Abstract
Blood vessel polarization in the apical-basal axis is important for directed secretion of proteins and lumen formation; yet, when and how polarization occurs in the context of angiogenic sprouting is not well understood. Here, we describe a novel topology for endothelial cells at the tip of angiogenic sprouts in several mammalian vascular beds. Two cells that extend filopodia and have significant overlap in space and time were present at vessel tips, both in vitro and in vivo. The cell overlap is more extensive than predicted for tip cell switching, and it sets up a longitudinal cell-cell border that is a site of apical polarization and lumen formation, presumably via a cord-hollowing mechanism. The extent of cell overlap at the tip is reduced in mice lacking aPKCζ, and this is accompanied by reduced distal extension of both the apical border and patent lumens. Thus, at least two polarized cells occupy the distal tip of blood vessel sprouts, and topology, polarization and lumenization along the longitudinal border of these cells are influenced by aPKCζ.
Keywords: Blood vessel polarity, Apical-basal polarity, Lumen formation, aPKCζ, Vessel sprout topology
INTRODUCTION
Developmental blood vessel formation initiates, via vasculogenesis, the differentiation and coalescence of endothelial cells to form cords and tubes. Complex networks subsequently form via angiogenesis, whereby endothelial cells sprout from parent vessels and anastomose to form new conduits (Carmeliet and Jain, 2011; Risau, 1997). Sprouting angiogenesis is thought to involve a single migratory cell at the tip that guides the sprout, along with a cell behind the tip cell that assumes a ‘stalk cell’ phenotype (Gerhardt et al., 2003). The distinction between tip and stalk cells results from cross-talk between VEGF-A and Notch signaling. In this binary paradigm, the tip cell experiences high VEGF and low Notch signaling, and high VEGF signaling elevates activity of the membrane-tethered Notch ligand Dll4. The stalk cell responds to tip cell Dll4 with elevated Notch signaling that downregulates VEGF signaling (Hellström et al., 2007; Siekmann and Lawson, 2007). As vessel networks expand, stalk cells move to the front and become new tip cells, revealing dynamic changes in phenotypes rather than fixed fates for endothelial cells in blood vessel sprouts (Arima et al., 2011; Jakobsson et al., 2010). By contrast, migration in the Drosophila trachea initially involves a group of cells with no clear tip cell, and cell intercalations allow a tip cell to emerge (Affolter and Caussinus, 2008; Ghabrial et al., 2003).
As blood vessels form, polarity in the apical (luminal) to basal (abluminal) axis is established (Lee and Bautch, 2011; Lizama and Zovein, 2013). This polarity is evident in large vessels and in capillaries, and is essential for lumen formation (Horvat et al., 1986; Iruela-Arispe and Davis, 2009). At early stages of mouse dorsal aorta development, apical-basal polarization leads to cord hollowing, whereby cells change shape to form a lumen (Strilić et al., 2009). Genetic loss of β1 integrin, which localizes basally, causes polarization and lumenization defects in arterioles (Zovein et al., 2010), and knockdown of the junctional marker VE-cadherin perturbs apical-basal polarity in vitro and in vivo (Lampugnani et al., 2010). However, where and how apical-basal polarity is first established during angiogenesis remains unclear.
Apical-basal polarity and lumen formation are linked in other epithelia (Datta et al., 2011), but little is known about their relationship during angiogenesis. Direct visualization of cell-cell borders in zebrafish segmental arteries revealed intercellular lumens, suggesting that both unicellular and multicellular tubes form; subsequent work showed that vessel anastomosis in zebrafish involves invagination to form a unicellular tube that is followed by cell rearrangements to form a multicellular tube (Blum et al., 2008; Herwig et al., 2011; Lenard et al., 2013). The zebrafish studies emphasize the importance of understanding the topology of the relevant cells in a sprout, yet the topology of endothelial cells in angiogenic sprouts is not well understood.
Here, we describe a novel endothelial cell topology in sprouts, with consistent overlap of at least two filopodia-extending cells at the tip. This topology creates a longitudinal cell-cell border that is a site of apical polarization and lumen formation in the sprouts. Genetic loss of aPKCζ affects vessel sprout tip topology and perturbs apical polarization and lumenogenesis, suggesting that these sprout tip properties are linked and might have functional significance.
