Abstract
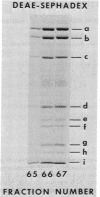
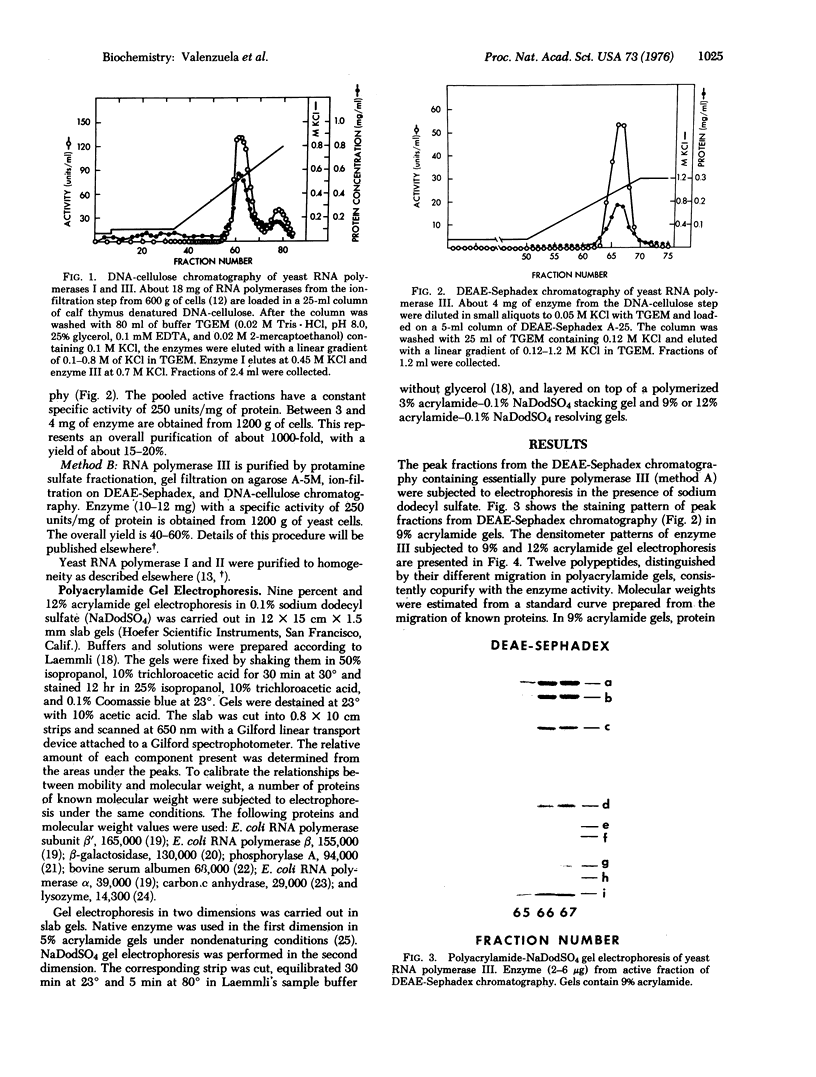
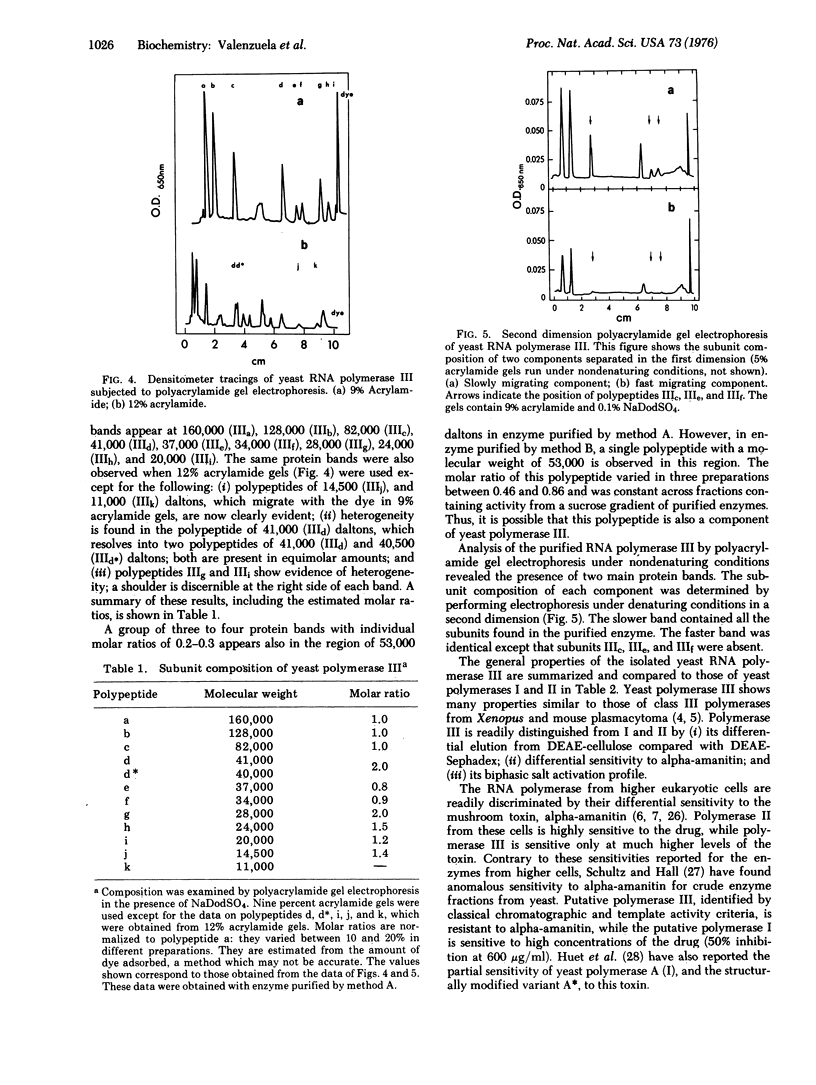
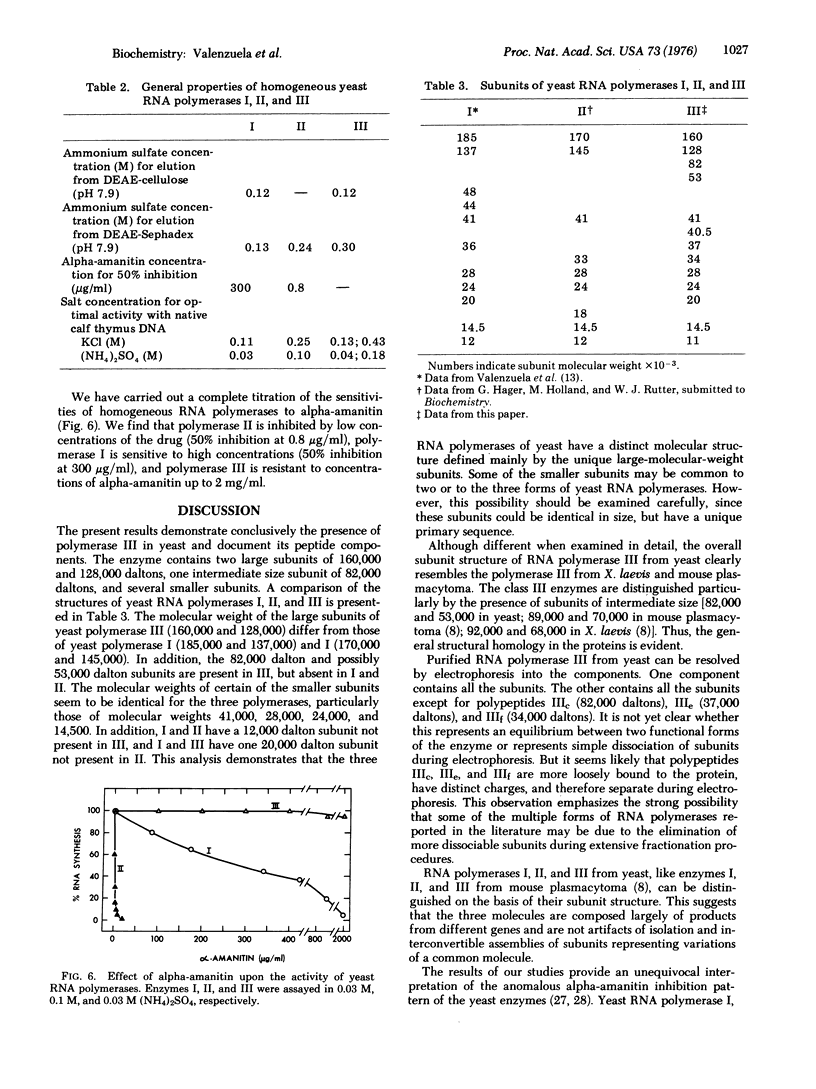
Homogeneous RNA polymerase III (RNA nucleotidyltransferase III) has been obtained from yeast. The subunit composition of the enzyme was examined by polyacrylamide gel electrophoresis in the presence of sodium dodecyl sulfate. The enzyme is composed of 12 putative subunits with molecular weights 160,000, 128,000, 82,000, 41,000, 40,500, 37,000, 34,000, 28,000, 24,000, 20,000, 14,500, and 11,000. The high-molecular-weight subunits and several of the smaller subunits of yeast RNA polymerase III are clearly different from those of enzymes I and II, indicating a distinct molecular structure. However, the molecular weights of some of the small subunits (41,000, 28,000, 24,000, and 14,500) appear to be identical to those of polymerases I and II. Thus, it is possible that the three classes of enzymes in yeast have some common subunits. As in other eukaryotes, yeast polymerase II is inhibited by relatively low concentrations of alpha-amanitin; however, contrary to what has been found in higher eukaryotes, yeast polymerase III is resistant (up to 2 mg/ml) to alpha-amanitin, while yeast polymerase I is sensitive to high concentrations of the drug (50% inhibition at 0.3 mg/ml). These results establish the existence of RNA polymerase III in yeast and provide a structural basis for the discrimination of the three functional polymerases in eukaryotes.
Full text
PDF

Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Adman R., Schultz L. D., Hall B. D. Transcription in yeast: separation and properties of multiple FNA polymerases. Proc Natl Acad Sci U S A. 1972 Jul;69(7):1702–1706. doi: 10.1073/pnas.69.7.1702. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Armstrong J. M., Myers D. V., Verpoorte J. A., Edsall J. T. Purification and properties of human erythrocyte carbonic anhydrases. J Biol Chem. 1966 Nov 10;241(21):5137–5149. [PubMed] [Google Scholar]
- Brogt T. M., Planta R. J. Characteristics of DNA-dependent RNA polymerase activity from isolated yeast nuclei. FEBS Lett. 1972 Jan 15;20(1):47–52. doi: 10.1016/0014-5793(72)80014-0. [DOI] [PubMed] [Google Scholar]
- Buhler J. M., Sentenac A., Fromageot P. Isolation, structure, and general properties of yeast ribonucleic acid polymerase A (or I). J Biol Chem. 1974 Sep 25;249(18):5963–5970. [PubMed] [Google Scholar]
- Burgess R. R. Separation and characterization of the subunits of ribonucleic acid polymerase. J Biol Chem. 1969 Nov 25;244(22):6168–6176. [PubMed] [Google Scholar]
- Chambon P. Eukaryotic nuclear RNA polymerases. Annu Rev Biochem. 1975;44:613–638. doi: 10.1146/annurev.bi.44.070175.003145. [DOI] [PubMed] [Google Scholar]
- Gornicki S. Z., Vuturo S. B., West T. V., Weaver R. F. Purification and properties of deoxyribonucleic acid-dependent ribonucleic acid polymerases from the slime mold Physarum polycephalum. J Biol Chem. 1974 Mar 25;249(6):1792–1798. [PubMed] [Google Scholar]
