Summary
Vascular development begins with formation of a primary capillary plexus that is later remodeled to give rise to the definitive vasculature. While the mechanism by which arterial and venous fates are acquired is well understood, little is known about when during vascular development arterial and venous vessels emerge and how their growth is regulated. Previously, we have demonstrated that a Hedgehog (HH)/ Vascular endothelial growth factor (VEGF) and Angiopoietin2 (ANG2) signaling pathway is essential for the development of the coronary vasculature. Here we use conditional gene targeting to identify the cell types that receive HH signaling and mediate coronary vascular development. We show that HH signaling to the cardiomyoblast is required for the development of coronary veins, while HH signaling to the perivascular cell (PVC) is necessary for coronary arterial growth. Moreover, the cardiomyoblast and PVC appear to be the exclusive cell types that receive HH signals, as ablation of HH signaling in both cell types leads to an arrest in coronary development. Finally, we present evidence suggesting that coronary arteries and veins may be derived from distinct lineages.
Keywords: Hedgehog (HH), Vascular endothelial growth factor (VEGF), Angiopoietin (Ang), heart development, coronary vascular development, myocardium, pericyte
Introduction
Vascular development is governed by two processes, vasculogenesis and angiogenesis. Vasculogenesis refers to the formation of blood vessels via de novo differentiation of either angioblast or hemangioblast precursors, while angiogenesis is defined as the growth or remodeling of established blood vessels. In general, vascular systems undergo a stereotyped pattern of development beginning with the formation of a primary capillary plexus that is later remodeled, giving rise to the mature vasculature. It is thought that the primary capillary plexus forms by vasculogenesis and is remodeled via angiogenesis (Flamme et al., 1997; Risau, 1997).
A critical component of capillary plexus remodeling is the emergence of a vascular tree composed of larger proximal and smaller distal vessels. In addition, it is thought that arterial and venous vessels differentiate during this remodeling process. Thus, the remodeling process yields many of the components of the mature circulatory system, including larger arteries and veins, medium sized arterioles and venules, and smaller capillaries (Risau, 1997). Interestingly, it has been reported that capillaries also acquire arterial and venous fates (Gerety et al., 1999; Shin et al., 2001; Wang et al., 1998).
The molecular mechanism underlying acquisition of arterial versus venous cell fates has been recently described. These analyses were based on the observation that arterial and venous endothelial cells differentially expressed ephrinB2 and its receptor, ephB4, respectively (Gerety et al., 1999; Shin et al., 2001; Wang et al., 1998). Intriguingly, while deletion of either ephrinB2 or ephB4 in mice severely affected vascular development, the differential expression of ephrinB2 and ephB4 was not affected, indicating that factors acting upstream of these genes control arterial versus venous identity (Lawson and Weinstein, 2002). Further work has identified these factors as components of the Notch signaling pathway. Notch1, 3, 4 and the notch ligand, Dll4, are expressed in arterial endothelial cells, are required for vascular development, and control ephrinB2 expression (Domenga et al., 2004; Duarte et al., 2004; Fischer et al., 2004; Krebs et al., 2000). Activation of Notch signaling is sufficient to promote acquisition of the arterial cell fate, and in the absence of Notch signaling, blood vessels initially form but all express the venous markers ephB4 and flt4 (Lawson et al., 2001).
Further analysis in zebrafish has identified that a Sonic Hedgehog (SHH)/Vascular Endothelial Growth Factor (VEGF) pathway acts upstream of Notch, functioning to select which endothelial cells will receive Notch signaling and take on the arterial fate. During somitogenesis, notochord derived SHH signals regulate expression of Vegf-A in the somitic mesoderm. In turn, VEGF-A activates the expression of Notch5 preferentially in dorsally situated endothelial precursors, thus restricting Notch signaling to cells that will give rise to the dorsal aorta (Lawson et al., 2002).
In addition to the role of Hedgehog (HH) signaling in arterial specification in zebrafish, several studies have provided evidence that HH signaling functions more broadly in vascular development. Mouse embryos lacking Smoothened (transducer of HH signaling) display defects in vasculogenesis, SHH promotes vascular plexus formation in cell culture, and activation of HH signaling in the adult mouse is sufficient to promote neovascularization in several different tissues (Kanda et al., 2003; Pola et al., 2001; Vokes et al., 2004) . Moreover, we have previously reported that a HH/VEGF/Angiopoietin2 (ANG2) signaling pathway is essential for coronary vascular development (Lavine et al., 2006).
Similar to other vascular systems, coronary development begins with the formation of a vascular network that is later remodeled to give rise to the mature coronary tree (Kattan et al., 2004; Morabito et al., 2002). Interestingly, the initial coronary vascular plexus consists of two sets of blood vessels located in different positions, the subepicardial mesenchyme and the myocardial wall. We have shown that HH signaling controls the growth of both sets of blood vessels via induction of Vegf-A, -B, -C, and Angiopoietin2 expression (Lavine et al., 2006). However, the mechanism by which HH signaling can coordinately control the development of both vascular structures is unclear.
We have proposed that HH signaling to two different cell types, cardiomyoblasts and perivascular cells, controls the growth of blood vessels located within the subepicardial mesenchyme and within the myocardial wall, respectively. Additionally, we hypothesized that subepicardial and intramyocardial blood vessels represent distinct vessel types. In this work, we identify the recipients of HH signaling in the embryonic heart using conditional gene targeting. We show that the cardiomyoblast and the perivascular cell are the functionally relevant targets of HH signaling during coronary vascular development. We demonstrate that HH signaling to the cardiomyoblast controls subepicardial blood vessel development, while HH signaling to the perivascular cell controls intramyocardial development. Furthermore, we show that subepicardial vessels are veins and intramyocardial vessels are arteries.
Materials and Methods
Mouse lines
Mlc2v-Cre (Chen et al., 1998a), Dermo1-Cre (Yu et al., 2003), Smof/f (Long et al., 2001), ephrinB2-LacZ (Wang et al., 1998), ephB4-LacZ (Gerety et al., 1999), and ROSA26R (Soriano, 1999) mice were maintained on a C57/Bl6J background or bred at least three generations onto C57/Bl6J. Littermate controls for conditional knockout experiments included animals with a genotype of Smof/+; Cre or Smof/f (and no Cre allele).
Whole Mount PECAM Immunohistochemistry
PECAM staining was performed as described (Lavine et al., 2005). Briefly, tissues were fixed in 4% PFA and dehydrated in a methanol series, incubated in methanol/hydrogen peroxide, rehydrated, and blocked in PBSST (5% goat serum/PBS 0.1% Triton X100). Primary antibody: rat anti-mouse PECAM (Pharminogen, 1:200). Biotinylated goat anti-rat IgG (Vector, 1:200) was used followed by Vectastain ABC-peroxidase reagent and DAB visualization (Vector). All antibody and ABC reagent dilutions were done in PBSST. Antibody and ABC reagent incubations were carried out at 4°C overnight. Following each overnight incubation, tissues were washed five times (1 hr each at 4°C) with PBSST. Following photography, PECAM stained hearts were paraffin embedded and sectioned. Paraffin sections (4μ) were then dewaxed, rehydrated, counterstained with hematoxylin (Sigma) and mounted. Whole mount specimens were photographed at 25X and histological sections at 400X magnification.
Blood vessel density was quantified by counting the number of vessels per unit area (10,000 square pixels). At least 6 biological specimens were examined per genotype. For quantitation of the number of vessels per 20X field, 3 representative fields were analyzed from 3 biological samples for each genotype. The number of subepicardial and intramyocardial blood vessels were quantified and plotted. The error bars represent 1 standard deviation from the mean.
LacZ staining was performed as described (Soriano, 1999).
Fluorescent Immunohistochemistry
12μ cryosections were cut from E12.5-E13.5 hearts and stained with primary antibodies to PECAM (R&D) 1:200, human CD4 (R&D) 1:200, CD45 (R&D) 1:200, Sca1 (R&D) 1:50, Cardiac Actin (Sigma) 1:400, VEGF-A (Santa Cruz) 1:200, VEGF-B (Santa Cruz) 1:200, VEGF-C (Santa Cruz) 1:200, and β-galactosidase (Abcam) 1:250. The following secondary antibodies were used at 1:200: anti-rat Alexa 555, anti-rabbit Alexa 488, anti-rabbit Alexa 647 (Molecular Probes), and anti-mouse IgM FITC (Vector Labs). Immunofluorescence was visualized on a Ziess Apotome Microscopy system. All specimens were photographed at 400X magnification.
For 3D reconstruction of PECAM staining, 40μ cryosections were cut from E12.5-E13.5 hearts and stained with a primary antibody to PECAM (R&D) 1:200 and anti-rat Alexa 555 secondary antibody (Molecular Bioprobes) 1:200. Using the Ziess Apotome system, 40 images, each spanning 1μ, were acquired per section and processed with Ziess Axiovision software to produce a compressed Z-stack image. Representative images are at 400X magnification.
