Abstract
Whereas low-valent late transition metal catalysis has become indispensible for chemical synthesis, homogeneous high-valent transition metal catalysis is underdeveloped, mainly due to the reactivity of high-valent transition metal complexes and the challenges associated with synthesizing them. In this manuscript, we report a mild carbon-carbon bond cleavage reaction by a Au(I) complex that generates a stable Au(III) cationic complex. Complementary to the well-established soft and carbophilic Au(I) catalyst, this Au(III) complex exhibits hard, oxophilic Lewis acidity. This is exemplified by catalytic activation of α,β-unsaturated aldehydes towards selective conjugate additions as well as activation of an unsaturated aldehyde-allene for a [2 + 2] cycloaddition reaction. The origin of the regioselectivity and catalytic activity was elucidated by X-ray crystallographic analysis of an isolated Au(III)-activated cinnamaldehyde intermediate. The concepts revealed in this study provide a strategy for accessing high-valent transition metal catalysis from readily available precursors.
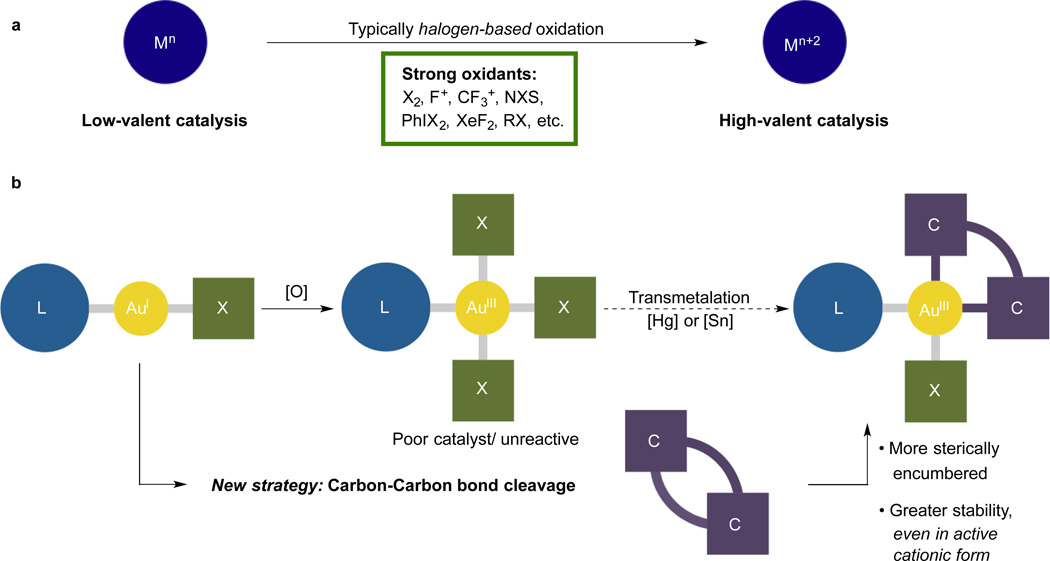
Transition metal catalysis has been developed into an efficient and selective strategy for organic transformations in modern chemistry. Low-valent late transition metal complexes enjoy particularly heavy use due to their stability and usefulness in forming important chemical bonds (C-C, C-O, C-N). However, low oxidation state, late metals struggle with other critical reactions including electrophilic C-H functionalization1, 2. Recent efforts have begun to unlock the potential of high-valent late transition metals, especially Pd(IV), to complement these shortcomings3–6. The major challenges thus far include the typical need for strong oxidants to access the high oxidation state, which limits the functional group tolerance, and the instability of the oxidized metal complexes, which often exist only as high-energy intermediates on the catalytic cycle (Fig. 1a). Put broadly, in order to fully explore the undoubtedly rich chemistry of high valent late metals, it will be essential to develop easily prepared, stable, tunable catalysts.
Figure 1. Routes to high-valent metal complexes.
a, Standard route to high-valent metal complexes using halogen-based oxidants. b, Hypothetically synthetic pathway for accessing Au(III)-C bond-stabilized Au(III) complexes.
In this regard, we have searched for a route to stable, catalytically active Au(III) complexes with the goal of complementing the ever-increasing library of Au(I)-catalyzed reactions. Whereas homogeneous Au(I) catalysis has seen great progress over the last decade7–11, Au(III) catalysis is still mainly limited to the use of inorganic Au(III) salts12. The synthetic challenge in forming stable, yet catalytically active, organometallic Au(III) complexes derives from the intrinsic high redox potential, leading to facile reduction of Au(III) complexes to Au(I) or metallic Au(0) species in the presence of electron-rich reagents13–18. In cases where the ligands are capable of stabilizing the highly oxidizing metal, the resulting complex is often rendered catalytically inert. For example, the oxidative addition product, L-AuX3, formed from L-AuX (X = halogen) is a poor catalyst by itself, and the abstraction of a halide to increase reactivity enables a facile reduction to the lower oxidation states15, 16. Moreover, while complexes of the type Au(III)(C^L)(X)(Y) are available through multi-step synthetic sequences19–21, their instability in the cationic form and the difficulties in readily tuning the coordination environment has severely limited their applications in catalysis.
