine responses, culminating in tissue injury, organ dysfunction, or even death [34]. The present study observed the nucleic NF-κB protein levels in the intestinal mucosa and found that it was slightly suppressed in the Ber group after CLP (data not shown), which means that berberine could attenuate NF-κB activation in rat intestine during CLP-induced polymicrobial sepsis. Further study is being conducted in our group.
In this present study, we found that CLP significantly increased the serum IL-6 and TNF-α concentration in all rats, and pretreatment with berberine alleviates the elevation of both TNF-α and IL-6 during CLP-induced sepsis which were similar to the recent findings [15,24]. Due to the mRNA levels of TLR2 and TLR4 and increased TLR9 after CLP were affected by pre-treatment with berberine, it is likely that berberine suppresses the inflammatory responses through the TLRs signaling pathway.
Tight junction proteins, including zonula occludens, occludin and claudins, are vital components of the intestinal epithelial paracellular barrier [35]. Research showed that intra-abdominal infection caused a marked decrease in the levels of tight junction proteins, resulting in loose interconnection of intestinal epithelial cells and increased intestinal permeability [36,37]. Our present study found that the level of tight junction (TJ) component protein ZO-1 was better maintained in the epithelial cells of the intestinal mucosa after berberine pretreatment. The expression of occludin and claudin-4 were also estimated and it appears that their levels decreased less after CLP with berberine pre-treatment (data not shown), which is consistent with the findings from literature [37]. Analysis on these tight junction proteins with more sensitive techniques may yield more detailed information. In addition to maintaining the expression of tight junction proteins, we also confirmed that berberine pre-treatment decreases cell death in the intestinal mucosa during sepsis. These effects likely contributed critically to the low intestinal permeability in the Ber group after CLP and we inferred that berberine protect the intestinal mucosal barrier by attenuating the decrease of tight junction protein and death of intestinal epithelial cell.
To our knowledge, most studies about the effects of berberine on intestinal mucosal barrier remain at vitro animal or cell experiment stage, there has been no report about its clinical relevance in humans by the mechanism we observed. Currently, Berberine is mainly used to treat gastrointestinal disorders and bacterial infection. Considering the high conservative Toll-like receptor signaling pathway, the regulatory effects of berberine on intestinal mucosal barrier may still function in humans. However, human immune system is far more complicated than the rat. Toll-like receptors are only one part of the initiation process. Whether the presence of other immune response networks stimulated in its downstream signaling cascade which leads to different outcomes between animal experiment and humans remains to be elucidated.
In China, gastrointestinal disorders and bacterial infection such as intestinal infection, bacterial dysentery is treated orally one to three times daily with berberine (100~300 mg). Intravenous administration is forbidden. Berberine is reported to responsible for the high incidence of gastrointestinal adverse events (34.5%) after oral administration (500 mg three times daily) to type 2 diabetic patients during the 13 weeks of berberine treatment [38]. These events included diarrhea (10.3%), constipation (6.9%), flatulence (19.0%) and abdominal pain (3.4%). After the dose of berberine was reduced to 300 mg three times daily, none of the patients were observed with pronounced (more than 50%) elevation in liver enzymes or creatinine. According to the report from 2010 NIH [39], for infectious diarrhea, 100 to 200 mg of berberine hydrochloride may be taken by mouth four times daily or as a single daily dose of 400 mg. Oral administration of berberine hydrochloride (500~1000 mg) is taken three times daily in a particular situation. Overall, further clinical trials are still needed to confirm the effective dose of berberine usage in humans.
In conclusion, pretreatment with berberine attenuates tissue injury and protects the intestinal mucosal barrier in early phase of sepsis and it is likely that the mechanisms of this preventive effect involve the TLRs signaling pathway.
ACKNOWLEDGEMENTS
The study was supported by the Chinese national natural science foundation grant #30901420 and 30972880.
