Abstract
Metformin has recently been introduced as an anti-cancer agent. In this study, we evaluated the effect of metformin and metformin/cisplatin on human gastric MKN-45 cell line. When we used metformin alone, it could inhibit proliferation and induce apoptosis, but it diminish anti-proliferative effects of cisplatin when they are used in combination. Further, we checked mRNA levels of survivin, mTOR, and Akt by real-time PCR. When MKN-45 cells were treated with metformin/cisplatin, the expression of survivin and mTOR were increased. The antagonistic effect of metformin on cisplatin could be through survivin and mTOR signaling pathways. Our results also suggest that interfering effect of metformin on cisplatin may be also through upregulation of Akt. Regarding the pivotal role of Akt in drug resistance, it may be reasonable to conclude that the antagonistic effect of metformin on cisplatin effect may be through this central mediator of drug resistance. Taken together, it seems that metformin is not a good option for sensitizing MKN-45 cell line to cisplatin, and in co-prescription of metformin and cisplatin in gastric cancer patients who suffer diabetes type 2, it should be highly cared.
Keywords: Metformin, Cisplatin, Gastric cancer, Survivin, Akt, mTOR
INTRODUCTION
Metformin is an oral drug traditionally used for the treatment of many diseases including type 2 diabetes1 and polycystic ovary syndrome2 and belongs to biguanides. Metformin is the most common antidiabetic agent in the world with the 48 million prescriptions in the US in 2010.3 It has been widely suggested that metformin works against diabetes by reducing glucose production in the liver. It is frequently reported that metformin stimulates the intracellular enzyme AMP-activated protein kinase (AMPK), leading to inhibition of gluconeogenesis in liver and increasing glucose uptake by peripheral tissues.4, 5 In addition, metformin is the only antidiabetic drug which can act against cardiovascular disorders.1
In addition, it has been suggested that metformin could decrease the risk of many cancers including colorectal, liver, and pancreas in the diabetic patients which are using this drug.6-9 Moreover, population studies have suggested that metformin decreases the incidence of cancer and cancer-related mortality in diabetic patients.6 In addition, recent studies have demonstrated that metformin could induce apoptosis and inhibit proliferation of many cancer cell types in vitro like breast and ovary cancer cells,10, 11 as well as tumorigenesis and tumor progression in vivo.12, 13 On the other hand, it has been demonstrated that metformin improves the response to chemotherapy in diabetic patients with breast cancer.14 The detailed mode of action of metformin in cancer cells has not yet understood very well. However, several reports have demonstrated that this therapeutic agent induces apoptosis through LKB1-mediated AMPK signaling pathway.15-17
Cisplatin is a chemotherapeutic agent which is usually used as a treatment for various types of cancers including sarcomas, lymphomas, germ cell tumors, and many carcinomas e.g. ovarian cancer, small cell lung cancer and gastric cancer.18 Cisplatin is an apoptotic agent and also crosslinks DNA in several different ways, interfering with cell division by mitosis.18 The major mechanism of cisplatin anticancer activity is interaction with purine bases in DNA, which forms DNA–protein, DNA–DNA inter-strand and intra-strand crosslinks that cause blocking proliferation of tumor cells and inducing apoptosis.19
The ability of metformin to improve the efficacy of chemotherapy by cisplatin is shown in some types of cancer cells such as adenocarcinoma cell lung cancer20 and ovarian carcinomas.21 In addition, it has been demonstrated that metformin acts synergistically with cisplatin in decreasing tumor size and inhibiting angiogenesis in mouse.9 All these findings make it a plausible candidate for combination with cisplatin-based therapy. On the other hand, Jaevotic et al.,22 and Chang et al.,23 reported that metformin antagonizes the cisplatin apoptotic effect through suppression of oxidative stress and inhibition of caspases activation in some cancer cells, which is not in line with the previous studies. Regarding these results and the facts that cisplatin is a drug traditionally used for the treatment of gastric cancer and the cytotoxic efficiency of cisplatin/metformin combination has not yet been clearly assessed in gastric cells; this question may be emerged if this drug antagonism could be a challenge for gastric cancer patients with diabetes type 2 which are treated with both cisplatin and metformin. The results may shed light on mechanistic detail of the combination use of these therapeutic agents in patients with gastric cancer and diabetes type 2 simultaneously.
