Abstract
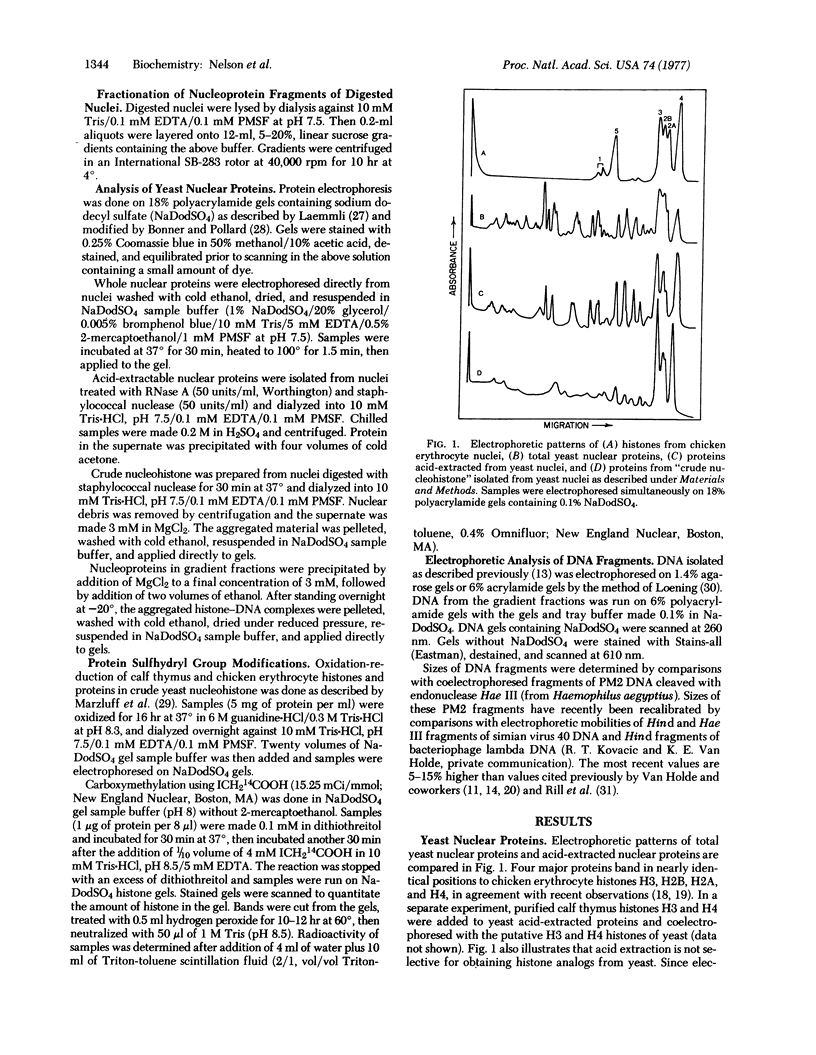
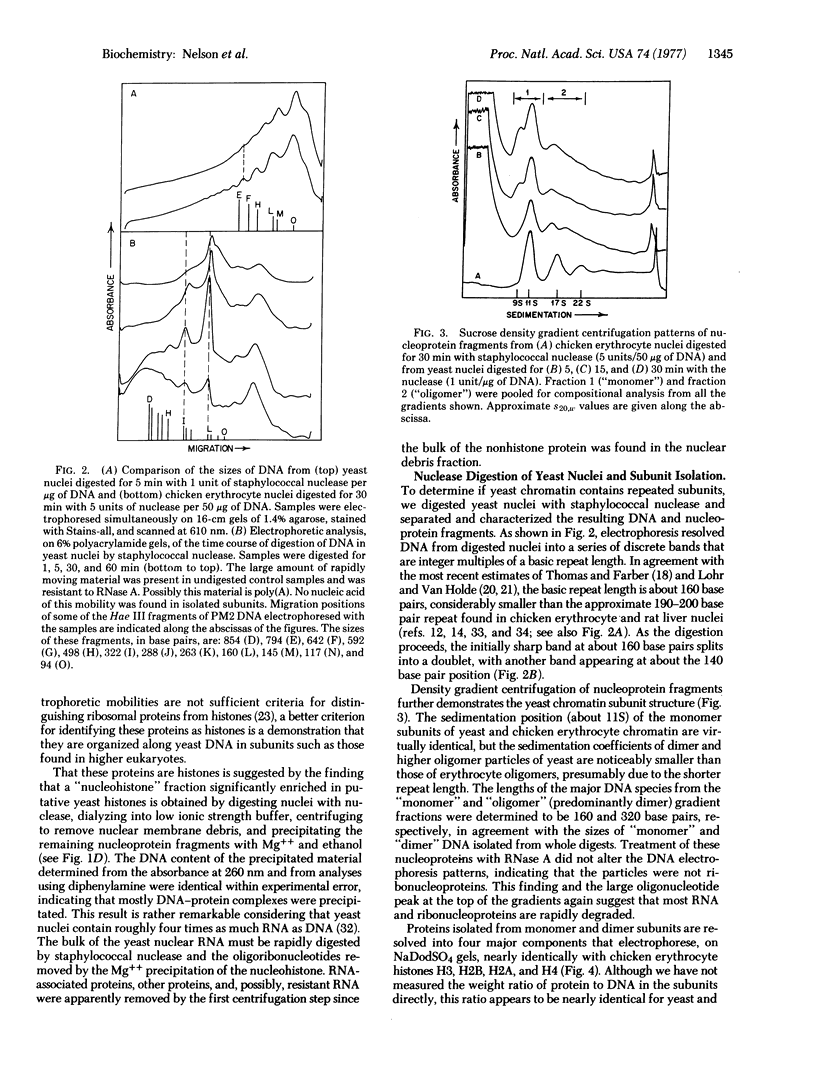
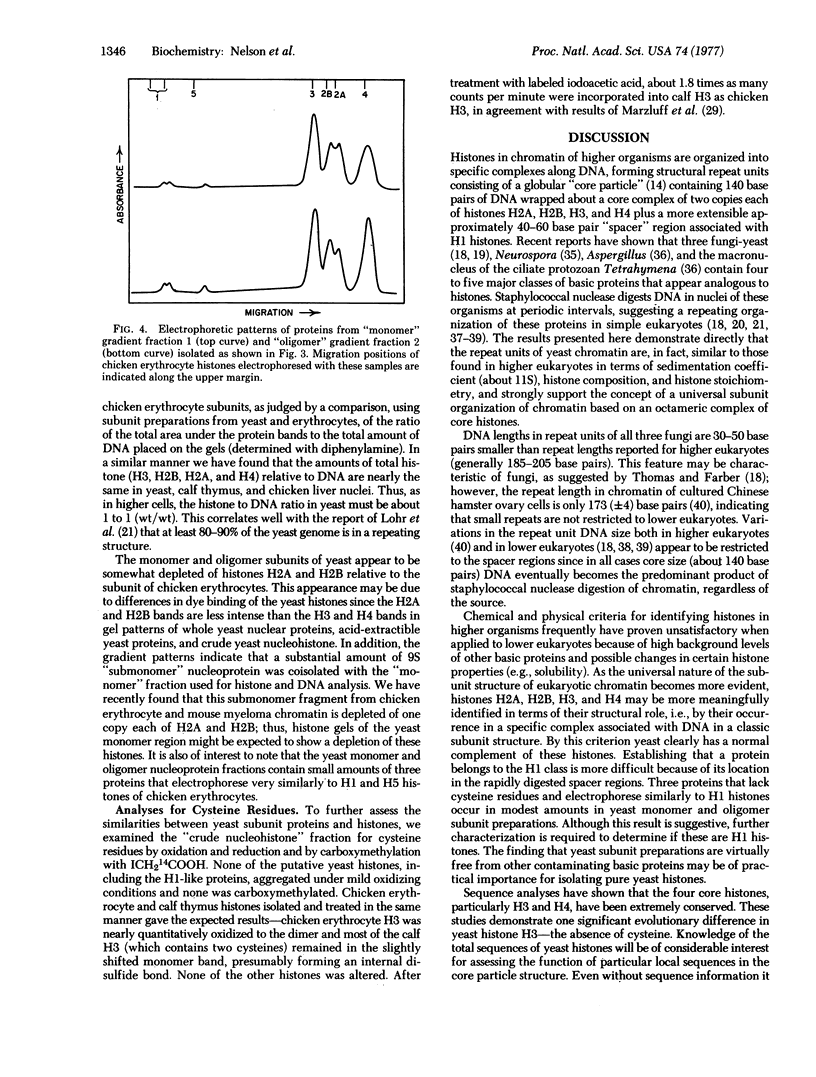
The organization of proteins along DNA in chromatin of Saccharomyces cerevisiae (baker's yeast) was examined by analyzing the DNA and nucleoprotein products obtained after digestion of yeast nuclei with staphylococcal nuclease. Yeast DNA is digested in situ at regularly spaced cleavage sites about 160 base pairs apart. Nucleoprotein fragments were resolved and isolated by centrifugation on linear, 5-20% sucrose gradients. The predominant 11S component appears to be identical to chromatin "subunits" or "nucleosomes" isolated from higher eukaryotes, containing a 150-160 base pair length of DNA and approximately equimolar amounts of four proteins that coelectrophorese with calf histones H2A, H2B, H3, and H4, plus small amounts of three proteins that electrophorese similarly to H1 histones. Thus, the structural organization of the yeast genome is similar to that of more developed organisms, except for the smaller total repeat length. None of the yeast subunit proteins, including the possible H1 proteins, contains cysteine.