To address this challenge, we hypothesized that a multidentate ligand scaffold with strong Au-C bonding energy might be able to stabilize cationic Au(III) organometallic complexes while maintaining catalytic activity. In designing the desired complex, we also hoped to avoid using strong oxidants to maximize the functional group compatibility of the protocol.
Based on the reported examples22, we reasoned that the transmetalation of Sn(biphenyl)(nBu)2 to L-AuX3 might afford a stable Au(III) complex with a bidentate ligand containing two strong Au-C bonds (Fig. 1b). The known stabilization of both neutral and cationic Au(I) complexes by NHCs (N-heterocyclic carbenes) inspired us to examine them as supporting ligands23. In an effort to access such compounds, attempts to perform the transmetalation of Sn(biphenyl)(nBu)2 to IPr-AuCl3 (2) (IPr = [1,3-bis(2,6-diisopropylphenyl)imidazol-2-ylidene]) gave us no desired product (Fig. 2). Although the replacement of IPrAuCl3 with (THT)AuCl3 (THT = tetrahydrothiophene) enabled access to complex 3 in good yield, the two step procedure was cumbersome, and did not meet the goals of being mild and straightforward.
Figure 2. Accessing gold(III) via oxidative addition of a carbon-carbon bond.
Attempted access to complex 3 via transmetalation of Sn(biphenyl)(nBu)2 with IPrAuCl3 2. Proposed pathway for the oxidative addition of IPrAu(I) complex with biphenylene/2,3,6,7-tetramethylbiphenylene. Coordination chemistry of IPrAu(III)(biphenyl) complex 3 and 6. X-ray structure of IPrAu(III)(biphenyl)Cl, complex 3 and [IPrAu(III)(biphenyl)(Me2NC(O)H)][SbF6] complex 6, SbF6− anion is omitted for clarity.
As an alternative, we imagined that insertion of an Au(I) complex into the strained C-C bond of biphenylene could achieve the desired oxidation and introduction of a stabilizing biphenyl ligand in a single step24–26 (Fig. 1b). The concept was appealing for its simplicity, even though oxidative addition to Au(I) complexes with mild oxidants is exceedingly rare, and in general requires either bimetallic complexes generating Au(II)-Au(II) intermediates18, 27–28 or complexes with specially designed ligands29–30. Moreover, no well-defined carbon-carbon bond cleavage reactions with Au(I) complexes have been reported. To investigate the feasibility of this strategy, the coordinatively unsaturated IPrAu(I)(SbF6) was generated in situ by reacting IPrAuCl with AgSbF6 resulting in the precipitation of AgCl in CD2Cl2 at room temperature. Reaction of the resulting electrophilic IPrAu(I)(SbF6) species with biphenylene resulted in the immediate and quantitative formation of the coordination complex, (IPrAu-biphenylene)(SbF6) 4 (Fig. 2), along with a small amount of the Au(III) aqua complex, [IPrAu(III)(biphenyl)(H2O)]SbF6 5, both of which were observed by 1H nuclear magnetic resonance (NMR) spectroscopy31. Nearly full conversion of the Au(I) cationic species to the desired Au(III) complex 5 was observed after 1.5 hours (see Supplementary Information, Figure S1 and Table S1). In contrast, most reported examples of this type of C-C bond cleavage requires more redox-active metals (e.g. Rh, Ir, Ni, Ru, Fe) and have been carried out under harsh reaction conditions24, 25, suggesting that the sterically unencumbered cationic IPrAu(I) complex might enjoy a comparatively fast rate of coordination and subsequent oxidative addition.
The oxidative addition of IPrAu(I)SbF6 with the more electron-rich 2,3,6,7-tetramethylbiphenylene (Me4-biphenylene) was also examined. Full conversion to [IPrAu(III)(Me4-biphenyl)(H2O)][SbF6] 7 was observed in 5 min at room temperature. When replacing AgSbF6 with AgOTf, the oxidative addition was complete after 6 hours to give [IPrAu(III)(Me4-biphenyl)OTf] 12. The longer reaction time required in the presence of the more coordinating anion is consistent with the hypothesis that coordination of biphenylene to a cationic coordinatively unsaturated Au(I) complex is the first step in the formation of 5. The thermodynamic driving force for this reaction comes from the cleavage of the strained biphenylene C-C bond (BDE = 65.4 kcal/mol) via oxidative addition to Au(I) yielding two Au-C(Ar) bonds24, 25. This unprecedented C-C bond cleavage by the linear IPrAu(I) cationic complex represents a facile approach for generating strong Au-C(Ar) bonds en route to stable Au(III) complexes. Treatment of 5 with nBu4NCl in dichloromethane resulted in the coordination of chloride and yielded IPrAu(III)(biphenyl)Cl 3 as a pale yellow powder in 80% isolated yield after column chromatography (Fig. 2).