Quantification of VEGF protein expression
VEGF-A, VEGF-B, and VEGF-C expression was quantified by measuring the immunofluorescent signal in both the myocardium and perivascular cells from control, Smomlc2v, and Smodermo1 E12.5 hearts. Regions containing myocardial and perivascular cells were identified by co-labeling with PECAM. Perivascular regions corresponded to cells within the immediate proximity of intramyocardial blood vessels, while myocardial areas corresponded to those not in the immediate vicinity of intramyocardial blood vessels. Average pixel intensity was measured in 9 total tissue sections for each genotype (3 sections each from 3 independent hearts). Pixel intensity was quantified using Adobe Photoshop software (and confirmed using other software packages including Metamorph and Canvas) and displayed as relative to control. Error bars represent one standard deviation from the mean. p-values were calculated using Student's T-test.
Whole Mount in situ Hybridization
Whole mount in situ hybridization was performed as previously described (Lavine et al., 2005). Tissues were photographed and then cryosectioned (16μ), mounted on slides and re-photographed. In situ probes:Vegf-A, Vegf-B, Vegf-C, and Ptc1 (Lavine et al., 2006). All comparisons shown are between littermates and all experiments were repeated at least 3 times. Cryosections are at 400X magnification.
Results
Subepicardial and intramyocardial blood vessels represent distinct venous and arterial lineages
Subepicardial and intramyocardial blood vessels represent two vascular subsets that appear to be preferentially regulated by HH signaling to different cell types. Activation of HH signaling in the myocardium promotes subepicardial blood vessel growth but not intramyocardial blood vessel growth (Lavine et al., 2006). These data suggest that subepicardial and intramyocardial blood vessels may represent distinct vascular lineages. Consistent with this hypothesis, corrosion casting of late gestation rat hearts has demonstrated that coronary arteries are positioned within the myocardial wall while veins are located closer to the epicardial surface (Ratajska et al., 2003), suggesting that subepicardial and intramyocardial blood vessels may represent venous and arterial blood vessels, respectively.
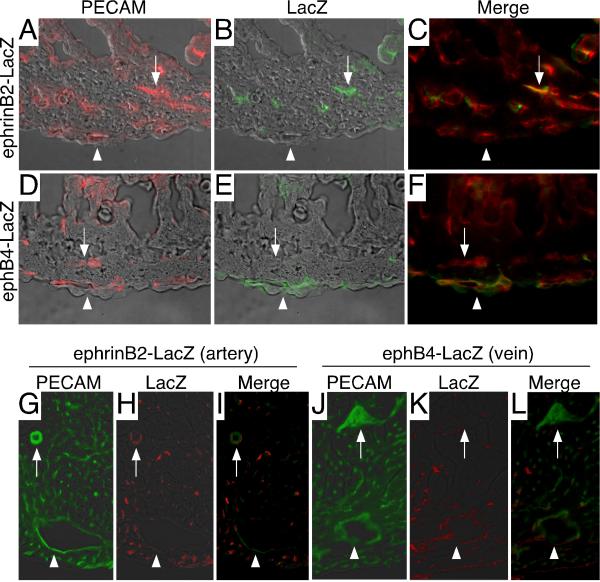
To identify arterial and venous blood vessels within the developing heart, we examined mice harboring LacZ inserted into either the ephrinB2 or ephB4 loci. EphrinB2-LacZ and ephB4-LacZ mice specifically express β-galactosidase in arteries and veins, respectively (Gerety et al., 1999; Wang et al., 1998). Immunofluorescent staining of E12.5 hearts with antibodies against the endothelial marker, PECAM (CD31), and β-galactosidase demonstrated that ephrinB2 is expressed within intramyocardial blood vessels and ephB4 is expressed within subepicardial blood vessels during the vascular plexus stage (Fig. 1A-F). Similar to the embryonic heart, immunofluorescent staining of adult hearts demonstrated that, in general, larger arteries are located deeper within the myocardial wall, while larger veins are positioned closer to the epicardial surface. In contrast, smaller arterial and venous blood vessels are dispersed throughout the myocardium (Fig. 1G-L). These analyses indicate that subepicardial and intramyocardial vessels represent coronary veins and arteries, respectively. Moreover, coronary artery and vein identity is specified during or prior to the vascular plexus stage, and the relative positions of larger coronary arteries and veins, established during vascular plexus development, are maintained in the adult heart.
Figure 1. Subepicardial and intramyocardial blood vessels represent venous and arterial lineages.
A-F, Immunofluorescent staining of cryosections for PECAM (A, D -red) and LacZ (B, E -green). Staining of E12.5 ephrinB2-LacZ mice (A-C) reveals that intramyocardial (arrow), but not subepicardial (arrowhead) blood vessels express the arterial marker, ephrinB2. Similar analysis of E12.5 ephB4-LacZ (D-F) mice demonstrates that subepicardial (arrowhead), but not intramyocardial, blood vessels (arrow) express the venous marker, ephB4. G-L, Immunofluorescent staining of cryosections from ephrinB2-LacZ (G-I) and ephB4-LacZ (J-L) adult hearts shows that the spatial relationship between larger arteries and veins is conserved in the adult heart. Green-PECAM (G, J) and Red-LacZ (H, K). C, F, I, and L are merges of fluorescent signals from A-B, D-E, G-H, and J-K, respectively.
The cardiomyoblast and perivascular cell (PVC) are the exclusive targets of HH signaling
In addition to the cardiomyoblast, Ptc1 is expressed in perivascular cells (PVCs), suggesting that PVCs are also targets of HH signaling. Consistent with this, Vegf-A, Vegf-B, and Vegf-C are also expressed in these cells (Lavine et al., 2006).
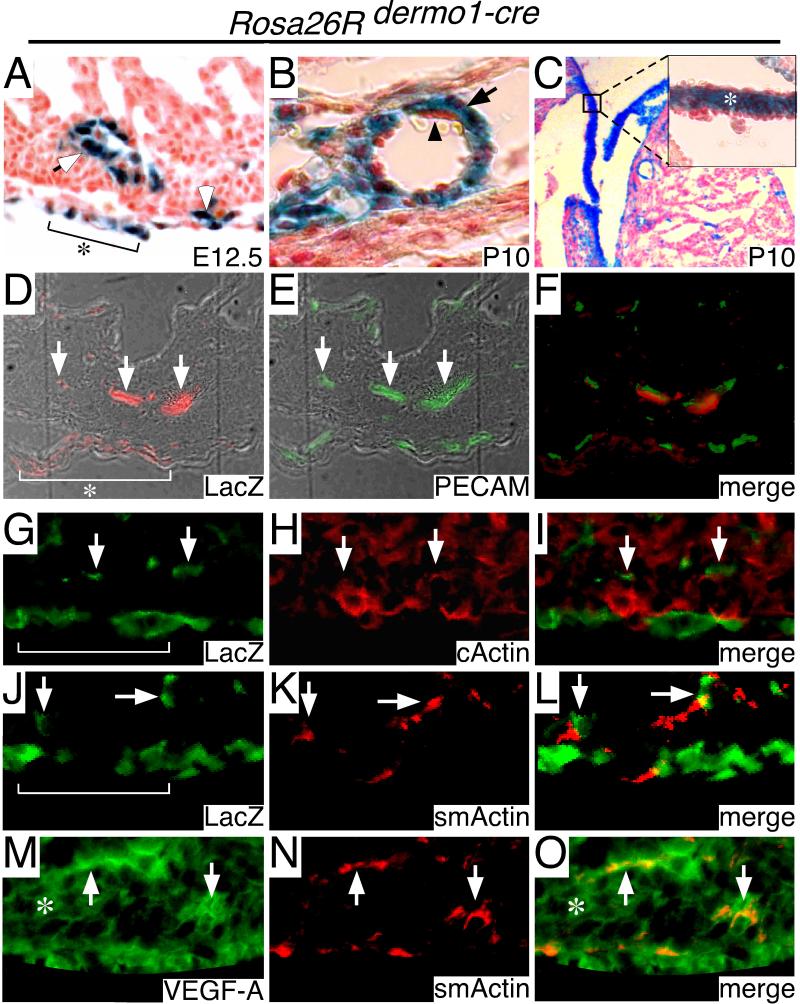
PVCs act as supporting cells for developing blood vessels and give rise to vascular smooth muscle and adventitial fibroblasts (Yoshida and Owens, 2005). Previously, we inserted the Cre-recombinase cDNA into the Dermo1 (Twist2) locus (Yu et al., 2003). Using the Rosa26-LacZ reporter mouse (Soriano, 1999), we have identified sites of Dermo1-cre activity in the developing heart. Beginning at E11.5, segmental regions of the epicardium display Dermo1-cre activity (data not shown). At E12.5, Dermo1-cre activity is present at epicardial sites that appear to be undergoing an epithelial mesenchymal transformation (EMT) and in cells located adjacent to developing intramyocardial blood vessels (Fig. 2A, D-F). Further characterization revealed that cells of the Dermo1-cre lineage express smooth muscle actin and do not express cardiac actin (Fig. 2G-L). Moreover, these cells express VEGF-A consistent with the notion that Dermo1-cre marks the PVC lineage (Fig. 2M-O). Given that PVCs and the subepicardial mesenchyme are derived from epicardium (Dettman et al., 1998; Merki et al., 2005; Mikawa and Gourdie, 1996; Vrancken Peeters et al., 1999), these data suggest that Dermo1-cre marks epicardial cells fated to undergo EMT that later take residence within either subepicardial mesenchymal or perivascular locations. This is consistent with proposed functions of Twist in EMT (Kang and Massague, 2004).
