Abstract
Purpose
To evaluate the growing skeleton for potential altered skeletalgenesis associated with antiangiogenesis therapy.
Patients and Methods
Knee radiographs and magnetic resonance imaging (MRI) were prospectively obtained on patients enrolled on two consecutive clinical trials using vandetanib, a potent oral (VEGF receptor 2) VEGFR-2 inhibitor alone or combined with dasatinib, a multiple tyrosine kinase inhibitor, in children with newly diagnosed diffuse intrinsic pontine glioma (DIPG).
Results
Fifty-nine patients (32 females) underwent 119 MRIs; 51 patients underwent 89 radiographs of the knees. The median age at enrollment was 6.2 years (range, 2.4–17.6 years). The dose of vandetanib ranged from 50 to 145 mg/m2/day. The median treatment duration was 205 days. Only two patients have not experienced disease progression after 18 and 60 months from diagnosis. MRI identified clinically significant premature physeal fusion in both knees of one patient, focal physeal thickening in one, osteonecrosis in eight patients (present at enrollment in one), and bony spicules crossing the physis in two patients (bilateral in one). MRI follow-up period averaged 5.3 months (range, 0–25.5 months; median, 3.5 months). Radiographs delineated normally fused physes in two patients but no cases of premature physeal fusion, osteonecrosis or bony spicules.
Conclusions
As MRI provided greater information than radiographs, and thus would be a more sensitive test to assess skeletalgenesis in pediatric patients.
Keywords: antiangiogenesis agents, chemotherapy, magnetic resonance imaging, pediatric brain tumors, skeletalgenesis, VEGF
INTRODUCTION
Angiogenesis is a critical process in tumorigenesis. Inhibitors of angiogenesis, particularly those targeting the vascular endothelial growth factor (VEGF) pathway, have demonstrated promising results in some adult [1–3] cancers. Several of these VEGF-targeting agents have been incorporated into pediatric clinical trials based on the key role of angiogenesis in different childhood cancers [4,5].
Diffuse intrinsic pontine glioma (DIPG) is the most lethal brain cancer in childhood with long-term survival of less than 10% [6]. Radiotherapy is the only standard treatment for children with DIPG as no chemotherapy regimen has shown benefit for affected children. Histologically, glioblastoma accounts for most DIPGs [7]. Angiogenesis is a hallmark of glioblastoma [8]. Therefore, we conducted two consecutive Phase I clinical trials using a potent antiangiogenic agent in combination with local radiotherapy in children with newly diagnosed DIPG. In the first study, vandetanib (AstraZeneca, Macclesfield, United Kingdom), an oral inhibitor of VEGF receptor 2 (VEGFR-2), EGFR, and RET, was administered during and after radiotherapy [9]. In the second study, patients received the combination of vandetanib and dasatinib, an inhibitor of platelet-derived growth factor receptor (PDGFR), Src, and c-kit during and after radiotherapy [10].
Treatment of children with cancer is particularly challenging due to the developmental and maturational changes ongoing during therapy. Preclinical studies have shown antiangiogenic agents to alter skeletalgenesis by blocking normal angiogenesis which is critical for endochondral ossification and longitudinal growth of long bones [11–13]. These results cause concern regarding potential toxicities in growing children yet, clinical reports of skeletal changes related to therapy are limited in children and young adults [14,15].
We included prospective radiographs and magnetic resonance imaging (MRI) of the knees in both clinical trials containing aVEGFR-2 inhibitor to assess for potential altered skeletalgenesis. Based upon our results, we recommend judicious MRI evaluation of skeletal changes in future studies using antiangiogenic therapy in children.
MATERIALS AND METHODS
Eligible patients for both studies (SJBG07 and SJBG09) had a newly diagnosed non-metastatic DIPG or other brainstem high-grade gliomas. Other eligibility criteria for both clinical trials were very similar and followed the standard for pediatric Phase I studies [9,10]. The age cutoff was between 2 and 21 years for SJBG07 and between 18 months and 21 years for SJBG09.
Patients received local radiotherapy (54 Gy) in both studies. In the first study (SJBG07), patients started vandetanib concurrently with radiotherapy and went on to receive this medication for a maximum of 2 years unless there was disease progression or unacceptable toxicity. In the second study, dasatinib was started concurrently with radiotherapy. Vandetanib was started on day 9 of therapy. Treatment with both medications lasted for up to 2 years unless there was disease progression or unacceptable toxicity: The doses of vandetanib and dasatinib ranged from 50 to 145 mg/m2/day and 50 to 85 mg/m2 twice a day, respectively.