Gastric cancer is the fourth common cancer and the second-leading cause of death in the world with more than 800 thousands death annually.24 Regarding the previous reports on anticancer efficacy of metformin and the importance of gastric cancer as the paramount cause of death, evaluation the effects of metformin, alone or in combination with other drugs, remains the main priority. In this study, we evaluated the combination effect of metformin and cisplatin on human gastric cancer MKN-45 cell line.
MATERIALS AND METHODS
Cell culture and treatment
The human gastric MKN-45 cell line was obtained from Pasteur Institute of Iran (Tehran, Iran). The mentioned cell line was cultured in RPMI 1640 medium (Invitrogen) supplemented with 10% fetal bovine serum (Invitrogen) in 5% CO2 at 37°C. The cultures were then treated with 5, 10, and 20 mM concentrations of metformin and 0.1, 1, 2.5, 5 and 10 μM concentrations of cisplatin (Sigma, St. Louis, Missouri, USA) for 24, 48 and 72 h.
Microculture tetrazolium (MTT) assay
Microculture tetrazolium (MTT) assay was used to evaluate the inhibitory effect of metformin and cisplatin on viability of MKN-45 cells. For this, seeded cells were plated in 96-well plates at density of 5000 cells/100 μl PRMI in each well and then exposed to either control or varying concentrations of metformin and/or cisplatin for 24, 48, and 72 hrs. Thereafter, the control medium and the treated media were replaced by MTT solution (0.5mg/ml) and DMSO (after 3 h), respectively. The color absorbance was finally read at wave length of 578 nm in an ELISA reader. The percentage cell viability was calculated as (%) = (ODexp/ODcon) ×100, where ODexp and ODcon are the optical densities of exposed and control cells, respectively.
Analysis of gene expression by real-time PCR
For real-time PCR, RNA was first extracted by Fast Pure RNA kit (Takara Bio Inc., Otsu, Japan) from cultured cells and quantified by Nanodrop ND-1000 (Nanodrop Technologies, Wilmington, Delaware, USA). Then, 1 μg of RNA from each sample was applied to reverse transcription, using the Prime Script RT reagent kit (Takara Bio). Real-time PCR was performed with a light cycler instrument (Roche Diagnostics, Germany), using SYBR Premix Ex Taq technology (Takara Bio Inc.). SYBR Green master mix (10μl) was added to 2 μl of cDNA samples, 0.5 μl of forward and reverse primers (10pmol) in water and 7 μl of nuclease-free water (Qiagen, Hilden, Germany) to conduct PCR in 20 μl of reaction mixture. Thermal cycling conditions involved an initial activation step for 30 s at 95°C, followed by 40 cycles including a denaturation step for 5 s at 95°C and a combined annealing/extension step for 20 s at 60°C. The sequences of primers are listed in Table 1. Melting curves were analyzed to validate single PCR product of each primer. Hypoxanthine phosphoribosyltransferase1 (HPRT1) was amplified as internal control and the fold change in relative expression of each target mRNA was calculated on the basis of comparative Ct (2-ΔΔCt) method.
Table 1.
Nucleotide sequences of the primers used for real-time RT-PCR
| Gene | Accession number | Forward primer (5’–3’) | Reverse primer (5’–3’) | Size |
|---|---|---|---|---|
| HPRT | NM_000194 | TGGACAGGACTGAACGTCTTG | CCAGCAGGTCAGCAAAGAATTTA | 111 |
| Akt | NM_005163 | AGCGACGTGGCTATTGTGAAG | GTACTCCCCTCGTTTGTGCAG | 51 |
| Survivin | NM_001168 | CCAGATGACGACCCCATAGAG | TTGTTGGTTTCCTTTGCAATTTT | 152 |
| mTOR | NM_004958 | AACTCCGAGAGATGAGTCAAGA | AGTTGGTCATAGAAGCGAGTAGA | 49 |
| Caspase-3 | NM_032991 | ATGGAAGCGAATCAATGGACTC | CTGTACCAGACCGAGATGTCA | 138 |
Quantification of apoptosis by flow cytometry
For quantization of apoptosis in MKN-45 cells treated with cisplatin and metformin, a double staining kit with Hoechst 33342 and propidium iodide (PI) was used (Invitrogen). After treatment of cells with desired concentrations of cisplatin, metformin, and combination of these drugs for 48h, the cells were harvested and washed in cold PBS and the cell density was adjusted to 0.5 ×106 cells/ml in PBS. For each sample, 1 ml volume was used. 1 μl of the Hoechst 33342 stock solution (5.0 mg/ml solution in water) and 1 μl of the propidium iodide stock solution (1.0 mg/ml solution in water) were added to each 1 ml of cell suspension. After 15 min, the stained cells were evaluated by flow cytometry instrument (PartecPasIII, Germany), using excitation/emission ∼350/461 and ∼535/617 nm for Hoechst 33342 and PI, respectively. The data were then analyzed, using FlowMax software.
