Abstract
AIM: Considerable attention is focused on polymorphisms in the gene encoding transforming growth factor-β1 (TGF-β1), a multifunctional cytokine that is in turn a potent growth inhibitor involved in wound healing and differentiation. In humans, it promotes the pathogenesis of organ fibrosis, atherosclerosis, cancer, autoimmune and inflammatory diseases, keloid disease, and hypertrophic scarring. For this reason, much emphasis has been placed on studies elucidating the impact of TGF-β1 and its gene variations for the susceptibility and pathogenesis of these diseases. Unfortunately, some studies have serious limitations.
METHODS: We have recently described a high-throughput method for investigation the Arg25Pro polymorphism of human TGF-β1 gene and showed that the frequency of the Pro25 allele is significantly associated with hepatic fibrogenesis. In this report, we describe two novel LightCycler (LC) techniques that facilitate the examination of the two other known alterations in the coding region of TGF-β1. We investigated whether these polymorphisms contribute to hepatitis-induced progression of fibrogenesis in Chinese and Caucasians.
RESULTS: In the Chinese ancestry, the gene polymorphisms at codons 25 and 263 were not found and the genetic variant at codon 10 is unlikely to confer susceptibility to hepatic fibrosis. Contrarily, in Caucasians TGF-β1 allelic variations are more frequent and the presence of prolines either in codon 25 or 10 is associated with the interindividual variability in developing more severe fibrosis during chronic hepatitis C infection.
CONCLUSION: In summary, these results confirm the hypothesis that TGF-β1 polymorphisms are associated with fibrosis progression in Caucasians chronically infected with hepatitis C.
Keywords: TGF-β1, Gene polymorphism, LightCycler, Viral hepatitis, Fibrosis
INTRODUCTION
Increasing evidence indicates that genetic factors determine the rate of progression of liver fibrosis[1]. However, much controversy surrounds the issue of the natural history of hepatitis C virus (HCV) infection concerning viral-related factors (e.g., viral load, genotype, quasispecies), host-related factors (age, sex, race, immune status) and external determinants (e.g., alcohol, smoking, drugs)[2-4]. During the last decade, several candidate genes were identified influencing the natural history of chronic liver diseases. In these studies, the selection of genes investigated was mainly based on biological plausibility. In the pathogenesis of liver fibrosis, transforming growth factor-β1 (TGF-β1) is believed to be the most potent master cytokine that promotes hepatic fibrosis by stimulating the synthesis and inhibition of degradation of a broad spectrum of extracellular matrix proteins[5,6]. Therefore, mechanisms increasing the fraction of biologically active TGF-β1 are potentially involved in the modulation of progression of liver fibrosis. In line with that hypothesis, it has been reported that TGF-β1 messenger RNA is increased in the liver of patients with chronic HCV infection[7]. Furthermore, these patients had a higher level of TGF-β1, both totally and biologically active forms compared to healthy controls suggesting that TGF-β1 indeed promotes hepatic fibrogenesis[8]. Moreover, complementary work from many laboratories demonstrated that aberrant expression of TGF-β leads to hepatic fibrosis and inflammation in a number of experimental models. Particularly, transgenic mice overexpressing TGF-β1 are prone to develop multiple tissue lesions including hepatic fibrosis[9-11]. Conversely, the biological inactivation of TGF-β and its signaling pathway were recently shown to be sufficient to prevent ongoing hepatic fibrosis as induced by administration of dimethylnitrosamine or by ligation of the common bile duct in rats[12-16]. On the other side, high doses of soluble TGF-β receptors sequestering TGF-β function were less antifibrogenic than lower doses in murine models of chronic liver injury[17].
Given that TGF-β1 currently occupies a central role in liver fibrogenesis, it is possible that also certain DNA sequence variants of TGF-β1 might confer genetic risk to hepatic fibrosis. To date, several TGF-β1 polymorphisms have been identified; three variations are located upstream of exon 1 (at positions -988, -800, and -509), an insertion/deletion of a cytosine residue in the 5’ untranslated region (at position +72), and three nucleotide substitutions in the gene’s coding region[18,19]. Of these, two are located in exon 1 changing the amino acid sequence of the signal peptide (at positions +869 and +915) resulting in variations of codon 10 (leucine to proline) or codon 25 (arginine to proline), respectively. The third polymorphism in codon 263 (in exon 5) results in the amino acid substitution threonine to isoleucine in the precursor part of the protein. In addition, an insertion/deletion of a cytosine residue located in intron 4 and several other single-nucleotide polymorphisms (SNPs) have been reported[20-22]. It has previously been discussed that the production of TGF-β1 is associated with some of these gene polymorphisms[18,23,24]. However, with regard to HCV induced liver fibrogenesis, the influence of TGF-β1 gene polymorphisms have yielded some contradictory results, possibly reflecting heterogeneity between different populations or other unknown independent risk factors[25-27]. In previous works we found that the heterozygosity of codon 25 predicts significantly faster fibrotic progression and more severe fibrosis than the homozygous Arg25 genotype[26,27]. In contrast, another study with a smaller sample size reported that individuals with the high TGF-β1-producing genotype (Arg25Arg) were more likely to have increased hepatic fibrosis compared with individuals of the low TGF-β1-producing genotype[25].
In this report, we describe two novel LightCycler (LC) techniques that facilitate the high-throughput analysis of the genetic TGF-β1 variants at codon 10 and 263. In combination with the recently developed method for discriminating the different sequence variations at codon 25[27], we have now established the methodology to screen the sequence variations within the coding region of human TGF-β1 on a large scale. All three assays are conducted in a LC system allowing target amplification and product analysis to be done consecutively. The concept of this genotyping platform is based on different pairs of fluorescently labeled oligonucleotide probes that specifically distinguish the different alleles by use of the fluorescence energy transfer (FRET) principle.