Figure 2. Dermo1-cre is active in the perivascular cell lineage.
A-C, LacZ staining of Dermo1-cre/Rosa26-LacZ hearts showing that at E12.5 (A) Dermo1-cre marks cells localized in patches of the epicardium that are undergoing EMT (bracket with asterisk, white arrowhead) and around intramyocardial blood vessels (open arrow). (B) LacZ staining of postnatal day 10 (P10) hearts demonstrating that Dermo1-cre expressing cells give rise to perivascular interstitial cells (black arrow), but not the vascular endothelium (black arrowhead). (C, insert) Dermo1-cre positive cells also give rise to the interstitium of the valvular leaflets (white asterisk). D-F, Immunofluorescent staining of crosections for LacZ (D, red) and PECAM (E, green) confirming that Dermo1-cre marks cells positioned immediately adjacent to the intramyocardial blood vessels (white arrow) and in patches of the epicardium (bracket with asterisk). Merge of D and E is shown in F. G-L, Immunofluorescent staining for LacZ (G, J -green), cardiac actin (H, red) and smooth muscle actin (K, red) demonstrating that Dermo1-cre marked cells (white arrows) express smooth muscle actin and not cardiac actin. Bracket denotes position of the epicardium. I, L merged images of G-H and J-K, respectively. M-O, Immunofluorescent staining for VEGF-A (M, green) and smooth muscle actin (N, red) revealing that smooth muscle cells located within the myocardial wall (white arrows) express VEGF-A. Asterisk denotes VEGF-A expressing myocardial cells. O, merged image of M-N.
LacZ staining of postnatal Dermo1-cre/Rosa26-LacZ hearts demonstrated LacZ activity in cells located within the interstitial space, including both vascular smooth muscle cells and fibroblasts (Fig. 2B). In addition, interstitial cells of the valve leaflets also displayed LacZ activity (Fig. 2C, insert). These data suggest that Dermo1-cre may be active in a precursor cell that gives rise to both the smooth muscle cell and fibroblast lineages.
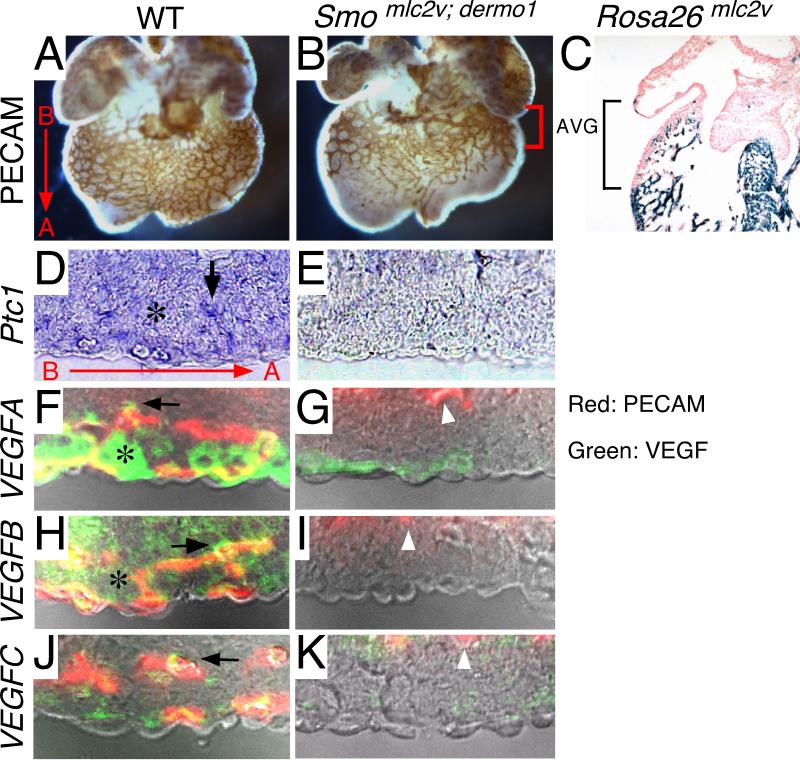
We have postulated that the cardiomyoblasts and perivascular cells are the relevant cell types that receive HH signaling during coronary development. To test this hypothesis, we concurrently deleted Smoothened (Smoflox) (Long et al., 2001) in both cardiomyoblasts and perivascular cells using Mlc2v-cre (Chen et al., 1998b) and Dermo1-cre, respectively. PECAM staining of E13.5 control and Smomlc2v; dermo1 CKO hearts revealed that loss of HH signaling in both cardiomyoblasts and perivascular cells severely disrupted coronary development. Smomlc2v; dermo1 CKO hearts failed to form a capillary plexus that significantly extended beyond the region surrounding the atrial-ventricular groove (Fig. 3A-B). Interestingly, myocardial cells adjacent to the atrial-ventricular groove are not targeted by Mlc2v-cre (Fig. 3C), potentially explaining why blood vessels are still present in the proximity of this region.
Figure 3. HH signaling to the cardiomyoblast and pericyte is essential for coronary development.
A-B, Whole mount PECAM immunohistochemistry demonstrating that compared to controls (B), Smomlc2v; dermo1 CKO hearts (A) display an arrest in coronary development. Bracket denotes atrial ventricular groove. C, β-galactosidase staining of an E12.5 Mlc2v-cre/ Rosa26R heart demonstrating that Mlc2v-cre does not efficiently target cardiomyoblasts located in the atrial ventricular grove (AVG, bracket). In contrast, Mlv2v-cre efficiently targets the ventricular myocardium (area below bracket). D-E, Cryosections of hearts stained with in situ probes for Ptc1 revealing that compared to controls, Smomlc2v; dermo1 CKO hearts lack both myocardial (asterisk) and perivascular (arrow) sources of Ptc1 expression. F-K, Immunofluorescent staining of cryosections for PECAM (red) and VEGF ligands (green) reveal that compared to controls, Smomlc2v; dermo1 CKO hearts display diminished expression of VEGF-A (F-G), VEGF-B (H-I), and VEGF-C (J-K). White arrowhead denotes (PECAM positive cells) endocardial cells that appear unaffected in these conditional mutants. Red arrow in A, D denotes orientation of tissue sections in relation to whole mount photographs (B- base and A- apex).
Consistent with loss of HH signaling, cryosections of Smomlc2v; dermo1 CKO hearts stained with a Ptc1 in situ probe showed loss of Ptc1 expression in both cardiomyoblasts and perivascular cells (Fig. 3D-E). Moreover, immunofluorescent analysis for VEGF-A, VEGF-B, and VEGF-C revealed that compared to controls, Smomlc2v; dermo1 CKO hearts expressed significantly lower levels of VEGF-A protein (Fig. 3F-G) and undetectable levels of VEGF-B and VEGF-C protein (Fig. 3H-K). In addition, whole mount in situ hybridization for Vegf-A, Vegf-B and Vegf-C demonstrated that compared to controls, Smomlc2v; dermo1 CKO hearts displayed reduced Vegf-A, Vegf-B and Vegf-C expression (Supp. Fig. 1). Together with the failure of vascular development in Smomlc2v; dermo1 CKO hearts, these data indicate that the cardiomyoblast and perivascular cells are the predominant cell types that receive HH signaling during coronary development.
HH signaling to the cardiomyoblast and perivascular cells are respectively necessary for coronary vein and artery development
Previously, we have shown that activation of HH signaling in the myocardium preferentially promotes subepicardial blood vessel growth with little effect on intramyocardial vessel growth, suggesting that subepicardial and intramyocardial blood vessel development is controlled by HH signaling to different cell types (Lavine et al., 2006). To test this hypothesis, we examined vascular development in hearts in which HH signaling was inactivated in either the myocardial or perivascular cell.
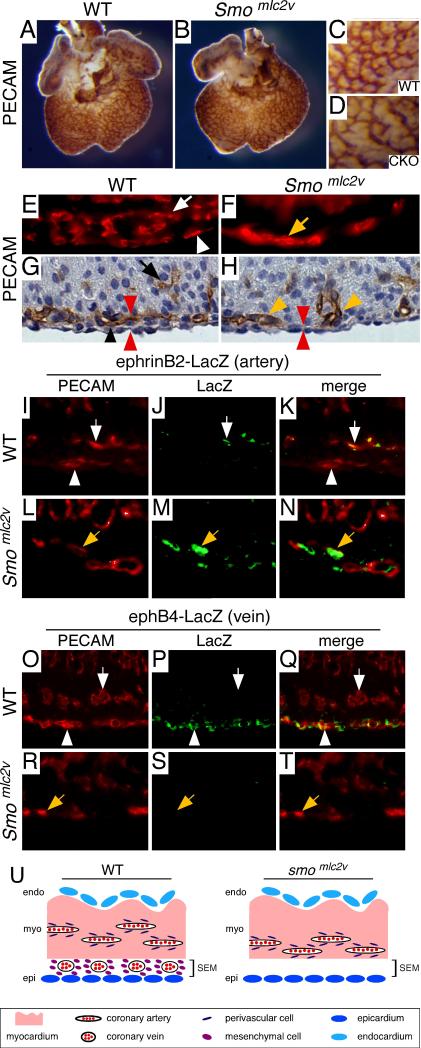
Similar to littermate controls, hearts lacking HH signaling in the myocardium (Smomlc2v CKO) contained a coronary plexus that encased the entire ventricle (Fig. 4A-B). However, in contrast to control hearts, the vascular plexus of Smomlc2v CKO hearts contained fewer vessels (Fig. 4C-D). Quantitation of blood vessel density revealed that compared to controls, Smomlc2v CKO hearts had fewer blood vessels per unit area (70.3 ± 3.1 and 40.7 ± 4.0, p<0.001).
