Abstract
Background
NOX‐2, the catalytic subunit of NADPH oxidase, has a key role in the formation of reactive oxidant species and is implicated in impairing flow‐mediated dilation (FMD). Dark chocolate exerts artery dilatation via down‐regulating NOX2‐mediated oxidative stress. The aim of this study was to investigate whether dark chocolate improves walking autonomy in peripheral artery disease (PAD) patients via an oxidative stress‐mediated mechanism.
Methods and Results
FMD, serum levels of isoprostanes, nitrite/nitrate (NOx) and sNOX2‐dp, a marker of blood NOX2 activity, maximal walking distance (MWD) and maximal walking time (MWT) were studied in 20 PAD patients (14 males and 6 females, mean age: 69±9 years) randomly allocated to 40 g of dark chocolate (>85% cocoa) or 40 g of milk chocolate (≤35% cocoa) in a single blind, cross‐over design. The above variables were assessed at baseline and 2 hours after chocolate ingestion. Dark chocolate intake significantly increased MWD (+11%; P<0.001), MWT (+15%; P<0.001), serum NOx (+57%; P<0.001) and decreased serum isoprostanes (−23%; P=0.01) and sNOX2‐dp (−37%; P<0.001); no changes of the above variables were observed after milk chocolate intake. Serum epicatechin and its methylated metabolite significantly increased only after dark chocolate ingestion. Multiple linear regression analysis showed that Δ of MWD was independently associated with Δ of MWT (P<0.001) and Δ of NOx (P=0.018). In vitro study demonstrated that HUVEC incubated with a mixture of polyphenols significantly increased nitric oxide (P<0.001) and decreased E‐selectin (P<0.001) and VCAM1 (P<0.001).
Conclusion
In PAD patients dark but not milk chocolate acutely improves walking autonomy with a mechanism possibly related to an oxidative stress‐mediated mechanism involving NOX2 regulation.
Clinical Trial Registration
URL: http://www.clinicaltrials.gov. Unique identifier: NCT01947712.
Keywords: antioxidant, atherosclerosis, chocolate, oxidant stress, peripheral vascular disease
Introduction
Peripheral arterial disease (PAD) represents a widespread atherosclerotic condition.1 More than 20% of adults older than 70 are affected by this disease in Western countries.2 It is characterized by a high rate of coronary and cerebral events.2 Intermittent Claudication (IC), the typical clinical manifestation of the disease, is identified by an impaired blood flow to the limbs during physical exercise.2 The atherosclerotic process in lower extremities of PAD patients is not confined to conduit vessels but also to microcirculation.3 Thus, a pivotal role in the onset of IC is played by resistant arteries, arterioles and capillaries that serve the skeletal muscle tissue distal to the site of stenosis.3–4 Endothelial dysfunction, reduced glucose oxidation, accumulation of toxic metabolites, impaired nitric oxide (NO) generation and oxidative stress seem to play a role2,4–7 among the factors contributing to reduce blood flow in PAD.
Previous study with antioxidant infusion provided evidence that oxidative stress is implicated in impairing walking distance autonomy (WDA) while its inhibition was associated with maximal walking distance (MWD) improvement.8
Cocoa is a polyphenol‐rich nutrient which elicits artery dilatation by reducing oxidative stress and increasing nitric oxide generation.9–10 In particular, dark chocolate is able to enhance artery dilatation via lowering activation of NOX2, the catalytic subunit of nicotinamide‐adenine dinucleotide phosphate (NADPH) oxidase,9,11 which has been shown in human as well in animal models to exert a vasoconstrictor activity.12–15
Our study hypothesis was that cocoa could improve WDA in PAD patients via an oxidative stress‐mediated mechanism. Thus, we performed an interventional study in which we measured the acute effect of dark chocolate on WDA, artery dilatation and NOX2‐mediated oxidative stress in a population affected by moderate‐severe PAD.
Materials and Methods
We performed an interventional trial in PAD patients to investigate the acute effect of 40 g of chocolate (dark versus milk), 2 hours after the assumption, on MWD, maximal walking time (MWT), ankle brachial index (ABI) at rest and post exercise, FMD, oxidative stress, as assessed by blood levels of soluble NOX2‐derivative peptide (sNOX2‐dp), a marker of NOX2 activation, serum isoprostanes and NO generation, as assessed by serum levels of nitrite/nitrate (NOx).
Twenty PAD patients at Fontaine stage IIb agreed to participate in the study, which was performed between January 2012 and September 2013. Patients had to be in a stable condition, without abrupt changes of walking distance and ABI in the month before the study.
They were randomly allocated to a treatment sequence with 40 g of dark chocolate (≥85% cocoa) or milk chocolate (≤35% cocoa) in a cross‐over, single‐blind design. There was at least 1 week washout between the 2 phases of the study. FMD, oxidative stress, serum levels of NOx and epicatechin (EC) were assessed at baseline, after a 24 hours abstinence from food rich in polyphenols, and 2 hours after ingestion of chocolate.
The schedule of the procedure was the following:
8:00 am: first blood samples (to analyze oxidative stress and epicatechin levels) collected after a fasting period of 8 hours.
8:15 am: first ABI at rest and FMD were performed.
9:00 am: first treadmill test was executed; 2 minutes after the measurement of the MWD and MWT, postexercise ABI was performed.
9:25 am: participants in the study received 40 g of chocolate (dark or milk); they had 15 minutes to eat chocolate.
11:25 am: second blood samples (to analyze oxidative stress and epicatechin levels) were collected.
11:30 am: second ABI at rest and FMD were performed.
11:50 am: second treadmill test was performed; 2 minutes after the measurement of the MWD and MWT, another postexercise ABI was performed.
No beverages were permitted during this period.
