Abstract
Pancreatic ductal adenocarcinoma (PDAC) is one of the most aggressive cancers with a grim overall 5-year survival rate of 5%. Advances in surgical techniques, critical care, molecular diagnosis, diagnostic imaging, endosonology and adjuvant therapy have improved outcomes; but still more needs to be achieved. There is an urgent need to discover new avenues that may impact survival. Radiofrequency ablation (RFA) has attracted attention as an adjunctive treatment in PDAC. A review of English literature in PubMed was done using the MESH terms for PDAC and RFA. All the articles were reviewed and core information was tabulated for reference. After a comprehensive review of all articles the data was evaluated to discover the role of RFA in PDAC management. Indications, contraindications, feasibility, success rate, safety, complications and impact on survival were reviewed and are discussed further. RFA appears to be an attractive option for non-metastatic locally advanced PDAC. RFA is feasible but has a significant morbidity. At the present time the integration of RFA into the management of pancreatic ductal adenocarcinoma is evolving. It should be considered as having a complimentary role to current standard therapy in the multimodal management care model. It is likely that indications and patient selection for pancreatic RFA will expand.
Keywords: Pancreatic cancer, Radiofrequency ablation
Core tip: Radiofrequency ablation of pancreatic cancer is rapidly emerging as an attractive adjunct in locally advanced inoperable disease and is a part of modern multimodal hepatobiliary teams. Due to technological advances, refinements in thermokinetic principles and ongoing advances in medicinal oncology; it is likely that the role of radiofrequency in management of pancreatic cancer is going to increase in future. In this article we summarize the current evidence of application of radiofrequency ablation in pancreatic cancer.
INTRODUCTION
Pancreatic ductal adenocarcinoma is the commonest form of pancreatic cancer and is characterized by delayed diagnosis, aggressive tumour biology and dismal survival. At presentation, only 10% of the tumours are potentially curable[1]. Currently, surgery is the only curative treatment which provides long-term survival benefit for patients with pancreatic cancer[2,3]. The median survival of untreated patients is 3-4 mo and less than 5% of patients are alive one year after diagnosis[4]. The 5 years survival rate after a combination of resection and adjuvant therapy does not exceed 30%. Patients with locally advanced and inoperable disease have limited options[5]. Stagnation in surgical and oncological advances has challenged the medical community to explore alternative avenues. While molecular and genetic advances may have a future impact, thermal ablative techniques are increasingly being explored since last decade.
RADIOFREQUENCY ABLATION OF PANCREAS
Principles
Radiofrequency ablation (RFA) is the commonest thermal ablative technique used to treat solid abdominal organ tumours. Apart from the thermal destructive effect of RFA, secondary anticancer immunity due to activation of tumour-specific T lymphocytes appears to play a role[6]. Increasing evidence suggests that RFA might stimulate anti-tumour immunity through an alternative pathway by inducing expression of heat shock protein 70[7].
The past
First animal application of pancreas RFA was done in 1999[8]. However, due to retroperitoneal location, distal bile duct traversing head of pancreas, proximity to major vascular structures and close relation to duodenum and stomach were the major hurdles which curtailed the widespread acceptance of RFA. The increased risk of thermal injury during RFA of pancreatic ductal adenocarcinoma also relates to its diffuse nature and vessel encasement[9]. Earlier reports of RFA of pancreatic adenocarcinoma quoted severe complications with unacceptable mortality[10]. Some serious complications of RFA of pancreas include gastro-intestinal haemorrhage, pancreatic fistula, biliary leak, portal vein thrombosis, pancreatic pseudocyst and sepsis[11,12].
The present
Thermokinetic principles: It was the systematic efforts of Manchester group that helped define and validate the thermokinetic principles[13]. Although the ideal temperature for optimal thermal ablation of the pancreatic adenocarcinoma has been validated in experimental model there is still lack of consensus on the optimal RFA parameters and standardization of operative technique[13]. In a porcine experiment, Fegrachi et al[14] has recommended a probe distance of 10 mm from duodenum and portomesenteric vessels along with continuous duodenal cooling with 100 mL/min saline at 5 °C[14]. Using these settings in six animals, they did not encounter major morbidity and there was no mortality at two weeks. The same group has also demonstrated that duodenal cooling does not affect the ablation efficacy[15]. Performing concomitant biliary and gastric bypass procedures can reduce some complications[9]. RFA of the distal pancreas cancer may be performed without duodenal cooling as the bile duct and duodenum are some distance away. Figure 1 shows general principles underlying the application of RFA in pancreatic lesions.