Figure 4. Myocardial HH signaling is required for coronary vein development.
A-D, Whole mount immunohistochemistry for PECAM demonstrating that both control (A) and Smo mlc2v CKO (B) hearts form a vascular plexus that encases the entire ventricle. High magnification of A-B demonstrating that the vascular plexus of Smomlc2v CKO (D) hearts is less dense than controls (C). E-F, 3D reconstructions of cryosections stained with antibodies to PECAM showing that while E13.5 control hearts (E) display both subepicardial (white arrowhead) and intramyocardial (white arrow) blood vessels, Smomlc2v CKO hearts (F) contain only a single layer of vasculature (yellow arrow). G-H, Histological sections of PECAM stained hearts reveal that compared to E13.5 controls (G), Smomlc2v CKO hearts (H) do not contain a blood vessels growing within the subepicardial space (red arrowheads) and only possess blood vessels growing with the myocardial wall (yellow arrow). I-T, Immunofluorescent staining for ephrinB2-LacZ (I-N) and ephB4-LacZ (O-T) E13.5 hearts with antibodies against PECAM (I, L, O, R -red) and LacZ (J, M, P, S -green). In contrast to control hearts (I-K, O-Q), which contain ephrinB2 expressing intramyocardial vessels (white arrows) and ephB4 expressing subepicardial vessels (white arrowheads) Smomlc2v CKO hearts (L-N, R-T) possess only a single set of vasculature expressing ephrinB2 (yellow arrows).
Immunofluorescent PECAM staining of cryosections revealed that while control hearts contained subepicardial and intramyocardial blood vessels, Smomlc2v CKO hearts contained only a single set of blood vessels (Fig. 4E-F). Histological analysis demonstrated that the vasculature of Smomlc2v CKO hearts appeared to be positioned within the outer region of the myocardial wall and no blood vessels were seen within the subepicardial space (Fig. 4G-H).
To further characterize the identity of blood vessels present in Smomlc2v CKO hearts, we bred the ephrinB2- and ephB4-LacZ alleles into the Smomlc2v CKO background. Immunofluorescent staining for PECAM and β-galactosidase demonstrated that, similar to controls, Smomlc2v CKO hearts contained blood vessels bearing ephrinB2 expression. However, no ephB4 expressing vasculature was detected, indicating that Smomlc2v CKO hearts contained arterial but lacked venous vasculature (Fig. 4I-T).
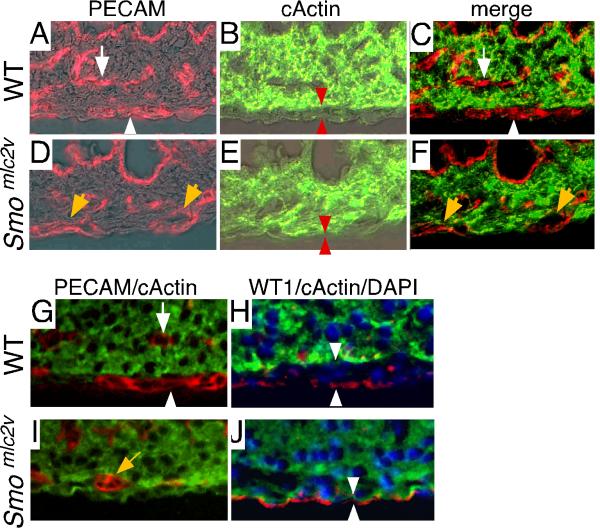
In addition to containing only arterial blood vessels, Smomlc2v CKO hearts lacked a subepicardial mesenchyme. Co-labeling with PECAM and cardiac actin antibodies confirmed that Smomlc2v CKO hearts only contained blood vessels located within the myocardial wall and lacked blood vessels growing within the subepicardial space (Fig. 5A-F). Immunohistochemistry for PECAM, cardiac actin, WT1, and DAPI demonstrated that not only do Smomlc2v CKO hearts lack subepicardial blood vessels, but they do not contain subepicardial mesenchyme (Fig. 5G-J). These data indicate that HH signaling to the cardiomyoblast is required for subepicardial vessel development and formation of the subepicardial mesenchyme. Furthermore, the mislocalization of intramyocardial vessels to the outer region of the myocardial wall raises the possibility that subepicardial and intramyocardial blood vessels may interact to coordinately regulate each other's growth and position.
Figure 5. Defective subepicardial mesenchyme development in Smomlc2v CKO hearts.
A-F, Immunofluorescent staining for cardiac actin (B, E green) and PECAM (A, D red) confirming that while control hearts (A-C) contain both subepicardial (white arrow head) and intramyocardial (white arrow) blood vessels, Smomlc2v CKO hearts (D-F) only contain blood vessels growing within the myocardial wall (yellow arrow). A-B and D-E fluorescent signals are superimposed on DIC images for orientation. C, F merge of fluorescent signals in A-B and D-E, respectively. Red arrowheads in B, E denote position of subepicardial mesenchyme. G-J, Immunofluorescent staining for PECAM (G, I red), cardiac (G-J green), WT1 (H, J red), and DAPI (H, J blue) demonstrating that compared to control hearts (G-H) that contain well developed subepicardial mesenchyme (denoted by white arrowheads, H), Smomlc2v CKO hearts (I-J) only contain blood vessels growing within the myocardial wall and fail to develop subepicardial mesenchyme (white arrowheads, J).
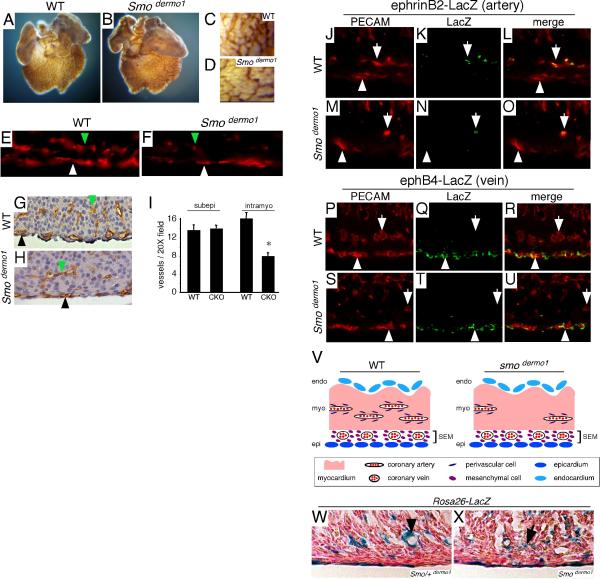
Examination of hearts that lacked HH signaling in the Dermo1-cre lineage (Smodermo1 CKO) revealed that similar to controls, Smodermo1 CKO hearts also contained a vascular plexus that encased the entire ventricle (Fig. 6A-B). However, similar to Smomlc2v CKO hearts, quantitation of blood vessel density revealed that Smodermo1 CKO hearts contained fewer blood vessels per 20X field compared to controls (71.7 ± 3.6 and 45.0 ± 5.9, p<0.001). Histological analysis demonstrated that Smodermo1 CKO hearts displayed specific defects in intramyocardial blood vessel development. Smodermo1 CKO hearts contained 8.0 ± 0.7 intramyocardial blood vessels/ 20X field, while control hearts contained 16.2 ± 1.3 intramyocardial blood vessels/ 20X field (p<0.0001). Interestingly, subepicardial blood vessel development remained intact in Smodermo1 CKO hearts (Fig. 6C-I).
Figure 6. Perivascular HH signaling is essential for coronary artery growth.
A-D, Whole mount immunohistochemistry for PECAM showing that both control (A) and Smodermo1 CKO (B) hearts contain a vascular plexus that encases the entire ventricle. High magnification demonstrating that the vascular plexus of Smodermo1 CKO hearts (D) is less dense compared to that of controls (C). E-F, 3D reconstructions of cryosections stained with antibodies to PECAM showing that Smodermo1 CKO hearts (F) contain a normal compliment of subepicardial blood vessels (white arrowhead) but fewer intramyocardial blood vessels (green arrowhead) compared to control hearts (E). G-H, Histological sections of PECAM stained control (G) and Smodermo1 CKO (H) hearts demonstrating that Smodermo1 CKO hearts contain similar numbers of subepicardial vessels (black arrowhead) but fewer intramyocardial vessels (green arrowhead) compared to controls. I, Quantitation of the number of subepicardial and intramyocardial vessels per 20X field in control and Smodermo1 CKO hearts. Asterisk indicates a statistically significant difference compared to controls (p<0.001). J-U, Immunofluorescent staining of ephrinB2-LacZ (J-O) and ephB4-LacZ (P-U) E13.5 hearts with antibodies against PECAM (J, M, P, S -red) and LacZ (K, N, Q, T -green). Compared to control hearts (J-L, P-R), Smodermo1 CKO hearts (M-O, S-U) contained fewer ephrinB2 expressing intramyocardial blood vessels (white arrows) but similar numbers of ephB4 expressing subepicardial blood vessels (white arrowheads). V-W, β-galactosidase staining for Rosa26-LacZ demonstrating that Dermo1-cre expressing cells are present in both control (W) and Smodermo1 CKO (X) hearts. In control hearts, Dermo1-cre expressing cells are present in a perivascular distribution (black arrowhead), while these cells are scattered throughout the heart in Smodermo1 CKO hearts (black arrow).
