Abstract
AIM: To comprehend the risk factors of recurrence of hepatocellular carcinoma (HCC) and its relationship with the infection patterns of hepatitis B virus (HBV).
METHODS: All materials of 270 cases of postoperative HCC were statistically analyzed by SPSS software. Recurrence and metastasis were classified into early ( ≤2 years) and late phase (>2 years). Risk factors for recurrence and metastasis after surgery in each group were analyzed.
RESULTS: Out of 270 cases of HCC, 162 cases were followed up in which recurrence and metastasis occurred in 136 cases. There were a lot of risk factors related to recurrence and metastasis of HCC; risk factors contributing to early phase recurrence were serum AFP level, vascular invasion, incisal margin and operative transfusion, gross tumor classification and number of intrahepatic node to late phase recurrence. The HBV infective rate of recurrent HCC was 94.1%, in which “HBsAg, HBeAb, HBcAb” positive pattern reached 45.6%. The proportion of HBV infection in solitary large hepatocellular carcinoma (SLHCC) evidently decreased compared to nodular hepatocellular carcinoma (NHCC) (P<0.05).
CONCLUSION: The early and late recurrence and metastasis after hepatectomy of HCC were associated with different risk factors. The early recurrence may be mediated by vascular invasion and remnant lesion, the late recurrence by tumor’s clinical pathology propert, as multicentric carcinogenesis or intrahepatic carcinoma de novo. HBV replication takes a great role in this process. From this study, we found that SLHCC has more satisfactory neoplasm biological behavior than NHCC.
Keywords: Hepatocellular carcinoma, Recurrence and metastasis, Risk factor, Hepatitis B virus
INTRODUCTION
Because of the advances in surgical techniques and perioperative management, hepatectomy of hepatocellular carcinoma (HCC) has become a safe operation with low operative mortality. However, the long-term prognosis remains poor due to a high incidence of recurrence and metastasis of HCC, ranging from 50% to 80%[1-3]. The effect of HCC’s therapy is also unpleasant; the high rate of recurrence and metastasis after HCC resection has always disturbed clinical surgeon nowadays. There are lots of reports based on the fact that intrahepatic recurrence or multicentric carcinogenesis de novo were found after resection of HCC[4,5]. Because different risk factors for recurrence and metastasis have been identified in previous articles[6], it is a controversy regarding the way of recurrence and metastasis, which need us continue to disclose its mechanism.
Hepatitis B virus (HBV) is a hepadnavirus, discussed worldwide as resulting in viral hepatitis in humans. HBV gets distributed at various ages, the infectious rate of HBV has reached 10% in China. Once it infects the human body, the HBV gets replicated and proliferated into hepatocytes, induces hepatocyte lesion and develops into chronic hepatitis. It has been reported that HBV is the prime cause for the development of HCC. In order to discuss the relationship between HBV and the recurrence and metastasis of HCC, we studied the clinical data of postoperative HCC and its follow-up.
MATERIALS AND METHODS
Population
The base population consisted of 270 consecutive patients who had undergone initial and curative hepatic resection for HCCs at Central South University Xiangya Hospital from 1992 to 2002. The indication of surgical resection and operative procedure was determined according to the decision criteria based on the liver function test including ascites, serum bilirubin level, and image data. Systematic resection of Couinaud’s segment was a preferred operative procedure if the patient’s liver functional reserve permitted. Curative resection was defined as removal of all recognizable tumors with a clear margin. As regards the tumor margin, resection with 1 cm was defined as curative so long as no tumor invasion was observed at the surgical cut surface[7]. By retrospectively investigating these data of 270 patients, 162 gained follow-up and finally entered into the analyses. One hundred and eight patients were excluded for the following reasons: data lacked follow-up for 96 and incomplete pathological analysis in 12. Out of 162 patients only 132 presented recurrence. We divided time of recurrence into early phase (amongst 2 years) and later phase (excluding 2 years); there were 107 cases of recurrence and metastasis in early phase and 29 cases in later phase. The baseline characteristics of these 162 patients classified according to viral infection pattern are presented in Table 1. Gross HCC classification was divided into small HCC (SHCC), solitary large HCC (SLHCC) and nodular HCC (NHCC) according to our previous study on HCC’s basic and clinical characteristics[8]. Various surgical procedures were classified as anatomical or non-anatomical resections. Anatomical resection involves resecting the tumor together with the tumor-containing portal vein and corresponding hepatic territory and includes hemihepatectomy (right or left), sectorectomy (right lateral, right paramedian, left medial, and the left lateral sector), and segmentectomy (resection of Couinaud’s segments). All other types of resection such as limited resection and tumor enucleation are classified as non-anatomical resection.