All subjects underwent a full medical history and physical examination. Subjects were excluded from the study if they had liver insufficiency, serious renal disorders (serum creatinine [mt]2.8 mg/dL), acute cerebrovascular disease, acute myocardial infarction, or if they were current smokers or taking antioxidants. All the participants in the study received a questionnaire to evaluate their fruit and vegetable intake.16
The number of PAD patients initially assessed for inclusion into the study was 32; after initial assessments 4 patients were excluded from the study for having serum creatinine [mt]2.8 mg/dL, 5 patients for current smoking, and 3 patients refused to participate in the study. No dropouts or missing data have been observed during the study.
Informed written consent was obtained from all subjects: the study was conformed to the ethical guidelines of the 1975 Declaration of Helsinki and was approved by the Ethical Committee of Sapienza University (ClinicalTrials.gov Identifier: NCT01947712).
Study Outcomes
The primary outcome of this study was to evaluate the effect of chocolate on endothelial function by FMD and on walking distance in PAD patients.
Randomization and Blinding
An individual not involved in the study, assigned codes to the study treatments, randomly allocated the participants to a treatment sequence with dark or milk chocolate, and kept the key in a sealed envelope. The randomization was carried out by a procedure based on a random numeric sequence. The authors and laboratory technicians were unaware of the treatment allocation.
ABI Measurement
ABI was calculated with the patient placed in the supine position, measuring the higher systolic pressure of the anterior or posterior tibial artery in each limb and dividing this pressure with the highest brachial systolic pressure.17
In patients with diabetes, we performed toe pressure determinations if ABI>1.3.
Treadmill Test
Treadmill test was performed using a treadmill speed of 3.5 km/h at 12% grade until maximal claudication pain.
FMD Measurements
FMD measurement was assessed as previously described.12
Serum Prostaglandin F2α‐III Assays
Quantification of isoprostanes was performed to measure serum prostaglandin F2α‐III by a previously described and validated enzyme immunoassay method.18 Intra‐assay and interassay coefficients of variation were 5.8% and 5.0%, respectively.
Analysis of Serum NOx
NO was measured in serum by measurement of metabolic end products ie, nitrite and nitrate (NOx) (Tema Ricerca). Intra‐ and inter‐assay coefficients of variation were 2.9% and 1.7%, respectively.
ELISA Detection of sNOX2‐dp
NOX2‐ derived peptide, a marker of NADPH oxidase activation, was detected in serum by the ELISA method as previously described by Pignatelli et al.19 The peptide was recognized by the specific monoclonal antibody against the amino acidic sequence (224 to 268) of the extramembrane portion of NOX2. Values were expressed as pg/mL, and intra‐assay and interassay coefficients of variation were 5.2% and 6%, respectively.
Serum Polyphenols/Metabolites Evaluation by High‐Performance Liquid Chromatography
For high‐performance liquid chromatography analysis, serum samples were prepared according to Schroeter et al.20 Briefly, samples were mixed with twice their volume of acidified methanol (100% vol/vol, −20°C, internal standard=3′ethylEC) and centrifuged at 17 000g for 15 minutes at 4°C. The supernatant was collected and the pellet was resuspended twice in methanol and centrifuged at 17 000g for 15 minutes at 4°C. After centrifugation, the supernatant was collected and the solvents were evaporated to dryness under nitrogen.
The determination of epicatechin, its metabolite EC‐3‐O‐methylether, catechin, and epigallocatechin‐3‐gallate was carried out using an Agilent 1200 Infinity series high‐performance liquid chromatography system equipped with an Eclipse plus C18 column (4.6 × 100 mm). All determinations were undertaken at 25°C. Separation of analytes was accomplished by using an acetonitrile gradient in 50 mmol/L methanolic (4% vol/vol) sodium acetate (pH 4.4) with a flow rate of 0.8 mL/min. Acetonitrile concentrations were linearly increased from 0% to 10% between minutes 5 and 20. Thereafter, acetonitrile concentrations were further increased in linear segments (20 to 28 minutes, 10% to 12%; 28 to 34 minutes, 12% to 20%; 34 to 41 minutes, 20% to 30%; 41 to 45 minutes, 30% to 71%) and held at 71% for 10 minutes. We used UV detection at 280 nm for sample analysis.
Chocolate Total Polyphenol Content
Chocolate was extracted by a modified method according to Smith.21 Total polyphenol content in dark and milk chocolate was determined by a modified Folin–Ciocalteu colorimetric method.22 Briefly, 0.1 mL of Folin–Ciocalteu reagent was added to 0.02 mL of dark or milk chocolate. After 5 minutes at room temperature, 0.05 mL of 20 g of sodium carbonate per milliliter was added and the reaction mixture was incubated at 37°C for 20 minutes. The absorbance at 765 nm was measured using an Asys UVM 340 microplate reader (Biochrom) and compared with a gallic‐acid calibration curve (100 to 1000 mg L−1). Results were expressed as mg gallic acid equivalent per kilogram. All experiments were performed in triplicate.
Human Umbilical Vein Endothelial Cells (HUVEC)
Human umbilical vein endothelial cells (HUVEC) were cultured as previously described.23 Briefly, cells were expanded (2000 cells/cm2) in complete medium (Endogro‐LS complete media kit, Millipore). Cell morphology and growth were monitored by light microscopy and assessed by Trypan Blue (Sigma). The culture was expanded until passage 5.
In Vitro Study
In vitro study was performed in HUVEC cells. We analyzed the effect of scalar doses of single polyphenols such as epicatechin (0.1 to 10 μmol/L), catechin (0.1 to 10 μmol/L), or epigallocatechin‐3‐gallate (0.1 to 10 μmol/L) or a mixture of them on HUVEC activation in 5 separate experiments.
