Abstract
The age-specific of occurrence of cervical cancer related to human papillomavirus genotypes HPV16 and HPV18, the two targeted by current HPV vaccines, is not well described. We therefore used data from two large, tissue-based HPV genotyping studies of cervical cancer, one conducted in New Mexico (USA) (n = 744) and an international study restricted to cancers (n = 1,729) from Europe, North America, and Australia to represent those regions with widely available cervical cancer screening facilities. HPV results were categorized as HPV16 or HPV18 positive (HPV16/18) versus other HPV genotype. We observed a decreasing proportion of HPV16/18-positive cancers with increasing age in the international study (ptrend < 0.001) and New Mexico study (ptrend < 0.001). There was no heterogeneity in the relationship between age of diagnosis and the proportion of HPV16/18-positive cancers between studies (p = 0.8). Combining results from the two studies (n = 2,473), the percentages of HPV16/18-positive cases were 77.0% (95%CI: 75.1%-78.9%) for women less than 65 years old and 62.7% (95%CI: 58.4%-66.9%) for women aged 65 and older (p < 0.001). In women who are under the age of 25 and have been vaccinated before becoming sexually active, the cervical cancer incidence is expected to be approximately 3.5 per million by 2020. HPV vaccination against HPV16/18 may have a greater impact on cervical cancers in women under 65 than in women aged 65 and older. These data will inform the age-specific impact of HPV vaccination and its integration with cervical cancer screening activities.
Introduction
Two prophylactic human papillomavirus (HPV) vaccines have been developed, Gardasil™ (Merck, West Point, PA, USA) and Cervarix™ (GSK, Rixensart, Belgium), and both have received U.S. Food-and-Drug Administration approval for women ages 9 to 26 years old. Both vaccines protect against infections by HPV16 and HPV18 (HPV16/18), the two HPV genotypes responsible for causing approximately 70-75% of all cervical cancer (1). Both HPV vaccines have shown better than 90% efficacy for preventing persistent infections and cervical precancerous lesions caused by HPV16/18 in uninfected women for up to 4-6 years with no evidence yet of waning immunity resulting in breakthrough events (2-6); up to 8 years of protection has been observed for a monovalent, research HPV16-only vaccine (7) and 8.4 years for Cervarix™ (8). However, because the current HPV vaccines do not prevent infections by all carcinogenic HPV genotypes, do not treat preexisting infections or related abnormalities caused by HPV16 or 18 (3, 4, 9, 10), and the duration of vaccine protection has not been established, cervical cancer screening will be required for the foreseeable future.
In order to project 1) what degree HPV vaccines will impact age-specific cervical cancer rates and 2) how screening might be affected and therefore may need to be altered to integrate the two interventions in safe and cost-effective manner (i.e., to re-balance the benefits and harms of screening (11)), age-specific data on the occurrence of HPV16/18-related cancer would be very useful. Notably, there is some evidence that HPV16/18-related cervical cancers occur on average at a younger age than cancers due to other HPV genotypes (1, 12). However, detailed, age-specific occurrence of HPV16/18-related cervical cancers has not been described. We therefore combined the data from two large studies, one from New Mexico (12) and one international study (1), to describe the age-specific patterns of HPV16/18-related cervical cancer in western countries, where vaccination and screening will need to be integrated in the near future.
Methods
We used data two large cervical cancer studies in this analysis. Both studies had large sample sizes and used tissue-based typing by well-validated assays conducted by expert labs. The two studies and their methods have been described in detail elsewhere (1, 12) and will only be summarized here. The institution review boards of the participating organizations approved these studies.
New Mexico Study
The New Mexico Surveillance, Epidemiology, and End Results Registry was used to ascertain all cases of invasive cervical cancer (n = 1,429) diagnosed from January 1, 1980, through September 30, 1999 (12). A total of 808 women (57%) had paraffin-embedded tissue blocks available for analysis, of which there were 744 HPV test results. The HPV genotyping of tissues by PGMY09/11 line blot assay (LBA) and SPF10/LiPA25 was described previously (12).
Retrospective International Survey and HPV Time Trends Study Group Study
A retrospective cross-sectional study was designed and coordinated to estimate the prevalence of HPV DNA genotypes in women with invasive cervical cancer during 1949–2009 (1). This present analysis was restricted to the 1,729 cases from Europe, North America, and Australia (1), where cervical cancer screening is generally available. The HPV genotyping of tissues by SPF10/LiPA25 was described previously (1).