By use of this robust and scaleable genotyping technique we corroborated and extended our previous work and determined the distributions of the respective allele frequencies at codon 10, 25 and 263 in Caucasians with chronic HCV infection and in healthy control subjects. We further compared the observed gene frequencies with those obtained in a Chinese cohort chronically infected with hepatitis B virus (HBV).
MATERIALS AND METHODS
Human subjects
The study population comprised 210 Caucasian patients with chronic HCV infection. They were recruited from the Department of Internal Medicine I, University Hospital of Regensburg (n = 141) and the Department of Medicine III, RWTH-University Hospital Aachen (n = 69). Fifty ethnically matched unrelated healthy controls were randomly recruited from the RWTH-University Hospital Aachen. The HCV diagnosis was based on positive anti-HCV assay (Abbott, Wiesbaden, Germany) and quantification of circulating HCV RNA by RT-PCR (Cobas Amplicor, Roche, Mannheim, Germany). Liver needle biopsies from 200 patients were stained and scored (stages F0-4) according to the guidelines by Desmet et al[28]. Further, we enrolled 90 Chinese patients with chronic HBV infection and 104 non-affected healthy controls from the Department of Laboratory Medicine, Changzheng Hospital, Second Military Medical University, Shanghai. HBV infection was demonstrated by positive serological assays for anti-HBc and HBsAg (Roche).
Analysis of TGF-β1 gene polymorphisms
Sequence variants at codons 10, 25 and 263 of human TGF-β1 gene (TGFB1) were determined in a LC-system (Roche). Genomic DNA was extracted from peripheral blood cells using the QIAamp DNA Blood Mini extraction kit (Qiagen, Hilden, Germany). The precise conditions for genotyping codon 25 of human TGF-β1 gene has been described in detail elsewhere[27]. Comparable assays were generated for genotyping at codon 10 and codon 263. A summary of primers, cycling conditions, and melting curve analysis is listed in Table 1, 2, and 3. In the final assays, approximately 80 ng genomic DNA were amplified in the presence of 0.5 µmol/L respective sense and reverse primers, 0.2 µmol/L sensor and anchor probes, 3 mmol/L MgCl2, 50 mL/L DMSO, and 1×LCTM FastStart DNA Master Hybridization Probes (Roche) including additionally 1 mmol/L MgCl2. The fluorescently labeled primers were obtained from Metabion (Martinsried, Germany), conventional PCR-primers were synthesized at MWG-Biotech AG (Ebersberg, Germany). The samples were initially heated for 10 min at 95 °C, and amplified for 45 cycles of 10 s at 95 °C, 10 s at 59 °C, and 20 s at 72 °C, respectively. The individual melting curves were recorded in 1 cycle of 95 °C for 1 min and 45 °C (codon 10) or 39 °C (codon 263) for 30 s, each with a temperature transition rate of 20 °C/s, and then ramping to 78 °C (codon 10) or 74 °C (codon 263) for 0 s with a transition rate of 0.2 °C/s. The exact compositions and locations of sensor and anchor probes are depicted in Figure 1.
Table 1.
LC Protocol 1 for Genotyping the TGF-β1 Gene Polymorphisms.
|
Codon 10 (Leu10Pro) |
||||
| Time (s) | Temp (°C) | Transition Rate (°C/s) | Cycles | |
| 600 | 95 | 20 | 1 | Denaturation |
| 10 | 95 | 20 | 45 | Cycling |
| 10 | 59 | 20 | ||
| 20 | 72 | 20 | ||
| 60 | 95 | 20 | 1 | Melting curve |
| Analysis | ||||
| 30 | 45 | 20 | ||
| 0 | 78 | 0.2 | ||
| 5-d(CTAGGTTATTTCCGTGGG)-3’ | Sense primer | |||
| 5-d(CCTTGGCGTAGTAGTCG)-3’ | Reverse primer | |||
| 5-d(TCCCCCATGCCGCCCTCCGGGCT-Fluo)-3’ | Anchor primer | |||
| 5-LCRed640-(CGGCTGCTGCCGCTGCT-P)-3’ | Sensor primer | |||
Table 2.
LC Protocol 2 for Genotyping the TGF-β1 Gene Polymorphisms.
|
Codon 25 (Arg25Pro) |
||||
| Time (s) | Temp (°C) | Transition Rate (°C/s) | Cycles | |
| 600 | 95 | 20 | 1 | Denaturation |
| 10 | 95 | 20 | 45 | Cycling |
| 10 | 59 | 20 | ||
| 20 | 72 | 20 | ||
| 60 | 95 | 20 | 1 | Melting curve |
| Analysis | ||||
| 30 | 40 | 20 | ||
| 0 | 80 | 0.2 | ||
| 5-d(CTAGGTTATTTCCGTGGG)-3’ | Sense primer | |||
| 5-d(CCTTGGCGTAGTAGTCG)-3’ | Reverse primer | |||
| 5-d(GCTACCGCTGCTGTGGCTACTGGTGCT-Fluo)-3 | Anchor primer | |||
| 5-LCRed640-(ACGCCTGGCCCGCCG-P)-3’ | Sensor primer | |||
Table 3.
LC Protocol 3 for Genotyping the TGF-β1 Gene Polymorphisms.
|
Codon 263 (Thr263Ile) |
||||
| Time (s) | Temp (°C) | Transition Rate (°C/s) | Cycles | |
| 600 | 95 | 20 | 1 | Denaturation |
| 10 | 95 | 20 | 45 | Cycling |
| 10 | 59 | 20 | ||
| 20 | 72 | 20 | ||
| 60 | 95 | 20 | 1 | Melting curve |
| Analysis | ||||
| 30 | 39 | 20 | ||
| 0 | 74 | 0.2 | ||
| 5-d(AAGCAGGGTTCACTACCGGC)-3’ | Sense primer | |||
| 5-d(AGGCCTCCATCCAGGCTACA)-3’ | Reverse primer | |||
| 5-LCRed640-(GAGAGGGCCCAGCATCTGCAAAGCT-P)-3 | Anchor primer | |||
| 5-d(ATGGCCACCCCGCT-Fluo)-3’ | Sensor primer | |||
Figure 1.