RESULTS
Metformin suppresses anti-cancer efficacy of cisplatin in MKN-45 cell line
To evaluate single and combinational effects of metformin and cisplatin in MKN-45 cell line, we treated this cell line with 5 and 10 mM metformin and 0.1, 1, 2.5, 5, and 10 μM cisplatin either alone or in combination at 48 and 72 h intervals. Our results demonstrated that both drugs could inhibit the viability of MKN-45 cell line when they were used separately. However, in combination treatment metformin suppressed the cytotoxicity effect of cisplatin (Fig. 1). The data were analyzed with the calcusin software and the combination indexes (CIs) were obtained as shown in Tables 2.
Figure 1.
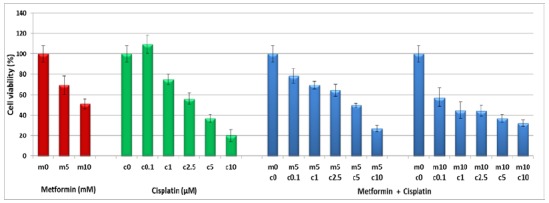
Evaluation of metformin, cisplatin and metformin /cisplatin on the viability of human adenocarcinoma cell lines MKN-45. The cell line was treated with different concentrations of metformin and/or cisplatin for 48 and 72 hrs and their viability was assessed using MTT assay. Results are expressed as a percentage of viability compared to the untreated control and are presented as mean±SD from three independent experiments (P<0.05 by one-way variance analysis)
Table 2.
Combined indexes for cisplatin and metformin in MKN-45 cell line
| Concentrations | Metformin/Cisplatin Combination Index (CI) | ||
|---|---|---|---|
| Metformin (mM) | Cisplatin(μM) | 48 hrs | 72 hrs |
| 5 | 0.1 | 1.304 | 1.591 |
| 5 | 1 | 1.205 | 1.470 |
| 5 | 2.5 | 1.099 | 1.852 |
| 5 | 5 | 1.309 | 1.945 |
| 5 | 10 | 1.147 | 1.914 |
| 10 | 0.1 | 1.416 | 1.291 |
| 10 | 1 | 1.133 | 1.056 |
| 10 | 2.5 | 1.403 | 1.537 |
| 10 | 5 | 1.078 | 1.694 |
| 10 | 10 | 1.183 | 2.447 |
Metformin reduces cisplatin-mediated apoptosis in MKN-45 cell line
To evaluate the effects of metformin, cisplatin and metformin/cisplatin on apoptosis in MKN-45 cell line, we used flow cytometry. As shown in Fig. 2, the analyzed data obtained from FlowMax software revealed that cisplatin could induce higher apoptosis compared to the untreated control either in concentration 1 μM (P<0.05) or 5 and 10 μM (P<0.01). However, the apoptosis ratio was reduced in the combination treatments as compared to the cisplatin alone. The induction of apoptosis in combination of metformin 10/cisplatin 5 was significantly lower than cisplatin alone at concentration of 5 μM (P<0.05). This data indicate that metformin antagonizes with cisplatin-induced apoptosis.
Figure 2.