An X-ray crystal structure of 3 reveals a Cs-symmetric distorted square-planar complex with one IPr carbene ligand, one chloride and two cyclometalation bonds from the biphenyl ligand defining the d8 Au(III) geometry shown in Fig. 2. The Au−C bond distances of the gold-biphenyl, Au(1)−C(1) and Au(1)−C(12), are 2.028(12) and 2.046(11) Å, respectively, are shorter than the gold-carbene bond length of 2.117(11) Å (see Supplementary Information). The relatively short bond distances exhibit the strong bonding energy between high oxidation Au(III) and its ligands. Consistent with our hypothesis, introduction of the cyclometallated biaryl ligand and one NHC ligand stabilized this high oxidation state Au(III) complex. The air- and moisture-stable complex 3 could be isolated and stored on the benchtop without any decomposition. Reaction of 3 with 1 equiv. of AgSbF6 caused immediate precipitation of AgCl in CD2Cl2 and the formation of 5 as observed by 1H NMR. This feature allows 3 to be used as a stable precatalyst of cationic Au(III).
To gain more insight on the coordination chemistry of cationic Au(III) complex with oxygen-based ligands, treatment of complex 5 with 1.1 equiv. of N,N-dimethylformamide (DMF) led to a substantial boost in stability, and allowed the isolation of [IPrAu(III)(biphenyl)(Me2NC(O)H)][SbF6] 6 via coordination of a lone pair of electrons on the carbonyl oxygen. As shown by the crystal structure of complex 6 (Fig. 2), the IPr, biphenyl and DMF ligands enforce a distorted square planar geometry around the Au(III) center, with the oxygen coordination from DMF in a distance of 2.140(3) Å. This finding suggests that the IPrAu(III)(biphenyl) cation exhibits a relatively hard, oxophilic Lewis-acidity, which is complementary to the soft Lewis acidity of cationic LAu(I) complexes. This was further exemplified by measuring the Lewis acidities of 1 and 3 by the Gutmann-Beckett method (see Supplementary Information, Figure S3). The DMF in 6 is located in the pocket created by the IPr and biphenyl ligands. This environment is substantially more crowded that observed in the linear IPrAuCl complex and effectively shields the DMF carbonyl group. The structural and electronic observations gained from this structure suggested a possible catalytic application of the IPrAu(III)(biphenyl) cation in selective 1,4-additions to α,β-unsaturated aldehydes.
Lewis acid-catalyzed 1,4-additions to α,β-unsaturated aldehydes remain challenging as 1,2-additions generally predominate32–34. Yamamoto and colleagues have reported a unique strategy for Lewis acid promoted 1,4-addition to a α,β-unsaturated aldehyde using aluminum reagents with sterically demanding ligands35. Although excellent 1,4-selectivity is obtained, stoichiometric amounts of Lewis acid appear to be necessary.
To obtain preliminary information regarding the reactivity of complex 5 prepared from 3 in Lewis acid catalysis, we initiated our experimental efforts by studying the Mukaiyama–Michael reaction with cinnamaldehyde and ketene silyl acetal 8 under ambient reaction condition. As an initial result, 1,4-adduct was obtained as the major product (1,4-/1,2-adduct = 75/25) in 98% NMR yield when catalyzed by IPrAu(III)(biphenyl)(SbF6) (see Supplementary Information, Table S2). Replacing AgSbF6 with AgOTf gave 1,4-selectivity exclusively and the product was isolated in 76% yield (1,4-/1,2-adduct = >98/2; Fig. 3a). With the optimized conditions in hand, silyl nitronate 9 was attempted as nucleophile instead of 8. The corresponding γ-nitro aldehyde was isolated in 78% yield with excellent selectivity (Fig. 3b). Employing trans-2-hexenal 18a as an aliphatic substrate also yielded the corresponding 1,4-adducts with good yield and excellent regioselectivity (Fig. 3a,b). Additionally, the catalyst loading could be decreased to 5 mol% without any loss of yield or selectivity: 10a was isolated in 77% yield with 1,4-/1,2-adduct >98/2 (see Supplementary Information, Table S2). The cationic IPrAu(III)(biphenyl) complex was essential for 1,4-selectivity in these reactions; in control experiments, the corresponding 1,2-adduct was mainly obtained using IPrAu(I)(OTf) and no product was obtained with Ag(OTf) alone. To maximize the convenience and accessibility of our new catalyst, we examined the possibility of in situ generation of the IPrAu(III) cation via oxidative addition with biphenylene, followed by its utilization as a catalyst in one-pot reactions (Fig. 3c). Indeed, combining IPrAu(I)Cl, AgOTf, and Me4-biphenylene (30 mol%) led to the formation of active catalyst IPrAu(III)(Me4-biphenyl)(OTf) 12 within 1 hour. Subsequent addition of cinnamaldehyde and silyl ketene acetal 8 led to the desired Mukaiyama−Michael adduct in 72% yield (1,4-/1,2-adduct = >98/2). Notably, the reactions do not suffer from using in situ generated catalyst as opposed to preformed Au(III) complex. This flexibility illustrates the power of using biphenylene to generate the Au(III) catalyst: other oxidizing agents are completely incompatible with the sensitive silyl ketene acetal and aldehyde functionalities. As an illustration, replacement of biphenylene with Br2 or PhICl2 for the oxidation of IPrAu(I)SbF6 yielded no 1,2-/1,4-adducts in the Mukaiyama−Michael reaction, and only 8% conversion to the 1,2-adduct was obtained with XeF2 (Fig. 3c).