ABBREVIATIONS
- TNF-α
Tumor necrosis factor α
- IL-6
Interleukin-6
- NF-κB
nuclear factor-kappa B
- CLP
cecal ligation and puncture
- NOD
Nucleotide oligomerization domain
References
- 1.Cribbs SK, Martin GS. Expanding the global epidemiology of sepsis. Crit Care Med. 2007;35:2646–2648. doi: 10.1097/01.CCM.0000288082.99980.90. [DOI] [PubMed] [Google Scholar]
- 2.Macdonald J, Galley HF, Webster NR. Oxidative stress and gene expression in sepsis. Br J Anaesth. 2003;90:221–232. doi: 10.1093/bja/aeg034. [DOI] [PubMed] [Google Scholar]
- 3.De-Souza DA, Greene LJ. Intestinal permeability and systemic infections in critically ill patients: effect of glutamine. Crit Care Med. 2005;33:1125–1135. doi: 10.1097/01.ccm.0000162680.52397.97. [DOI] [PubMed] [Google Scholar]
- 4.Doig CJ, Sutherland LR, Sandham JD, Fick GH, Verhoef M, Meddings JB. Increased intestinal permeability is associated with the development of multiple organ dysfunction syndrome in critically ill ICU patients. Am J Respir Crit Care Med. 1998;158:444–451. doi: 10.1164/ajrccm.158.2.9710092. [DOI] [PubMed] [Google Scholar]
- 5.Yu P, Martin CM. Increased gut permeability and bacterial translocation in Pseudomonas pneumonia-induced sepsis. Crit Care Med. 2000;28:2573–2577. doi: 10.1097/00003246-200007000-00065. [DOI] [PubMed] [Google Scholar]
- 6.Madara JL. Warner-Lambert/Parke-Davis Award lecture. Pathobiology of the intestinal epithelial barrier. Am J Pathol. 1990;137:1273–1281. [PMC free article] [PubMed] [Google Scholar]
- 7.Suzuki T. Regulation of intestinal epithelial permeability by tight junctions. Cell Mol Life Sci. 2013;70:631–659. doi: 10.1007/s00018-012-1070-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Turner JR. Intestinal mucosal barrier function in health and disease. Nat Rev Immunol. 2009;9:799–809. doi: 10.1038/nri2653. [DOI] [PubMed] [Google Scholar]
- 9.Cao M, Wang P, Sun C, He W, Wang F. Amelioration of IFN-γ and TNF-α-induced intestinal epithelial barrier dysfunction by berberine via suppression of MLCK-MLC phosphorylation signaling pathway. PLoS One. 2013;8:e61944. doi: 10.1371/journal.pone.0061944. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Kong DX, Li XJ, Tang GY, Zhang HY. How many traditional Chinese medicine components have been recognized by modern Western medicine? A chemoinformatic analysis and implications for finding multicomponent drugs. Chem Med Chem. 2008;3:233–236. doi: 10.1002/cmdc.200700291. [DOI] [PubMed] [Google Scholar]
- 11.Zeng XH, Zeng XJ, Li YY. Efficacy and safety of berberine for congestive heart failure secondary to ischemic or idiopathic dilated cardiomyopathy. Am J Cardiol. 2003;92:173–176. doi: 10.1016/s0002-9149(03)00533-2. [DOI] [PubMed] [Google Scholar]
- 12.Zhang Q, Piao XL, Piao XS, Lu T, Wang D, Kim SW. Preventive effect of Coptis chinensis and berberine on intestinal injury in rats challenged with lipopolysaccharides. Food Chem Toxicol. 2011;49:61–69. doi: 10.1016/j.fct.2010.09.032. [DOI] [PubMed] [Google Scholar]
- 13.Dvorák Z, Vrzal R, Maurel P, Ulrichová J. Differential effects of selected natural compounds with anti-inflammatory activity on the glucocorticoid receptor and NF-kappaB in HeLa cells. Chem Biol Interact. 2006;159:117–128. doi: 10.1016/j.cbi.2005.10.105. [DOI] [PubMed] [Google Scholar]
- 14.Hsiang CY, Wu SL, Cheng SE, Ho TY. Acetaldehyde-induced interleukin-1beta and tumor necrosis factor-alpha production is inhibited by berberine through nuclear factor-kappaB signaling pathway in HepG2 cells. J Biomed Sci. 2005;12:791–801. doi: 10.1007/s11373-005-9003-4. [DOI] [PubMed] [Google Scholar]
