Abstract
Ornithine decarboxylase (ODC) is the key rate limiting enzyme in the polyamine synthesis pathway and it is overexpressed in a variety of cancers. We found that polyamine synthesis and modulation of ODC signaling occurs at early stages of pancreatic precursor lesions and increases as the tumor progresses in Kras activated p48Cre/+-LSL-KrasG12D/+ mice. Interest in use of the ODC inhibitor Eflornithine (DFMO) as a cancer chemopreventive agent has increased in recent years since ODC was shown to be transactivated by the c-myc oncogene and to cooperate with the ras oncogene in malignant transformation of epithelial tissues. We tested the effects of DFMO on pancreatic intraepithelial neoplasms (PanINs) and their progression to pancreatic ductal adenocarcinoma (PDAC) in genetically engineered Kras mice. The KrasG12D/+ mice fed DFMO at 0.1 and 0.2 % in the diet showed a significant inhibition (p<0.0001) of PDAC incidence compared with mice fed control diet. Pancreatic tumor weights were decreased by 31–43% (p<0.03–0.001) with both doses of DFMO. DFMO at 0.1 and 0.2 % caused a significant suppression (27 and 31%, P<0.02–0.004) of PanIN 3 lesions (carcinoma in situ). DFMO-treated pancreas exhibited modulated ODC pathway components along with decreased proliferation and increased expression of p21/p27 as compared with pancreatic tissues derived from mice fed control diet. In summary, our preclinical data indicate that DFMO has potential for chemoprevention of pancreatic cancer and should be evaluated in other PDAC models and in combination with other drugs in anticipation of future clinical trials.
Keywords: Eflornithine, DFMO, Ornithine decarcoxylase (ODC), PanIN lesions, pancreatic cancer, Kras, genetically engineered mouse (GEM), chemoprevention
Introduction
Pancreatic cancer (PC) is the fourth leading cause of cancer mortality in both men and women in the U.S (1) and the seventh leading cause of cancer deaths world-wide. Despite tremendous scientific effort for over three decades, PC still remains an almost uniformly lethal devastating disease with <6 % five-year survival. The incidence of PC has been increasing over the past 10 years. In 2013, about 46,420 people (23,530 men and 22,890 women) were expected to be diagnosed with PC and 39,590 people (20,170 men and 19,420 women) were expected to die of PC in the USA (1). Worldwide, 338,000 people were diagnosed with PC in 2012 (2). The Pancreatic Action Network estimates that by 2020, PC will become the second leading cause of cancer-related deaths in the USA. Lack of early diagnosis and effective interventions are the major factors in the poor prognosis and dismal survival rates (3–5). Frontline chemotherapeutic agents for pancreatic cancer treatment provide a marginal survival benefit. So far, a range of targeted therapies has failed to improve survival significantly in many clinical trials. Identifying successful intervention strategies using preclinical models that mimic human PC progression would help in the development of novel agents for PC prevention and treatment. The PC precursors, pancreatic intraepithelial neoplasias (PanINs), progress slowly over many years to development of invasive PC (3–5). Hence, it is important to develop novel strategies to delay or inhibit progression of PanIN to PC. The K-rasG12D genetically engineered mouse (GEM) is an excellent PC model that shows development of lesions closely resembling human PanINs with progression to pancreatic ductal carcinoma (PDAC) and it has been used successfully for chemoprevention studies.
Activation of ornithine Decarboxylase (ODC), the key regulatory enzyme in polyamine biosynthesis, and the consequent increase in concentrations of polyamines (putresine, spermine and spermidine) are associated with tumor promotion and progression. ODC is modulated by important genes of the polyamine biosynthesis pathway, such as increased arginase (Arg1) and ornithine aminotransferase (Oat), and decreased ODC antizyme (Oaz) and spermidine/spermine N(1)-acetyltransferase (Sat1). A link between polyamine levels and tumor grade and stage is well established in colorectal cancers (6,7). Elevated levels of ODC and polyamines also have been found in PC (8). Pancreatic adenocarcinoma cell lines have been shown previously to have elevated ODC activities (9). Although it is clear that ODC over-expression is associated with advanced PC development, it has not been established whether overexpression of ODC is involved in the initiation or progression of pancreatic carcinogenesis.
Clinical and preclinical studies have demonstrated that eflornithine (DFMO), an ODC inhibitor, is a potential chemopreventive agent for several cancers (10–16). However, expression of ODC during progression of PC and the chemopreventive efficacy of DFMO in a preclinical model that recapitulates human PC have not yet been evaluated. The rationale for the use of DFMO as a cancer chemopreventive agent has been strengthened in recent years because ODC has been shown to be transactivated by the c-myc oncogene in various cell and tissue types and to cooperate with the ras oncogene in malignant transformation of epithelial tissues. To address the role of ODC in pancreatic carcinogenesis, we first compared ODC activity in pancreatic tumor tissue from normal mice and that from 2 month-, 6 month- and 10 month-old p48Cre/+-LSL-KrasG12D/+ mice. We confirmed overexpression of ODC in pancreatic cancer tissues as the mice age and then tested the ODC inhibitor DFMO as a chemopreventive. DFMO inhibited tumor progression and modulated several components of ODC signaling and cell proliferation, supporting future testing of DFMO in clinical trials for PC prevention.
Materials and methods
Mouse model, diet and handling
Generation of p48Cre/+; LSL-KrasG12D/+ mice expressing the activated KrasG12D oncogene has been described previously (17). All animal research was performed under the animal protocols approved by the University of Oklahoma Health Sciences Center (OUHSC) institutional animal care and use committee (IACUC). Animals were housed in ventilated cages under standardized conditions (21°C, 60% humidity, 12-h light/12-h dark cycle, 20 air changes/h) in the OUHSC rodent barrier facility. Semi-purified modified AIN-76A diet ingredients were purchased from Bioserv, Inc., NJ. Eflornithine (DFMO) was procured from the NCI-DCP chemoprevention drug repository. DFMO (0.1% and 0.2%) was premixed with small quantities of casein and then blended into the diet using a Hobart Mixer. The rationale for selecting 0.2% DFMO was based on earlier experiments; this dose is equivalent to ≤40% of the maximum tolerated dose when it is given in semi-purified AIN-76A diet. The lower dose (0.1%) chosen is comparable to doses of DFMO used in clinical trials. Both control and experimental diets were prepared weekly and stored in the cold room. Mice were allowed ad libitum access to the respective diets and to automated tap water purified by reverse osmosis.
Breeding and genotyping analysis
LSL-KrasG12D/+ and p48Cre/+ mice were maintained in a C57BL/6 heterozygous genetic background and bred. Offspring of activated p48Cre/+.LSL-KrasG12D/+ and C5BL/6 wild type mice were generated at required quantities. The genotype of each pup was confirmed by tail DNA extraction and polymerase chain reaction (PCR). Briefly, genomic DNA was isolated from tail tissue samples using the mini-prep kit (Invitrogen, Carlsbad, CA). PCR was performed for K-ras and Cre genes using the following conditions: denaturation at 95°C for 5 min, followed by 35 cycles at 95°C for 1 minute, 60°C for 1 minute and 72°C for 1 minute. Oligonucleotide primer sequences used were as follows: K-ras 5′-CCTTTACAAGCGCACGCAGAG-3′ sense, 5′-AGCTAGCCACCATGGCTTGAGTAAGTCTGCA-3′ anti-sense; and p48Cre 5′-ACCGTCAGTACGTGAGATATCTT-3′ sense and 5-ACCTGAAGATGTTCGCGATTATCT-3′ antisense. PCR products were separated on a 2 % agarose gel. Successful recombination yields were 550 and 350-bp products for Kras and Cre genes, respectively (17).
