Abstract
OBJECTIVE
Gut microbiome dysbiosis is associated with numerous diseases, including type 1 diabetes. This pilot study determines how geographical location affects the microbiome of infants at high risk for type 1 diabetes in a population of homogenous HLA class II genotypes.
RESEARCH DESIGN AND METHODS
High-throughput 16S rRNA sequencing was performed on stool samples collected from 90 high-risk, nonautoimmune infants participating in The Environmental Determinants of Diabetes in the Young (TEDDY) study in the U.S., Germany, Sweden, and Finland.
RESULTS
Study site–specific patterns of gut colonization share characteristics across continents. Finland and Colorado have a significantly lower bacterial diversity, while Sweden and Washington state are dominated by Bifidobacterium in early life. Bacterial community diversity over time is significantly different by geographical location.
CONCLUSIONS
The microbiome of high-risk infants is associated with geographical location. Future studies aiming to identify the microbiome disease phenotype need to carefully consider the geographical origin of subjects.
Introduction
The Environmental Determinants of Diabetes in the Young (TEDDY) study was formed to investigate environmental factors that trigger type 1 diabetes in genetically at-risk children (1). The gut microbiome is of interest, as several studies (2–6) have shown that dysbiosis of the microbiome is associated with type 1 diabetes autoimmunity. The composition of the fecal microbiome is dependent on numerous external factors, including geographical location (7–11). This work presents the first geographical assessment of the gut microbiome in these genetically higher-risk children.
Research Design and Methods
The TEDDY study prospectively observes children at six clinical centers in Europe (Finland, Sweden, and Germany) and the U.S. (Colorado, Washington state, and Georgia/Florida) (1). A total of 1,129 stool samples from 90 children, 15 from each study site, were collected monthly starting, on average, at 151.1 days after birth (SE 5.5 days after birth) until the average last sampling at 537 days after birth (SE 4.5 days after birth). Samples were collected at home and mailed to a TEDDY repository within 72 h, with ice packs during the summer months (1,12). Fecal sample storage at room temperature for up to 72 h does not affect bacterial composition by >10% (13). Subjects were determined to have the highest risk HLA class II genotype (DR4-DQA1*030×-DQB1*0302/DR3-DQA1*0501-DQB1*0201) by genotyping of cord blood (1), but neither autoantibodies nor disease developed during the sample collection period. Clinical data were collected on gestational age, delivery mode, sex, and early feeding practices (age at first introduction to formula, and duration of exclusive and any breastfeeding), and later on diet (age at first introduction to oats, gluten, milk products, cow milk, and solid food) (1).
DNA was isolated from frozen stool samples as previously described (13). Extracted DNA was purified using the PowerClean DNA Kit (MO BIO Laboratories, Inc., Carlsbad, CA). 16S rRNA amplification, sequencing using a barcoded Illumina approach, sequence analysis, read trimming, and taxonomic classification were performed as previously described (14).
Samples with <10,000 reads and any operational taxonomic units with <50 reads in at least one sample were removed from the data set. This resulted in an average of 102,147 reads per sample (SE 1,151 reads per sample), of which on average 43.9% (SE 0.1%) were successfully classified at the genus level. The relative abundances of bacterial genera were calculated as the percentage of classified reads. Sequences that did not map to known genera were clustered to each other at 95% similarity. The original sequences were submitted to MG-RAST under project identification #3229. The bacterial diversity of each sample was determined by calculating the Shannon diversity index (SDI).
Data analysis was performed using R statistical software version 3.0.0 (15) or SAS version 9.3 (SAS Institute Inc., Cary, NC). Demographic, clinical, and dietary variables were assessed by site. Categorical variables were analyzed using Pearson χ2 test or Fisher exact test. Continuous variables were tested using the one-way ANOVA or Kruskal-Wallis test for differences in means. Generalized estimating equations for longitudinal correlated data were used to assess the association between geographical location and bacterial abundance and diversity adjusting for demographic, clinical, and dietary variables. Separate models were examined for each bacterial genus under study. A permutation test and the F statistic were used to determine whether SDI differed among the six TEDDY study sites, as previously described (16). P values <0.05 were considered significant.