Figure 1.
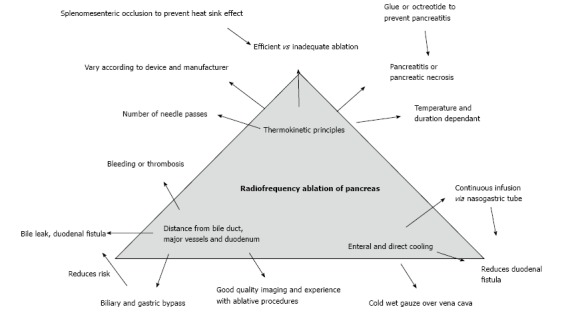
Principles of pancreatic radiofrequency ablation.
Technical approaches: The pancreas can be accessed directly by an open laparotomy, endosco-pically via transgastric or transduodenal approach and percutaneously by a posterior retroperitoneal approach. Endoscopic ultrasound guided RFA (EUS-RFA) appears attractive as it avoids surgery. In a study involving ten adult mini pigs, Kim et al[16] has demonstrated safety, feasibility and efficacy for pancreatic body and tail EUS-RFA. In a study involving five Yucatan pigs, Gaidhane et al[17] have demonstrated that EUS-RFA of pancreatic head was well tolerated with minimal pancreatitis. Pai et al[18] has reported EUS-RFA on eight patients with pancreatic cystic or neuroendocrine tumours with good results and acceptable safety profile. At the 2010 annual conference of International Hepatopancreaticobiliary Association, we presented a report of percutaneous RFA in a patient with local recurrence following a Whipple’s operation for a lower bile duct cholangiocarcinoma[19]. We performed duodenal cooling via a nasogastric tube and splenomesenteric occlusion to reduce heat sink effect. This patient survived for nine months after RFA.
Multimodal cancer care: RFA is increasingly recognized as an attractive adjunct treatment modality in reducing tumour burden and compliments other adjuvant therapies with potential for improved palliation. Although the effectiveness of RFA have been estimated by reductions in carbohydrate antigen 19-9, improvement of abdominal/back pain and/or non-progression of tumour on repeat interval imaging, such end points are surrogate measurements only. The desired endpoint is ultimately improvement in survival. RFA has shown to improve survival in patients with locally advanced inoperable pancreatic cancer[20,21]. Concomitant octreotide, antiproteases and chemotherapy (systemic or transarterial liver directed) or local application of radioactive seeds could also modify the clinical response. It is evident from the current reports that RFA should not be done in an obviously resectable pancreatic cancer or a metastatic disease. While RFA of pancreas cancer may not be worthwhile in this clinical context, RFA of liver metastases from pancreatic cancer have been attempted in the setting of multimodal approach. Park et al[22] have reported a retrospective review of RFA ablation for liver metastases from pancreatic ductal adenocarcinoma. They performed RFA on 34 patients over a period of seven years including patients with less than six liver lesions and size ≤ 3 cm and excluding patients with extrahepatic metastatic disease. Median survival time was 14 mo. Patients with oligometastatic disease showed improved survival after RFA compared to patients without liver metastases and no treatment. Huang et al[23] reported a median survival of 11 mo with transarterial chemoembolization plus RFA and/or 125I radioactive seed implantation on unresectable pancreatic cancer in a series of 71 patients. In this study the one-year survival was 32.4% for all patients and 25.5% for patients with liver metastases. Multiple case series of RFA application have been published and they generally testify its safety and feasibility. Table 1 provides details of thermokinetic principles applied by various authors and Table 2 summarizes outcomes with reference to survival and morbidity/mortality. RFA appears to have a role in treating locally advanced disease; however heterogeneity in the current reports makes it difficult to draw any robust recommendation about RFA applicability. RFA is being explored for improved palliation in malignant obstructive jaundice. Endobiliary RFA along with self-expanding metal stents is reported to be safe, feasible and associated with improved stent patency rates in patients with malignant biliary obstruction[33]. In the first in vivo study involving 22 patients with locally advanced pancreatic cancer, Arcidiacono et al[34] demonstrated feasibility and safety of endoscopic ultrasound guided cryothermal ablation with technical success in 16 patients (72.8%) and median post-ablation survival of 6 mo. They described late complications of jaundice, duodenal stricture and cystic fluid collection in four patients. Keane et al[35] conducted a systematic review on novel ablative methods in locally advanced pancreatic cancer and concluded that despite proven safety, feasibility and reproducibility; the benefit of ablative techniques on long term survival remains to be confirmed in large prospective randomized studies. Figure 2 shows the past, the present and the future of RFA application in pancreatic cancer.