Ornithine Decarboxylase (ODC) assay
p48Cre/+-LSL-KrasG12D/+ GEM (n=6/group) at 2, 6 and 10 months of age, along with wild type mice, were killed and pancreata were snap frozen in liquid nitrogen for further analysis. ODC activity in the pancreata of these mice was measured as release of CO2 from L-[1-14C]-ornithine. Briefly, pancreas samples were trypsinized, washed with phosphate-buffered saline (PBS), and lysed in 100 μL of ODC lysis buffer [25 mmol/L Tris-HCl (pH 7.4), 0.1 mmol/L EDTA, 0.1 % Triton X-100, 0.1 mmol/L pyridoxyl-5-phosphate, 1 mmol/L dithiothreitol (DTT), and 1x protease inhibitors]. Lysates were frozen at −80°C, thawed on ice, and centrifuged at 12,000 × g for 20 minutes at 4°C to remove cell debris. Supernatants were transferred to new Eppendorf tubes, and 100 μL (5mg/mL protein) aliquots were transferred to stoppered glass vials containing 10 uL (55 nMol) of L-[1-14C]ornithine hydrochloride (Thermo Scientific) in triplicate. ODC assay buffer (110 μL) containing [1-14C]ornithine hydrochloride was added and incubated for 1 hour at 37°C. Reactions were terminated with an equal volume of 10 % trichloroacetic acid (TCA) injected with a syringe through the rubber stopper top and left for another 20 min. Released 14CO2 was captured with a filter paper, which was soaked with 80 μL sodium hydroxide and inserted through a hole in the stopper top (Kontes Glass, Vineland, NH). The filter paper was transferred to a scintillation vial, 5 mL of scintillation fluid was added, and liquid scintillation counting was done for 1 minute. ODC activity was determined as picomoles of CO2 released (calculated from cpm) per milligram of protein per hour.
Preclinical efficacy assay
Male and female p48Cre/+-LSL-KrasG12D/+ mice were used in the efficacy study. Briefly, five week-old mice were selected and randomized so that average body weights in each group were equal (n = 30–34/group for p48Cre/+-LSL-KrasG12D/+ mice and n = 12/group for C57BL/6 wild-type mice) and were fed AIN-76A diet for one week. At 6 weeks of age, mice were fed AIN-76A experimental diets containing 0 %, 0.1 % or 0.2 % DFMO in the diet until termination of the study. Mice were checked routinely for signs of weight loss or any signs of toxicity or any abnormalities. Body weight of each animal was measured once weekly for the first 6 weeks and then once a month until termination. After 265 days (38 weeks) on experimental diets, all mice were euthanized by CO2 asphyxiation at 44 weeks (~300 days) of age and necropsied; pancreata were collected from all groups, weighed and snap frozen in liquid nitrogen for further analysis. Pancreata required for histopathology and immunohistochemistry (IHC) to identify PanIN lesions and PDAC and for evaluation of various molecular markers were fixed (head to tail) in 10 % neutral-buffered formalin as previously described (17–19).
Analysis of PanIN Lesions and PDAC
Normal and tumor pancreatic tissues were fixed in 10 % neutral buffered formalin for 24–48 h and then processed and embedded in paraffin according to standard protocols. Tissue sections (4 μm) of each pancreas stained with Hematoxylin and Eosin (H&E) were evaluated histologically by a pathologist blinded to the experimental groups. PanIN lesions and carcinomas were classified according to histopathology criteria as previously described (17–19). To quantify the progression of PanIN lesions, the total number of ductal lesions and their grades were determined. The relative proportion of each PanIN lesion grade to the overall number of analyzed ducts was recorded for each animal. Similarly, pancreatic carcinoma and normal appearing pancreatic tissue were evaluated for all the animals. The incidence of PDAC is defined as the percentage of mice with PDAC as previously described by us (17–19). Briefly, We evaluate pancreas sections derived from top, middle and bottom portion of the tissue block (from head to tail) from the GEM mice (N=30–34 mice/group male and female). The efficacy endpoints used in this study were inhibition of PanINs and PDAC. The criterion for invasive disease incidence is groups of adenocarcinoma cells (at least 3%) invading with adenomatous stroma streaming around the cells.
Immunohistochemistry and Immunofluorescence
5-μm fixed sections were incubated with primary antibodies in a hybridization chamber for 1 h at room temperature or overnight at 4 °C. The primary antibodies for proliferating cell nuclear antigen (PCNA), Cav-1, β-catenin and p21 were from Santa Cruz Biotechnology ); those for Ki67 and cyclin D1 were from Cell Signaling; those for p16 were from LS Bio and those for Rb were from Abcam. Following incubations with primary antibody, sections were incubated for 1 h with anti-mouse or anti-rabbit secondary antibodies, as appropriate for each primary, then visualized with diaminobenzidine (DAB) and counterstained with Hematoxylin for IHC and counterstained with DAPI (4′,6-diamidino-2-phenylindole) for immunohistofluorescence (IHF). Slides were observed under an Olympus microscope 1X701 and digital computer images were recorded with an Olympus DP70 camera.
Quantitative real-time PCR analysis
Total RNA from pancreatic samples (consisting of normal, PanIN and PDAC) was extracted using an RNA Kit for isolation of total cellular RNA (Invitrogen) as per the manufacturer’s instructions. Equal quantities of DNA-free RNA were used in reverse transcription reactions for making cDNA using SuperScript reverse transcriptase (Invitrogen). The real-time PCR was carried out in a 25-μL reaction volume using 3 μL of a 1:10 cDNA dilution containing SYBR Green master mix (BioRad) and primers for PCNA, caveolin-1 (Cav-1), ODC antizyme (Oaz), Ornithine aminotransferase (Oat), spermidine/spermine N(1)-acetyltransferase (Sat), arginase (Arg1), spermine synthase (Sms), spermidine synthase (Srm) (Supplementary Table 1). All PCRs were done in a Biorad iCycler iQ real-time PCR detection system and fluorescence threshold values (Ct) were calculated. Relative mRNA levels were assessed by standardization to actin. Results are expressed as a fold difference in gene expression.
Cell lines
The human MiaPaCa 2 cancer cell line was obtained from the American Type Culture Collection (ATCC) and maintained in Dulbecco’s modified Eagle medium (DMEM) (Life Technologies Inc.) supplemented with 10% fetal bovine serum and 1% penicillin-streptomycin in a humidified chamber at 37°C and 5% CO2. The cell line was authenticated by short-tandem repeat analysis by ATCC. MiaPaCa 2 cell line was initially expanded and cryopreserved within 1 month of receipt. Cells were typically used for 3 months, and at that time a fresh vial of cryopreserved cells was used. All experiments were carried out with cells grown to 70% to 80% confluence. To assess growth inhibition and molecular markers, we applied sub-toxic concentrations of DFMO, ranging from 2 to 4 mmol/L.