We prospectively assessed physeal development in study participants by acquiring serial MRI on either 1.5 T (AVANTO; intermediate field strength) or 3 T (TRIO, Skyra; high field strength) units (Siemens Healthcare, Malvern, PA). Sequences obtained were: sagittal T1 tse 3 mm 160 field of view, coronal proton density PD tse 3 mm 160 field of view, T2 DESS with sagittal 3D reconstruction, and coronal short tau inversion recovery (STIR). An antero-posterior radiograph of the knees also was performed at the time of the MRI.
Two experienced pediatric radiologists provided consensus review and coding of all radiographs and MRIs. Data recorded during imaging reviews included the status of physeal development according to the grading system developed by Laor et al. [16] (Table I), presence or absence of premature physeal fusion, cartilaginous canals or bony spicules, osteonecrosis and its distribution (epiphyseal, metaphyseal, and diaphyseal), growth recovery lines, joint effusion, and non-ossifying fibroma. Any change in these findings on follow-up studies was recorded. Radiographs were assessed for the same findings as the MRIs.
TABLE I.
Laor Classification of Physeal Patency as Defined by MR*
| Category 1: open physis and visible metaphyseal stripe |
| Category 2: partially open physis and visible metaphyseal stripe |
| Category 3: fused physis and visible metaphyseal stripe |
| Category 4: fused physis and undetectable metaphyseal stripe |
Ref. [16].
The study was approved by the Institutional Review Board. Signed informed consent was obtained from parents or guardians and as appropriate, assent was obtained from the patients. All data were managed according to the Health Insurance Portability and Accountability Act (HIPAA) of 1996.
RESULTS
Between June 2007 and April 2010, 60 patients were enrolled on these two studies; 33 were girls. One patient was on-study for only 3 days and thus is excluded from further discussion and analysis leaving a study cohort of 59 (32 girls). The median age at enrollment was 6.4 years (range, 2.1–17.2 years). The median treatment duration was 205 days. During the first 6 weeks of therapy, all but three patients also received dexamethasone in doses ranging from 2 to 16 mg/m2 for a median of 14 days (range, 2–98 days). Only two patients have not experienced disease progression after 18 and 60 months from diagnosis.
Radiographic Findings
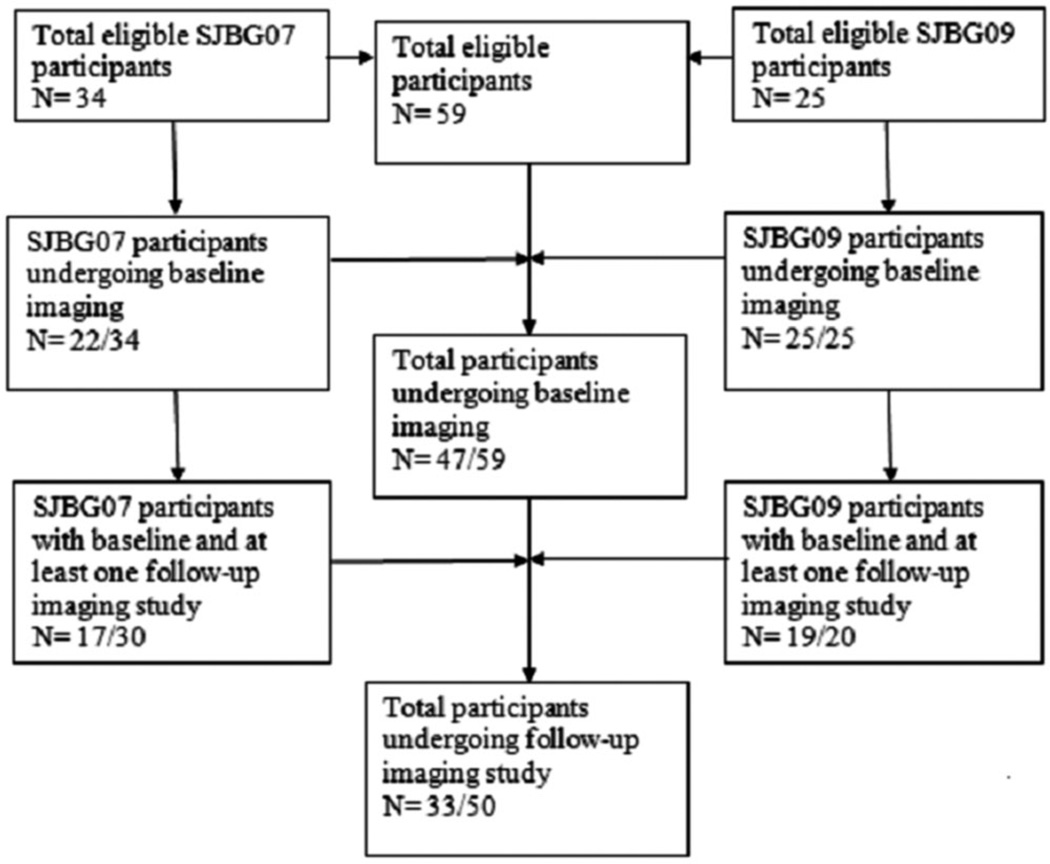
At least one antero-posterior radiograph of the knees was obtained in 51 of the 59 patients enrolled (Fig. 1); a total of 89 radiographs were obtained. Forty-seven patients underwent at least baseline radiographs, of whom 33 also underwent follow-up studies; four patients underwent follow-up studies without having baseline radiographs obtained. Average length of radiographic follow-up was 3.2 months (range: 0–13 months; median, 3.5 months).
