Significance
The inhibitory protein cytotoxic T-lymphocyte-associated antigen-4 (CTLA-4) is recognized as a crucial regulator of autoimmunity, but its precise mechanism of action is not yet fully understood. CTLA-4 can down-regulate expression of the costimulatory ligands CD80 and CD86 on antigen presenting cells, thereby reducing T-cell CD28 engagement. Here we demonstrate that quantitative changes in the level of CD28 engagement have functional consequences for T-cell differentiation toward follicular helper T cells (TFHs). These findings link CTLA-4 control of T-cell responses with the generation of high-affinity class-switched antibody responses. This generates an advanced conceptual framework for understanding the linked nature of CTLA-4 and CD28 functions and the role of this pathway in influencing autoimmunity.
Keywords: T cell, costimulation, CD28, CTLA-4, autoimmunity
Abstract
Cytotoxic T-lymphocyte-associated antigen-4 (CTLA-4) is an essential regulator of T-cell responses, and its absence precipitates lethal T-cell hyperactivity. However, whether CTLA-4 acts simply to veto the activation of certain clones or plays a more nuanced role in shaping the quality of T-cell responses is not clear. Here we report that T cells in CTLA-4–deficient mice show spontaneous T-follicular helper (TFH) differentiation in vivo, and this is accompanied by the appearance of large germinal centers (GCs). Remarkably, short-term blockade with anti–CTLA-4 antibody in wild-type mice is sufficient to elicit TFH generation and GC development. The latter occurs in a CD28-dependent manner, consistent with the known role of CTLA-4 in regulating the CD28 pathway. CTLA-4 can act by down-regulating CD80 and CD86 on antigen presenting cells (APCs), thereby altering the level of CD28 engagement. To mimic reduced CD28 ligation, we used mice heterozygous for CD28, revealing that the magnitude of CD28 engagement is tightly linked to the propensity for TFH differentiation. In contrast, other parameters of T-cell activation, including CD62L down-regulation and Ki67 expression, were relatively insensitive to altered CD28 level. Altered TFH generation as a result of graded reduction in CD28 was associated with decreased numbers of GC B cells and a reduction in overall GC size. These data support a model in which CTLA-4 control of immunity goes beyond vetoing T-cell priming and encompasses the regulation of TFH differentiation by graded control of CD28 engagement.
Control of the magnitude and nature of adaptive immune responses is critical for health. The cytotoxic T-lymphocyte-associated antigen-4 (CTLA-4)/CD28 axis has long been known to control the magnitude of T-cell responses, however whether it also influences their nature has not been clear. Early studies suggested that CD28 may be particularly important for Th2 differentiation (1, 2), although others identified roles for CD28 in both Th1 and Th2 responses (3, 4). It is known that CD28 is an absolute requirement for the differentiation of follicular helper T cells (TFHs) that support germinal center (GC) formation (5, 6). However, these studies generally make use of CD28-deficient T cells, and therefore, results may reflect a failure of the cells to properly activate, proliferate, or survive, particularly given the known contribution of CD28 to these processes.
A key outstanding question is whether CD28 costimulation in vivo is more complex than a binary checkpoint for T-cell priming. It is clear that expression of costimulatory ligands on antigen presenting cells (APCs) fluctuates in response to environmental stimuli, being up-regulated by inflammatory cytokines and TLR agonists and down-regulated by Treg-expressed CTLA-4 (7–11). Thus, variable levels of costimulatory ligands will be available for CD28 binding depending on the microenvironmental context. However, whether this simply alters the number of T cells that achieve the required threshold to commit to a response or whether it influences the nature of the response is not clear. Effective immune homeostasis appears to be reliant on maintaining an appropriate level of CD28 engagement. For example, basal expression of CD28 ligands, in particular dendritic cell-expressed CD86 (12), is critical to Treg homeostasis (13, 14), whereas excessive CD28 engagement in the absence of CTLA-4 results in lethal autoimmunity (15, 16). Nonetheless, distinguishing whether these effects are simply quantitative is not straightforward. Thus, although levels of CD28 ligands are clearly variable in vivo, our understanding of the impact of altering the level of CD28 engagement is still incomplete.
To explore the impact of varying levels of CD28 ligation, we have used the CTLA-4–deficient mouse as a model of excessive CD28 stimulation. In these mice, we observed a striking skewing toward TFH differentiation, with induction of IL-21 and spontaneous formation of GCs. In a complementary approach, we used CD28 heterozygosity to decrease T cell CD28 expression: This revealed that the level of CD28 engagement is tightly coupled to the level of inducible T-cell costimulator (ICOS) induction, TFH generation, and GC formation, whereas other parameters of T-cell activation were less affected. Finally, we demonstrate that induction of the microRNA cluster miR17-92, recently linked with TFH differentiation (17, 18), varies proportionally with APC costimulatory ligand expression and is modulated by CTLA-4 deficiency or blockade. Collectively, these data suggest that the CTLA-4/CD28 axis provides quantitative and qualitative control of T-cell help for humoral immunity.
Results
CTLA-4 Deficiency or Blockade Augments TFH Numbers.