Figure 3. Examples of selective Au(III)-catalyzed 1,4-additions.
a, Mukaiyama-Michael addition. b, Nitronate Michael-addition. c, In situ generation of IPrAu(III)(Me4-biphenyl) catalyst for Mukaiyama−Michael addition. d, One-pot tandem Au(I)/Au(III) and Au(III)/Au(III)-catalyzed reactions. [a] NMR yield.
The ability to generate the Au(III) catalyst in situ under mild conditions further suggested to us the alluring possibility of performing successive Au(I)- and Au(III)-catalyzed reactions in a single pot. Thus, we explored a one-pot tandem reaction starting from propargyl alcohol 13 (Fig. 3d). First, the IPrAu(I)(SbF6) catalyzed Meyer–Schuster rearrangement of propargyl alcohol 13 gave an unsaturated aldehyde. Subsequent oxidative addition of biphenylene afforded the IPrAu(III) cation 5, which catalyzed the Mukaiyama–Michael addition of silyl ketene acetal 8 to provide the final product in 57% overall yield (1,4-/1,2-adduct = >98/2). The ability to carry out two distinct reactions with different oxidation state catalysts originating from a single precursor is a remarkable feature of the newly developed methods.
Futhermore, a successive Au(III)-catalyzed ring-opening and Mukaiyama-Michael reaction was conducted. In the first step, 5 activated the ring-opening of the cyclopropene 14, which reacted with furan, to form a functionalized conjugated trienal (Fig. 3d)36. After removal of the excess furan, 5 catalyzed the Mukaiyama–Michael addition of 8 to obtain the final product in 53% isolated yield (>98% 1,4-selectivity).
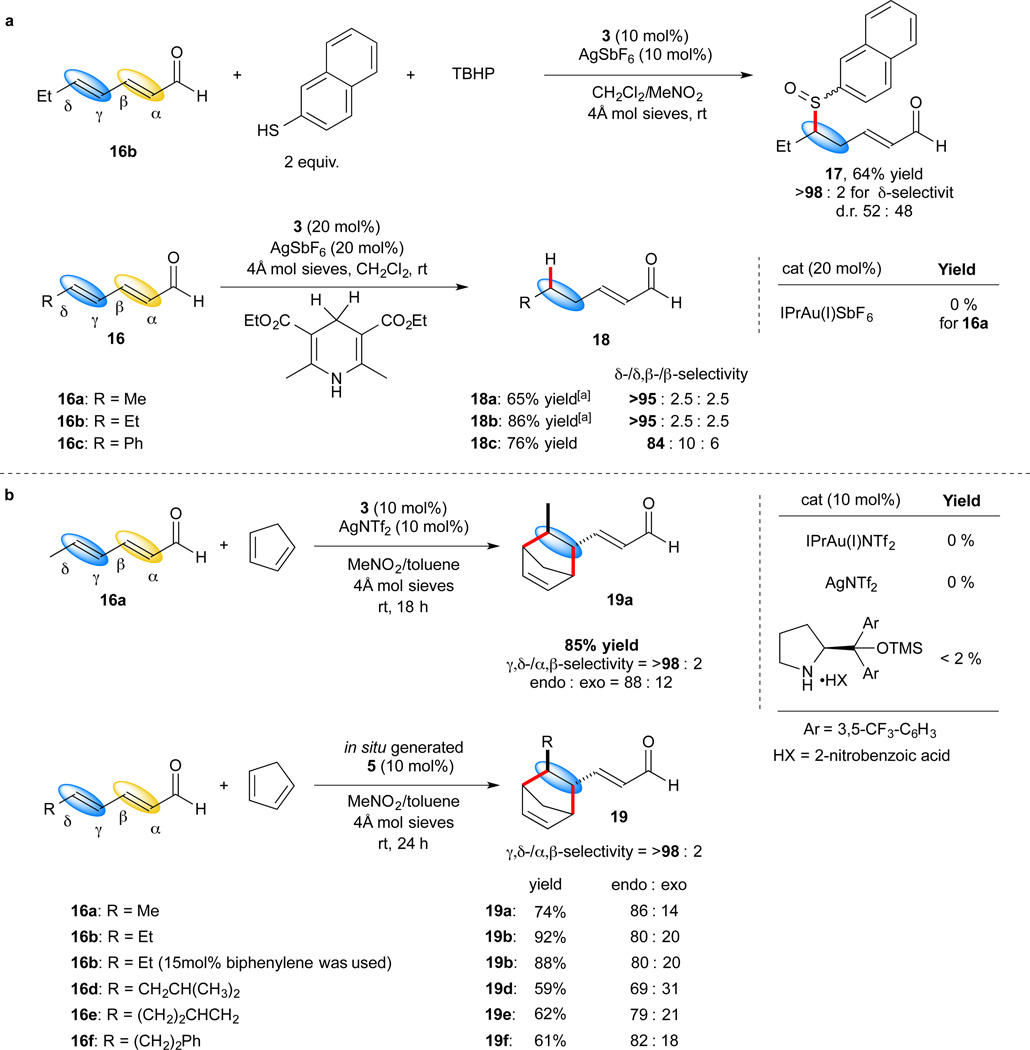
To demonstrate the generality of this Au(III) catalyst for obtaining remote selectivity, we next performed 1,6-selective thiol addition and reduction reactions of α,β,γ,δ-diunsaturated aldehydes. We hypothesized that the bulky catalyst ‘aldehyde pocket’ should partially shield the proximal double bond, thus promoting nucleophilic addition at the γ,δ-double bond. 1,6-additions to α,β,γ,δ-diunsaturated aldehydes are challenging and especially thiol additions and Hantzsch ester mediated reductions are known to proceed with low selectivity to provide a mixture of products.37, 38
For the thiol addition (Fig. 4a), 1,6-addition of naphthalene thiol to 16b and subsequent oxidation was carried out. With the use of 10 mol% 3 and AgSbF6, the oxidized 1,6-addition product 17 was obtained with 64% yield and exclusive 1,6-selectivity in the presence of TBHP as an oxidant. For the reduction reaction with Hantzsch ester (Fig 4a), catalyst 5 also demonstrated excellent selectivity for the remote reduction of α,β-γ,δ-diunsaturated aldehydes 16 to give the α,β-unsaturated aldehydes 18, further showcasing the unique regioselectivity obtained with the Au(III) catalyst compared to traditional bulky Lewis and Brønsted acid catalysts (See Supplementary Information, Table S5 and e.g. ref. 32–34).
Figure 4. Remote selectivity in gold(III) catalyzed addtions to dienals.