Protein extraction and Western blot assay
Pancreata (normal+PanINs+PDAC) harvested from mice fed diets with or without DFMO, or MiaPaCa 2 cells treated with or without DFMO in the presence or absence of spermidine (polyamine), were homogenized and lysed in ice-cold lysis buffer. After a brief vortexing, the lysates were collected by centrifugation at 12,000 × g for 15 minutes at 4°C, and protein concentrations were measured with the Bio-Rad Protein Assay reagent (Hercules, CA). An aliquot (50 μg protein/lane) of the total protein was separated with 10 % sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) and transferred to nitrocellulose membranes. After blocking with 5 % milk powder, membranes were probed for expression of ODC, c-myc, p21 and α-tubulin in hybridizing solution [1:500, in Tris-buffered saline (TBS)-Tween 20 solution] using the respective primary antibodies and then probed with the appropriate horseradish peroxidase (HRP)-conjugated secondary antibodies. Detection was performed using the SuperSignal® West Pico Chemiluminescence procedure (Pierce, Rockford, IL). All Western blot experiments were repeated two times. Image J software was used for quantification of the blots.
Statistical Analysis
The data are presented as means ± SE. Differences in body weights were analyzed by ANOVA. Statistical differences between control and treated groups were evaluated using Fisher’s exact test for PDAC incidence and unpaired t-test with Welch’s correction was used for PanINs and PDAC lesions. Differences between groups are considered significant at p<0.05.
Results
Activation of the ornithine decarboxylase pathway during progression of pancreatic cancer
p48Cre/+-LSL-KrasG12D/+ and wild type mice were analyzed for the expression of ODC pathway components (Fig. 1A–F). ODC catalyzes the first step in the polyamine biosynthetic pathway, the decarboxylation of ornithine to putrescine. ODC is modulated by other important componetnts of this pathway, such as arginase (Arg1), Oat, Oaz and Sat1; and its activation can lead to tumor promotion and progression (Fig. 1G). As the mice aged from 2 to 6 to 10 months, we observed a significant increase in the ODC activity (Fig. 1A). ODC activity was > 2-fold elevated in the pancreas of 10 month-old GEM mice compared with control pancreas (Fig. 1A). Similarly, other important ODC-regulating enzymes were modulated in the tumors compared with normal pancreas as determined by quantitative real time PCR (Fig. 1B–F). The ODC inhibitory enzyme Oaz and the polyamine-degrading enzyme Sat1 were down-regulated significantly in the pancreatic tumors compared with normal pancreas. The Arg and Oat required for increased ODC were increased in the tumors.
Figure 1.
A. ODC activity in the wild type (WT) and p48Cre/+-LSL-KrasG12D/+ GEM mice. ODC activity increased in the GEM mice as they aged from 2 to 10 months. B–F. mRNA expression of ODC (B), Oaz (C), Arg1 (D), Oat (E) and Sat1 (F) in normal and tumor-bearing pancreas. A significant increase in ODC, Arg1 and Oat and a decrease in Oaz and Sat1 mRNA expression were observed in the pancreatic tumors compared with normal pancreas. G) Schematic representation of polyamine biosynthesis. H. Experimental design for evaluation of DFMO efficacy in PC prevention in male and female p48Cre/+-LSL-KrasG12D/+ mice. At 6-weeks of age, groups of mice (30–34/group for activated p48Cre/+-LSL-KrasG12D/+ or 12/group for wild-type) were fed AIN76-A diets containing 0, 0.1% or 0.2% DFMO continuously for 38 weeks and each pancreas was evaluated histopathologically for marker expression as described in the text. I–J. Effect of DFMO on body weight (BW; means ± SE) at termination of the experiment. No statistically significant difference was observed between control and DFMO-treated p48Cre/+-LSL-KrasG12D/+ or wild type mice.
General health of animals treated with DFMO
The experimental protocol for evaluating DFMO in pancreatic cancer progression is summarized in Figure 1H. p48Cre/+-LSL-KrasG12D/+ mice fed AIN-76A or DFMO diets had steady body weight gain. As shown in Fig. 1 (I–J), there was no significant difference in body weight in the mice fed either AIN-76A or AIN-76A diets supplemented with either 0.1% or 0.2% DFMO. None of the animals fed the DFMO diets exhibited any observable toxicity or any gross morphologic changes to liver, spleen, kidney or lung.
Effect of DFMO diet on pancreatic tumor weight and PDAC Incidence in genetically engineered mice
Pancreatic weight is a simple marker to assess the progression of tumor. A significant increase in pancreas weight (0.8 g to 1.5 g) was observed in p48Cre/+-LSL-KrasG12D/+ mice compared with wild-type mice (~200–300 mg). Figure 2A shows the H&E staining of the pancreatic tumors with and without DFMO treatment. As summarized in Figures 2B and 2C, DFMO treatment caused a significant decrease in the weight of pancreatic tumors in p48Cre/+-LSL-KrasG12D/+ mice. In GEM fed 0.1 or 0.2% DFMO, the pancreas weights were 0.75 ± 0.1 g and 0.86 ± 0.01 g, respectively, in males and 0.65 ± 0.08 g and 0.61 ± 0.04 g in females. The pancreatic tumor weights were decreased by 31–43% (p<0.03–0.001) with both doses of DFMO in male and female mice.
Figure 2.
A. Histopathologic analysis of untreated and DFMO-treated pancreas using H&E staining. Pancreas from untreated animals showing poorly differentiated adenocarcinoma with some of the cells invading stroma (left panel). Pancreas from animals treated with DFMO showing PanIN lesions (right panels). B–C. Effect of DFMO on pancreas weight at the termination of the experiment in male (B) and female (C) mice. Both doses of DFMO significantly reduced the pancreatic tumor weights. D–E. Effect of DFMO on the incidence of PDAC in male (D) and female (E) mice.
Extensive histopathologic analysis of the pancreas using H&E-stained slides revealed no microscopic pathologic alterations in wild-type mice fed either AIN-76A or DFMO-supplemented diets. In contrast, AIN76A diet-fed male and female p48Cre/+LSL-KrasG12D/+ mice demonstrated >80 and 60% incidence of PDAC (% of mice with PDAC), respectively (Fig. 2D and 2E). DFMO treatment at 0.1 and 0.2 % decreased PDAC incidence to 10 and 6%, respectively, in male and to 16 and 9 %, respectively, in female GEM (Fig. 2D and 2E).
Inhibition of PanIN Lesion Progression and % Carcinoma by DFMO
Histological analysis showed 100 % penetrance of pancreatic precursor PanIN lesions in the GEM fed AIN76A or DFMO-supplemented diets. The number of PanIN 1, 2, and 3 lesions in male GEM fed AIN76-A diet were (means ± SE): 180 ± 28, 162 ± 20, and 155 ± 15, respectively; in the mice fed 0.1 % DFMO, PanIN 1, 2, and 3 numbers were 375 ± 59, 225 ± 42 and 115 ± 33; and in mice fed 0.2 % DFMO they were 260 ± 33, 192 ± 20 and 109 ± 17, respectively (Fig. 3A). The number of PanIN 1, 2, and 3 lesions in female GEM fed AIN76A diet were 183 ± 23, 178 ± 13 and 240 ± 19, respectively; in the mice fed 0.1 % DFMO, the numbers were 255 ± 36, 220 ± 21 and 188 ± 28; and in mice fed 0.2 % DFMO, they were 280 ± 49, 195 ± 24 and 158 ± 16, respectively (Fig. 3B). The number of PanIN 3 lesions or carcinoma in situ was suppressed by 21–33% in the DFMO-treated groups (Fig. 3A, B). An increase in the number of PanIN 1 and 2 lesions was observed in pancreas of DFMO-supplemented mice, suggesting a potential blockade of further progression of these lesions to carcinoma in situ and PDAC. Pancreas of GEM fed AIN76 A diet showed 30 ± 6 % (male; Fig. 3C) and 12 ± 4 % (female; Fig. 3D) PDAC within the pancreas. The carcinoma % within the pancreas was inhibited significantly (> 60 % in males and > 50 % in females; Fig. 3 panels C and D) by both doses of DFMO in GEM. Up to 23% of the pancreas from mice treated with DFMO was normal appearing (i.e., free from PanIN lesions and carcinoma) whereas only up to 5% was normal appearing in the untreated GEM (Fig. 3, panels E and F). Although DFMO significantly inhibited PDAC incidence and carcinoma spread, tumor outgrowth was observed in the treatment groups.