Mice lacking CTLA-4 exhibit a lethal CD28-dependent lymphoproliferative syndrome with evidence of skewing to Th2 (19). However, the impact of CTLA-4 deficiency on TFH differentiation has not previously been examined. We therefore analyzed spleen (Fig. 1) and lymph node (Fig. S1) CD4 T cells for expression of a panel of TFH markers. CD4 T cells from CTLA-4–deficient mice expressed elevated levels of the TFH markers PD-1 and CXCR5 (Fig. 1A). IL-21, the archetypal cytokine associated with TFH differentiation, was up-regulated at the mRNA (Fig. S1C) and protein (Fig. 1B) level in conventional T cells (Tconv) from CTLA-4–deficient mice compared with age-matched wild-type controls. Consistent with augmented TFH differentiation, B cells with a GC phenotype (Fas+GL-7+) were readily detectable in the lymph nodes (LNs) and spleens of CTLA-4–deficient mice (Fig. 1C and Fig. S1), and immunohistological analysis revealed spontaneous GC formation (Fig. 1D). The systemic immune dysregulation associated with CTLA-4 deficiency makes it hard to establish whether features observed in these animals reflect a direct effect of CTLA-4 deficiency or a secondary effect of disease. Indeed, the T-cell compartment in CTLA-4–deficient mice shows evidence of extensive activation (Fig. S2). We therefore performed short-term CTLA-4 blockade studies in wild-type mice. Remarkably, short-term blockade of CTLA-4 was sufficient to induce the appearance of TFH cells (Fig. 2A), GC B cells (Fig. 2B), and immunohistologically evident GCs (Fig. 2C) in the spleen. Similar effects were noted in the LNs (Fig. S3 A and B). There was marked Bcl6 up-regulation in CD4 T cells from anti–CTLA-4 Ab-injected mice (Fig. 2D). GC formation could be detected as early as 3 d following anti–CTLA-4 Ab injection (Fig. S4A), and injection of control Ab did not induce GC formation (Fig. S4B). Importantly, GC formation was CD28-dependent, as anti–CTLA-4 Ab did not elicit spontaneous GC formation in CD28-deficient mice (Fig. S4C). To assess whether GC formation following CTLA-4 blockade was associated with autoantibody production, we first used serum from CTLA-4–deficient mice to confirm a suitable target autoantigen. Serum from CTLA-4–deficient, but not wild-type mice, stained stomach sections from Rag2−/− mice (Fig. S5A), consistent with the previous demonstration that deficiency of CTLA-4 in Treg triggers the production of antiparietal cell antibodies (Abs) and autoimmune gastritis (9). Interestingly, serum from mice treated with anti–CTLA-4 Ab also showed reactivity to Rag2−/− stomach sections, whereas serum from control-treated mice did not (Fig. S5B). In addition, anti-dsDNA Abs were detected in mice treated with anti–CTLA-4 Ab (Fig. S5C). Collectively, these data indicate that the CTLA-4 pathway controls TFH development in a CD28-dependent manner and that persistent CTLA-4–mediated regulation is required to prevent the spontaneous emergence of TFHs, GCs, and autoantibodies.
Fig. 1.
CTLA-4 deficiency elicits spontaneous T-cell help for GCs. (A) Representative staining and collated data showing the proportion of TFH (CXCR5+PD-1+) in splenic CD4 T cells from 15- to 19-d CTLA-4−/− mice or CTLA-4+/− littermate controls. (B) Spleen cell suspensions from 15- to 18-d CTLA-4−/− or CTLA-4+/+ mice were restimulated and stained for CD4 and intracellular Foxp3 and IL-21. Plots show representative IL-21 staining and collated data. n = 5–7, ***P < 0.001. (C) Spleen cells from 15- to 19-d CTLA-4−/− mice or CTLA-4+/− littermate controls were stained for the GC B-cell markers FAS and GL-7. Plots are gated on CD19+ cells, and representative staining and collated data are shown. (D) Frozen spleen sections from 17-d CTLA-4−/− mice or CTLA-4+/− littermate controls were stained for CD4 or PNA (blue) and IgD (brown). F, follicle; GC, germinal center; T, t zone.
Fig. 2.
CTLA-4 blockade triggers TFH differentiation and GC formation. BALB/c mice were treated with 500 μg of anti–CTLA-4 Ab (or control hamster IgG) every 2 d, and spleens were harvested on day 5 or 11. Representative FACS plots and pooled data for TFH frequency in gated CD4+ cells (A) and GC B-cell frequency in gated CD19+ cells (B). ***P < 0.001, *P < 0.05; ns, not significant. (C) Immunohistological staining of frozen spleen sections showing CD4 (blue) and IgD (brown) (Top) or PNA (blue) and IgD (brown) (Bottom). Images are from one experiment (n = 3) and are representative of three independent experiments with similar results. (D) CD4 T cells from mice treated for 11 d with anti–CTLA-4 Abs or control Abs were stained for expression of Bcl6, Gata3, and Tbet. Plots are gated on CD4+Foxp3– cells. Collated data depict fold increase in transcription factor mean fluorescence intensity (MFI) in CTLA-4 Ab-treated mice relative to control Ab-treated mice. n = 4, ***P < 0.001.
Quantitative Effect of CD28 on TFH Development.
Because the phenotype of CTLA-4−/− mice is likely due to excessive CD28 engagement (20, 21), we hypothesized that the GC response should also be influenced by directly affecting levels of CD28 on T cells. To probe whether changing the amount of CD28 engagement on Tconv altered their ability to support GC formation, we performed adoptive transfer experiments comparing CD28+/+, CD28+/−, and CD28−/− T cells. In this way, we were able to alter the level of CD28 available for ligation on the T cells, as CD28 expression was markedly lower on CD28+/− T cells compared with CD28+/+ T cells (Fig. S6A). Importantly, heterozygosity, rather than total deficiency, offers a better model of raising or lowering the level of CD28 engagement that could result from Treg-mediated ligand down-regulation on APCs. To ensure the absence of background GCs at the start of the experiment, CD28−/− mice were used as recipients; thus, in this system, the ability to support GC formation is restricted to the adoptively transferred T cells. DO11 T cells that were CD28+/+, CD28+/−, or CD28−/− were transferred, and recipient mice were immunized intraperitoneally with alum-precipitated hapten-conjugated ovalbumin (NP-OVA).
DO11 T cells from CD28+/− mice showed a markedly reduced propensity to acquire a TFH phenotype (based on coexpression of CXCR5 and PD1) (Fig. 3A) compared with their CD28+/+ counterparts, and this correlated with a decreased number of B cells bearing a GC B-cell phenotype (Fig. 3B). We noted that average GC size was also decreased in mice receiving CD28+/− T cells (Fig. 3C) and that Ab titres were lower than in recipients of CD28+/+ T cells (Fig. S7A). A major pathway implicated in the generation of GC responses is the ICOS pathway, the absence of which is associated with a defect in GC formation (22–25). T cells from CD28+/− mice also showed a graded decrease in ICOS expression, with ICOS levels being intermediate compared with CD28+/+ and CD28−/− T cells (Fig. 3D). Interestingly, not all activation markers were similarly affected by CD28 heterozygosity: CD62L down-regulation in CD28+/− T cells was similar to that seen in wild-type T cells, and Ki67 levels were comparable, indicating equivalent proliferation (Fig. 3 E and F). Analysis of Cell Trace profiles at early time points confirmed the similar proliferative response of CD28+/+ and CD28+/− T cells (Fig. S7B). It should be noted that the percentage and absolute number of DO11 T cells recovered was broadly equivalent for CD28+/+ and CD28+/− cells but very low for CD28−/− cells, consistent with poor T-cell survival in the complete absence of CD28 (Fig. 3G). Taken together, these data reveal that T-cell activation in the context of reduced CD28 engagement has a marked effect on TFH differentiation, while minimally altering CD62L down-regulation, proliferation, and survival.