a, δ-selective thiol addition and reduction reactions. b, γ, δ-selective Diels−Alder reaction and in situ generation of IPrAu(III)(biphenyl) catalyst for Diels−Alder reactions.
Moreover, we also performed the Diels–Alder reaction of 2,4-hexadienal 16 with cyclopentadiene39. Only the γ,δ-functionalized product 19a was obtained in 85% yield (endo:exo = 88:12) employing IPrAu(III)(biphenyl)(NTf2) as catalyst (Fig. 4b). γ,δ-selective Diels–Alder reactions are unprecedented since α,β-selectivity normally predominates in such Lewis acid catalyzed reactions40. Notably, neither IPrAu(I)(NTf2), Ag(NTf2) nor organocatalysts provided any product with 16a and cyclopentadiene (see Supplementary Information, Table S3) further illustrating the novel reactivity of this gold(III)-based catalytic system. Furthermore, in situ generation of Au(III) catalyst from Au(I) (Fig 3c and 4b) was also used for the Diels–Alder reaction. Several substrates were shown to provide products in good yield and excellent regioselectivity. The products was isolated in up to 92% yield exclusively as the γ,δ-adducts with a endo:exo of up to 86/14 employing [IPrAu(III)(biphenyl)(H2O)]SbF6 generated in situ from IPrAu(I)(SbF6) and biphenylene. To increase the practicality of the developed procedure we also showed that the amount of biphenylene oxidant can be lowered to 15 mol% (1.5 equivalents as compared to Au(I)). Using these conditions, the in situ formed Au(III) catalyst produced product 19b without significant decline in yield and selectivity.
Whereas [2 + 2] cycloadditions of α,β-unsaturated carbonyl compounds and allenes have been carried out with photochemical conditions41 there are no examples of Lewis acid catalysis performing such reactions. Therefore, we next attempted to carry out the intramolecular [2 + 2] cycloaddition of cis-unsaturated aldehyde-allene 20 (Fig. 5). When 3 with AgSbF6 was employed, only [2 + 2] cycloadduct was obtained in 70% yield (cis : trans = 89 : 11). Investigations showed that neither IPrAu(I)(SbF6), Ag(SbF6), MeAlCl2, hυ, nor organocatalysts catalyzed the [2 + 2] cycloaddition (see Supplementary Information, Table S4); as such, this [2 + 2] cycloaddition is an unprecedented reaction unique to the developed Au(III) catalytic system.
Figure 5. Gold(III) catalyzed [2 + 2] cycloaddition of a allene-aldehyde.
The solid state structure of the key intermediate [IPrAu(III)(biphenyl)(η1-cinnamaldehyde)][SbF6] 22 was determined by X-ray crystallography as illustrated in Fig. 6. This Au(III)-cinnamaldehyde complex displays a distorted square planar geometry with a η1−-coordination cinnamaldehyde. The s-trans-cinnamaldehyde is buried in the pocket created by the IPr and biphenyl ligands. One of the 2,6-diisopropyl phenyl units is tilted away from the cinnamaldehyde plane due to steric hindrance. As a result, the carbonyl moiety is effectively shielded from nucleophilic attack and by this means, the IPrAu(III)(biphenyl) cation exhibits excellent remote selectivity as a catalyst in additions to unsaturated aldehydes.
Figure 6. A model for the obtained selectivity.