Figure 3.
A–B. Effect of DFMO on PanIN multiplicity in male (panel A) and female (panel B) GEM mice (means ± SE). C–D. Effect of DFMO on the percentage of pancreas with carcinoma (C-male, D-female). E–F. Effect of DFMO on the percentage of normal appearing pancreas (E-male, F-female). The data in the panels were analyzed by unpaired ‘t’-test with Welch’s correction; values are considered statistically significant at p < 0.05.
Modulation of ornithine decarboxylase Signaling by DFMO in pancreatic cancer
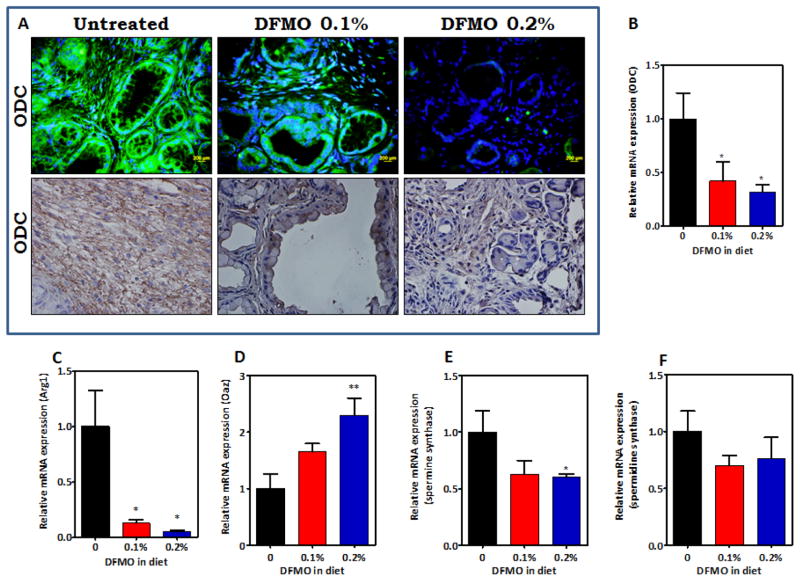
Activation of the ODC pathway is critical in polyamine biosynthesis. ODC pathway signaling molecules were measured via immunohistochemistry and quantitative real time PCR in pancreatic tissues. As shown in Figure 4A, markedly decreased ODC staining was observed in PanIN lesions and carcinoma in p48Cre/+-LSL-KrasG12D/+ mice fed DFMO-supplemented diets compared with the pancreatic tissues from mice fed AIN-76A diet alone. The expression of ODC and its related signaling molecules in the pancreas (consisting normal pancreas, PanINs and PDAC) was analyzed further using real time quantitative PCR assays. Snap frozen pancreatic tissues were analyzed for fold change in mRNA expression of ODC, arginase (Arg), OAZ and Sat1 (Fig. 4 B–D). Total RNA was isolated, reverse-transcribed into cDNA and quantified with a real-time PCR assay. As shown in Figure 4B–E, compared with pancreas from AIN-76A-fed p48Cre/+-LSL-KrasG12D/+ mice, the pancreatic tissues of DFMO-fed GEM showed significantly decreased (more than 2-fold; P < 0.05) expression of ODC and Arg mRNA (Figure 4 B, C) and an increase (more than 2-fold) in Oaz mRNA (Fig. 4 D). Expression of spermine synthase (Sms) and spermidine synthase (Srm) was reduced upon DFMO treatment (Fig. 4E–F). The decrease in the ODC signaling mRNAs in the DFMO treatment groups is likely to be due to the greater amount of normal pancreas present in the treatment groups.
Figure 4.
A. Effect of DFMO on ODC expression in pancreatic tumors. Immunohistochemical and Immunofluorescence analyses were performed with paraffin-embedded and micro-sectioned pancreatic tissues as described in the Methods section. A significantly decreased expression of ODC was seen with DFMO treatment. B–F. Effect of DFMO on ODC (B), Arg1 (C), Oaz (D), Spermine synthase (E) and Spermidine synthase (F) mRNA expression in pancreatic tumors as determined by quantitative real time PCR. DFMO treatment led to decreased ODC, Arg1, spermine synthase, spermidine synthase and increased Oaz. (* - p<0.01, ** - p<0.001)
DFMO inhibits tumor cell proliferation and induces p21, p27 and p53
To determine the effects of DFMO on tumor cell proliferation in pancreatic tissues of the GEM, PCNA, Ki67, p21, p27, cyclin D1, Rb, p16 and p53 were analyzed using IHC/IHF and real time PCR approaches (Fig. 5A, B, supplementary Fig. 1). IHC staining showed that PanIN lesions and carcinoma were labeled positively for PCNA and Ki67 in the p48Cre/+-LSL-KrasG12D/+ mice fed the AIN-76A diet alone (Fig. 5A, supplementary Fig. 1). Markedly decreased numbers of PCNA- and Ki67-positive cells and decreased proliferative index were observed in mice fed a DFMO-supplemented diet (Fig. 5A, B, supplementary Fig. 1). IHC analysis of p21 revealed a significant increase in labeling with the higher DFMO dose compared with untreated pancreatic tumor tissues (Fig. 5A, B). We also measured the relative mRNA levels of PCNA, p21 and p27 in pancreatic tissues by quantitative real time-PCR (qRT-PCR) and found that DFMO significantly decreased the expression of PCNA and increased p21 mRNA in pancreatic tissues of the GEM. The higher dose of DFMO also led to an increase in p53 and p27 expression in the pancreatic tissues (Fig. 5B). These results suggest that the inhibition of tumor progression may be associated with decreased tumor cell proliferation (Fig. 5A, B) and are consistent with our in vivo tumor inhibition findings. Further, we have observed a decrease in cyclin D1 and an increase in the tumor suppressor Rb upon DFMO treatment. Higher p16 expression was observed in the normal appearing pancreatic tissues from the DFMO-treated pancreatic tumors compared with the untreated tumors (Supplementary Fig. 1).
Figure 5.
Effect of DFMO on pancreatic tumor cell proliferation. A. Immunohistochemical analysis for PCNA and p21 was performed on paraffin-embedded and micro-sectioned pancreatic tissues as described in the Methods section. A significantly decreased expression of PCNA and increased p21 expression was seen with DFMO treatment. B. Effect of DFMO on the PCNA index and on mRNA expression of PCNA, p21, p53 and p27 as determined with quantitative real time-PCR. A significantly decreased PCNA index and increased p21, p27 and p53 mRNA expression were seen in the DFMO-treated pancreatic tumor tissue. C. Effect of DFMO on c-Myc protein expression. A decrease in c-Myc expression was seen upon DFMO treatment.