RESULTS AND DISCUSSION
Blood vessel sprout tips are composed of two endothelial cells in close register
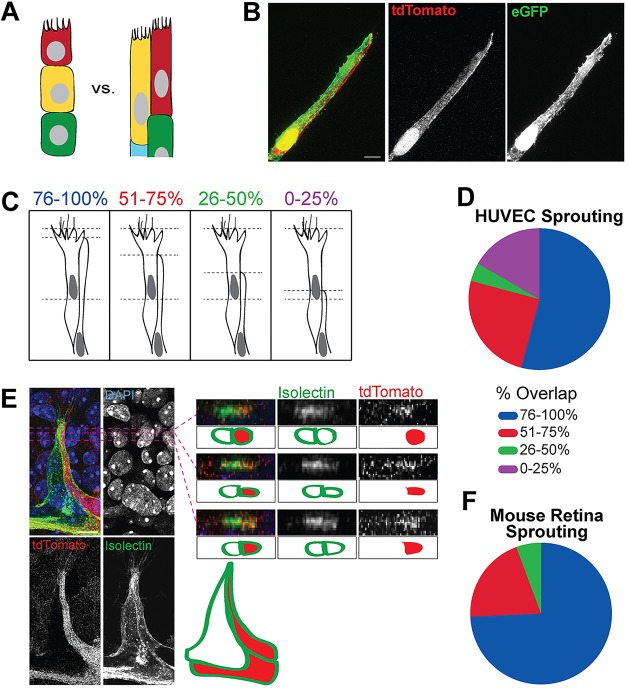
The widely accepted model of angiogenic sprouting describes a single cell, the tip cell, at the leading edge of vessel sprouts. By contrast, using several mammalian angiogenesis models, we observed that sprouts often had multiple overlapping cells at the tip (Fig. 1A,B). To determine the frequency of overlapping endothelial cells at sprout tips, we used a sprouting assay and human umbilical vein endothelial cells (HUVECs) (Nakatsu et al., 2007). HUVECs expressing either eGFP (green) or tdTomato (red) were mixed prior to embedding to create mosaic spouts (Fig. 1B). We measured overlap by calculating the percentage of overlap of the trailing endothelial cell (minor tip cell) with the leading endothelial cell (major tip cell), starting from the proximal edge of the major tip cell nucleus (Fig. 1C). The majority of HUVEC sprouts had two endothelial cells with significant overlap at the tip, with 77% of sprouts having >50% overlap (n=24; Fig. 1D).
Fig. 1.
Multiple endothelial cells occupy the tip position in blood vessel sprouts. (A) Common inferred endothelial cell topology in sprouts (left), and the observed topology of overlapping cells (right). (B) Sprout consisting of HUVECs infected with either GFP-expressing lentivirus or tdTomato-expressing lentivirus and mixed prior to assay. (C) Schematic of percentage cell overlap measurements. (D) Frequency of topologies with indicated percentage of cell overlap in HUVEC sprouting assay. (E) Left, mosaic vessel sprout from P6 mouse retina of Ub-CreER×tdTomatoflox/+ cross, stained with isolectin (green, endothelial marker) and DAPI (blue, nuclear marker). Right, orthogonal views and diagrams show overlap of endothelial cells near the tip. (F) Frequency of topologies with indicated percentage of tip cell overlap in mouse retina.
To determine whether this topology is present in vivo, we examined sprouts in postnatal mouse retinal vessels. We generated mosaic vessels using mice carrying a ubiquitously expressed inducible Cre driver and a tdTomato excision reporter. The minor tip cell overlapped the major tip cell in retinal vessels, with >75% of the major tip cell length overlapped in 75% of sprouts (Fig. 1E,F; n=35). To determine whether this was a consistent sprout topology, we analyzed blood vessels from the embryonic back. Mosaic vessels were generated using an endothelial cell-selective Cre driver [VEcad-Cre (Alva et al., 2006)] and the excision reporter. Embryonic back sprouts had overlapping endothelial cells, with 83% of sprouts having >50% overlap (supplementary material Fig. S1). Taken together, these results indicate that two endothelial cells consistently overlap at the tip of mammalian angiogenic sprouts in vitro and in vivo.