X-ray structure of [IPrAu(III)(biphenyl)(η1-cinnamaldehyde)][SbF6] complex 22. SbF6− anion is omitted for clarity.
Exploration of the unique catalytic abilities of high valent late transition metals has been hampered by difficulties in accessing stable complexes with well-controlled reactivity. Nowhere is this more evident than in the case of Au(III), which lags in its infancy compared to the ever-expanding field of Au(I) catalysis. In sharp contrast to previous methods for accessing Au(III), which rely on strong halogen-based oxidants, we have discovered that stable and catalytically active Au(III) complexes can be obtained by the mild oxidative addition of biphenylene to cationic IPrAu(I). While oxidative additions with gold(I) have been previously viewed as kinetically challenging,42–43 this striking carbon-carbon bond cleavage proceeds under surprisingly mild reaction conditions, even when compared to previously reported oxidative additions of biphenylene. The resulting IPrAu(III)(biphenyl) catalyst shows good reactivity as a hard Lewis acid catalyst, which is complementary to the soft Lewis acidity exhibited by Au(I)-catalysts. The catalytic reactivity is exemplified by six reactions of α,β-unsaturated or α,β-γ,δ-diunsaturated aldehydes: Mukaiyama−Michael additions, nitronate Michael additions, a thiol addition, Hantzsch ester reductions, Diels-Alder reactions and a [2 + 2] cycloaddition all proceed good yield and excellent selectivity. Whereas commonly employed Au(III) catalysts like AuCl3 exhibit harsh, nonselective acidity44, these new catalysts possess intermediate reactivity and a sterically defined binding pocket. The ligand environment of the new catalysts, characterized by X-ray crystallography, imparts exquisite 1,4- over 1,2-selectivity in the case of the Michael additions, δ-selectivity in the thiol addition and reduction reactions as well as γ,δ-selectivity in the Diels-Alder reactions of dienals. None of these reactions are feasible with gold(I)-based catalysts nor traditional bulky Lewis and Brønsted acid catalysts (see Supplementary Information, Table S5 and e.g. ref. 32–34). Furthermore, the catalysts can be generated in situ from commercially available reagents, thereby eliminating the need for specialized organometallic techniques. The mild nature of the oxidation from Au(I) to Au(III) even permits successive Au(I)- and Au(III)-catalyzed reactions in a single reaction vessel. In other words, one can take a single precursor and switch between two different reaction manifolds by simple in situ catalyst modification. In view of all these features, the methods presented herein should aid in unlocking the potential of high oxidation state gold catalysis.
Full Methods and any associated references are available in the online version of the paper at www.nature.com/nature.
Supplementary Material
Acknowledgments
We gratefully acknowledge the NIHGMS (RO1 GM073932) for financial support. C.-Y.Wu thanks the Taiwan National Science Council for a postdoctoral fellowship (2011–2012). T.H. thanks the Uehara Memorial Foundation for a postdoctoral fellowship. C.B.J. is grateful to The Lundbeck Foundation for a postdoctoral fellowship. We thank Dr. Antonio DiPasquale (the University of California, Berkeley College of Chemistry X-ray Crystallography Facility) for X-ray crystallographic data collection and acknowledge the support from NIH Shared Instrumentation Grant S10-RR027172. We thank Dr. Hsueh-Ju Liu for his generous donation of the biphenylene.
Footnotes
Supplementary Information is linked to the online version of the paper at www.nature.com/nature.
Author Contributions C.-Y.Wu initiated and developed the organometallic study. C.-Y.Wu and T.H. developed the Lewis acid catalysis. C.-Y.Wu, T.H. and C.B.J. optimized the Lewis acid catalysis study. C.-Y.Wu, T.H. and C.B.J. performed the experiments. C.-Y.Wu, T.H. and C.B.J. and F.D.T wrote the manuscript.
Author Information X-ray crystallographic data have been deposited in the Cambridge Crystallographic Data Centre database (http://www.ccdc.cam.ac.uk/) under code CCDC 1002525-1002527. Reprints and permissions information is available at www.nature.com/ reprints. The authors declare no competing financial interests. Readers are welcome to comment on the online version of this article at www.nature.com/nature.
