Abstract
Taurodontism (i.e. enlarged pulp chamber with concomitant apical displacement of the root bi/trifurcation) is considered a dental anomaly with relatively low incidence in contemporary societies, but it represents a typical trait frequently found in Neandertal teeth. Four hypotheses can be envisioned to explain the high frequency in Neandertals: adaptation to a specific occlusal loading regime (biomechanical advantage), adaptation to a high attrition diet, pleiotropic or genetic drift effects. In this contribution we used finite element analysis (FEA) and advanced loading concepts based on macrowear information to evaluate whether taurodontism supplies some dental biomechanical advantages. Loads were applied to the digital model of the lower right first molar (RM1) of the Neandertal specimen Le Moustier 1, as well as to the digital models of both a shortened and a hyper-taurodontic version of Le Moustier RM1. Moreover, we simulated a scenario where an object is held between teeth and pulled in different directions to investigate whether taurodontism might be useful for para-masticatory activities. Our results do not show any meaningful difference among all the simulations, pointing out that taurodontism does not improve the functional biomechanics of the tooth and does not favour para-masticatory pulling activities. Therefore, taurodontism should be considered either an adaptation to a high attrition diet or most likely the result of pleiotropic or genetic drift effects. Finally, our results have important implications for modern dentistry during endodontic treatments, as we observed that filling the pulp chamber with dentine-like material increases tooth stiffness, and ultimately tensile stresses in the crown, thus favouring tooth failure.
Keywords: biomechanics, finite element analysis (FEA), Homo neanderthalensis, taurodontism, teeth
Introduction
Neandertal teeth are peculiar for the high frequency of specific dental traits, such as large, shovel-shaped incisors, premolars with complex occlusal morphology, lower premolars with asymmetrical occlusal outline, upper molars (usually M1) with expanded hypocone, lower molars with anterior fovea distally bordered by a mid-trigonid crest, and molars (both uppers and lowers) with enlarged pulp chambers or taurodont (‘bull-toothed’) roots (Bailey, 2002, 2004, 2006; Kupczik & Hublin, 2010; Benazzi et al. 2011a). With regard to the last feature, Gorjanović-Kramberger (1907, 1908) was the first to observe in the Neandertal Krapina fossil sample that molars (and, to a lesser extent, premolars) show apically positioned root bifurcation resulting in an enlargement of the body (or stem) of the root at the expense of the root branches. Due to the similarity of this prismatic root with the condition seen in the teeth of ungulates, Keith (1913) introduced the term ‘taurodontism’, and subsequently Shaw JCM (1928) Taurodont teeth in South African races. J Anat 62, 476-498. Classified taurodont teeth according to the relative displacement of the floor of the pulp chamber into hypo-, meso- and hyper-taurodont forms.
Taurodontism is an anatomical developmental anomaly seen in permanent and primary teeth, caused by the failure or delay of Hertwig's epithelial sheath diaphragm to invaginate at the proper horizontal level (Hamner et al. 1964; Wright, 2007). Even though the specific molecular signals that induce the invagination of the epithelial sheath are still unknown, it has been noted that this process influences root length (Kovacs, 1971): fast narrowing of the epithelial sheath produces short and tapered roots, whereas delay or failure of the invagination produces long roots (or even pyramidal roots), as often found in Neandertal molars (e.g. Lebel & Trinkaus, 2002; Macchiarelli et al. 2008; Walker et al. 2008; Dąbrowski et al. 2013).
Kupczik & Hublin (2010) provided a comprehensive morphometric analysis of this feature comparing European Neandertals with Late Pleistocene humans from Northwest Africa (Aterian) and recent Homo sapiens, confirming the high frequency of taurodontism in Neandertal mandibular molars and pointing out the larger M1 and M2 root surface areas in Neandertals than in recent modern humans. Owing to the link between masticatory function and root surface area (Spencer, 2003; Kupczik & Dean, 2008) their results suggest that Neandertal molars were subjected to elevated occlusal forces exercised to comminute hard food items. Therefore, taurodontism might be a masticatory functionally related feature (i.e. an adaptive biomechanical advantage) useful to withstand a specific occlusal loading regime. Indeed, it has been suggested that the Neandertal cranio-facial morphology (e.g. marked mid-facial prognathism, posteriorly positioned zygomatic root) is an adaptation to resist strong bite forces, mainly applied to the anterior teeth (e.g. Hylander, 1977; Rak, 1986; Trinkaus, 1987; Smith & Paquette, 1989; Spencer & Demes, 1993; Wang et al. 2010). However, Antón (1994) showed that bite force production in Neandertals was smaller than in modern humans (for both anterior and posterior teeth). Likewise, O'Connor et al. (2005) pointed out that Neandertal's bite force was not exceptionally large when considering cranial size (even less efficient than many modern humans), a result later supported by Clement et al. (2012), who suggested that Neandertals were similar to other Late Pleistocene hominins at least with regard to the anterior dental loadings.
In this contribution we aim to evaluate whether taurodontism confers biomechanical advantages in Neandertals. We used finite element analysis (FEA) and advanced loading concepts based on macrowear information (Kullmer et al. 2009; Benazzi et al. 2011b, 2012, 2013a,b, 2014) to load the lower right first molar (RM1) of Le Moustier 1. The analysis was carried out on the digital volumetric model of the original specimen and on the digital models of a shortened and a hyper-taurodontic version of Le Moustier RM1 to emphasize the differences (in displacements and stress distribution), if any, between the original and two extreme manipulated root shapes. Moreover, we investigated whether taurodontism might be useful for intensive use of the molar as a tool (i.e. para-masticatory activities; Fiorenza & Kullmer, 2013), simulating a scenario where an object is held between teeth and pulled in different directions.
Materials and methods
We obtained permission from the Museum für Vor- und Frühgeschichte, Staatliche Museen zu Berlin (Germany) to micro-CT scan the Neandertal specimen Le Moustier 1. Le Moustier 1 was selected because of the almost complete upper and lower dentition and because the molars are only slightly worn, possessing a well-developed wear facet pattern. Micro-CT scanning was carried out with a BIR ACTIS high-resolution CT scanner using the following scan parameters: 130 kV, 100 μA, 0.25 mm brass, and 5000 views per rotation. Volume data were reconstructed using isometric voxels of 66.8 μm for the mandible and 30.6 μm for the maxilla.