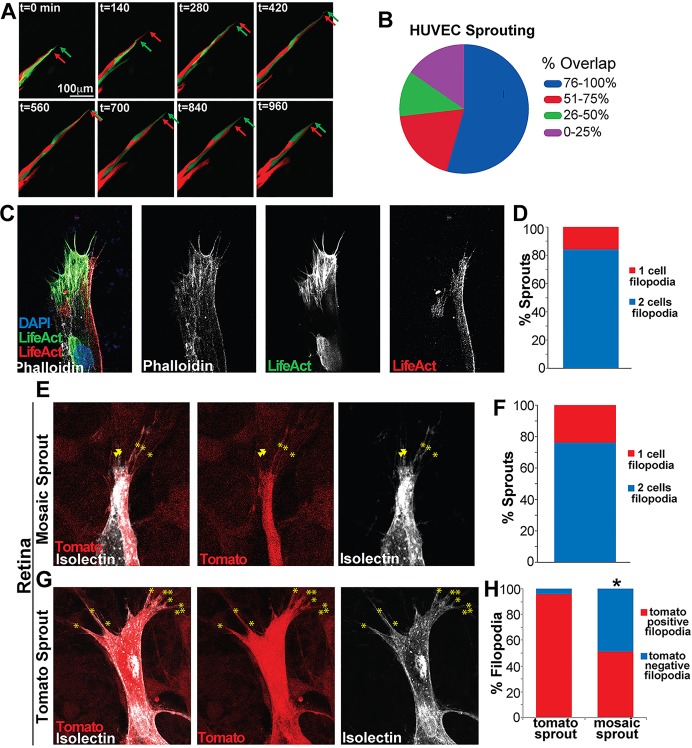
Tip cells switch place with a stalk cell every 4-7 h, suggesting that tip cells and stalk cells overlap for some time as a new cell ‘overtakes’ the current tip cell (Arima et al., 2011; Jakobsson et al., 2010). We hypothesized that the overlap needed for switching did not explain the prevalence of observed endothelial cell overlap. To further examine behaviors at the tip, we live-imaged labeled HUVEC sprouts (Fig. 2A). Consistent with previous studies, we observed tip cell switching approximately every 5.5 h, based on positions of endothelial cell nuclei (not shown) (Arima et al., 2011; Jakobsson et al., 2010). By contrast, tip cell overlap was consistent (n=12 movies, 100 time points each; Fig. 2B), and overlap was maintained continuously through multiple instances of nuclear switching. These results demonstrate that endothelial cell overlap at sprout tips is a major topology and is not confined to periods of tip cell switching. Because endothelial cell overlap did not appear linked to tip cell switching, we analyzed the relationship of the leading edge of distal endothelial cells relative to their nuclei. Surprisingly, the movement of a tip cell's leading edge did not correlate with its nuclear movement (r=0.08, n=6 cells, 100 time points; supplementary material Fig. S2A). These results demonstrate that the leading edges of endothelial cells at the tip stay in close proximity and maintain overlap, and that this topology is largely independent of the relative nuclear positions of the relevant cells.
Fig. 2.
Endothelial cell dynamics lead to tip cell overlap. (A) Time-lapse imaging of HUVEC sprout. Red and green arrows, tips of tdTomato- and GFP-expressing cells, respectively. (B) Frequency of topologies with indicated percentage of tip cell overlap in live-imaged HUVEC sprouts. (C) Mosaic HUVEC sprout with individual cells expressing either LifeAct-GFP or LifeAct-RFP. (D) Percentage of mosaic sprouts with filopodia of one color (red) or two colors (blue), n=20 sprouts. (E,G) Vessel sprouts from P6 mouse retina of Ub-CreER×tdTomatoflox/+ cross, stained with isolectin (white, endothelial marker). Asterisks, tomato-positive filopodia; arrowheads, tomato-negative filopodia. (E) Sprout with labeled and unlabeled cells at tip. (F) Percentage of sprouts with only red filopodia (red) or red and negative filopodia (blue), n=20 sprouts. (G) Sprout with only labeled cells at tip. (H) Percentage of filopodia that are tomato-positive (red) or tomato-negative (blue) in sprouts of indicated compositions. Statistical comparisons using χ2. *P≤0.001.
To maintain cellular overlap, endothelial cells at sprout tips must either sustain approximately equal forward velocities and/or alternate velocities at high rates. We assessed velocities of the major and minor tip cell relative to each other and found that the cells overall maintained similar relative velocities, while alternating maximal velocity at approximately half of the measured time points (12 movies, 100 time points each; supplementary material Fig. S2B,C). These results suggest that both similar velocities and small alternations in velocity maintain significant overlap at sprout tips.
A hallmark of tip cells is the formation of actin-rich filopodia that extend from the cell body. To determine whether both cells at the tip extended filopodia, we infected HUVECs with the actin-binding protein LifeAct, labeled with either GFP or RFP, and mixed prior to sprouting. Mosaic sprout analysis showed that over 80% of mosaic sprout tips had filopodia of two colors, indicating that both cells at the tip extended filopodia (n=20 sprouts; Fig. 2C,D). Counts showed that the cell further ahead, or major tip cell, had on average more filopodia than its neighbor (supplementary material Fig. S2D). We next examined mosaic retinas produced as described and found that 76% of mosaic sprouts had two cells contributing filopodia (n=20 sprouts; Fig. 2E,F). Control sprouts with all tip cells labeled with tdTomato showed >95% labeling of filopodia, indicating that the 50% labeling seen in mosaic sprouts reflected contributions of multiple cells (Fig. 2G,H). Finally, to study the status of Notch signaling in retinal sprout tips, we examined expression of the Notch ligand Dll4. We documented sprout tips in which cells with a longitudinal border had differential Dll4 staining (for example, see supplementary material Fig. S2E), suggesting that the two tip cells do not have equivalent Notch status.