Table 1.
Baseline characteristics in 270 patients (mean±SD).
| Variable factors | All patients (n = 270) | Recurrent patients (n = 136) |
Recurrent patients (n = 136) |
|
| Early phase (n = 107) | Late phase (n = 29) | |||
| Age (yr) | 46.9±13.0 | 4.0±11.5 | 44.2±11.1 | 47.7±12.9 |
| Sex (male/female) | 247/23 | 126/10 | Aug-99 | 27/2 |
| Gross tumorclassification (small/solitary/nodular) | 66/168/36 | 35/77/24 | 23/63/21 | 12/14/3 |
| Edmondson classification (I-II/III-IV) | 128/142 | 65/71 | 46/61 | 19/10 |
| Tumor capsule (present/absent) | 55/215 | 23/113 | 13/94 | 10/19 |
| Vascular invasion (present/absent) | 145/125 | 79/57 | 62/45 | 7/12 |
| Alpha-fetoprotein (positive/negative) | 141/129 | 85/51 | 75/32 | 10/19 |
| Hepatocirrhosis (positive/negative) | 200/70 | 105/31 | 82/25 | 23/6 |
| Type of liver resection (non-anatomical/anatomical) | 149/121 | 78/58 | 59/48 | 19/10 |
| Incisal margin (≤1 cm/>1 cm) | 22/258 | 15/131 | 14/93 | 1/28 |
| HBV infection (positive/negative) | 239/31 | 128/8 | 102/5 | 26/3 |
| Blood transfusion (yes/no) | 199/71 | 100/36 | 75/32 | 25/4 |
| Survival time (d) | – | 575.9±499.6 | 384.7±313.1 | 1248.5±450.9 |
| Recurrent time (d) | – | 435.6±417.9 | 23.1±199.6 | 1072.2±396.9 |
Note: Small HCC (SHCC), solitary large HCC (SLHCC), nodular HCC (NHCC).
Follow-up and end point
After discharge, monthly follow-up by tumor marker (alpha-fetoprotein, AFP) and ultrasound (US) as well as helical computed tomographic (CT) scan for every 4 mo were conducted for 1 year. Then, we screened patients by tumor marker measurement and US for every 2 mo and helical CT for every 6 mo thereafter, and hepatic angiography when recurrence was suspected. Recurrence was diagnosed based on the combined findings of these clinical examinations. The end point of this study was time-to-recurrence, which was defined as the period between surgery and the diagnosis of recurrence and metastasis. Patients who died for reasons not related to HCC were censored at the time of death. All follow-up data were summarized by the end of April 2004.
Variables analyzed
Discrete variables were computed directly, whereas continuous variables were classified into binary or polytomous categorical data. Cut-off points for AFP were 20 μg/L, and maximal tumor diameter were determined according to operative records, which gave the best discrimination between time-to-recurrence curves among several stratifications[5].