Table 1.
Case series on radiofrequency ablation of pancreatic ductal adenocarcinoma-themokinetic principles
| Ref. | n | Age (yr) | Tumour size (cm) | Thermokinetics |
| Matsui et al[24] | 20 | 59 | 5.3 | 15 min at 50 °C in 2 × 2 × 2 cc field |
| Date et al[25] | 1 | 58 | 3 | RITA probe, 90 °C for 10 min each |
| Hadjicostas et al[26] | 4 | 70 | 8.5 (3-12) | Cooltip© RFA for shorter duration of 2-8 min with 17-gauge electrode |
| Varshney et al[27] | 3 | 58 | 6.5 | 4200 W of energy was delivered using a saline perfused needle with the aim of producing a 3 cm diameter necrosis |
| Wu et al[28] | 16 | 67 | 51 | Cooltip© RFA probe with up to 200 W energy, 12 min and tip temperature < 30 °C. A 5 mm safe distance between probe and major vessel |
| Spiliotis et al[20] | 12 | 67 | 3.5 | Cooltip© 17-gauge RFA electrode which achieved 80-90 °C. Cooltip© at < 10 min each |
| Casadei et al[29] | 3 | 66 | 4.7 | Cooltip© ablation at 90 °C for 5 min each |
| Girelli et al[11] | 50 | 65 | 4 | RITA system was used. Initial temperature of 105 °C (first 25 patients) was reduced to 90 °C after interim review |
| Zou3 et al[30] | 32 | 68 | 4-122 | 17 gauge electrode at 100-150 W energy with tip temperature of 90-100 °C for 12 min each After RFA, 125Iodine seed was implanted |
| Ikuta et al[31] | 1 | 60 | 4 | Cooltip© 17-gauge RFA electrode for 3-4 min each and a temperature of 99 °C |
82% tumors were > 5 cm;
Mean/median size not mentioned;
Simultaneous
I seed implantation. RFA: Radiofrequency ablation.
Table 2.
Case series on radiofrequency ablation of pancreatic ductal adenocarcinoma-outcomes and comments
| Ref. | Survival | Morbidity and mortality | Comments |
| Matsui et al[24] | 3 mo (median) | Morbidity (10%)-septic shock and gastrointestinal bleeding Mortality (5%)-patient with septic shock | All patients had a laparotomy |
| Date et al[25] | 3 mo (overall) | Patient developed polyuria. No major complication | Single patient |
| Hadjicostas et al[26] | 7 mo (median) | No major complications occurred | Sandostatin was administered prophylactically. Palliative bypass procedures were performed. One patient had significant pain relief |
| Varshney et al[27] | 7 mo (mean) | Self-limiting complications occurred in two patients | One patient had percutaneous CT guided RFA. All patients had endobiliary stenting All patients received 7 d of antibiotics |
| Later this group has updated their results in 10 patients with 10% morbidity and no mortality. Eight patients received post RFA chemotherapy. One patient developed a 2 cm pseudocyst. Overall survival range was 9-36 mo[32] | |||
| Wu et al[28] | Not reported | Pancreatic fistula 18.8% (3/16). Overall morbidity 43%. Mortality 25% Massive and mortal gastrointestinal bleeding occurred in 3 patients | Initially performed only for body and tail lesions. Later expanded for head of pancreas lesions, but had 50% mortality in this group 50% patients had relief of back pain 5 patients had liver metastases 5 mm distance to portal vein may not be safe |
| Spiliotis et al[20] | 33 mo (mean) | Overall morbidity 25% and nil mortality | Mean survival without RFA was 13 mo RFA in parallel to palliative therapy provided survival benefit for patients with unresectable pancreatic cancer |
| Casadei et al[29] | 4 mo (mean) | 3 patients developed ascites 1 patient developed biliary fistula | Prospective study. Included 3 patients Complete necrosis achieved in all patients All patients had a laparotomy and double bypass. Study was stopped at interim analysis |
| Girelli et al[11] | Not reported | Abdominal complications occurred in 24%. 30 d mortality 2%. Three patients with surgery related complicated required reoperation | Prospective study RFA was the only treatment in 19 patients All patients received antibiotics, octreotide and gabexate mesilate. Reduction of RFA temperature from 105 °C to 900 °C resulted in significant reduction in complications |
| Later this group has updated their experience of 107 patients (Cantore et al[21]). They performed a group wise comparison between upfront RFA vs RFA following primary therapy and concluded that RFA following primary treatment improves survival (14.7 mo vs 25.6 mo) | |||
| Zou1 et al[30] | 17.6 mo (mean) | Three patients experienced complications, but no mortality | Somatostatin analogues were used post-operatively The overall 12 mo survival was 65.6% |
| Ikuta et al[31] | Alive at 18 mo | No complications | Laparotomy with bypass procedure followed by chemoradiotherapy to induce pancreatic fibrosis. This was followed by second laparotomy and RFA |
Simultaneous 125I seed implantation. CT: Computerized tomography; RFA: Radiofrequency ablation.