Results
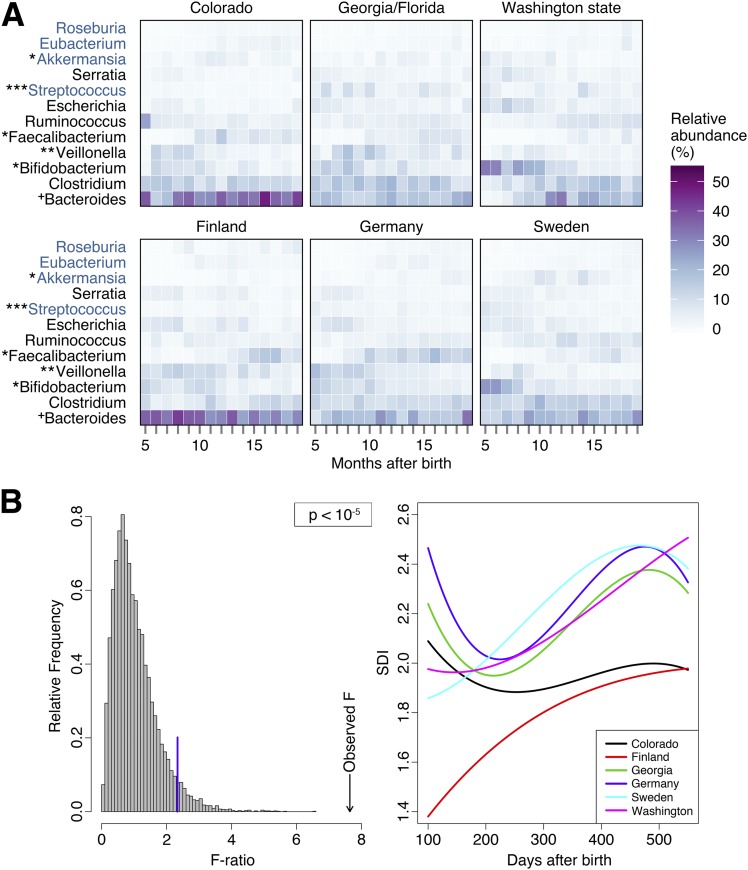
Sex (P = 0.0092) and age at first introduction to oats (P = 0.0042), gluten (P = 0.0001), and milk products (P < 0.0001) were the only clinical characteristics significantly different by geographical location. To characterize the development of the gut microbiome over time, 16S rRNA sequencing read values of bacterial genera were grouped according to age of subject (in months) at the time of sample collection (Fig. 1A). Bacteroides was the predominant genera at all sites (average abundance 22.7%, SE 0.7%). The abundance of Bifidobacterium (P = 0.0172), Veillonella (P = 0.0048), Faecalibacterium (P = 0.0122), Streptococcus (P = 0.0003), and Akkermansia (P = 0.0196) was significantly different by geographical location after adjusting for significant clinical and dietary variables. Although Bacteroides abundance was not significantly different (P = 0.0530) by site, Colorado had a significantly higher abundance than all other sites (P = 0.0126), except Finland.
Figure 1.
A: A heat map of the relative abundance of the most abundant bacterial genera shows a distinct pattern of development at each study site. 16S rRNA read values were grouped according to age of subject (in months) at the time of sample collection. If a subject had more than one sample within 1 month, the read values were averaged to prevent over-representation of a single individual. Bacterial genera denoted in black font are represented in the top 10 most abundant genera at all sites, and those denoted in blue font represent genera from the 10 most abundant at only some sites. Symbols indicate a statistically significant difference in bacterial abundance by geographical site after adjusting for age at stool collection and other significant covariates, as follows: *P < 0.05, **P < 0.01, ***P < 0.001, +P = 0.053. B: Changes in the SDI of genus-level microbial communities over time differ significantly at each site (P < 10−5). Diversity remains significantly different after adjusting for mode of delivery and age at first introduction to milk products (P = 0.0045). The left panel depicts a histogram of 10,000 F statistics obtained after randomly permuting the site labels. The blue line indicates the 95% quartile of this F statistic, and the arrow indicates the observed F statistic. The right panel depicts, for every site, a polynomial of degree 3 adjusted to the observed SDI, days after birth.
The permutation test of the SDI of bacterial genera identified at each site showed that all sites differ from each other across time (Fig. 1B). Furthermore, the difference remained significant after adjustment for delivery mode and age at first introduction to milk products (P = 0.0045). Colorado and Finland had a significantly lower SDI than all other sites (P = 0.0258). Georgia/Florida and Germany had a more diverse profile, characterized by a relative abundance of Clostridium, Bifidobacterium, and Veillonella of >8.0% each (Fig. 1A). In Sweden and Washington state, the fecal profile was dominated by Bifidobacterium until 8 and 10 months of age, respectively, and the overall abundance was significantly higher than in Colorado and Finland (P = 0.0199).