HUVEC were incubated 60 minutes at 37°C with polyphenols and stimulated for 10 minutes with endothelial growth factor (10 ng/mL). After 60 minutes of incubation, supernatants were removed by gentle washing. Afterwards, cultures were left in an incubator for 2 hours with basal medium and harvested by trypsin. Then, HUVEC culture supernatants were collected for the analysis of soluble vascular adhesion molecule‐1, sE‐selectin, and NOx. The enzyme immunoassay method (Tema Ricerca) was used to determine the soluble vascular adhesion molecule‐1 and sE‐selectin concentration. Values were expressed as ng/mL; intra‐assay and interassay coefficients of variation were 5.2% and 6%, respectively. Concentration of NOx in the supernatant was assessed as reported above.
Sample Size Determination
Since the primary outcome of this study was to evaluate the effect of chocolate on endothelial function by FMD in PAD patients, we computed the minimum sample size with respect to a 2‐tailed, 1‐sample Student t test with Welch correction, considering, on the basis of data from a previous pilot study (data not shown): (1) a difference for FMD variation in PAD to be detected between the groups after dark and milk chocolate treatments δ: 3%; (2) SD of the paired differences: 3.0%; (3) type I error probability α: 0.05 and power 1‐β: 0.90. FMD was expressed as a change in poststimulus diameter and evaluated as a percentage increase of the baseline diameter; FMD variation was defined as continuous measure. This resulted in n=13 patients, which was increased to n=20.
Statistical Methods
Continuous variables are reported as mean±SD unless otherwise indicated. The crossover study data were analyzed for the assessment of treatment and period effects, by performing a split‐plot ANOVA with one between‐subject factor (treatment sequence) and 2 within‐subject factors (period 1 versus 2; pre‐ versus post‐treatment). The full model was considered, allowing for the assessment of all main effects and interactions. Pairwise comparisons were corrected by the Bonferroni correction factor; the results of paired differences were replicated and confirmed by appropriate nonparametric tests (Wilcoxon test). Bivariate analysis was performed with the Spearman linear regression test. Multiple linear regression analysis was performed using a forward selection. A value of P<0.05 was considered statistically significant. All analyses were carried out with SPSS 18.0 software for Windows (SPSS, Chicago, IL).
Results
Clinical characteristics of PAD patients are reported in Table 1. Total and single polyphenol content was significantly higher in dark compared to milk chocolate (Table 2) (P<0.001).
Table 1.
Clinical Characteristics of PAD Patients
| Variables | PAD (n=20) |
|---|---|
| Mean age, y* | 69±9 |
| Males/females | 14/6 |
| Hypertension, % (n) | 85% (17) |
| Diabetes mellitus, % (n) | 30% (6) |
| Dyslipidemia, % (n) | 90% (18) |
| Former smokers, % (n) | 80% (16) |
| CHD | 40% (8) |
| Previous stroke | 15% (3) |
| BMI* | 27±3 |
| Glycemia, mg/dL* | 114±23 |
| Pharmacological treatments, % (n) | |
| ACE‐inhibitors | 60% (12) |
| Oral antidiabetic drugs | 20% (4) |
| Insulin | 20% (4) |
| Statin | 100% (20) |
| Antiplatelets | 95% (19) |
| Oral anticoagulants | 5% (1) |
ACE indicates angiotensin‐converting enzyme; BMI, body–mass index; CHD, coronary heart disease; PAD, peripheral artery disease.
Data are expressed as mean±SD.
Table 2.
Total Polyphenols Content in Dark and Milk Chocolate
| Compounds | Dark | Milk | P Value |
|---|---|---|---|
| Total polyphenols, mg/L GAE | 799 | 296 | <0.001 |
| Epicatechin, μg/mL | 0.59 | 0.16 | <0.001 |
| Catechin, μg/mL | 0.32 | 0.13 | <0.001 |
| EGCG, μg/mL | 1.8 | 0.28 | <0.001 |
EGCG indicates epigallocatechin‐3‐gallate; GAE, gallic acid equivalent.
Compared to baseline, there was no difference of serum epicatechin and its metabolite EC‐3‐O‐methylether (Figure 1A) and epigallocatechin‐3‐gallate (Figure 1B) levels 2 hours after milk chocolate ingestion; conversely, a significant increase of serum catechin was detected (Figure 1B). Compared to baseline, serum levels of epicatechin and its metabolite EC‐3‐O‐methylether (Figure 1A), catechin (Figure 1B), and epigallocatechin‐3‐gallate (Figure 1B) increased 2 hours after dark chocolate intake. A chromatogram representative of serum epicatechin and catechin before and after dark chocolate intake is reported in Data S1.
Figure 1.
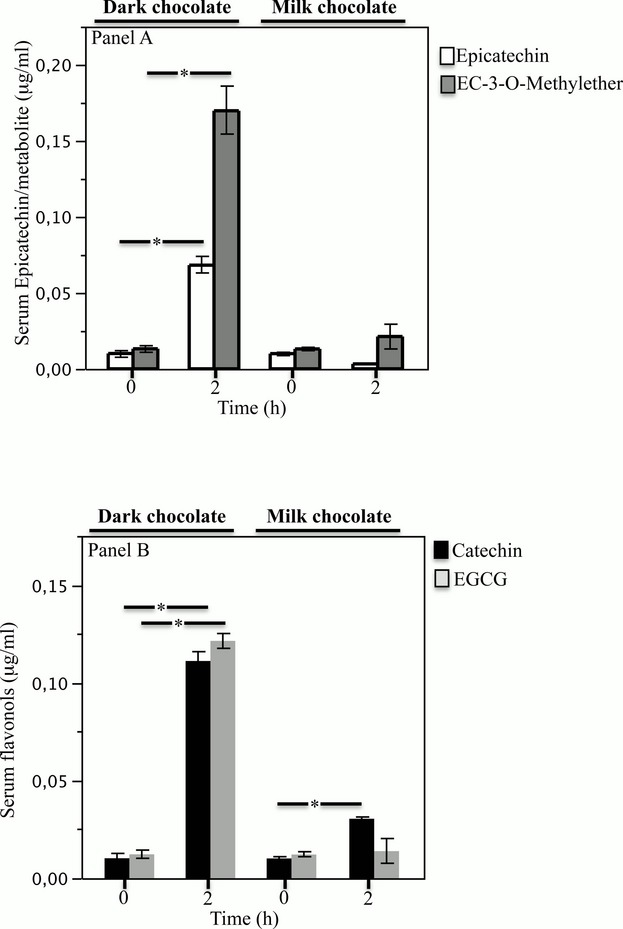
Serum epicatechin and EC‐3‐O‐methylether (A) determination at baseline and after 2 hours of dark or milk chocolate intake. Serum catechin and epigallocatechin‐3‐gallate (EGCG) (B) determination at baseline and after 2 hours of dark or milk chocolate intake (n=20) (*P[lt]0.001). Data are expressed as mean±SE.