The size of the digital models was reduced by cropping the micro-CT image stack mesially and distally to the socket of the RM1 (for the mandible) and RP4-RM1 (for the maxilla). Segmentation of the RM1 dental and supporting tissues (enamel, dentine, pulp, alveolar and cortical bone), as well as RP4-RM1 dental tissues was carried out in avizo 7 (Visualization Sciences Group Inc.). Periodontal ligament (PDL) for the RM1 was modelled with an interface of 0.2 mm between the root surface and the alveolar bone, an average value among those found in the scientific literature (e.g. Hohmann et al. 2011; Xia et al. 2013). As recent in vitro studies suggest that the enamel-dentine junction (EDJ) plays an important role in accommodating mechanical stresses (Zaytsev & Panfilov, 2014), the EDJ was modelled as 100-μm-thick layer between enamel and dentine (Marshall et al. 2001).
The final refinement of the digital model (hereafter called RM1-original) was carried out in geomagic studio 2012 (Geomagic, Inc.) (Fig.1a). In the same software the pulp of RM1-original was digitally modified to simulate a reduced (hereafter called RM1-reduced; Fig.1b) and a hyper-taurodont (hereafter called RM1-hyper; Fig.1c) pulp chamber. In both cases, the position and morphology of the pulp apices and pulp roof were preserved, and just the bifurcation was moved upward (for RM1-reduced) and downward (for RM1-hyper) while constraining its relative position with regard the the pulp floor. As taurodontism is a continuous trait (in term of expression), we have selected two representative conditions that might account for a large range of variation in Neandertal and H. sapiens pulp shape. In detail, we considered data about root stem volume (i.e. the volume of the root between the cervical plane and the plane at bifurcation) published by Kupczik & Hublin (2010) for Neandertals (417.11 ± 145.70 mm3) and H. sapiens (237.90 ± 58.52 mm3) to guide our simulations. Starting from a root stem volume of 327.74 mm3 (RM1-original; pulp volume = 87.42 mm3, which is close to the Neandertal mean, 79.9 ± 34.47 mm3, computed by Kupczik & Hublin, 2010), the volume obtained for RM1-reduced was 269.45 mm3 (within the range of recent H. sapiens variability; pulp volume = 72.06 mm3), whereas the volume obtained for RM1-hyper was 512.41 mm3 (within the upper range of Neandertal variability; pulp volume = 118.09 mm3).
Figure 1.

Dental tissues and supporting structures for the lower right first molar Le Moustier 1 (RM1-original), as well as for two further versions where the pulp chamber of the RM1 was both reduced, to simulate the Homo sapiens condition (RM1-reduced), and increased, to simulate a hyper-taurodontic root (RM1-hyper). EDJ, enamel-dentine junction; PDL, periodontal ligament.
As suggested in previous contributions (e.g. Benazzi et al. 2011b, 2013a,b, 2014), a preliminary step for FEA requires the identification of contact areas between the RM1 and the antagonistic teeth during a two-body interaction, i.e. tooth–tooth contact, which might be more damaging to the tooth crown than food–tooth contacts, because more localized stresses increase (Lucas, 2004). The dental surface model of the RM1 and RP4-RM1 were imported into occlusal fingerprint analyser (OFA) software (Fig.2a). Thanks to collision detection, deflection and break-free algorithms, the OFA software allows one model to be moved against the other, selecting automatically the colliding triangles of the triangulated crown surface model (Benazzi et al. 2011b, 2012, 2014; Kullmer et al. 2013). The contact areas identified during maximum intercuspation were selected for FEA, thus giving information about the position where occlusal loads should be applied (red areas in Fig.2b). For loading direction, perpendicular loads to the contact areas were applied because we assumed that during maximum intercuspation contact, compressive forces act between complementary wear facet pairs (Hattori et al. 2009; Benazzi et al. 2011b). As in the digital versions of Le Moustier 1 RM1 (namely, RM1-original, RM1-reduced and RM1-hyper) the occlusal surface was not modified, all simulations were characterized by the same loading conditions (and boundary constraints; see below).
Figure 2.

(a) Collision detection for Le Moustier 1 RM1 with the antagonists RP4-RM1 in the occlusal fingerprint analyser software (OFA) during maximum intercuspation contact. (b) The RP4-RM1 are transparent to show the collision (red spots) in the occlusal surface of the RM1. (c) The FE mesh of RM1-original consisting of 939 380 10-node tetrahedral elements. B, buccal; D, distal; L, lingual; M, mesial;PDL, periodontal ligament.
The surface models were imported into hyperworks Software (Altair Engineering, Inc.), where volumetric meshes (for enamel, dentine, EDJ, pulp, PDL, cortical and alveolar bone) were created using 10-node tetrahedral elements (Fig.2c; Supporting Information Table S1). Information for material properties (elastic modulus – E, and the Poisson's ratio) was collected from the literature (Table1). As the E-modulus of EDJ is not constant but is a monotonic decrease from the enamel side of the EDJ to the dentine (Marshall et al. 2001), an average value between enamel and dentine was computed. All the biological materials represented in the models were considered homogeneous, linearly elastic and isotropic. Although this is an evident simplification for enamel (owing to the highly anisotropic stiffness characteristics of the enamel), it has been suggested that dentine can be modelled as an isotropic hard tissue (Kinney et al. 1999), and that the mechanical properties of dentine depend strongly on the shape of the specimen under investigation (Zaytsev et al. 2012). As we aim to explore the pattern of stress distribution in the root (which is basically made of dentine), and because in all simulations we used the same material parameters, the same boundary conditions (see below) and the same morphology for enamel and EDJ, the results will ultimately be driven by the different shapes of the root.
Table 1.