To confirm that decreased intramyocardial blood vessel number in Smodermo1 CKO hearts represented a deficit in arterial blood vessel development, we bred the ephrinB2- and ephB4-LacZ alleles into the Smodermo1 CKO background. Immunohistochemistry for PECAM and β-galactosidase revealed that Smodermo1 CKO hearts contained fewer ephrinB2 expressing vessels but normal numbers of ephB4 expressing vessels, indicating that Smodermo1 CKO hearts display defects specific to the arterial vasculature (Fig. 6J-U).
One potential mechanism explaining the selective loss of coronary arteries in Smodermo1 CKO hearts is that HH signaling to perivascular cells is necessary for their survival. As a result, the absence of perivascular cells would lead to failure of arterial blood vessel development. To examine whether this might be the case, we bred the Rosa26-LacZ reporter into the Smodermo1 CKO background. β-galactosidase staining for Rosa26-LacZ revealed that both control and Smodermo1 CKO hearts contained Dermo1-Cre expressing cells (Fig. 6W-X). However, while control hearts demonstrated a perivascular distribution of Dermo1 expressing cells, these cells were scattered throughout the myocardium of Smodermo1 CKO hearts. These data demonstrate that HH signaling to perivascular cells is not required for their survival, suggesting that HH signaling to this cell type may control arterial blood vessel growth within the myocardial wall by promoting the local expression of pro-angiogenic factors. Alternatively, HH signaling to perivascular cells may be necessary for proper differentiation of this cell type.
HH signaling to the cardiomyoblast and perivascular cell is required for the expression of specific VEGF ligands
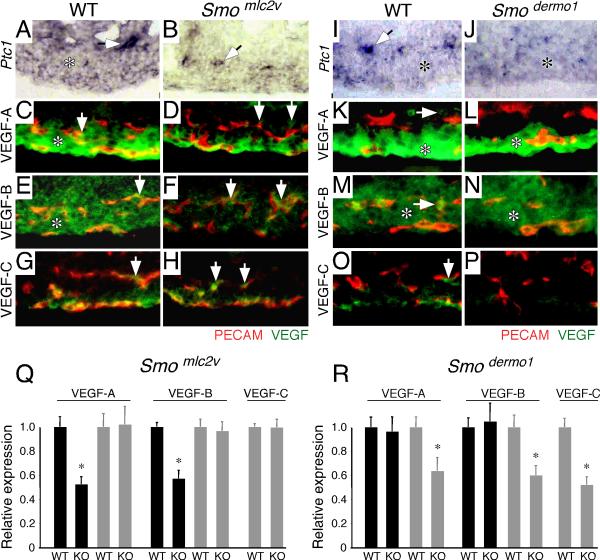
We have previously shown that HH signaling promotes coronary vascular development by inducing Vegf-A, Vegf-B, Vegf-C, and Ang2 expression (Lavine et al., 2006). To determine whether HH signaling to the cardiomyoblast regulates a specific subset of these factors, E13.5 control and Smomlc2v CKO hearts were analyzed by in situ hybridization for Ptc1 and by immunohistochemistry for VEGF-A, VEGF-B, and VEGF-C.
Compared to controls, Smomlc2v CKO hearts displayed diminished levels of Ptc1 in the myocardium of Smomlc2v CKO hearts. However, perivascular Ptc1 expression was present in Smomlc2v CKO hearts (Fig. 7A-B). Similar to that of Ptc1, immunofluorescent analysis with antibodies against VEGF-A and VEGF-B confirmed the loss of myocardial expression and retention of perivascular expression of VEGF-A and VEGF-B protein in Smomlc2v CKO hearts (Fig. 7C-F). Immunolabeling for VEGF-C demonstrated that both control and Smomlc2v CKO hearts displayed comparable perivascular VEGF-C protein expression (Fig. 7G-H). Quantitative analysis of fluorescent intensity confirmed that compared to controls, Smomlc2v CKO hearts had significant (p<0.001) reductions in myocardial VEGF-A and VEGF-B expression (Fig. 7Q). In situ hybridization for Vegf-A, Vegf-B and Vegf-C confirmed reductions in myocardial expression of Vegf-A and Vegf-B in Smomlc2v CKO hearts (Supp. Fig. 1).
Figure 7. Myocardial and perivascular HH signaling is required for tissue specific VEGF ligand expression.
A-B, Cryosections of hearts stained with in situ probes for Ptc1. While control hearts (A) contain expression of Ptc1 in both cardiomyoblasts (asterisk) and perivascular cells (arrow), Smomlc2v CKO hearts (B) only express Ptc1 in perivascular cells (arrow). C-H, Immunofluorescent staining of cryosections for PECAM (red) and VEGF ligands (green). Compared to control hearts (C, E), which express VEGF-A and VEGF-B in both cardiomyoblasts (asterisk) and perivascular cells (arrow), Smomlc2v CKO hearts (D, F) only express VEGF-A and VEGF-B in perivascular cells (arrow). VEGF-C is expressed in perivascular cells of both control (G) and Smomlc2v CKO (H) hearts. I-J, Cryosections of hearts stained with an in situ probe for Ptc1. While control hearts (I) express Ptc1 in both cardiomyoblasts (asterisk) and perivascular cells (arrow), Smodermo1 CKO hearts (J) only express these transcripts in the cardiomyoblast (asterisk). K-P, Immunofluorescent staining of cryosections for PECAM (red) and VEGF ligands (green). Compared to control hearts (K, M), which express VEGF-A and VEGF-B protein in both cardiomyoblasts (asterisk) and perivascular cells (arrow), Smodermo1 CKO hearts (L, N) only express VEGF-A and VEGF-B in cardiomyoblasts (asterisk). VEGF-C is expressed in perivascular cells of control (O), but not Smodermo1 CKO (P) hearts. Q-R, Quantitative analysis of VEGF expression demonstrating statistically significant alterations in VEGF ligand expression in Smomlc2v CKO (Q) and Smodermo1 CKO (R) hearts compared to control hearts. Black bars represent myocardial expression and grey bars represent perivascular expression. Asterisk indicates a statistically significant difference compared to controls (p<0.01).
These analyses indicate that HH signaling to the cardiomyoblast regulates Vegf-A and Vegf-B expression in a cell autonomous manner. This is consistent with our previous finding that activation of HH signaling in the cardiomyoblast can upregulate Vegf-A expression cell autonomously (Lavine et al., 2006). Together with the finding that HH signaling to the myocardium is essential for subepicardial vessel growth, these data support the conclusion that HH signaling to the myocardium controls subepicardial vessel development by regulating myocardial VEGF-A and VEGF-B expression.
Analysis of Smomlc2v CKO hearts revealed that while subepicardial vessel development was disrupted, intramyocardial vessel development was intact. Moreover VEGF-A and VEGF-B expression was specifically lost from the myocardium but retained in PVCs of Smomlc2v CKO hearts. These data suggest that perivascular expression of VEGF ligands regulates intramyocardial blood vessel development. Given that disruption of HH signaling in PVCs leads to specific defects in intramyocardial vessel development, we hypothesized that HH signaling to PVCs regulates expression of VEGF ligands in these cells. This hypothesis is supported by the loss of both cardiomyoblast and perivascular sources of VEGF ligand expression in Smomlc2v; dermo1 CKO hearts (Fig. 3).
Consistent with loss of perivascular HH signaling, in situ hybridization for Ptc1 revealed decreased expression in perivascular cells of Smodermo1 CKO hearts compared to controls. Myocardial Ptc1 expression was unchanged in Smodermo1 CKO hearts (Fig. 7I-J). Similarly, immunofluorescent analysis of VEGF-A, VEGF-B, and VEGF-C protein expression confirmed specific loss of VEGF-A and VEGF-B expression in perivascular cells of Smodermo1 CKO hearts. Myocardial VEGF-A and VEGF-B protein expression was unaffected (Fig. 7K-N). VEGF-C, which is specifically expressed in perivascular cells, could not be detected in the ventricles of Smodermo1 CKO hearts (Fig. 7O-P). Quantitative anaylsis of fluorescent intensity confirmed that, compared to controls, Smodermo1 CKO hearts had statistically significant reductions in perivascular VEGF-A, VEGF-B, and VEGF-C expression (Fig. 7R). In situ hybridization for Vegf-A, Vegf-B and Vegf-C confirmed reductions in perivascular expression of these factors in Smodermo1 CKO hearts (Supp. Fig. 1).