Fig. 3.
Quantitative effect of CD28 on TFH and GC B-cell development. DO11 CD28+/+, DO11 CD28+/−, or DO11 CD28−/− T cells (2 × 105) were injected into CD28−/− mice that were immunized i.p. with 200 μg of NP-OVA/alum 24 h later. At day 8, spleens were harvested for analysis. (A) Percentage of TFH (CXCR5+PD-1+) within gated CD4+DO11+ T cells. (B) Percentage of GC B cells (Fas+GL-7+) within the CD19+ gate. (C) Average GC size based on staining of spleen sections for PNA and IgD. Each point represents the mean of >60 GCs scored in an individual spleen. (D) ICOS, (E) CD62L, and (F) Ki67 expression on CD4+DO11+ T cells. (G) Percentage and absolute number of DO11 T cells. Data for A–G are compiled from two experiments (n = 4–6).
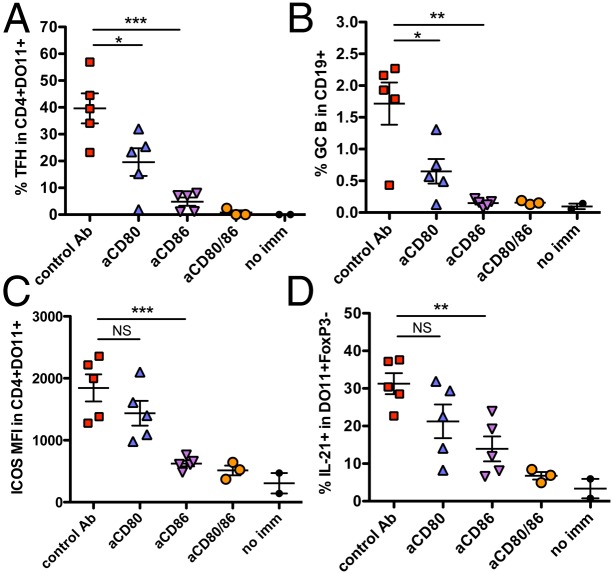
CD86 Is the Dominant Ligand for TFH Development.
Because the acquisition of a TFH phenotype, and support for GC formation, was clearly modulated by CD28 signaling, we sought to identify the CD28 ligand driving this process. Adoptive transfer experiments were performed using CD28+/+ DO11 T cells, and blocking Abs were injected against CD80, CD86, or both. The capacity of the CD80 and CD86 Ab to block their respective ligands was verified in vitro (Fig. S8A). Blockade of CD80 had only a modest effect on the frequency of TFH cells (Fig. 4A), and this was associated with a marked but incomplete reduction in GC B cells (Fig. 4B). In contrast, in the presence of CD86 Ab, the percentage of TFH cells was severely reduced—virtually to the same extent as when CD86 and CD80 were both blocked (Fig. 4A). The frequency of GC B cells was similarly impaired with CD86 blockade, to a comparable extent as with the simultaneous blockade of both ligands (Fig. 4B). The pattern of ICOS expression showed the same trend (Fig. 4C), whereas the data suggested a higher degree of redundancy between ligands for IL-21 production (Fig. 4D). Effects of ligand blockade on production of IFNγ and IL-17 and on CD62L expression are shown in Fig. S8B. Collectively, these experiments revealed that CD86 was the dominant CD28 ligand for generating the TFH cells that support GC development.
Fig. 4.
CD86 is the dominant ligand for TFH and GC development. CD28−/− mice were injected i.v. with DO11 WT T cells (0.2 × 106). One day later, mice were injected i.p. with 200 μg of NP-OVA/alum or PBS (no imm) and received two doses of anti-CD80 Ab, anti-CD86 Ab, or both i.p., as indicated (day 1 and day 5). At day 8, spleen cells were analyzed by flow cytometry. (A) Percentage of TFH (CXCR5+PD-1+) within gated CD4+DO11+ T cells. (B) Percentage of GC B cells (Fas+GL-7+) in gated CD19+ cells. (C) MFI ICOS expression within gated CD4+DO11+ T cells. (D) Percentage IL-21+ cells within CD4+DO11+Foxp3– cells after restimulation and intracellular staining. Data are compiled from 2 experiments (n = 3–5). ***P < 0.001, **P < 0.01, *P < 0.05; NS, not significant.
Graded Control of the microRNA-17–92 Cluster by CD28.
The miR-17–92 cluster has recently been shown to promote TFH generation (17, 18) and is induced in mouse T cells stimulated with anti-CD3 and anti-CD28 (26). We found that naïve T cells stimulated with anti-CD3 and anti-CD28 up-regulated higher levels of miR-17, an indicator miR for expression of this cluster, than those stimulated with anti-CD3 alone and that lack of CD28 stimulation could not be substituted by provision of IL-2 (Fig. 5A). To assess the impact of CD28 engagement using natural ligands rather than Abs, we activated naïve T cells in the presence of purified splenic B cells from CD80CD86+/+, CD80CD86+/−, or CD80CD86−/− mice, which express graded levels of CD80 and CD86 (Fig. S6B). This revealed a clear dose-dependent effect of CD80 and CD86 on miR-17 expression (Fig. 5B). Additional experiments revealed that deficiency or blockade of CTLA-4 led to increased T-cell miR-17 expression (Fig. S9). These data suggest that by altering expression levels of costimulatory ligands, CTLA-4 has the capacity to influence key controllers of the TFH differentiation program.
Fig. 5.

Graded control of the microRNA-17–92 cluster by CD28. (A) BALB/c CD4+CD25− T cells were stimulated for 1, 2, or 3 d (D1, D2, D3) with anti-CD3 alone or in the presence of anti-CD28 or 20 ng/mL of IL-2. mRNA was extracted, and levels of miR-17 expression were assessed. Graph shows fold change relative to naive CD4+CD25− T cells. Graph shows one experiment and is representative of three independent experiments with similar results. (B) BALB/c CD4+CD25− T cells were cultured for 3 d in the presence of anti-CD3, with costimulation being provided by splenic CD19+ B cells isolated from CD80CD86+/+, CD80CD86+/−, or CD80CD86−/− mice. T cells were reisolated at day 3, and miR-17 mRNA levels were assessed. Graph shows collated data from three independent experiments. Statistical analysis was performed by one-way ANOVA. *P < 0.05, **P < 0.01, ***P < 0.001.