A significant difference for treatments (dark versus milk chocolate) was found with respect to FMD (P=0.003; Figure 2A), sNOX2‐dp release (P=0.04; Figure 2B), Knox (P=0.03; Figure 2C), serum 8‐iso‐prostaglandin F2α‐III (P=0.018; Figure 2D), MWD (P=0.01; Figure 2A), Maximal Walking Time (MWT) (P=0.006; Figure 2B), and postexercise ABI (P=0.04; Figure 2D) from the ANOVA performed on crossover study data.
Figure 2.
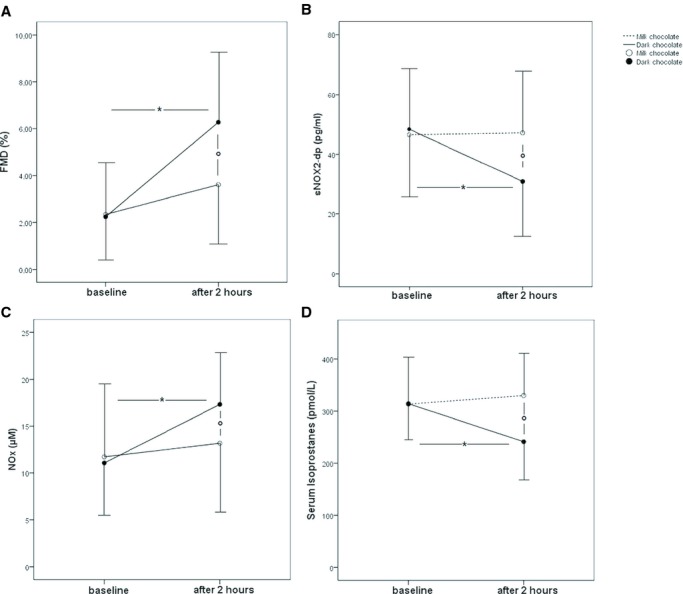
Flow‐mediated dilatation (FMD) (A), serum soluble NOX2‐derived peptide (sNOX2‐dp) (B), serum nitrite/nitrate (Knox) levels (C), and serum isoprostanes (D) before and 2 hours after intake of dark or milk chocolate in peripheral artery disease (PAD) patients (n=20). Data are expressed as mean±SD. *P[lt]0.05 for within‐groups analysis; °P[lt]0.05 for between‐groups analysis. NOX indicates nitrite/nitrate.
Compared to baseline, MWD and MWT increased after dark chocolate intake (from 110.7±64.5 to 122.2±61.5 m, P<0.001, and from 124.8±60.8 to 142.2±62.0 seconds, P<0.001, respectively) but not after milk chocolate intake (from 115.8±71.9 to 109.1±65.1 m, P=0.231, and from 124.5±60.1 to 125.4±64.1 seconds, P=0.783, respectively) (Figure 3A and 3B). Conversely, no significant effect was found after dark and milk chocolate intake for ABI at rest (Figure 3C). Furthermore, no significant difference in postexercise ABI was observed in the analysis within groups with the 2 treatments (Figure 3D).
Figure 3.
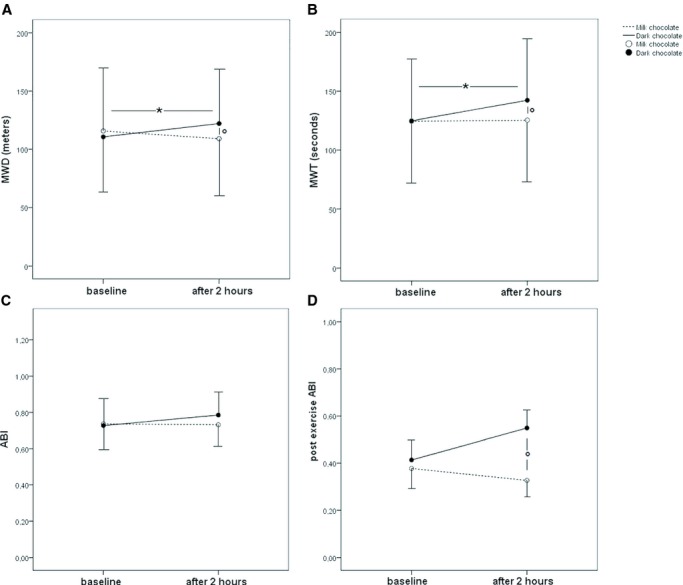
Maximal walking distance (MWD) (A), maximal walking time (MWT) (B), ankle brachial index (ABI) (C), and postexercise ABI (D) before and 2 hours after intake of dark or milk chocolate in peripheral artery disease patients (n=20). Data are expressed as mean±SD. *P[lt]0.05 for within‐groups analysis; °P[lt]0.05 for between‐groups analysis.