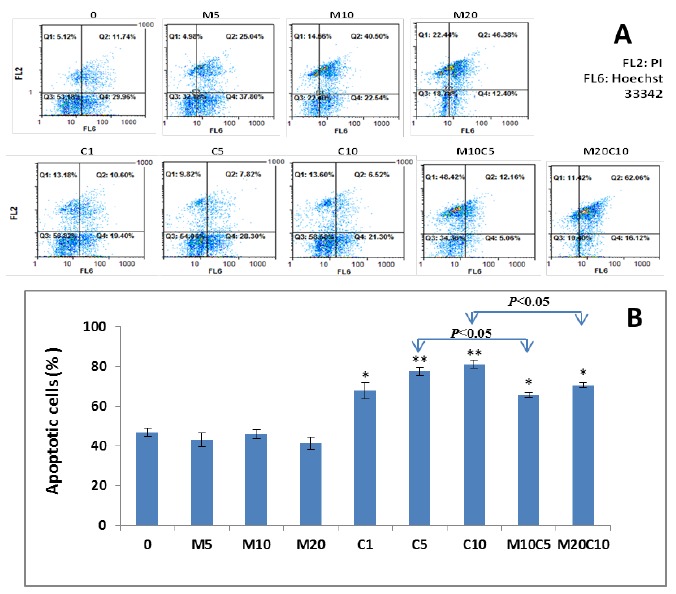
Effect of metformin, cisplatin and metformin/cisplatin on apoptosis in MKN-45 cells. A) Hoechst 33342 stains the condensed chromatin in apoptotic cells more brightly than the chromatin in normal cells and Propidium Iodide (PI) is only permeate to dead cells (FL2: PI, FL6: Hoechst 33342). As shown in figure, there is significant change in the apoptosis rate of metformin, cisplatin and metformin/cisplatin treated MKN-45 cells compared with the control. B) Quantified values of apoptosis in MKN-45 cell line treated with metformin, cisplatin and metformin/cisplatin. As shown in this figure, the cisplatin-induced apoptosis at concentration of 5 μM is significantly higher than the combination of metformin 10 /cisplatin 5. Statistically different values of *P<0.05 and **P<0.01 are determined compared with the control
Effects of Survivin, Akt and mTOR expressions on antagonistic behavior of metformin and cisplatin
To evaluate molecular mechanisms of antagonistic behavior of metformin and cisplatin in MKN-45 cell line, the expressions of survivin, Akt, and mTOR were assessed in cells treated with the mentioned concentrations of metformin and cisplatin at transcriptional level. Previous reports demonstrated that survivin has a paramount role in resistance of MKN-45 to chemotherapy with cisplatin.25 As shown in Fig. 3, our real-time PCR results indicated that treatment with metformin or cisplatin for 48h significantly reduced the expression of survivin. However, metformin/cisplatin leads to an increase in the expression of survivin compared to the treatment with cisplatin (P<0.05).
Figure 3.
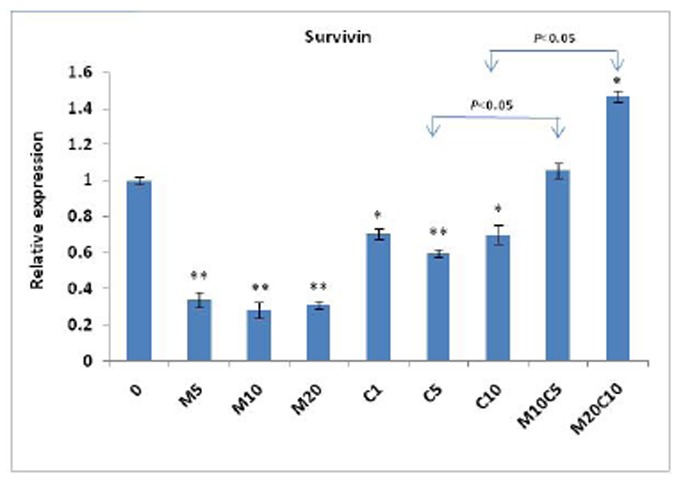
Effect of metformin, cisplatin and metformin/cisplatin on transcriptional levels of the survivin gene measured by real-time PCR. Data are shown as fold change in relative expression compared with HPRT1 on the basis of comparative Ct (2-ΔΔCt) method. As shown in fig, treatment with metformin or cisplatin for 48 h significantly reduces expression of survivin. However, metformin/cisplatin leads to increased expression of survivin compared to treatment with cisplatin. Values are shown as mean ± SD. Statistically different values of *P<0.05 and **P<0.01 are determined compared with the control
We further evaluated transcriptional expression of Akt and mTOR, key mediators of resistance to chemotherapy by cisplatin, to see if these genes are involved in antagonistic behavior of metformin on cisplatin. Our results indicated that the expression of Akt was significantly increased (P<0.05) in the cells treated with metformin/cisplatin, compared to both drugs separately (Fig. 4). This finding was also observed in the case of mTOR expression (P<0.05), which may be a further document for antagonistic behavior of metformin and cisplatin (Fig. 5).
Figure 4.
