Abstract
Background
Heart failure, a leading cause of hospitalization among elderly people, disproportionately afflicts African-American and other non-White populations. Studies of health care interventions often do not include these groups in proportion to numbers in the patient population. Our objective was to assess whether a randomized controlled effectiveness trial enrolled patients by ethnicity/race, gender, and age in proportion to those eligible.
Methods
We conducted a randomized controlled trial comparing nurse management and usual care among ambulatory heart failure patients at the four hospitals in East and Central Harlem, New York. We incorporated culturally sensitive and age-appropriate strategies to enroll a demographically representative group into the trial. Recruitment proceeded in several steps: identifying patients with billing code and visit criteria, documenting systolic dysfunction, obtaining clinician permission and correct addresses, contacting patients, and enrolling eligible patients. We assessed differences by ethnicity/race and gender at successive steps in the recruitment process, and differences between enrollees and refusals regarding overall health, evaluation of medical care, and difficulty receiving care.
Results
We enrolled 406 ambulatory patients by ethnicity/race and gender in proportion to the numbers eligible to be contacted (46% African-American/Black, 33% Hispanic, and 47% female). Among patients contacted, however, those 18 through 74 years were 2.0 to 3.3 times more likely than those ≥75 years to enroll (p < 0.001).
Conclusions
The recruitment strategy successfully enrolled patients by ethnicity/race, gender, and age through 74 years, but not those ≥75 years. Registries of patients who refuse to enroll in trials could provide guidance for clinical and public policy.
Keywords: randomized controlled trial, recruitment, effectiveness trial, congestive heart failure, nurse management
Clinical trials of health care interventions have not included certain groups in proportion to their numbers in the patient population. From 1991 through 2000, for example, 40% of the randomized controlled trials of acute myocardial infarction or unstable angina excluded people >75 years. This age group accounted for only 9% of people in these trials, but 37% of all patients with myocardial infarctions.1 Similarly, women comprised 25% of enrollees in cardiovascular trials, but 43% of patients with myocardial infarction.1 Although clinical studies have generally not assessed whether rates of recruiting ethnic minority patients have differed from those of Whites,2 the majority of patients with heart failure enrolled into trials of investigational drugs have historically been middle-aged, male, and White.3 The resulting lack of information about important patient subgroups has raised the question of whether the findings of studies are applicable to groups underrepresented in trials. This situation has posed a dilemma for clinicians seeking evidence to guide patient management and policy makers wishing to base regulatory standards and insurance coverage on evidence-based guidelines.4–6
In recognition of these considerations, the federal government has adopted policies to promote greater representation of certain groups in clinical trials. Since 1994, the National Institutes of Health has required that the clinical research projects that it funds include women and minority groups, though no such requirements pertain to elderly people.6 Guidelines of the Food and Drug Administration since 1989 have called for studies of pharmaceuticals to include all age groups, including older people who might benefit.5 The success of studies implementing these policies will therefore influence the evidence likely to be available for future clinical and public policy makers.
This article reports on enrollment into a recent federally funded effectiveness trial, one with broad inclusion and minimal exclusion criteria designed to be representative of and generalizable to patients with the target condition. This randomized controlled trial evaluated the effectiveness of nurse management to improve the quality of care for heart failure patients with systolic dysfunction. Because the prevalence of this condition is higher among African-Americans and increases with age, we conducted the study in East and Central Harlem, New York City. This inner-city area's population of about 225,000, primarily African-American (56%) and Hispanic (35%), in the year 2000 had mortality rates due to heart disease among the highest in the city.7–10 Women comprised 53% and people >65 years comprised 11% of this population. We examined how successful our efforts had been to enroll people representative of the race or ethnicity, gender, and age distribution of the underlying eligible patient pool. The results may guide others' recruitment efforts and in turn have implications for the availability of future research results that are generalizable and able to better inform clinical practice and public policy.
