Abstract
Neutrophils play important roles in many inflammatory diseases. The migration of neutrophils to the inflammatory site is tightly regulated by specific chemokines, of which interleukin-8 (IL-8) and leukotriene B4 (LTB4) constitute key mediators by binding to the surface receptors CXCR1/2 and BLT1, respectively. Oligonucleotides (ODN) containing CpG motifs mediate potent immunomodulatory effects through binding to Toll-like receptor 9. So far, knowledge on how ODN can affect neutrophil migration during inflammation is lacking. This study demonstrates that several novel CpG ODN significantly down-regulate the surface expression of CXCR1/2 and BLT1. In addition, the ODN significantly blocked IL-8-induced and LTB4-induced neutrophil migration in vitro, as well as leucocyte migration in vivo demonstrated in mice by intravital microscopy and in a model of airway inflammation. The down-regulation of CXCR1 is rapid, occurring 15 min after ODN stimulation, and can be mediated through an endosomally independent mechanism. Inhibition of the IL-8 and LTB4 pathways may provide new opportunities of therapeutic intervention using ODN to reduce neutrophil infiltration during inflammation.
Keywords: chemotaxis, inflammation, neutrophils, oligonucleotide
Introduction
Toll-like receptor 9 (TLR9) detects unmethylated CpG DNA, which are common in the genomes of bacteria and DNA viruses, but under-represented and methylated in the vertebrate genome. CpG DNA are taken up into the cell by receptor-mediated endocytosis and interact with TLR9 in endosomes.1 The natural ligand for TLR9, bacterial/viral CpG DNA, can be mimicked through single-stranded oligonucleotides (ODN) containing one or more unmethylated CpG motifs (CpG ODN) that can induce an immune response without the actual infection. These immunomodulatory properties of CpG ODN make them interesting tools to use in immunotherapy, and they have been evaluated with promising outcomes as therapy of various diseases such as cancer, allergy, ulcerative colitis and autoimmunity.2,3 The mechanism by which TLR9 interacts with nucleic acid and becomes activated in live cells is not well understood. However, it is known that depending on the sequence and length, as well as on the number and positions of CpG motifs in an ODN, distinct immunomodulatory profiles can be observed. For example, ODN containing Poly-G sequences are known to induce high levels of interferon-α.4
Neutrophils play a critical role in the innate immune response to bacterial infections by mediating the phagocytosis and destruction of microorganisms, as well as producing cytokines, chemokines and growth factors that shape the immune response.
Previous studies have shown that bacterial DNA can induce activation of neutrophils through a non-CpG and TLR9-independent mechanism that does not require DNA internalization, in addition to having TLR9-dependent effects.5 Bacterial DNA has been shown to induce cytokine secretion, delay apoptosis and increase the phagocytic properties of neutrophils.6,7
During inflammation there is excessive neutrophil recruitment, in which neutrophil-derived proteinases and reactive oxygen species are capable of inflicting damage to surrounding tissues.8 Indeed, neutrophil-mediated tissue damage underlies and contributes to the pathology of a range of inflammatory diseases.8 Interleukin-8 (IL-8) has a key role in the recruitment and activation of neutrophils during inflammation. It reacts with neutrophils via two distinct types of receptor, CXC chemokine receptor 1 (CXCR1) and CXCR2.9 CXCR1 is specific for IL-8, while CXCR2 also reacts with other chemokines.9 Increasing evidence indicates that CXCR1 could have a dominant role in mediating IL-8 chemotaxis of polymorphonuclear neutrophils (PMN) that express both receptors.10 The IL-8/CXCR1/2 pathway has been shown to have a key role in inflammatory diseases such as ulcerative colitis, ischaemia/reperfusion injury and chronic obstructive pulmonary disease, as well as in tumour progression.11–14 For this reason, the IL-8/CXCR1/2 pathway constitutes a major target for anti-inflammatory therapy through identification of CXCR1/2 antagonists, as well as IL-8 blocking antibodies.15,16
Another potent chemoattractant is leukotriene B4 (LTB4), which binds with high affinity to the receptor BLT1, which is abundantly expressed on the surface of neutrophils.17 The LTB4/BLT1 pathway is important for the recruitment of inflammatory cells into the sites of inflammation in various diseases such as asthma and arthritis.17,18 It has been indicated to have a specifically important role in severe persistent asthma, since LTB4 production in general is resistant to corticosteroid treatment.17 In addition, it has been shown that corticosteroids can up-regulate BLT1 expression on corticosteroid-resistant inflammatory cells, and that the LTB4/BLT1 pathway may in this way be an important pathway for the pathogenesis of inflammatory allergic diseases that do not respond to corticosteroids.17,19
The aim of this study was to investigate the effect of a novel set of ODN with distinct lengths and sequences on the PMN surface expression of CXCR1, CXCR2 and BLT1 as well as their influence on IL-8-induced or LTB4-induced chemotaxis. Inhibiting the IL-8/CXCR1/CXCR2 as well as LTB4/BLT1 pathways may be a significant step in reducing neutrophil infiltration in numerous inflammatory disorders.
Materials and methods
Oligonucleotides
Oligonucleotides were synthesized by Biomers.net (Ulm, Germany). The ODN were adjusted with PBS (Invitrogen, Carlsbad, CA) pH 7·4 to reach a stock concentration of 500 μm and stored at −20° until used. The ODN used in this study are listed in Table1.
Table 1.
Oligonucleotides included in the study
| Oligonucleotide ID1 | Properties | Mer |
|---|---|---|
| DX9022 | CpG oligonucleotide, without poly-G flanks | 22 |
| IDX9031 | CpG oligonucleotide, without poly-G flanks | 21 |
| IDX9052 | CpG oligonucleotide, with poly-G flanks | 13 |
| IDX9054 | CpG oligonucleotide, with poly-G flanks | 14 |
| IDX9059 | CpG oligonucleotide, with poly-G flank | 15 |
| IDX9096 | CpG oligonucleotide, without poly-G flanks | 18 |
The identification (ID), properties and the nucleotide length (mer) of the oligonucleotides included in the study are given.