The pairwise comparisons showed that sNOX2‐dp and serum isoprostanes significantly decreased after dark chocolate (from 48.25±21.7 to 30.9±20.6 pg/mL, P<0.001, and from 314.2±86.6 to 241.3±75.4 pg/mL, P=0.01, respectively), while no changes were observed after milk chocolate (from 46.3±22.3 to 46.9±22.9 pg/mL, P=0.909, and from 313.5±57.5 to 330.0±79.8 pmol/L, P=0.482, respectively), (Figure 2B and 2D); conversely, FMD and Knox significantly increased after dark chocolate intake (from 2.3±2.2% to 6.3±2.7%, P<0.001 and from 11.0±5.8 to 17.3±5.8 μmol/L, P=0.001, respectively) (Figure 2A and 2C). No changes were observed after milk chocolate intake for FMD and Knox (from 2.3±2.5% to 3.6±3.2%, P=0.064, and from 11.7±7.8 to 13.1±7.57 μmol/L, P=0.353, respectively), (Figure 2A and 2C).
A linear correlation analysis showed that Δ (expressed by difference of values between before and after chocolate intake) of MWD correlated with Δ of sNOX2‐dp (r=−0.477; P=0.002), Δ of Knox (r=0.439; P=0.005), Δ of MWT (r=0.706; P[lt]0.001). Furthermore, Δ of sNOX2‐dp correlated with Δ of FMD (r=−0.399; P=0.01) and Δ of Knox (r=−0.469; P=0.002). Multiple linear regression analysis showed that the only independent predictive variables associated with Δ MWD in PAD patients were Δ of MWT (SE=0.148; standardized coefficient β=0.518; P<0.001) and Δ of Knox (SE=0.043; standardized coefficient β=0.315; P=0.018), with R2=41.7%.
In Vitro Study
We performed an in vitro study with a mixture of polyphenols composed of epicatechin (0.1 to 10 μmol/L), catechin (0.1 to 10 μmol/L), and epigallocatechin‐3‐gallate (0.1 to 10 μmol/L) at the concentration relatively similar to that found in serum of patients after dark chocolate ingestion.
HUVEC stimulated with endothelial growth factor released a higher amount of soluble vascular adhesion molecule‐1 (Figure 4A, P<0.0001), E‐selectin (Figure 2B, *P<0.0001) and a decreased amount of Knox production (Figure 4C, P<0.0001) versus unstimulated HUVEC.
Figure 4.
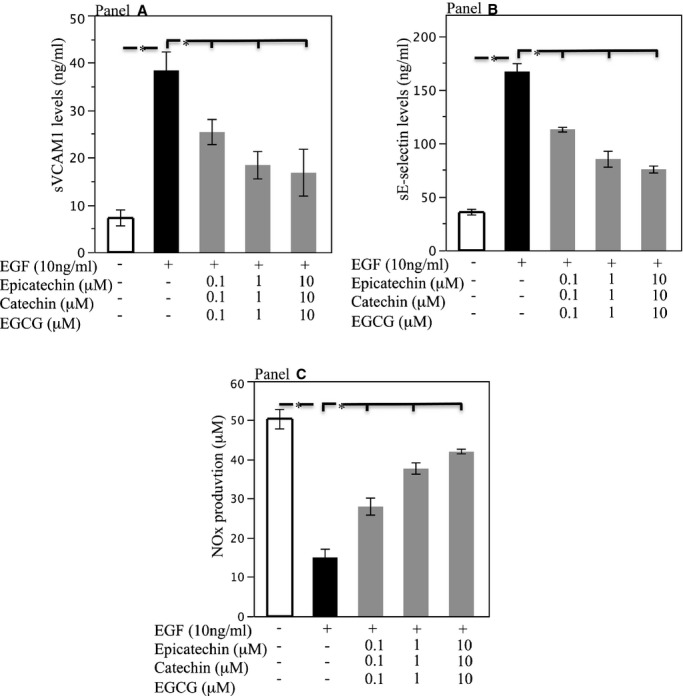
Soluble vascular adhesion molecule‐1 (A), sE‐selectin (B), and Knox (C) levels released by HUVEC after stimulation with EGF (black bar), and after incubation with scalar doses of mixture of polyphenols composed of epicatechin (0.1 to 10 μmol/L), catechin (0.1 to 10 μmol/L), and epigallocatechin‐3‐gallate (EGCG) (0.1 to 10 μmol/L) (light gray bars), (n=5) (*P[lt]0.001). Data are expressed as mean±SE. EGF indicates endothelial growth factor; HUVEC, human umbilical vein endothelial cells; Knox, nitrite/nitrate.
In HUVEC pre‐incubated with single polyphenols, no changes of the above variables were detected (not shown). Conversely, endothelial growth factor–stimulated HUVEC incubated with a mixture of polyphenols showed significant reduction of soluble vascular adhesion molecule‐1 and sE‐selectin levels and an increase of Knox (Figure 4A through 4C). Significant changes of these variables were already observed with concentrations of single polyphenols detected in serum 2 hours after dark chocolate intake.
Discussion
This study provides the first evidence that short‐term intake of dark chocolate by PAD patients is associated with a significant increase of walking autonomy, an effect that may be related to a downregulation of NOX2‐mediated oxidative stress.
The novel finding of the present study is the improvement of MWD and MWT after 2 hours by ingestion of dark but not milk chocolate in PAD patients. The different effect of dark and milk chocolate on walking autonomy supports the hypothesis that polyphenol content may be responsible for this effect, because dark chocolate is richer in polyphenol compared to milk chocolate.24 Accordingly, total polyphenol content was much higher in dark compared to milk chocolate; furthermore, although a small increase of serum catechin was detected after milk chocolate, the increase of serum polyphenols and in particular of the methylate metabolite of epicathecin was much more evident after dark chocolate.
The scientific background of our research was based on a previous study that documented an acute increase of artery dilatation after dark chocolate intake in smokers.9 Such positive effect was attributed to an enhanced generation of NO, which is a powerful vasodilating molecule rapidly inactivated by reactive oxygen species.3,25 Thus, the vasodilating property of dark chocolate was explained by a sequence of events including downregulation of NOX2‐mediated oxidative stress and eventually enhanced NO generation.