Methods
We evaluated our recruitment strategy for the randomized controlled trial of the effectiveness of nurse management to improve functioning and reduce hospitalizations among patients with systolic dysfunction. During this trial, nurses counseled patients to improve self-management of the condition and interacted with physicians and other clinicians to improve prescribing of appropriate medications and dosages.
Our recruitment strategy involved a multistep process to identify and recruit heart failure patients with documented systolic dysfunction into the trial. Although we did not adopt specific targets for enrollment of these underrepresented groups, we incorporated strategies to improve enrollment, especially of minority patients, as described below.
All four hospitals in East and Central Harlem, the area's major providers, collaborated in the trial: one large private academic medical center, one minority-owned small private community hospital, and two medium-sized municipal hospitals. We applied the same multistep process to identify and recruit patients at two neighborhood health centers. Because we identified only seven patients from the neighborhood health centers with documented systolic dysfunction, however, and because either the patients or their clinicians refused permission to participate, we have excluded those patients from this analysis.
Local Clinicians' Support
To improve clinicians' support for the study, we initially worked with local clinical leaders to operationalize national evidence-based clinical guidelines and develop a clinical protocol consistent with local circumstances.11– 13 A small multidisciplinary, multi-specialty planning group prepared a draft protocol to guide the nurses' interactions with physicians and nurses' counseling to improve patients' self-management. At a meeting of clinicians from the participating sites, clinicians representing each site refined and approved the protocol. The protocol provided the basis for gaining clinicians' acceptance of the study and permission to recruit their patients into the trial. The key clinical leaders who helped to develop the protocol built relationships with the study team and encouraged their patients and other clinicians to participate in the study. The trial had the following criteria for eligibility: adults age ≥18 years; systolic dysfunction documented on a cardiac test of left ventricular function; English or Spanish speaking; community dwelling at enrollment; and currently a patient in a general medicine, geriatrics, or cardiology clinic or office at one of the participating sites. The following exclusion criteria applied: medical conditions that prevented a patient's interacting with the nurse, including blindness, deafness, or cognitive impairment; medical conditions that called for individualized management that might differ from the standard protocol, namely pregnancy and renal dialysis; and receipt of procedures that corrected systolic dysfunction, such as heart transplantation.
After development of the protocol, we held group meetings with clinicians at each participating clinic to obtain their input about the study, gain their endorsement, and encourage them to speak positively about the study with their patients. Following briefings at each site, all the 150 clinicians, 137 physicians and 13 physician assistants or nurse practitioners in the participating clinics and offices gave written overall permission to recruit their patients.
Recruitment Process
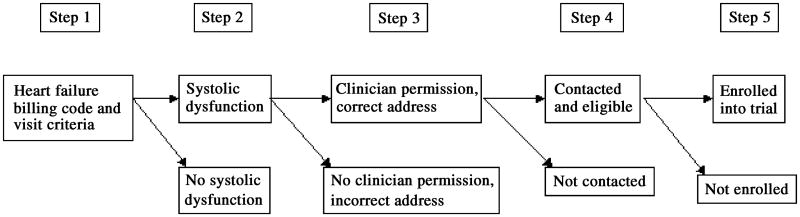
Starting with services billed from each hospital, we identified patients with International Classification of Diseases-Version 9-Clinical Modification (ICD9-CM) and diagnosis-related group (DRG) codes for heart failure from March 1999 through February 2001 who had had at least one clinic or office visit during the year in general medicine, geriatrics, or cardiology (Step 1) (Fig. 1). We next checked the cardiac test results of these patients, including echocardiography, radionuclide ventriculography, myocardial stress sestamibi/thallium testing, and left-heart catheterization, to identify those with impaired systolic dysfunction, defined as a left ventricular ejection fraction <40%, or moderately/severely reduced systolic dysfunction (Step 2). We then sought permission from clinicians to recruit their specific patients and mailed a letter signed by the physician coordinator at the provider site to each of the approved patients (Step 3). The letter briefly described the study and indicated that we would be contacting the person.
Fig 1.