Statistical and Analytic Methods
Cancers were classified as positive for HPV16/18 or for other HPV genotypes. We made the a priori assumption that in the context of detecting multiple HPV infections (<10%; (1, 12)), if HPV16 and/or HPV18 were detected, these genotypes were the cause of the cancer. This undoubtedly led to slight over-attribution of cancer to these HPV genotypes (12). Age at diagnosis was categorized as <25 years, by 5 year age groups from 25 to 79, and 80 and older.
We used the following statistical procedures and tests in our analysis: 1) exact 95% confidence intervals (95%CI) were calculated for the overall and age group-specific proportions of HPV16/18-positive cervical cancers; 2) Fisher's exact test was used to compare the overall and age group-specific proportions of HPV16/18-positive cervical cancers between studies; and 3) a non-parametric test for linear trend was used to test for an association of age with the proportion of HPV16/18-positive cervical cancers and of multiple HPV types detected (12).
We projected the impact of HPV vaccination on the incidence of cervical cancer in women under 40 in the U.S. To do this, we used the recently reported incidence rates by age (13), based on data from the National Program of Cancer Registries and the Surveillance, Epidemiology, and End Results Program covering 92% of the U.S. population, and the estimated fraction of vaccine-preventable HPV16/18 cervical cancers by age determined in this analysis. We made the following assumptions: 1) full efficacy for HPV vaccination i.e., young women were vaccinated before sexual initiation; 2) HPV vaccines protected women against the HPV vaccine types until the age at which there would be insufficient time to develop cervical cancer from an incident HPV16/18 infection, approximately 20 years duration; and 3) there is no significant cross-protection against other carcinogenic HPV genotypes (5, 14)(vs. the main effects of HPV16/18 protection). We project the impact of HPV vaccination on the incidence rate of all cervical cancer and cervical cancers of squamous cell carcinoma and adenocarcinoma histology, thereby excluding possible rare, non-HPV related cancers, benign tumors, and misclassified disease.
STATA Version 11.1 (StataCorp, College Station, Texas, USA) and SAS Version 9.2 (SAS Institute, Inc., Cary, NC, USA) were used for statistical tests. A p value of < 0.05 was considered significant.
Results
Table 1 shows the age group-specific occurrence of HPV16/18-positive cervical cancer for the two studies individually and combined. Overall, the proportion of HPV16/18-positive in both studies was similar, 74.8% (95%CI: 72.7%-76.9%) for the international study and 72.5% (95%CI: 69.1%-75.8%) for the New Mexico study (p = 0.24). In both studies, the proportion of HPV16/18-related cervical cancer decreased in older age groups (ptrend< 0.001 for both), from approximately 75%-80% in age groups up to 60-64 years and then approximately 60-65% in age groups 65-69 years and older. Comparing the two studies by age groups, there were only 3 of 13 age groups with statistically significant differences in the proportion of HPV16/18-positive cases (international vs. New Mexico): <25 years (100% vs. 60%, p = 0.02), 55-59 years (81.0% vs. 64.4%, p = 0.01), and 65-69 years (72.5% vs. 44.4%, p < 0.001).
Table 1.
Age group-specific prevalence of human papillomavirus (HPV) type 16 (HPV16) or HPV18 vs. other HPV genotypes in cervical cancers diagnosed in western countries (Europe, North America, and Australia) (1) (A), in New Mexico, USA (12) (B), and combining data from the two studies (C). ptrend, a non-parametric test of trend of the fraction of HPV16 or HPV18 with increasing age; p, Fisher's exact test for age-group differences in the fraction of HPV16 or HPV18 between the two studies (Bold type indicates statistical significance [p<0.05]).