Endothelial cells polarize along a longitudinal border at blood vessel sprout tips
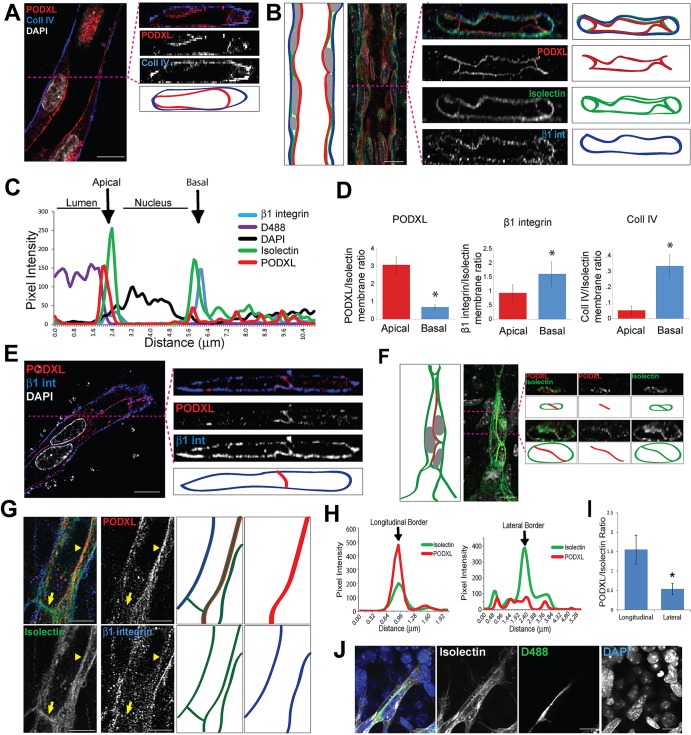
The observation that sprouting vessels have two extensively overlapping cells at the tip, rather than a single tip cell, led us to investigate apical-basal polarity at sprout tips. Apical surfaces are established at the border between two cells in the dorsal aorta (Strilić et al., 2009), so we hypothesized that a longitudinal border created by cell overlap in angiogenic sprouts is associated with an apical surface. First, we developed a quantitative method for analyzing vessel polarity using mature vessels. We stained lumenized HUVEC sprouts with the apical marker podocalyxin (PODXL/Podxl) and the basal marker collagen IV (Coll IV; Col4a – Mouse Genome Informatics) and found them localized to the apical (luminal) or basal (abluminal) surface (Fig. 3A), consistent with a previous study (Lampugnani et al., 2010). Likewise, mouse postnatal retinal vessels had PODXL (apical) and β1 integrin (basal) properly localized in lumenized vessels behind the vascular front (Fig. 3B). To quantitatively examine apical-basal polarization, we generated line plots of pixel intensity, using isolectin to define both apical and basal surfaces (Fig. 3C). PODXL pixel intensity at the apical surface is significantly higher than at the basal surface, whereas collagen IV and β1 integrin have significantly higher pixel intensities at the basal surface (Fig. 3D).
Fig. 3.
Polarity markers localize in sprout tips and define a longitudinal apical border. (A) Lumenized HUVEC sprout stained for collagen IV (blue, basal), podocalyxin (PODXL, red, apical), and DAPI (white, nuclear). Left, en face view of compressed z-stacks; right, orthogonal z-planes through horizontal line after deconvolution analysis. (B) Lumenized retinal vein behind vascular front stained for isolectin (green, endothelial), PODXL (red, apical), β1 integrin (blue, basal) and DAPI (white, nuclear). Left, en face view of compressed z-stacks; right, orthogonal z-planes through horizontal line after deconvolution analysis. (C) Line-scan of marker intensity from the luminal space of the vessel outwards; isolectin (green, endothelial), PODXL (red, apical), β1 integrin (blue, basal), dextran perfusion (D488, purple, patent lumen), and DAPI (black, nuclear). Black arrows, intensity peaks corresponding to apical and basal areas. (D) Average ratio of basal to apical pixel intensity for PODXL (n=26), collagen IV (n=12) and β1 integrin (n=200), normalized to isolectin. Statistical comparisons versus Isolectin by Student's t-test±s.e.m. *P≤0.05. (E) Unlumenized HUVEC sprout tip, stained with β1 integrin (blue, basal), PODXL (red, apical) and DAPI (white, nuclear). Note polarization near the sprout tip. Left, en face view of compressed z-stacks; right, orthogonal z-planes through horizontal line after deconvolution analysis. (F) Retinal sprout tip stained with PODXL (red, apical), isolectin (green, endothelial) and DAPI (white, nuclear). Left, en face view of compressed z-stacks; right, orthogonal z-planes through horizontal lines after deconvolution analysis. Note PODXL localization at the longitudinal cell-cell border. (G) Retinal sprout tip stained with PODXL (red, apical), Isolectin (green, endothelial) and β1 integrin (blue, basal). Arrow, lateral cell-cell border; arrowhead, longitudinal cell-cell border. (H) Line scan of marker intensity at the indicated borders. Black arrows, intensity peaks for isolectin. (I) Relative PODXL intensity in longitudinal versus lateral borders in retinal sprouts. Error bars±s.e.m., Student's t-test. *P≤0.05. (J) Retinal sprout perfused with FITC-D488 (green), then stained with isolectin (white, endothelial) and DAPI (blue, nuclear). Note perfusion along longitudinal border. (A,B,E-G) Diagrams show approximate marker localization. Scale bars: 10 µm in A,B,E,F,J; 5 µm in G.