Fig. 1.
Patient cohort for radiographic and magnetic resonance (XR/MRI) imaging of the knees.
Physes were open in all but two patients (ages 15 and 16 years) (Table II) and did not change during the study period regardless of patients’ ages. Growth recovery lines, indicative of longitudinal growth, were seen in 13 patients. We found no radiographic evidence of premature physeal fusion, physeal widening or osteonecrosis in any patient. Seven patients were found to have a non-ossifying fibroma, all of which were present at baseline and persisted at follow-up. One patient was assessed as having increased joint fluid which was noted at 8-month follow-up. Subjective bony demineralization was found in 17, in eight of whom, demineralization developed over time.
TABLE II.
Imaging Findings Identified by in Study Patients by Radiographs Compared to Those Detected by MR
| XR | MRI | |
|---|---|---|
| Imaging finding (patients) | (51) | (59) |
| Physeal status: open/closed | 49/2 | 55/4 |
| Premature physeal fusiona | 0 | 1 |
| Osteonecrosisa | 0 | 8 |
| Non-ossifying fibroma | 7 | 11 |
| Growth lines | 13 | 48 |
| Increased joint fluid | 1 | 7 |
| Demineralization | 17 | 0 |
| Bony spicules crossing physisa | 0 | 2 |
| Cartilaginous canals | 0 | 19 |
| Focal physeal thickeninga | 0 | 1 |
| Synovial proliferation | 0 | 2 |
Findings that are of potential clinical importance in monitoring for dyskeletogenesis.
MRI Findings
On 59 patients, we performed a total of 119 MRI studies (Fig. 1). All 59 underwent at least baseline MRI (20 were of both knees) and 50 underwent at least one follow-up MRI between weeks 16 and 19 of therapy; six patients completed three MRIs; three patients completed four MRIs. Ninety-one studies were performed on a 3.0 T unit and 28 studies on a 1.5 T unit; MRI field strength was not known for one study. The average length of MRI follow-up was 5.3 months (range: 0–25.5 months; median, 3.5 months). Nine patients had no MRI follow-up after initial imaging.
By MRI, we found no progression in the state of physeal maturity between the baseline and last follow-up study amongst these patients. Most patients had open, Laor categories 1 and 2 physes (Table II). Physes were classified as having Laor category 1 in 54 patients. One patient was classified as category 2, three as category 3, one patient as category 3 in the femur and category 4 in the tibia. Growth recovery lines were present at some imaging time point in all but 12 patients imaged.