Full text
PDF

Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Axel R. Cleavage of DNA in nuclei and chromatin with staphylococcal nuclease. Biochemistry. 1975 Jul;14(13):2921–2925. doi: 10.1021/bi00684a020. [DOI] [PubMed] [Google Scholar]
- Blobel G., Potter V. R. Nuclei from rat liver: isolation method that combines purity with high yield. Science. 1966 Dec 30;154(3757):1662–1665. doi: 10.1126/science.154.3757.1662. [DOI] [PubMed] [Google Scholar]
- Bonner W. M., Pollard H. B. The presence of F3-F2a1 dimers and F1 oligomers in chromatin. Biochem Biophys Res Commun. 1975 May 5;64(1):282–288. doi: 10.1016/0006-291x(75)90250-8. [DOI] [PubMed] [Google Scholar]
- De Kloet S. R., Andrean B. A. Methylated nucleosides in polyadenylate-containing yeast messenger ribonucleic acid. Biochim Biophys Acta. 1976 Apr 2;425(4):401–408. doi: 10.1016/0005-2787(76)90004-6. [DOI] [PubMed] [Google Scholar]
- De Klt S. R. The formation of ribonucleic acid in yeast: hybridization of high molecular weight RNA species to yeast DNA. Arch Biochem Biophys. 1970 Feb;136(2):402–412. doi: 10.1016/0003-9861(70)90211-0. [DOI] [PubMed] [Google Scholar]
- Felden R. A., Sanders M. M., Morris N. R. Presence of histones in Aspergillus nidulans. J Cell Biol. 1976 Mar;68(3):430–439. doi: 10.1083/jcb.68.3.430. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Franco L., Johns E. W., Navlet J. M. Histones from baker's yeast. Isolation and fractionation. Eur J Biochem. 1974 Jun 1;45(1):83–89. doi: 10.1111/j.1432-1033.1974.tb03532.x. [DOI] [PubMed] [Google Scholar]
- Goff C. G. Histones of Neurospora crassa. J Biol Chem. 1976 Jul 10;251(13):4131–4138. [PubMed] [Google Scholar]
- Gorovsky M. A., Keevert J. B. Subunit structure of a naturally occurring chromatin lacking histones F1 and F3. Proc Natl Acad Sci U S A. 1975 Sep;72(9):3536–3540. doi: 10.1073/pnas.72.9.3536. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kornberg R. D. Chromatin structure: a repeating unit of histones and DNA. Science. 1974 May 24;184(4139):868–871. doi: 10.1126/science.184.4139.868. [DOI] [PubMed] [Google Scholar]
- Leighton T., Leighton F., Dill B., Stock J. The similarities of ribosomal and basic chromosomal proteins from fungi. Biochim Biophys Acta. 1976 May 19;432(3):381–394. doi: 10.1016/0005-2787(76)90148-9. [DOI] [PubMed] [Google Scholar]
- Loening U. E. The fractionation of high-molecular-weight ribonucleic acid by polyacrylamide-gel electrophoresis. Biochem J. 1967 Jan;102(1):251–257. doi: 10.1042/bj1020251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lohr D., Kovacic R. T., Van Holde K. E. Quantitative analysis of the digestion of yeast chromatin by staphylococcal nuclease. Biochemistry. 1977 Feb 8;16(3):463–471. doi: 10.1021/bi00622a020. [DOI] [PubMed] [Google Scholar]
- Lohr D., Van Holde K. E. Yeast chromatin subunit structure. Science. 1975 Apr 11;188(4184):165–166. doi: 10.1126/science.1090006. [DOI] [PubMed] [Google Scholar]
- Lusby E. W., Jr, de Kloet S. R. The heavy DNA satellite of yeast and its relationship to the ribosomal RNA genes. Arch Biochem Biophys. 1976 May;174(1):187–191. doi: 10.1016/0003-9861(76)90337-4. [DOI] [PubMed] [Google Scholar]
- Marzluff W. F., Jr, Sanders L. A., Miller D. M., McCarty K. S. Two chemically and metabolically distinct forms of calf thymus histone F3. J Biol Chem. 1972 Apr 10;247(7):2026–2033. [PubMed] [Google Scholar]
- McLaughlin C. S., Warner J. R., Edmonds M., Nakazato H., Vaughan M. H. Polyadenylic acid sequences in yeast messenger ribonucleic acid. J Biol Chem. 1973 Feb 25;248(4):1466–1471. [PubMed] [Google Scholar]