Figure 2.
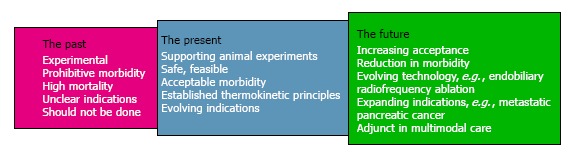
The past, the present and the future of pancreatic radiofrequency ablation.
The future
At the present time the integration of RFA into the management of pancreatic ductal adenocarcinoma is evolving. It should be considered as having a complimentary role to current standard therapy in the multimodal management care model. It is likely that indications and patient selection for pancreatic RFA will expand.
Footnotes
P- Reviewer: Bradley EL, Fusai G, Ogura T, Tandon R S- Editor: Ji FF L- Editor: A E- Editor: Lu YJ
Conflict-of-interest: None to declare.
Open-Access: This article is an open-access article which was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution Non Commercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: http://creativecommons.org/licenses/by-nc/4.0/
Peer-review started: October 5, 2014
First decision: November 3, 2014
Article in press: December 31, 2014
References
- 1.Warshaw AL, Fernández-del Castillo C. Pancreatic carcinoma. N Engl J Med. 1992;326:455–465. doi: 10.1056/NEJM199202133260706. [DOI] [PubMed] [Google Scholar]
- 2.Wagner M, Redaelli C, Lietz M, Seiler CA, Friess H, Büchler MW. Curative resection is the single most important factor determining outcome in patients with pancreatic adenocarcinoma. Br J Surg. 2004;91:586–594. doi: 10.1002/bjs.4484. [DOI] [PubMed] [Google Scholar]
- 3.Singh SM, Longmire WP, Reber HA. Surgical palliation for pancreatic cancer. The UCLA experience. Ann Surg. 1990;212:132–139. doi: 10.1097/00000658-199008000-00003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Jemal A, Murray T, Ward E, Samuels A, Tiwari RC, Ghafoor A, Feuer EJ, Thun MJ. Cancer statistics, 2005. CA Cancer J Clin. 2005;55:10–30. doi: 10.3322/canjclin.55.1.10. [DOI] [PubMed] [Google Scholar]
- 5.Vulfovich M, Rocha-Lima C. Novel advances in pancreatic cancer treatment. Expert Rev Anticancer Ther. 2008;8:993–1002. doi: 10.1586/14737140.8.6.993. [DOI] [PubMed] [Google Scholar]
- 6.Dromi SA, Walsh MP, Herby S, Traughber B, Xie J, Sharma KV, Sekhar KP, Luk A, Liewehr DJ, Dreher MR, et al. Radiofrequency ablation induces antigen-presenting cell infiltration and amplification of weak tumor-induced immunity. Radiology. 2009;251:58–66. doi: 10.1148/radiol.2511072175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Teng LS, Jin KT, Han N, Cao J. Radiofrequency ablation, heat shock protein 70 and potential anti-tumor immunity in hepatic and pancreatic cancers: a minireview. Hepatobiliary Pancreat Dis Int. 2010;9:361–365. [PubMed] [Google Scholar]
- 8.Goldberg SN, Mallery S, Gazelle GS, Brugge WR. EUS-guided radiofrequency ablation in the pancreas: results in a porcine model. Gastrointest Endosc. 1999;50:392–401. doi: 10.1053/ge.1999.v50.98847. [DOI] [PubMed] [Google Scholar]
- 9.Siriwardena AK. Radiofrequency ablation for locally advanced cancer of the pancreas. JOP. 2006;7:1–4. [PubMed] [Google Scholar]
- 10.Elias D, Baton O, Sideris L, Lasser P, Pocard M. Necrotizing pancreatitis after radiofrequency destruction of pancreatic tumours. Eur J Surg Oncol. 2004;30:85–87. doi: 10.1016/j.ejso.2003.10.013. [DOI] [PubMed] [Google Scholar]
- 11.Girelli R, Frigerio I, Salvia R, Barbi E, Tinazzi Martini P, Bassi C. Feasibility and safety of radiofrequency ablation for locally advanced pancreatic cancer. Br J Surg. 2010;97:220–225. doi: 10.1002/bjs.6800. [DOI] [PubMed] [Google Scholar]