- 15.Jeong HW, Hsu KC, Lee JW, Ham M, Huh JY, Shin HJ, Kim WS, Kim JB. Berberine suppresses proinflammatory responses through AMPK activation in macrophages. Am J Physiol Endocrinol Metab. 2009;296:E955–E964. doi: 10.1152/ajpendo.90599.2008. [DOI] [PubMed] [Google Scholar]
- 16.Kuo CL, Chi CW, Liu TY. The anti-inflammatory potential of berberine in vitro and in vivo. Cancer Lett. 2004;203:127–137. doi: 10.1016/j.canlet.2003.09.002. [DOI] [PubMed] [Google Scholar]
- 17.Lee DU, Kang YJ, Park MK, Lee YS, Seo HG, Kim TS, Kim CH, Chang KC. Effects of 13-alkyl-substituted berberine alkaloids on the expression of COX-II, TNF-alpha, iNOS, and IL-12 production in LPS-stimulated macrophages. Life Sci. 2003;73:1401–1412. doi: 10.1016/s0024-3205(03)00435-1. [DOI] [PubMed] [Google Scholar]
- 18.Amasheh M, Fromm A, Krug SM, Amasheh S, Andres S, Zeitz M, Fromm M, Schulzke JD. TNFalpha-induced and berberineantagonized tight junction barrier impairment via tyrosine kinase, Akt and NFkappaB signaling. J Cell Sci. 2010;123:4145–4155. doi: 10.1242/jcs.070896. [DOI] [PubMed] [Google Scholar]
- 19.Cui HS, Hayasaka S, Zhang XY, Hayasaka Y, Chi ZL, Zheng LS. Effect of berberine on barrier function in a human retinal pigment epithelial cell line. Jpn J Ophthalmol. 2007;51:64–67. doi: 10.1007/s10384-006-0395-0. [DOI] [PubMed] [Google Scholar]
- 20.Ma X, Jiang Y, Wu A, Chen X, Pi R, Liu M, Liu Y. Berberine attenuates experimental autoimmune encephalomyelitis in C57 BL/6 mice. PLoS One. 2010;5:e13489. doi: 10.1371/journal.pone.0013489. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Cario E. Bacterial interactions with cells of the intestinal mucosa: Toll-like receptors and NOD2. Gut. 2005;54:1182–1193. doi: 10.1136/gut.2004.062794. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Williams DL, Ha T, Li C, Kalbfleisch JH, Ferguson DA., Jr Early activation of hepatic NFkappaB and NF-IL6 in polymicrobial sepsis correlates with bacteremia, cytokine expression, and mortality. Ann Surg. 1999;230:95–104. doi: 10.1097/00000658-199907000-00014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Otero-Antón E, González-Quintela A, López-Soto A, López-Ben S, Llovo J, Pérez LF. Cecal ligation and puncture as a model of sepsis in the rat: influence of the puncture size on mortality, bacteremia, endotoxemia and tumor necrosis factor alpha levels. Eur Surg Res. 2001;33:77–79. doi: 10.1159/000049698. [DOI] [PubMed] [Google Scholar]
- 24.Li GX, Wang XM, Jiang T, Gong JF, Niu LY, Li N. Berberine prevents damage to the intestinal mucosal barrier during early phase of sepsis in rat through mechanisms independent of the NOD-like receptors signaling pathway. Eur J Pharmacol. 2014;730:1–7. doi: 10.1016/j.ejphar.2014.02.006. [DOI] [PubMed] [Google Scholar]
- 25.Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods. 2001;25:402–408. doi: 10.1006/meth.2001.1262. [DOI] [PubMed] [Google Scholar]
- 26.Takeuchi O, Hoshino K, Kawai T, Sanjo H, Takada H, Ogawa T, Takeda K, Akira S. Differential roles of TLR2 and TLR4 in recognition of gram-negative and gram-positive bacterial cell wall components. Immunity. 1999;11:443–451. doi: 10.1016/s1074-7613(00)80119-3. [DOI] [PubMed] [Google Scholar]