| A. de Sanjose et al., 2010 | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| HPV Status | All ages | <25 | 30-34 | 35-39 | 45-49 | 50-54 | 55-59 | 60-64 | 65-69 | 70-74 | 75-79 | ≥80 | ptrend |
| HPV16 or HPV18 | 1294 | 13 | 91 | 144 | 165 | 152 | 153 | 119 | 110 | 68 | 39 | 36 | <0.001 |
| 100% | 1.0% | 7.0% | 11.1% | 12.7% | 11.7% | 11.8% | 9.2% | 8.5% | 5.3% | 3.0% | 2.8% | ||
| 74.8% | 100% | 83.3% | 82.3% | 72.3% | 73.4% | 81.0% | 72.4% | 72.5% | 60.1% | 61.1% | 63.2% | ||
| Other HPV Genotypes | 435 | 0 | 18 | 31 | 63 | 55 | 36 | 46 | 41 | 45 | 25 | 21 | |
| 100.0% | 0.0% | 4.2% | 7.1% | 14.5% | 12.6% | 8.3% | 10.5% | 9.5% | 10.4% | 5.7% | 4.8% | ||
| 25.2% | 0.0% | 16.7% | 17.7% | 27.7% | 26.6% | 19.0% | 27.6% | 27.5% | 39.9% | 38.9% | 36.8% | ||
| Total | 1729 | 13 | 109 | 175 | 228 | 207 | 189 | 165 | 151 | 113 | 64 | 57 | |
| B. Wheeler et al., 2009 | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| HPV Status | All ages | <25 | 30-34 | 35-39 | 45-49 | 50-54 | 55-59 | 60-64 | 65-69 | 70-74 | 75-79 | ≥80 | ptrend |
| HPV16 or HPV18 | 535 | 9 | 70 | 80 | 59 | 42 | 38 | 51 | 20 | 23 | 18 | 17 | <0.001 |
| 100% | 1.7% | 13.1% | 15.0% | 11.0% | 7.9% | 7.1% | 9.5% | 3.7% | 4.3% | 3.4% | 3.2% | ||
| 71.9% | 60.0% | 78.7% | 75.5% | 78.7% | 79.2% | 64.4% | 77.3% | 44.4% | 59.0% | 62.1% | 56.7% | ||
| Other HPV Genotypes | 209 | 6 | 19 | 26 | 16 | 11 | 21 | 15 | 25 | 16 | 11 | 13 | |
| 100.0% | 2.9% | 9.1% | 12.4% | 7.7% | 5.3% | 10.0% | 7.2% | 12.0% | 7.7% | 5.3% | 6.2% | ||
| 28.1% | 40.0% | 21.3% | 24.5% | 21.3% | 20.8% | 35.6% | 22.7% | 55.6% | 41.0% | 37.9% | 43.3% | ||
| Total | 744 | 15 | 89 | 106 | 75 | 53 | 59 | 66 | 45 | 39 | 29 | 30 | |
| 0.018 | 0.46 | 0.17 | 0.64 | 0.48 | 0.01 | 0.51 | <0.001 | 1.0 | 1.0 | 1.0 | |||
| C. Combined | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| HPV Status | All ages | <25 | 30-34 | 35-39 | 45-49 | 50-54 | 55-59 | 60-64 | 65-69 | 70-74 | 75-79 | ≥80 | ptrend |
| HPV16 or HPV18 | 1829 | 22 | 161 | 224 | 224 | 194 | 191 | 170 | 130 | 91 | 57 | 53 | <0.001 |
| 100% | 1.2% | 8.8% | 12.2% | 12.2% | 10.6% | 10.4% | 9.3% | 7.1% | 5.0% | 3.1% | 2.9% | ||
| 74.0% | 78.6% | 81.2% | 79.7% | 73.9% | 74.6% | 77.1% | 73.8% | 66.1% | 59.8% | 61.4% | 60.9% | ||
| Other HPV Genotypes | 644 | 6 | 37 | 57 | 79 | 66 | 57 | 61 | 66 | 61 | 36 | 34 | |
| 100.0% | 0.9% | 5.8% | 8.9% | 12.3% | 10.2% | 8.8% | 9.4% | 10.3% | 9.5% | 5.6% | 5.3% | ||
| 26.0% | 21.4% | 18.8% | 20.3% | 26.1% | 25.4% | 22.9% | 26.2% | 33.9% | 40.2% | 38.6% | 39.1% | ||
| Total | 2473 | 28 | 198 | 281 | 303 | 260 | 248 | 231 | 196 | 152 | 93 | 87 | |
However, we did not find an overall difference between studies in the age group-specific proportion of HPV16/18-positive cancers (p = 0.8). The percentage of multiple HPV genotypes detected in the international cases was 6.5% and in the New Mexico cases was 8.3% (p = 0.1). There was no trend with age for the International cases (ptrend = 0.8) or New Mexico cases (ptrend = 0.3).
We therefore combined the data across studies for greater power (n = 2,473). The age groups with the highest percentage of HPV16/18-postive cases were 30-34 years (81.2%) and 40-44 years (80.1%). The age groups with the lowest percentage of HPV16/18-positive cases were 70-74 years (59.8%) and 80 years and older (60.9%). The percentage of multiple HPV genotypes detected was 7.0% and there was no trend across age groups (ptrend = 0.3).