In contrast to Smomlc2v CKO hearts, which demonstrate an absence of subepicardial blood vessels and reduced myocardial VEGF ligand expression, Smodermo1 CKO hearts showed a quantitative decrease in intramyocardial blood vessels despite diminished perivascular VEGF expression. Given that subepicardial and intramyocardial blood vessels express identical VEGFRs (Lavine et al., 2006), these data suggest that myocardial sources of VEGF-A and VEGF-B preferentially promote subepicardial vessel growth, but also can support intramyocardial vessel growth to a lesser degree. However, perivascular sources of VEGF-A, VEGF-B, and VEGF-C specifically control intramyocardial vessel growth, as no subepicardial vessels are present in the ventricles of Smomlc2v CKO hearts (Fig. 4).
Coronary arteries and veins may be derived from distinct vascular lineages
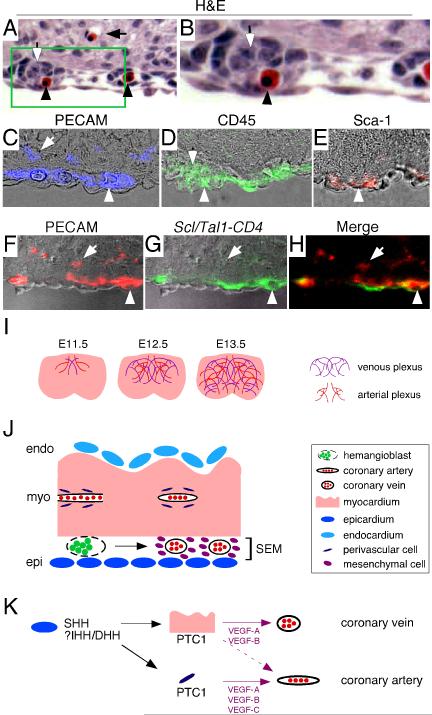
The finding that HH signaling to different tissues differentially controls coronary artery and vein development raises the possibility that coronary arteries and veins may develop via distinct mechanisms and potentially may be derived from different endothelial cell precursors. Consistent with this hypothesis, examination of H&E stained histological sections of E12.5 hearts revealed that while coronary arteries only contained red blood cells, coronary veins contained both red blood cells and rosette-like clusters of undefined cells located within the vascular lumen (Fig. 8A-B). These clusters of cells were reminiscent of hemangioblast precursors present within vascular channels of the yolk sac (Ema and Rossant, 2003).
Figure 8. Coronary arteries and veins represent distinct vascular lineages.
A, H&E stained section of an E12.5 heart demonstrating that coronary veins (black arrowhead) contain both red blood cells and rosettes of undefined cells (open arrow) while coronary arteries (black arrow) only contain red blood cells. B, high magnification of the area enclosed by the box in A. C-E, Immunofluorescent staining of E12.5 hearts demonstrating that while both coronary veins (C, white arrowhead) and coronary arteries (C, white arrow) express PECAM, only coronary veins express CD45 (D, white arrowhead) and Sca-1 (E, white arrow head). Cells located within coronary veins express CD45 (D, open arrow) indicating that they are of hematopoietic origin. Blue-PECAM, Green-CD45, and Red-Sca-1. F-H, Immunofluorescent staining of Scl/Tal1-CD4 knockin E12.5 hearts demonstrating that Scl/Tal1 is expressed in coronary veins (white arrowhead) but not in coronary arteries (white arrow). Red-PECAM, green-CD4 (Scl/Tal1). H, merge of fluorescent signals in F, G. C-H, Immunofluorescent signals are superimposed on DIC images. A and C-H, 400X magnification. I, Schematic depicting that the developing coronary vascular plexus is composed of two distinct subsets of blood vessels, coronary arteries and veins. J, Model describing the origins of the coronary arterial and venous lineages. Coronary veins are derived from hemangioblasts, while coronary arteries are likely derived from endothelial cells. K, Model describing the signaling events that coordinately control coronary artery and vein development. Perivascular HH signaling controls coronary artery growth by regulating perivascular expression of VEGF-A, VEGF-B, and VEGF-C. Myocardial HH signaling controls myocardial VEGF-A and VEGF-B expression, which is required for coronary vein growth, and in combination with perivascular VEGF expression, positively regulates coronary artery growth.
To determine whether hemangioblasts are present within coronary veins, we examined the expression of the hemangioblast markers, CD45, SCA1 (Bailey et al., 2004), and SCL/TAL1 (Chung et al., 2002) in E12.5 hearts. In contrast to PECAM, which marks both coronary veins and arteries, CD45 expression could only be detected in coronary veins (Fig. 8C-D). Consistent with a hematopoietic origin, the clusters of cells located within the lumen of coronary veins also expressed CD45 (Fig. 8D). Furthermore, cells located within coronary veins expressed Sca-1 (Fig. 8E). In addition, the definitive hemangioblast marker, Scl/Tal1, was specifically expressed in coronary veins but not in coronary arteries (Fig. 8F-H). These data demonstrate that hemangioblasts or hemangioblast-like cells reside within coronary veins. Moreover, coronary veins are likely derived from these cells and potentially grow via a vasculogenic mechanism. In contrast, coronary arteries likely grow via a distinct mechanism as they do not express markers of the hemangioblast lineage.
Discussion
HH signaling coordinately regulates coronary artery and vein development
Previously, we hypothesized that HH signaling to the cardiomyoblast and perivascular cell regulates coronary vascular development (Lavine et al., 2006). Through conditional gene targeting, we have now shown that the cardiomyoblast and perivascular cells are the relevant targets of HH signaling. Furthermore, we have demonstrated that myocardial and perivascular HH signaling controls the development of distinct vascular subtypes. HH signaling to the cardiomyoblast is required for coronary vein development and HH signaling to the perivascular cell specifically supports coronary artery development. Moreover, coronary veins likely arise from hemangioblasts, while coronary arteries are likely derived from another cell type, possibly from previously established vasculature (Fig. 8I-K).
The ability of HH ligands to coordinately control the development of coronary arteries and veins by signaling to distinct cell types implies that coronary artery and vein growth can be uncoupled by differentially altering the ability of cardiomyoblasts and perivascular cells to receive HH signaling. While there is no evidence that this occurs during the vascular plexus stage, it may be important during the remodeling stage, as in the adult, coronary arteries and veins are not always located in the same positions and do not always follow the same routes (Icardo and Colvee, 2001).
In addition, the ability of HH signaling to differentially promote the development of distinct vessel types by signaling to different tissues may be important beyond differentially promoting artery and vein growth. It is possible that differential HH signaling may promote distinct patterns of vascular growth. Perivascular HH signaling may promote angiogenic growth, such as vascular sprouting, via localized production of VEGF ligands. In contrast, HH signaling to the cardiomyoblast likely produces a more diffuse pattern of VEGF expression and thus may promote a vasculogenic or vascular plexus pattern of growth. Consistent with this, subepicardial vessels likely develop from hemangioblasts possibly through vasculogenesis.
Coronary arterial and venous lineages are established during the vascular plexus stage
Vascular development is thought to proceed through a stereotyped series of events beginning with the formation of a vascular plexus that is later remodeled, giving rise to the mature vasculature. While much is known about the signaling mechanisms that confer arterial and venous cell fates, it remains unclear when this signaling occurs and when such fates are established.
Studies analyzing spatiotemporal patterns of ephrinB2 and ephB4 expression have produced contrasting results. In the yolk sac, arterial and venous cell fates appear to be established during the vascular plexus stage. However, in the embryonic head region, arterial and venous gene expression is not observed until the remodeling phase (Gerety et al., 1999; Wang et al., 1998).
In the embryonic heart, we have observed that coronary arterial and venous fates are established either during or prior to the vascular plexus stage. Thus, the coronary vascular plexus is not merely a network of capillaries, but rather consists of two distinct and superimposed sets of arterial and venous blood vessels. Interestingly, the spatial relationship between these two sets of blood vessels is conserved in the adult heart, indicating that processes which pattern the vascular plexus may influence the organization of the mature vasculature.
Vascular plexus remodeling
The molecular mechanisms that control vascular remodeling are largely unknown. However, a key observation that has been made is that classical axon guidance factors can influence vascular remodeling and patterning (Carmeliet, 2003). Netrin signaling through the UNC5b receptor acts as a repulsive cue during vascular morphogenesis. Loss of Unc5b leads to ectopic vascular sprouting and subsequent defects in vascular patterning (Lu et al., 2004). Similarly, Plexin signaling through Semaphorin receptors also serves as a repulsive cue, as mutations in plexinD1 result in ectopic vascular sprouting (Torres-Vazquez et al., 2004). Consistent with a role in vascular remodeling, plexinD1−/− and semaphorin3C−/− mice display profound defects in aortic arch remodeling (Gitler et al., 2004). It is of note to mention that Neuropilin (Nrp) is a co-receptor for both Semaphorin and VEGF receptors, and Nrp1 and Nrp2 knockout mice display severe vascular growth and pattern phenotypes (Kawasaki et al., 1999; Takashima et al., 2002; Yuan et al., 2002).