- 12.Pezzilli R, Ricci C, Serra C, Casadei R, Monari F, D’Ambra M, Corinaldesi R, Minni F. The problems of radiofrequency ablation as an approach for advanced unresectable ductal pancreatic carcinoma. Cancers (Basel) 2010;2:1419–1431. doi: 10.3390/cancers2031419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Date RS, Biggins J, Paterson I, Denton J, McMahon RF, Siriwardena AK. Development and validation of an experimental model for the assessment of radiofrequency ablation of pancreatic parenchyma. Pancreas. 2005;30:266–271. doi: 10.1097/01.mpa.0000153334.65729.a6. [DOI] [PubMed] [Google Scholar]
- 14.Fegrachi S, Molenaar IQ, Klaessens JH, Besselink MG, Offerhaus JA, van Hillegersberg R. Radiofrequency ablation of the pancreas: two-week follow-up in a porcine model. Eur J Surg Oncol. 2014;40:1000–1007. doi: 10.1016/j.ejso.2013.11.001. [DOI] [PubMed] [Google Scholar]
- 15.Fegrachi S, Molenaar IQ, Klaessens JH, Besselink MG, Offerhaus JA, van Hillegersberg R. Radiofrequency ablation of the pancreas with and without intraluminal duodenal cooling in a porcine model. J Surg Res. 2013;184:867–872. doi: 10.1016/j.jss.2013.04.068. [DOI] [PubMed] [Google Scholar]
- 16.Kim HJ, Seo DW, Hassanuddin A, Kim SH, Chae HJ, Jang JW, Park do H, Lee SS, Lee SK, Kim MH. EUS-guided radiofrequency ablation of the porcine pancreas. Gastrointest Endosc. 2012;76:1039–1043. doi: 10.1016/j.gie.2012.07.015. [DOI] [PubMed] [Google Scholar]
- 17.Gaidhane M, Smith I, Ellen K, Gatesman J, Habib N, Foley P, Moskaluk C, Kahaleh M. Endoscopic Ultrasound-Guided Radiofrequency Ablation (EUS-RFA) of the Pancreas in a Porcine Model. Gastroenterol Res Pract. 2012;2012:431451. doi: 10.1155/2012/431451. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Pai M, Senturk H, Lakhtakia S, Reddy DN, Cicinnati V, Kabar I, Beckebaum S, Jin Z, Wang D, Yang J, et al. Endoscopic Ultrasound Guided Radiofrequency Ablation (EUS-RFA) for Cystic Neoplasms and Neuroendocrine Tumours of the Pancreas. Gastrointest Endosc. 2013;77:AB143–AB144. [Google Scholar]
- 19.Shelat VG, JK Low, W Woon. Radiofrequency ablation of pancreatic cancer-overview. HPB. 2012;14(Suppl. 2):107–287. [Google Scholar]
- 20.Spiliotis JD, Datsis AC, Michalopoulos NV, Kekelos SP, Vaxevanidou A, Rogdakis AG, Christopoulou AN. Radiofrequency ablation combined with palliative surgery may prolong survival of patients with advanced cancer of the pancreas. Langenbecks Arch Surg. 2007;392:55–60. doi: 10.1007/s00423-006-0098-5. [DOI] [PubMed] [Google Scholar]
- 21.Cantore M, Girelli R, Mambrini A, Frigerio I, Boz G, Salvia R, Giardino A, Orlandi M, Auriemma A, Bassi C. Combined modality treatment for patients with locally advanced pancreatic adenocarcinoma. Br J Surg. 2012;99:1083–1088. doi: 10.1002/bjs.8789. [DOI] [PubMed] [Google Scholar]
- 22.Park JB, Kim YH, Kim J, Chang HM, Kim TW, Kim SC, Kim PN, Han DJ. Radiofrequency ablation of liver metastasis in patients with locally controlled pancreatic ductal adenocarcinoma. J Vasc Interv Radiol. 2012;23:635–641. doi: 10.1016/j.jvir.2012.01.080. [DOI] [PubMed] [Google Scholar]