Steps in the Recruitment Process.
Three trained lay recruiters with bachelor degrees who were bilingual in English and Spanish sought to contact these patients to further describe the study and determine their eligibility (Step 4), and for those interested, to schedule an appointment to obtain their informed consents and enroll them into the trial (Step 5). From September 2000 through January 2002, the recruiters telephoned the patients at least 15 times at different times of the day and week. For wrong numbers, the recruiters searched telephone records and contacted any next of kin in the providers' records for current telephone numbers. If these methods were not successful, the recruiters approached patients when they came for scheduled visits with their clinicians. Finally, we sent a telegram to patients whom we were still unable to reach. The Institutional Review Boards at each hospital approved the study methods.
We used findings and recommendations from published articles and consulted with local researchers and community leaders to develop a minority recruitment strategy.14–18 We first sent bilingual letters to patients that included the following information: their clinicians had given us permission to contact them; they would receive no experimental drugs or tests as part of the study; we would pay their transportation plus $50 for the interview and up to a total of $125 for later interviews; we would keep all their personal information confidential; and the study results might help improve care for people with their heart condition. The letter was written at a 6th grade reading level.
To follow up, we used only bilingual, African-American or Latino recruiters experienced in health care settings, whom we had trained to give simple, clear information; emphasize that patients' usual clinicians would remain in charge of their care; welcome patients to bring a relative or friend to the recruitment session; and be flexible and accommodating in scheduling times, including evenings, for both telephone and in-person conversations. We also arranged for taxi service, if the patient wished. As part of their training, recruiters interviewed mock patients who acted out common reasons for resistance, including fearing experimentation, mistrusting the medical system, being too busy, and misunderstanding the research protocol, and we gave feedback on ways to address these challenges. We held weekly recruiter meetings to discuss recruitment progress, highlight successful techniques, and brainstorm about ways to handle difficult situations that had arisen.
Analysis
We analyzed whether patients' ethnicity (non-Hispanic African-American or Black, Hispanic, non-Hispanic White, other), gender, or age varied across the five sequential steps of the recruitment process (Fig. 2): medical billing and visit criteria (Step 1); documented systolic dysfunction (Step 2); clinician permission and valid address (Step 3); recruiter contact and eligibility confirmation (Step 4); and enrollment into the trial (Step 5). We used t-tests to assess differences in mean age and chi-squared tests to assess differences in race/ethnicity and gender for patients who did and did not proceed from each step to the subsequent step in the recruitment process. We estimated a multivariate logistic regression to assess differences in these characteristics between patients who were contacted and eligible but not enrolled (Step 4) and those who chose to enroll (Step 5). To better communicate the meaning of the results, we converted odds ratios from the logistic regression to risk ratios using published methods.19 Finally, we asked all people who were contacted three questions regarding their overall health, evaluation of medical care, and difficulty receiving care. We compared responses to these questions among enrollees and patients who refused enrollment.
Fig 2.

Age distribution at different steps in the eligibility and recruitment process. [Color figure can be viewed in the online issue, which is available at www.interscience.wiley.com.]
Results
Of the 4,030 patients with heart failure billings who met the criteria for clinic or office visits in the first step, 1,762 (43.7%) met the second-step criteria for documented systolic dysfunction (Table 1). At the third step, clinicians gave permission for recruitment, and we obtained correct addresses to send letters to 1,548 (87.9%) of the 1,762 patients. Clinicians' reasons for refusing permission included patients' cognitive impairment and conditions requiring individualized management. Recruiters attempted to contact the remaining 1,548 patients from the four hospitals: 202 (13.0%) had died, 334 (21.6%) were unreachable, 349 (22.5%) were ineligible, and 663 (42.8%) were eligible, alive, and contacted (Step 4) (Table 2). Of these 663, 264 (39.8%) refused or did not keep recruitment appointments, and 399 (60.2%) were enrolled (Step 5). An additional 7 patients recruited separately in February 2002, for a total of 406 patients, are not included in this analysis.