In addition to Netrin and Semaphorin, Ephrins have also been implicated in the control of vascular patterning. The ephrinB2 ligand is expressed on arterial endothelial cells and signals to its receptor, ephB4, which is expressed on venous endothelial cells. Also, similar to Netrin and Semaphorin signaling, Ephrin signaling serves as a repulsive cue. Loss of either ephrinB2 or ephB4 leads to vascular patterning defects affecting both arterial and venous lineages, suggesting that interactions between arterial and venous blood vessels are critical for proper vascular patterning (Gerety et al., 1999; Wang et al., 1998). Consistent with this, deletion of HH signaling in the myocardium (Smomlc2v CKO) not only resulted in loss of coronary veins, but also led to mislocalization of coronary arteries.
Implications for therapeutic neovascularization
Previously, we and others have demonstrated that activation of HH signaling in the adult heart can promote the formation of new coronary vessels (Kusano et al., 2005; Lavine et al., 2006). Furthermore, HH induced neovascularization protected the heart from ischemic insult and preserved cardiac function following myocardial infarction, implicating the HH pathway as a potentially important therapeutic target for treating ischemic heart disease (Kusano et al., 2005). Similar to the mechanism by which HH signaling regulates coronary development, activation of HH signaling promoted blood vessel growth in the adult heart by inducing expression of VEGF and Angiopoietin expression (Kusano et al., 2005; Lavine et al., 2006).
Interestingly, forced expression of Shh in the adult heart led to the growth of multiple vascular types, including capillaries and larger blood vessels. Moreover, the HH receptor and target of signaling, Ptc1, was expressed in both cardiomyocytes and in cells surrounding the vasculature (Kusano et al., 2005). The ability of HH signaling to induce growth of multiple blood vessel types and expression of Ptc1 in several different cell types suggests that, like during coronary development, HH signaling differentially promotes the growth of distinct vascular types by signaling to different tissues.
The potential to selectively trigger the growth of particular blood vessel types would have profound implications for therapeutic intervention. Therapies could be rationally tailored to specific diseases and/or individual patients based on the type of vasculature that would be most efficacious. Further understanding of whether and how HH signaling controls the growth of distinct blood vessel types in the adult heart may provide the first steps toward designing such strategies.
Supplementary Material
Supplemental Figure 1. HH signaling to the myocardium and perivascular cell differentially regulates Vegf ligand expression. A-F, Whole mount in situ hybridization for Vegf-A (A, B), Vegf-B (C, D), and Vegf-C (E, F) showing that compared to controls (A, C, E), Smomlc2v; dermo1 CKO hearts (B, D, F) display decreases to Vegf-A, Vegf-B and Vegf-C expression. G-L, Cryosections of hearts stained with in situ probes Vegf-A (G-I), and Vegf-B (J-L). While control hearts (G, J) contain expression of Vegf-A and Vegf-B in both cardiomyoblasts (asterisk) and perivascular cells (arrowhead), Smomlc2v CKO hearts (H, K) only express these transcripts in perivascular cells (arrowhead). In contrast, Smodermo1 CKO hearts (I, L) only express Vegf-A and Vegf-B in the cardiomyoblast (asterisk). M-P, Whole mount in situ hybridization for Vegf-C demonstrating that compared controls (M), Smomlc2v CKO hearts (N) express similar levels of Vegf-C. Compared to controls (O), Smodermo1 CKO hearts (P) display diminished Vegf-C expression.
Acknowledgements
We thank K. Chien for providing Mlc2v-Cre mice and D. Anderson for ephrinB2-LacZ and ephB4-LacZ mice. This work was initiated with a grant from the American Heart Association and funded by NIH grant HL076664, American Heart Association grant 0415469Z, and a generous contribution from the Virginia Friedhofer Charitable Trust.
References
- Bailey AS, Jiang S, Afentoulis M, Baumann CI, Schroeder DA, Olson SB, Wong MH, Fleming WH. Transplanted adult hematopoietic stems cells differentiate into functional endothelial cells. Blood. 2004;103:13–9. doi: 10.1182/blood-2003-05-1684. [DOI] [PubMed] [Google Scholar]
- Carmeliet P. Blood vessels and nerves: common signals, pathways and diseases. Nat Rev Genet. 2003;4:710–20. doi: 10.1038/nrg1158. [DOI] [PubMed] [Google Scholar]
- Chen J, Kubalak SW, Chien KR. Ventricular muscle-restricted targeting of the RXRalpha gene reveals a non-cell-autonomous requirement in cardiac chamber morphogenesis. Development. 1998a;125:1943–9. doi: 10.1242/dev.125.10.1943. [DOI] [PubMed] [Google Scholar]
- Chen J, Kubalak SW, Minamisawa S, Price RL, Becker KD, Hickey R, Ross J, Jr., Chien KR. Selective requirement of myosin light chain 2v in embryonic heart function. J Biol Chem. 1998b;273:1252–6. doi: 10.1074/jbc.273.2.1252. [DOI] [PubMed] [Google Scholar]
- Chung YS, Zhang WJ, Arentson E, Kingsley PD, Palis J, Choi K. Lineage analysis of the hemangioblast as defined by FLK1 and SCL expression. Development. 2002;129:5511–20. doi: 10.1242/dev.00149. [DOI] [PubMed] [Google Scholar]
- Dettman RW, Denetclaw W, Jr., Ordahl CP, Bristow J. Common epicardial origin of coronary vascular smooth muscle, perivascular fibroblasts, and intermyocardial fibroblasts in the avian heart. Dev Biol. 1998;193:169–81. doi: 10.1006/dbio.1997.8801. [DOI] [PubMed] [Google Scholar]
- Domenga V, Fardoux P, Lacombe P, Monet M, Maciazek J, Krebs LT, Klonjkowski B, Berrou E, Mericskay M, Li Z, et al. Genes Dev. 2004;Notch3 is required for arterial identity and maturation of vascular smooth muscle cells.18:2730–5. doi: 10.1101/gad.308904. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Duarte A, Hirashima M, Benedito R, Trindade A, Diniz P, Bekman E, Costa L, Henrique D, Rossant J. Dosage-sensitive requirement for mouse Dll4 in artery development. Genes Dev. 2004;18:2474–8. doi: 10.1101/gad.1239004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ema M, Rossant J. Cell fate decisions in early blood vessel formation. Trends Cardiovasc Med. 2003;13:254–9. doi: 10.1016/s1050-1738(03)00105-1. [DOI] [PubMed] [Google Scholar]
- Fischer A, Schumacher N, Maier M, Sendtner M, Gessler M. The Notch target genes Hey1 and Hey2 are required for embryonic vascular development. Genes Dev. 2004;18:901–11. doi: 10.1101/gad.291004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Flamme I, Frolich T, Risau W. Molecular mechanisms of vasculogenesis and embryonic angiogenesis. J Cell Physiol. 1997;173:206–10. doi: 10.1002/(SICI)1097-4652(199711)173:2<206::AID-JCP22>3.0.CO;2-C. [DOI] [PubMed] [Google Scholar]
- Gerety SS, Wang HU, Chen ZF, Anderson DJ. Symmetrical mutant phenotypes of the receptor EphB4 and its specific transmembrane ligand ephrin-B2 in cardiovascular development. Mol Cell. 1999;4:403–14. doi: 10.1016/s1097-2765(00)80342-1. [DOI] [PubMed] [Google Scholar]
- Gitler AD, Lu MM, Epstein JA. PlexinD1 and semaphorin signaling are required in endothelial cells for cardiovascular development. Dev Cell. 2004;7:107–16. doi: 10.1016/j.devcel.2004.06.002. [DOI] [PubMed] [Google Scholar]
- Icardo JM, Colvee E. Origin and course of the coronary arteries in normal mice and in iv/iv mice. J Anat. 2001;199:473–82. doi: 10.1046/j.1469-7580.2001.19940473.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kanda S, Mochizuki Y, Suematsu T, Miyata Y, Nomata K, Kanetake H. Sonic hedgehog induces capillary morphogenesis by endothelial cells through phosphoinositide 3-kinase. J Biol Chem. 2003;278:8244–9. doi: 10.1074/jbc.M210635200. [DOI] [PubMed] [Google Scholar]
- Kang Y, Massague J. Epithelial-mesenchymal transitions: twist in development and metastasis. Cell. 2004;118:277–9. doi: 10.1016/j.cell.2004.07.011. [DOI] [PubMed] [Google Scholar]
- Kattan J, Dettman RW, Bristow J. Formation and remodeling of the coronary vascular bed in the embryonic avian heart. Dev Dyn. 2004;230:34–43. doi: 10.1002/dvdy.20022. [DOI] [PubMed] [Google Scholar]