Cell preparation and in vitro stimulation with ODN
Whole blood from healthy blood donors was used for preparation of PMN. The study was approved by the local ethics committee. PMN were isolated by density centrifugation using Polymorphprep™ (Axis-Schield, Oslo, Norway) according to the manufacturer's instruction. Red blood cells were lysed using ACK lysing buffer (Bioconcept, Allschwill, Switzerland). The cells were then further washed in PBS, and the viability and the cell number were determined by counting the cells in Trypan blue (Sigma Aldrich, Stockholm, Sweden). The isolation resulted in a purity of ≥ 88% PMN as analysed by FACS. Thereafter, the cells were re-suspended in complete cell medium consisting of RPMI-1640 (Sigma Aldrich) supplemented with 10% heat inactivated fetal calf serum (Invitrogen), 2 mm l-glutamine, 100 U/ml penicillin, 100 μg/ml streptomycin, 10 mm HEPES (Sigma Aldrich) and 5 μg/ml gentamicin (Invitrogen). The PMN were cultured in 96-well culture tissue plates [Becton Dickinson (BD), Franklin Lakes, NJ] at a concentration of 2 × 106 cells/ml with 0·5, 10 or 25 μm ODN or with medium alone as a negative control in a total volume of 200 μl/well. The cells were incubated for the indicated times at 37° in a humidified cell culture incubator (Thermo Scientific, Waltham, MA) with 5% CO2 in air, after which the cells were stained with fluorochrome-labelled antibodies (BD) against CXCR1, CXCR2 and BLT1 and the expression of the receptors was analysed using flow cytometry. In some experiments, the supernatants were collected and used for cytokine analysis using cytometric bead array, see below. In a number of experiments PMN were pre-incubated with chloroquine (Sigma Aldrich) or a monoclonal antibody against Mac-1 (BD) for 30 min before stimulation with ODN as described below.
Flow cytometry
Cells incubated with ODN were harvested, washed in PBS and resuspended in PBS supplemented with 2% fetal calf serum. The cells were stained for the granulocyte marker CD66b together with CXCR1, CXCR2 or BLT1 using fluorochrome-labelled mouse monoclonal antibodies (Becton Dickinson, San Jose, CA) for 30 min at 4°. The antibodies used were compared with isotype-matched controls (BD). After washing in PBS, the cells were analysed by a FACSarray flow cytometer (BD) and the data were analysed using the FACSarray software system (BD). A minimum of 15 000 CD66b+ gated PMN were analysed per sample.
Kinetic evaluation of the effect of ODN on CXCR1 surface expression
Human PMN from healthy blood donors were stimulated with 10 μm ODN or with medium alone for various time-points (15 min, 30 min, 1 hr, 2 hr and 3 hr). Cells were subsequently harvested and fixed at each time-point in 2% paraformaldehyde, after which they were analysed for CXCR1 expression by flow cytometry as described above.
Cytokine analysis
Culture supernatants from peripheral blood mononuclear cells stimulated with ODN were analysed for the presence of the cytokines IL-8 and tumour necrosis factor-α (TNF-α) using a cytometric bead array flex kit (BD) according to the manufacturer's instructions on a FACSArray flow cytometer (BD). The data were analysed using FCAP Array software (BD).
Chloroquine assay
Polymorphonuclear neutrophils isolated from healthy blood donors were pre-treated with 0·5, 5, 10, 50 and 500 μg/ml chloroquine (Sigma Aldrich) for 30 min at 37° before being stimulated with 10 μm ODN for an additional 3 hr. Surface expression of CXCR1 was then analysed by flow cytometry as described above.
Blocking Mac-1 complex
Polymorphonuclear neutrophils isolated from healthy blood donors were pre-treated with 10 μg/ml anti-Mac-1 monoclonal antibody (BD) for 30 min at 37° before being stimulated with 10 μm ODN for an additional 3 hr. Surface expression of CXCR1 was then analysed by flow cytometry as described above.
Chemotaxis assay
Chemotaxis of PMN was investigated using the QCM™ 3-μm 24-well colorimetric chemotaxis assay (Millipore, Temecula, CA) according to the manufacturer's instructions. Briefly, PMN were prepared from whole blood from healthy donors and resuspended in complete RPMI medium as described above. PMN were pre-incubated in 48-well plates (BD) at a concentration of 1 × 106 cells/ml, using 250 μl cell suspension per well, with 0·5, 10 or 25 μm ODN for 1 hr at 37° in a humidified cell culture incubator (Thermo Scientific) with 5% CO2. The cells were then washed in medium and transferred to the top inserts of a 24-well cell migration plate assembly with a 3-μm pore size (Millipore). To the lower chamber, complete cell medium containing 100 ng/ml recombinant human IL-8 (Invitrogen) or 500 nm LTB4 were added. In the IL-8-induced chemotaxis assay, 10 μg/ml of an anti-IL-8 monoclonal antibody (BD) was included in the experiment as a control for the specificity of the migration. The cells were then allowed to migrate through the filter for 3 hr at 37° in a humidified cell culture incubator (Thermo Scientific) with 5% CO2. Thereafter, the cells from the lower chamber, i.e. migrated cells, were detected by incubation with the cell viability stain WST-1 for 1 hr followed by quantification by measuring the absorbance at 450 nm using a microplate reader (Tecan, Männedorf, Switzerland).
Intravital microscopy of leucocyte recruitment
C57BL/6 specific pathogen-free mice from Scanbur AB, Sollentuna, Sweden, were kept in an animal room with controlled temperature (21 ± 2°), light–dark cycles of 12 hr each, and were allowed free access to food and water. All experiments were approved by the Stockholm Ethical Committee for Animal Experimentation, Sweden.
The animals were given IDX9059 subcutaneously, 50 μg/100 μl/mouse 20 min before induction of inflammation. The mice were anaesthetized by an intraperitoneal injection of 0·15–0·20 ml of a mixture of ketamine (Ketalar®; Pfizer AB, Sollentuna, Sweden; 25 mg/ml) and xylazine (Narcoxyl vet.®; Intervet International B.V., Boxmeer, the Netherlands; 5 mg/ml). The left jugular vein was cannulated with polyethylene tubing (PE10) for continuous administration of anaesthesia. A ventral incision was made on the right scrotum and one testis was withdrawn. The cremaster muscle was dissected free of fascia, incised and pinned out flat on a transparent pedestal to allow trans-illumination. The testis was then pinned to the side. The preparation was kept moist and warm by continuous superfusion of a 37° temperature-controlled bicarbonate buffer, maintaining physiological levels of temperature, pH and gas tensions. Leucocyte extravasation was induced by addition of platelet activating factor (PAF; Sigma-Aldrich, St Louis, MO; 100 nm) to the superfusion solution for 60 min. Measurements of rolling, adhesion and transmigration were made before and after stimulation. Video recordings were obtained from 20–50-μm-wide well-defined venules. Rolling was determined as the number of leucocytes passing a reference line perpendicular to blood flow during 30 seconds. Cells within the vessel were classified as adherent if they remained stationary for more than 30 seconds. Transmigrated cells were counted in the extravascular tissue within a distance of 70 μm from the studied vessel. All microscopic observations were made using a Leitz Orthoplan intravital microscope with a Leitz SW25 water immersion objective (Leitz GMBH, Wetzlar, Germany). Images were televised and recorded using Panasonic WV-1550/G (Panasonic, Osaka, Japan) video camera.