Discussion
Multiple studies demonstrate the association between the CTLA-4/CD28 axis and autoimmunity (27–29). More recently, a link between TFH differentiation and autoimmunity has emerged (30, 31). Accordingly, overproduction of TFH in mice with a roquin mutation is associated with severe autoimmune disease (32, 33), and elevations in T cells with a TFH phenotype have been noted in systemic lupus erythematosus (34), myasthenia gravis (35, 36), and rheumatoid arthritis (37) (refs. 30, 31 and the references therein). Our data show that control of immunity via the CTLA-4/CD28 axis and control of TFH generation are fundamentally linked.
TFH cells provide a key link between T-cell activation and the ability to generate high-affinity class-switched Abs via GCs. The association between TFH and autoimmunity has sparked an increasing interest in understanding how TFH generation and function is regulated. It has been reported that a population of Qa1-restricted CD8 T cells expressing CXCR5 can regulate TFH numbers in a manner dependent on perforin expression (38). More recently, a subset of regulatory T cells termed TFR (T-follicular regulatory) has been identified that enter the GC and have the capacity to limit TFH and GC B-cell numbers (39–41). Thus, the magnitude of TFH and GC responses is likely controlled by specialized Treg.
A major portion of CTLA-4 function can be attributed to its role in Treg (9, 42) and CTLA-4 expression, along with IL-2 repression, is the minimal requirement to confer Treg-like suppressive activity (43). One mechanism of action of CTLA-4 is the down-regulation of costimulatory ligands on APCs (7–10), which can occur via a process of transendocytosis (11). This results in decreased availability of ligands for CD28-mediated costimulation of T cells. Accordingly, Treg-expressed CTLA-4 can directly control CD28 signaling in Tconv by restricting CD28 ligand availability. Intriguingly, Tconv can also use CTLA-4 to mediate transendocytosis (11), and we (44) and others (45) have shown that Tconv-expressed CTLA-4 can elicit regulation in a cell-extrinsic manner. This suggests a common mechanism of action for CTLA-4 regardless of the cell type on which it is expressed. Thus, CTLA-4, on both Treg and Tconv, can down-regulate costimulatory ligands and thereby decrease T-cell CD28 stimulation. Although one major impact of CTLA-4 function is clearly to prevent self-reactive T-cell activation, additional functions may include its impact on T-cell differentiation and B-cell responses as indicated here.
The mechanism used by TFR to regulate the GC response is not yet clear. Notably, TFRs express CTLA-4 at high levels (39), suggesting they are well placed to use the CTLA-4 pathway to elicit suppression. Indeed, TFRs were clearly detectable in GC induced by anti–CTLA-4 Ab treatment (Fig. S10), consistent with the idea that despite their correct positioning TFRs are unable to regulate GCs in the absence of CTLA-4. Furthermore, in one study, TFRs were reported to express low levels of CD25 (39), suggesting a decreased capacity to use IL-2 sequestration (46) as a suppressive mechanism. Indeed, consuming local IL-2 may be of limited value in regulating the TFH responses, as IL-2 itself inhibits TFH differentiation (47–50).
It has been shown that TFRs limit the outgrowth of nonantigen-specific B cells in the GC (39), thereby regulating GC size. Indeed, the deletion of B cells that are unable to elicit T-cell help is a key selection step within the GC that permits affinity maturation of the humoral response. The importance of such deletion is exemplified by the lymphoproliferative disease that affects mice in which Fas-mediated apoptosis is blocked in GC B cells (51). The simplest explanation for the capacity of TFR to control GC size is that they limit TFH number and thereby restrict the ability of T cells to rescue GC B cells from death. Because TFH homeostasis is tightly linked to the availability of costimulatory ligands on B cells (52, 53), it is easy to envisage how TFR could use the CTLA-4 pathway to control TFH by down-regulating costimulatory ligand expression on B cells.
TFHs are endowed with the capacity to solicit CD86 expression on B cells via production of IL-21 (54) or IL-4 (55). Thus, high-affinity B cells that competitively capture antigen from follicular dendritic cells, and productively engage with TFHs in the light zone of GCs, may be rewarded with cytokines that up-regulate their CD86 expression. This allows them to offer CD28 engagement to the T cells providing help, likely augmenting IL-21 production and beginning a feedback loop that will ensure entry of the clone into the long-lived plasma cell or memory B-cell pool.
That a complete block in CD28 signaling abrogates the GC response has been known for some time. We previously reported (6) that this reflected a requirement for CD28 to up-regulate CXCR5 on T cells, the chemokine receptor that allows them to respond to CXCL13 expressed in the B-cell follicle. Accordingly, in mice where CD28 signaling was blocked by transgenic expression of CTLA-4–Ig, T cells failed to up-regulate CXCR5; however, CXCR5 up-regulation, follicular migration, and GC formation could be restored if CD80/86 blockade was bypassed by injection of agonistic anti-CD28 (6). Thus, CD28 plays a critical role in resetting the T-cell chemokine receptor balance that permits T-cell localization to the B-cell follicles. Here we move beyond the simple presence or absence of costimulation and instead explore how the strength of CD28 signaling alters T-cell fate. We demonstrate that sensing of costimulatory ligands in vivo is analog and not digital—that is, it is not all or nothing but reflects the overall level of CD28 engagement. Commitment to a TFH phenotype appears to require a higher level of CD28 engagement than commitment to proliferation or CD62L down-regulation. This may reflect the fact that such commitment also licenses the B-cell response and therefore serves as an additional checkpoint in the adaptive immune response. Indeed, our data suggest that the level of CD28 signaling directly couples the magnitude of the T-helper response to the magnitude of the B-cell response.
In addition to CD28-mediated induction of CXCR5, ICOS-derived signals are required to enhance T-cell motility to facilitate entry to the B-cell follicle (56). Accordingly, the failure of ICOS-deficient T cells to enter B-cell follicles cannot be overcome by transgenic expression of CXCR5 (56). The division of labor between CD28 and ICOS in control of T-cell help for B cells has been a topic of intense debate. We and others previously suggested a hierarchy of costimulatory interactions in which CD28 operates upstream of ICOS (57, 58). This is consistent with CD28 being expressed on naïve T cells while ICOS is induced following T-cell activation (59). Indeed, it was noted that restricting ICOS function to downstream of CD28 was key to maintaining two-signal control of T-cell responses (60). We now extend this concept and suggest that CD28 and ICOS function are inherently coupled. Our data demonstrate that the strength of CD28 signaling in vivo translates directly into the level of ICOS surface expression induced on the T cell. This provides an elegant mechanism linking CD28 engagement to the ICOS-dependent activation of PI3K known to be critical for the generation of TFHs (61, 62). Thus, neither CD28 nor ICOS alone is sufficient to induce the TFH program, consistent with microarray data obtained following independent engagement of these receptors (63). Rather, our data suggest that CD28 and ICOS function together to confer the TFH phenotype. The long-range linkage disequilibrium reported at the CD28 and ICOS loci (64) may lock together variants that function coordinately to couple T- and B-cell function.