- Kawasaki T, Kitsukawa T, Bekku Y, Matsuda Y, Sanbo M, Yagi T, Fujisawa H. A requirement for neuropilin-1 in embryonic vessel formation. Development. 1999;126:4895–902. doi: 10.1242/dev.126.21.4895. [DOI] [PubMed] [Google Scholar]
- Krebs LT, Xue Y, Norton CR, Shutter JR, Maguire M, Sundberg JP, Gallahan D, Closson V, Kitajewski J, Callahan R, et al. Notch signaling is essential for vascular morphogenesis in mice. Genes Dev. 2000;14:1343–52. [PMC free article] [PubMed] [Google Scholar]
- Kusano KF, Pola R, Murayama T, Curry C, Kawamoto A, Iwakura A, Shintani S, Ii M, Asai J, Tkebuchava T, et al. Sonic hedgehog myocardial gene therapy: tissue repair through transient reconstitution of embryonic signaling. Nat Med. 2005;11:1197–204. doi: 10.1038/nm1313. [DOI] [PubMed] [Google Scholar]
- Lavine KJ, White AC, Park C, Smith CS, Choi K, Long F, Hui C, Ornitz DM. Fibroblast growth factor signals regulate a wave of Hedgehog activation that is essential for coronary vascular development. Genes Dev. 2006;20:1651–1661. doi: 10.1101/gad.1411406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lavine KJ, Yu K, White AC, Zhang X, Smith C, Partanen J, Ornitz DM. Endocardial and epicardial derived FGF signals regulate myocardial proliferation and differentiation in vivo. Dev Cell. 2005;8:85–95. doi: 10.1016/j.devcel.2004.12.002. [DOI] [PubMed] [Google Scholar]
- Lawson ND, Scheer N, Pham VN, Kim CH, Chitnis AB, Campos-Ortega JA, Weinstein BM. Notch signaling is required for arterial-venous differentiation during embryonic vascular development. Development. 2001;128:3675–83. doi: 10.1242/dev.128.19.3675. [DOI] [PubMed] [Google Scholar]
- Lawson ND, Vogel AM, Weinstein BM. sonic hedgehog and vascular endothelial growth factor act upstream of the Notch pathway during arterial endothelial differentiation. Dev Cell. 2002;3:127–36. doi: 10.1016/s1534-5807(02)00198-3. [DOI] [PubMed] [Google Scholar]
- Lawson ND, Weinstein BM. Arteries and veins: making a difference with zebrafish. Nat Rev Genet. 2002;3:674–82. doi: 10.1038/nrg888. [DOI] [PubMed] [Google Scholar]
- Long F, Zhang XM, Karp S, Yang Y, McMahon AP. Genetic manipulation of hedgehog signaling in the endochondral skeleton reveals a direct role in the regulation of chondrocyte proliferation. Development. 2001;128:5099–108. doi: 10.1242/dev.128.24.5099. [DOI] [PubMed] [Google Scholar]
- Lu X, Le Noble F, Yuan L, Jiang Q, De Lafarge B, Sugiyama D, Breant C, Claes F, De Smet F, Thomas JL, et al. The netrin receptor UNC5B mediates guidance events controlling morphogenesis of the vascular system. Nature. 2004;432:179–86. doi: 10.1038/nature03080. [DOI] [PubMed] [Google Scholar]
- Merki E, Zamora M, Raya A, Kawakami Y, Wang J, Zhang X, Burch J, Kubalak SW, Kaliman P, Belmonte JC, et al. Epicardial retinoid X receptor alpha is required for myocardial growth and coronary artery formation. Proc Natl Acad Sci U S A. 2005;102:18455–60. doi: 10.1073/pnas.0504343102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mikawa T, Gourdie RG. Pericardial mesoderm generates a population of coronary smooth muscle cells migrating into the heart along with ingrowth of the epicardial organ. Dev Biol. 1996;174:221–32. doi: 10.1006/dbio.1996.0068. [DOI] [PubMed] [Google Scholar]
- Morabito CJ, Kattan J, Bristow J. Mechanisms of embryonic coronary artery development. Curr Opin Cardiol. 2002;17:235–41. doi: 10.1097/00001573-200205000-00005. [DOI] [PubMed] [Google Scholar]
- Pola R, Ling LE, Silver M, Corbley MJ, Kearney M, Blake Pepinsky R, Shapiro R, Taylor FR, Baker DP, Asahara T, et al. The morphogen Sonic hedgehog is an indirect angiogenic agent upregulating two families of angiogenic growth factors. Nat Med. 2001;7:706–11. doi: 10.1038/89083. [DOI] [PubMed] [Google Scholar]
- Ratajska A, Ciszek B, Sowinska A. Embryonic development of coronary vasculature in rats: corrosion casting studies. Anat Rec A Discov Mol Cell Evol Biol. 2003;270:109–16. doi: 10.1002/ar.a.10011. [DOI] [PubMed] [Google Scholar]
- Risau W. Mechanisms of angiogenesis. Nature. 1997;386:671–4. doi: 10.1038/386671a0. [DOI] [PubMed] [Google Scholar]
- Shin D, Garcia-Cardena G, Hayashi S, Gerety S, Asahara T, Stavrakis G, Isner J, Folkman J, Gimbrone MA, Jr., Anderson DJ. Expression of ephrinB2 identifies a stable genetic difference between arterial and venous vascular smooth muscle as well as endothelial cells, and marks subsets of microvessels at sites of adult neovascularization. Dev Biol. 2001;230:139–50. doi: 10.1006/dbio.2000.9957. [DOI] [PubMed] [Google Scholar]
- Soriano P. Generalized lacZ expression with the ROSA26 Cre reporter strain. Nat Genet. 1999;21:70–1. doi: 10.1038/5007. [DOI] [PubMed] [Google Scholar]
- Takashima S, Kitakaze M, Asakura M, Asanuma H, Sanada S, Tashiro F, Niwa H, Miyazaki Ji J, Hirota S, Kitamura Y, et al. Targeting of both mouse neuropilin-1 and neuropilin-2 genes severely impairs developmental yolk sac and embryonic angiogenesis. Proc Natl Acad Sci U S A. 2002;99:3657–62. doi: 10.1073/pnas.022017899. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Torres-Vazquez J, Gitler AD, Fraser SD, Berk JD, Van NP, Fishman MC, Childs S, Epstein JA, Weinstein BM. Semaphorin-plexin signaling guides patterning of the developing vasculature. Dev Cell. 2004;7:117–23. doi: 10.1016/j.devcel.2004.06.008. [DOI] [PubMed] [Google Scholar]
- Vokes SA, Yatskievych TA, Heimark RL, McMahon J, McMahon AP, Antin PB, Krieg PA. Hedgehog signaling is essential for endothelial tube formation during vasculogenesis. Development. 2004;131:4371–80. doi: 10.1242/dev.01304. [DOI] [PubMed] [Google Scholar]
- Vrancken Peeters MP, Gittenberger-de Groot AC, Mentink MM, Poelmann RE. Smooth muscle cells and fibroblasts of the coronary arteries derive from epithelial mesenchymal transformation of the epicardium. Anat Embryol (Berl) 1999;199:367–78. doi: 10.1007/s004290050235. [DOI] [PubMed] [Google Scholar]
- Wang HU, Chen ZF, Anderson DJ. Molecular distinction and angiogenic interaction between embryonic arteries and veins revealed by ephrin-B2 and its receptor Eph-B4. Cell. 1998;93:741–53. doi: 10.1016/s0092-8674(00)81436-1. [DOI] [PubMed] [Google Scholar]
- Yoshida T, Owens GK. Molecular determinants of vascular smooth muscle cell diversity. Circ Res. 2005;96:280–91. doi: 10.1161/01.RES.0000155951.62152.2e. [DOI] [PubMed] [Google Scholar]
- Yu K, Xu J, Liu Z, Sosic D, Shao J, Olson EN, Towler DA, Ornitz DM. Conditional inactivation of FGF receptor 2 reveals an essential role for FGF signaling in the regulation of osteoblast function and bone growth. Development. 2003;130:3063–74. doi: 10.1242/dev.00491. [DOI] [PubMed] [Google Scholar]
- Yuan L, Moyon D, Pardanaud L, Breant C, Karkkainen MJ, Alitalo K, Eichmann A. Abnormal lymphatic vessel development in neuropilin 2 mutant mice. Development. 2002;129:4797–806. doi: 10.1242/dev.129.20.4797. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplemental Figure 1. HH signaling to the myocardium and perivascular cell differentially regulates Vegf ligand expression. A-F, Whole mount in situ hybridization for Vegf-A (A, B), Vegf-B (C, D), and Vegf-C (E, F) showing that compared to controls (A, C, E), Smomlc2v; dermo1 CKO hearts (B, D, F) display decreases to Vegf-A, Vegf-B and Vegf-C expression. G-L, Cryosections of hearts stained with in situ probes Vegf-A (G-I), and Vegf-B (J-L). While control hearts (G, J) contain expression of Vegf-A and Vegf-B in both cardiomyoblasts (asterisk) and perivascular cells (arrowhead), Smomlc2v CKO hearts (H, K) only express these transcripts in perivascular cells (arrowhead). In contrast, Smodermo1 CKO hearts (I, L) only express Vegf-A and Vegf-B in the cardiomyoblast (asterisk). M-P, Whole mount in situ hybridization for Vegf-C demonstrating that compared controls (M), Smomlc2v CKO hearts (N) express similar levels of Vegf-C. Compared to controls (O), Smodermo1 CKO hearts (P) display diminished Vegf-C expression.