The studied variables could be classified as host-, cancer-, or surgery-related (Table 1). The host-related factors were age, sex, viral markers (HBsAg-, HBeAg-, HBsAb-, HBeAb-, HBcAb-). HBV infection pattern was graded according to the difference of viral markers positive expression. Fibrosis was assessed using non-cancerous parts of the resected specimens. The stage of liver fibrosis was classified into four categories (no, mild, moderate, and cirrhosis) to represent the severity of liver disease. Cancer-related factors were gross classification of the tumor[6,9], tumor multiplicity, maximum diameter of tumor, microscopically assessed vascular invasion, intrahepatic metastasis, microscopically assessed presence and/or invasion of a tumor capsule, tumor cell differentiation, serum AFP level. Multiple tumor nodules and intrahepatic metastasis were differentiated based on macro- and microscopical findings. In brief, multiple tumors were classified into intrahepatic metastases if they were (a) tumors apparently growing from portal venous tumor thrombi, or (b) multiple satellite nodules surrounding a main tumor with similar or poorer degree of cell differentiation. Surgery-related factors included type of resection (anatomical vs non-anatomical) and surgical free margin (>1 cm vs <1 cm).
Statistical analysis
The overall hazard function for recurrence and metastasis was also estimated to visually depict chronological change in the recurrence rate after hepatectomy. We investigated factors contributing to early and late phase recurrence, separately, setting 2 years as the cut-off between the early and late phases[7]. For this purpose, we first conducted a multiple regression analysis by setting the different baseline hazard functions for respective institutions and by censoring patients without recurrence at 2 years after surgery at this time point. Then, we performed a landmark analysis on recurrence-free status at 2 years after surgery. Finally, the relationship between the viral infection patterns was investigated using the Mantel-Haenszel trend test on data from patients showing only one of two hepatitis markers. As additional sets of analyses, we performed the same analyses exclusively in patients with solitary tumor. Likewise, we also performed analyses dividing patients with and without liver cirrhosis. All statistical analyses in the present study were performed using the Statistical Package for Social Sciences (SPSS11.0 software).
RESULTS
Analysis of the recurrence and metastasis of HCC
Among the 162 cases of follow-up, the median follow-up time was 1742 d (range 7-4200 d). Recurrence and metastasis were observed in 136 patients. There was a patient who survived more than 11 years. Overall cumulative recurrence and metastasis rate curves for all patients are shown in Figure 1, and Figure 2 depicts the overall recurrence and metastasis at each year after operation, which peaked at the first year postoperatively, then gradually decreased year by year. The overall cumulative recurrence and metastasis rate were 32.7% (200 d), 59.2% (400 d), 64.5% (600 d), 69.8% (800 d), 73.5% (1000 d), 80.3% (1200 d), 84.1% (1400 d); there were 46.3% (1st year), 36.8% (2nd year), 32.7% (3rd year), 24.3% (4th year) and 12.4% (5th year) respectively from 1st to 5th year.
Figure 1.
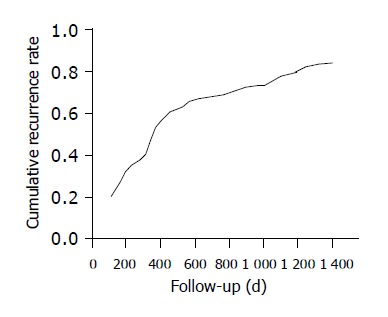
Curve of overall cumulative recurrence and metastasis rate metastasis rate present in 136 HCC patients.
Figure 2.
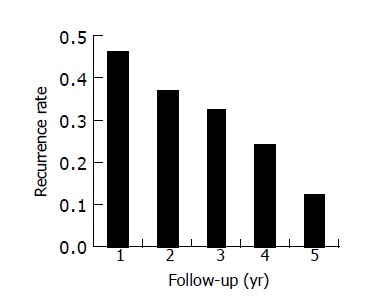
Bar graph of recurrence and metastasis rate present in 136 patients at each year.
Factors contributing to early (≤2 years) phase recurrence and metastasis
Recurrence and metastasis before 2 years was observed in 107 patients. Multiple regression analysis identified four variables as contributing to early phase recurrence and metastasis: Serum AFP (alpha-fetoprotein) level, vascular invasion, incisal margin and operative transfusion (Table 2). As a result, the causes of recurrence and metastasis for early phase were the factors correlated to surgery.
Table 2.