Ovalbumin-induced airway inflammation
Female BALB/c mice (8 weeks), obtained from B&K Sollentuna, Stockholm, Sweden, were used in the experiment. All experiments were approved by the Regional Animal Research Ethics Committee in Umeå. Mice were sensitized intraperitoneally with 200 μl of 50 μg/ml ovalbumin (OVA; chicken egg albumin grade V, Sigma)/aluminium hydroxide gel on day 0 and 12. On days 23, 26, 30 and 33, mice were challenged in the lungs by inhalation of aerosolized OVA for 30 min using a Batelle exposure chamber. Aerosols were generated by a compressed-air nebulizer (Collison six-jet) at airflow 7·4 l/min using a nebulizer concentration of 10 mg/ml OVA dissolved in PBS (Sigma). The control group with non-sensitized animals received only aerosolized OVA at days 23, 26, 30 and 33. There was also a control group of sensitized mice that did not receive aerosol challenge. IDX9059 (1247 μg/μl) was administered by intranasal instillation on days 16 and 21 in 40 μl PBS giving a dose of ∼ 50 μg/mice (49·88 μg/mice). The two sensitized sham-treatment groups were instilled with PBS in the same total volume as for the treatment groups at days 16 and 21. Mice were killed by cervical dislocation 42 hr after the last OVA aerosol challenge. The trachea was cannulated with polyethylene tubing (BD, Sparks, MD) and bronchoalveolar lavage was performed using 4 × 1 ml aliquots of ice-cold Hanks’ balanced salt solution (Sigma). The bronchoalveolar lavage fluid was centrifuged (400 g, 10 min, 4°), after which the cells were resuspended in 0·4 ml PBS and the total number of leucocytes was counted using Trypan blue exclusion in a Bürker chamber. Duplicate Cytospin (Cytospin 3, Shandon, Runcorn, UK) preparations of bronchoalveolar lavage fluid cells were stained with May–Grünewald–Giemsa stain for differential counts, using standard morphological criteria.
Results
Polymorphonuclear neutrophils are one of the major leucocytes that are attracted to inflamed tissues. The binding of IL-8 and LTB4 to their individual receptors CXCR1/CXCR2, and BLT1, respectively, plays a key role in the recruitment of neutrophils to the inflammation site.20,21 CpG containing ODN have been shown to affect innate and adaptive immune responses including antigen presentation, expression of co-stimulatory molecules and induction of cytokines,1 which makes them interesting therapeutic agents for use in different disease contexts. In this study, we wanted to investigate the effect of a novel set of ODN on the surface expression of CXCR1, CXCR2 and BLT1 on human PMN, as well as their effect on PMN migration.
ODN reduce surface expression of CXCR1 and CXCR2 on human PMN
Six ODN with different structural properties (Table) were investigated for their effect on CXCR1 and CXCR2 surface expression using PMN from healthy blood donors. PMN were stimulated with ODN for 3 hr after which they were analysed by flow cytometry. Four of the six ODN tested (IDX9022, IDX9052, IDX9054 and IDX9059) induced a significant dose-dependent reduction in CXCR1 surface expression (Fig.1a, b), whereas two of the ODN tested (IDX9031 and IDX9096) were unable to decrease CXCR1 surface expression to a significant degree (Fig.1a, b). The most potent ODN in reducing CXCR1 surface expression were IDX9054 and IDX9052, which at 25 μm reduced the surface expression of CXCR1 by as much as 62% (Fig.1b). Surface expression of CXCR2 was also down-regulated in response to ODN, especially in response to IDX9052 and IDX9054. Stimulating PMN with IDX9052 and IDX9054 decreased CXCR2 surface expression by 60% and 44%, respectively, when used at 25 μm (Fig.1c).
Figure 1.
Oligonucleotides (ODN) reduce surface expression of CXCR1 and CXCR2 on human polymorphonuclear neutrophils (PMN). PMN from healthy blood donors were stimulated with 0·5, 10 μm or 25 μm of ODN or with medium alone (untreated) for 3 hr. Cells were subsequently harvested and analysed for CXCR1 (a and b) or CXCR2 (c) surface expression by flow cytometry. (a) FACS histograms show the expression of CXCR1 of PMN incubated with 25 μmIDX9054 or IDX9031 compared with medium alone. The fold changes in mean fluorescence intensity (MFI) of CXCR1+ (b) or CXCR2+ (c) CD66b+ PMN were calculated by normalizing the MFI of corresponding untreated cells to 1 (dotted line). Results are presented as mean values ± SEM (n = 5–9). *P < 0·05, **P < 0·01 and ***P < 0·001 by one-way analysis of variance with Dunnett's post hoc correction versus untreated cells.
ODN stimulation rapidly reduces the surface expression of CXCR1
To investigate the kinetics for CXCR1 down-regulation, human PMN were stimulated with 10 μm of IDX9059 for 15 min, 30 min, 1 hr, 2 hr and 3 hr after which CXCR1 expression was investigated with flow cytometry. Already after 15 min, a small decrease in CXCR1 surface expression could be observed, and after 30 min there was a significant down-regulation of CXCR1 that reached maximum level at around 3 hr (Fig.2a). The expression of CXCR1 was also investigated after 20 hr of stimulation with IDX9059, but no significant difference in the levels of CXCR1 was found compared with 3 hr of stimulation (Fig.2b). No difference in cell viability, analysed by trypan blue staining, was evident between 3 and 20 hr incubation, with a cell viability always above 95%.
Figure 2.
The reduction in CXCR1 surface expression starts already after 15 min of oligonucleotide (ODN) stimulation. (a) Polymorphonuclear neutrophils (PMN) from healthy blood donors were stimulated with 10 μm of the ODN IDX9059 or with medium alone (untreated) for 15 min, 30 min, 1 hr, 2 hr or 3 hr. Cells were subsequently harvested and fixed at each time-point and analysed for CXCR1 surface expression by flow cytometry. The fold changes in mean fluorescence intensity (MFI) of CXCR1+ CD66b+ PMN was calculated by normalizing the MFI of corresponding untreated cells to 1. (b) PMN from healthy blood donors were stimulated with 10 μm of the ODN IDX9059 for 3 or 20 hr, after which the expression of CXCR1 was analysed with flow cytometry. Results are presented as mean values ± SEM (n = 4). ***P < 0·001 by two-way analysis of variance with Bonferroni's post hoc correction versus untreated cells.
ODN down-regulate CXCR1 expression through endosomal TLR9-dependent and -independent pathways
To investigate the role of endosomal TLR9 in the reduction of surface CXCR1 expression induced by ODN, PMN were pre-treated with chloroquine before being stimulated with ODN. Chloroquine is a 4-aminoquinoline drug, which blocks endosomal fusion and acidification and prevents endosomal TLR9 activation and downstream metabolic signalling pathways. Chloroquine could dose-dependently inhibit the reduction in CXCR1 surface expression induced by IDX9059 and IDX9022 (Fig.3a); 10 μg/ml of Chloroquine almost completely blocked the decrease in CXCR1 surface expression induced by these two ODN (Fig.3a). Surprisingly, Chloroquine could not block the reduction in CXCR1 surface expression induced by either IDX9054 or IDX9052 (Fig.3a). Doses of 50 and 500 μg/ml of Chloroquine were also tested, but these concentrations of Chloroquine negatively affected the cell viability. These results indicate that some ODN, such as IDX9059 and IDX9022, mediate their CXCR1-reducing effect through an endosomal TLR9-dependent mechanism, while other ODN can down-regulate CXCR1 independently of endosomal TLR9, suggesting the involvement of an alternative mechanism.