Elastic properties of isotropic materials
| Material | E*(GPa) | Poisson's ratio | References |
|---|---|---|---|
| Enamel | 84.1 | 0.3 | Magne (2007) |
| Dentine | 18.6 | 0.31 | Ko et al. (1992) |
| EDJ | 51.35 | 0.3 | Average between enamel and dentine |
| Pulp | 0.002 | 0.45 | Rubin et al. (1983) |
| PDL | 0.0689 | 0.45 | Holmes et al. (1996) |
| Alveolar bone | 11.5 | 0.3 | Dejak et al. (2007) |
| Cortical bone | 13.7 | 0.3 | Ko et al. (1992) |
EDJ, enamel-dentine junction; PDL, periodontal ligament.
Elastic modulus.
For each version of the Le Moustier RM1 we also evaluated whether filling the pulp chamber with dentine affects the pattern of stress distribution in the tooth (hereafter called RM1-original-F, RM1-reduced-F and RM1-hyper-F). This scenario reflects not only the physiological deposition of secondary dentine in the pulp chamber observed during individual life, but also the normal practice in endodontic treatment of root canal filling of taurodontic teeth (e.g. Bharti et al. 2009; Marques-da-Silva et al. 2010).
Boundary constraints were applied to the medial and distal cut surfaces of the mandible section. The medial nodes were restrained only in linguo-buccal translation, whereas the distal nodes were restrained both in mesio-distal and supero-inferior translation (Benazzi et al. 2012). A uniform pressure of 150 N was distributed on the RM1 occlusal surface according to the proportion of the occlusal contact areas detected in the OFA software. We applied 150 N because it is an average force among the loading conditions found in the literature for modern human (Cheng et al. 2010; Fu et al. 2010). Even though this value is uncertain, we note that our goal was to detect patterns of stress distribution in the tooth (which is not affected by the loading value because at each material point of the model the stress is linearly proportional to the force applied; i.e. stress and displacement can be scaled according to load factor), rather than to predict realistic loads that cause, e.g. fractures in the root.
Moreover, for each version of Le Moustier RM1 (RM1-original, RM1-reduced and RM1-hyper) we simulated a scenario wherein an object, having elastic modulus similar to rubber (E = 0.05 GPa) and Poisson's ratio = 0.3, is embedded on the occlusal surface of the RM1. The embedded material is much softer than the enamel and dentine so that it does not increase the stiffness of the entire tooth (Poisson's ratio value has no influence). A resultant force of 150 N was applied perpendicular to the object surface (i.e. parallel to the z-axis), reflecting a scenario that mimics soft tissue processing behaviour, e.g. food in between teeth during occlusion (load case 1 – LC1; Fig.3a). To investigate whether taurodontism might be useful for para-masticatory activities, further simulations were carried out pulling the embedded object either buccally (i.e. towards the cheek) or mesially (i.e. towards the anterior dentition). Specifically, loads were applied either parallel to the x-axis (load case 2 – LC2; Fig.3b) or parallel to the y-axis (load case 3 – LC3; Fig.3c) with a resultant force of 30 N computed as the product between the perpendicular loading (150 N) and the coefficient of friction of 0.2, a value found for wet conditions (Li & Zhou, 2002).
Figure 3.

An object was embedded on the occlusal surface of the RM1 and then a force was applied perpendicular to the object surface (load case 1 – LC1), parallel to the x-axis (i.e. buccally; load case 2 – LC2) and parallel to the y-axis (i.e. mesially; load case 3 – LC3).
For the six versions of Le Moustier RM1 (hence including the models with filled pulp chamber) we evaluated the displacement of the tooth and the pattern of maximum principal stresses. Maximum principal stresses are presented graphically using colour maps, with red areas showing highest tensile stresses and blue areas showing the lowest value (negative values suggest compression).
Results for the embedding object were qualitatively and quantitatively compared according to the first maximum principal stresses criterion only.
Due to the small sample size, no statistical test was used to assess the significance of the results. However, to facilitate the comparison of the patterns of stress distribution (for maximum principal stresses), we set the range limit of the colour maps at −6 to 6 MPa.
Results
The ultimate displacements of the six digital models of Le Moustier RM1 are low and very similar to each other when loaded during a maximum intercuspation contact situation (Table2). The largest displacement is found on the x-axis (bucco-lingual direction), probably due to the contact areas on the lingual cusps (which are higher than the buccal ones), which concur to create a resultant occlusal force slightly lingually directed. We observe a slight decrease of displacement values when the pulp chamber is filled with dentine material, suggesting that a filled pulp chamber increases tooth stiffness.
Table 2.
Ultimate displacement in x-, y- and z-axes and the magnitude (mm) of the Le Moustier RM1 digital versions under a uniform pressure of 150 N
| Specimen | ux | uy | uz | Magnitude |
|---|---|---|---|---|
| RM1-original | −8.401E-03 | 3.035E-03 | −7.056E-03 | 1.138E-02 |
| RM1-original-F* | −8.307E-03 | 3.004E-03 | −6.989E-03 | 1.126E-02 |
| RM1-reduced | −8.656E-03 | 3.058E-03 | −7.199E-03 | 1.167E-02 |
| RM1-reduced-F* | −8.556E-03 | 3.031E-03 | −7.133E-03 | 1.154E-02 |
| RM1-hyper | −8.644E-03 | 2.933E-03 | −7.161E-03 | 1.160E-02 |
| RM1-hyper-F* | −8.557E-03 | 2.886E-03 | −7.087E-03 | 1.148E-02 |
The pulp chamber is filled with material with dentine properties.
Similarly, the distributions of maximum principal stress for RM1-original, RM1-reduced and RM1-hyper are very similar to each other (Fig.4a). Tensile stresses are observed in the grooves of the occlusal surface, mainly in the lingual groove between the metaconid and entoconid, whereas the buccal half experiences compressive stresses. The root is mainly under compression, and the only difference observed among the three simulations involves a slight reduction of tensile stresses in the root bifurcation of RM1-hyper. It is worthwhile noting that modelling the EDJ as 100-μm-thick layers between enamel and dentine decreases tensile stresses in the crown (but not the general pattern of stress distribution) as compared with simulations that do not include the EDJ (Supporting Information Fig. S1).