- Huet J., Buhler J. M., Sentenac A., Fromageot P. Dissociation of two polypeptide chains from yeast RNA polymerase A. Proc Natl Acad Sci U S A. 1975 Aug;72(8):3034–3038. doi: 10.1073/pnas.72.8.3034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Lindell T. J., Weinberg F., Morris P. W., Roeder R. G., Rutter W. J. Specific inhibition of nuclear RNA polymerase II by alpha-amanitin. Science. 1970 Oct 23;170(3956):447–449. doi: 10.1126/science.170.3956.447. [DOI] [PubMed] [Google Scholar]
- Pong S. S., Loomis W. F., Jr Multiple nuclear ribonucleic acid polymerases during development of Dictyostelium discoideum. J Biol Chem. 1973 Jun 10;248(11):3933–3939. [PubMed] [Google Scholar]
- Ponta H., Ponta U., Wintersberger E. DNA-dependent RNA polymerases from yeast. Partial characterization of three nuclear enzyme activities. FEBS Lett. 1971 Nov 1;18(2):204–208. doi: 10.1016/0014-5793(71)80445-3. [DOI] [PubMed] [Google Scholar]
- Roeder R. G. Multiple forms of deoxyribonucleic acid-dependent ribonucleic acid polymerase in Xenopus laevis. Isolation and partial characterization. J Biol Chem. 1974 Jan 10;249(1):241–248. [PubMed] [Google Scholar]
- Roeder R. G., Rutter W. J. Multiple forms of DNA-dependent RNA polymerase in eukaryotic organisms. Nature. 1969 Oct 18;224(5216):234–237. doi: 10.1038/224234a0. [DOI] [PubMed] [Google Scholar]
- Schultz L. D., Hall B. D. Transcription in yeast: alpha-amanitin sensitivity and other properties which distinguish between RNA polymerases I and III. Proc Natl Acad Sci U S A. 1976 Apr;73(4):1029–1033. doi: 10.1073/pnas.73.4.1029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schwartz L. B., Sklar V. E., Jaehning J. A., Weinmann R., Roeder R. G. Isolation and partial characterization of the multiple forms of deoxyribonucleic acid-dependent ribonucleic acid polymerase in the mouse myeloma, MOPC 315. J Biol Chem. 1974 Sep 25;249(18):5889–5897. [PubMed] [Google Scholar]