Figure 3.
The decrease in CXCR1 surface expression can be mediated through both Toll-like receptor 9 (TLR9) -dependent and -independent mechanisms and do not involve MAC-1, interleukin-8 (IL-8) or tumour necrosis factor-α (TNF-α). Polymorphonuclear neutrophils (PMN) from healthy blood donors were pre-incubated for 30 min with 0·5, 5 or 10 μg/ml of Chloroquine (a) (n = 4) or with a monoclonal anti-MAC1 antibody (b) (n = 3) before being stimulated with 10 μm of oligonucleotides (ODN) or with medium alone (untreated) for 3 hr. Cells were subsequently harvested and analysed for CXCR1 surface expression by flow cytometry. The fold changes in mean fluorescence intensity (MFI) of CXCR1+ CD66b+ PMN were calculated by normalizing the MFI of corresponding untreated cells to 1 (dotted line). (c) PMN from healthy blood donors were stimulated with 10 μm of the ODN IDX9059 or with medium alone (untreated) for 20 hr after which the supernatants were collected and analysed for the presence of IL-8 using cytometric bead array (n = 4). Results are presented as mean values ±SEM. *P < 0·05 by one-way analysis of variance with Dunnett's post hoc correction versus untreated cells.
Reduction in CXCR1 surface expression is independent of Mac-1, IL-8 and TNF-α
It has previously been shown that bacterial DNA can bind to the Mac-1 (CD11b/CD18) complex on the neutrophil cell surface and be internalized into the cells.22 To determine whether Mac-1-mediated internalization of the ODN is required for the reduction of CXCR1 surface expression, PMN from healthy blood donors were pre-treated with a monoclonal antibody against the Mac-1 complex before being stimulated with ODN. No difference in the down-regulation of CXCR1 surface expression was seen with or without blocking the Mac-1 complex, indicating that this surface receptor is not involved in down-regulation of CXCR1 surface expression (Fig.3b).
It is further known that IL-8 can induce internalization of its own receptors, CXCR1 and CXCR2, by an autocrine mechanism, and that the receptors are later re-expressed at the cell surface within a few hours.23 Down-modulation of CXCR1 and CXCR2 has also been demonstrated in response to TNF-α.24 To exclude the possibility that the down-modulation of CXCR1 and CXCR2 by ODN is due to an increased production of IL-8 or TNF-α, supernatants collected from PMN 20 hr after stimulation with ODN were analysed for the presence of IL-8 and TNF-α. No difference in the levels of IL-8 could be seen in supernatants from untreated and ODN-treated PMN (Fig.3c) and the levels of TNF-α was below the detection limit in all samples (data not shown), indicating that the down-regulation of CXCR1 and CXCR2 by ODN is not the result of induction of either IL-8 or TNF-α.
ODN reduce expression of the BLT1 receptor on the surface of PMN
Leukotriene B4 is another important PMN chemoattractant, which mediates its effect by binding to the receptor BLT1 on the PMN cell surface.17 To investigate if the ODN, in addition to reducing CXCR1 and CXCR2 surface expression, could decrease surface expression of BLT1, PMN from healthy blood donors were stimulated with ODN for 3 hr after which the surface expression of BLT1 was analysed with flow cytometry. A dose-dependent reduction in BLT1 surface expression was seen 3 hr after stimulation with ODN, with the most potent ODN being IDX9052 and IDX9054, which decreased the surface expression of BLT1 by as much as 78% and 80%, respectively (Fig.4).
Figure 4.
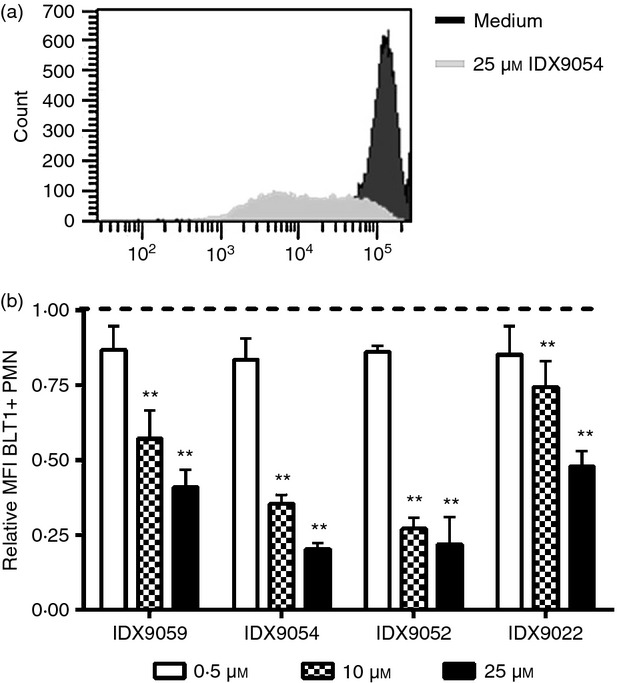
Oligonucleotides (ODN) decrease surface expression of the leukotriene B4 (LTB4) receptor BLT1. Human polymorphonuclear neutrophils (PMN) from healthy blood donors were stimulated with 0·5, 10 or 25 μm of ODN or with medium alone (untreated) for 3 hr. Cells were subsequently harvested and analysed for BLT1 surface expression by flow cytometry. (a) FACS histograms show the expression of BLT1 of PMN incubated with 25 μmIDX9054 compared with medium alone. The fold change in mean fluorescence intensity (MFI) of BLT1+ CD66b-positive PMN was calculated by normalizing the MFI of corresponding untreated cells to 1 (dotted line). Results are presented as mean ± SEM (n = 5). **P < 0·01 by one-way analysis of variance with Dunnett's post hoc correction versus untreated cells.
ODN inhibit IL-8-induced and LTB4-induced chemotaxis
To investigate if the reduction in CXCR1 and CXCR2 surface expression induced by our CpG ODN also resulted in decreased IL-8-mediated migration of PMN in vitro, PMN from healthy blood donors were pre-incubated for 1 hr with CpG ODN. The cells were then allowed to migrate towards IL-8 in a chemotaxis assay for 3 hr. A dose-dependent reduction in the number of migrated PMN was seen for IDX9022, IDX9052, IDX9054 and IDX9059, with the most efficient compound being IDX9052, which at a concentration of 10 μm totally blocked the migration of PMN to a level below the limit of detection (Fig.5a). IDX9031 induced a smaller decrease in PMN migration that was not dose-dependent (Fig.5a), which is in agreement with the observation that IDX9031 was less efficient in down-regulating CXCR1 and CXCR2 (Fig.). Repeated experiments significantly showed that IDX9052, IDX9054 and IDX9059 were potent inhibitors of IL-8-induced PMN migration and that the migration was IL-8 specific, as blocking IL-8 with an anti-IL-8 monoclonal antibody significantly inhibited the PMN chemotaxis (Fig.5b). In addition, inhibition (P < 0·01) of LTB4 induced PMN chemotaxis by ODN could also be demonstrated (P < 0·01), with the most potent ODN again being IDX9052, IDX9054 and IDX9059 (Fig.5c).