Effect of DFMO on Cav-1 and β-catenin
C-myc activation leads to increased proliferation and down-regulates p21, which can lead to tumor growth. To examine the effect of DFMO on the expression of c-myc, we analyzed the pancreatic tissues (containing normal pancreas, PanINs and PDAC) and MiaPaCa cells by western immunoblotting. As shown in Figure 5C, the DFMO treatment caused a non-significant reduction in the expression of c-myc, with no dose-dependency. This reduction most likely is due to higher amounts of normal pancreas in treatment groups. We observed no significant change in the ODC protein expression in the DFMO-treated and polyamine+DFMO-treated MiaPaCa cells. The c-myc expression was unchanged in the lower dose treatment, with only a slight increase in high dose group; and cells treated with polyamines with or without DFMO showed no change in c-myc protein. Moreover, we found that p21 was increased to some extent upon DFMO treatment (Supplementary Fig. 2). Since caveolin-1 (Cav-1) can transport polyamines into the cells (20), we analyzed cav-1 protein expression by IHC/IHF and also measured mRNA expression by real time PCR (Fig. 6). A substantial decrease in cav-1 was observed upon DFMO treatment as compared with untreated mouse pancreatic tumor tissues (Fig. 6). Similarly, we observed a decrease in the β-catenin protein and mRNA expressions in the treatment groups (Fig. 6). These results are consistent with our in vivo tumor inhibition findings and suggest that the inhibition of tumor progression may be associated with the modulation of molecular markers related to the ODC pathway.
Figure 6.
Effect of DFMO on cav-1 and β-catenin. Immunohistochemistry (top row of each set), Immunofluorescence (bottom row of each set) and mRNA analysis (bar graphs to the right) revealed a decrease in the cav-1 expression with both doses of DFMO; however, only the high dose (0.2%) caused a decrease in β-catenin expression.
Discussion
Polyamines are essential for cell division and growth of neoplasia. Overexpression of ODC and increased polyamines are observed in tumor samples from various human cancers including PC (7, 21). ODC activity was 3.6-fold elevated in human adenocarcinomas and 3.9-fold elevated in neuroendocrine tumors compared with control pancreas (8). We observed increased ODC activity in p48Cre/+-LSL-KrasG12D/+ mice, a well-established model that simulates the step-wise progression of human PC and which has been used extensively by us and others for studies of PC chemoprevention. As the mice aged, the ODC activity increased significantly (Fig. 1A), suggesting that tumor cell proliferation is occurring along with the tumor progression. We evaluated mRNA expression for several constituents of the ODC pathway in the tumors as compared with normal pancreas. ODC antizyme (Oaz), which regulates polyamine synthesis by binding to and inhibiting ODC, and Sat1, a rate-limiting enzyme in the catabolic pathway of polyamine metabolism, were decreased significantly in the pancreatic tumors compared with normal pancreas (Fig. 1). Arginase and Oat, which increase polyamine levels by increasing the ODC activity, also were increased in the tumors (Fig. 1). These results are consistent with previous reports on polyamine biosynthesis in different cancers (22) and are consistent with a role for polyamine biosynthesis in PC progression.
Previous studies have shown cytostatic effects of the ODC inhibitor DMFO against several tumors (23–25) but not in pancreatic cancers. Clinical and preclinical studies have shown that DFMO has significant chemopreventive effects against several cancers (10–16); and DFMO has been employed successfully for chemoprevention of colon, prostate, skin and cervical cancers (26–31). However, no in vivo chemoprevention studies had been reported with DFMO using GEM that mimic human PC.
Inhibition of ODC activity by DFMO caused decreased cell growth and increased apoptosis in pancreatic tumor-derived cell lines (8). DFMO also was examined in hamster H2T cells, an experimental xenograft model of pancreatic carcinogenesis, both in vitro and in vivo by Marx et al. (32). In vitro they showed that cytotoxicity and Inhibition of polyamine biosynthesis increased with prolonged DFMO treatment, suggesting that continuous use of DFMO for PC prevention might improve efficacy. In the in vivo studies, H2T cells were injected into the cheek pouch of Syrian golden hamsters and DFMO treatment (3% in drinking water) decreased tumor size and inhibited growth of the PC by as much as 50% of control (32). However that xenograft model lacks classic initiation and promotion steps that characterize human PC.
We have extended these findings by showing the potential of dietary DFMO (0.1% and 0.2%) to prevent progression of PC in the p48Cre/+-LSL-KrasG12D/+ mice. We found that DFMO inhibits formation of PanIN lesions and their progression to ductal adenocarcinoma. DFMO (0.1 and 0.2%) caused significant inhibition of PDAC incidence in the male and female GEM (Fig. 2). A 21–33% suppression of PanIN 3 lesions (carcinoma in situ) was observed with DFMO treatment, and carcinoma % was inhibited by >60% in males and by >50 in females (Fig. 3). There was no dose-dependency observed with DFMO treatment; instead, the lower dose was associated with higher amounts of normal-appearing pancreas (Fig. 3).
We observed DFMO-induced inhibition of ODC and modulation of several components of polyamine biosynthesis in the pancreatic tumors. The DFMO treatment led to a significant decrease in expression of ODC, Arg1, Sms, Srm and increased Oaz expression (Fig. 4). DFMO caused a significant decrease in tumor progression, with a large amount of the pancreas still appearing normal (Fig 3). The significant reduction in pancreatic ODC mRNA most likely was du to the reduction in the overall tumor area (PanINs and PDAC) in the pancreata of mice exposed to DFMO. The pancreas tissue of untreated mice consists of greater numbers of tumor and stromal cells in comparison with the DFMO treatment groups (Fig. 2, 3). Mechanistically, although DFMO inhibits ODC activity, few studies have reported that ODC mRNA is reduced upon DFMO treatment (33–35). ODC is a critical transcription target of the Myc oncogene, which regulates checkpoints that guard against tumorigenesis; and is an effective target for cancer chemoprevention (33). Overexpression of Myc at levels found in cancer is sufficient to drive normal quiescent cells into the cycle and to accelerate their rates of cell cycle traverse (36). These responses are, at least in part, dependent upon Myc’s ability to down-regulate the expression of the cyclin-dependent kinase (Cdk) inhibitors p27Kip1 and p21Cip1 (37–39). In other systems, DFMO treatment has been associated with a reduction in c-Myc and an increase in p16 and p27 (40–42). Consistent with these reports, we observed a decrease in expression of c-myc upon DFMO treatment, although it did not reach statistical significance, and a significant increase in expression of p21, p27 and the tumor suppressor p53 (Fig. 5).
Cav1 has been predicted to help in the uptake of polyamines into cells (20). Clinical studies have shown that Cav-1 expression is associated with pancreatic tumor progression and poor prognosis for patient survival (43–44). In the present study, overexpression of Cav-1 was correlated with an increase in proliferation markers (Fig. 6). The high expression of the proliferation markers Ki67, PCNA, Cav-1 and β-catenin observed in KrasG12D/+ mouse pancreatic lesions was inhibited by dietary DFMO (Fig. 5 and 6). The exact mechanism by which DFMO influences Cav-1 is not known; however, our previous studies on colon cancer cell lines exposed to lovastatin or pancreatic cancer inhibition by atorvastatin showed significantly suppressed Cav-1 expression and its localization to membrane lipid rafts (19, 20).