Figure 4.
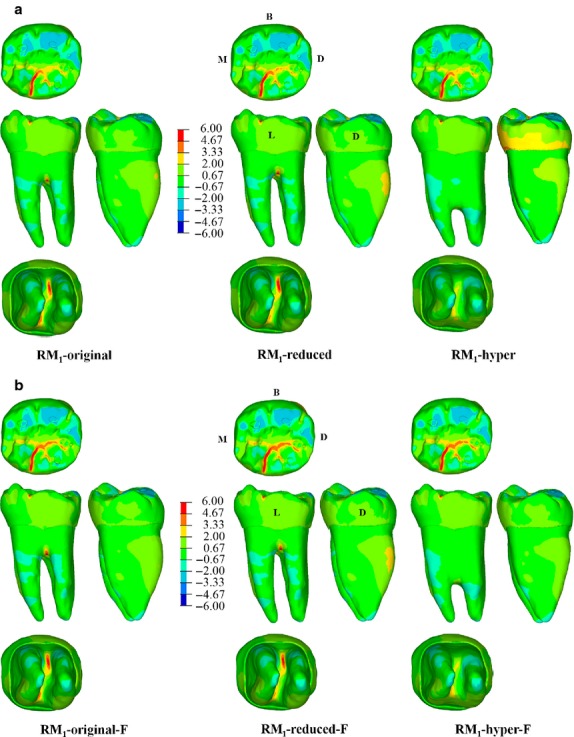
The maximum principal stress (MPa) distribution for specimen RM1-original, RM1-reduced (i.e. the bifurcation was moved upward) and RM1-hyper (i.e. the bifurcation was moved downward) with either unfilled (a) or filled (b) pulp chamber (i.e. RM1-original-F, RM1-reduced-F and RM1-hyper-F). Blue spots in the occlusal surface (compressive stress) represent the contact areas with the antagonistic teeth during maximum intercuspation (where the load was applied), whereas red spots represent tensile stresses. B, buccal; D, distal; L, lingual; M, mesial.
The pattern of stress distribution is also similar among the models with the pulp filled with dentine (Fig.4b). In the three simulations (RM1-original-F, RM1-reduced-F and RM1-hyper-F), tensile stresses are concentrated in the grooves of the lingual aspect of the occlusal surface, constrained between the mid-trigonid crest and the posterior fovea. Again, tensile stresses in the root bifurcation slightly decrease in specimen RM1-hyper-F. Noteworthy, Fig.4 also shows that when the pulp chamber is filled with dentine the magnitude of tensile stresses increases in the enamel (up to ∼25%; Supporting Information Fig. S2), indicating that a stiffer root negatively affects the crown. As in this simulation we applied only 150 N on the occlusal surface, according to Pérez-González et al. (2011) the failure criterion for brittle material, the increased tensile stresses do not reach the level of failure, but this might happen if the occlusal load is augmented.
Finally, a similar pattern of stress distribution was also obtained for RM1-original, RM1-reduced and RM1-hyper when an object is embedded on the occlusal surface of the RM1 and loaded separately along the z-axis (LC1), x-axis (LC2) and y-axis (LC3) (Fig.5). For LC1, all models show lower tensile stresses than during maximum intercuspation contact, presumably because the load was less localized and more uniformly distributed on the whole occlusal surface. For LC2, all models experience tensile stresses on the lingual half of the root. For LC3, the stresses are concentrated on the occlusal surface and on the distal half of the root, where RM1-hyper shows lower magnitude than either RM1-original or RM1-reduced.
Figure 5.

The maximum principal stress (MPa) distribution for specimen RM1-original, RM1-reduced and RM1-hyper when loads are applied on the embedded object (see Fig.3) parallel to the z-axis (LC1), the x-axis (i.e. buccally; LC2) and the y-axis (i.e. mesially; LC3). Red spots represent tensile stresses. B, buccal; D, distal; L, lingual; M, mesial.
Discussion and Conclusions
In modern societies, taurodontism has been reported to account for less than about 5% of the human population, even though a wide discrepancy in the prevalence of the trait has been observed due to ethnic variations and different criteria used to interpret this trait (Jafarzadeh et al. 2008). Conversely, we observe a high frequency of taurodontism in Neandertal teeth, at least European Neandertals (Kupczik & Hublin, 2010), suggesting that the trait might confer biomechanical advantages for withstanding and distributing the occlusal loadings produced during the power stroke.
Using a digital approach that allows the same specimen to be compared (Le Moustier RM1) with its original morphology and different root morphologies, our results suggest that taurodontism does not improve dental biomechanics. In all simulations (including those with the pulp chamber filled with dentine), there are no meaningful differences in displacement and stress distribution in RM1-reduced and RM1-hyper digital models. The difference in tensile stress magnitude observed in the bifurcation is so low and localized that it is unlikely to require evolution towards taurodontism. Similarly, taurodontism does not seem to favour para-masticatory activities, as all simulations show the same patterns and almost the same magnitude of maximum principal stresses (except for a decrease in tensile stresses in RM1-hyper relative to load case 3 – LC3, i.e. pressure in the Y direction).