Figure 5.
Oligonucleotides (ODN) inhibit interleuin-8 (IL-8) and leukotriene B4 (LTB4) -induced chemotaxis. (a) Human polymorphonuclear neutrophils (PMN) from healthy blood donors were pre-incubated with 0·5, 10 or 25 μm of ODN or with medium alone [untreated (untr)] for 1 hr after which the cells were investigated for their ability to migrate towards IL-8 in the presence of free ODN (n = 3) (a) or free oligonucleotides were washed away and the cells were investigated for their ability to migrate towards IL-8 (n = 5) (b) or LTB4 (n = 4–6) (c) in a chemotaxis assay for 3 hr. As a control for specificity, a monoclonal antibody against IL-8 (α-IL-8) was included in the assay (b). Results are presented as the mean number of migrated PMN ± SEM. *P < 0·05 and **P < 0·01 by one-way analysis of variance with Dunnett's post hoc correction versus untreated cells. (d,e) The mean fluorescence intensity (MFI) of CXCR1 (d), CXCR2 (e) and BLT1 (f) surface expression on PMN after stimulation with ODN were plotted against the number of PMN that migrated towards IL-8 (d and e) or LTB4 (f) in the chemotaxis assay. The curve fit (r2) is specified in the figures. ***P < 0·001 by linear regression analysis.
The surface expression of CXCR1 and CXCR2 correlated with the number of migrated PMN in the IL-8-induced chemotaxis assay (Fig.5d, e), as did the surface expression of BLT1 and the number of migrated PMN in the LTB4-induced chemotaxis assay (Fig.5f).
ODN inhibit leucocyte migration in vivo
By using intravital microscopy on the cremaster muscle of mice we investigated the ex vivo effect of ODN on the migration of leucocytes before and after addition of the chemotactic substance PAF. Rolling, sticking and transmigration were studied using time-lapse video microscopy and the recordings were later used for quantification of cells.
Treatment with IDX9059 significantly inhibited both the rolling and the adherence of leucocytes to the endothelial wall in the absence of an inflammatory stimulus (Fig.6a). After application of the chemotactic PAF, there was as expected a high increase in trans-migrating cells from a basal value of 1·7 cells/field to 23·7 cells/field (Fig.6). In agreement with this, there was a reduction of rolling cells from 22·3 cells/field to 8·3 cells/field, and adhering cells remained at about 15 cells/field. In the presence of PAF, the addition of IDX9059 significantly lowered the number of adherent and transmigrating cells, with rolling cells being reduced by 68%, adherent cells by 55% and transmigrating cell by 72% (Fig.6b).
Figure 6.
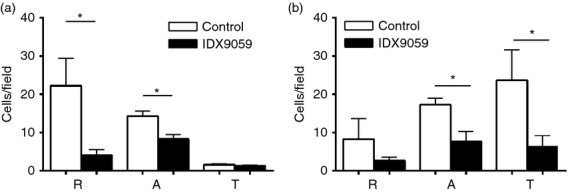
IDX9059 inhibit leucocyte migration in vivo. Leucocyte migration was analysed in vivo by chemotactic intravital microscopy in the cremaster muscle of mice. Cells per field before (a) and after (b) addition of platelet activating factor (PAF) were analysed using time-lapse video recordings. The animals were given IDX9059 subcutaneously, 50 μg/100 μl/mouse, c. 20 min before induction of inflammation. R = rolling cells, A = adhering cells and T = transmigrated cells. Results are presented as mean ± SEM,n = 4. *P < 0·05 by Student's t-test.
ODN reduced OVA-induced airway inflammation in mice
To study the effect of ODN on the migration of cells during acute inflammation, intranasal administration of IDX9059 was investigated in a mouse model of OVA-induced airway inflammation. The OVA-induced allergic asthma model is a widely used model to reproduce the airway eosinophilia, pulmonary inflammation and elevated IgE levels found during asthma. Treatment with IDX9059 significantly reduced the migration of inflammatory cells into the lung with a significant decrease in both the amount of infiltrating PMN and lymphocytes compared with PBS-treated mice (Fig.7). These results suggest that IDX9059 is a potent inhibitor of in vivo PMN migration as determined in this murine model of airway inflammation.
Figure 7.
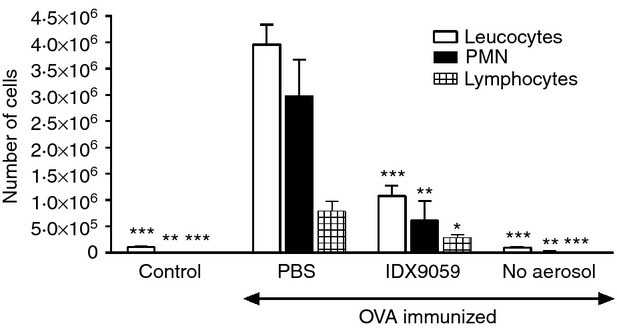
IDX9059 inhibit polymorphonuclear neutrophils (PMN) migration in a mouse model of ovalbumin (OVA) -induced airway inflammation. Mice were sensitized to OVA by intraperitoneal injection of OVA/alum on days 0 and 12. On days 23, 26, 30 and 33, mice were challenged in the lungs by inhalation of aerosolized OVA. At days 16 and 21, 50 μg/animal of IDX9059 were instilled intranasally. PBS was used as control for the ODN treatment. Mice that were not immunized with OVA but treated with PBS and exposed to OVA aerosol were included as controls, as well as mice that were immunized with OVA but not exposed to OVA aerosol (no aerosol). The total numbers of leucocytes, PMN and lymphocytes were analysed in bronchoalveolar lavage fluid 48 hr after the last OVA aerosol and are presented as mean ± SEM. *P < 0·05, **P < 0·01 and ***P < 0·001 by one-way analysis of variance using Dunnett's post hoc correction compared with PBS-treated control mice.
Discussion
Immunomodulatory CpG ODN have the potential to be used for activating protective immunity against several diseases such as allergy and cancer, as well as to function as vaccine adjuvants.1 The CpG ODN can exert their immunomodulatory effects by inducing cytokine secretion and by affecting the expression of cell surface receptors on leucocytes. PMN are one of the major cells mediating tissue damage during an inflammatory response. They migrate from the blood to the site of inflammation in response to locally produced chemoattractants. One of the major mediators of PMN migration is the CXC chemokine IL-8, which binds to two different receptors, CXCR1 and CXCR2, expressed on the cell surface. LTB4 is another important PMN chemoattractant which mediates its effect by binding to the cell surface receptor BLT1. In this study we investigated the effect of our ODN on the surface expression of CXCR1, CXCR2 and BLT1, and on the migration of PMN in vitro and in vivo.