We next asked where along the proximal-distal sprout axis apical-basal polarity is present. We analyzed HUVEC sprouts in distal areas without detectable lumens and found the apical marker PODXL at the longitudinal cell-cell border between the two distal-most cells and the basal marker β1 integrin localized to the non-apical surface near the sprout tip (Fig. 3E). This indicates that cells are polarized in the apical-basal plane at or near the sprout tip, and that the polarization plane is perpendicular to the longitudinal border. To determine whether sprouts were polarized comparably in vivo, we analyzed mouse retinal vessels and also found PODXL localized to the longitudinal cell-cell border at the sprout tip (Fig. 3F). To determine whether PODXL localization was specific to the longitudinal cell-cell border, we analyzed retinal vessel sprout tips in which a lateral border, defined as perpendicular to the sprout axis, was also evident (Fig. 3G). Line scan analysis revealed significantly higher levels of PODXL at the longitudinal border relative to the lateral border (Fig. 3H,I). These results show that the longitudinal cell-cell border in sprout tips at the vascular front is a primary location of apical polarization.
As lumens form between two cells in zebrafish intersegmental vessels and in the mouse aorta (Herwig et al., 2011; Strilić et al., 2009), we hypothesized that endothelial cell overlap within angiogenic sprouts allows for lumen formation along the apically polarized longitudinal border. When FITC-conjugated dextran (D488) was perfused to visualize patent lumens in retinal vessels, the signal localized to the longitudinal border, almost to the tip of the sprout (Fig. 3J). Thus, lumens are present between two overlapping endothelial cells in sprouts, suggesting that longitudinal borders promote lumen formation.
Loss of aPKCζ disrupts endothelial cell overlap and lumen formation in vessel sprouts
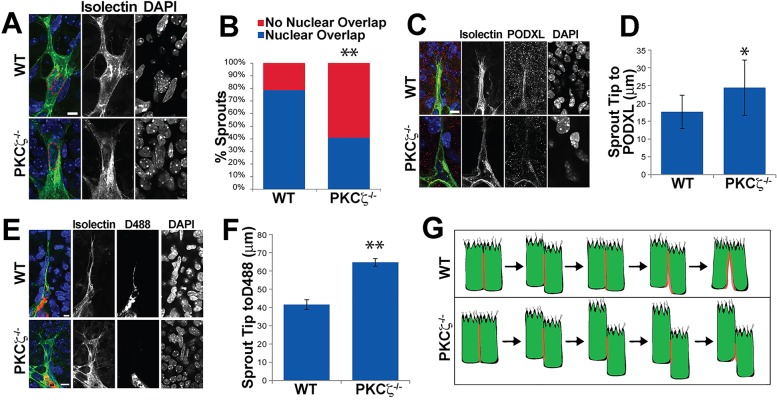
Our finding that the longitudinal border of endothelial cells in sprout tips is apically polarized and has lumens led us to investigate the effects of disrupting a putative molecular regulator of polarization on each parameter. Atypical protein kinase C (aPKC) is required for apical polarity and lumen formation in other epithelia (Bryant et al., 2010; Horne-Badovinac et al., 2001; Whyte et al., 2010). Mammals have two PKC isoforms, aPKCλ (Prkci – Mouse Genome Informatics) and aPKCζ (Prkcz – Mouse Genome Informatics) (Ono et al., 1988; Selbie et al., 1993). aPKCζ knockdown reduces lumen formation in vitro (Koh et al., 2008) and aPKCλ is involved in spatial regulation of VEGF signaling in post-natal retinal vessels (Nakayama et al., 2013). We examined the effects of genetic loss of aPKCζ (Leitges et al., 2001) on polarization and lumenization of post-natal retinal vessels. Measurements of overlap of the two distal-most nuclei, which correlate with cellular overlap (supplementary material Fig. S3A), revealed that aPKCζ−/− retinas had significantly fewer overlapping nuclei compared with wild-type littermate controls (aPKCζ+/+) (Fig. 4A,B). The distance from the sprout tip to the first detected PODXL staining was significantly increased in aPKCζ−/− retinas relative to controls, suggesting that sprout tip apical polarization is compromised in mutant retinas (Fig. 4C,D). Perfusion labeling showed that the distance from sprout tip to labeled lumen was significantly higher in aPKCζ−/− retinas compared with controls (Fig. 4E,F), showing a defect in lumen formation in mutant vessels. By contrast, overall apical-basal polarity was not disrupted (supplementary material Fig. S3B-D). Taken together, these results link endothelial cell overlap at the tip, apical polarization and lumen formation, and suggest a role for aPKCζ in these aspects of vascular development.
Fig. 4.