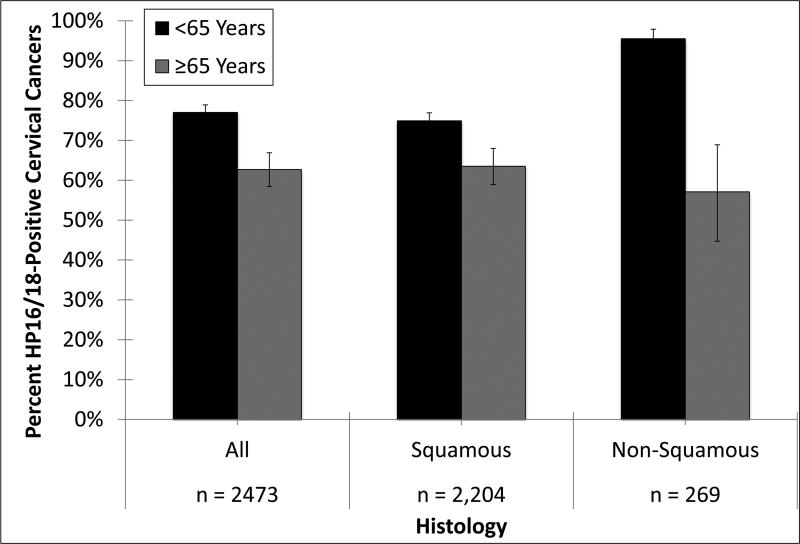
As a post-hoc analysis, we categorized age of diagnosis as less than 65 years and 65 years and older (Figure 1). Percentages of HPV16/18-positive cases were 77.0% (95%CI: 75.1%-78.9%) for women less than 65 years old and 62.7% (95%CI: 58.4%-66.9%) for women aged 65 and older (p < 0.001). The corresponding prevalence ratio of HPV16/18 to other HPV genotypes for less than 65 versus 65 and older was 1.23 (95%CI: 1.15-1.32). Restricted to squamous cell carcinoma histology only (n = 2,204), the percentages of HPV16/18-positive cases were 74.9% (95%CI: 72.8%-76.9%) for women less than 65 years old and 63.5% (95%CI: 58.9%-68.0%) for women aged 65 and older (p < 0.001). The corresponding prevalence ratio was 1.18 (95%CI: 1.09-1.27). Restricted to non-squamous histology (i.e., primarily adenocarcinoma)(n = 269), the percentages of HPV16/18-positive cases were 95.5% (95%CI: 91.6%-97.9%) for women less than 65 years old and 57.1% (95%CI: 44.7%-68.9%) for women aged 65 and older (p < 0.001). The corresponding prevalence ratio was 1.67 (95%CI: 1.36-2.05).
Figure 1.
The proportions of HPV16/18-positive cancers by age (<65 years vs. 65 and older) and histology type, using data from two previous studies of HPV genotyping of cervical cancers (1,12).
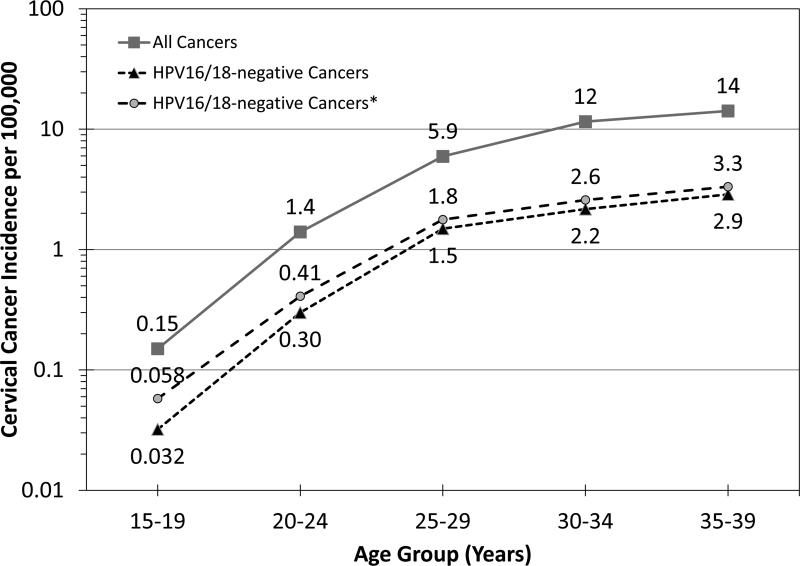
In Figure 2, we projected what the age group-specific profile of cervical cancer in the U.S. in women <40 years of age (13) might be without HPV16/18 in the population. The risks of cervical cancer incidence will decrease from 1.5 per million (0.15 per 100,000) to 0.32-0.58 per million in women aged 15-19 years and from 14 per million to 3.0-4.1 per million in women aged 20-24 years. Similar relative reductions are expected in the older age groups.