Several of the novel ODN (IDX9022, IDX9052, IDX9054 and IDX9059) could significantly reduce the expression of CXCR1 on the surface of PMN to below 50% of what was seen for untreated cells. Down-regulation of CXCR2 surface expression could also be demonstrated but was mainly observed with two of the ODN (IDX9052 and IDX9054), and the reduction was not as substantial as for CXCR1. This indicates that these two receptors may be regulated through different mechanisms, which have been described previously by others.25 Furthermore, neutrophil transmigration appears to depend more on CXCR1 than on CXCR2,10 suggesting that a reduction in CXCR1 expression is more likely to have a greater impact at inhibiting neutrophil migration. A major reduction in BLT1 surface expression was also observed following stimulation with ODN. In addition, both the reduction in CXCR1 and CXCR2 as well as BLT1 surface expression correlated with inhibition of IL-8 or LTB4-induced PMN chemotaxis, which implies that the reduced PMN migration induced by the ODN is the result of decreased receptor expression.
The reduction in CXCR1 surface expression was rapid following ODN IDX9059 incubation and was already apparent after 15 min, with > 50% reduction in receptor levels observed after 1 hr. This fast decrease in CXCR1 surface expression is probably the result of a direct effect of the ODN on the PMN, and not the release of secondary mediators such as cytokines. This is an important point because neutrophils are terminally differentiated cells and therefore short lived, favouring the desire for agents that are capable of inducing rapid immunomodulatory effects. Hayashi et al.6 demonstrated a down-regulation of CXCR1 expression on PMN by CpG DNA, which was only induced if the cells were pre-treated with granulocyte–macrophage colony-stimulating factor. In this study, the ODN reduced surface expression of CXCR1 and CXCR2 in the absence of cytokine pre-treatment, underlining their potent immunomodulatory properties. It has previously been reported that IL-8 can induce the internalization of CXCR1 and CXCR2, and that both receptors can be down-modulated by TNF-α.23,24 However, no induction of either IL-8 or TNF-α was noted following stimulation with the ODN compounds, which may support a direct effect on the PMN.
Two of the ODN tested (IDX9031 and IDX9096) did not mediate any reduction in either CXCR1 or CXCR2 surface expression despite the fact that they both contain CpG motifs, demonstrating that solely a CpG motif is not enough to induce this effect, but that other motifs are probably of importance. The ODN that were most potent in down-regulating CXCR1, CXCR2 and BLT1 surface expression all have a sequence comprising a G-flank in one or both ends. This suggests that poly-G sequences might be of importance for mediating an effective reduction in CXCR1, CXCR2 and BLT1 surface expression. This is in agreement with previous studies showing enhanced immunomodulatory effect with poly-G-containing ODN in human leucocytes.26 In line with the data presented here it has previously been shown that the activation of neutrophils by bacterial DNA can occur by non-CpG ODN and through TLR9-independent mechanisms.27 Our results using Chloroquine blocking experiments would indicate that the reduction in surface CXCR1 expression induced by the ODN can be mediated either through endosomally dependent or endosomally independent mechanisms depending on the sequence of the ODN suggesting that a surface receptor may be involved. It has previously been demonstrated that neutrophil activation by bacterial DNA does not require DNA internalization and that bacterial DNA can bind to the neutrophil cell surface, supporting an interaction with a neutrophil surface receptor.28 Furthermore, it has been reported that ODN can bind to the Mac-1 complex (CD11b/CD18) on the surface of neutrophils.22 However, our experiment using anti-Mac-1 monoclonal antibodies to block the MAC-1 complex did not affect the reduction of CXCR1 surface expression induced by our ODN compounds, indicating that the MAC-1 receptor is not involved in this mechanism. Interestingly, it was recently reported that functional TLR9 can be expressed on the surface of human primary PMN,29 and binding of our ODN to surface TLR9 may be one explanation of why we see CXCR1 down-regulation despite blocking endosomal signalling.
It has previously been demonstrated that CXCR1 and CXCR2 surface expression can be down-regulated through several mechanisms. As mentioned above, IL-8-directed neutrophil activation and migration have been shown to be regulated by the internalization and subsequent re-expression of CXCR1 and CXCR2. However, other immunomodulators such as bacterial lipopolysaccharide down-regulate CXCR1 and CXCR2 through proteolytic cleavage of the receptors from the cell surface by the action of metalloproteinases.24 It is believed that lipopolysaccharide-mediated down-regulation of CXCR1 and CXCR2 may serve as a mechanism of immune evasion used by bacteria to interfere with the migration of neutrophils to the sites of bacterial infection. By which mechanism the CXCR1 and CXCR2 receptors are down-regulated in response to the ODN presented here remains to be investigated.
Leucocyte extravasation is the movement of leucocytes out of the circulation, towards the site of inflammation. This process can be divided into to several steps, i.e. rolling, adhesion and transmigration, and the leucocyte recruitment is halted whenever any of these steps is suppressed.30 During the first step, leucocytes bind reversibly to the vascular endothelium; however, this interaction cannot anchor the leucocyte against the force of the blood flow, making the leucocytes roll along the endothelium. If the expression of adhesion molecules and their receptors is increased by pro-inflammatory signals a tighter binding arrests the rolling and allows the leucocyte to squeeze between the endothelial cells into the underlying tissue, where they migrate along a concentration gradient of chemokines to the inflammation site.30
To study the effect of ODN on cell migration in vivo we applied chemotactic PAF on a cremaster mouse model of intravital microscopy. PAF are known to induce IL-8 production31,32 and IL-8 can enhance the response to PAF,33 showing that these two molecules are tightly link during an inflammatory response. By time-lapse video recordings we could demonstrate a significant inhibition of migrating leucocytes in vivo in response to IDX9059. The inhibitory effect of IDX9059 was seen as a reduction in both the number of rolling, adhering and transmigrating cells.
In vivo efficacy of IDX9059 could be further demonstrated in a mouse model of OVA-induced airway inflammation, in which IDX9059 significantly inhibited the infiltration of PMN into the site of inflammation, i.e. into the lung. An inhibitory effect was seen on both PMN and lymphocyte infiltration, which was in agreement with our preliminary data showing down-regulation of chemokine receptors also on lymphocytes by our CpG ODN (data not shown).