DFMO treatment results in the depletion of polyamines leading to a significant decrease in the steady-state levels of transcription factors such as c-myc. It is suggested that regulation of c-myc expression may be one of the mechanisms by which polyamines modulate cell growth. A few studies have reported a decrease in c-myc upon DFMO treatment (40–42, 45,46). However, a number of signaling mechanisms, especially those involving Wnt, can contribute to regulation of c-myc and polyamines (22, 47). In the presence of Wnt, β-catenin target genes including c-myc are expressed. Myc expression, in turn, leads to the expression of ODC, the first enzyme in polyamine metabolism. Diminished β-catenin leads to reduced c-Myc (48). Wnt signaling also regulates ODC antizyme (OAZ), a protein that regulates ODC activity by targeting its degradation. In addition, peroxisome-proliferator-activated receptor-gamma (PPAR-γ), which activates spermidine/spermine N1-acetyltransferase (SSAT) transcription, is repressed by active KRAS. However, in normal cells and tissues, most of the KRAS protein is inactive as a signaling molecule. Therefore, in normal cells and tissues, Wnt signaling and inactive KRAS lead to reduced proliferation, increased apoptosis and reduced neoplasia. Under tumor conditions, ODC expression is elevated by altered Wnt signaling, in part due to inhibition of OAZ. Studies also support the existence of oncogenic mutations in KRAS that suppress polyamine catabolism (22). Consequently, altered Wnt signaling and KRAS together can act to promote neoplasia by increasing polyamine biosynthesis and suppressing polyamine catabolism. Additionally, Myc overexpression is able to down-regulate the expression of the Cdk inhibitors p27Kip1 and p21Cip1 (37–39). In the present studies, DFMO is shown to suppress the PC progression by modulating ODC signaling, decreasing proliferation, altering Wnt signaling and increasing p21, p27, Rb and p53 (Supplementary Fig. 3).
Although DFMO inhibited PDAC in Kras mice, there was still some tumor outgrowth in animals treated with both tested doses, indicating potential limitations to the use of DFMO in human clinical trials. Some possible reasons for this tumor escape include the following: 1) Although endogenous polyamines are reduced upon DFMO treatment, dietary polyamines are readily available exogenously. Thus, we think that one of the reasons for outgrowth of tumors even after DFMO treatment is the availability of exogenous polyamines through these diets. 2) It is possible that drug resistance occurs as a result of decreased intracellular drug accumulation due to overexpression of certain drug resistant efflux pumps. It may be possible to test this possibility with short term in vitro culturing of the outgrown tumors. 3) Another potential mechanism for DFMO resistance relates to the alternative mechanisms available for the synthesis of polyamines within the tumor cells, including alterations of the agmatine mechanism (Fig 1G). As evident from Fig 4 E&F, although the ODC inhibition is irreversible, polyamines are still available from these alternate pathways and may lead to the tumor outgrowth. 4) Another potential explanation for tumor outgrowth is dependent on ratio of DFMO and ODC concentrations. The half-life (t1/2) of DFMO in the serum is 2 to 4 hrs and it is eliminated in 3.2 to 3.6 hrs. The ODC enzyme has turn-over rate or t1/2 of 30 minutes. Thus, any observed persistent biological effects of DFMO are questionable. Previously, we and others have shown dose-response effects of DFMO in other organ site cancers. The failure to detect dose-dependent effects of DFMO on PDAC in this study suggests that both doses tested were on the plateau of the dose-response curve and thus that tumor outgrowth was not due to insufficient DFMO. However, the ratio of DFMO to ODC concentration also may vary at different time points throughout the period of the experiment. Although mice are fed DFMO in the diet, DFMO levels in the serum may vary periodically depending on the dietary intake by the mice. This possibility can be tested by determining the levels of DFMO in serum as a function of time and correlating average, maximal and nadir levels with anti-tumor efficacy. 5) It also is possible that tumor outgrowth is related to the species or strain of animal used. Effects of DFMO should be tested in other models of PC to see if tumor outgrowth occurs. 6) Finally, it should also be noted that, there are several other signaling mechanisms apart from the ODC pathway that can drive the tumorigenesis process. Thus, developing combinations of DFMO with other agents having different tumor inhibitory mechanisms may improve the efficacy.
Since the Kras GEM model shows step-wise progression of precursor lesions to carcinoma as occurs in humans, and is well optimized for studies of PC chemoprevention (5, 17–19, 49,50), our findings suggest (a) that ornithine decarboxylase plays a key role in progression of PanIN lesions to PC; and (b) that low dose DFMO may have potential for chemoprevention in patients at high risk for PC. Patients with chronic pancreatitis, those with PanIN lesions, Intrapapillary mucinous neoplasms, diabetes, or those of older age might be evaluated for polyamine levels to evaluate their candidacy for such chemoprevention. Further evaluation also is warranted of DFMO effects on tumors at an earlier stage and in combination with other agents to increase the efficacy.
Supplementary Material
Acknowledgments
Grant Support: This work was supported by the National Cancer Institute N01-CN-53300 to C.V. Rao.
The authors thank the University of Oklahoma Health Sciences Center Rodent Barrier Facility staff. We also want to thank Dr. Julie Sando for valuable suggestions and editorial help.
Abbrevations
- ODC
ornithine decarboxylase
- PC
pancreatic cancer
- PDAC
pancreatic ductal adenocarcinoma
- PanIN
pancreatic intraepithelial neoplasia
- DFMO
eflornithine (difluoromethylornithine)
- GEM
genetically engineered mouse
- Arg1
arginase
- Oat
ornithine aminotransferase
- Oaz
ODC antizyme
- Sat1
spermidine/spermine N(1)-acetyltransferase
- PCR
Polymerase chain reaction
- PBS
phosphate-buffered saline
- DTT
dithiothreitol
- IHC
immunohistochemistry
- H&E
Hematoxylin and Eosin
- DAB
diaminobenzidine
- IHF
immunohistofluorescence
- PCNA
proliferating cell nuclear antigen
- Cav-1
caveolin-1
- Sms
spermine synthase
- Srm
spermidine synthase
- Ct
fluorescence threshold values
- DMEM
Dulbecco’s modified Eagle medium
- SDS-PAGE
sodium dodecyl sulfate-polyacrylamide gel electrophoresis
- TBS
Tris-buffered saline
- HRP
horseradish peroxidase
- qRT-PCR
quantitative real time-PCR
- Cdk
cyclin-dependent kinase
- PPAR-γ
peroxisome-proliferator-activated receptor-gamma
- SSAT
spermidine/spermine N1-acetyltransferase
Footnotes
Conflict of interest: The authors declare no conflict of interest.