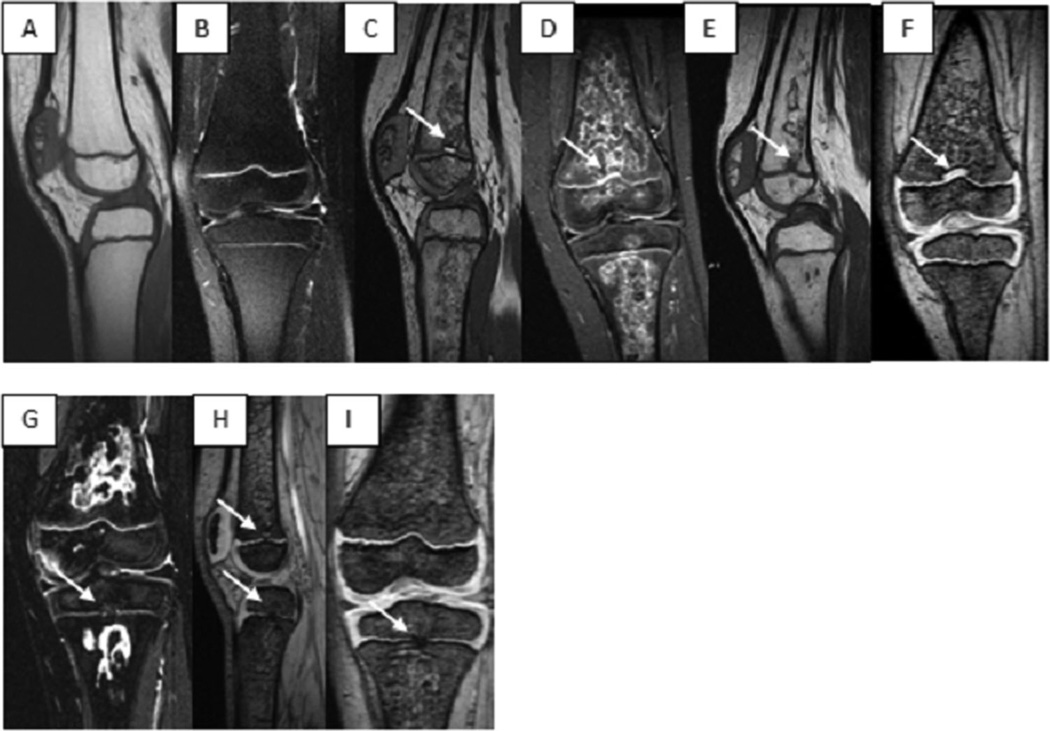
Additional MRI findings included development of cartilaginous canals extending into the metaphyses in 19 patients and resolution of cartilaginous canal in one. Tiny cartilaginous canals were found to cross the physis in two patients. Bony spicules crossing the physis developed in two patients (bilaterally in one). One patient developed a small area of premature fusion of the cartilaginous growth plate (approximately 2% of total area) demonstrated by MRI preceded by focal physeal thickening and in concert with an unusual lacey pattern of presumed osteonecrosis (Fig. 2). This patient had received vandetanib alone. In two patients, a subjective decrease in physeal signal intensity was noted at follow-up imaging.
Fig. 2.
Six-year-old female who developed extensive ostenecrosis and focal physeal fusion while on therapy. This patient had received 10 mg/m2 dexamethasone and 50 mg/m2/day vandetanib. A: Normal baseline appearance of the knee on sagittal T1- and (B) coronal STIR. Three-month follow-up shows extensive diffuse intramedullary lacey pattern on the sagittal (C) and coronal (D) images outlined with edema and involving the epiphysis, metaphysis, and diaphysis. Focal physeal thickening is indicated by the arrow (E) and (F) 7-month follow-up showing increase in size of the focal femoral physeal thickening (arrows), persistence of diffuse epiphyseal, metaphyseal, diaphyseal, and patellar changes of presumed osteonecrosis. Note that the involved areas of intramedullary changes appear to be better demarcated and the interstices somewhat thicker. G–I: Nine-month follow-up MR demonstrating focal central physeal bridging tibia and femur (arrows) in the site of previous cartilaginous in growth on the femur. Note thickening of interstices of intramedullary changes and some degree of regression of the extent of involvement of osteonecrosis.
Osteonecrosis was present at baseline imaging and persisted at follow-up in one patient. Seven other patients developed osteonecrosis in 11 knees after initiation of therapy. The appearance of osteonecrotic lesions was characterized not by well-defined geographic margins that are typically recognized as osteonecrosis [17], but rather by an overall lacey pattern of signal abnormality (Fig. 2) associated with varying degrees of surrounding edema. Shifting distribution of osteonecrosis was seen in one case and healing was demonstrated at the second follow-up imaging in one other patient. None of the patients with findings of osteonecrosis was symptomatic.
Non-ossifying fibromas were noted in 13 knees of 11 patients at baseline. At first follow-up in seven knees of six patients, two non-ossifying fibromas regressed and one developed during the time interval between the two imaging studies. Increased joint fluid was noted in seven patients, four of them at baseline with resolution of the increased fluid at follow-up in three of the four. Synovial proliferation was demonstrated in two patients.