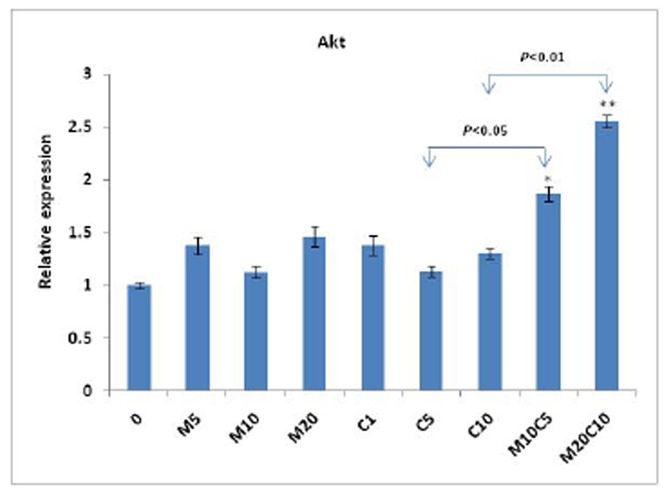
Effect of metformin, cisplatin and metformin/cisplatin on transcriptional levels of the Akt gene measured by real-time PCR. As shown in figure, when the cell line was treated with metformin/cisplatin, the expression of Akt is significantly increased compared to drugs separately. Values are shown as mean ± SD. Statistically different values of *P<0.05 and **P<0.01 are determined compared with the control
Figure 5.
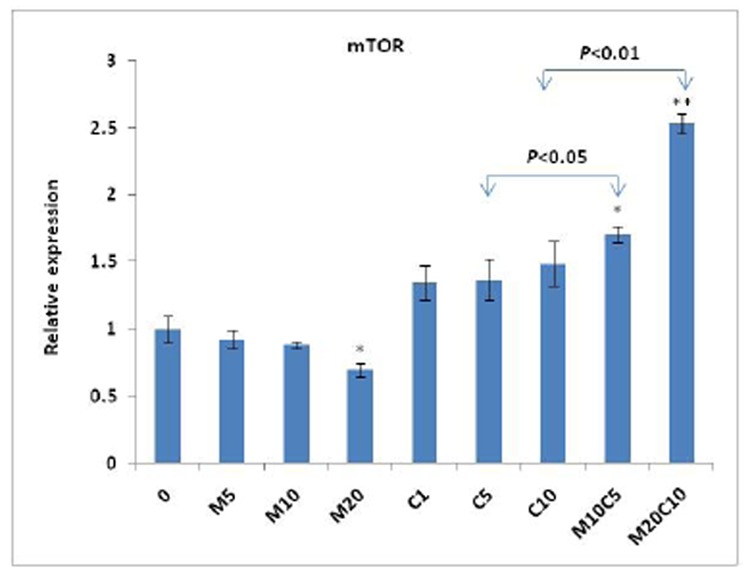
Effect of metformin, cisplatin and metformin/cisplatin on transcriptional levels of the mTOR gene measured by real-time PCR. Treatment with metformin/cisplatin results inan increase in the expression of mTOR compared to treatment with cisplatin. Values are shown as mean±SD. Statistically different values of *P<0.05 and **P<0.01 are determined compared with the control
DISCUSSION
Frequent studies have recently shown that metformin could decrease the risk of many cancers including colorectal and pancreatic cancers.6-9 In addition, the effect of metformin on inhibition of proliferation and induction of apoptosis has been reported in vitro in breast and ovarian cancer cells 10, 11 and in vivo against tumorigenesis and angiogenesis.12, 13 It has also been reported in some studies that metformin could enhance the anti-cancer effect of cisplatin in breast and ovarian cancers.9, 14, 21 On the other hand, Jaevotic et al., showed that metformin could diminish the antineoplastic effect of SHY5Y, C6, U251, L929, and HL-60 cell lines through suppression of oxidative stress and inhibition of caspases activation 22 which is not in line with previous reports. Thus, it is reasonable to conclude that the effect of metformin on cisplatin may be dependent to the cell type and possibly to the type of cancer. In this study, we evaluated the combinational effect of metformin and cisplatin on gastric cancer MKN-45 cell line. As a result of MTT and flow cytometry, metformin antagonizes the effect of cisplatin in MKN-45 cell line. Moreover, this study suggests that this antagonism may be through up-regulation in transcription of survivin. Survivin is detected during mitosis and specifically binds to terminal effector cell death proteases, caspase 3 and caspase 7, thus decreases apoptosis.25 It has been reported that over expression of survivin suppresses etoposide-induced apoptosis.26 Furthermore, it has been reported that survivin mRNA level in patients with esophageal cancer who respond to chemotherapy is much less than patients who are resistant to chemotherapy.27 In line with this observation, other reports suggest that survivin mRNA and protein expression are correlated with unfavorable prognosis in many malignancies.28-30 Hence, it is concluded that survivin expression has a paramount importance in resistance to chemotherapy.