Multiple regression analysis of recurrence and metastasis of 136 HCC patients.
| Variable |
Results on early phase |
Results on late phase |
||||
| Regression coefficient | t | P | Regression coefficient | t | P | |
| AFP level | -0.328 | -3.693 | 0.000 | -0.280 | -1.306 | 0.208 |
| Hepatocirrhosis | 0.024 | 0.274 | 0.785 | -0.377 | -1.530 | 0.143 |
| Type of liver resection | 0.023 | 0.260 | 0.795 | 0.245 | 1.235 | 0.233 |
| Incisal margin | 0.217 | 2.485 | 0.015 | 0.106 | 0.420 | 0.679 |
| Blood transfusion | -0.223 | 2.553 | 0.012 | 0.268 | 1.266 | 0.222 |
| Gross tumor classification | -0.051 | -0.583 | 0.561 | -0.518 | -2.433 | 0.026 |
| Vascular invasion | 0.266 | 3.013 | 0.003 | -0.361 | -1.562 | 0.136 |
| Tumor capsule | 0.023 | 0.259 | 0.797 | -0.281 | -1.111 | 0.281 |
| Edmondson classification | 0.126 | 1.444 | 0.152 | 0.065 | 0.292 | 0.774 |
Note: Dependent variable was the time of recurrence and metastasis (d).
Factors contributing to late (>2 years) phase recurrence and metastasis
In the same way, recurrence and metastasis was observed in 29 patients. Factors related to late phase recurrence and metastasis were gross tumor classification and number of intrahepatic node (Table 2). NHCC had higher recurrence and metastasis rate than SLHCC (P = 0.026). It was coincident to the results of our prophase study on the SLHCC characterization at basic and clinical[10].
Relationship on the HBV infection with recurrence and metastasis of HCC
It has been confirmed by lots of epidemiological survey that HCC genesis is closely related to the HBV infection. Our further study disclosed that the HBV infection has related to the recurrence and metastasis of HCC. In this paper, the infective rate was 88.5% on 270 cases of HCC, but 94.1% on 136 cases of recurrence and metastasis of HCC. Among the 162 cases of follow-up, there were 128 recurrences with HBV infection (147 cases) and only 8 recurrences without HBV infection (15 cases). Statistic comparison had high significant difference (χ2 = 11.501, P = 0.001); it prompted that the HBV infection was an important cause of recurrence and metastasis of HCC.
The HBV infective pattern was different in various clinical classifications of HCC. Contrast analysis of the HBV infective pattern among 270 HCC patients that accepted operation with 136 HCC patients showed that recurrence and metastasis occurred; we found that HBV infection not only related to the HCC genesis and recurrence, but also that their infective pattern varied (t = 2.407, P = 0.037, Table 3). Further comparison of the infective pattern of recurrent SLHCC and recurrent NHCC, results showed the HBV infection pattern of recurrent NHCC, which was obviously different compared with SLHCC, and the proportion of recurrent NHCC with HBV infection was obviously step-up than recurrent SLHCC (P<0.05, Table 4).
Table 3.
Statistics of the HBV infective pattern.
| Serum markers |
All patients (n = 270) |
Recurrent patients (n = 136) |
||
| Positive number | Ratio (%) | Positive number | Ratio (%) | |
| HBsAg+HBeAg+HBcAb+ | 20 | 7.4 | 14 | 10.3 |
| HBsAg+HBeAb+HBcAb+ | 118 | 43.7 | 62 | 45.6 |
| HBsAg+HBcAb+ | 57 | 21.1 | 32 | 23.5 |
| HBsAb+ | 9 | 3.3 | 6 | 4.4 |
| HBcAb+ | 2 | 0.7 | 2 | 1.5 |
| HBeAb+HBcAb+ | 7 | 2.6 | 1 | 0.7 |
| HBsAg+ | 16 | 5.9 | 9 | 6.6 |
| HBsAb+HBeAb+HBcAb+ | 2 | 0.7 | 0 | 0.0 |
| HBsAg+HBeAb+ | 6 | 2.2 | 1 | 0.7 |
| HBsAb+HBeAb+ | 2 | 0.7 | 1 | 0.7 |
| HBV negative | 31 | 11.5 | 8 | 5.9 |
Table 4.