Although the CD28-dependent up-regulation of ICOS is likely to be a key step in driving TFH generation, it is not the sole mediator of the response. Accordingly, CD28−/− mice overexpressing ICOS as a consequence of roquin mutation do not form spontaneous GCs (although GCs can be induced by immunization) (60). Thus, overstimulation of CD28, as a consequence of CTLA-4 deficiency or blockade (Figs. 1 and 2), induces a distinct phenotype from overexpression of ICOS. Other mechanisms by which CD28 may promote TFH generation include regulation of the microRNA cluster 17–92, as shown here. Recent data revealed that expression of the miR-17–92 cluster is critical for the differentiation and function of TFH (17, 18). Indeed, overexpression of miR-17–92 promotes TFH generation (17), and transgenic expression of miR-17–92 in T cells can drive spontaneous TFH differentiation and GC formation, similar to our findings with CTLA-4 deficiency and blockade (18). The functionally relevant targets of the miR-17–92 cluster are still unclear, but the mechanism of action is likely to include direct and indirect repression of genes that antagonize TFH differentiation. Control of genes that regulate the PI3K pathway (e.g., the PI3K antagonist PTEN and the AKT phosphatase PHLPP2) may be particularly important (17, 18), consistent with the well-recognized role of PI3K in the GC response (61, 62).
The relative contribution of CD86 and CD80 to T-cell–dependent Ab responses is still unclear. The dominant role for CD86 identified here is consistent with the demonstration that CD86−/− mice show a more profound defect in GC formation than CD80−/− mice (65). Given the sequential interaction of T cells with dendritic cells (DCs) and B cells during humoral responses, this could reflect distinct cell type-specific or kinetic expression patterns between ligands. Of note, studies have documented the importance of either CD86 (52) or CD80 (53) on B cells for TFH generation and GC responses. It is possible that the nature and context of the antigenic challenge dictate the relative requirement for CD80 versus CD86, consistent with the capacity of strong adjuvants to restore the GC response in CD86−/− mice (65).
Collectively our data demonstrate that fine-tuning the degree of CD28 engagement—for example, by CTLA-4–dependent down-regulation of costimulatory ligands on DCs or GC B cells—results in tailored modulation of the TFH response. Thus, T-cell–dependent humoral immunity is tightly controlled by the CTLA-4 pathway.
Materials and Methods
Mice.
BALB/c and DO11.10 mice were from The Jackson Laboratory and CD28−/− mice from Taconic Laboratories. BALB/c CTLA-4−/− mice were kindly provided by A. Sharpe (Harvard, Boston, MA). CD80CD86−/− mice were from the Mutant Mouse Regional Resource Center. Mice were housed in individually vented cages at University College London or within the University of Birmingham Biological Services Unit. Experiments were performed in accordance with the relevant Home Office project and personal licenses following institutional ethical approval (University of Birmingham and University College London).
Flow Cytometry.
Cells were stained with Ab against CD4 (RM4-5; BD Biosciences), ICOS (7E.17G9), PD-1 (RMP1-30; Biolegend), CXCR5 (SPRCL5), CD19 (ID3; BD Biosciences), FAS (Jo2; BD Biosciences), GL7 (GL7; BD Biosciences), CD3 (17A2), Foxp3 (FJK-16s), IL-21 (mhalx21), DO11.10 TCR (KJ1-26), CD62L (MEL-14), Ki67 (B56; BD Biosciences), CD28 (37.51; BD Biosciences), Bcl6 (K112-91), Gata3 (L50-823; BD Biosciences), and Tbet (4B10, Biolegend). All Abs were purchased from eBioscience unless otherwise stated. For IL-21 measurement, cells were stimulated with phorbol myristate acetate (PMA) and ionomycin for 5 h in the presence of brefeldin A. For IL-21, Foxp3, and Ki67 staining, cells were fixed and permeabilized (eBioscience).
RT-PCR.
Cells were purified by sorting (MoFlo, Dako Cytomation), and RNA was isolated using RNAzol B (Biogenesis), reverse transcribed and interrogated for IL-21 (TaqMan, Applied Biosystems) using the Stratagene MX3000P real-time PCR detection system. Gene expression was normalized to β-actin levels.
Immunohistochemistry and Confocal Microscopy.
Acetone-fixed frozen 5-μm spleen sections were stained with sheep anti-IgD (Abcam) and rat anti-CD4 (BD Biosciences) or biotinylated peanut agglutinin (PNA; Vector Labs). Second step reagents were HRP-conjugated donkey anti-sheep IgG (Binding Site), biotinylated rabbit anti-rat Igs, followed by Streptavidin–ABComplex–alkaline phosphate (Vector Labs). Staining was visualized using FastBlue and DAB (3,3′-diaminobenzidine; Sigma-Aldrich). We incubated 5-μm cryostat sections from rag2−/− stomach with serum samples from CTLA-4–deficient, wild-type, or Ab-treated mice, as indicated. Bound Ab was detected with anti-mouse IgG-555, and sections were counterstained with DAPI. Images were captured by confocal microscopy (Zeiss LSM 510 Meta).
In Vivo Experiments.
BALB/c mice 9–10 wk old were injected with anti–CTLA-4 blocking Ab (4F10) or hamster IgG i.p. every 2 d. For adoptive transfer experiments, DO11 T cells (2-7 × 105) from DO11 WT, DO11 CD28+/−, or DO11 CD28−/− mice were injected i.v. into CD28−/− recipients. Where indicated, cells were labeled with Cell Trace Violet. One day later, recipients were immunized i.p. with 200 μg of NP-OVA/alum. Anti-CD80 (clone 16–10A1), anti-CD86 (clone GL-1) blocking Ab, or both (100 μg each Ab per injection) were injected twice i.p. where indicated (first dose immediately before antigen administration, second dose 4 d postimmunization). Control Ab-treated mice received 100 μg of rat IgG and 100 μg of hamster IgG. All injections were carried out in the morning, in the absence of anesthesia and analgesia, and mice were returned immediately to the home cage following the procedure. The welfare of experimental animals was monitored regularly (typically immediately postprocedure, then at least every 2–3 d). No adverse events were noted during these experiments.
miRNA Analysis.