It is currently obvious that metformin activates AMPK.31 AMPK, in phosphorylated status, inhibits mTOR through TSC1 and TSC2.31 Inhibition of mTOR is observed in our study as well. As we know, mTOR inhibition leads to inhibition of survivin and thus, mTOR could decrease cell viability and induce apoptosis through suppression of survivin expression. Even though metformin treatment causes a reduction in mTOR and survivin expression in this study, the combination treatments with metformin and cisplatin increase the expression of the mentioned genes. So, it may be reasonable to conclude that antagonistic behavior of metformin and cisplatin may be dependent on the survivin and mTOR induction. In the case of an increase in the expression of survivin in combinational treatment, it should be noticed that the previous study showed that an increase in the expression of survivin in MKN-45 cell line made it resistant to the chemotherapy.25
In addition, our study suggests that antagonistic effect of metformin on cisplatin-induced cell toxicity could also be through increase in the expression of Akt. Akt is a serin-threonin kinase which has a paramount importance in cell survival and apoptosis. This protein exerts its antiapoptotic effect directly through phosphorylation and inactivation of Bad, forkhead transcription factors, c-raf, and caspase 932-35 and indirectly through activation of NF-κB, and prosurvival proteins.36 Akt is highly active in chemoresistant tumors37 and it is suggested that inhibition of this pathway sensitizes cancer cells to chemotherapy.38, 39 In line with these findings, we demonstrated that antiapoptotic activity of metformin in combination treatment with cisplatin may be through inhibition of cisplatin-induced reduction of Akt expression. The results of the present study suggest that the cells treated with combination of drugs show an increase in the expression level of Akt as compared to the treatment with metformin or cisplatin. Likewise, the theory that protective effect of metformin is through suppression of Akt expression has been confirmed with Akt specific inhibitors which result in suppression of antiapoptotic effect of metformin.31 Finally, regarding the results of previous reports and the present study, it seems that metformin may not be a suitable drug for sensitization of most cancers to cisplatin, and co-prescription of metformin and cisplatin in gastric cancer patients who suffer diabetes type 2 should be highly cared. Further in vitro and in vivo studies are required to enlighten detailed action mode of combinational effects of these therapeutic agents on gastric cancer, as well as other cancers.
Footnotes
CONFLICT OF INTEREST
All authors declare no conflict of interest.
References
- 1.Basevi V, et al. Comment on: American Diabetes Association. Standards of medical care in diabetes--2011. Diabetes Care. 2011;34(Suppl. 1):S11–S61. doi: 10.2337/dc11-0174. [DOI] [PMC free article] [PubMed] [Google Scholar]; Diabetes Care. 2011;34(5):e53. doi: 10.2337/dc11-0174. author reply e54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Moll E, van der Veen F, van Wely M. The role of metformin in polycystic ovary syndrome: a systematic review. Hum Reprod Update. 2007;13(6):527–37. doi: 10.1093/humupd/dmm026. [DOI] [PubMed] [Google Scholar]
- 3.Ravandi F, et al. Effective treatment of acute promyelocytic leukemia with all-trans-retinoic acid, arsenic trioxide, and gemtuzumab ozogamicin. J Clin Oncol. 2009;27(4):504–10. doi: 10.1200/JCO.2008.18.6130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Correia S, et al. Mechanisms of action of metformin in type 2 diabetes and associated complications: an overview. Mini Rev Med Chem. 2008;8(13):1343–54. doi: 10.2174/138955708786369546. [DOI] [PubMed] [Google Scholar]