Compare the HBV infective pattern between SLHCC and NHCC.
| Serum markers |
All patients (n = 270) |
Recurrent patients (n = 136) |
χ2 | P | ||
| SLHCC ratio (%) | NHCC ratio (%) | SLHCC ratio (%) | NHCC ratio (%) | |||
| HBsAg+HBeAg+HBcAb+ | 40.0 | 13.3 | 33.3 | 25.0 | 4.244 | 0.039 |
| HBsAg+HBeAb+HBcAb+ | 56.3 | 12.5 | 53.1 | 12.2 | 0.003 | 0.955 |
| HBsAg+HBcAb+ | 70.2 | 12.8 | 73.1 | 11.5 | 0.079 | 0.778 |
| HBsAb+ | 71.4 | 14.3 | 60.0 | 20.0 | 1.833 | 0.176 |
| HBeAb+HBcAb+ | 75.0 | 12.5 | 0.0 | 100.0 | 4.876 | 0.000 |
| HBsAg+ | 63.2 | 5.3 | 50.0 | 12.5 | 4.867 | 0.027 |
| HBV negative | 69.6 | 4.3 | 87.5 | 0.0 | 4.877 | 0.027 |
DISCUSSION
Recurrence and metastasis of HCC could divide into intrahepatic recurrence and extrahepatic metastasis, but 90% of recurrence were intrahepatic, which could be expressed in mono-, poly- and omni-hepatic dissemination. Extrahepatic metastasis means primary lesion of HCC spreading to distant location, it can also accompany with intrahepatic recurrence. Multi-center carcinogenesis of HCC was closely related to postoperative recurrence. It has reported that recurrent rate reach to peak amongst 2 year, about 62-82%. This time after hepatectomy was the most risky period for recurrence and metastasis[11]. Therefore, we divided the recurrent time into early and late phase according to the point of 2 years postoperatively. In this study, recurrent risk factors at early phase were serum AFP level, vascular invasion, incisal margin and operative transfusion. Serum AFP was synthesized on embryo hepatocyte, it was not expressed on normal hepatocytes, but was expressed specifically on hepatic cancer cells. Now serum AFP level usually assayed as utility data contributing to judgment of operative effect on whether it descents to normal level (<20 μg/L). It has reported that serum AFP level would be descended to negative in 18% postoperative patients, and obvious descended in 40%[12]. Nowadays image examination such as B-US, CT scan, magnetic resonance imaging and digital subtraction angiography are limited, even the LR-CT with ultra-iodized oil image through hepatic artery can only find tumor >0.5 cm, and it is difficult to find the micro carcinoma <0.3 cm. So it is important for the surveillance of the recurrence and metastasis of postoperative HCC. Serum AFP level will be changed pre- and post-operation at HCC patients. Our material shows 85 cases occurred positive expression.
Surgery-related vascular invasion, incisal margin and operative transfusion were immediate causes for postoperative recurrence and metastasis at early phase, it means the HCC incision was not radical, and remnant carcinoma continues to grow after operation. As the way of enlarged incisal margin peremptory to prevent postoperative recurrence and metastasis is undesirable, it could not reduce the recurrence and metastasis but lead the patient to liver function failure or death[13-15]. Our experiences are to adopt limited hepatectomy, incisal margin is kept in 1-2 cm especially for the tumor with capsule or pseudocapsule. Owing to abundant liver blood supply, operative transfusion is usually inevitable even though there are lots of hemostasia measures to manipulate hepatic hemorrhage on operation. Investigation has made clear that operative hetero-transfusion could bring out many troubles[16,17]. Our study detected the recurrent rate obviously increased in whom accepted transfusion compared to non-transfusion (P = 0.012), indicated operative transfusion is auxo-action for recurrence and metastasis at the early phase of postoperation. The mechanism maybe: Thrombosis is the advantage of cancer cell implantation, growth factors are released to reinforce cancer cell growth directly or indirectly, to inhibit the NK cell’s activity and result in the function of NK immunocytes decreased, to accelerate the growth of remnant cancer cells. Therefore surgeon should weigh the merits and demerits while performing HCC hepatectomy, adopt acrobatic hepatectomy technique, reduced operative hepatic bleeding and blood transfusion as far as possible, to adopt self-transfusion when necessary. Preoperative vascular invasion or carcinoma thrombosis are fundamental cause for postoperative recurrence and metastasis, leading to decrease of postoperative survival rate. Above all, the micro vascular invasion under microscopy could not be judged, which directly lead to recurrence and metastasis at early phase after hepatectomy[18].