CD4+CD25– T cells and CD19+ B cells were purified from LNs and spleens of BALB/c mice, respectively, by magnetic separation (Miltenyi Biotec). T cells (5 × 104 cells per well) were activated with 2 μg/mL of plate-bound anti-CD3 Ab alone or in combination with 5 μg/mL of anti-CD28 or 20 ng/mL of IL-2. In some experiments, T cells activated with anti-CD3 in the presence of B cells (2:1 B:T ratio). Three days later, B cells were removed by magnetic cell separation, and T cells were snap-frozen for RNA extraction. For ex vivo miR-17 analysis, splenic CD4 T cells were isolated from age-matched WT, CTLA-4+/−, CTLA-4−/−, or anti–CTLA-4 Ab-treated mice. Total RNA was extracted using a mirVana miRNA Isolation Kit (Ambion, Life Technologies) and converted to cDNA with a TaqMan MicroRNA Reverse Transcription Kit. The miRNA expression levels of mouse miR-17 were established by real-time PCR using Taqman primers (Taqman, Life Technologies) and a 7500 Fast RT-PCR system (Applied Biosystems, Life Technologies). Expression was normalized against RNA from freshly isolated CD4+CD25– T cells and the sno202 housekeeping gene, with the ΔΔ cyclic threshold method used to calculate relative fold change.
Statistics.
Statistical analysis was performed using Graphpad Prism version 5, and P values were calculated by two-tailed, unpaired t test for the means with a 95% confidence interval. Analysis of more than two samples was performed by one-way ANOVA.
Supplementary Material
Acknowledgments
This work was funded by a Medical Research Council Senior Fellowship (to L.S.K.W.). L.W. and R.K. are funded by Diabetes UK.
Footnotes
The authors declare no conflict of interest.
This article is a PNAS Direct Submission.
This article contains supporting information online at www.pnas.org/lookup/suppl/doi:10.1073/pnas.1414576112/-/DCSupplemental.
References
- 1.King CL, Stupi RJ, Craighead N, June CH, Thyphronitis G. CD28 activation promotes Th2 subset differentiation by human CD4+ cells. Eur J Immunol. 1995;25(2):587–595. doi: 10.1002/eji.1830250242. [DOI] [PubMed] [Google Scholar]
- 2.Rulifson IC, Sperling AI, Fields PE, Fitch FW, Bluestone JA. CD28 costimulation promotes the production of Th2 cytokines. J Immunol. 1997;158(2):658–665. [PubMed] [Google Scholar]
- 3.Seder RA, Germain RN, Linsley PS, Paul WE. CD28-mediated costimulation of interleukin 2 (IL-2) production plays a critical role in T cell priming for IL-4 and interferon gamma production. J Exp Med. 1994;179(1):299–304. doi: 10.1084/jem.179.1.299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Schweitzer AN, Borriello F, Wong RC, Abbas AK, Sharpe AH. Role of costimulators in T cell differentiation: Studies using antigen-presenting cells lacking expression of CD80 or CD86. J Immunol. 1997;158(6):2713–2722. [PubMed] [Google Scholar]
- 5.Ferguson SE, Han S, Kelsoe G, Thompson CB. CD28 is required for germinal center formation. J Immunol. 1996;156(12):4576–4581. [PubMed] [Google Scholar]
- 6.Walker LS, et al. Compromised OX40 function in CD28-deficient mice is linked with failure to develop CXC chemokine receptor 5-positive CD4 cells and germinal centers. J Exp Med. 1999;190(8):1115–1122. doi: 10.1084/jem.190.8.1115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Oderup C, Cederbom L, Makowska A, Cilio CM, Ivars F. Cytotoxic T lymphocyte antigen-4-dependent down-modulation of costimulatory molecules on dendritic cells in CD4+ CD25+ regulatory T-cell-mediated suppression. Immunology. 2006;118(2):240–249. doi: 10.1111/j.1365-2567.2006.02362.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Onishi Y, Fehervari Z, Yamaguchi T, Sakaguchi S. Foxp3+ natural regulatory T cells preferentially form aggregates on dendritic cells in vitro and actively inhibit their maturation. Proc Natl Acad Sci USA. 2008;105(29):10113–10118. doi: 10.1073/pnas.0711106105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Wing K, et al. CTLA-4 control over Foxp3+ regulatory T cell function. Science. 2008;322(5899):271–275. doi: 10.1126/science.1160062. [DOI] [PubMed] [Google Scholar]
- 10.Schmidt EM, et al. Ctla-4 controls regulatory T cell peripheral homeostasis and is required for suppression of pancreatic islet autoimmunity. J Immunol. 2009;182(1):274–282. doi: 10.4049/jimmunol.182.1.274. [DOI] [PubMed] [Google Scholar]
- 11.Qureshi OS, et al. Trans-endocytosis of CD80 and CD86: A molecular basis for the cell-extrinsic function of CTLA-4. Science. 2011;332(6029):600–603. doi: 10.1126/science.1202947. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Bar-On L, Birnberg T, Kim KW, Jung S. Dendritic cell-restricted CD80/86 deficiency results in peripheral regulatory T-cell reduction but is not associated with lymphocyte hyperactivation. Eur J Immunol. 2011;41(2):291–298. doi: 10.1002/eji.201041169. [DOI] [PubMed] [Google Scholar]
- 13.Salomon B, et al. B7/CD28 costimulation is essential for the homeostasis of the CD4+CD25+ immunoregulatory T cells that control autoimmune diabetes. Immunity. 2000;12(4):431–440. doi: 10.1016/s1074-7613(00)80195-8. [DOI] [PubMed] [Google Scholar]
- 14.Tang Q, et al. Cutting edge: CD28 controls peripheral homeostasis of CD4+CD25+ regulatory T cells. J Immunol. 2003;171(7):3348–3352. doi: 10.4049/jimmunol.171.7.3348. [DOI] [PubMed] [Google Scholar]
- 15.Tivol EA, et al. Loss of CTLA-4 leads to massive lymphoproliferation and fatal multiorgan tissue destruction, revealing a critical negative regulatory role of CTLA-4. Immunity. 1995;3(5):541–547. doi: 10.1016/1074-7613(95)90125-6. [DOI] [PubMed] [Google Scholar]
- 16.Waterhouse P, et al. Lymphoproliferative disorders with early lethality in mice deficient in Ctla-4. Science. 1995;270(5238):985–988. doi: 10.1126/science.270.5238.985. [DOI] [PubMed] [Google Scholar]