Table 1. Eligibility Determination and Recruitment.
| Patients # (%)a | |
|---|---|
| Step 1: Heart failure billing code and visit criteria met | 4,030 |
| Step 2: Systolic dysfunction documented | 1,762 (43.7) |
| Step 3: Clinician permission and address correct | 1,548 (87.9) |
| Step 4: Contacted and eligible | 663 (42.8) |
| Step 5: Enrolled | 399 (60.2) |
Each percentage uses as the denominator the number of patients eligible to continue in the previous step. An additional seven patients recruited separately in February 2002 are not included in this analysis.
Table 2. Results of Recruiters' Contacts.
| Category | Patients # (%) n = 1, 548 | |
|---|---|---|
| Dead | 202 (13.1) | |
| Unreachable | 334 (21.6) | |
| Ineligible | 349 (22.5) | |
| No longer patient at site/lived outside area | 136 (39.0) | |
| Cognitive impairment | 72 (20.6) | |
| Institutionalized | 40 (11.5) | |
| Medical condition (pregnant, dialysis, heart transplant/no longer systolic dysfunction) | 31 (8.9) | |
| Disability (hearing, vision, homebound) | 26 (7.4) | |
| In another study | 22 (6.3) | |
| Language other than English and Spanish | 13 (3.7) | |
| Other | 9 (2.6) | |
| Contacted and eligible for enrollment | 663 (42.8) | |
| Enrolled | 399 (60.2) | |
| Refused | 228 (34.4) | |
| Did not keep enrollment appointments | 36 (5.4) |
Race or Ethnicity
In bivariate, unadjusted analyses, patients eligible at the second step of eligibility determination and recruitment, having documented systolic dysfunction, did not differ significantly in race or ethnicity from those who did not (p = 0.09). At the third step, however, non-Hispanic White patients were less likely than others to receive clinicians' permission for recruitment and have valid addresses, (6.7% non-Hispanic White versus 32.5% Hispanic and 36.4% for Black patients [p<0.0001]). Non-Hispanic Whites at this step were significantly older than other racial or ethnic groups. At the last two steps, those contacted and found to be eligible and those enrolled into the trial, race, or ethnicity did not differ significantly (p = 0.38 and p = 0.32, respectively).
Gender
Patients eligible at the second step, having documented systolic dysfunction, did differ significantly in gender from those who did not. Women were less likely than men to have documented systolic dysfunction, 34.6% compared with 54.5% (p<0.0001). Our data did not address whether women were less likely to receive a test to measure systolic dysfunction, or whether given a test, they were less likely to have impaired function. Patients did not differ significantly in gender at the third step, receiving clinicians' permission or having valid addresses (p = 0.11); at the next step, being contacted and eligible (p = 0.93); or at the last step, being enrolled into the study (p = 0.13).
Age
Patients differed significantly in age at each of the successive steps as follows (p<0.04): no documented systolic dysfunction, mean 67.9 years (95% confidence interval [CI] 67.3, 68.6) versus documented systolic dysfunction, mean 64.6 years (CI 63.9, 65.3); no clinician permission or invalid address, mean 68.7 years (CI 66.7, 70.7) versus permission or valid address, mean 64.1 years (CI 63.3, 64.8); not contacted or ineligible, mean 65.6 years (CI 64.6, 66.6) versus contacted and eligible, mean 62.0 years (CI 61.0, 63.1); and refused or did not keep recruitment appointments, mean 66.4 years (CI 64.8, 68.1) versus enrolled into the trial, mean 59.1 years (CI 57.8, 60.5) (Fig. 2). Compared to those enrolled into the trial, people who had died, were ineligible, or refused to participate were significantly older (p<0.0001). Among those ineligible, people who were cognitively impaired, had prohibitive disabilities (blind, deaf, homebound) or could not speak English or Spanish were significantly older than those enrolled. Those who could not be reached or did not keep recruitment appointments, however, did not differ significantly in age from those who enrolled (p = 0.46). The age of people enrolled did not differ significantly by hospital.