Conclusions
This study highlights the great variability in the composition and diversity of gut microbiomes among the six TEDDY study sites. So far, studies examining the association between the gut microbiome and type 1 diabetes autoimmunity have focused on small geographical regions (2–6). Country- and lifestyle-specific factors are a major player in shaping the composition of the gut community (7–11), but the extent to which they affect the microbiome of infants at high risk for type 1 diabetes was previously unknown. Our study is the first to investigate the fecal microbial profile of high-risk children across two continents and multiple countries.
Our data suggest that children at high risk for type 1 diabetes have study site–specific patterns of gut colonization showing intercontinental similarities but intracontinental differences. Geographical origin significantly associated with the diversity of bacterial communities and the relative abundance of numerous bacterial genera (Fig. 1A). Low bacterial diversity was characteristic of subjects from Finland and Colorado. Previous studies have reported similar reductions in microbiome diversity in northern European infants compared with infants from southern European countries (Sweden vs. Spain and Finland vs. Germany, respectively) (9,10). While Finland has the highest incidence of type 1 diabetes in children (17), it remains to be seen whether SDI is associated with disease incidence. Perplexing differences, not explained by clinical characteristics, exist between the neighboring countries Sweden and Finland. Compared with their Swedish neighbors, Finnish subjects had a significantly higher abundance of Bacteroides (P = 0.0508) and Veillonella (P = 0.0160), and a lower abundance of Bifidobacterium (P = 0.0199), Akkermansia (P = 0.0014), and Ruminococcus (P = 0.0248). The fecal profile of Swedish subjects was more similar to those from Washington state (both groups of subjects were dominated by Bifidobacteria at early time points), suggesting a more typical colonization pattern of the infant gut (18).
Clearly, microbiome diversity varies with geographical location, even in a population of homogenous HLA class II genotypes. Geography represents a culmination of underlying environmental and cultural factors that, on their own, are difficult to account for. The prime source of variability in these data remains unknown because significant differences in microbiome composition exist even after adjusting for numerous early-life and dietary variables. Future studies must carefully consider the impact of geographical location on the microbiome of children who are genetically at higher risk for type 1 diabetes as location may confound analyses of disease-associated microbiome states. Furthermore, whether the microbiome is causative or merely an indicator of underlying type 1 diabetes etiology, these microbiome differences suggest a geographically tailored approach to diagnostics or preventative therapies.
Supplementary Material
Article Information
Funding. This study was funded by grants U01-DK-63829, U01-DK-63861, U01-DK-63821, U01-DK-63865, U01-DK-63863, U01-DK-63836, U01-DK-63790, UC4-DK-63829, UC4-DK-63861, UC4-DK-63821, UC4-DK-63865, UC4-DK-63863, UC4-DK-63836, and UC4-DK-95300, and Contract No. HHSN267200700014C from the National Institute of Diabetes and Digestive and Kidney Diseases; the National Institute of Allergy and Infectious Diseases; the National Institute of Child Health and Human Development; the National Institute of Environmental Health Sciences; JDRF; and the Centers for Disease Control and Prevention. This work was supported in part by National Institutes of Health/National Center for Advancing Translational Sciences Clinical and Translational Science Awards to the University of Florida (UL1-TR-000064) and the University of Colorado (UL1-TR-001082).
Duality of Interest. No potential conflicts of interest relevant to this article were reported.
Author Contributions. K.M.K. participated in the study design, carried out data analysis, drafted the manuscript, and read and approved the final manuscript. A.N.A. participated in the study conception and design, carried out data analysis, helped draft the manuscript, and read and approved the final manuscript. A.G.D.-R. and J.R.F. participated in the study design, carried out data analysis, and read and approved the final manuscript. K.A.G. participated in the study design, helped to draft the manuscript, and read and approved the final manuscript. L.G.L.-N. carried out data analysis, and read and approved the final manuscript. K.V. participated in the study design and data analysis, and read and approved the final manuscript. G.C. participated in the study design and data analysis. O.S., A.G.Z., M.J.R., A.L., W.H., J.-X.S., J.P.K., B.A., D.A.S., and M.A.A. conceived of the study, participated in the study design and acquisition of data, and read and approved the final manuscript. E.W.T. conceived of the study; participated in its design, data acquisition, and data analysis; helped to draft the manuscript; and read and approved the final manuscript. E.W.T. is the guarantor of this work and, as such, had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Footnotes
A complete list of the TEDDY Study Group can be found in the Supplementary Data online.