References
- 1.Hickman AJ, Sanford MS. High-valent organometallic copper and palladium in catalysis. Nature. 2012;484:177–185. doi: 10.1038/nature11008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Furuya T, Kamlet AS, Ritter T. Catalysis for fluorination and trifluoromethylation. Nature. 2011;473:470–477. doi: 10.1038/nature10108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Lee E, et al. A fluoride-derived electrophilic late-stage fluorination reagent for PET imaging. Science. 2011;334:639–642. doi: 10.1126/science.1212625. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Furuya T, et al. Mechanism of C-F reductive elimination from palladium(IV) fluorides. J. Am. Chem. Soc. 2010;132:3793–3807. doi: 10.1021/ja909371t. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Whitfield SR, Sanford MS. Reactivity of Pd(II) complexes with electrophilic chlorinating reagents: Isolation of Pd(IV) products and observation of C−Cl bond-forming reductive elimination. J. Am. Chem. Soc. 2007;129:15142–15143. doi: 10.1021/ja077866q. [DOI] [PubMed] [Google Scholar]
- 6.Alsters PL, et al. Rigid five- and six-membered C,N,N'-bound aryl-, benzyl-, and alkylorganopalladium complexes: sp2 vs. sp3 carbon-hydrogen activation during cyclopalladation and palladium(IV) intermediates in oxidative addition reactions with dihalogens and alkyl halides. Organometallics. 1993;12:1831–1844. [Google Scholar]
- 7.Wang Y-M, Lackner AD, Toste FD. Development of catalysts and ligands for enantioselective gold catalysis. Acc. Chem. Res. 2014;47:889–901. doi: 10.1021/ar400188g. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Krause N, Winter C. Gold-catalyzed nucleophilic cyclization of functionalized allenes: A powerful access to carbo- and heterocycles. Chem. Rev. 2011;111:1994–2009. doi: 10.1021/cr1004088. [DOI] [PubMed] [Google Scholar]
- 9.Corma A, Leyva-Pérez A, Sabater MJ. Gold-catalyzed carbon-heteroatom bond-forming reactions. Chem. Rev. 2011;111:1657–1712. doi: 10.1021/cr100414u. [DOI] [PubMed] [Google Scholar]
- 10.Hamilton GL, Kang EJ, Mba M, Toste FD. A powerful chiral counterion strategy for asymmetric transition metal catalysis. Science. 2007;317:496–499. doi: 10.1126/science.1145229. [DOI] [PubMed] [Google Scholar]
- 11.Gorin DJ, Toste FD. Relativistic effects in homogeneous gold catalysis. Nature. 2007;446:395–403. doi: 10.1038/nature05592. [DOI] [PubMed] [Google Scholar]
- 12.Schmidbaur H, Schier A. Gold(III) compounds for homogeneous catalysis: Preparation, reaction conditions, and scope of application. Arabian J. Sci. Eng. 2012;37:1187–1225. [Google Scholar]
- 13.Oliver-Meseguer J, et al. Small gold clusters formed in solution give reaction turnover numbers of 107 at room temperature. Science. 2012;338:1452–1455. doi: 10.1126/science.1227813. [DOI] [PubMed] [Google Scholar]
- 14.Leyva-Pérez A, Corma A. Similarities and differences between the “relativistic” triad gold, platinum, and mercury in catalysis. Angew. Chem. Int. Ed. 2012;51:614–635. doi: 10.1002/anie.201101726. [DOI] [PubMed] [Google Scholar]
- 15.Gaillard S, et al. Synthetic and structural studies of [AuCl3(NHC)] complexes. Organometallics. 2010;29:394–402. [Google Scholar]
- 16.de Frémont P, Singh R, Stevens ED, Petersen JL, Nolan SP. Synthesis, characterization and reactivity of N-heterocyclic carbene gold(III) complexes. Organometallics. 2007;26:1376–1385. [Google Scholar]
- 17.Hashmi ASK, Blanco MC, Fischer D, Bats JW. Gold catalysis: Evidence for the in situ reduction of gold(III) During the cyclization of allenyl carbinols. Eur. J Org. Chem. 2006:1387–1389. [Google Scholar]
- 18.Wolf WJ, Winston MS, Toste FD. Exceptionally fast carbon–carbon bond reductive elimination from gold(III) Nature Chem. 2013;6:159–164. doi: 10.1038/nchem.1822. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Vicente J, Bermúdez M-D, Carrión F-J, Jones PG. Synthesis and reactivity of some nitroaryl complexes of HgII and AuIII - synthesis of a substituted biphenyl by C-C coupling-crystal structure of [Hg(C6H4NO2-3,O n Bu-6)2] Chem. Ber. 1996;129:1395–1399. [Google Scholar]
- 20.Roşca D-A, Smith DA, Hughes DL, Bochmann M. A Thermally stable gold(III) hydride: synthesis, reactivity, and reductive condensation as a route to gold(II) complexes. Angew. Chem. Int. Ed. 2012;51:10643–10646. doi: 10.1002/anie.201206468. [DOI] [PubMed] [Google Scholar]
- 21.Hashmi ASK. Fire and ice: A Gold(III) monohydride. Angew. Chem. Int. Ed. 2012;51:12935–12936. doi: 10.1002/anie.201208603. [DOI] [PubMed] [Google Scholar]
- 22.Usón R, Vicente J, Cirac JA, Chicote MT. Synthesis and reactivity of dibenzometallole complexes of gold(III) and platinum(II) J. Organomet. Chem. 1980;198:105–112. [Google Scholar]
- 23.Pyykkö P, Runeberg N. Comparative theoretical study of N-heterocyclic carbenes and other ligands bound to AuI. Chem. Asian J. 2006;1:623–628. doi: 10.1002/asia.200600181. [DOI] [PubMed] [Google Scholar]
- 24.Jones WD. Mechanistic studies of transition metal-mediated C–C bond activation. Top. Curr. Chem. 2013;491:1–31. doi: 10.1007/128_2013_491. [DOI] [PubMed] [Google Scholar]
- 25.Perthuisot C, et al. Cleavage of the carbon–carbon bond in biphenylene using transition metals. J. Mol. Catal. A: Chem. 2002;189:157–168. [Google Scholar]
- 26.Darmon JM, et al. Oxidative addition of carbon–carbon bonds with a redox-active bis(imino)pyridine iron complex. J. Am. Chem. Soc. 2012;134:17125–17137. doi: 10.1021/ja306526d. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.For examples see: Levin LD, Toste FD. Gold-catalyzed allylation of aryl boronic acids: accescing cross-coupling reactivity with gold. Angew. Chem. Int. Ed. 2014;53:6211–6215. doi: 10.1002/anie.201402924. and references herein.