In multivariate analysis of whether someone contacted and eligible enrolled in the trial, a person in age group from 18 through 74 years was 2.0 to 3.3 times more likely than a person 75 years or older to enroll (p<0.001). Neither gender (p = 0.08) nor race/ethnicity (p = 0.17 to 0.91) had a significant effect.
Refusals
We asked the people who refused to participate to answer three questions about their overall health, their evaluation of their medical care at the recruitment site, and their difficulty in getting medical care when they needed it. Of the 228 people who refused, 85, 84, and 81 answered the respective questions. Although the respondents' replies evaluating their medical care or difficulty getting care did not differ significantly from those of trial enrollees (p = 0.68), the respondents reported significantly poorer health than trial enrollees (p<0.0001). Of these respondents, 64% reported fair or poor health compared to 13% of the enrollees.
Conclusions
A culturally sensitive approach to gain the participation of inner-city, minority patients with heart failure into a randomized controlled trial proved to be successful. The trial to evaluate nurse management enrolled African-American and Hispanic patients in proportion to their numbers at the four hospitals in East and Central Harlem. The study engaged clinical leaders in developing the protocol that the nurses used, and gained the strong support of clinicians. We tailored the recruitment process to be convenient and understandable to patients and to address possible concerns about experimentation and mistrust of the health care system.
The study also succeeded in enrolling both women and men in proportion to the numbers eligible to be contacted at the four hospitals which ranged from a tertiary academic medical center to a small community hospital and two large municipal hospitals. Women were less likely to have documented systolic dysfunction, a situation consistent with findings from other investigators.2,20,21
We were not as successful in enrolling people 75 years and older. Despite keeping exclusion criteria to a minimum, the trial enrolled significantly fewer patients in this age group at successive steps of the recruitment process. Many of these cases were unavoidable. Clinicians were more likely to refuse permission to recruit these patients, often because of complicating comorbidities, and patients 75 years and older were more likely to have cognitive impairments, which excluded them from the study. Even among those eligible and contacted, however, patients and their families were more likely to refuse to participate. This pattern occurred in spite of the fact that participation in the study entailed at the most only one in-person meeting with the nurse, which could have been scheduled to coincide with a regular physician visit. The responses of people who refused suggest that their poorer health may have played a role.
We could identify no other randomized controlled trials involving some variant of nurse management for heart failure patients that identified and recruited patients from ambulatory clinics or offices. Seven randomized trials in the United States whose enrollees averaged from 65 to 76 years identified or recruited patients as inpatients, sometimes with the requirement that they be at least 70 years old.22–25 Current or recent inpatients and their families may be more likely to agree to participate, perhaps because they feel more at risk of the condition's complications and more open to approaches to reduce that risk.
Our trial had certain limitations. Our trial of nurse management involved no invasive or experimental procedures, a matter that we highlighted in our recruitment process. It is therefore unclear whether the methods effective in recruiting ambulatory patients of both genders, African-American and Latinos patients, and older adults through 74 years for this trial would be successful with invasive or experimental interventions. Another is the lack of any comparison between our recruitment strategy and any other. Such comparisons, involving variations in approach, are an appropriate subject for further research.
Evidence-based guidance for clinical practice and public policy requires that clinical studies enroll people representative of the underlying patient group. As the U.S. population ages, improving recruitment efforts to better represent the older age groups has assumed increasing importance. Clinicians often lack evidence to guide decisions about medical and surgical management. Like other insurers, the Medicare program also requires better information on effectiveness and safety to determine whether or not to cover specific medical interventions, especially as it moves to cover complex interventions, such as disease management (Public Law #108-173).
Given current policy focus on studying interventions to reduce health-related disparities among different populations, it is promising that the simple steps used by this multisite randomized controlled effectiveness trial successfully enrolled people by race/ethnicity and gender in proportion to the patient population. Other researchers may wish to adapt these methods to their circumstances.