The clinical classification of HCC was the independence related risk factor during the late phase of postoperation. On the basement of previous abundant basic and clinical research, we divided HCC into small HCC (SHCC), solitary large HCC (SLHCC) and nodular HCC (NHCC) according to its clinical pathology propert. Analyzed 29 cases of recurrent data at late phase in contrast with 107 cases at early phase, we found the proportion of SLHCC decreased obviously than NHCC (58.9% vs 48.3%), there was a significant difference (χ2 = 3.929, P = 0.047). It indicated SLHCC has more satisfactory neoplasm biological behavior than NHCC, which coincided with the results of our prophase research[10].
Chronic HBV infection is the main cause of HCC genesis in China. We statistically analyzed the materials of in-patients accepted hepatectomy at our hospital, the HBV infective rate reached 90%. The closed relation between HBV infection and HCC could manifest in the concordance on their epidemiologic character, such as local distribution, sex, age and family history[19]. The main HBV infective pattern of 270 patients was positive “HBsAg, HBeAb, HBcAb” viral markers. Positive HBsAg shows those patients existing with HBV infection, positive HBeAb and HBcAb shows HBV replicated in hepatocytes at lower level. If HBeAb presented long-term positive, the HBV-DNA has been integrated with host hepatocyte chromosome DNA, kept latentiation ever since[20,21]. Our data on 136 cases of recurrence and metastasis shows that the pattern of positive HBcAb was distinctly enhanced. We could explain that the HCC patients with HBV infection have lower immunological function, with decreased resistant ability, HBV promoted recurrence and metastasis by various kinds of pathway as activating HBx function, integrating host cell DNA, destructing apoptosis of normal hepatocyte, inactivating P53 gene, facilitating extracellular matrix degradation[22-24].
Among the recurrent HCC, the HBV infective pattern of NHCC was evidently different from that of SLHCC, furthermore, the proportion of NHCC with HBV infection was evidently increased (P<0.05). It is interpreted that NHCC more easily combines with HBV reproduction than SLHCC in the course of recurrence and metastasis. So the prognosis that NHCC is worse than SLHCC could be elucidated from another point of view.
In conclusion, this study provides an epidemiological evidence that intrahepatic recurrence and metastasis of HCC after hepatic resection has two obvious etiologies. The early recurrence may be mediated by vascular invasion and remnant lesion, the late recurrence by tumor’s clinical pathology propert, as multicentric carcinogenesis or intrahepatic carcinoma de novo. So we could prevent recurrence and metastasis of HCC in the following way: to adopt limited hepatectomy and acrobatic hepatectomy technique, to reduce operative hepatic bleeding and blood transfusion as far as possible, to control HBV infection in HCC patients. In this way, we can increase postoperative survival rate of HCC on whole.