- 17.Baumjohann D, et al. The microRNA cluster miR-17∼92 promotes TFH cell differentiation and represses subset-inappropriate gene expression. Nat Immunol. 2013;14(8):840–848. doi: 10.1038/ni.2642. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Kang SG, et al. MicroRNAs of the miR-17∼92 family are critical regulators of T(FH) differentiation. Nat Immunol. 2013;14(8):849–857. doi: 10.1038/ni.2648. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Khattri R, Auger JA, Griffin MD, Sharpe AH, Bluestone JA. Lymphoproliferative disorder in CTLA-4 knockout mice is characterized by CD28-regulated activation of Th2 responses. J Immunol. 1999;162(10):5784–5791. [PubMed] [Google Scholar]
- 20.Mandelbrot DA, McAdam AJ, Sharpe AH. B7-1 or B7-2 is required to produce the lymphoproliferative phenotype in mice lacking cytotoxic T lymphocyte-associated antigen 4 (CTLA-4) J Exp Med. 1999;189(2):435–440. doi: 10.1084/jem.189.2.435. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Tai X, Van Laethem F, Sharpe AH, Singer A. Induction of autoimmune disease in CTLA-4-/- mice depends on a specific CD28 motif that is required for in vivo costimulation. Proc Natl Acad Sci USA. 2007;104(34):13756–13761. doi: 10.1073/pnas.0706509104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.McAdam AJ, et al. ICOS is critical for CD40-mediated antibody class switching. Nature. 2001;409(6816):102–105. doi: 10.1038/35051107. [DOI] [PubMed] [Google Scholar]
- 23.Tafuri A, et al. ICOS is essential for effective T-helper-cell responses. Nature. 2001;409(6816):105–109. doi: 10.1038/35051113. [DOI] [PubMed] [Google Scholar]
- 24.Dong C, et al. ICOS co-stimulatory receptor is essential for T-cell activation and function. Nature. 2001;409(6816):97–101. doi: 10.1038/35051100. [DOI] [PubMed] [Google Scholar]
- 25.Dong C, Temann UA, Flavell RA. Cutting edge: Critical role of inducible costimulator in germinal center reactions. J Immunol. 2001;166(6):3659–3662. doi: 10.4049/jimmunol.166.6.3659. [DOI] [PubMed] [Google Scholar]
- 26.de Kouchkovsky D, et al. microRNA-17-92 regulates IL-10 production by regulatory T cells and control of experimental autoimmune encephalomyelitis. J Immunol. 2013;191(4):1594–1605. doi: 10.4049/jimmunol.1203567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Ueda H, et al. Association of the T-cell regulatory gene CTLA4 with susceptibility to autoimmune disease. Nature. 2003;423(6939):506–511. doi: 10.1038/nature01621. [DOI] [PubMed] [Google Scholar]
- 28.Keir ME, Sharpe AH. The B7/CD28 costimulatory family in autoimmunity. Immunol Rev. 2005;204:128–143. doi: 10.1111/j.0105-2896.2005.00242.x. [DOI] [PubMed] [Google Scholar]
- 29.Gough SC, Walker LS, Sansom DM. CTLA4 gene polymorphism and autoimmunity. Immunol Rev. 2005;204:102–115. doi: 10.1111/j.0105-2896.2005.00249.x. [DOI] [PubMed] [Google Scholar]
- 30.Tangye SG, Ma CS, Brink R, Deenick EK. The good, the bad and the ugly—TFH cells in human health and disease. Nat Rev Immunol. 2013;13(6):412–426. doi: 10.1038/nri3447. [DOI] [PubMed] [Google Scholar]
- 31.Ma CS, Deenick EK. Human T follicular helper (Tfh) cells and disease. Immunol Cell Biol. 2014;92(1):64–71. doi: 10.1038/icb.2013.55. [DOI] [PubMed] [Google Scholar]
- 32.Vinuesa CG, et al. A RING-type ubiquitin ligase family member required to repress follicular helper T cells and autoimmunity. Nature. 2005;435(7041):452–458. doi: 10.1038/nature03555. [DOI] [PubMed] [Google Scholar]
- 33.Linterman MA, et al. Follicular helper T cells are required for systemic autoimmunity. J Exp Med. 2009;206(3):561–576. doi: 10.1084/jem.20081886. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Simpson N, et al. Expansion of circulating T cells resembling follicular helper T cells is a fixed phenotype that identifies a subset of severe systemic lupus erythematosus. Arthritis Rheum. 2010;62(1):234–244. doi: 10.1002/art.25032. [DOI] [PubMed] [Google Scholar]
- 35.Saito R, et al. Altered expression of chemokine receptor CXCR5 on T cells of myasthenia gravis patients. J Neuroimmunol. 2005;170(1-2):172–178. doi: 10.1016/j.jneuroim.2005.09.001. [DOI] [PubMed] [Google Scholar]
- 36.Tackenberg B, et al. Clonal expansions of CD4+ B helper T cells in autoimmune myasthenia gravis. Eur J Immunol. 2007;37(3):849–863. doi: 10.1002/eji.200636449. [DOI] [PubMed] [Google Scholar]
- 37.Wang J, et al. High frequencies of activated B cells and T follicular helper cells are correlated with disease activity in patients with new-onset rheumatoid arthritis. Clin Exp Immunol. 2013;174(2):212–220. doi: 10.1111/cei.12162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Kim HJ, Verbinnen B, Tang X, Lu L, Cantor H. Inhibition of follicular T-helper cells by CD8(+) regulatory T cells is essential for self tolerance. Nature. 2010;467(7313):328–332. doi: 10.1038/nature09370. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Linterman MA, et al. Foxp3+ follicular regulatory T cells control the germinal center response. Nat Med. 2011;17(8):975–982. doi: 10.1038/nm.2425. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Chung Y, et al. Follicular regulatory T cells expressing Foxp3 and Bcl-6 suppress germinal center reactions. Nat Med. 2011;17(8):983–988. doi: 10.1038/nm.2426. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Wollenberg I, et al. Regulation of the germinal center reaction by Foxp3+ follicular regulatory T cells. J Immunol. 2011;187(9):4553–4560. doi: 10.4049/jimmunol.1101328. [DOI] [PubMed] [Google Scholar]