Our recruitment experience also supports the importance of registries of patients who refuse to enroll in clinical trials. Through such registries, researchers can initially compare the characteristics of people who did and did not enroll to assess the generalizability of the results.26 By following both groups over time and documenting their health-related outcomes and costs, one might also gain insight into whether an intervention's results would have differed among those outside the trial. In the absence of better representation of people 75 years and older and other groups in trials, analysis of registry data might offer some guidance to policy makers.
Acknowledgments
This project was supported by grant number R01 HS01402 from the U.S. Agency for Healthcare Research and Quality. The manuscript has not been published elsewhere.
The findings and conclusions in this paper are those of the authors, and do not necessarity represent the views of the National Center for Health Statistics, Centers for Disease Control and Prevention.
References
- 1.Lee PY, Alexander KP, Hammill BG, et al. Representation of elderly persons and women in published randomized trials of acute coronary syndromes. JAMA. 2001;286:708–713. doi: 10.1001/jama.286.6.708. [DOI] [PubMed] [Google Scholar]
- 2.Ness RB, Nelson DB, Kumanyika SK, et al. Evaluating minority recruitment into clinical studies: how good are the data? Ann Epidemiol. 1997;7:472–478. doi: 10.1016/s1047-2797(97)00080-x. [DOI] [PubMed] [Google Scholar]
- 3.Jessup M, Brozena S. Heart failure. N Engl J Med. 2003;348:2007–2018. doi: 10.1056/NEJMra021498. [DOI] [PubMed] [Google Scholar]
- 4.International Longevity Center. Clinical trials and older persons: the need for greater representation. Issue Brief. 2002 Nov-Dec; [Google Scholar]
- 5.United States, Food and Drug, Administration, Center for Drug Evaluation and Research. Guidance for industry: guideline for the study of drugs likely to be used in the elderly. Rockville: Nov, 1989. [Google Scholar]
- 6.United States, National Institutes of Health. [Accessed November 6, 2003];NIH guidelines on the inclusion of women and minorities as subjects in clinical research: notice. 1994 Mar 28; RIN 0905-ZA18. 59 Federal Register 11146, Part VIII. http://frwebgate4.access.gpo.gov/cgi-bin/waisgate.cgi?WAISdocID.
- 7.New York City, Department of City Planning. Community district profiles: Manhattan community district 10. Central Harlem: [Accessed March 2, 2004]. http://www.nyc.gov/html/dcp/pdf/lucds/mn10profile.pdf. [Google Scholar]
- 8.New York City, Department of City Planning. Community district profiles: Manhattan community district 11. East Harlem: [Accessed March 2, 2004]. http://www.nyc.gov/html/dcp/pdf/lucds/mn11profile.pdf. [Google Scholar]
- 9.New York City, Department of Health and Mental Hygiene. Community health profiles. Central Harlem: [Accessed December 12, 2003]. http://www.nyc.gov/html/doh/pdf/data/2003nhp-manhattana.pdf. [Google Scholar]
- 10.New York City, Department of Health and Mental Hygiene. Community health profiles. East Harlem: [Accessed December 12, 2003]. http://www.nyc.gov/html/doh/pdf/data/2003nhp-manhattana.pdf. [Google Scholar]
- 11.Konstam MA, Dracup K, Baker DW, et al. Heart failure: evaluation and care of patients with left-ventricular systolic dysfunction. Rockville: U.S: Agency for Health Care Policy and Research, Department of Health and Human Services; Jun, 1994. [Google Scholar]
- 12.Packer M, Cohn N Steering Committee and Membership of the Advisory Council to Improve Outcomes Nationwide in Heart Failure. Consensus recommendations for the management of chronic heart failure. Am J Cardiol. 1999;83:1A–38A. [PubMed] [Google Scholar]