Loss of aPKCζ disrupts endothelial cell overlap and lumen formation. (A,C,E) Sprouts of indicated genotypes stained for isolectin (green, endothelial cells), DAPI (blue, nuclear), and PODXL (red, apical) or D488 (red, perfusion). (B) Relative amount of nuclear overlap in sprout tips of indicated genotypes. Statistical comparisons using χ2. **P≤0.01. (D,F) Distance from sprout tip to PODXL (D) or to D488 (F). Error bars±s.e.m., Student's t-test. *P≤0.05; **P≤0.01. (G) Model showing proposed sprout tip topology in different genetic backgrounds, and proposed relationship between topology, polarization and lumen formation.
Conclusions
This work shows that angiogenesis involves the intimate association of at least two filopodia-extending endothelial cells at sprout tips of mammalian vessels, in a HUVEC sprouting assay, in the embryonic back and in the postnatal retina. This association is maintained by similar velocities and by alternation of maximal velocities, and it is associated with a longitudinal apical border and lumens (Fig. 4G). Genetic loss of aPKCζ results in small but significant changes in cell overlap, polarization and lumen location, suggesting that these processes influence each other and receive input from aPKC. These surprising results suggest that the paradigm of a single endothelial cell at the sprout tip requires revision, and this revised model has several implications. Our model implies a cord-hollowing mechanism of lumen formation at the tip of angiogenic sprouts. It also suggests that the apical-basal polarity axis is initially set up perpendicular to the sprout axis, which is its ultimate position. The model also indicates that the two endothelial cells at the tip have a complex, or interchanging, relationship in terms of Notch signaling, which was modeled on the basis of a single tip cell and a binary interaction with the stalk. The finding that two filopodia-extending endothelial cells occupy the tip suggests a non-binary Notch signaling scenario between tip and stalk cells, whereas differential Notch ligand expression and filopodia numbers in the tip cells suggest cross-talk between the cells at the tip of vascular sprouts.
MATERIALS AND METHODS
HUVEC sprouting angiogenesis assay
HUVECs were maintained per distributor instructions (Lonza) and prepared for sprouting assay and subsequent antibody staining as described (Nakatsu et al., 2007). See supplementary material Table S1 for a list of antibodies used. For mosaic experiments, HUVECs were infected with cytoplasmic eGFP or cytoplasmic tdTomato, or with LifeAct-GFP or LifeAct-RFP lentivirus 2-3 d prior to the assay, as described.
Mouse strains and tissue preparations
All experiments involving animals were performed with approval of the University of North Carolina, Chapel Hill Institutional Animal Care and Use Committee. For mosaically labeled postnatal retinal vessels, [Tg(UBC-cre/ERT2), JAX #007001] mice that express Cre ubiquitously and inducibly were mated with R26R tdTomato [Gt(ROSA)26Sortm14(CAG-tdTomato)Hze, JAX #007914] reporter mice. Tamoxifen was injected at a dose of 0.25 mg/ml in vehicle at P3, with retina harvest at P6. Retinas were fixed, processed and stained with isolectin GS-IB4 conjugated to Alexa Fluor 647 (Molecular Probes, #132450; 1:100) as previously described (Gerhardt et al., 2003). See supplementary material Table S1 for a list of antibodies used. Dextran perfusion labeling was performed as described (Gerhardt et al., 2003). For mosaically labeled embryonic back vessels, VECad-Cre mice [Tg(Cdh5-cre)7Mlia, JAX #006137] (Alva et al., 2006) were mated to tdTomato reporter mice. Embryos were dissected at E12.5 and fixed in 4% PFA overnight at 4 C. The embryonic skin layer was dissected from the back just dorsal to the somites, cranially from the cervical vertebrae through the caudal tail vertebrae. Tissue was blocked in 1% Triton X-100 and 3% goat serum in PBS overnight at 4 C, washed 3×5 min in PBS, then incubated in rat anti-mouse CD31 (BD Pharmingen, #553370; 1:100) overnight at 4 C. After 4×5 min wash in PBS, tissues were incubated in donkey anti-rat Alexa Fluor 488 (Invitrogen, A-21208; 1:500) overnight at 4°C. Tissue was washed 5×5 min in PBS, flattened and mounted on slides in Fluoromount (Southern Biotech) with coverslips.
Image acquisition and analysis
Fixed images were taken using a Leica DMI 6000B confocal microscope and z-stacks were processed with deconvolution software (Huygens Essential Software v. 3.5). Apical and basal membranes were determined in orthogonal views using line plot analysis of polarity markers relative to isolectin. Borders were defined as lateral or longitudinal borders if they were within 30 degrees of perpendicular or parallel to the sprout axis, respectively. Live imaging was performed using an Olympus FV10 microscope or an Olympus VivaView Incubator Fluorescence Microscope. Images were acquired every 10 min for 16-20 h.
Supplementary Material
Acknowledgements
We thank members of the Bautch lab for productive discussions, Kelsey Harvey for technical support and Jessica Heinz for embryo preparations.
Footnotes
Competing interests
The authors declare no competing financial interests.