Excluding the biomechanical hypothesis, we are left with three potential explanations for the high frequency of taurodontism in Neandertals: either a selective advantage to prolong tooth longevity in a high attrition diet, and ultimately to preserve the health of the periodontium (Coon, 1962; Blumberg et al. 1971), or pleiotropic or genetic drift effects, as it was observed that mutation of some genes might affect root formation (e.g. Steele-Perkins et al. 2003). The former hypothesis (which might take place either by filling in the pulp chamber with secondary dentin or delaying the exposure of the bifurcation during the physiological eruption of worn teeth, which is required to maintain the occlusion between antagonists; Coon, 1962; Blumberg et al. 1971) is attractive, but it is at odd when considering that Late Pleistocene H. sapiens, including modern humans from pre-industrialized societies, were characterized by heavy tooth wear, a condition that changed little until very recently (Molnar, 1972; Kaifu, 2000; Kaifu et al. 2003; Kaidonis, 2008; Benazzi et al. 2013a). Despite that, modern humans do not show the high frequency of taurodontism typical of Neandertals (Kupczik & Hublin, 2010). Rather, nowadays taurodontism is recognized as a clinical challenge during endodontic treatment, owing to the variation in the size and shape of the pulp chamber, the complexity of the root canal system, the apically positioned canal orifices, requiring special handling during cavity preparation, root canal filling as well as tooth extraction (e.g. Jafarzadeh et al. 2008; Bharti et al. 2009; Manjunatha & Kovvuru, 2010; Marques-da-Silva et al. 2010; Bronoosh et al. 2012; Simsek et al. 2013; Radwan & Kim, 2014). Therefore, even though we cannot rule out the selective advantage of taurodontism for coping with a high attrition environment, it is reasonable to assume that the high frequency of this trait in Neandertals is the result of either the side effect of genetic conditions related to other phenotypic expressions under positive selection (pleiotropy), or (mechanically neutral) genetic drift effects that may have started with Homo heidelbergensis groups or even earlier. Indeed, relatively high percentages of hypotaurodontism were found in the Sima de los Huesos hominins (Martinón-Torres et al. 2012). The Pleistocene, particularly the late Middle Pleistocene and Late Pleistocene, were characterized by climatically unstable glacial periods with episodes of icecap and permafrost expansion, which ultimately modified the geography of Europe (van Andel & Tzedakis, 1996; Fletcher et al. 2010; Sanchez Goñi & Harrison, 2010). During these cold and dry conditions, Neandertal population sizes severely decreased (probably as a result of high local mortality rates) and environmental constraints reduced movements of populations between Europe, Central Asia and the Near East (Hublin, 2009; Hublin & Roebroeks, 2009). Periodic isolations, demographic collapses, and the retraction into warmer refugia might have resulted in genetic bottlenecks and drift effects, thus explaining the reduced morphological variability observed in European Neandertals (Weaver et al. 2007) and ultimately the high frequency of taurodontism in this group.
Clinical relevance
A final remark, with important implications for modern dentistry, concerns the results we obtained when filling the pulp chamber with dentine. This digital simulation, which mirrors general protocols followed by dentists during endodontic treatments when filling root canals with various sealing materials (e.g. amalgam, composite resins, mineral trioxide aggregate, calcium phosphate cement), shows that a filled pulp increases tooth stiffness (Table2), and ultimately tensile stresses in the crown (Fig.4b), augmenting the risk of tooth fracture. Indeed, several dentists have emphasized the risk of tooth fracture following endodontic treatments (e.g. Batur et al. 2013; Taha et al. 2014; Tennert et al. 2014), pointing out that intact natural teeth show superior fracture strength than endodontically treated teeth (Pradeep et al. 2013). Furthermore, we observed that the EDJ might play an important role to decrease tensile stresses in the crown, as also argued by other scholars (e.g. Zaslansky et al. 2006; Shimizu & Macho, 2007; Zaytsev & Panfilov, 2014). Therefore, we suggest preserving tooth structure wherever possible (ideally, using more elastic restorative materials that decrease tooth stiffness) to reduce tensile stresses in the crown and the resulting risk of tooth failure.
Acknowledgments
We are grateful to the Museum für Vor- und Frühgeschichte in Berlin and in particular to Almut Hoffmann for providing access to the original specimen and allowing the acquisition of μCT data of Le Moustier 1 skull fragments and mandible. The data are stored in the digital database of the Max-Planck Institute for Evolutionary Anthropology in Leipzig. This is publication no. 73 of the DFG Research Unit 771 ‘Function and performance enhancement in the mammalian dentition – phylogenetic and ontogenetic impact on the masticatory apparatus’.
Authors' contributions
S.B.: study concepts and design, and microCT data segmentation. S.B., H.N.N.: 3D volumetric mesh and finite element analysis (FEA). S.B., O.K.: occlusal fingerprint analysis (OFA). S.B., H.N.N., O.K., J.J.H.: data interpretation and writing of manuscript.
Supporting Information
Additional Supporting Information may be found in the online version of this article:
The maximum principal stress (MPa) distribution for specimen RM1-original, RM1-reduced (i.e. the bifurcation was moved upward) and RM1-hyper (i.e. the bifurcation was moved downward) without modelling the enamel-dentine junction (EDJ).
Difference of the maximum principal stress (%) between the crowns of the models with non-filled/filled pulp chambers based on 12 homologous nodes selected on the occlusal grooves.
Numbers of nodes and tetrahedral elements for each specimen.