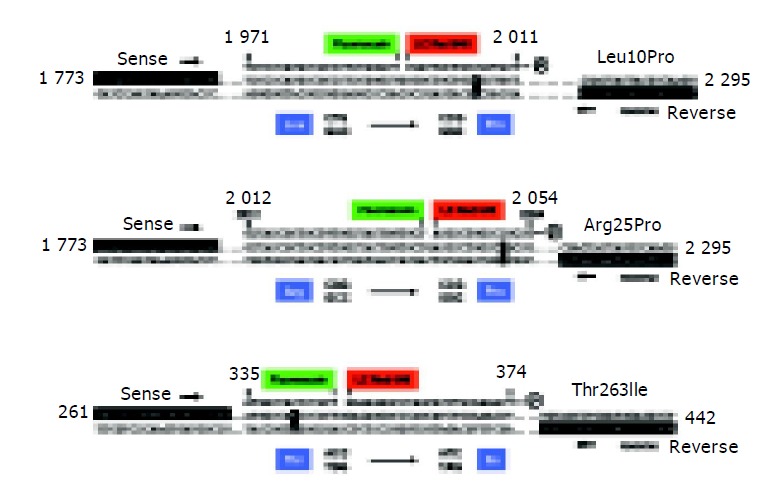
Molecular analysis of the coding TGF-β1 allele variations. For analysis of the Leu10Pro (A), Arg25Pro (B) and Thr263Ile (C) TGF-β1 polymorphisms, corresponding sites are amplified using specific combinations of sense and reverse primers. Subsequently, fluorescently labeled hybridization probes are annealed to the amplicons allowing discrimination of the different alleles by melting curve analysis. Nucleotide positions are given according to GenBank accession nos. X05839 (Leu10Pro, Arg25Pro) and X05844 (Thr263Ile), respectively.
Sequence analysis
Representative amplification products were purified by gel electrophoresis and sequenced with the Big Dye® Terminator Cycle Sequencing Ready Reaction kit (Applied Biosystems, Weiterstadt, Germany). Briefly, sequence reactions were performed in a final volume of 20 µL containing approximately 100 ng amplicon, 8 µL of BigDye® Terminator Reaction Mix, and 40 ng sequencing primer. The cycling conditions were: initial denaturation at 95 °C for 5 min, followed by 50 cycles at 95 °C for 30 s, 55 °C for 20 s, and 60 °C for 4 min. Products were purified on Centri-Sep spin columns (Princeton Separations, Adelphia, NJ), dried in a Speed Vac system, and resuspended in 20 µL template suppression reagent (Applied Biosystems). The reaction products were then denatured at 95 °C for 2 min and separated on the ABI PRISM 310 DNA Genetic Analyzer (Applied Biosystems).
Calculation of gene frequencies and statistics
The consistency of genotype frequencies with Hardy-Weinberg equilibrium was checked and visualized with the help of the triangular de Finetti diagram employing a software developed by Wienker and Strom[29]. The influence of TGF-β1 genotypes on the stage of fibrosis between groups was evaluated using χ2 analysis. In comparisons, P values ≤0.05 were considered significant.
RESULTS
TGF-β1 genotyping
We have recently described a real-time PCR detection method for discriminating the codon 25 variants of human TGF-β1 gene. The assay couples rapid PCR with fluorescent hybridization probes, allowing target amplification and product analysis to be done consecutively[27]. In a first step, the target is amplified by PCR and further processed for mutation detection. This is achieved by the nearby hybridization of two labeled probes within the amplified sequence exchanging FRET. One of the probes is a tightly binding anchor probe whereas the adjacent sensor probe covers the region of sequence variations. During melting of the annealed probes, differentiation of the different allelic variants are detected by their characteristic melting profiles; a homozygous sample results in a single peaked melting curve with a particular melting point for each allele, and a heterozygous allele constellation results in two peaks. The accuracy and speed of the genotyping protocol inspired us to develop comparable methods for analysis of the two other known polymorphic sites within the coding region (codons 10 and 263) of the TGF-β1 gene. The individual protocols were optimized for reagent concentrations, cycling conditions, and melting point analysis. In the final protocols (Table 1-3), different fluorescein- and LC-Red640-labeled hybridization probes are allowed to bind to the amplified product (Figure 1) and the different variants are identified in melting curve analysis. Typically, DNA homozygous for one variant results in a single peak with a different melting temperature than the other sequence variant and a heterozygous sample induces a biphasic melting curve with the combined peaks (Figures 2A and 3A). To demonstrate the accuracy of the two novel LC-techniques, we sequenced and analyzed representative amplicons by gel electrophoresis revealing that the amplicons (1) were derived from the TGF-β1 gene, (2) contained the predicted allele constellation (Figures 2B and 3B), and (3) had the correct sizes (Figures 2C and 3C).
Figure 2.
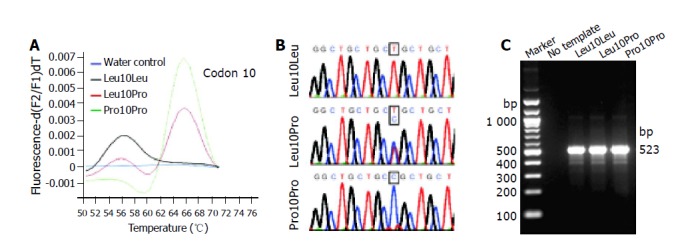
LC analysis of the Leu10Pro polymorphism. A: The derivative melting curves of a representative LC analysis for different samples are plotted. Genotypes identified are: Leu10Leu (black line), Leu10Pro (red line), and Pro10Pro (green line). The different allelic variants of codon 10 show typical melting temperatures at Tm = 56 °C (Leu10) and Tm = 67 °C (Pro25), respectively. A no-template control is shown in blue; B: Sequencing was performed to confirm the determined genotypes; C: Representative LC products were separated in 2 g/L agarose gel to demonstrate the correct size of synthesized amplicons (523 bp). The sizes of selected molecular weight markers are indicated at the left margin.