Deceased 17 June 2012.
This article contains Supplementary Data online at http://care.diabetesjournals.org/lookup/suppl/doi:10.2337/dc14-0850/-/DC1.
References
- 1.TEDDY Study Group The Environmental Determinants of Diabetes in the Young (TEDDY) study: study design. Pediatr Diabetes 2007;8:286–298 [DOI] [PubMed] [Google Scholar]
- 2.Murri M, Leiva I, Gomez-Zumaquero JM, et al. Gut microbiota in children with type 1 diabetes differs from that in healthy children: a case-control study. BMC Med 2013;11:46. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.de Goffau MC, Luopajärvi K, Knip M, et al. Fecal microbiota composition differs between children with β-cell autoimmunity and those without. Diabetes 2013;62:1238–1244 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Giongo A, Gano KA, Crabb DB, et al. Toward defining the autoimmune microbiome for type 1 diabetes. ISME J 2011;5:82–91 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Brown CT, Davis-Richardson AG, Giongo A, et al. Gut microbiome metagenomics analysis suggests a functional model for the development of autoimmunity for type 1 diabetes. PLoS One 2011;6:e25792. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Endesfelder D, zu Castell W, Ardissone A, et al. Compromised gut microbiota networks in children with anti-islet cell autoimmunity. Diabetes 2014;63:2006–2014 [DOI] [PubMed] [Google Scholar]
- 7.Yatsunenko T, Rey FE, Manary MJ, et al. Human gut microbiome viewed across age and geography. Nature 2012;486:222–227 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Echarri PP, Graciá CM, Berruezo GR, et al. Assessment of intestinal microbiota of full-term breast-fed infants from two different geographical locations. Early Hum Dev 2011;87:511–513 [DOI] [PubMed] [Google Scholar]
- 9.Fallani M, Young D, Scott J, et al.; Other Members of the INFABIO Team . Intestinal microbiota of 6-week-old infants across Europe: geographic influence beyond delivery mode, breast-feeding, and antibiotics. J Pediatr Gastroenterol Nutr 2010;51:77–84 [DOI] [PubMed] [Google Scholar]
- 10.Grześkowiak Ł, Grönlund MM, Beckmann C, Salminen S, von Berg A, Isolauri E. The impact of perinatal probiotic intervention on gut microbiota: double-blind placebo-controlled trials in Finland and Germany. Anaerobe 2012;18:7–13 [DOI] [PubMed] [Google Scholar]
- 11.De Filippo C, Cavalieri D, Di Paola M, et al. Impact of diet in shaping gut microbiota revealed by a comparative study in children from Europe and rural Africa. Proc Natl Acad Sci U S A 2010;107:14691–14696 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Vehik K, Fiske SW, Logan CA, et al.; TEDDY Study Group . Methods, quality control and specimen management in an international multicentre investigation of type 1 diabetes: TEDDY. Diabetes Metab Res Rev 2013;29:557–567 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Roesch LF, Casella G, Simell O, et al. Influence of fecal sample storage on bacterial community diversity. Open Microbiol J 2009;3:40–46 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Fagen JR, Giongo A, Brown CT, Davis-Richardson AG, Gano KA, Triplett EW. Characterization of the relative abundance of the citrus pathogen Ca. Liberibacter asiaticus in the microbiome of its insect vector, Diaphorina citri, using high throughput 16S rRNA sequencing. Open Microbiol J 2012;6:29–33 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.R Core Team R: a language and environment for statistical computing [Internet], 2014. Version 3.0.0. Vienna, Austria, R Foundation for Statistical Computing. Available from http://www.R-project.org. Accessed 5 April 2013
- 16.León-Novelo L, Kemppainen KM, Ardissone A, et al.; TEDDY Study Group . Two applications of permutation tests in biostatistics. Bol Soc Mat Mex 2013;19:255–266 [PMC free article] [PubMed] [Google Scholar]
- 17.Borchers AT, Uibo R, Gershwin ME. The geoepidemiology of type 1 diabetes. Autoimmun Rev 2010;9:A355–A365 [DOI] [PubMed] [Google Scholar]
- 18.Scholtens PA, Oozeer R, Martin R, Amor KB, Knol J. The early settlers: intestinal microbiology in early life. Annu Rev Food Sci Technol 2012;3:425–447 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.