References
- van Andel TH. Tzedakis PC. Palaeolithic landscapes of Europe and environs, 150 000–25 000 years ago: an overview. Quat Sci Rev. 1996;15:481–500. [Google Scholar]
- Antón SC. Ciochon RL. Biomechanical and other perspectives on the Neandertal face. In: Corruccini RS, editor; Integrative Paths to the Past. Englewood Cliffs: Prentice Hall; 1994. pp. 677–695. [Google Scholar]
- Bailey SE. A closer look at Neanderthal postcanine dental morphology: the mandibular dentition. Anat Rec. 2002;269:148–156. doi: 10.1002/ar.10116. [DOI] [PubMed] [Google Scholar]
- Bailey SE. A morphometric analysis of maxillary molar crowns of Middle-Late Pleistocene hominins. J Hum Evol. 2004;47:183–198. doi: 10.1016/j.jhevol.2004.07.001. [DOI] [PubMed] [Google Scholar]
- Bailey SE. Beyond shovel shaped incisors: Neandertal dental morphology in a comparative context. Period Biol. 2006;108:253–267. [Google Scholar]
- Batur YB. Erdemir U. Sancakli HS. The long-term effect of calcium hydroxide application on dentin fracture strength of endodontically treated teeth. Dent Traumatol. 2013;29:461–464. doi: 10.1111/edt.12037. [DOI] [PubMed] [Google Scholar]
- Benazzi S. Coquerelle M. Fiorenza L, et al., editors. Comparison of dental measurement systems for taxonomic assignment of first molars. Am J Phys Anthropol. 2011a;144:342–354. doi: 10.1002/ajpa.21409. [DOI] [PubMed] [Google Scholar]
- Benazzi S. Kullmer O. Grosse IR, et al., editors. Using occlusal wear information and finite element analysis to investigate stress distributions in human molars. J Anat. 2011b;219:259–272. doi: 10.1111/j.1469-7580.2011.01396.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Benazzi S. Kullmer O. Grosse IR, et al., editors. Brief communication: comparing loading scenarios in lower first molar supporting bone structure using 3D Finite Element Analysis. Am J Phys Anthropol. 2012;147:128–134. doi: 10.1002/ajpa.21607. [DOI] [PubMed] [Google Scholar]
- Benazzi S. Nguyen HN. Schulz D, et al., editors. The evolutionary paradox of tooth wear: simply destruction or inevitable adaptation? PLoS ONE. 2013a;8:e62263. doi: 10.1371/journal.pone.0062263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Benazzi S. Nguyen HN. Kullmer O, et al., editors. Unravelling the functional biomechanics of dental features and tooth wear. PLoS ONE. 2013b;8:e69990. doi: 10.1371/journal.pone.0069990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Benazzi S. Grosse IR. Gruppioni G, et al., editors. Comparison of occlusal loading conditions in a lower second premolar using three-dimensional finite element analysis. Clin Oral Investig. 2014;18:369–375. doi: 10.1007/s00784-013-0973-8. [DOI] [PubMed] [Google Scholar]
- Bharti R. Chandra A. Tikku AP, et al., editors. ‘Taurodontism’ an endodontic challenge: a case report. J Oral Sci. 2009;51:471–474. doi: 10.2334/josnusd.51.471. [DOI] [PubMed] [Google Scholar]
- Blumberg JE. Hylander WL. Goepp RA. Taurodontism: a biometric study. Am J Phys Anthropol. 1971;34:243–255. doi: 10.1002/ajpa.1330340208. [DOI] [PubMed] [Google Scholar]
- Bronoosh P. Haghnegahdar A. Dehbozorgi M. Prevalence of taurodontism in premolars and molars in the South of Iran. J Dent Res Dent Clin Dent Prospects. 2012;6:21–24. doi: 10.5681/joddd.2012.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheng YY. Cheung WL. Chow TW. Strain analysis of maxillary complete denture with three-dimensional finite element method. J Prosthet Dent. 2010;103:309–318. doi: 10.1016/S0022-3913(10)60064-9. [DOI] [PubMed] [Google Scholar]
- Clement AF. Hillson SW. Aiello LC. Tooth wear, Neanderthal facial morphology and the anterior dental loading hypothesis. J Hum Evol. 2012;62:367–376. doi: 10.1016/j.jhevol.2011.11.014. [DOI] [PubMed] [Google Scholar]
- Coon CS. The Origin of Races. New York: Alfred A Knopf, Inc; 1962. [Google Scholar]
- Dąbrowski P. Nowaczewska W. Stringer CB, et al., editors. A Neanderthal lower molar from Stajnia Cave, Poland. Homo. 2013;64:89–103. doi: 10.1016/j.jchb.2013.01.001. [DOI] [PubMed] [Google Scholar]
- Dejak B. Mlotkowski A. Romanowicz M. Strength estimation of different designs of ceramic inlays and onlays in molars based on the Tsai-Wu failure criterion. J Prosthet Dent. 2007;98:89–100. doi: 10.1016/S0022-3913(07)60042-0. [DOI] [PubMed] [Google Scholar]
- Fiorenza L. Kullmer O. Dental wear and cultural behavior in Middle Paleolithic humans from the Near East. Am J Phys Anthropol. 2013;152:107–117. doi: 10.1002/ajpa.22335. [DOI] [PubMed] [Google Scholar]
- Fletcher WJ. Sánchez Goñi MF. Allen JRM, et al., editors. Millennial-scale variability during the last glacial in vegetation records from Europe. Quat Sci Rev. 2010;29:2839–2864. [Google Scholar]
- Fu G. Deng F. Wang L, et al., editors. The three-dimension finite element analysis of stress in posterior tooth residual root restored with postcore crown. Dent Traumatol. 2010;26:64–69. doi: 10.1111/j.1600-9657.2009.00829.x. [DOI] [PubMed] [Google Scholar]
- Gorjanović-Kramberger D. Die Kronen und Wurzeln der Mahlzähne primigenius und ihre genetische Bedeutung. Anat Anz. 1907;31:97–134. [Google Scholar]
- Gorjanović-Kramberger D. Über prismatische Molarwurzeln rezent er und diluvialer Menschen. Anat Anz. 1908;32:401–413. [Google Scholar]
- Hamner JE. Witkop CJ. Metro PS. Taurodontism; report of a case. Oral Surg Oral Med Oral Pathol. 1964;18:409–418. doi: 10.1016/0030-4220(64)90097-0. [DOI] [PubMed] [Google Scholar]
- Hattori Y. Satoh C. Kunieda T, et al., editors. Bite forces and their resultants during forceful intercuspation clenching in humans. J Biomech. 2009;42:1533–1538. doi: 10.1016/j.jbiomech.2009.03.040. [DOI] [PubMed] [Google Scholar]