DISCUSSION
The discrepancy between radiographic findings and those demonstrated by MRI in the current study is of great importance in defining methods to monitor the development of potential skeletal toxicities as several new anticancer agents with similar mechanisms of action have been incorporated into clinical trials in children with cancer. Combination studies of conventional chemotherapy with agents that target the VEGF pathway (e.g., bevacizumab and sorafenib) have been recently completed in children with cancer [18–20]. MRI is well known to be exquisitely sensitive for assessing cartilage and bone marrow changes [21,22], traumatic injuries as well as osteonecrotic changes not demonstrable radiographically [23,24]. Using prospectively acquired radiographs and MRI of the knees, we identified disturbance of physeal development in a single patient but osteonecrosis in eight patients, seven having developed while on therapy, none of which were apparent on radiographs. We showed that radiographs may provide insufficient evaluation of the potential bone toxicity of agents used in the treatment of children with cancer.
Solid tumors are known to parasitize host tissues via stimulation of endothelial signaling responses through vascular recruitment. Such angiogenic pathways permit tumor access to host nutrients and oxygen. Antiangiogenic agents block development of tumor neovascularity but are non-selective and thus block vascularization required for normal skeletalgenesis and endochondral ossification as well as induce early apoptosis of chondrocytes [11].
Because of evidence of altered skeletalgenesis demonstrated in preclinical models using VEGF-targeted therapies [11,25–32], we prospectively monitored our patients for cartilaginous growth plate abnormalities using both knee radiographs and MRI. We found changes in growth plates to be uncommon after the use of vandetanib with and without dasatinib. However, MRI delineated findings not assessable by radiographs such as osteonecrosis, and demonstrated more physeal abnormalities than shown radiographically. MRI also identified more non-ossifying fibromas and more instances of increased joint fluid than did radiographs. Because of its increased sensitivity to skeletal and soft tissue changes, we believe that MRI is superior to radiographs for monitoring children for potential skeletal developmental toxicity associated with VEGF-targeting agents.
MRI imaging can critically assess musculoskeletal anatomy and development. STIR images [33] can readily depict physeal patency. Multiple MRI sequences for assessing cartilage are available and include T1-weighted spin echo with fat saturation, FLASH 2-D, and DESS sequences. Though a more complex and costly modality than radiographs, MRI is a valuable imaging method for children in that it involves no ionizing radiation and exquisitely depicts bone and soft tissue detail not demonstrated using radiography.
The sensitivity of MRI provides for visualization of not only pathologic findings but also normal findings not commonly recognized [25]. Such imaging findings must be assessed longitudinally for their clinical significance. One such example is that of cartilaginous canals, also termed vascular canals. These are normal structures in newborns providing nutritional support to the cartilaginous femoral head via an extensive network of blood vessels throughout the cartilaginous femoral head [26,27]. These canals gradually regress and eventually disappear as the secondary center of ossification which becomes the femoral head enlarges and the articular cartilage thickens [28,29]. Thus far, the exact point of maturation at which these canals disappear is uncertain. Since the vast majority of patients with DIPG are young, we might expect the presence of vascular channels, as we saw. Whether or not treatment with antiangiogenic agents alters the prevalence, duration, and function of these channels has yet to be determined.
Glucocorticoids are known to alter physeal development and maturation by both direct and indirect effect on chondrocytes and chondrogenesis [30,31,34]. However, their potential interaction with an antiangiogenic agent and subsequent effect on developing bone is as yet unknown. Our findings underscore the importance of prospective MRI screening of patients receiving antiangiogenic agents and reiterate a report by Fangusaro et al. These investigators reported three children treated with bevacizumab who developed osteonecrosis. Two of the three patients underwent imaging due to joint pain (one in a wrist and one in both knees). Radiographs were either normal or unhelpful but MRI demonstrated osteonecrosis in all three patients [35].
MRI-determined osteonecrosis is a well-reported toxicity of therapy in patients treated for acute lymphocytic leukemia [36]. However, similar studies of well-characterized patient cohorts are not available for pediatric patients with brain tumors. At present, we are unable to explain the unusual lacey, very irregular and in some cases transient pattern of osteonecrosis. Whether or not these findings represent osteonecrosis as we know it or perhaps a manifestation of altered vascularity or altered angiogenesis is uncertain. An additional possibility is that this appearance results from the interaction of glucocorticoids with antiangiogenesis agents.