References
- 1.American Cancer Society. Cancer facts and figures 2014. Atlanta, GA: 2014. [Google Scholar]
- 2.Ferlay J, Soerjomataram I, Ervik M, Dikshit R, Eser S, Mathers C, et al. GLOBOCAN 2012 v1.0, Cancer Incidence and Mortality Worldwide: IARC CancerBase No. 11 [Internet] Lyon, France: International Agency for Research on Cancer; 2013. Available from: http://globocan.iarc.fr. [Google Scholar]
- 3.Mazur PK, Siveke JT. Genetically Engineered mouse models of pancreatic cancer:Unravelling tumor biology and progressing translational oncology. Gut. 2011 doi: 10.1136/gutjnl-2011-300756. [DOI] [PubMed] [Google Scholar]
- 4.Yachida S, Jones S, Bozic I, Antal T, Leary R, Fu B, et al. Distant metastasis occurs late during the genetic evolution of pancreatic cancer. Nature. 2011;467:1114–17. doi: 10.1038/nature09515. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Mohammed A, Janakiram NB, Lightfoot S, Gali H, Vibhudutta A, Rao CV. Early Detection and Prevention of Pancreatic Cancer: Use of Genetically Engineered Mouse Models and advanced Imaging Technologies. Cur Med Chem. 2012;19:3701–13. doi: 10.2174/092986712801661095. [DOI] [PubMed] [Google Scholar]
- 6.Weiss TS, Bernhardt G, Buschauer A, Thasler WE, Dolgner D, Zirngibl H, et al. Polyamine levels of human colorectal adenocarcinomas are correlated with tumor stage and grade. Int J Colorectal Dis. 2002;17:381–87. doi: 10.1007/s00384-002-0394-7. [DOI] [PubMed] [Google Scholar]
- 7.Hu HY, Liu XX, Jiang CY, Lu Y, Liu SL, Bian JF, et al. Ornithine decarboxylase gene is overexpressed in colorectal carcinoma. World J Gastroenterol. 2005;11:2244–48. doi: 10.3748/wjg.v11.i15.2244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Subhi AL, Tang B, Balsara BR, Altomare DA, Testa JR, Cooper HS, et al. Loss of methylthioadenosine phosphorylase and elevated ornithine decarboxylase is common in pancreatic cancer. Clin Cancer Res. 2004;10:7290–96. doi: 10.1158/1078-0432.CCR-04-0972. [DOI] [PubMed] [Google Scholar]
- 9.Black 0, Jr, Chang BK. Ornithine decarboxylase enzyme activity in human and hamster pancreatic tumor cell lines. Cancer Lett. 1982;17:87–93. doi: 10.1016/0304-3835(82)90113-6. [DOI] [PubMed] [Google Scholar]
- 10.Meyskens FL, Jr, Gerner EW. Development of difluoromethylornithine as a chemoprevention agent. Clin Cancer Res. 1999;5:945–51. [PubMed] [Google Scholar]
- 11.Meyskens FL, Jr, McLaren CE, Pelot D, Fujikawa-Brooks S, Carpenter PM, Hawk E, et al. Difluoromethylornithine plus sulindac for the prevention of sporadic colorectal adenomas: a randomized placebo-controlled, double-blind trial. Cancer Prev Res. 2008;1:32–38. doi: 10.1158/1940-6207.CAPR-08-0042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Sintoneau AR, Gerner EW, Nagle R, Ziogas A, Fujikawa-Brooks S, Yerushalmi H, et al. The effect of difluoromethylornithine on decreasing prostate size and polyamines in men: results of a year-long phase IIb randomized placebo-controlled chemoprevention trial. Cancer Epidemiol Biomarkers Prev. 2008;17:292–99. doi: 10.1158/1055-9965.EPI-07-0658. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Witherspoon M, Chen Q, Kopelovich L, Gross SS, Lipkin SM. Unbiased metabolite profiling indicates that a diminished thymidine pool is the underlying mechanism of colon cancer chemoprevention by alpha-difluoromethylornithine. Cancer Discov. 2013;3:1072–81. doi: 10.1158/2159-8290.CD-12-0305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Kreul SM, Havighurst T, Kim K, Mendonça EA, Wood GS, Snow S, et al. A phase III skin cancer chemoprevention study of DFMO: long-term follow-up of skin cancer events and toxicity. Cancer Prev Res. 2012;5:1368–74. doi: 10.1158/1940-6207.CAPR-12-0233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Rao CV, Tokumo K, Rigotty J, Zang E, Kelloff G, Reddy BS. Chemoprevention of colon carcinogenesis by dietary administration of piroxicam, alpha-difluoromethylornithine, 16 alpha-fluoro-5-androsten-17-one, and ellagic acid individually and in combination. Cancer Res. 1991;51(17):4528–34. [PubMed] [Google Scholar]
- 16.Meyskens FL, Jr, Gerner EW, Emerson S, Pelot D, Durbin T, Doyle K, et al. Effect of alpha-difluoromethylornithine on rectal mucosal levels of polyamines in a randomized, double-blinded trial for colon cancer prevention. J Natl Cancer Inst. 1998;90:1212–18. doi: 10.1093/jnci/90.16.1212. [DOI] [PubMed] [Google Scholar]
- 17.Mohammed A, Janakiram NB, Brewer M, Ritchie RL, Marya A, Lightfoot S, et al. Antidiabetic Drug Metformin Prevents Progression of Pancreatic Cancer by Targeting in Part Cancer Stem Cells and mTOR Signaling. Transl Oncol. 2013;6:649–59. doi: 10.1593/tlo.13556. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Mohammed A, Janakiram NB, Li Q, Madka V, Ely M, Lightfoot S, et al. The Epidermal Growth Factor Receptor Inhibitor Gefitinib Prevents the Progression of Pancreatic Lesions to Carcinoma in a Conditional LSL-KrasG12D/+ Transgenic Mouse Model. Cancer Prev Res. 2010;3:1417–26. doi: 10.1158/1940-6207.CAPR-10-0038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Mohammed A, Qian L, Janakiram NB, Lightfoot S, Steele VE, Rao CV. Atorvastatin delays progression of pancreatic lesions to carcinoma by regulating PI3/AKT signaling in p48Cre/+-LSL-KrasG12D/+ mice. Int J Cancer. 2012;131:1951–62. doi: 10.1002/ijc.27456. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Guruswamy S, Rao CV. Synergistic effects of lovastatin and celecoxib on caveolin-1 and its downstream signaling molecules: implications for colon cancer prevention. Int J Oncol. 2009;35:1037–43. doi: 10.3892/ijo_00000418. [DOI] [PubMed] [Google Scholar]
- 21.Löser C, Fölsch UR, Paprotny C, Creutzfeldt W. Polyamine concentrations in pancreatic tissue, serum, and urine of patients with pancreatic cancer. Pancreas. 1990;5:119–27. doi: 10.1097/00006676-199003000-00001. [DOI] [PubMed] [Google Scholar]
- 22.Gerner EW, Meyskens FL., Jr Polyamines and cancer: old molecules, new understanding. Nature Reviews Cancer. 2004;4:781–92. doi: 10.1038/nrc1454. [DOI] [PubMed] [Google Scholar]
- 23.Grossie VB, Jr, Ota DM, Ajani JA, Nishioka K. Effects of intravenous a-difluoromethylornithine on the polyamine levels of normal tissue and a transplantable fibrosarcoma. Cancer Res. 1987;47:1836–40. [PubMed] [Google Scholar]
- 24.Kingsnorth AN. The chemotherapeutic potential of polyamine antimetabolites. Ann R Coll Surg Engl. 1986;68:76–81. [PMC free article] [PubMed] [Google Scholar]
- 25.Tutton PJM, Barkla DH. Comparison of the effects of an ornithine decarboxylase inhibitor on the intestinal epithelium and on intestinal tumors. Cancer Res. 1986;46:6091–94. [PubMed] [Google Scholar]