- Hohmann A. Kober C. Young P, et al., editors. Influence of different modeling strategies for the periodontal ligament on finite element simulation results. Am J Orthod Dentofacial Orthop. 2011;139:775–783. doi: 10.1016/j.ajodo.2009.11.014. [DOI] [PubMed] [Google Scholar]
- Holmes DC. Diaz-Arnold AM. Leary JM. Influence of post dimension on stress distribution in dentin. J Prosthet Dent. 1996;75:140–147. doi: 10.1016/s0022-3913(96)90090-6. [DOI] [PubMed] [Google Scholar]
- Hublin JJ. Out of Africa: modern human origins special feature: the origin of Neandertals. Proc Natl Acad Sci U S A. 2009;106:16022–16027. doi: 10.1073/pnas.0904119106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hublin JJ. Roebroeks W. Ebb and flow or regional extinctions? On the character of Neandertal occupation of northern environments. Pal Evol. 2009;8:503–509. [Google Scholar]
- Hylander WL. Graber TM. In: Orofacial Growth and Development. Dahlberg AA, editor; Chicago: Mouton Publishers; 1977. pp. 129–169. [Google Scholar]
- Jafarzadeh H. Azarpazhooh A. Mayhall JT. Taurodontism: a review of the condition and endodontic treatment challenges. Int Endod J. 2008;41:375–388. doi: 10.1111/j.1365-2591.2008.01388.x. [DOI] [PubMed] [Google Scholar]
- Kaidonis JA. Tooth wear: the view of the anthropologist. Clin Oral Investig. 2008;12:21–26. doi: 10.1007/s00784-007-0154-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaifu Y. Was extensive tooth wear normal in our ancestors? A preliminary examination in the genus Homo. Anthropol Sci. 2000;108:371–385. [Google Scholar]
- Kaifu Y. Kasai K. Townsend GC, et al., editors. Tooth wear and the ‘design’ of the human dentition: a perspective from evolutionary medicine. Am J Phys Anthropol Suppl. 2003;37:47–61. doi: 10.1002/ajpa.10329. [DOI] [PubMed] [Google Scholar]
- Keith A. Problems relating to the teeth of the earlier forms of prehistorical man. Proc R Soc Med. 1913;6:103–119. doi: 10.1177/003591571300601018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kinney JH. Balooch M. Marshall GW, et al., editors. A micromechanics model of the elastic properties of human dentine. Arch Oral Biol. 1999;44:813–822. doi: 10.1016/s0003-9969(99)00080-1. [DOI] [PubMed] [Google Scholar]
- Ko CC. Chu CS. Chung KH, et al., editors. Effects of posts on dentin stress distribution in pulpless teeth. J Prosthet Dent. 1992;68:421–427. doi: 10.1016/0022-3913(92)90404-x. [DOI] [PubMed] [Google Scholar]
- Kovacs I. A systematic description of dental roots. In: Dahlberg AA, editor. Dental Morphology and Evolution. Chicago: The University of Chicago Press; 1971. pp. 211–256. [Google Scholar]
- Kullmer O. Benazzi S. Fiorenza L, et al., editors. Technical note: Occlusal Fingerprint Analysis (OFA): quantification of tooth wear pattern. Am J Phys Anthropol. 2009;139:600–605. doi: 10.1002/ajpa.21086. [DOI] [PubMed] [Google Scholar]
- Kullmer O. Benazzi S. Schulz D, et al., editors. Dental arch restoration using tooth macrowear patterns with application to Rudapithecus hungaricus, from the late Miocene of Rudabánya, Hungary. J Hum Evol. 2013;64:151–160. doi: 10.1016/j.jhevol.2012.10.009. [DOI] [PubMed] [Google Scholar]
- Kupczik K. Dean MC. Comparative observations on the tooth root morphology of Gigantopithecus blacki. J Hum Evol. 2008;54:196–204. doi: 10.1016/j.jhevol.2007.09.013. [DOI] [PubMed] [Google Scholar]
- Kupczik K. Hublin J-J. Mandibular molar root morphology in Neanderthals and Late Pleistocene and recent Homo sapiens. J Hum Evol. 2010;59:525–541. doi: 10.1016/j.jhevol.2010.05.009. [DOI] [PubMed] [Google Scholar]
- Lebel S. Trinkaus E. Middle Pleistocene human remains from the Bau de l'Aubesier. J Hum Evol. 2002;43:659–685. doi: 10.1006/jhev.2002.0598. [DOI] [PubMed] [Google Scholar]
- Li H. Zhou ZR. Wear behavior of human teeth in dry and artificial saliva conditions. Wear. 2002;249:980–984. [Google Scholar]
- Lucas PW. Dental Functional Morphology. How Teeth Work. Cambridge: Cambridge University Press; 2004. [Google Scholar]
- Macchiarelli R. Bondioli L. Mazurier A. Nelson GC. Virtual dentitions: touching the hidden evidence. In: Irish JD, editor; Technique and Application in Dental Anthropology. Cambridge: Cambridge University Press; 2008. pp. 426–448. [Google Scholar]
- Magne P. Efficient 3D finite element analysis of dental restorative procedures using micro-CT data. Dent Mater. 2007;23:539–548. doi: 10.1016/j.dental.2006.03.013. [DOI] [PubMed] [Google Scholar]
- Manjunatha BS. Kovvuru SK. Taurodontism – a review on its etiology, prevalence and clinical considerations. J Clin Exp Dent. 2010;2:187–190. [Google Scholar]
- Marques-da-Silva B. Baratto-Filho F. Abuabara A, et al., editors. Multiple taurodontism: the challenge of endodontic treatment. J Oral Sci. 2010;52:653–658. doi: 10.2334/josnusd.52.653. [DOI] [PubMed] [Google Scholar]
- Marshall GW., Jr Balooch M. Gallagher RR, et al., editors. Mechanical properties of the dentinoenamel junction: AFM studies of nanohardness, elastic modulus, and fracture. J Biomed Mater Res. 2001;54:87–95. doi: 10.1002/1097-4636(200101)54:1<87::aid-jbm10>3.0.co;2-z. [DOI] [PubMed] [Google Scholar]
- Martinón-Torres M. Bermúdez de Castro JM. Gómez-Robles A, et al., editors. Morphological description and comparison of the dental remains from Atapuerca-Sima de los Huesos site (Spain) J Hum Evol. 2012;62:7–58. doi: 10.1016/j.jhevol.2011.08.007. [DOI] [PubMed] [Google Scholar]
- Molnar S. Tooth wear and culture: a survey of tooth functions among some prehistoric populations. Curr Anthrop. 1972;13:511–526. [Google Scholar]
- O'Connor CF. Franciscus RG. Holton NE. Bite force production capability and efficiency in Neandertals and modern humans. Am J Phys Anthropol. 2005;127:129–151. doi: 10.1002/ajpa.20025. [DOI] [PubMed] [Google Scholar]