Although the size of our study cohort is relatively small, this study represents the largest prospective pediatric clinical evaluation of changes in skeletalgenesis during antiangiogenic therapy. Its strengths also lie in the standardized imaging review of prospectively acquired MRIs and radiographs, regardless of whether or not the patient complained of clinical symptoms that could be related to osteonecrosis. Further, the study design provided the ability to assess the sensitivity of imaging modalities in skeletalgenesis. Despite these strengths, the short 4-month follow-up period precludes determination of potential long-term effects of exposure to antiangiogenic agents on skeletal development in these children.
Stronger field strength MRI units (e.g., 3 T) can provide increased sensitivity and spatial resolution compared to lower strength magnets (e.g., 1.5 T), thereby allowing for visualization of some findings that may be very subtle on lower strength magnets [37–39]. Whether or not the sensitivity of 3.0 T MRI is any greater in defining the characteristics we captured compared with 1.5 T is under study [37–39]. As 24% of the studies reported were performed on 1.5 T MRI, our results may under-report cartilaginous abnormalities.
We found few concerning growth plate abnormalities in our study. However, the findings of one case of focal physeal fusion and several unexpected cases of osteonecrosis justify the use of MRI over radiographs when monitoring young children for potential treatment-associated altered skeletalgenesis. The short 4-month follow-up for most of our patients precludes comments regarding potential long-term effects of anti-VEGF therapy on growing bones. However, we have demonstrated that MRI monitoring is more sensitive and informative than radiographs. Long-term MRI monitoring of patients will be needed as use of antiangiogenic agents increases.
ACKNOWLEDGEMENTS
This work was supported in part by grant US National Institutes of Health Cancer Center Support (CORE) Grant P30 CA-21765, a Center of Excellence grant from the State of Tennessee, the American Lebanese Syrian Associated Charities (ALSAC), the Noyes Brain Tumor Foundation, Musicians Against Childhood Cancer (MACC), AstraZeneca, and The Cure Starts Now Foundation. We thank Sandra Gaither for manuscript preparation.
Footnotes
Conflict of interest: Nothing to declare.
REFERENCES
- 1.Kreisl TN, Kim L, Moore K, et al. Phase II trial of single-agent bevacizumab followed by bevacizumab plus irinotecan at tumor progression in recurrent glioblastoma. J Clin Oncol. 2009;27:740–745. doi: 10.1200/JCO.2008.16.3055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Motzer RJ, Rini BI, Bukowski RM, et al. Sunitinib in patients with metastatic renal cell carcinoma. JAMA. 2006;295:2516–2524. doi: 10.1001/jama.295.21.2516. [DOI] [PubMed] [Google Scholar]
- 3.Zondor SD, Medina PJ. Bevacizumab: An angiogenesis inhibitor with efficacy in colorectal and other malignancies. Ann Pharmacother. 2004;38:1258–1264. doi: 10.1345/aph.1D470. [DOI] [PubMed] [Google Scholar]
- 4.Widemann BC, Kim A, Fox E, et al. A phase I trial and pharmacokinetic study of the Raf kinase and receptor tyrosine kinase inhibitor sorafenib in children with refractory solid tumors or refractory leukemias. Clin Cancer Res. 2012;18:6011–6022. doi: 10.1158/1078-0432.CCR-11-3284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Glade Bender JL, Adamson PC, Reid JM, et al. Phase I trial and pharmacokinetic study of bevacizumab in pediatric patients with refractory solid tumors: A children’s oncology group study. J Clin Oncol. 2008;26:399–405. doi: 10.1200/JCO.2007.11.9230. [DOI] [PubMed] [Google Scholar]
- 6.Hargrave D, Bartels U, Bouffet E. Diffuse brainstem glioma in children: Critical review of clinical trials. Lancet Oncol. 2006;7:241–248. doi: 10.1016/S1470-2045(06)70615-5. [DOI] [PubMed] [Google Scholar]