Footnotes
Supported by the National Key Technologies R and D Program, No. 2001BA703B04 and Hunan Province Developing Planning Committee, No. 2001907
Science Editor Li WZ Language Editor Elsevier HK
References
- 1.Chen XP, Qiu FZ, Wu ZD, Zhang BX. Chinese experience with hepatectomy for huge hepatocellular carcinoma. Br J Surg. 2004;91:322–326. doi: 10.1002/bjs.4413. [DOI] [PubMed] [Google Scholar]
- 2.Chen WT, Chau GY, Lui WY, Tsay SH, King KL, Loong CC, Wu CW. Recurrent hepatocellular carcinoma after hepatic resection: prognostic factors and long-term outcome. Eur J Surg Oncol. 2004;30:414–420. doi: 10.1016/j.ejso.2004.01.013. [DOI] [PubMed] [Google Scholar]
- 3.Shimozawa N, Hanazaki K. Longterm prognosis after hepatic resection for small hepatocellular carcinoma. J Am Coll Surg. 2004;198:356–365. doi: 10.1016/j.jamcollsurg.2003.10.017. [DOI] [PubMed] [Google Scholar]
- 4.Huo TI, Lui WY, Wu JC, Huang YH, King KL, Loong CC, Lee PC, Chang FY, Lee SD. Deterioration of hepatic functional reserve in patients with hepatocellular carcinoma after resection: incidence, risk factors, and association with intrahepatic tumor recurrence. World J Surg. 2004;28:258–262. doi: 10.1007/s00268-003-7182-6. [DOI] [PubMed] [Google Scholar]
- 5.Imamura H, Matsuyama Y, Tanaka E, Ohkubo T, Hasegawa K, Miyagawa S, Sugawara Y, Minagawa M, Takayama T, Kawasaki S, et al. Risk factors contributing to early and late phase intrahepatic recurrence of hepatocellular carcinoma after hepatectomy. J Hepatol. 2003;38:200–207. doi: 10.1016/s0168-8278(02)00360-4. [DOI] [PubMed] [Google Scholar]
- 6.Tung-Ping Poon R, Fan ST, Wong J. Risk factors, prevention, and management of postoperative recurrence after resection of hepatocellular carcinoma. Ann Surg. 2000;232:10–24. doi: 10.1097/00000658-200007000-00003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Cha C, Fong Y, Jarnagin WR, Blumgart LH, DeMatteo RP. Predictors and patterns of recurrence after resection of hepatocellular carcinoma. J Am Coll Surg. 2003;197:753–758. doi: 10.1016/j.jamcollsurg.2003.07.003. [DOI] [PubMed] [Google Scholar]
- 8.Yang LY, Wang W, Peng JX, Yang JQ, Huang GW. Differentially expressed genes between solitary large hepatocellular carcinoma and nodular hepatocellular carcinoma. World J Gastroenterol. 2004;10:3569–3573. doi: 10.3748/wjg.v10.i24.3569. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Hui AM, Takayama T, Sano K, Kubota K, Akahane M, Ohtomo K, Makuuchi M. Predictive value of gross classification of hepatocellular carcinoma on recurrence and survival after hepatectomy. J Hepatol. 2000;33:975–979. doi: 10.1016/s0168-8278(00)80131-2. [DOI] [PubMed] [Google Scholar]
- 10.Wang W, Yang LY, Huang GW, Lu WQ, Yang ZL, Yang JQ, Liu HL. Genomic analysis reveals RhoC as a potential marker in hepatocellular carcinoma with poor prognosis. Br J Cancer. 2004;90:2349–2355. doi: 10.1038/sj.bjc.6601749. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Hanazaki K, Kajikawa S, Shimozawa N, Mihara M, Shimada K, Hiraguri M, Koide N, Adachi W, Amano J. Survival and recurrence after hepatic resection of 386 consecutive patients with hepatocellular carcinoma. J Am Coll Surg. 2000;191:381–388. doi: 10.1016/s1072-7515(00)00700-6. [DOI] [PubMed] [Google Scholar]