- Pérez-González A. Iserte-Vilar JL. González-Lluch C. Interpreting finite element results for brittle materials in endodontic restorations. Biomed Eng Online. 2011;2011:10–44. doi: 10.1186/1475-925X-10-44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pradeep P. Kumar VS. Bantwal SR, et al., editors. Fracture strength of endodontically treated premolars: an in-vitro evaluation. J Int Oral Health. 2013;5:9–17. [PMC free article] [PubMed] [Google Scholar]
- Radwan A. Kim SG. Treatment of a hypertaurodontic maxillary second molar in a patient with 10 taurodonts: a case report. J Endod. 2014;40:140–144. doi: 10.1016/j.joen.2013.08.017. [DOI] [PubMed] [Google Scholar]
- Rak Y. The Neanderthal: a new look at an old face. J Hum Evol. 1986;15:151–164. [Google Scholar]
- Rubin C. Krishnamurthy N. Capilouto E, et al., editors. Stress analysis of the human tooth using a three-dimensional finite element model. J Dent Res. 1983;62:82–86. doi: 10.1177/00220345830620021701. [DOI] [PubMed] [Google Scholar]
- Sanchez Goñi MF. Harrison SP. Millennial-scale climate variability and vegetation changes during the Last Glacial: concepts and terminology. Quat Sci Rev. 2010;29:2823–2827. [Google Scholar]
- Shimizu D. Macho GA. Functional significance of the microstructural detail of the primate dentino-enamel junction: a possible example of exaptation. J Hum Evol. 2007;52:103–111. doi: 10.1016/j.jhevol.2006.08.004. [DOI] [PubMed] [Google Scholar]
- Simsek N. Keles A. Ocak MS. Endodontic treatment of hypertaurodontism with multiple bilateral taurodontism. J Conserv Dent. 2013;16:477–479. doi: 10.4103/0972-0707.117497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith FH. Paquette SP. The adaptive basis of Neandertal facial form, with some thoughts on the nature of modern human origins. In: Trinkaus E, editor. The Emergence of Modern Humans. Cambridge: Cambridge University Press; 1989. pp. 181–210. [Google Scholar]
- Spencer MA. Tooth-root form and function in platyrrhine seed-eaters. Am J Phys Anthropol. 2003;122:325–335. doi: 10.1002/ajpa.10288. [DOI] [PubMed] [Google Scholar]
- Spencer MA. Demes B. Biomechanical analysis of masticatory system configuration in Neandertals and Inuits. Am J Phys Anthropol. 1993;91:1–20. doi: 10.1002/ajpa.1330910102. [DOI] [PubMed] [Google Scholar]
- Steele-Perkins G. Butz KG. Lyons GE, et al., editors. Essential role for NFI-C/CTF transcription-replication factor in tooth root development. Mol Cell Biol. 2003;23:1075–1084. doi: 10.1128/MCB.23.3.1075-1084.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Taha N. Palamara J. Messer H. Fracture strength and fracture patterns of root-filled teeth restored with direct resin composite restorations under static and fatigue loading. Oper Dent. 2014;39:181–188. doi: 10.2341/13-006-L. [DOI] [PubMed] [Google Scholar]
- Tennert C. Eismann M. Goetz F, et al., editors. A temporary filling material used for coronal sealing during endodontic treatment may cause tooth fractures in large class II cavities in vitro. Int Endod J. 2014 doi: 10.1111/iej.12280. doi: 10.1111/iej.12280. [DOI] [PubMed] [Google Scholar]
- Trinkaus E. The Neandertal face: evolutionary and functional perspectives on a recent hominid face. J Hum Evol. 1987;16:429–443. [Google Scholar]
- Walker MJ. Gibert J. López MV, et al., editors. Late Neandertals in Southeastern Iberia: sima de las palomas del cabezo gordo, Murcia, Spain. Proc Natl Acad Sci U S A. 2008;105:20631–20636. doi: 10.1073/pnas.0811213106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang Q. Wright BW. Smith A, et al., editors. Mechanical impact of incisor loading on the primate midfacial skeleton and its relevance to human evolution. Anat Rec. 2010;293:607–617. doi: 10.1002/ar.21123. [DOI] [PubMed] [Google Scholar]
- Weaver TD. Roseman CC. Stringer CB. Were neandertal and modern human cranial differences produced by natural selection or genetic drift? J Hum Evol. 2007;53:135–145. doi: 10.1016/j.jhevol.2007.03.001. [DOI] [PubMed] [Google Scholar]
- Wright T. The molecular control of and clinical variations in root formation. Cells Tissues Organs. 2007;186:86–93. doi: 10.1159/000102684. [DOI] [PubMed] [Google Scholar]
- Xia Z. Jiang F. Chen J. Estimation of periodontal ligament's equivalent mechanical parameters for finite element modeling. Am J Orthod Dentofacial Orthop. 2013;143:486–491. doi: 10.1016/j.ajodo.2012.10.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zaslansky P. Friesem AA. Weiner S. Structure and mechanical properties of the soft zone separating bulk dentin and enamel in crowns of human teeth: insight into tooth function. J Struct Biol. 2006;153:188–199. doi: 10.1016/j.jsb.2005.10.010. [DOI] [PubMed] [Google Scholar]
- Zaytsev D. Panfilov P. Deformation behavior of human enamel and dentin-enamel junction under compression. Mater Sci Eng C Mater Biol Appl. 2014;34:15–21. doi: 10.1016/j.msec.2013.10.009. [DOI] [PubMed] [Google Scholar]
- Zaytsev D. Grigoriev S. Panfilov P. Deformation behavior of human dentin under uniaxial compression. Int J Biomater. 2012;2012:854539. doi: 10.1155/2012/854539. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
The maximum principal stress (MPa) distribution for specimen RM1-original, RM1-reduced (i.e. the bifurcation was moved upward) and RM1-hyper (i.e. the bifurcation was moved downward) without modelling the enamel-dentine junction (EDJ).
Difference of the maximum principal stress (%) between the crowns of the models with non-filled/filled pulp chambers based on 12 homologous nodes selected on the occlusal grooves.
Numbers of nodes and tetrahedral elements for each specimen.


