Abstract
Objective
In a multicenter study of newly diagnosed ALS patients without a reported family history of ALS, we are prospectively investigating whether markers of oxidative stress (OS) are associated with disease progression.
Methods
An extensive structured telephone interview ascertained environmental, lifestyle, dietary and psychological risk factors associated with OS. Detailed assessments were performed at baseline and at 3 to 6 month intervals during the ensuing 30 months. Our biorepository includes DNA, plasma, urine, and skin.
Results
355 patients were recruited. Subjects were enrolled over a 36 month-period at 16 sites. To meet the target number of subjects, the recruitment period was prolonged and additional sites were included. Demographic and disease characteristics were similar between 477 eligible/non-enrolled and enrolled patients, with the only difference being type of health insurance among enrolled patients. Sites were divided into 3 groups by the number of enrolled subjects. Comparing these 3 groups, the Columbia site had fewer “definite ALS” diagnoses.
Conclusion
This is the first prospective, interdisciplinary, in-depth, multicenter epidemiological investigation of OS related to ALS progression and was accomplished by an aggressive recruitment process. The baseline demographic and disease features of the study sample are now fully characterized.
Keywords: ALS, Oxidative Stress, Disease Progression, Survival, Epidemiology
INTRODUCTION
Diverse environmental and lifestyle factors associated with amyotrophic lateral sclerosis (ALS), include aging, environmental exposures, viral infections, physical exertion, body habitus, diet, smoking, and occupation (1). Many of these factors are associated with oxidative stress (OS). OS is defined as a disruption in the pro- and anti-oxidant balance with increases occurring in reactive oxygen and nitrogen species. In ALS, OS is intimately linked to a series of cellular events in motor neurons that apparently contribute to neuronal degeneration and death (2-4). Much of what is hypothesized concerning the cause of ALS comes from molecular studies of specific SOD1 mutations linked to ALS (2, 5, 6). These studies also point to a role for neuronal OS in the etiology of ALS. However, few data are available to determine whether the presence of OS biomarkers or exposure to agents related to OS mechanisms are associated with ALS progression; this forms the overall hypothesis of our study.
While the onset of ALS is insidious, once diagnosed, disease progression is both observable and measurable. Progression is highly variable: some patients have a rapid disease course, whereas others have a much slower course. Here, we describe the ALS COSMOS study, which was designed to determine whether markers of OS, measured either directly in blood or urine or indirectly by structured interviews, are associated with ALS disease progression. We describe the study structure, methods, recruitment, and demographic and disease characteristics of this multicenter case-only cohort study in patients with ALS without family history.
PATIENTS AND METHODS
IRB Approval
The study protocol, including informed consent procedures and HIPAA compliance was approved by the Institutional Review Board (IRB) of each participating site and of Columbia University.
Development of the Structured Questionnaire
Our instrument was developed with the advice of outside ALS experts (Drs. Lorene Nelson, Edward Kasarskis, and Ammar Al-Chalabi) in 2003. We first used this instrument to query diagnostic, demographic, familial, lifestyle and other risk factors in 91 sALS cases and 121 controls, participating in a genetic epidemiological study funded by the MDA. Out of 91 patients, 67 living patients agreed to be enrolled in the ALS COSMOS study.
We evaluated the test-retest reliability of the questionnaire for in-person versus telephone administration in 29 ALS cases and 29 controls. Cases and controls were randomly assigned to either an in-person interview followed by a telephone interview or to a telephone interview followed by an in-person interview. For most items, reliability was in the good to excellent range (kappa ≥ .85) (7). For items pertaining to neurological history and physical activity, the reliability was in the moderate range (kappa ranging from .62 to .65) (8). To improve overall reliability, redundant and confusing questions were eliminated from the final instrument.
Participating Sites and Study Enrollment
Originally 11 study sites were selected based on experience with ALS studies and previous collaboration with Columbia ALS studies (7, 8). The site locations are geographically dispersed throughout the United States. Although we planned to recruit 1 to 2 new patients every month per site for a two-year recruitment period, enrollment was slower than expected. We therefore added 5 sites at the end of the 2nd year (see Figure 1 and the ALS COSMOS sites listed at the end of the manuscript) and extended the enrollment period by 12 months for a total of 3 years. To encourage and support site-based recruitment, monthly conference calls and biannual investigators’ meetings were held with the principal investigators and coordinators during the course of the study.
Figure 1.
This is a multicenter design consisting of 16 ALS clinic study sites. The distribution of sites is fairly evenly dispersed throughout the Unites States.
New Patient Log
Each study site was asked to collect de-identified data on all new ALS patients at their clinic, including demographics, insurance type, referral source, disease duration after symptom onset to the diagnosis, the El Escorial Criteria (EEC) for ALS diagnostic categories, participation in a clinical trial and use of alternative therapy. Screening data were used to determine: (1) overall eligibility of patients at the site; (2) reasons for non-participation among eligible patients and (3) whether enrolled patients represented the overall patient population.
Patient Eligibility
ALS was diagnosed as “definite,” “probable,” or “possible” according to the EEC/Airlie House revision, widely used in current clinical trials (9). We added the new Awaji Criteria, which increases the chance of early diagnosis due to its acceptance of electrophysiological criteria as equivalent to the clinical criteria (10). Bulbar-onset or spinal-onset ALS, predominantly upper motor neuron (PLS type) or lower motor neuron (PMA type), were all included. Inclusion and exclusion criteria are shown in Table 1.
Table 1.