Figure 3.
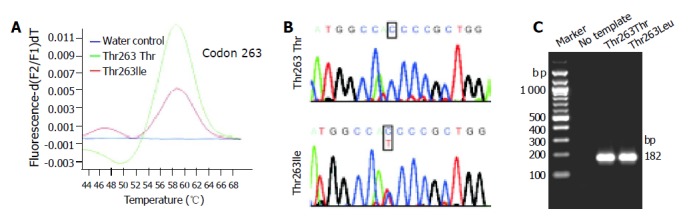
LC analysis of the Thr263Ile polymorphism. A: The derivative melting curves of a representative LC analysis for different samples are plotted. Genotypes identified are: Thr263Thr (green line) and Thr263Ile (red line). The different allelic variants of codon 263 show typical melting temperatures at Tm = 47 °C (Ile263) and Tm = 59 °C (Thr263), respectively. A no-template control is shown in blue; B: Sequencing was performed to confirm the determined genotypes; C: Representative LC products were separated in 8 g/L agarose gels to demonstrate the correct size of synthesized amplicons (182 bp). The sizes of selected molecular weight markers are indicated at the left margin.
Genotype frequencies in subjects of Caucasian and Chinese ancestry
The summarized TGF-β1 genotyping data of the two study populations are presented in Table 4. Although the genotype frequencies of all polymorphisms are in Hardy-Weinberg equilibrium, it is obvious that the distributions of the three gene variants are significantly dependent on ethnicity. In 104 healthy control subjects and in 90 patients with HBV infection taken from China the genetic polymorphisms at codons 25 and 263 were not observed. In contrast, 40 of 260 Caucasian subjects (15.4%) were heterozygous at codon 25 and 19 (7.3%) were heterozygous at codon 263. None of the tested samples were homozygous for proline or isoleucine at codon 25 or codon 263, respectively. The frequencies of TT, TC, and CC genotypes at codon 10 in the Chinese population among 194 subjects were 50 (25.8%), 87 (44.8%) and 57 (29.4%), respectively. In the group of 260 Caucasians the observed values were 106 (40.8%), 127 (48.8%) and 27 (10.4%), again demonstrating that both populations are divergent for these nucleotide substitutions. Interestingly, all 40 individuals heterozygous at codon 25 (Arg25Pro) were heterozygous (Leu10Pro) or homozygous for Pro (Pro10Pro) at codon 10. None of the subjects heterozygous at codon 25 was homozygous for Leu at codon 10. Hence, it is possible that the two prolines at codons 25 and 10 encoded by exon 1 form a defined haplotype in Caucasians that tends to be more frequent than the linkage of a proline at codon 25 together with a leucine at codon 10.
Table 4.
TGF-β1 gene polymorphisms in subjects of Chinese and Caucasian origin.
| Ethnic group | n | Subjects | Codon 25 | n | Codon 10 | n | Codon 263 | n |
| Chinese | 194 | Controls | Arg/Arg | 104 | Leu/Leu | 29 | Thr/Thr | 104 |
| Leu/Pro | 49 | |||||||
| Pro/Pro | 26 | |||||||
| Patients | Arg/Arg | 90 | Leu/Leu | 21 | Thr/Thr | 90 | ||
| Leu/Pro | 38 | |||||||
| Pro/Pro | 31 | |||||||
| Caucasians | 260 | Controls | Arg/Arg | 42 | Leu/Leu | 19 | Thr/Thr | 19 |
| Leu/Pro | 20 | Thr/Thr | 17 | |||||
| Thr/Ile | 3 | |||||||
| Pro/Pro | 3 | Thr/Thr | 2 | |||||
| Thr/Ile | 1 | |||||||
| Arg/Pro | 8 | Leu/Leu | 0 | |||||
| Leu/Pro | 7 | Thr/Thr | 7 | |||||
| Pro/Pro | 1 | Thr/Ile | 1 | |||||
| Patients | Arg/Arg | 178 | Leu/Leu | 87 | Thr/Thr | 85 | ||
| Thr/Ile | 2 | |||||||
| Leu/Pro | 80 | Thr/Thr | 74 | |||||
| Thr/Ile | 6 | |||||||
| Pro/Pro | 11 | Thr/Thr | 7 | |||||
| Thr/Ile | 4 | |||||||
| Arg/Pro | 32 | Leu/Leu | 0 | |||||
| Leu/Pro | 20 | Thr/Thr | 20 | |||||
| Pro/Pro | 12 | Thr/Thr | 10 | |||||
| Thr/Ile | 2 |
TGF-β1 genetic polymorphisms in assessing interindividual fibrosis variability in chronic hepatitis
The respective genotype frequencies for each polymorphic site were calculated for the Chinese and Caucasian populations (Table 5). We found no significant differences in the genotype frequencies between the individual control groups and patients suffering from chronic HBV (Chinese) or HCV (Caucasians) infection, respectively. There was only a slight, but not significant increase (P = 0.169; χ2 = 1.89) of the C allele (Pro) at codon 10 in Chinese subjects infected by HBV compared to non-infected Chinese controls.
Table 5.