Figure 2.
An estimation of the impact of human papillomavirus (HPV) vaccination on the annual incidence of cervical cancer (per 100,000) in U.S., women under the age of 40 years, assuming vaccination before exposure to HPV16 and 18 (HPV16/18) and protective immunity until age 40. The incidence rates were derived based on recently published rates of cervical cancer in the U.S. (15) and the age group-specific fraction of HPV16 /18 cervical cancers (1,12). The rates of HPV16 /18 –negative cancers in women less than 20 years and 20-24 were based on the fraction of non-HPV16/18 cervical cancers in women under the age of 25. * indicates the estimated fraction for HPV16/18-negative cancers assuming no impact of HPV16/18 vaccination on cervical cancers with an unknown histology.
Discussion
We can only anticipate the HPV-vaccine impact based on cervical cancer annual rates and HPV genotype-specific attribution of cervical cancers, or wait many years to observe the changes in cancer rates first in the youngest and then later in older women. We therefore empirically projected the impact of removing HPV16 and HPV18 from the population on the age-specific rates of HPV16/18-related cancers in the western countries, using the best data available from two large case series with high-quality HPV genotyping of tissues blocks.
In the U.S., where HPV vaccination coverage is poor (15), a broad recommendation for changes in cervical cancer screening in HPV-vaccinated women is not currently appropriate (16). However, we suggest, based on the presented data, that when individual vaccination can be verified through electronic medical records or vaccine registries, or if there is high-population coverage under the age 15 (because women are unlikely to have been exposed to HPV since most will not yet be sexually active (17)), it will be rational to delay screening until at least age 25. The benefits of screening in unvaccinated women under the age of 25 are already questionable given the very low incidence of cancer (18) in this age group. Time trends of cervical cancer incidence in the U.S. in women aged 20-24 have only declined by approximately 40% over the last 40 years (18), suggesting that the widespread introduction of screening in women aged <25 years over that period of time had a minor impact on cancer occurrence. Many European Union countries do not initiate screening until the age of 25 or 30 (19, 20). Likewise, Canadian guidelines now recommend against cervical cancer screening in those under 25 years of age (21). The population risks in these western countries are comparable to the U.S., indirectly affirming that there is little cancer prevention benefit in screening these young women, even without vaccination. Finally, there is a lack of evidence that screening in this age group prevents these rapidly developing cancers occurring in these very young women (22).
Based on this analysis, effective HPV vaccination in primarily unexposed women would be projected to ultimately reduce the incidence of cervical cancer in these vaccinated cohorts of young women up to 80%, or 3-4 per million in women aged 20-24 years, a cancer risk that does not typically warrant population-based screening. By comparison, the age-adjusted, all-ages cancer incidences in the U.S. in 2009 (23) were 7.5 per million for vaginal cancer, 23.8 per million for vulvar cancer, and 12.0 per million for male breast cancer, none for which we routinely screen. Likewise, we now do not screen women under the age of 21 years for cervical cancer (23-25) and it may be very reasonable to delay screening until the age of 25 years in those confirmed or highly likely to have been vaccinated before the age of 15 years given the lack of benefit and the real potential for harms from screening women aged 21-24 years.
In addition, the potential harms of screening these young women are great. HPV infections, and associated cytologic abnormalities, are common in this age group. Testing positive can lead to unnecessary clinical follow-up and treatment, which has been linked to an increased risk of negative reproductive outcomes such as preterm delivery (24).
We noted that the effect of age on the proportion of HPV16/18-positive cervical cancer appeared more like a step function with a decrease around age 65. Although the age-specific effect can be rationalized as a function of the overall greater carcinogenicity of HPV16 and HPV18 compared to the other, weaker carcinogenic HPV genotypes, one would anticipate a more gradual effect over a range of age. It is unclear why this shift occurs at age 65 but it was observed in both sets of independently collected data, lending credence to the observation. One possible contributor to this effect is that screening, which typically stops around age 65, may influence the HPV genotype distribution in cancers. Slowly developing tumors caused by infections by other carcinogenic HPV genotypes might develop after screening has stopped while HPV16 and HPV18-related tumors are more aggressive and cause tumors or precancerous lesions that are detected by screening and treated at an earlier age.