In this study, we show that a similar effect may also be detected in patients with severe atherosclerosis, such as those with PAD, as FMD significantly increased after dark chocolate consumption. The vasodilatation effect of cocoa is attributed to its high content of polyphenols; thus, ingestion of pure polyphenol is associated with artery dilatation similar to that observed with flavonol‐rich cocoa.26
The vasodilating capacity of dark chocolate could be dependent on the polyphenol antioxidant effect that has been documented in humans by reduction of markers of oxidative stress as well as by an increase of its plasma antioxidant property.26–27 In accordance with this effect, patients with PAD, when given dark chocolate, disclosed short term changes of oxidative stress as shown by reduced serum isoprostanes, NOX2 activity, and enhanced generation of NO. These changes are biologically plausible because previous studies have shown that polyphenols inhibit NOX2‐derived oxidative stress and, in turn, upregulate NO generation9,11,28; this latter effect is likely to play a major role in the WDA changes as it was independently associated with MWD increase by dark chocolate. These data may lead to speculation that the enhanced NO generation could be responsible for artery dilatation and eventually improve WDA. Indirect support for this hypothesis may be provided by in vitro experiments demonstrating that polyphenols contained in the chocolate reduced the release of adhesive molecules and, overall, increased NO generation by stimulated HUVEC. This experiment was conducted using catechin and epicatechin concentrations relatively close to that achieved in blood circulation after dark chocolate intake. Prior studies provided conflicting results as to whether free polyphenols may be detected after dark chocolate consumption. Thus, while Actis‐Goreta et al.29 and Ottaviani et al.30 did not find either catechin or epicatechin after chocolate intake, 2 previous reports and the present one showed detectable free polyphenols in blood circulation.31–32 This difference may depend on the study protocol as, differently from other studies, Actis‐Goreta29 and Ottaviani30 included healthy volunteers who followed a diet not containing polyphenols in the 24 to 48 hours preceding the intervention.
It is of note that catechin or epicatechin increased NO generation by endothelial cells only if used in combination, suggesting a synergism among polyphenols in functionally affecting endothelial cells.
This hypothesis is in accordance with previous reports indicating that the antioxidant effect exhibited by dark chocolate is explained by a synergism among polyphenols more than by the effect of single polyphenols.33
However, there are still uncertainties on whether NO is implicated in the improvement of walking autonomy. Thus, supplementation of Knox, by diet (beet root), or by sodium nitrate as potential sources of NO has been studied to improve exercise performance in healthy subjects and PAD patients.34 In particular, Kenjale et al.35 demonstrated that dietary nitrate supplementation enhanced exercise tolerance in PAD patients, as shown by enhancement in peak of walking distance and claudication pain‐free time but not ABI, supporting the hypothesis that NO increased peripheral tissue oxygenation in the area of hypoxia. However, a large‐prospective, double‐blind, placebo‐controlled study36 with prolonged administration of an NO‐donating agent (NCX 4016) did not improve walking distance in PAD.
The study has implications and limitations. It should be considered a proof‐of‐concept study that is potentially useful to understand the mechanism of disease related to IC but not transferable to clinical practice because of small sample size and the design of the study. Further study, in fact, should be done to assess whether similar changes may be detected with long‐term dark chocolate administration. The study is also limited by its single‐blinded design and the lack of a placebo group.
Some data interpretation is based essentially on indirect evidence. Thus, we have only indirect evidence that vasodilatation is the mechanism accounting for walking autonomy increase, because direct analysis of peripheral circulation has not been done. We also have indirect evidence that NOX2 inhibition plays a key role in the upregulation of NO, but a specific NOX2 inhibitor should be used to explore this issue.
In conclusion, the results of this study suggest that short‐term administration of dark chocolate improves walking autonomy with a mechanism involving its high content of polyphenols and perhaps mediated by an oxidative stress mechanism, which ultimately leads to enhanced NO generation. Evaluation of walking distance by long‐term dark chocolate administration is mandatory to assess whether dark chocolate may be a novel approach for the treatment of IC in PAD patients.
Sources of Funding
This study was supported by a grant from the Sapienza University of Rome (Ricerche Universitarie 2012 prot. C26A12BPZN) (funds to L. Loffredo).
Disclosures
None.