- 23.Huang ZM, Pan CC, Wu PH, Zhao M, Li W, Huang ZL, Yi RY. Efficacy of minimally invasive therapies on unresectable pancreatic cancer. Chin J Cancer. 2013;32:334–341. doi: 10.5732/cjc.012.10093. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Matsui Y, Nakagawa A, Kamiyama Y, Yamamoto K, Kubo N, Nakase Y. Selective thermocoagulation of unresectable pancreatic cancers by using radiofrequency capacitive heating. Pancreas. 2000;20:14–20. doi: 10.1097/00006676-200001000-00002. [DOI] [PubMed] [Google Scholar]
- 25.Date RS, Siriwardena AK. Radiofrequency ablation of the pancreas. II: Intra-operative ablation of non-resectable pancreatic cancer. A description of technique and initial outcome. JOP. 2005;6:588–592. [PubMed] [Google Scholar]
- 26.Hadjicostas P, Malakounides N, Varianos C, Kitiris E, Lerni F, Symeonides P. Radiofrequency ablation in pancreatic cancer. HPB (Oxford) 2006;8:61–64. doi: 10.1080/13651820500466673. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Varshney S, Sewkani A, Sharma S, Kapoor S, Naik S, Sharma A, Patel K. Radiofrequency ablation of unresectable pancreatic carcinoma: feasibility, efficacy and safety. JOP. 2006;7:74–78. [PubMed] [Google Scholar]
- 28.Wu Y, Tang Z, Fang H, Gao S, Chen J, Wang Y, Yan H. High operative risk of cool-tip radiofrequency ablation for unresectable pancreatic head cancer. J Surg Oncol. 2006;94:392–395. doi: 10.1002/jso.20580. [DOI] [PubMed] [Google Scholar]
- 29.Casadei R, Ricci C, Pezzilli R, Serra C, Calculli L, Morselli-Labate AM, Santini D, Minni F. A prospective study on radiofrequency ablation locally advanced pancreatic cancer. Hepatobiliary Pancreat Dis Int. 2010;9:306–311. [PubMed] [Google Scholar]
- 30.Zou YP, Li WM, Zheng F, Li FC, Huang H, Du JD, Liu HR. Intraoperative radiofrequency ablation combined with 125 iodine seed implantation for unresectable pancreatic cancer. World J Gastroenterol. 2010;16:5104–5110. doi: 10.3748/wjg.v16.i40.5104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Ikuta S, Kurimoto A, Iida H, Aihara T, Takechi M, Kamikonya N, Yamanaka N. Optimal combination of radiofrequency ablation with chemoradiotherapy for locally advanced pancreatic cancer. World J Clin Oncol. 2012;3:12–14. doi: 10.5306/wjco.v3.i1.12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Singh V, Varshney S, Sewkani A, Varshney R, Deshpande G, Shaji P, Jat A. Radiofrequency ablation of unresectable pancreatic carcinoma: 10-year experience from single centre. Pancreatology. 2011;11(Suppl 1):52. [Google Scholar]
- 33.Steel AW, Postgate AJ, Khorsandi S, Nicholls J, Jiao L, Vlavianos P, Habib N, Westaby D. Endoscopically applied radiofrequency ablation appears to be safe in the treatment of malignant biliary obstruction. Gastrointest Endosc. 2011;73:149–153. doi: 10.1016/j.gie.2010.09.031. [DOI] [PubMed] [Google Scholar]
- 34.Arcidiacono PG, Carrara S, Reni M, Petrone MC, Cappio S, Balzano G, Boemo C, Cereda S, Nicoletti R, Enderle MD, et al. Feasibility and safety of EUS-guided cryothermal ablation in patients with locally advanced pancreatic cancer. Gastrointest Endosc. 2012;76:1142–1151. doi: 10.1016/j.gie.2012.08.006. [DOI] [PubMed] [Google Scholar]
- 35.Keane MG, Bramis K, Pereira SP, Fusai GK. Systematic review of novel ablative methods in locally advanced pancreatic cancer. World J Gastroenterol. 2014;20:2267–2278. doi: 10.3748/wjg.v20.i9.2267. [DOI] [PMC free article] [PubMed] [Google Scholar]