- 13.American College of Cardiology/American Heart Association Guidelines for the Evaluation and Management of Chronic Heart Failure in the Adult. A report of the ACC/AHA task force on practice guidelines (Committee to revise the 1995 guidelines for the evaluation and management of heart failure) 2001 [Google Scholar]
- 14.Corbie-Smith G, Thomas S, Williams M, et al. Attitudes and beliefs of African-Americans toward participation in medical research. J Gen Intern Med. 1999;14:537–546. doi: 10.1046/j.1525-1497.1999.07048.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Gorelick PB, Harris Y, Burnett M, et al. The recruitment triangle: reasons why African Americans enroll, refuse to enroll, or voluntarily withdraw from a clinical trial: an interim report from the African-American antiplatelet stroke prevention study (AAASPS) J Natl Med Assoc. 1998;90:141–145. [PMC free article] [PubMed] [Google Scholar]
- 16.Roberson NL. Clinical trial participation: viewpoints from racial/ethnic groups. Cancer. 1994;74(suppl 9):2687–2691. doi: 10.1002/1097-0142(19941101)74:9+<2687::aid-cncr2820741817>3.0.co;2-b. [DOI] [PubMed] [Google Scholar]
- 17.Shavers-Hornaday VL, Lynch CF, Burmeister LF, et al. Why are African Americans under-represented in medical research studies? impediments to participation. Ethn Health. 1997;2:31–45. doi: 10.1080/13557858.1997.9961813. [DOI] [PubMed] [Google Scholar]
- 18.Swanson GM, Ward AJ. Recruiting minorities into clinical trials: toward a participant-friendly system. J Natl Cancer Inst. 1995;87:1747–1759. doi: 10.1093/jnci/87.23.1747. [DOI] [PubMed] [Google Scholar]
- 19.Zhang J, Yu KF. What's the relative risk? a method of correcting the odds ratio in cohort studies of common outcomes. JAMA. 1998;280:1690–1691. doi: 10.1001/jama.280.19.1690. [DOI] [PubMed] [Google Scholar]
- 20.Senni M, Redfield MM. Heart failure with preserved systolic function: a different natural history? J Am Coll Cardiol. 2001;38:1277–1282. doi: 10.1016/s0735-1097(01)01567-4. [DOI] [PubMed] [Google Scholar]
- 21.Vasan RS, Larson MG, Benjamin EJ, et al. Congestive heart failure in subjects with normal versus reduced left ventricular ejection fraction. J Am Coll Cardiol. 1999;33:1948–1955. doi: 10.1016/s0735-1097(99)00118-7. [DOI] [PubMed] [Google Scholar]
- 22.McAlister FA, Lawson FME, Teo KK, et al. A systematic review of randomized trials of disease management programs in heart failure. Am J Med. 2001;110:378–384. doi: 10.1016/s0002-9343(00)00743-9. [DOI] [PubMed] [Google Scholar]
- 23.Naylor MD, Brooten D, Campbell R, et al. Comprehensive discharge planning and home follow-up of hospitalized elders: a randomized clinical trial. JAMA. 1999;281:613–620. doi: 10.1001/jama.281.7.613. [DOI] [PubMed] [Google Scholar]
- 24.Oddone EZ, Weinberger M, Giobbie-Hurder A, et al. Enhanced access to primary care for patients with congestive heart failure. Veterans affairs cooperative study group on primary care and hospital readmission. Eff Clin Pract. 1999;2:201–209. [PubMed] [Google Scholar]
- 25.Rich MW, Beckham V, Wittenberg C, et al. A multidisciplinary intervention to prevent the readmission of elderly people with congestive heart failure. N Engl J Med. 1995;333:1190–1195. doi: 10.1056/NEJM199511023331806. [DOI] [PubMed] [Google Scholar]
- 26.Feit F, Brooks MM, Sopko G, et al. BARI Investigators. Long-term clinical outcome in the bypass angioplasty revascularization investigation registry: comparison with the randomized trial. Circulation. 2000;101:2795–2802. doi: 10.1161/01.cir.101.24.2795. [DOI] [PubMed] [Google Scholar]