- Moll R., Wintersberger E. Synthesis of yeast histones in the cell cycle. Proc Natl Acad Sci U S A. 1976 Jun;73(6):1863–1867. doi: 10.1073/pnas.73.6.1863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morris N. R. Nucleosome structure in Aspergillus nidulans. Cell. 1976 Jul;8(3):357–363. doi: 10.1016/0092-8674(76)90147-1. [DOI] [PubMed] [Google Scholar]
- Noll M. Differences and similarities in chromatin structure of Neurospora crassa and higher eucaryotes. Cell. 1976 Jul;8(3):349–355. doi: 10.1016/0092-8674(76)90146-x. [DOI] [PubMed] [Google Scholar]
- Noll M. Subunit structure of chromatin. Nature. 1974 Sep 20;251(5472):249–251. doi: 10.1038/251249a0. [DOI] [PubMed] [Google Scholar]
- Olins A. L., Olins D. E. Spheroid chromatin units (v bodies). Science. 1974 Jan 25;183(4122):330–332. doi: 10.1126/science.183.4122.330. [DOI] [PubMed] [Google Scholar]
- Once more into the breach. Nature. 1970 Jul 4;227(5253):1–1. doi: 10.1038/227001a0. [DOI] [PubMed] [Google Scholar]
- Oosterhof D. K., Hozier J. C., Rill R. L. Nucleas action on chromatin: evidence for discrete, repeated nucleoprotein units along chromatin fibrils. Proc Natl Acad Sci U S A. 1975 Feb;72(2):633–637. doi: 10.1073/pnas.72.2.633. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pardon J. F., Worcester D. L., Wooley J. C., Tatchell K., Van Holde K. E., Richards B. M. Low-angle neutron scattering from chromatin subunit particles. Nucleic Acids Res. 1975 Nov;2(11):2163–2176. doi: 10.1093/nar/2.11.2163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Retèl J., Planta R. J. On the mechanism of the biosynthesis of ribosomal RNA in yeast. Biochim Biophys Acta. 1970 Dec 14;224(2):458–469. doi: 10.1016/0005-2787(70)90578-2. [DOI] [PubMed] [Google Scholar]
- Rill R. L., Oosterhof D. K., Hozier J. C., Nelson D. A. Heterogeneity of chromatin fragments produced by micrococcal nuclease action. Nucleic Acids Res. 1975 Sep;2(9):1525–1538. doi: 10.1093/nar/2.9.1525. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schultz L. D., Hall B. D. Transcription in yeast: alpha-amanitin sensitivity and other properties which distinguish between RNA polymerases I and III. Proc Natl Acad Sci U S A. 1976 Apr;73(4):1029–1033. doi: 10.1073/pnas.73.4.1029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shaw B. R., Herman T. M., Kovacic R. T., Beaudreau G. S., Van Holde K. E. Analysis of subunit organization in chicken erythrocyte chromatin. Proc Natl Acad Sci U S A. 1976 Feb;73(2):505–509. doi: 10.1073/pnas.73.2.505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sollner-Webb B., Felsenfeld G. A comparison of the digestion of nuclei and chromatin by staphylococcal nuclease. Biochemistry. 1975 Jul;14(13):2915–2920. doi: 10.1021/bi00684a019. [DOI] [PubMed] [Google Scholar]
- Thomas J. O., Furber V. Yeast chromatin structure. FEBS Lett. 1976 Jul 15;66(2):274–280. doi: 10.1016/0014-5793(76)80521-2. [DOI] [PubMed] [Google Scholar]
- Varshavsky A. J., Bakayev V. V., Georgiev G. P. Heterogeneity of chromatin subunits in vitro and location of histone H1. Nucleic Acids Res. 1976 Feb;3(2):477–492. doi: 10.1093/nar/3.2.477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Whitlock J. P., Jr, Simpson R. T. Removal of histone H1 exposes a fifty base pair DNA segment between nucleosomes. Biochemistry. 1976 Jul 27;15(15):3307–3314. doi: 10.1021/bi00660a022. [DOI] [PubMed] [Google Scholar]
- Wintersberger U., Smith P., Letnansky K. Yeast chromatin. Preparation from isolated nuclei, histone composition and transcription capacity. Eur J Biochem. 1973 Feb 15;33(1):123–130. doi: 10.1111/j.1432-1033.1973.tb02663.x. [DOI] [PubMed] [Google Scholar]
- de KLOET S., van WERMESKERKEN R., KONINGSBERGER V. V. Studies on protein synthesis by protoplasts of Saccharomyces carlsbergensis. I. The effect of ribonuclease on protein synthesis. Biochim Biophys Acta. 1961 Feb 12;47:138–143. doi: 10.1016/0006-3002(61)90838-1. [DOI] [PubMed] [Google Scholar]