Author contributions
J.C.P. helped design experiments, performed experiments, analyzed and interpreted data and contributed to writing the manuscript. C.E.W. helped perform some experiments. M.L. provided a key reagent. V.L.B. conceptualized and designed experiments, interpreted data and wrote and edited the manuscript.
Funding
This work was supported by the National Institutes of Health [R01 HL43174, HL86564 and HL116719 to V.L.B.]. Deposited in PMC for release after 12 months.
Supplementary material
Supplementary material available online at http://dev.biologists.org/lookup/suppl/doi:10.1242/dev.110296/-/DC1
References
- Affolter M. and Caussinus E. (2008). Tracheal branching morphogenesis in Drosophila: new insights into cell behaviour and organ architecture. Development 135, 2055-2064 10.1242/dev.014498 [DOI] [PubMed] [Google Scholar]
- Alva J. A., Zovein A. C., Monvoisin A., Murphy T., Salazar A., Harvey N. L., Carmeliet P. and Iruela-Arispe M. L. (2006). VE-Cadherin-Cre-recombinase transgenic mouse: a tool for lineage analysis and gene deletion in endothelial cells. Dev. Dyn. 235, 759-767 10.1002/dvdy.20643 [DOI] [PubMed] [Google Scholar]
- Arima S., Nishiyama K., Ko T., Arima Y., Hakozaki Y., Sugihara K., Koseki H., Uchijima Y., Kurihara Y. and Kurihara H. (2011). Angiogenic morphogenesis driven by dynamic and heterogeneous collective endothelial cell movement. Development 138, 4763-4776 10.1242/dev.068023 [DOI] [PubMed] [Google Scholar]
- Blum Y., Belting H.-G., Ellertsdottir E., Herwig L., Lüders F. and Affolter M. (2008). Complex cell rearrangements during intersegmental vessel sprouting and vessel fusion in the zebrafish embryo. Dev. Biol. 316, 312-322 10.1016/j.ydbio.2008.01.038 [DOI] [PubMed] [Google Scholar]
- Bryant D. M., Datta A., Rodríguez-Fraticilli A. E., Peränen J., Martín-Belmonte F. and Mostov K. E. (2010). A molecular network for de novo generation of the apical surface and lumen. Nat. Cell Biol. 12, 1035-1045 10.1038/ncb2106 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carmeliet P. and Jain R. K. (2011). Principles and mechanisms of vessel normalization for cancer and other angiogenic diseases. Nat. Rev. Drug Discov. 10, 417-427 10.1038/nrd3455 [DOI] [PubMed] [Google Scholar]
- Datta A., Bryant D. M. and Mostov K. E. (2011). Molecular regulation of lumen morphogenesis. Curr. Biol. 21, R126-R136 10.1016/j.cub.2010.12.003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gerhardt H., Golding M., Fruttiger M., Ruhrberg C., Lundkvist A., Abramsson A., Jeltsch M., Mitchell C., Alitalo K., Shima D. et al. (2003). VEGF guides angiogenic sprouting utilizing endothelial tip cell filopodia. J. Cell Biol. 161, 1163-1177 10.1083/jcb.200302047 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ghabrial A., Luschnig S., Metzstein M. M. and Krasnow M. A. (2003). Branching morphogenesis of the drosophila tracheal system. Ann. Rev. Cell Dev. Biol. 19, 623-647 10.1146/annurev.cellbio.19.031403.160043 [DOI] [PubMed] [Google Scholar]
- Hellström M., Phng L.-K., Hofmann J. J., Wallgard E., Coultas L., Lindblom P., Alva J., Nilsson A.-K., Karlsson L., Gaiano N. et al. (2007). Dll4 signalling through Notch1 regulates formation of tip cells during angiogenesis. Nature 445, 776-780 10.1038/nature05571 [DOI] [PubMed] [Google Scholar]
- Herwig L., Blum Y., Krudewig A., Ellertsdottir E., Lenard A., Belting H.-G. and Affolter M. (2011). Distinct cellular mechanisms of blood vessel fusion in the zebrafish embryo. Curr. Biol. 21, 1942-1948 10.1016/j.cub.2011.10.016 [DOI] [PubMed] [Google Scholar]
- Horne-Badovinac S., Lin D., Waldron S., Schwarz M., Mbamalu G., Pawson T., Jan Y.-N., Stainier D. Y. R. and Abdelilah-Seyfried S. (2001). Positional cloning of heart and soul reveals multiple roles for PKCλ in zebrafish organogenesis. Curr. Biol. 11, 1492-1502 10.1016/S0960-9822(01)00458-4 [DOI] [PubMed] [Google Scholar]