References
- 1.Tendera M, Aboyans V, Bartelink ML, Baumgartner I, Clement D, Collet JP, Cremonesi A, De Carlo M, Erbel R, Fowkes FG, Heras M, Kownator S, Minar E, Ostergren J, Poldermans D, Riambau V, Roffi M, Rother J, Sievert H, van Sambeek M, Zeller T. ESC guidelines on the diagnosis and treatment of peripheral artery diseases: document covering atherosclerotic disease of extracranial carotid and vertebral, mesenteric, renal, upper and lower extremity arteries: the Task Force on the Diagnosis and Treatment of Peripheral Artery Diseases of the European Society of Cardiology (ESC). Eur Heart J. 2011; 32:2851-2906. [DOI] [PubMed] [Google Scholar]
- 2.Hirsch AT, Haskal ZJ, Hertzer NR, Bakal CW, Creager MA, Halperin JL, Hiratzka LF, Murphy WR, Olin JW, Puschett JB, Rosenfield KA, Sacks D, Stanley JC, Taylor LM, Jr, White CJ, White J, White RA, Antman EM, Smith SC, Jr, Adams CD, Anderson JL, Faxon DP, Fuster V, Gibbons RJ, Hunt SA, Jacobs AK, Nishimura R, Ornato JP, Page RL, Riegel B. ACC/AHA 2005 practice guidelines for the management of patients with peripheral arterial disease (lower extremity, renal, mesenteric, and abdominal aortic): a collaborative report from the American Association for Vascular Surgery/Society for Vascular Surgery, Society for Cardiovascular Angiography and Interventions, Society for Vascular Medicine and Biology, Society of Interventional Radiology, and the ACC/AHA Task Force on Practice Guidelines (writing committee to develop guidelines for the management of patients with peripheral arterial disease): endorsed by the American Association of Cardiovascular and Pulmonary Rehabilitation; National Heart, Lung, and Blood Institute; Society for Vascular Nursing; Transatlantic Inter‐Society Consensus; and Vascular Disease Foundation. Circulation. 2006; 113:e463-e654. [DOI] [PubMed] [Google Scholar]
- 3.Allen JD, Giordano T, Kevil CG. Nitrite and nitric oxide metabolism in peripheral artery disease. Nitric Oxide. 2012; 26:217-222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Brevetti G, Corrado S, Martone VD, Di Donato A, Silvestro A, Vanni L. Microcirculation and tissue metabolism in peripheral arterial disease. Clin Hemorheol Microcirc. 1999; 21:245-254. [PubMed] [Google Scholar]
- 5.Coutinho T, Rooke TW, Kullo IJ. Arterial dysfunction and functional performance in patients with peripheral artery disease: a review. Vasc Med. 2011; 16:203-211. [DOI] [PubMed] [Google Scholar]
- 6.Loffredo L, Carnevale R, Cangemi R, Angelico F, Augelletti T, Di Santo S, Calabrese CM, Della Volpe L, Pignatelli P, Perri L, Basili S, Violi F. NOX2 up‐regulation is associated with artery dysfunction in patients with peripheral artery disease. Int J Cardiol. 2013; 165:184-192. [DOI] [PubMed] [Google Scholar]
- 7.Loffredo L, Marcoccia A, Pignatelli P, Andreozzi P, Borgia MC, Cangemi R, Chiarotti F, Violi F. Oxidative‐stress‐mediated arterial dysfunction in patients with peripheral arterial disease. Eur Heart J. 2007; 28:608-612. [DOI] [PubMed] [Google Scholar]
- 8.Loffredo L, Pignatelli P, Cangemi R, Andreozzi P, Panico MA, Meloni V, Violi F. Imbalance between nitric oxide generation and oxidative stress in patients with peripheral arterial disease: effect of an antioxidant treatment. J Vasc Surg. 2006; 44:525-530. [DOI] [PubMed] [Google Scholar]
- 9.Loffredo L, Carnevale R, Perri L, Catasca E, Augelletti T, Cangemi R, Albanese F, Piccheri C, Nocella C, Pignatelli P, Violi F. NOX2‐mediated arterial dysfunction in smokers: acute effect of dark chocolate. Heart. 2011; 97:1776-1781. [DOI] [PubMed] [Google Scholar]
- 10.Heiss C, Kleinbongard P, Dejam A, Perre S, Schroeter H, Sies H, Kelm M. Acute consumption of flavanol‐rich cocoa and the reversal of endothelial dysfunction in smokers. J Am Coll Cardiol. 2005; 46:1276-1283. [DOI] [PubMed] [Google Scholar]
- 11.Carnevale R, Loffredo L, Pignatelli P, Nocella C, Bartimoccia S, Di Santo S, Martino F, Catasca E, Perri L, Violi F. Dark chocolate inhibits platelet isoprostanes via NOX2 down‐regulation in smokers. J Thromb Haemost. 2012; 10:125-132. [DOI] [PubMed] [Google Scholar]
- 12.Violi F, Sanguigni V, Carnevale R, Plebani A, Rossi P, Finocchi A, Pignata C, De Mattia D, Martire B, Pietrogrande MC, Martino S, Gambineri E, Soresina AR, Pignatelli P, Martino F, Basili S, Loffredo L. Hereditary deficiency of gp91(phox) is associated with enhanced arterial dilatation: results of a multicenter study. Circulation. 2009; 120:1616-1622. [DOI] [PubMed] [Google Scholar]
- 13.Violi F, Pignatelli P, Pignata C, Plebani A, Rossi P, Sanguigni V, Carnevale R, Soresina A, Finocchi A, Cirillo E, Catasca E, Angelico F, Loffredo L. Reduced atherosclerotic burden in subjects with genetically determined low oxidative stress. Arterioscler Thromb Vasc Biol. 2013; 33:406-412. [DOI] [PubMed] [Google Scholar]
- 14.Bendall JK, Rinze R, Adlam D, Tatham AL, de Bono J, Wilson N, Volpi E, Channon KM. Endothelial Nox2 overexpression potentiates vascular oxidative stress and hemodynamic response to angiotensin II: studies in endothelial‐targeted Nox2 transgenic mice. Circ Res. 2007; 100:1016-1025. [DOI] [PubMed] [Google Scholar]
- 15.Jung O, Schreiber JG, Geiger H, Pedrazzini T, Busse R, Brandes RP. Gp91phox‐containing NADPH oxidase mediates endothelial dysfunction in renovascular hypertension. Circulation. 2004; 109:1795-1801. [DOI] [PubMed] [Google Scholar]