TGF-β1 Allelic Frequencies.
| Polymorphic site |
Chinese |
Caucasians |
||||
| Total (n = 194) | Controls(n = 104) | Patients(n = 90) | Total(n = 260) | Controls(n = 50) | Patients (n = 210) | |
| Codon 10 (T/C) | 0.50/0.50 | 0.51/0.49 | 0.44/0.56 | 0.65/0.35 | 0.65/0.35 | 0.65/0.35 |
| Codon 25 (G/C) | 1/0 | 1/0 | 1/0 | 0.92/0.08 | 0.92/0.08 | 0.92/0.08 |
| Codon 263 (C/T) | 1/0 | 1/0 | 1/0 | 0.96/0.04 | 0.95/0.05 | 0.97/0.03 |
To elucidate the impact of the different allelic TGF-β1 variants and hepatic fibrogenesis in Caucasians, we compared the obtained genotype distributions between patients with different degrees of fibrosis (F0-2 vs F3-4) (Table 6). In summary, this analysis revealed that (1) patients with at least one proline at codon 10 are at a higher risk to have more pronounced fibrosis than patients without a proline (χ2 = 4.39; P = 0.036); (2) the severity of fibrosis correlates with the appearance of proline at codon 25 (χ2 = 5.97; P = 0.015), and (3) the Thr263Ile polymorphism had no significant impact on the interindividual variability in fibrosis progression.
Table 6.
Genotype distribution of codon 10, 25, and 263 polymorphisms and grade of fibrosis in caucasian patients (n = 200) with chronic hepatitis C infection.
| Codon | Genotype | Total | F0-2 | F3-4 |
| 10 | Leu/Leu | 82 | 70 | 12 |
| Leu/Pro | 95 | 69 | 26 | |
| Pro/Pro | 23 | 17 | 6 | |
| 25 | Arg/Arg | 169 | 137 | 32 |
| Arg/Pro | 31 | 19 | 12 | |
| Pro/Pro | 0 | 0 | 0 | |
| 263 | Thr/Thr | 186 | 145 | 41 |
| Thr/Ile | 14 | 11 | 3 | |
| Ile/Ile | 0 | 0 | 0 |
DISCUSSION
Chronic HBV and HCV infections are one of the most common infectious diseases leading to high morbidity and mortality due to the development of liver fibrosis/cirrhosis and hepatocellular carcinoma. At least four million people in the USA are believed to be chronically infected with HCV and nearly 10 000 HCV-related deaths occur each year in the USA[30-32]. Worldwide, it is estimated that nearly half a billion people are affected. However, epidemiological studies revealed that the progression of liver fibrosis in patients exposed to HCV infection is highly variable and that host factors including age, alcohol intake, intravenous drug abuse, sexual activity and gender are independent risk factors[33-36]. Previous studies during the last few years suggested that a strong genetic component affects the natural course of viral infections[37]. Based on biological plausibility, numerous candidate genes and their allelic variations were analyzed as potential biomarkers influencing the disease progression in chronic HCV[1,25]. Unfortunately, some of these studies have yielded contradictory results[1]. In particular, the lack of well-matched controls, the uncritical comparison of results obtained in different ethnicities, and the analysis of populations with unbalanced ethnic admixture are apparently the cause for some conflicting reports. Further, a critical factor to consider is the sample size implicating the statistical power of genetic association studies. Most often, a strong inverse relationship between the effort necessary to determine genetic polymorphism and sample size exists. Some of the molecular techniques that facilitate the analysis of variants are labor-intensive and are only appropriate for small-scale studies. These methods include restriction fragment length polymorphism (RFLP) analysis, amplification refractory mutation system (ARMS)-PCR, specialized 5’ nuclease assays, and single-strand conformational polymorphism (SSCP) analysis. Recently, high-throughput genotyping platforms, including SNP mapping, fluorescent dye-based genotyping technologies, DNA chip-based microarrays, and mass spectrometry genotyping technologies are developed for large-scale application.
The clarification of genetic polymorphisms in assessing interindividual variability in stage, grade and rate of progression of chronically injured livers in well-designed studies and the development of adequate genotyping methods are one objective of the recently created German Network of Competence for Viral Hepatitis (Hep-Net) sponsored by the German Ministry of Education and Research[38].
Recently, we described a new procedure discriminating between different allelic variants at codon 25 of the human TGF-β1 gene[27] and demonstrated that in Caucasian patients with severe hepatic fibrosis the Pro25 allele is twice as frequent as in subjects with mild fibrosis. We have analyzed this polymorphism since TGF-β1 is the most dominant fibrogenic cytokine in hepatic fibrosis contributing to the increased synthesis and deposition of a broad spectrum of extracellular matrix molecules including fibronectin, collagens type I, III, and IV, tenascin, elastin, osteonectin, biglycan, and decorin[6].
In the present study, we established two novel LC techniques allowing differentiation of the two other TGF-β1 allelic variants at codons 10 and 263. We extended our previous investigations and elucidated the possible relationship between the gene’s coding polymorphisms and the stage of liver fibrosis in Chinese and Caucasian patients with HBV or HCV infection, respectively. In the different ancestries, we found high differences of allelic frequencies at all three codons. All subjects from China carried the G-allele at codon 25 encoding arginine. This finding along with the recent demonstration that the variability at this site is also absent in populations from Korea and Japan[39-41] suggests that this polymorphism is generally not found in populations of Asia. Further, we observed no genetic alteration at position 263 in subjects from China. Moreover, the allele frequencies at codon 10 were different between the two ethnic groups (Leu 0.514 vs 0.652; Pro 0.486 vs 0.348) again demonstrating that it is of utmost importance to resolve issues regarding TGF-β1 gene polymorphisms in each of these subpopulations separately. In the Asian cohort we observed a slight increase of the Pro allele at codon 10 in patients suffering from HBV infection. However, the observed difference was of modest magnitude and not statistically significant indicating that this polymorphism is unlikely to be associated with the susceptibility for HBV infection in China.