- 7.Broniscer A, Baker JN, Baker SJ, et al. Prospective collection of tissue samples at autopsy in children with diffuse intrinsic pontine glioma. Cancer. 2010;116:4632–4637. doi: 10.1002/cncr.25405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Kleihues P, Burger PC, Aldape KD, et al. Glioblastoma. In: Louis DN, Ohgaki H, Wiestler OD, Cavenee WK, editors. WHO classification of tumors of the central nervous system. Lyon: IARC Press; 2007. pp. 33–49. [Google Scholar]
- 9.Broniscer A, Baker JN, Tagen M, et al. Phase I study of vandetanib during and after radiotherapy in children with diffuse intrinsic pontine glioma. J Clin Oncol. 2010;28:4762–4768. doi: 10.1200/JCO.2010.30.3545. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Broniscer A, Baker SD, Baker JN, et al. A phase I study of the combination of vandetanib and dasatinib administered during and after radiotherapy (RT) in children. 2011:iii98. [Google Scholar]
- 11.Zelzer E, Olsen BR. Multiple roles of vascular endothelial growth factor (VEGF) in skeletal development, growth, and repair. Curr Top Dev Biol. 2005;65:169–187. doi: 10.1016/S0070-2153(04)65006-X. [DOI] [PubMed] [Google Scholar]
- 12.Patyna S, Arrigoni C, Terron A, et al. Nonclinical safety evaluation of sunitinib: A potent inhibitor of VEGF, PDGF, KIT, FLT3, and RET receptors. Toxicol Pathol. 2008;36:905–916. doi: 10.1177/0192623308326151. [DOI] [PubMed] [Google Scholar]
- 13.Kabata T, Matsumoto T, Yagishita S, et al. Vascular endothelial growth factor in rabbits during development of corticosteroid-induced osteonecrosis: A controlled experiment. J Rheumatol. 2008;35:2383–2390. doi: 10.3899/jrheum.070838. [DOI] [PubMed] [Google Scholar]
- 14.Smith AR, Hennessy JM, Kurth MA, et al. Reversible skeletal changes after treatment with bevacizumab in a child with cutaneovisceral angiomatosis with thrombocytopenia syndrome. Pediatr Blood Cancer. 2008;51:418–420. doi: 10.1002/pbc.21597. [DOI] [PubMed] [Google Scholar]
- 15.Modak S, Cheung NK, Abramson SJ, et al. Lack of early bevacizumab-related skeletal radiographic changes in children with neuroblastoma. Pediatr Blood Cancer. 2009;52:304–305. doi: 10.1002/pbc.21776. [DOI] [PubMed] [Google Scholar]
- 16.Laor T, Chun GF, Dardzinski BJ, et al. Posterior distal femoral and proximal tibial metaphyseal stripes at MR imaging in children and young adults. Radiology. 2002;224:669–674. doi: 10.1148/radiol.2243011259. [DOI] [PubMed] [Google Scholar]
- 17.Karimova EJ, Kaste SC. MR imaging of osteonecrosis of the knee in children with acute lymphocytic leukemia. Pediatr Radiol. 2007;37:1140–1146. doi: 10.1007/s00247-007-0579-x. [DOI] [PubMed] [Google Scholar]
- 18.Gururangan S, Chi SN, Young PT, et al. Lack of efficacy of bevacizumab plus irinotecan in children with recurrent malignant glioma and diffuse brainstem glioma: A pediatric brain tumor consortium study. J Clin Oncol. 2010;28:3069–3075. doi: 10.1200/JCO.2009.26.8789. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Grunewald TG, Greulich N, Kontny U, et al. Targeted therapeutics in treatment of children and young adults with solid tumors: An expert survey and review of the literature. Klin Padiatr. 2012;224:124–131. doi: 10.1055/s-0032-1301930. [DOI] [PubMed] [Google Scholar]
- 20.Inaba H, Rubnitz JE, Coustan-Smith E, et al. Phase I pharmacokinetic and pharmacodynamic study of the multikinase inhibitor sorafenib in combination with clofarabine and cytarabine in pediatric relapsed/refractory leukemia. J Clin Oncol. 2011;29:3293–3300. doi: 10.1200/JCO.2011.34.7427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Thapa MM, Iyer RS, Khanna PC, et al. MRI of pediatric patients: Part 1, normal and abnormal cartilage. AJR Am J Roentgenol. 2012;198:W450–W455. doi: 10.2214/AJR.10.7280. [DOI] [PubMed] [Google Scholar]