- 12.Huo TI, Huang YH, Lui WY, Wu JC, Lee PC, Chang FY, Lee SD. Selective prognostic impact of serum alpha-fetoprotein level in patients with hepatocellular carcinoma: analysis of 543 patients in a single center. Oncol Rep. 2004;11:543–550. [PubMed] [Google Scholar]
- 13.Jaeck D, Bachellier P, Oussoultzoglou E, Weber JC, Wolf P. Surgical resection of hepatocellular carcinoma. Post-operative outcome and long-term results in Europe: an overview. Liver Transpl. 2004;10:S58–S63. doi: 10.1002/lt.20041. [DOI] [PubMed] [Google Scholar]
- 14.Cho A, Okazumi S, Miyazawa Y, Makino H, Miura F, Ohira G, Yoshinaga Y, Tohma T, Kudo H, Matsubara K, et al. Limited resection based on reclassification of segment 8 of the liver. Hepatogastroenterology. 2004;51:575–576. [PubMed] [Google Scholar]
- 15.Vauthey JN, Pawlik TM, Abdalla EK, Arens JF, Nemr RA, Wei SH, Kennamer DL, Ellis LM, Curley SA. Is extended hepatectomy for hepatobiliary malignancy justified? Ann Surg. 2004;239:722–730; discussion 730-732. doi: 10.1097/01.sla.0000124385.83887.d5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Kooby DA, Jarnagin WR. Surgical management of hepatic malignancy. Cancer Invest. 2004;22:283–303. doi: 10.1081/cnv-120030217. [DOI] [PubMed] [Google Scholar]
- 17.Rui JA, Zhou L, Liu FD, Chu QF, Wang SB, Chen SG, Qu Q, Wei X, Han K, Zhang N, et al. Major hepatectomy without blood transfusion: report of 51 cases. Chin Med J (Engl) 2004;117:673–676. [PubMed] [Google Scholar]
- 18.Poon RT, Fan ST, Ng IO, Lo CM, Liu CL, Wong J. Different risk factors and prognosis for early and late intrahepatic recurrence after resection of hepatocellular carcinoma. Cancer. 2000;89:500–507. [PubMed] [Google Scholar]
- 19.Chen CH, Chen YY, Chen GH, Yang SS, Tang HS, Lin HH, Lin DY, Lo SK, Du JM, Chang TT, et al. Hepatitis B virus transmission and hepatocarcinogenesis: a 9 year retrospective cohort of 13676 relatives with hepatocellular carcinoma. J Hepatol. 2004;40:653–659. doi: 10.1016/j.jhep.2003.12.002. [DOI] [PubMed] [Google Scholar]
- 20.Ohata K, Hamasaki K, Toriyama K, Ishikawa H, Nakao K, Eguchi K. High viral load is a risk factor for hepatocellular carcinoma in patients with chronic hepatitis B virus infection. J Gastroenterol Hepatol. 2004;19:670–675. doi: 10.1111/j.1440-1746.2004.03360.x. [DOI] [PubMed] [Google Scholar]
- 21.Chen JD, Liu CJ, Lee PH, Chen PJ, Lai MY, Kao JH, Chen DS. Hepatitis B genotypes correlate with tumor recurrence after curative resection of hepatocellular carcinoma. Clin Gastroenterol Hepatol. 2004;2:64–71. doi: 10.1016/s1542-3565(03)00293-3. [DOI] [PubMed] [Google Scholar]
- 22.Pál J, Somogyi C, Szmolenszky A A, Szekeres G, Sípos J, Hegedüs G, Martzinovits I, Molnár J, Németh P. Immunohistochemical assessment and prognostic value of hepatitis B virus X protein in chronic hepatitis and primary hepatocellular carcinomas using anti-HBxAg monoclonal antibody. Pathol Oncol Res. 2001;7:178–184. doi: 10.1007/BF03032346. [DOI] [PubMed] [Google Scholar]
- 23.Feitelson MA, Zhu M, Duan LX, London WT. Hepatitis B x antigen and p53 are associated in vitro and in liver tissues from patients with primary hepatocellular carcinoma. Oncogene. 1993;8:1109–1117. [PubMed] [Google Scholar]
- 24.Théret N, Musso O, Turlin B, Lotrian D, Bioulac-Sage P, Campion JP, Boudjéma K, Clément B. Increased extracellular matrix remodeling is associated with tumor progression in human hepatocellular carcinomas. Hepatology. 2001;34:82–88. doi: 10.1053/jhep.2001.25758. [DOI] [PubMed] [Google Scholar]