In accordance, frequencies of the three investigated TGF-β1 genotypes did not differ between Caucasian patients with chronic HCV infection and control subjects confirming that these TGF-β polymorphisms are not per se associated with an increased risk of chronic hepatitis infections. However, in Caucasians the presence of a C rather than a G at codon 25 changing arginine to proline predispose to more severe fibrosis. Furthermore, patients have an elevated risk when a proline is present at codon 10 suggesting that TGF-β1 gene polymorphisms contribute to the interindividual disease variability.
The observed association between the leucine to proline substitution at codon 10 and the severity of fibrosis might be more indirect since both amino acids are apolar. Therefore, it is unlikely that the polymorphism affects the biological function and biochemical properties of the signal peptide and up to now there are no reports available demonstrating a different processing or synthesis of TGF-β1 for these modifications. Rather, the finding that all subjects genotyped for at least one proline at codon 10 were also heterozygous at codon 25 suggests that the prolines at codon 25 and codon 10 form a defined haplotype in Caucasians.
In general, our findings are somewhat unexpected because (1) experimental evidence has suggested that leukocytes taken from individuals with the Arg25Arg genotype had a significantly higher mean production of TGF-β1 than individuals with the Arg25Pro genotype[18] and (2) previous work of our laboratory has demonstrated that the median value of the TGF-β1 in platelet-free plasma from fibrotic patients carrying the homozygous Arg genotype were higher than in heterozygous patients[26]. As a consequence, it would be more presumable when the Arg25Arg genotype had more impact on hepatic fibrogenesis. In this regard, a recent report demonstrating that the sequestering of TGF-β1 by a soluble TGF-β receptor in an experimental animal model of chronic liver injury was less pronounced when the antagonist was given at higher doses is most interesting[17]. Both findings correspondingly show that high TGF-β1 concentrations per se are not decisive for the issue of fibrogenesis. Although speculative, a potential mechanism underlying the varying susceptibility for fibrosis might be a different tendency of both genotypes to increase the hepatic fraction of biologically active TGF-β1 in injured liver. Theoretically, it might also be possible that the suppression of the immune status by TGF-β1 in the high producer phenotype is more pronounced and that this dose-dependent modulation influences the overall susceptibility to the virus. However, this hypothesis is in contrast to our findings that reveal no differences in the frequencies of the individual TGF-β1 genotypes between patients chronically infected with HBV or HCV and the corresponding control groups. Since we have as yet no direct explanation how the amino acid substitutions at codon 10 and 25 contribute to hepatic fibrogenesis, further studies will determine at which level they modulate the biological activity of TGF-β1 function. We have not correlated our data to surrogate markers for liver fibrosis (e.g., serum hyaluronic acid), since these markers are known to be predominantly altered in patients with severe fibrosis or cirrhosis. However, the histological scoring as the gold standard for evaluating the degree of fibrosis was found to be strongly dependent on gene frequencies within the TGF-β1 gene.
In summary, our data support the hypothesis that TGF-β1 polymorphisms contribute to the susceptibility for hepatic fibrosis in chronic HCV infection. The established TGF-β1 genotyping methodology and the integration of our work into the German Network of Excellence for Viral Hepatitis (Hep-Net) will allow analyzing large numbers of HBV- and HCV-samples from academic centers and private practice institutions in the near future. This will undoubtedly provide the basis for a better design of studies aimed to understand the impact of human diversity in the pathogenesis of liver diseases. We hope that this report encourages investigators to enter and participate in large-scale collaboration studies.
ACKNOWLEDGEMENTS
Dr. Hao Wang was financed by a stipend from the Germany United Society of Clinical Chemistry and Laboratory Medicine.
Footnotes
Supported by the Grants From the Federal Ministry of Education and Research of Germany (Network of Competence in Medicine Hep-Net) and the Natural Science Foundation of China, No. 30270605
Science Editor Li WZ Language Editor Elsevier HK
References
- 1.Bataller R, North KE, Brenner DA. Genetic polymorphisms and the progression of liver fibrosis: a critical appraisal. Hepatology. 2003;37:493–503. doi: 10.1053/jhep.2003.50127. [DOI] [PubMed] [Google Scholar]
- 2.Seeff LB. Natural history of chronic hepatitis C. Hepatology. 2002;36:S35–S46. doi: 10.1053/jhep.2002.36806. [DOI] [PubMed] [Google Scholar]
- 3.Marcellin P, Asselah T, Boyer N. Fibrosis and disease progression in hepatitis C. Hepatology. 2002;36:S47–S56. doi: 10.1053/jhep.2002.36993. [DOI] [PubMed] [Google Scholar]