Increased infiltration of PMN to the inflammatory site is seen in many inflammatory disorders. For example, histological examination of biopsies from patients with ulcerative colitis revealed the presence of a large number of PMN.34 Additionally, the intestinal mucosa of patients with ulcerative colitis has increased expression of both IL-8 and its receptors CXCR1 and CXCR2.34 Moreover, asthmatic patients as well as patients with chronic obstructive pulmonary disease have increased infiltration of neutrophils and higher levels of IL-8 and its receptors in the bronchial mucosa.35,36 The LTB4/BLT1 pathway also has an important role in the pathogenesis of severe persistent asthma, and LTB4 production is, in general, resistant to corticosteroid treatment. In fact, it has been shown that corticosteroids can up-regulate BLT1 and it is thought that the LTB4/BLT1 pathway is contributing to the development of inflammation in allergic diseases that do not respond to corticosteroids.17 Furthermore, the IL-8/CXCR1/CXCR2 pathway as well as the LTB4/BLT1 pathway also appears to be involved in the pathogenesis of disease such as multiple sclerosis,37–39 cancer14 and in ischaemia and reperfusion injury.34 For this reason, the IL-8/CXCR1/CXCR2 pathway as well as the LTB4/BLT1 pathway constitute major targets for anti-inflammatory therapy, and several CXCR1/CXCR2 antagonists, and IL-8 blocking antibodies as well as LTB4 inhibitors are under clinical development.15,16,40,41 The fact that the ODN presented here can down-regulate the surface expression of CXCR1, CXCR2 and BLT1 simultaneously and inhibit PMN migration both in vitro and in vivo makes them attractive as potential agents for use in anti-inflammatory therapy.
Acknowledgments
We wish to thank Professor Lennart Lindbom and Ellinor Kenne for excellent support and help with the intravital microscopy experiments and Professor Anders Bucht and Linda Svensson for valuable advice and support with the experiments using the mice model of OVA-induced airway inflammation.
Glossary
- CXCR
CXC chemokine receptor
- LTB4
leukotriene B4
- ODN
oligonucleotides
- PAF
platelet activating factor
Author contributions
CA planned, conducted and analysed parts of the experiments and wrote the paper. LGA planned parts of the experiments and suggested improvements to the manuscript. OVS advised on the design of the study and the experimental strategy and suggested improvements to the manuscript. AZ designed the study and wrote the paper.
Disclosures
The authors of this paper are current or former employees of InDex Pharmaceuticals.
References
- Krieg AM. Therapeutic potential of Toll-like receptor 9 activation. Nat Rev Drug Discov. 2006;5:471–84. doi: 10.1038/nrd2059. [DOI] [PubMed] [Google Scholar]
- Wilson HL, Dar A, Napper SK, Marianela Lopez A, Babiuk LA, Mutwiri GK. Immune mechanisms and therapeutic potential of CpG oligodeoxynucleotides. Int Rev Immunol. 2006;25:183–213. doi: 10.1080/08830180600785868. [DOI] [PubMed] [Google Scholar]
- Musch E, Lutfi T, von Stein P, Zargari A, Admyre C, Malek M, Lofberg R, von Stein OD. Topical treatment with the Toll-like receptor agonist DIMS0150 has potential for lasting relief of symptoms in patients with chronic active ulcerative colitis by restoring glucocorticoid sensitivity. Inflamm Bowel Dis. 2012;19:283–92. doi: 10.1002/ibd.23019. [DOI] [PubMed] [Google Scholar]
- Hanagata N. Structure-dependent immunostimulatory effect of CpG oligodeoxynucleotides and their delivery system. Int J Nanomedicine. 2012;7:2181–95. doi: 10.2147/IJN.S30197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- El Kebir D, Jozsef L, Filep JG. Neutrophil recognition of bacterial DNA and Toll-like receptor 9-dependent and -independent regulation of neutrophil function. Arch Immunol Ther Exp (Warsz) 2008;56:41–53. doi: 10.1007/s00005-008-0008-3. [DOI] [PubMed] [Google Scholar]
- Hayashi F, Means TK, Luster AD. Toll-like receptors stimulate human neutrophil function. Blood. 2003;102:2660–9. doi: 10.1182/blood-2003-04-1078. [DOI] [PubMed] [Google Scholar]
- Jozsef L, Khreiss T, Filep JG. CpG motifs in bacterial DNA delay apoptosis of neutrophil granulocytes. FASEB J. 2004;18:1776–8. doi: 10.1096/fj.04-2048fje. [DOI] [PubMed] [Google Scholar]
- Fialkow L, Wang Y, Downey GP. Reactive oxygen and nitrogen species as signaling molecules regulating neutrophil function. Free Radic Biol Med. 2007;42:153–64. doi: 10.1016/j.freeradbiomed.2006.09.030. [DOI] [PubMed] [Google Scholar]
- Nasser MW, Raghuwanshi SK, Malloy KM, Gangavarapu P, Shim JY, Rajarathnam K, Richardson RM. CXCR1 and CXCR2 activation and regulation. Role of aspartate 199 of the second extracellular loop of CXCR2 in CXCL8-mediated rapid receptor internalization. J Biol Chem. 2007;282:6906–15. doi: 10.1074/jbc.M610289200. [DOI] [PubMed] [Google Scholar]
- Hammond ME, Lapointe GR, Feucht PH. IL-8 induces neutrophil chemotaxis predominantly via type I IL-8 receptors. J Immunol. 1995;155:1428–33. [PubMed] [Google Scholar]
- MacDermott RP. Chemokines in the inflammatory bowel diseases. J Clin Immunol. 1999;19:266–72. doi: 10.1023/a:1020583306627. [DOI] [PubMed] [Google Scholar]
- Frangogiannis NG, Entman ML. Chemokines in myocardial ischemia. Trends Cardiovasc Med. 2005;15:163–9. doi: 10.1016/j.tcm.2005.06.005. [DOI] [PubMed] [Google Scholar]
- Pease JE, Sabroe I. The role of interleukin-8 and its receptors in inflammatory lung disease: implications for therapy. Am J Respir Med. 2002;1:19–25. doi: 10.1007/BF03257159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Waugh DJ, Wilson C. The interleukin-8 pathway in cancer. Clin Cancer Res. 2008;14:6735–41. doi: 10.1158/1078-0432.CCR-07-4843. [DOI] [PubMed] [Google Scholar]
- Busch-Petersen J. Small molecule antagonists of the CXCR2 and CXCR1 chemokine receptors as therapeutic agents for the treatment of inflammatory diseases. Curr Top Med Chem. 2006;6:1345–52. doi: 10.2174/15680266106061345. [DOI] [PubMed] [Google Scholar]
- Yamagata T, Ichinose M. Agents against cytokine synthesis or receptors. Eur J Pharmacol. 2006;533:289–301. doi: 10.1016/j.ejphar.2005.12.046. [DOI] [PubMed] [Google Scholar]