- 42.Walker LS. Treg and CTLA-4: Two intertwining pathways to immune tolerance. J Autoimmun. 2013;45:49–57. doi: 10.1016/j.jaut.2013.06.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Yamaguchi T, et al. Construction of self-recognizing regulatory T cells from conventional T cells by controlling CTLA-4 and IL-2 expression. Proc Natl Acad Sci USA. 2013;110(23):E2116–E2125. doi: 10.1073/pnas.1307185110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Wang CJ, et al. Cutting edge: Cell-extrinsic immune regulation by CTLA-4 expressed on conventional T cells. J Immunol. 2012;189(3):1118–1122. doi: 10.4049/jimmunol.1200972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Corse E, Allison JP. Cutting edge: CTLA-4 on effector T cells inhibits in trans. J Immunol. 2012;189(3):1123–1127. doi: 10.4049/jimmunol.1200695. [DOI] [PubMed] [Google Scholar]
- 46.Pandiyan P, Zheng L, Ishihara S, Reed J, Lenardo MJ. CD4+CD25+Foxp3+ regulatory T cells induce cytokine deprivation-mediated apoptosis of effector CD4+ T cells. Nat Immunol. 2007;8(12):1353–1362. doi: 10.1038/ni1536. [DOI] [PubMed] [Google Scholar]
- 47.Johnston RJ, Choi YS, Diamond JA, Yang JA, Crotty S. STAT5 is a potent negative regulator of TFH cell differentiation. J Exp Med. 2012;209(2):243–250. doi: 10.1084/jem.20111174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Ballesteros-Tato A, et al. Interleukin-2 inhibits germinal center formation by limiting T follicular helper cell differentiation. Immunity. 2012;36(5):847–856. doi: 10.1016/j.immuni.2012.02.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Pepper M, Pagán AJ, Igyártó BZ, Taylor JJ, Jenkins MK. Opposing signals from the Bcl6 transcription factor and the interleukin-2 receptor generate T helper 1 central and effector memory cells. Immunity. 2011;35(4):583–595. doi: 10.1016/j.immuni.2011.09.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Choi YS, et al. ICOS receptor instructs T follicular helper cell versus effector cell differentiation via induction of the transcriptional repressor Bcl6. Immunity. 2011;34(6):932–946. doi: 10.1016/j.immuni.2011.03.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Hao Z, et al. Fas receptor expression in germinal-center B cells is essential for T and B lymphocyte homeostasis. Immunity. 2008;29(4):615–627. doi: 10.1016/j.immuni.2008.07.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Salek-Ardakani S, et al. B cell-specific expression of B7-2 is required for follicular Th cell function in response to vaccinia virus. J Immunol. 2011;186(9):5294–5303. doi: 10.4049/jimmunol.1100406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Good-Jacobson KL, Song E, Anderson S, Sharpe AH, Shlomchik MJ. CD80 expression on B cells regulates murine T follicular helper development, germinal center B cell survival, and plasma cell generation. J Immunol. 2012;188(9):4217–4225. doi: 10.4049/jimmunol.1102885. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Attridge K, et al. IL-21 promotes CD4 T cell responses by phosphatidylinositol 3-kinase-dependent upregulation of CD86 on B cells. J Immunol. 2014;192(5):2195–2201. doi: 10.4049/jimmunol.1302082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Marshall-Clarke S, Tasker L, Heaton MP, Parkhouse RM. A differential requirement for phosphoinositide 3-kinase reveals two pathways for inducible upregulation of major histocompatibility complex class II molecules and CD86 expression by murine B lymphocytes. Immunology. 2003;109(1):102–108. doi: 10.1046/j.1365-2567.2003.01638.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Xu H, et al. Follicular T-helper cell recruitment governed by bystander B cells and ICOS-driven motility. Nature. 2013;496(7446):523–527. doi: 10.1038/nature12058. [DOI] [PubMed] [Google Scholar]
- 57.Walker LS, et al. Established T cell-driven germinal center B cell proliferation is independent of CD28 signaling but is tightly regulated through CTLA-4. J Immunol. 2003;170(1):91–98. doi: 10.4049/jimmunol.170.1.91. [DOI] [PubMed] [Google Scholar]
- 58.Wong SC, Tan AH, Lam KP. Functional hierarchy and relative contribution of the CD28/B7 and ICOS/B7-H2 costimulatory pathways to T cell-mediated delayed-type hypersensitivity. Cell Immunol. 2009;256(1-2):64–71. doi: 10.1016/j.cellimm.2009.01.009. [DOI] [PubMed] [Google Scholar]
- 59.Hutloff A, et al. ICOS is an inducible T-cell co-stimulator structurally and functionally related to CD28. Nature. 1999;397(6716):263–266. doi: 10.1038/16717. [DOI] [PubMed] [Google Scholar]
- 60.Linterman MA, et al. Roquin differentiates the specialized functions of duplicated T cell costimulatory receptor genes CD28 and ICOS. Immunity. 2009;30(2):228–241. doi: 10.1016/j.immuni.2008.12.015. [DOI] [PubMed] [Google Scholar]
- 61.Gigoux M, et al. Inducible costimulator promotes helper T-cell differentiation through phosphoinositide 3-kinase. Proc Natl Acad Sci USA. 2009;106(48):20371–20376. doi: 10.1073/pnas.0911573106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Rolf J, et al. Phosphoinositide 3-kinase activity in T cells regulates the magnitude of the germinal center reaction. J Immunol. 2010;185(7):4042–4052. doi: 10.4049/jimmunol.1001730. [DOI] [PubMed] [Google Scholar]
- 63.Wakamatsu E, Mathis D, Benoist C. Convergent and divergent effects of costimulatory molecules in conventional and regulatory CD4+ T cells. Proc Natl Acad Sci USA. 2013;110(3):1023–1028. doi: 10.1073/pnas.1220688110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Butty V, et al. Signatures of strong population differentiation shape extended haplotypes across the human CD28, CTLA4, and ICOS costimulatory genes. Proc Natl Acad Sci USA. 2007;104(2):570–575. doi: 10.1073/pnas.0610124104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Borriello F, et al. B7-1 and B7-2 have overlapping, critical roles in immunoglobulin class switching and germinal center formation. Immunity. 1997;6(3):303–313. doi: 10.1016/s1074-7613(00)80333-7. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.