We also cannot rule out that the change in the HPV16/18-attributable fraction of cervical cancer at age 65 is due to a cohort effect. We did not have enough cases to do an age-period-cohort analysis to really understand the origins of this effect. Stratification on women born before and after 1930 showed that in both studies cancer cases in women over the age of 65 years of age had a lower fraction of HPV16/18 than those under the age of 65 (data not shown), which may suggest that the year a woman was born does not explain the observed decrease in the HPV16/18-attributable fraction in women aged 65 and older.
The age-specific effects observed in this analysis should be considered in mathematical modeling of the impact of HPV vaccination. There may be a greater impact of HPV16/18 vaccination on the occurrence of cervical cancer in women under the age of 65 than in women aged 65 and older. This translates into the possibility of a greater number of years of life (and quality adjusted life years) saved from HPV vaccination than if the fraction of HPV16/18 was constant with age. Our data also emphasize the importance of vaccination in young women before sexual initiation for maximum population impact. Finally, other changes to screening beyond changing the age of initiation of screening to age 25 might be considered, given the reduction in cancer risk and positive predictive value of screening tests for cervical precancer in the absence of the two most carcinogenic HPV types, to maintain an appropriate balance of benefits and harms of screening. When vaccination against 7 carcinogenic HPV genotypes becomes available, we anticipate that further adjustments in how and when women are screened will be needed.
Acknowledgements
Retrospective International Survey and HPV Time Trends Study Group:
Australia: Suzanne M. Garland, Sepher Tabrizi (The Royal Women's Hospital, The University of Melbourne).
Bosnia Herzegovina: Ermina Iljazovic (University Clinical Center Tuzla BiH).
Croatia: Magdalena Grce, Ivan Sabol (RudjerBoskovic Institutem), Sonja Dzebro, Mara Dominis (Clinical Hospital Merkur).
Czech Republic: Ivo Steiner (Faculty of Medicine and Faculty Hospital, Hradec Kralove), Vladimir Vonka (Institute of Hematology and Blood Transfusion).
France: Christine Clavel (CHU Reims, Laboratoire Pol Bouin, Hôpital Maison Blanche), Massimo Tommasino (International Agency for Research on Cancer).
Greece: Maria Tzardi (Medical School of University of Crete), Theodoros Agorastos (Aristotle University of Thessaloniki).
Italy: Luciano Mariani, Ferdinando Marandino (Regina Elena Cancer Institute).
Poland: Andrzej Marcin Nowakowski (Medical University of Lublin).
Portugal: Eugenia Cruz (Centro Regional de Oncologia Coimbra, InstitutoPortuguês de Oncologia); Manuela Lacerda, Manuel Sobrinho-Simoes (Institute of Molecular Pathology and Immunology of the University of Porto); Ana Felix (Instituto Portugues de Oncologia de Lisboa Francisco Gentil).
Spain: Enrique Lerma (Hospital de la Santa Creu i Sant Pau, Barcelona); Antonio Llombart-Bosch, Morelva Toro (Facultad de Medicina, Universidad de Valencia); Belen Lloveras (Hospital del Mar, Barcelona); Ana Puras, Rosa Guarch (Hospital Virgen del Camino, Pamplona); Miguel Andújar (Complejo Hospitalario Universitario Insular Marteno-Infantil, Las Palmas de Gran Canaria); Jaume Ordi (Hospital Clínic i Provincial de Barcelona); Adela Pelayo (Clínica San Carlos, Madrid); Julio Velasco, Cristina Pérez (Hospital San Agustín, & IUOPA (Oncologic and Universitary Institute of Principality of Asturias)); Maria Alejo (Hospital General de l’Hospitalet de Llobregat, Barcelona); Laia Alemany, F. Xavier Bosch, Vanesa Camón, Xavier Castellsagué, Omar Clavero, Silvia de Sanjosé, Ana Esteban, Nuria Guimerà, Yolanda Florencia, Joellen Klaustermeier, Nubia Muñoz, Bea Quirós, Cristina Rajo, Sara Tous, Marleny Vergara (IDIBELL, Institut Català d’Oncologia-Catalan Institute of Oncology, Hospitalet de Llobregat, Barcelona).
The Netherlands: Chris J.L.M Meijer, Peter J.F Snijders (Vrije Universiteit Medical Center); Wim G.V. Quint, Jean-Paul Brunsveld, Anco C. Molijn, Daan T. Geraets (DDL Diagnostic Laboratory).
United States of America: Esther Oliva (Massachusetts General Hospital); Thomas C. Wright (New York Presbyterian Hospital, Columbia University Medical Center).