- 28.Fackler JP., Jr Metal-metal bond formation in the oxidative addition to dinuclear gold(I) species. Implications from dinuclear and trinuclear gold chemistry for the oxidative addition process generally. Polyhedron. 1997;16:1. [Google Scholar]
- 29.Guenther J, Mallet-Ladeira S, Estevez L, Miqueu K, Amgoune A, Bourissou D. Activation of aryl halides at gold(I): practical synthesis of (P,C) cyclometalated gold(III) complexes. J. Am. Chem. Soc. 2014;136:1778–1781. doi: 10.1021/ja412432k. [DOI] [PubMed] [Google Scholar]
- 30.Joost M, Zeineddine A, Estévez L, Mallet-Ladeira S, Miqueu K, Amgoune A, Bourissou D. Facile oxidative addition of aryl iodides to gold(I) by ligand design: bending turns on reactivity. J. Am. Chem. Soc. doi: 10.1021/ja506978c. [DOI] [PubMed] [Google Scholar]
- 31.Weber SG, Rominger F, Straub BF. Isolated silver intermediate of gold precatalyst activation. Eur. J. Inorg. Chem. 2012:2863–2867. [Google Scholar]
- 32.North M, Usanov DL, Young C. Lewis acid catalyzed asymmetric cyanohydrin synthesis. Chem. Rev. 2008;108:5146–5226. doi: 10.1021/cr800255k. [DOI] [PubMed] [Google Scholar]
- 33.Yamamoto H, editor. Lewis acids in organic synthesis. Wiley: Weinheim; 2000. pp. 1–995. [Google Scholar]
- 34.Mahrwald R. Diastereoselection in Lewis-acid-mediated aldol reactions. Chem. Rev. 1999;99:1095–1120. doi: 10.1021/cr980415r. [DOI] [PubMed] [Google Scholar]
- 35.Maruoka K, Imoto H, Saito S, Yamamoto H. Virtually complete blocking of α,β-unsaturated aldehyde carbonyls by complexation with aluminum tris(2,6-diphenylphenoxide) J. Am. Chem. Soc. 1994;116:4131–4132. [Google Scholar]
- 36.Hadfield MS, Lee A-L. Gold(I)-catalyzed syntheis of conjugated trienes. Chem. Commun. 2011;47:1333–1335. doi: 10.1039/c0cc04217j. [DOI] [PubMed] [Google Scholar]
- 37.Akagawa K, Nishi N, Sen J, Kudo K. Peptide-catalyzed consecutive 1,6- and 1,4-additions of thiols to α,β,γ,δ-unsaturated aldehydes. Org. Biomol. Chem. 2014;12:3581–3585. doi: 10.1039/c4ob00565a. [DOI] [PubMed] [Google Scholar]
- 38.Akagawa K, Sen J, Kudo K. Peptide-catalyzed regio- and enantioselective reduction of α,β,γ,δ-diunsaturated aldehydes. Angew. Chem. Int. Ed. 2013;52:11585–11588. doi: 10.1002/anie.201305004. [DOI] [PubMed] [Google Scholar]
- 39.Ahrendt KA, Borths CJ, MacMillan DWC. New strategies for organic synthesis: the first highly enantioselective organocatalytic Diels-Alder reaction. J. Am. Chem. Soc. 2000;122:4243–4244. [Google Scholar]
- 40.Hayashi Y, Okamura D, Umemiya S, Uchimaru T. Organocatalytic 1,4-addition reaction of α,β,γ,δ-diunsaturated aldehydes versus 1,6-addition reaction. ChemCatChem. 2012;4:959–962. [Google Scholar]
- 41.Alcaide B, Almendros P, Arancillo C. Exploiting [2 + 2] cycloaddition chemistry: achievements with allenes. Chem. Soc. Rev. 2010;39:783–816. doi: 10.1039/b913749a. [DOI] [PubMed] [Google Scholar]
- 42.Hashmi ASK, et al. On Homogeneous gold/palladium catalytic systems. Adv. Synth. Catal. 2012;354:133–134. [Google Scholar]
- 43.Livendahl M, Goehry C, Maseras F, Echavarren AM. Rationale for the sluggish oxidative addition of aryl halides to Au(I) Chem. Commun. 2014;50:1533–1536. doi: 10.1039/c3cc48914k. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Hashmi ASK, Frost TM, Bats JW. Highly selective gold-catalyzed arene synthesis. J. Am. Chem. Soc. 2000;122:11553–11554. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.