- 5.Zhou G, et al. Role of AMP-activated protein kinase in mechanism of metformin action. J Clin Invest. 2001;108(8):1167–74. doi: 10.1172/JCI13505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Evans JM, et al. Metformin and reduced risk of cancer in diabetic patients. BMJ. 2005;330(7503):1304–5. doi: 10.1136/bmj.38415.708634.F7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Chong CR, Chabner BA. Mysterious metformin. Oncologist. 2009;14(12):1178–81. doi: 10.1634/theoncologist.2009-0286. [DOI] [PubMed] [Google Scholar]
- 8.Lee MS, et al. Type 2 diabetes increases and metformin reduces total, colorectal, liver and pancreatic cancer incidences in Taiwanese: a representative population prospective cohort study of 800,000 individuals. BMC Cancer. 2011;11:20. doi: 10.1186/1471-2407-11-20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Rattan R, et al. Metformin suppresses ovarian cancer growth and metastasis with enhancement of cisplatin cytotoxicity in vivo. Neoplasia. 2011;13(5):483–91. doi: 10.1593/neo.11148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Wang Y, et al. [Effects of antidiabetic drug metformin on human breast carcinoma cells with different estrogen receptor expressing in vitro] Xi Bao Yu Fen Zi Mian Yi Xue Za Zhi. 2011;27(3):253–6. [PubMed] [Google Scholar]
- 11.Inoki K, Zhu T, Guan KL. TSC2 mediates cellular energy response to control cell growth and survival. Cell. 2003;115(5):577–90. doi: 10.1016/s0092-8674(03)00929-2. [DOI] [PubMed] [Google Scholar]
- 12.Buzzai M, et al. Systemic treatment with the antidiabetic drug metformin selectively impairs p53-deficient tumor cell growth. Cancer Res. 2007;67(14):6745–52. doi: 10.1158/0008-5472.CAN-06-4447. [DOI] [PubMed] [Google Scholar]
- 13.Huang X, et al. Important role of the LKB1-AMPK pathway in suppressing tumorigenesis in PTEN-deficient mice. Biochem J. 2008;412(2):211–221. doi: 10.1042/BJ20080557. [DOI] [PubMed] [Google Scholar]
- 14.Jiralerspong S, et al. Metformin and Pathologic Complete Responses to Neoadjuvant Chemotherapy in Diabetic Patients With Breast Cancer. Journal of Clinical Oncology. 2009;27(20):3297–3302. doi: 10.1200/JCO.2009.19.6410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Zhuang Y, Miskimins WK. Cell cycle arrest in Metformin treated breast cancer cells involves activation of AMPK, downregulation of cyclin D1, and requires p27Kip1 or p21Cip1. J Mol Signal. 2008;3:18. doi: 10.1186/1750-2187-3-18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Zakikhani M, et al. Metformin is an AMP kinase-dependent growth inhibitor for breast cancer cells. Cancer Res. 2006;66(21):10269–73. doi: 10.1158/0008-5472.CAN-06-1500. [DOI] [PubMed] [Google Scholar]
- 17.Parkin DM. International variation. Oncogene. 2004;23(38):6329–40. doi: 10.1038/sj.onc.1207726. [DOI] [PubMed] [Google Scholar]
- 18.Pruefer FG, et al. Participation of Omi Htra2 serine-protease activity in the apoptosis induced by cisplatin on SW480 colon cancer cells. J Chemother. 2008;20(3):348–54. doi: 10.1179/joc.2008.20.3.348. [DOI] [PubMed] [Google Scholar]
- 19.Jordan P, Carmo-Fonseca M. Molecular mechanisms involved in cisplatin cytotoxicity. Cell Mol Life Sci. 2000;57(8-9):1229–35. doi: 10.1007/PL00000762. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Lin CC, et al. Metformin enhances cisplatin cytotoxicity by suppressing signal transducer and activator of transcription-3 activity independently of the liver kinase B1-AMP-activated protein kinase pathway. Am J Respir Cell Mol Biol. 2013;49(2):241–50. doi: 10.1165/rcmb.2012-0244OC. [DOI] [PubMed] [Google Scholar]
- 21.Gotlieb WH, et al. In vitro metformin anti-neoplastic activity in epithelial ovarian cancer. Gynecol Oncol. 2008;110(2):246–50. doi: 10.1016/j.ygyno.2008.04.008. [DOI] [PubMed] [Google Scholar]
- 22.Janjetovic K, et al. Metformin reduces cisplatin-mediated apoptotic death of cancer cells through AMPK-independent activation of Akt. Eur J Pharmacol. 2011;651(1-3):41–50. doi: 10.1016/j.ejphar.2010.11.005. [DOI] [PubMed] [Google Scholar]