Footnotes
Disclosures: Dr. Castle is compensated for his service on a data and safety monitoring board for Merck. He has received HPV tests and testing for research at a reduced or no cost from Qiagen, Roche, Norchip, and MTM. Dr. Castle is a consultant for BD, Cepheid, and GE Healthcare, and has received an honorarium from Roche. Dr. Quint has received funding through DDL Diagnostic Laboratory from GlaxoSmithKline to conduct HPV vaccine studies. Drs. Bosch, De Sanjose, and Alemany Vilches are involved in vaccine trials organized by GlaxoSmithKline and Merck / Sanofi Pasteur MSD, investigators-driven research partially sponsored by GlaxoSmithKline and Merck / Sanofi Pasteur MSD. All have received travel support to attend conferences from GlaxoSmithKline, Sanofi Pasteur MSD or Qiagen. Dr. Bosch has received consulting fees from GSK and Sanofi Pasteur MSD, serves on their speakers’ bureau, and provides expert testimony for GSK. He is serving a guest editor for Vaccine for a series of international reviews. Dr. Bosch is the Editor of HPV Today. Dr. Wheeler has received funding through her institution (the University of New Mexico Health Sciences Center) from GlaxoSmith Kline and Merck and Co., Inc. to conduct HPV vaccine studies and similarly through her institution has received reagents and equipment to perform HPV genotyping from Roche Molecular Systems. Dr. Myers has received compensation for consultation from Merck and GSK. He has received research support from Merck, GSK, and Gen-Probe. He also serves as an advisor for National Breast and Cervical Cancer Early Detection Program. Drs. Castle and Myers served as Steering Committee members on guidelines for the use of molecular testing for cervical cancer screening (Steering Committee).
Reference List
- 1.de SS, Quint WG, Alemany L, et al. Human papillomavirus genotype attribution in invasive cervical cancer: a retrospective cross-sectional worldwide study. Lancet Oncol. 2010;11(11):1048–56. doi: 10.1016/S1470-2045(10)70230-8. [DOI] [PubMed] [Google Scholar]
- 2.Romanowski B, de Borba PC, Naud PS, et al. Sustained efficacy and immunogenicity of the human papillomavirus (HPV)-16/18 AS04-adjuvanted vaccine: analysis of a randomised placebo-controlled trial up to 6.4 years. Lancet. 2009;374(9706):1975–85. doi: 10.1016/S0140-6736(09)61567-1. [DOI] [PubMed] [Google Scholar]
- 3.Quadrivalent vaccine against human papillomavirus to prevent high-grade cervical lesions. N Engl J Med. 2007;356(19):1915–27. doi: 10.1056/NEJMoa061741. [DOI] [PubMed] [Google Scholar]
- 4.Garland SM, Hernandez-Avila M, Wheeler CM, et al. Quadrivalent vaccine against human papillomavirus to prevent anogenital diseases. N Engl J Med. 2007;356(19):1928–43. doi: 10.1056/NEJMoa061760. [DOI] [PubMed] [Google Scholar]
- 5.Paavonen J, Naud P, Salmeron J, et al. Efficacy of human papillomavirus (HPV)-16/18 AS04-adjuvanted vaccine against cervical infection and precancer caused by oncogenic HPV types (PATRICIA): final analysis of a double-blind, randomised study in young women. Lancet. 2009;374(9686):301–14. doi: 10.1016/S0140-6736(09)61248-4. [DOI] [PubMed] [Google Scholar]
- 6.Dillner J, Kjaer SK, Wheeler CM, et al. Four year efficacy of prophylactic human papillomavirus quadrivalent vaccine against low grade cervical, vulvar, and vaginal intraepithelial neoplasia and anogenital warts: randomised controlled trial. BMJ. 2010;341:c3493. doi: 10.1136/bmj.c3493. doi: 10.1136/bmj.c3493.:c3493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Rowhani-Rahbar A, Mao C, Hughes JP, et al. Longer term efficacy of a prophylactic monovalent human papillomavirus type 16 vaccine. Vaccine. 2009;27(41):5612–9. doi: 10.1016/j.vaccine.2009.07.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Roteli-Martins CM, Naud P, De BP, et al. Sustained immunogenicity and efficacy of the HPV-16/18 AS04-adjuvanted vaccine: up to 8.4 years of follow-up. Hum Vaccin Immunother. 2012;8(3):390–7. doi: 10.4161/hv.18865. [DOI] [PubMed] [Google Scholar]
- 9.Hildesheim A, Herrero R, Wacholder S, et al. Effect of human papillomavirus 16/18 L1 viruslike particle vaccine among young women with preexisting infection: a randomized trial. JAMA. 2007;298(7):743–53. doi: 10.1001/jama.298.7.743. [DOI] [PubMed] [Google Scholar]