- 4.Hoofnagle JH. Course and outcome of hepatitis C. Hepatology. 2002;36:S21–S29. doi: 10.1053/jhep.2002.36227. [DOI] [PubMed] [Google Scholar]
- 5.Friedman SL. Molecular regulation of hepatic fibrosis, an integrated cellular response to tissue injury. J Biol Chem. 2000;275:2247–2250. doi: 10.1074/jbc.275.4.2247. [DOI] [PubMed] [Google Scholar]
- 6.Gressner AM, Weiskirchen R, Breitkopf K, Dooley S. Roles of TGF-beta in hepatic fibrosis. Front Biosci. 2002;7:d793–d807. doi: 10.2741/A812. [DOI] [PubMed] [Google Scholar]
- 7.Castilla A, Prieto J, Fausto N. Transforming growth factors beta 1 and alpha in chronic liver disease. Effects of interferon alfa therapy. N Engl J Med. 1991;324:933–940. doi: 10.1056/NEJM199104043241401. [DOI] [PubMed] [Google Scholar]
- 8.Nelson DR, Gonzalez-Peralta RP, Qian K, Xu Y, Marousis CG, Davis GL, Lau JY. Transforming growth factor-beta 1 in chronic hepatitis C. J Viral Hepat. 1997;4:29–35. doi: 10.1046/j.1365-2893.1997.00124.x. [DOI] [PubMed] [Google Scholar]
- 9.Sanderson N, Factor V, Nagy P, Kopp J, Kondaiah P, Wakefield L, Roberts AB, Sporn MB, Thorgeirsson SS. Hepatic expression of mature transforming growth factor beta 1 in transgenic mice results in multiple tissue lesions. Proc Natl Acad Sci USA. 1995;92:2572–2576. doi: 10.1073/pnas.92.7.2572. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Kanzler S, Lohse AW, Keil A, Henninger J, Dienes HP, Schirmacher P, Rose-John S, zum Büschenfelde KH, Blessing M. TGF-beta1 in liver fibrosis: an inducible transgenic mouse model to study liver fibrogenesis. Am J Physiol. 1999;276:G1059–G1068. doi: 10.1152/ajpgi.1999.276.4.G1059. [DOI] [PubMed] [Google Scholar]
- 11.Ueberham E, Löw R, Ueberham U, Schönig K, Bujard H, Gebhardt R. Conditional tetracycline-regulated expression of TGF-beta1 in liver of transgenic mice leads to reversible intermediary fibrosis. Hepatology. 2003;37:1067–1078. doi: 10.1053/jhep.2003.50196. [DOI] [PubMed] [Google Scholar]
- 12.Qi Z, Atsuchi N, Ooshima A, Takeshita A, Ueno H. Blockade of type beta transforming growth factor signaling prevents liver fibrosis and dysfunction in the rat. Proc Natl Acad Sci USA. 1999;96:2345–2349. doi: 10.1073/pnas.96.5.2345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Ueno H, Sakamoto T, Nakamura T, Qi Z, Astuchi N, Takeshita A, Shimizu K, Ohashi H. A soluble transforming growth factor beta receptor expressed in muscle prevents liver fibrogenesis and dysfunction in rats. Hum Gene Ther. 2000;11:33–42. doi: 10.1089/10430340050016139. [DOI] [PubMed] [Google Scholar]
- 14.George J, Roulot D, Koteliansky VE, Bissell DM. In vivo inhibition of rat stellate cell activation by soluble transforming growth factor beta type II receptor: a potential new therapy for hepatic fibrosis. Proc Natl Acad Sci USA. 1999;96:12719–12724. doi: 10.1073/pnas.96.22.12719. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Arias M, Sauer-Lehnen S, Treptau J, Janoschek N, Theuerkauf I, Buettner R, Gressner AM, Weiskirchen R. Adenoviral expression of a transforming growth factor-beta1 antisense mRNA is effective in preventing liver fibrosis in bile-duct ligated rats. BMC Gastroenterol. 2003;3:29. doi: 10.1186/1471-230X-3-29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Dooley S, Hamzavi J, Breitkopf K, Wiercinska E, Said HM, Lorenzen J, Ten Dijke P, Gressner AM. Smad7 prevents activation of hepatic stellate cells and liver fibrosis in rats. Gastroenterology. 2003;125:178–191. doi: 10.1016/s0016-5085(03)00666-8. [DOI] [PubMed] [Google Scholar]
- 17.Yata Y, Gotwals P, Koteliansky V, Rockey DC. Dose-dependent inhibition of hepatic fibrosis in mice by a TGF-beta soluble receptor: implications for antifibrotic therapy. Hepatology. 2002;35:1022–1030. doi: 10.1053/jhep.2002.32673. [DOI] [PubMed] [Google Scholar]
- 18.Awad MR, El-Gamel A, Hasleton P, Turner DM, Sinnott PJ, Hutchinson IV. Genotypic variation in the transforming growth factor-beta1 gene: association with transforming growth factor-beta1 production, fibrotic lung disease, and graft fibrosis after lung transplantation. Transplantation. 1998;66:1014–1020. doi: 10.1097/00007890-199810270-00009. [DOI] [PubMed] [Google Scholar]
- 19.Cambien F, Ricard S, Troesch A, Mallet C, Générénaz L, Evans A, Arveiler D, Luc G, Ruidavets JB, Poirier O. Polymorphisms of the transforming growth factor-beta 1 gene in relation to myocardial infarction and blood pressure. The Etude Cas-Témoin de l'Infarctus du Myocarde (ECTIM) Study. Hypertension. 1996;28:881–887. doi: 10.1161/01.hyp.28.5.881. [DOI] [PubMed] [Google Scholar]
- 20.Park BL, Han IK, Lee HS, Kim LH, Kim SJ, Shin HD. Identification of novel variants in transforming growth factor-beta 1 (TGFB1) gene and association analysis with bone mineral density. Hum Mutat. 2003;22:257–258. doi: 10.1002/humu.9170. [DOI] [PubMed] [Google Scholar]
- 21.Langdahl BL, Knudsen JY, Jensen HK, Gregersen N, Eriksen EF. A sequence variation: 713-8delC in the transforming growth factor-beta 1 gene has higher prevalence in osteoporotic women than in normal women and is associated with very low bone mass in osteoporotic women and increased bone turnover in both osteoporotic and normal women. Bone. 1997;20:289–294. doi: 10.1016/s8756-3282(96)00363-8. [DOI] [PubMed] [Google Scholar]