- 22.Strouse PJ. Overview of musculoskeletal imaging. In: Slovis TL, Caffey J, editors. Caffey’s pediatric diagnostic imaging. Mosby/Elsevier; 2008. p. 2831. [Google Scholar]
- 23.Imhof H, Breitenseher M, Trattnig S, et al. Imaging of a vascular necrosis of bone. Eur Radiol. 1997;7:180–186. doi: 10.1007/s003300050131. [DOI] [PubMed] [Google Scholar]
- 24.Zibis AH, Karantanas AH, Roidis NT, et al. The role of MR imaging in staging femoral head osteonecrosis. Eur J Radiol. 2007;63:3–9. doi: 10.1016/j.ejrad.2007.03.029. [DOI] [PubMed] [Google Scholar]
- 25.Laor T, Jaramillo D. MR imaging insights into skeletal maturation: What is normal? Radiology. 2009;250:28–38. doi: 10.1148/radiol.2501071322. [DOI] [PubMed] [Google Scholar]
- 26.Trueta J. The normal vascular anatomy of the human femoral head during growth. J Bone Joint Surg Br. 1957;39-B:358–394. doi: 10.1302/0301-620X.39B2.358. [DOI] [PubMed] [Google Scholar]
- 27.Haines RW. Cartilage canals. J Anat. 1933;68:45–64. [PMC free article] [PubMed] [Google Scholar]
- 28.Hughes LO, Aronson J, Smith HS. Normal radiographic values for cartilage thickness and physeal angle in the pediatric hip. J Pediatr Orthop. 1999;19:443–448. doi: 10.1097/00004694-199907000-00005. [DOI] [PubMed] [Google Scholar]
- 29.Rissech C, Schaefer M, Malgosa A. Development of the femur—Implications for age and sex determination. Forensic Sci Int. 2008;180:1–9. doi: 10.1016/j.forsciint.2008.06.006. [DOI] [PubMed] [Google Scholar]
- 30.DeLuca F. Impaired growth plate chondrogenesis in children with chronic illnesses. Pediatr Res. 2006;59:625–629. doi: 10.1203/01.pdr.0000214966.60416.1b. [DOI] [PubMed] [Google Scholar]
- 31.Silvestrini G, Mocetti P, Di GR, et al. Localization of the glucocorticoid receptor mRNA in cartilage and bone cells of the rat. An in situ hybridization study. Eur J Histochem. 2003;47:245–252. doi: 10.4081/834. [DOI] [PubMed] [Google Scholar]
- 32.Hall AP, Westwood FR, Wadsworth PF. Review of the effects of anti-angiogenic compounds on the epiphyseal growth plate. Toxicol Pathol. 2006;34:131–147. doi: 10.1080/01926230600611836. [DOI] [PubMed] [Google Scholar]
- 33.Jaramillo D, Hoffer FA. Cartilaginous epiphysis and growth plate: Normal and abnormal MR imaging findings. AJR Am J Roentgenol. 1992;158:1105–1110. doi: 10.2214/ajr.158.5.1566676. [DOI] [PubMed] [Google Scholar]
- 34.Chrysis D, Ritzen EM, Savendahl L. Growth retardation induced by dexamethasone is associated with increased apoptosis of the growth plate chondrocytes. J Endocrinol. 2003;176:331–337. doi: 10.1677/joe.0.1760331. [DOI] [PubMed] [Google Scholar]
- 35.Fangusaro J, Gururangan S, Jakacki RI, et al. Bevacizumab-associated osteonecrosis of the wrist and knee in three pediatric patients with recurrent central nervous system (CNS) tumors. J Clin Oncol. 2013;31:e24–e27. doi: 10.1200/JCO.2012.43.6733. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Karimova EJ, Rai SN, Howard SC, et al. Femoral head osteonecrosis in pediatric and young adult patients with leukemia or lymphoma. J Clin Oncol. 2007;25:1525–1531. doi: 10.1200/JCO.2006.07.9947. [DOI] [PubMed] [Google Scholar]
- 37.Friedrich KM, Reiter G, Kaiser B, et al. High-resolution cartilage imaging of the knee at 3T: Basic evaluation of modern isotropic 3D MR-sequences. Eur J Radiol. 2011;78:398–405. doi: 10.1016/j.ejrad.2010.01.008. [DOI] [PubMed] [Google Scholar]
- 38.Milewski MD, Smitaman E, Moukaddam H, et al. Comparison of 3D vs. 2D fast spin echo imaging for evaluation of articular cartilage in the knee on a 3T system scientific research. Eur J Radiol. 2012;81:1637–1643. doi: 10.1016/j.ejrad.2011.04.072. [DOI] [PubMed] [Google Scholar]
- 39.Mosher TJ. Musculoskeletal imaging at 3T: Current techniques and future applications. Magn Reson Imaging Clin N Am. 2006;14:63–76. doi: 10.1016/j.mric.2005.12.002. [DOI] [PubMed] [Google Scholar]