- 27.Ozinsky A, Underhill DM, Fontenot JD, Hajjar AM, Smith KD, Wilson CB, Schroeder L, Aderem A. The repertoire for pattern recognition of pathogens by the innate immune system is defined by cooperation between toll-like receptors. Proc Natl Acad Sci U S A. 2000;97:13766–13771. doi: 10.1073/pnas.250476497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Poltorak A, He X, Smirnova I, Liu MY, Van Huffel C, Du X, Birdwell D, Alejos E, Silva M, Galanos C, Freudenberg M, Ricciardi-Castagnoli P, Layton B, Beutler B. Defective LPS signaling in C3H/HeJ and C57BL/10ScCr mice: mutations in Tlr4 gene. Science. 1998;282:2085–2088. doi: 10.1126/science.282.5396.2085. [DOI] [PubMed] [Google Scholar]
- 29.Ivory CP, Prystajecky M, Jobin C, Chadee K. Toll-like receptor 9-dependent macrophage activation by Entamoeba histolytica DNA. Infect Immun. 2008;76:289–297. doi: 10.1128/IAI.01217-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Shang L, Fukata M, Thirunarayanan N, Martin AP, Arnaboldi P, Maussang D, Berin C, Unkeless JC, Mayer L, Abreu MT, Lira SA. Toll-like receptor signaling in small intestinal epithelium promotes B-cell recruitment and IgA production in lamina propria. Gastroenterology. 2008;135:529–538. doi: 10.1053/j.gastro.2008.04.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Gribar SC, Sodhi CP, Richardson WM, Anand RJ, Gittes GK, Branca MF, Jakub A, Shi XH, Shah S, Ozolek JA, Hackam DJ. Reciprocal expression and signaling of TLR4 and TLR9 in the pathogenesis and treatment of necrotizing enterocolitis. J Immunol. 2009;182:636–646. doi: 10.4049/jimmunol.182.1.636. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Ghosh S, May MJ, Kopp EB. NF-kappa B and Rel proteins: evolutionarily conserved mediators of immune responses. Annu Rev Immunol. 1998;16:225–260. doi: 10.1146/annurev.immunol.16.1.225. [DOI] [PubMed] [Google Scholar]
- 33.Wu CF, Bi XL, Yang JY, Zhan JY, Dong YX, Wang JH, Wang JM, Zhang R, Li X. Differential effects of ginsenosides on NO and TNF-alpha production by LPS-activated N9 microglia. Int Immunopharmacol. 2007;7:313–320. doi: 10.1016/j.intimp.2006.04.021. [DOI] [PubMed] [Google Scholar]
- 34.Yu M, Shao D, Feng X, Duan M, Xu J. Effects of ketamine on pulmonary TLR4 expression and NF-kappa-B activation during endotoxemia in rats. Methods Find Exp Clin Pharmacol. 2007;29:395–399. doi: 10.1358/mf.2007.29.6.1119165. [DOI] [PubMed] [Google Scholar]
- 35.Liu H, Li M, Wang P, Wang F. Blockade of hypoxia-inducible factor-1α by YC-1 attenuates interferon-γ and tumor necrosis factor-α-induced intestinal epithelial barrier dysfunction. Cytokine. 2011;56:581–588. doi: 10.1016/j.cyto.2011.08.023. [DOI] [PubMed] [Google Scholar]
- 36.Costantini TW, Loomis WH, Putnam JG, Drusinsky D, Deree J, Choi S, Wolf P, Baird A, Eliceiri B, Bansal V, Coimbra R. Burn-induced gut barrier injury is attenuated by phosphodiesterase inhibition: effects on tight junction structural proteins. Shock. 2009;31:416–422. doi: 10.1097/SHK.0b013e3181863080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Costantini TW, Deree J, Loomis W, Putnam JG, Choi S, Baird A, Eliceiri BP, Bansal V, Coimbra R. Phosphodiesterase inhibition attenuates alterations to the tight junction proteins occludin and ZO-1 in immunostimulated Caco-2 intestinal monolayers. Life Sci. 2009;84:18–22. doi: 10.1016/j.lfs.2008.10.007. [DOI] [PubMed] [Google Scholar]
- 38.Yin J, Xing H, Ye J. Efficacy of berberine in patients with type 2 diabetes mellitus. Metabolism. 2008;57:712–717. doi: 10.1016/j.metabol.2008.01.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.National Toxicology Program. Toxicology and carcinogenesis studies of goldenseal root powder (Hydrastis Canadensis) in F344/N rats and B6C3F1 mice (feed studies) Natl Toxicol Program Tech Rep Ser. 2010;(562):1–188. [PubMed] [Google Scholar]