- 10.Haupt RM, Wheeler CM, Brown DR, et al. Impact of an HPV6/11/16/18 L1 virus-like particle vaccine on progression to cervical intraepithelial neoplasia in seropositive women with HPV16/18 infection. Int J Cancer. 2011;129(11):2632–42. doi: 10.1002/ijc.25940. [DOI] [PubMed] [Google Scholar]
- 11.Saslow D, Solomon D, Lawson HW, et al. American Cancer Society, American Society for Colposcopy and Cervical Pathology, and American Society for Clinical Pathology screening guidelines for the prevention and early detection of cervical cancer. CA Cancer J Clin. 2012;62(3):147–72. doi: 10.3322/caac.21139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Wheeler CM, Hunt WC, Joste NE, et al. Human papillomavirus genotype distributions: implications for vaccination and cancer screening in the United States. J Natl Cancer Inst. 2009;101(7):475–87. doi: 10.1093/jnci/djn510. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Benard VB, Watson M, Castle PE, Saraiya M. Cervical carcinoma rates among young females in the United States. Obstet Gynecol. 2012;120(5):1117–23. doi: 10.1097/aog.0b013e31826e4609. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Wheeler CM, Kjaer SK, Sigurdsson K, et al. The impact of quadrivalent human papillomavirus (HPV; types 6, 11, 16, and 18) L1 virus-like particle vaccine on infection and disease due to oncogenic nonvaccine HPV types in sexually active women aged 16-26 years. J Infect Dis. 2009;199(7):936–44. doi: 10.1086/597309. [DOI] [PubMed] [Google Scholar]
- 15.National and state vaccination coverage among adolescents aged 13 through 17 years --- United States, 2010. MMWR Morb Mortal Wkly Rep. 2011;60:1117–23. 1117–23. [PubMed] [Google Scholar]
- 16.Saslow D, Solomon D, Lawson HW, et al. American Cancer Society, American Society for Colposcopy and Cervical Pathology, and American Society for Clinical Pathology screening guidelines for the prevention and early detection of cervical cancer. CA Cancer J Clin. 2012;62(3):147–72. doi: 10.3322/caac.21139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Mosher WD, Chandra A, Jones J. Sexual behavior and selected health measures: men and women 15-44 years of age, United States, 2002. Adv Data. 2005;(362):1–55. [PubMed] [Google Scholar]
- 18.Castle PE, Carreon JD. Practice improvement in cervical screening and management: symposium on management of cervical abnormalities in adolescents and young women. J Low Genit Tract Dis. 2010;14(3):238–340. doi: 10.1097/LGT.0b013e3181d95ad6. [DOI] [PubMed] [Google Scholar]
- 19.Arbyn M, Anttila A, Jordan J, et al. European Guidelines for Quality Assurance in Cervical Cancer Screening. Second edition--summary document. Ann Oncol. 2010;21(3):448–58. doi: 10.1093/annonc/mdp471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Anttila A, Ronco G. Description of the national situation of cervical cancer screening in the member states of the European Union. Eur J Cancer. 2009;45(15):2685–708. doi: 10.1016/j.ejca.2009.07.017. [DOI] [PubMed] [Google Scholar]
- 21.Recommendations on screening for cervical cancer. CMAJ. 2013;185(1):35–45. doi: 10.1503/cmaj.121505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Sasieni P, Castanon A, Cuzick J. Effectiveness of cervical screening with age: population based case-control study of prospectively recorded data. BMJ. 2009;339:b2968. doi: 10.1136/bmj.b2968. doi: 10.1136/bmj.b2968.:b2968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Surveillance, Epidemiology, and End Results (SEER) Program (www.seer.cancer.gov) SEER*Stat Database: Incidence - SEER 9 Regs Research Data, Nov 2011 Sub (1973-2009) <Katrina/Rita Population Adjustment> - Linked To County Attributes - Total U.S., 1969-2010 Counties, National Cancer Institute, DCCPS, Surveillance Research Program, Surveillance Systems Branch, released April 2012, based on the November 2011 submission. 2009.
- 24.Arbyn M, Kyrgiou M, Simoens C, et al. Perinatal mortality and other severe adverse pregnancy outcomes associated with treatment of cervical intraepithelial neoplasia: meta-analysis. BMJ. 2008;337:a1284. doi: 10.1136/bmj.a1284. doi: 10.1136/bmj.a1284.:a1284. [DOI] [PMC free article] [PubMed] [Google Scholar]