- Ohnishi H, Miyahara N, Gelfand EW. The role of leukotriene B4 in allergic diseases. Allergol Int. 2008;57:291–8. doi: 10.2332/allergolint.08-RAI-0019. [DOI] [PubMed] [Google Scholar]
- Kim ND, Chou RC, Seung E, Tager AM, Luster AD. A unique requirement for the leukotriene B4 receptor BLT1 for neutrophil recruitment in inflammatory arthritis. J Exp Med. 2006;203:829–35. doi: 10.1084/jem.20052349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stankova J, Turcotte S, Harris J, Rola-Pleszczynski M. Modulation of leukotriene B4 receptor-1 expression by dexamethasone: potential mechanism for enhanced neutrophil survival. J Immunol. 2002;168:3570–6. doi: 10.4049/jimmunol.168.7.3570. [DOI] [PubMed] [Google Scholar]
- Kobayashi Y. The role of chemokines in neutrophil biology. Front Biosci. 2008;13:2400–7. doi: 10.2741/2853. [DOI] [PubMed] [Google Scholar]
- Tager AM, Luster AD. BLT1 and BLT2: the leukotriene B4 receptors. Prostaglandins Leukot Essent Fatty Acids. 2003;69:123–34. doi: 10.1016/s0952-3278(03)00073-5. [DOI] [PubMed] [Google Scholar]
- Benimetskaya L, Loike JD, Khaled Z. Mac-1 (CD11b/CD18) is an oligodeoxynucleotide-binding protein. Nat Med. 1997;3:414–20. doi: 10.1038/nm0497-414. [DOI] [PubMed] [Google Scholar]
- Samanta AK, Oppenheim JJ, Matsushima K. Interleukin 8 (monocyte-derived neutrophil chemotactic factor) dynamically regulates its own receptor expression on human neutrophils. J Biol Chem. 1990;265:183–9. [PubMed] [Google Scholar]
- Khandaker MH, Mitchell G, Xu L. Metalloproteinases are involved in lipopolysaccharide- and tumor necrosis factor-α-mediated regulation of CXCR1 and CXCR2 chemokine receptor expression. Blood. 1999;93:2173–85. [PubMed] [Google Scholar]
- Feniger-Barish R, Ran M, Zaslaver A, Ben-Baruch A. Differential modes of regulation of cxc chemokine-induced internalization and recycling of human CXCR1 and CXCR2. Cytokine. 1999;11:996–1009. doi: 10.1006/cyto.1999.0510. [DOI] [PubMed] [Google Scholar]
- Bartz H, Mendoza Y, Gebker M, Fischborn T, Heeg K, Dalpke A. Poly-guanosine strings improve cellular uptake and stimulatory activity of phosphodiester CpG oligonucleotides in human leukocytes. Vaccine. 2004;23:148–55. doi: 10.1016/j.vaccine.2004.05.020. [DOI] [PubMed] [Google Scholar]
- Trevani AS, Chorny A, Salamone G, Vermeulen M, Gamberale R, Schettini J, Raiden S, Geffner J. Bacterial DNA activates human neutrophils by a CpG-independent pathway. Eur J Immunol. 2003;33:3164–74. doi: 10.1002/eji.200324334. [DOI] [PubMed] [Google Scholar]
- Fuxman Bass JI, Gabelloni ML, Alvarez ME, Vermeulen ME, Russo DM, Zorreguieta A, Geffner JR, Trevani AS. Characterization of bacterial DNA binding to human neutrophil surface. Lab Invest. 2008;88:926–37. doi: 10.1038/labinvest.2008.59. [DOI] [PubMed] [Google Scholar]
- Lindau D, Mussard J, Wagner BJ. Primary blood neutrophils express a functional cell surface Toll-like receptor 9. Eur J Immunol. 2013;43:2101–13. doi: 10.1002/eji.201142143. [DOI] [PubMed] [Google Scholar]
- Picker LJ. Control of lymphocyte homing. Curr Opin Immunol. 1994;6:394–406. doi: 10.1016/0952-7915(94)90118-x. [DOI] [PubMed] [Google Scholar]
- Matsumoto K, Hashimoto S, Gon Y, Nakayama T, Horie T. Proinflammatory cytokine-induced and chemical mediator-induced IL-8 expression in human bronchial epithelial cells through p38 mitogen-activated protein kinase-dependent pathway. J Allergy Clin Immunol. 1998;101:825–31. doi: 10.1016/S0091-6749(98)70311-2. [DOI] [PubMed] [Google Scholar]
- Denault S, April MJ, Stankova J. Transcriptional activation of the interleukin-8 gene by platelet-activating factor in human peripheral blood monocytes. Immunology. 1997;91:297–302. doi: 10.1046/j.1365-2567.1997.00213.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wozniak A, Betts WH, Murphy GA, Rokicinski M. Interleukin-8 primes human neutrophils for enhanced superoxide anion production. Immunology. 1993;79:608–15. [PMC free article] [PubMed] [Google Scholar]
- Bizzarri C, Beccari AR, Bertini R, Cavicchia MR, Giorgini S, Allegretti M. ELR+ CXC chemokines and their receptors (CXC chemokine receptor 1 and CXC chemokine receptor 2) as new therapeutic targets. Pharmacol Ther. 2006;112:139–49. doi: 10.1016/j.pharmthera.2006.04.002. [DOI] [PubMed] [Google Scholar]
- Qiu Y, Zhu J, Bandi V, Guntupalli KK, Jeffery PK. Bronchial mucosal inflammation and upregulation of CXC chemoattractants and receptors in severe exacerbations of asthma. Thorax. 2007;62:475–82. doi: 10.1136/thx.2006.066670. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Papi A, Luppi F, Franco F, Fabbri LM. Pathophysiology of exacerbations of chronic obstructive pulmonary disease. Proc Am Thorac Soc. 2006;3:245–51. doi: 10.1513/pats.200512-125SF. [DOI] [PubMed] [Google Scholar]
- Bielecki B, Mazurek A, Wolinski P, Glabinski A. Treatment of multiple sclerosis with methylprednisolone and mitoxantrone modulates the expression of CXC chemokine receptors in PBMC. J Clin Immunol. 2008;28:122–30. doi: 10.1007/s10875-007-9142-7. [DOI] [PubMed] [Google Scholar]
- Neu IS, Metzger G, Zschocke J, Zelezny R, Mayatepek E. Leukotrienes in patients with clinically active multiple sclerosis. Acta Neurol Scand. 2002;105:63–6. doi: 10.1034/j.1600-0404.2002.00070.x. [DOI] [PubMed] [Google Scholar]
- Bartosik-Psujek H, Stelmasiak Z. The levels of chemokines CXCL8, CCL2 and CCL5 in multiple sclerosis patients are linked to the activity of the disease. Eur J Neurol. 2005;12:49–54. doi: 10.1111/j.1468-1331.2004.00951.x. [DOI] [PubMed] [Google Scholar]
- Hutchinson JH. Leukotriene B4 inhibitors: US2009054466; US2009227603 and US2009253684. Expert Opin Ther Pat. 2010;20:707–14. doi: 10.1517/13543771003657180. [DOI] [PubMed] [Google Scholar]
- Chapman RW, Minnicozzi M, Celly CS. A novel, orally active CXCR1/2 receptor antagonist, Sch527123, inhibits neutrophil recruitment, mucus production, and goblet cell hyperplasia in animal models of pulmonary inflammation. J Pharmacol Exp Ther. 2007;322:486–93. doi: 10.1124/jpet.106.119040. [DOI] [PubMed] [Google Scholar]