- 22.Watanabe Y, Kinoshita A, Yamada T, Ohta T, Kishino T, Matsumoto N, Ishikawa M, Niikawa N, Yoshiura K. A catalog of 106 single-nucleotide polymorphisms (SNPs) and 11 other types of variations in genes for transforming growth factor-beta1 (TGF-beta1) and its signaling pathway. J Hum Genet. 2002;47:478–483. doi: 10.1007/s100380200069. [DOI] [PubMed] [Google Scholar]
- 23.Grainger DJ, Heathcote K, Chiano M, Snieder H, Kemp PR, Metcalfe JC, Carter ND, Spector TD. Genetic control of the circulating concentration of transforming growth factor type beta1. Hum Mol Genet. 1999;8:93–97. doi: 10.1093/hmg/8.1.93. [DOI] [PubMed] [Google Scholar]
- 24.El-Gamel A, Awad MR, Hasleton PS, Yonan NA, Hutchinson JA, Campbell CS, Rahman AH, Deiraniya AK, Sinnott PJ, Hutchinson IV. Transforming growth factor-beta (TGF-beta1) genotype and lung allograft fibrosis. J Heart Lung Transplant. 1999;18:517–523. doi: 10.1016/s1053-2498(98)00024-2. [DOI] [PubMed] [Google Scholar]
- 25.Powell EE, Edwards-Smith CJ, Hay JL, Clouston AD, Crawford DH, Shorthouse C, Purdie DM, Jonsson JR. Host genetic factors influence disease progression in chronic hepatitis C. Hepatology. 2000;31:828–833. doi: 10.1053/he.2000.6253. [DOI] [PubMed] [Google Scholar]
- 26.Gewaltig J, Mangasser-Stephan K, Gartung C, Biesterfeld S, Gressner AM. Association of polymorphisms of the transforming growth factor-beta1 gene with the rate of progression of HCV-induced liver fibrosis. Clin Chim Acta. 2002;316:83–94. doi: 10.1016/s0009-8981(01)00738-0. [DOI] [PubMed] [Google Scholar]
- 27.Tag CG, Mengsteab S, Hellerbrand C, Lammert F, Gressner AM, Weiskirchen R. Analysis of the transforming growth factor-beta1 (TGF-beta1) codon 25 gene polymorphism by LightCycler-analysis in patients with chronic hepatitis C infection. Cytokine. 2003;24:173–181. doi: 10.1016/j.cyto.2003.08.007. [DOI] [PubMed] [Google Scholar]
- 28.Desmet VJ, Gerber M, Hoofnagle JH, Manns M, Scheuer PJ. Classification of chronic hepatitis: diagnosis, grading and staging. Hepatology. 1994;19:1513–1520. [PubMed] [Google Scholar]
- 29.Elston RC, Forthofer R. Testing for Hardy-Weinberg equilibrium in small samples. Biometrics. 1977;33:536–542. [Google Scholar]
- 30.National Institutes of Health Consensus Development Conference Statement: Management of hepatitis C: 2002--June 10-12, 2002. Hepatology. 2002;36:S3–20. doi: 10.1053/jhep.2002.37117. [DOI] [PubMed] [Google Scholar]
- 31.Wong JB, McQuillan GM, McHutchison JG, Poynard T. Estimating future hepatitis C morbidity, mortality, and costs in the United States. Am J Public Health. 2000;90:1562–1569. doi: 10.2105/ajph.90.10.1562. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Alter MJ, Kruszon-Moran D, Nainan OV, McQuillan GM, Gao F, Moyer LA, Kaslow RA, Margolis HS. The prevalence of hepatitis C virus infection in the United States, 1988 through 1994. N Engl J Med. 1999;341:556–562. doi: 10.1056/NEJM199908193410802. [DOI] [PubMed] [Google Scholar]
- 33.Peters MG, Terrault NA. Alcohol use and hepatitis C. Hepatology. 2002;36:S220–S225. doi: 10.1053/jhep.2002.36811. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Edlin BR. Prevention and treatment of hepatitis C in injection drug users. Hepatology. 2002;36:S210–S219. doi: 10.1053/jhep.2002.36809. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Terrault NA. Sexual activity as a risk factor for hepatitis C. Hepatology. 2002;36:S99–S105. doi: 10.1053/jhep.2002.36797. [DOI] [PubMed] [Google Scholar]
- 36.Poynard T, Bedossa P, Opolon P. Natural history of liver fibrosis progression in patients with chronic hepatitis C. The OBSVIRC, METAVIR, CLINIVIR, and DOSVIRC groups. Lancet. 1997;349:825–832. doi: 10.1016/s0140-6736(96)07642-8. [DOI] [PubMed] [Google Scholar]
- 37.Weatherall D, Clegg J, Kwiatkowski D. The role of genomics in studying genetic susceptibility to infectious disease. Genome Res. 1997;7:967–973. doi: 10.1101/gr.7.10.967. [DOI] [PubMed] [Google Scholar]
- 38.Manns MP, Meyer S, Wedemeyer H. The German network of excellence for viral hepatitis (Hep-Net) Hepatology. 2003;38:543–544. doi: 10.1002/hep.510380302. [DOI] [PubMed] [Google Scholar]
- 39.Lee JG, Ahn C, Yoon SC, Park JH, Eo HS, No JJ, Kim KH, Lee EJ, Hwang YH, Hwang DY, et al. No association of the TGF-beta1 gene polymorphisms with the renal progression in autosomal dominant polycystic kidney disease (ADPKD) patients. Clin Nephrol. 2003;59:10–16. doi: 10.5414/cnp59010. [DOI] [PubMed] [Google Scholar]
- 40.Ohtsuka T, Yamakage A, Yamazaki S. The polymorphism of transforming growth factor-beta1 gene in Japanese patients with systemic sclerosis. Br J Dermatol. 2002;147:458–463. doi: 10.1046/j.1365-2133.2002.04947.x. [DOI] [PubMed] [Google Scholar]
- 41.Suzuki S, Tanaka Y, Orito E, Sugauchi F, Hasegawa I, Sakurai M, Fujiwara K, Ohno T, Ueda R, Mizokami M. Transforming growth factor-beta-1 genetic polymorphism in Japanese patients with chronic hepatitis C virus infection. J Gastroenterol Hepatol. 2003;18:1139–1143. doi: 10.1046/j.1440-1746.2003.03161.x. [DOI] [PubMed] [Google Scholar]