- 26.Laukaitis CM, Gerner EW. DFMO: targeted risk reduction therapy for colorectal neoplasia. Best Pract Res Clin Gasteroenterol. 2011;25:495–506. doi: 10.1016/j.bpg.2011.09.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Esmat AY, Refaie FM, Shaheen MH, Said MM. Chemoprevention of prostate carcinogenesis by DFMO and/or finasteride treatment in male Wistar rats. Tumori. 2002;88:513–21. doi: 10.1177/030089160208800616. [DOI] [PubMed] [Google Scholar]
- 28.Gupta S, Ahmad N, Marengo SR, MacLennan GT, Greenberg NM, Mukhtar H. Chemoprevention of prostate carcinogenesis by alpha-difluoromethylornithine in TRAMP mice. Cancer Res. 2000;60:5125–33. [PubMed] [Google Scholar]
- 29.Bailey HH, Kim K, Verma AK, Sielaff K, Larson PO, Snow S, et al. A randomized, double-blind, placebo-controlled phase 3 skin cancer prevention study of {alpha}-difluoromethylornithine in subjects with previous history of skin cancer. Cancer Prev Res. 2010;3:35–47. doi: 10.1158/1940-6207.CAPR-09-0096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Vlastos AT, West LA, Atkinson EN, Boiko I, Malpica A, Hong WK. Results of a phase II double-blinded randomized clinical trial of difluoromethylornithine for cervical intraepithelial neoplasia grades 2 to 3. Clin Cancer Res. 2005;11:390–96. [PubMed] [Google Scholar]
- 31.Basuroy UK, Gerner EW. Emerging concepts in targeting the polyamine metabolic pathway in epithelial cancer chemoprevention and chemotherapy. J Biochem. 2006;139:27–33. doi: 10.1093/jb/mvj022. [DOI] [PubMed] [Google Scholar]
- 32.Marx M, Townsend CM, Jr, Barranco SC, Glass EJ, Thompson JC. Treatment of hamster pancreatic cancer with alpha-difluoromethylornithine, an inhibitor of polyamine biosynthesis. J Natl Cancer Inst. 1987;79:543–48. [PubMed] [Google Scholar]
- 33.Nilsson JA, Keller UB, Baudino TA, Yang C, Norton S, Old JA, et al. Targeting ornithine decarboxylase in Myc-induced lymphomagenesis prevents tumor formation. Cancer Cell. 2005;7:433–44. doi: 10.1016/j.ccr.2005.03.036. [DOI] [PubMed] [Google Scholar]
- 34.Rounbehler RJ, Li W, Hall MA, Yang C, Fallahi M, Cleveland JL. Targeting ornithine decarboxylase impairs development of MYCN-amplified neuroblastoma. Cancer Res. 2009;69(2):547–53. doi: 10.1158/0008-5472.CAN-08-2968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Wan X, Jiang B, Liu YS, Liu HW, Wu GY, Chen R. Effect of alpha-difluoromethylornithine on the expression of ODC mRNA in the cortex and hippocampus in rats after cerebral ischemia reperfusion. Zhong Nan Da Xue Xue Bao Yi Xue Ban. 2005;30(5):579–82. [PubMed] [Google Scholar]
- 36.Bouchard C, Staller P, Eilers M. Control of cell proliferation by Myc. Trends Cell Biol. 1998;8:202–06. doi: 10.1016/s0962-8924(98)01251-3. [DOI] [PubMed] [Google Scholar]
- 37.Baudino TA, Maclean KH, Brennan J, Parganas E, Yang C, Aslanian A, et al. Myc-mediated proliferation and lymphomagenesis, but not apoptosis, are compromised by E2f1 loss. Mol Cell. 2003;11:905–14. doi: 10.1016/s1097-2765(03)00102-3. [DOI] [PubMed] [Google Scholar]
- 38.Herold S, Wanzel M, Beuger V, Frohme C, Beul D, Hillukkala T, et al. Negative regulation of the mammalian UV response by Myc through association with Miz-1. Mol Cell. 2002;10:509–21. doi: 10.1016/s1097-2765(02)00633-0. [DOI] [PubMed] [Google Scholar]
- 39.Vlach J, Hennecke S, Alevizopoulos K, Conti K, Amati B. Growth arrest by the cyclin-dependent kinase inhibitor p27Kip1 is abrogated by c-Myc. EMBO J. 1996;15:6595–04. [PMC free article] [PubMed] [Google Scholar]
- 40.Tao L, Kramer PM, Wang W, Yang S, Lubet RA, Steele VE, Pereira MA. Altered expression of c-myc, p16 and p27 in rat colon tumors and its reversal by short-term treatment with chemopreventive agents. Carcinogenesis. 2002;23(9):1447–54. doi: 10.1093/carcin/23.9.1447. [DOI] [PubMed] [Google Scholar]
- 41.Raul F. Revival of 2-(difluoromethyl)ornithine (DFMO), an inhibitor of polyamine biosynthesis, as a cancer chemopreventive agent. Biochem Soc Trans. 2007;35:353–5. doi: 10.1042/BST0350353. [DOI] [PubMed] [Google Scholar]
- 42.Liu L, Li L, Rao JN, Zou T, Zhang HM, Boneva D, Bernard MS, Wang JY. Polyamine-modulated expression of c-myc plays a critical role in stimulation of normal intestinal epithelial cell proliferation. Am J Physiol Cell Physiol. 2005;288(1):C89–99. doi: 10.1152/ajpcell.00326.2004. [DOI] [PubMed] [Google Scholar]
- 43.Suzuoki M, Miyamoto M, Kato K, Hiraoka K, Oshikiri T, Nakakubo Y, et al. Impact of caveolin-1 expression on prognosis of pancreatic ductal adenocarcinoma. Br J Cancer. 2002;87:1140–44. doi: 10.1038/sj.bjc.6600619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Hehlgans S, Eke I, Haase M, Baretton G, Cordes N. Caveolin-1 mediated radioresistance of 3D grown pancreatic cancer cells. Radiother Oncol. 2009;92:363–70. doi: 10.1016/j.radonc.2009.07.004. [DOI] [PubMed] [Google Scholar]
- 45.Klinken SP, Holmes KL, Morse HC, 3rd, Thorgeirsson SS. Transcriptional and post-transcriptional regulation of c-myc, c-myb, and p53 during proliferation and differentiation of murine erythroleukemia cells treated with DFMO and DMSO. Exp Cell Res. 1988;178(2):185–98. doi: 10.1016/0014-4827(88)90390-4. [DOI] [PubMed] [Google Scholar]
- 46.Celano P, Berchtold CM, Giardiello FM, Casero RA., Jr Modulation of growth gene expression by selective alteration of polyamines in human colon carcinoma cells. Biochem Biophys Res Commun. 1989;165(1):384–90. doi: 10.1016/0006-291x(89)91082-6. [DOI] [PubMed] [Google Scholar]
- 47.Gerner GW. Impact of dietary amino acids and polyamines on intestinal carcinogenesis and chemoprevention in mouse models. Biochem Soc Trans. 2007;35:322–5. doi: 10.1042/BST0350322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Juan J, Muraguchi T, Iezza G, Sears RC, McMahon M. Diminished WNT -> β-catenin -> c-MYC signaling is a barrier for malignant progression of BRAFV600E-induced lung tumors. Genes Dev. 2014;28(6):561–75. doi: 10.1101/gad.233627.113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Mohammed A, Janakiram NB, Brewer M, Duff A, Lightfoot S, Brush RS, et al. Endogenous n-3 Polyunsaturated Fatty Acids delay progression of Pancreatic Ductal Adenocarcinoma in Fat-1. P48Cre/+-LSL-KrasG12D/+ mice. Neoplasia. 2012;14:1249–59. doi: 10.1593/neo.121508. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Rao CV, Mohammed A, Janakiram NB, Qian L, Ritchie RL, Lightfoot S, et al. Inhibition of Pancreatic Intraepithelial Neoplasia Progression to Carcinoma by Nitric Oxide-Releasing Aspirin in p48Cre/+-LSL-KrasG12D/+ Mice. Neoplasia. 2012;14:778–87. doi: 10.1593/neo.121026. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.