- Seery V. L., Fischer E. H., Teller D. C. A reinvestigation of the molecular weight of glycogen phosphorylase. Biochemistry. 1967 Oct;6(10):3315–3327. doi: 10.1021/bi00862a042. [DOI] [PubMed] [Google Scholar]
- Sklar V. E., Schwartz L. B., Roeder R. G. Distinct molecular structures of nuclear class I, II, and III DNA-dependent RNA polymerases. Proc Natl Acad Sci U S A. 1975 Jan;72(1):348–352. doi: 10.1073/pnas.72.1.348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ullmann A., Goldberg M. E., Perrin D., Monod J. On the determination of molecular weight of proteins and protein subunits in the presence of 6 M guanidine hydrochloride. Biochemistry. 1968 Jan;7(1):261–265. doi: 10.1021/bi00841a031. [DOI] [PubMed] [Google Scholar]
- Weber K., Osborn M. The reliability of molecular weight determinations by dodecyl sulfate-polyacrylamide gel electrophoresis. J Biol Chem. 1969 Aug 25;244(16):4406–4412. [PubMed] [Google Scholar]
- Weinmann R., Raskas H. J., Roeder R. G. Role of DNA-dependent RNA polymerases II and III in transcription of the adenovirus genome late in productive infection. Proc Natl Acad Sci U S A. 1974 Sep;71(9):3426–3439. doi: 10.1073/pnas.71.9.3426. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weinmann R., Roeder R. G. Role of DNA-dependent RNA polymerase 3 in the transcription of the tRNA and 5S RNA genes. Proc Natl Acad Sci U S A. 1974 May;71(5):1790–1794. doi: 10.1073/pnas.71.5.1790. [DOI] [PMC free article] [PubMed] [Google Scholar]