- 16.Martinez‐Gonzalez MA, Fernandez‐Jarne E, Serrano‐Martinez M, Wright M, Gomez‐Gracia E. Development of a short dietary intake questionnaire for the quantitative estimation of adherence to a cardioprotective Mediterranean diet. Eur J Clin Nutr. 2004; 58:1550-1552. [DOI] [PubMed] [Google Scholar]
- 17.Aboyans V, Criqui MH, Abraham P, Allison MA, Creager MA, Diehm C, Fowkes FG, Hiatt WR, Jonsson B, Lacroix P, Marin B, McDermott MM, Norgren L, Pande RL, Preux PM, Stoffers HE, Treat‐Jacobson D. Measurement and interpretation of the ankle‐brachial index: a scientific statement from the American Heart Association. Circulation. 2012; 126:2890-2909. [DOI] [PubMed] [Google Scholar]
- 18.Hoffman SW, Roof RL, Stein DG. A reliable and sensitive enzyme immunoassay method for measuring 8‐isoprostaglandin F2 alpha: a marker for lipid peroxidation after experimental brain injury. J Neurosci Methods. 1996; 68:133-136. [DOI] [PubMed] [Google Scholar]
- 19.Pignatelli P, Carnevale R, Cangemi R, Loffredo L, Sanguigni V, Stefanutti C, Basili S, Violi F. Atorvastatin inhibits gp91phox circulating levels in patients with hypercholesterolemia. Arterioscler Thromb Vasc Biol. 2010; 30:360-367. [DOI] [PubMed] [Google Scholar]
- 20.Schroeter H, Heiss C, Balzer J, Kleinbongard P, Keen CL, Hollenberg NK, Sies H, Kwik‐Uribe C, Schmitz HH, Kelm M. (‐)‐Epicatechin mediates beneficial effects of flavanol‐rich cocoa on vascular function in humans. Proc Natl Acad Sci USA. 2006; 103:1024-1029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Smith CR, Tschinkel WR. Ant fat extraction with a Soxhlet extractor. Cold Spring Harb Protoc. 2009; 2009 [DOI] [PubMed] [Google Scholar]
- 22.Mosca L, De Marco C, Visioli F, Cannella C. Enzymatic assay for the determination of olive oil polyphenol content: assay conditions and validation of the method. J Agric Food Chem. 2000; 48:297-301. [DOI] [PubMed] [Google Scholar]
- 23.Menna C, De Falco E, Pacini L, Scafetta G, Ruggieri P, Puca R, Petrozza V, Ciccone AM, Rendina EA, Calogero A, Ibrahim M. Axitinib affects cell viability and migration of a primary foetal lung adenocarcinoma culture. Cancer Invest. 2014; 32:13-21. [DOI] [PubMed] [Google Scholar]
- 24.Langer S, Marshall LJ, Day AJ, Morgan MR. Flavanols and methylxanthines in commercially available dark chocolate: a study of the correlation with nonfat cocoa solids. J Agric Food Chem. 2011; 59:8435-8441. [DOI] [PubMed] [Google Scholar]
- 25.Violi F, Marino R, Milite M, Loffredo L. Nitric oxide and its role in lipid peroxidation. Diabetes Metab Res Rev. 1999; 15:283-288. [DOI] [PubMed] [Google Scholar]
- 26.Monahan KD. Effect of cocoa/chocolate ingestion on brachial artery flow‐mediated dilation and its relevance to cardiovascular health and disease in humans. Arch Biochem Biophys. 2012; 527:90-94. [DOI] [PubMed] [Google Scholar]
- 27.Corti R, Flammer AJ, Hollenberg NK, Luscher TF. Cocoa and cardiovascular health. Circulation. 2009; 119:1433-1441. [DOI] [PubMed] [Google Scholar]
- 28.Pignatelli P, Di Santo S, Buchetti B, Sanguigni V, Brunelli A, Violi F. Polyphenols enhance platelet nitric oxide by inhibiting protein kinase C‐dependent NADPH oxidase activation: effect on platelet recruitment. FASEB J. 2006; 20:1082-1089. [DOI] [PubMed] [Google Scholar]
- 29.Actis‐Goretta L, Leveques A, Giuffrida F, Romanov‐Michailidis F, Viton F, Barron D, Duenas‐Paton M, Gonzalez‐Manzano S, Santos‐Buelga C, Williamson G, Dionisi F. Elucidation of (‐)‐epicatechin metabolites after ingestion of chocolate by healthy humans. Free Radic Biol Med. 2012; 53:787-795. [DOI] [PubMed] [Google Scholar]
- 30.Ottaviani JI, Momma TY, Kuhnle GK, Keen CL, Schroeter H. Structurally related (‐)‐epicatechin metabolites in humans: assessment using de novo chemically synthesized authentic standards. Free Radic Biol Med. 2012; 52:1403-1412. [DOI] [PubMed] [Google Scholar]
- 31.Flammer AJ, Hermann F, Sudano I, Spieker L, Hermann M, Cooper KA, Serafini M, Luscher TF, Ruschitzka F, Noll G, Corti R. Dark chocolate improves coronary vasomotion and reduces platelet reactivity. Circulation. 2007; 116:2376-2382. [DOI] [PubMed] [Google Scholar]
- 32.Schroeter H, Heiss C, Balzer J, Kleinbongard P, Keen CL, Hollenberg NK, Sies H, Kwik‐Uribe C, Schmitz HH, Kelm M. (‐)‐Epicatechin mediates beneficial effects of flavanol‐rich cocoa on vascular function in humans. Proc Natl Acad Sci USA. 2006; 103:1024-1029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Pignatelli P, Di Santo S, Carnevale R, Violi F. The polyphenols quercetin and catechin synergize in inhibiting platelet CD40 l expression. Thromb Haemost. 2005; 94:888-889. [DOI] [PubMed] [Google Scholar]
- 34.Lidder S, Webb AJ. Vascular effects of dietary nitrate (as found in green leafy vegetables and beetroot) via the nitrate‐nitrite‐nitric oxide pathway. Br J Clin Pharmacol. 2013; 75:677-696. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Kenjale AA, Ham KL, Stabler T, Robbins JL, Johnson JL, Vanbruggen M, Privette G, Yim E, Kraus WE, Allen JD. Dietary nitrate supplementation enhances exercise performance in peripheral arterial disease. J Appl Physiol (1985). 2011; 110:1582-1591. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Gresele P, Migliacci R, Arosio E, Bonizzoni E, Minuz P, Violi F. Effect on walking distance and atherosclerosis progression of a nitric oxide‐donating agent in intermittent claudication. J Vasc Surg. 2012; 56:1622-1628. [DOI] [PubMed] [Google Scholar]