- 23.Chang J, et al. Protective Effect of Metformin Against Cisplatin-Induced Ototoxicity in an Auditory Cell Line. J Assoc Res Otolaryngol. 2013 doi: 10.1007/s10162-013-0431-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Scartozzi M, et al. Chemotherapy for advanced gastric cancer: across the years for a standard of care. Expert Opin Pharmacother. 2007;8(6):797–808. doi: 10.1517/14656566.8.6.797. [DOI] [PubMed] [Google Scholar]
- 25.Ikeguchi M, Liu J, Kaibara N. Expression of survivin mRNA and protein in gastric cancer cell line (MKN-45) during cisplatin treatment. Apoptosis. 2002;7(1):23–29. doi: 10.1023/a:1013556727182. [DOI] [PubMed] [Google Scholar]
- 26.Tamm I, et al. IAP-Family Protein Survivin Inhibits Caspase Activity and Apoptosis Induced by Fas (CD95), Bax, Caspases, and Anticancer Drugs. Cancer Research. 1998;58(23):5315–5320. [PubMed] [Google Scholar]
- 27.Kato J, et al. Expression of survivin in esophageal cancer: correlation with the prognosis and response to chemotherapy. Int J Cancer. 2001;95(2):92–5. doi: 10.1002/1097-0215(20010320)95:2<92::aid-ijc1016>3.0.co;2-9. [DOI] [PubMed] [Google Scholar]
- 28.Adida C, et al. Anti-apoptosis gene, survivin, and prognosis of neuroblastoma. The Lancet. 1998;351(9106):882–883. doi: 10.1016/S0140-6736(05)70294-4. [DOI] [PubMed] [Google Scholar]
- 29.Monzo M, et al. A novel anti-apoptosis gene: Re-expression of survivin messenger RNA as a prognosis marker in non-small-cell lung cancers. J Clin Oncol. 1999;17(7):2100–4. doi: 10.1200/JCO.1999.17.7.2100. [DOI] [PubMed] [Google Scholar]
- 30.Sarela AI, et al. Expression of the antiapoptosis gene, survivin, predicts death from recurrent colorectal carcinoma. Gut. 2000;46(5):645–50. doi: 10.1136/gut.46.5.645. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Vazquez-Martin A, et al. The antidiabetic drug metformin: a pharmaceutical AMPK activator to overcome breast cancer resistance to HER2 inhibitors while decreasing risk of cardiomyopathy. Annals of Oncology. 2009;20(3):592–595. doi: 10.1093/annonc/mdn758. [DOI] [PubMed] [Google Scholar]
- 32.Datta SR, et al. Akt phosphorylation of BAD couples survival signals to the cell-intrinsic death machinery. Cell. 1997;91(2):231–41. doi: 10.1016/s0092-8674(00)80405-5. [DOI] [PubMed] [Google Scholar]
- 33.Brunet A, et al. Akt promotes cell survival by phosphorylating and inhibiting a Forkhead transcription factor. Cell. 1999;96(6):857–68. doi: 10.1016/s0092-8674(00)80595-4. [DOI] [PubMed] [Google Scholar]
- 34.Zimmermann S, Moelling K. Phosphorylation and regulation of Raf by Akt (protein kinase B) Science. 1999;286(5445):1741–4. doi: 10.1126/science.286.5445.1741. [DOI] [PubMed] [Google Scholar]
- 35.Cardone MH, et al. Regulation of cell death protease caspase-9 by phosphorylation. Science. 1998;282(5392):1318–21. doi: 10.1126/science.282.5392.1318. [DOI] [PubMed] [Google Scholar]
- 36.Romashkova JA, Makarov SS. NF-[kappa]B is a target of AKT in anti-apoptotic PDGF signalling. Nature. 1999;401(6748):86–90. doi: 10.1038/43474. [DOI] [PubMed] [Google Scholar]
- 37.Yuan TL, Cantley LC. PI3K pathway alterations in cancer: variations on a theme. Oncogene. 2008;27(41):5497–510. doi: 10.1038/onc.2008.245. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Hayakawa J, et al. Inhibition of BAD Phosphorylation Either at Serine 112 via Extracellular Signal-regulated Protein Kinase Cascade or at Serine 136 via Akt Cascade Sensitizes Human Ovarian Cancer Cells to Cisplatin. Cancer Research. 2000;60(21):5988–5994. [PubMed] [Google Scholar]
- 39.Winograd-Katz SE, Levitzki A. Cisplatin induces PKB//Akt activation and p38MAPK phosphorylation of the EGF receptor. Oncogene. 2006;25(56):7381–7390. doi: 10.1038/sj.onc.1209737. [DOI] [PubMed] [Google Scholar]