- Horvat R., Hovorka A., Dekan G., Poczewski H. and Kerjaschki D. (1986). Endothelial cell membranes contain podocalyxin–the major sialoprotein of visceral glomerular epithelial cells. J. Cell Biol. 102, 484-491 10.1083/jcb.102.2.484 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Iruela-Arispe M. L. and Davis G. E. (2009). Cellular and molecular mechanisms of vascular lumen formation. Dev. Cell 16, 222-231 10.1016/j.devcel.2009.01.013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jakobsson L., Franco C. A., Bentley K., Collins R. T., Ponsioen B., Aspalter I. M., Rosewell I., Busse M., Thurston G., Medvinsky A. et al. (2010). Endothelial cells dynamically compete for the tip cell position during angiogenic sprouting. Nat. Cell Biol. 12, 943-953 10.1038/ncb2103 [DOI] [PubMed] [Google Scholar]
- Koh W., Mahan R. D. and Davis G. E. (2008). Cdc42- and Rac1-mediated endothelial lumen formation requires Pak2, Pak4 and Par3, and PKC-dependent signaling. J. Cell Sci. 121, 989-1001 10.1242/jcs.020693 [DOI] [PubMed] [Google Scholar]
- Lampugnani M. G., Orsenigo F., Rudini N., Maddaluno L., Boulday G., Chapon F. and Dejana E. (2010). CCM1 regulates vascular-lumen organization by inducing endothelial polarity. J. Cell Sci. 123, 1073-1080 10.1242/jcs.059329 [DOI] [PubMed] [Google Scholar]
- Lee C. Y. and Bautch V. L. (2011). Ups and downs of guided vessel sprouting: the role of polarity. Physiology 26, 326-333 10.1152/physiol.00018.2011 [DOI] [PubMed] [Google Scholar]
- Leitges M., Sanz L., Martin P., Duran A., Braun U., García J. F., Camacho F., Diaz-Meco M. T., Rennert P. D. and Moscat J. (2001). Targeted disruption of the zetaPKC gene results in the impairment of the NF-kappaB pathway. Mol. Cell 8, 771-780 10.1016/S1097-2765(01)00361-6 [DOI] [PubMed] [Google Scholar]
- Lenard A., Ellertsdottir E., Herwig L., Krudewig A., Sauteur L., Belting H.-G. and Affolter M. (2013). In vivo analysis reveals a highly stereotypic morphogenetic pathway of vascular anastomosis. Dev. Cell 25, 492-506 10.1016/j.devcel.2013.05.010 [DOI] [PubMed] [Google Scholar]
- Lizama C. O. and Zovein A. C. (2013). Polarizing pathways: balancing endothelial polarity, permeability, and lumen formation. Exp. Cell Res. 319, 1247-1254 10.1016/j.yexcr.2013.03.028 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakatsu M. N., Davis J. and Hughes C. C. (2007). Optimized fibrin gel bead assay for the study of angiogenesis. J. Vis. Exp. 186 10.3791/186 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakayama M., Nakayama A., van Lessen M., Yamamoto H., Hoffmann S., Drexler H. C. A., Itoh N., Hirose T., Breier G., Vestweber D. et al. (2013). Spatial regulation of VEGF receptor endocytosis in angiogenesis. Nat. Cell Biol. 15, 249-260 10.1038/ncb2679 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ono Y., Fujii T., Ogita K., Kikkawa U., Igarashi K. and Nishizuka Y. (1988). The structure, expression, and properties of additional members of the protein kinase C family. J. Biol. Chem. 263, 6927-6932. [PubMed] [Google Scholar]
- Risau W. (1997). Mechanisms of angiogenesis. Nature 386, 671-674 10.1038/386671a0 [DOI] [PubMed] [Google Scholar]
- Selbie L. A., Schmitz-Peiffer C., Sheng Y. and Biden T. J. (1993). Molecular cloning and characterization of PKC iota, an atypical isoform of protein kinase C derived from insulin-secreting cells. J. Biol. Chem. 268, 24296-24302. [PubMed] [Google Scholar]
- Siekmann A. F. and Lawson N. D. (2007). Notch signalling limits angiogenic cell behaviour in developing zebrafish arteries. Nature 445, 781-784 10.1038/nature05577 [DOI] [PubMed] [Google Scholar]
- Strilić B., Kucera T., Eglinger J., Hughes M. R., McNagny K. M., Tsukita S., Dejana E., Ferrara N. and Lammert E. (2009). The molecular basis of vascular lumen formation in the developing mouse aorta. Dev. Cell 17, 505-515 10.1016/j.devcel.2009.08.011 [DOI] [PubMed] [Google Scholar]
- Whyte J., Thornton L., McNally S., McCarthy S., Lanigan F., Gallagher W. M., Stein T. and Martin F. (2010). PKCζ regulates cell polarisation and proliferation restriction during mammary acinus formation. J. Cell Sci. 123, 3316-3328 10.1242/jcs.065243 [DOI] [PubMed] [Google Scholar]
- Zovein A. C., Luque A., Turlo K. A., Hofmann J. J., Yee K. M., Becker M. S., Fassler R., Mellman I., Lane T. F. and Iruela-Arispe M. L. (2010). beta1 integrin establishes endothelial cell polarity and arteriolar lumen formation via a Par3-dependent mechanism. Dev. Cell 18, 39-51 10.1016/j.devcel.2009.12.006 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.