Inclusion and Exclusion Criteria.
|
Inclusion Criteria:
|
| • Disease duration of 18 months or less after symptom onset. |
| • At least age 20 years and older. |
| • A reliable family caregiver who gives independent Informed Consent for providing the information from structured interviews. |
| • English fluency due to intensive structured interviews and diet questionnaires |
| • The capacity to consent (by performing a capacity assessment (39), i.e. patient able to understand and sign the Informed Consent approved by the IRB at each study site and also sign the form for HIPAA regulations. |
| • Willing to return to the site for follow-up examinations. |
|
Exclusion Criteria: |
| • Familial ALS (fALS) based on a clear history of disease in first-degree relatives or a positive fALS molecular test results, such as SOD1 or C9ORF72 mutations. |
| • Already participating in clinical trials prior to enrollment. Those who participate in a trial after enrollment in this study remain eligible for follow up. |
| • Major active neurological diseases other than ALS or a history of major neurological diseases. |
| • Major unstable medical diseases that require active medical treatment (e.g., active cancer, dialysis) in the past 6 months. |
Enrollment, Case Ascertainment and Study Assessment
Clinical Assessment
Table 2 shows the study schedule of assessments for baseline and follow-up visits. Table 3 details items on enrollment and case ascertainment forms. Clinical assessment included the ALS Functional Rating Scale-Revised (ALSFRS-R), forced vital capacity (FVC), and a comprehensive neurological examination. The primary outcome for the study was defined as death or tracheostomy/long-term mechanical ventilation, endpoints used in most ALS clinical trials (7, 11-15). All of the study sites had participated in ALS clinical trials, and the evaluators and coordinators at each site were highly experienced with the measures used in this study. Detailed clinical assessments were performed at baseline and 3 months following enrollment, thereafter at 6 month intervals for 24 months or until death/outcome or study drop out. Survival status for all enrolled patients was confirmed at Month 30 after enrollment.
Table 2.
Procedure Schedule at Individual Sites and the Columbia University Coordinating Center Coordinating Center (CUCC).
| Study Activity | Month 0 Baseline | 3 | 6 | 12 | 18 | 24 End | 30 |
|---|---|---|---|---|---|---|---|
| Done at individual study sites | |||||||
| Eligibility | X | ||||||
| Informed Consent | X | ||||||
| Medical Evaluation & Assessment | X | X | X | X | X | X | |
| Physical Exam | X | X | |||||
| Neurological Exam | X | X | |||||
| Cognitive Screening | X | X | |||||
| Forced Vital Capacity | X | X | X | X | X | X | |
| ALSFRS-R | X | X | X | X | X | X | |
| Blood and urine | X | X | X | X | X | X | |
| Drug list | X | X | X | X | X | X | |
| Final Survival | X | ||||||
| Web-based data entry | X | X | X | X | X | X | X |
| Done by the CUCC | |||||||
| Baseline interview | X | ||||||
| Follow up interview | X | X | X | X | X | ||
| Brief FFQ | X | X | X | X | |||
| Psychological interview | X | X | X | X | X | X | |
Table 3.
Description of Case Enrollment and Ascertainment Data Collection.
| Items | Number of Items Administered |
|---|---|
| Subject initials | 1 |
| Capacity Assessment Record | 13 |
| Case Eligibility Form | 12 |
| Enrollment Form | 4 |
| ALS Center physician | 1 |
| Date of birth | 1 |
| Date of enrollment | 1 |
| Date of diagnosis | 1 |
| Date of symptom onset | 1 |
| Duration between symptom onset and enrollment | 1 |
| Anatomical region of onset | 1 |
| LMN and UMN Signs | 54 |
| Diagnostic Testing | 5 |
| Familial ALS | 6 |
| Atypical Features | 4 |
| Disease Progression | 2 |
| Diagnostic Certainty | 1 |
| PLS or PMA | 1 |
| Total items (exam or question) | 110 |
Cognitive Impairment
To screen patients for fronto-temporal dementia (FTD), we used the UCSF Cognitive Test Battery for FTD, which is comprised of the ALS Cognitive Behavioral Screen (ALS-CBS) (16), either the Abrahams phonemic verbal fluency or the Controlled Oral Word Association Test, Frontal Behavioral Inventory (FBI) and Center for Neurological Study (CNS) Lability Scale (see Table 4). The assessment involved both patients and caregivers as outlined in Table 4 and averaged 20 to 30 minutes. FTD screening was performed at enrollment and month 12 (17). Special training for cognitive assessment was given to all the evaluators, and a refresher training video (developed by one of the co-authors, J. Murphy) was made available to the evaluators as well.
Table 4.
University of California San Francisco (UCSF) Cognitive Test Battery for Fronto-Temporal Dementia (FTD).
| 1) ALS Cognitive Behavioral Screen (ALS-CBS) (16) |
| a. Patient's portion |
| b. Caregiver's portion |
| 2) Either the Abrahams phonemic verbal fluency or the Controlled Oral Word Association Test (to be used in place of the Written Verbal Fluency Test if significant hand weakness is present) (40) |
| 3) Frontal Behavioral Inventory (FBI) (41) –Caregiver only. |
| 4) Center for Neurological Study (CNS) Lability Scale (42) |
Biospecimen Collection and Biomarker Analyses
Overnight fasting blood samples were obtained. Blood was immediately processed to obtain aliquots of white blood cells, plasma and whole blood. First void urine specimens were also obtained. Biospecimens were collected at the baseline visit and at each follow-up visit at the clinical site. All samples were stored at -80°C and shipped on a quarterly basis on dry ice to the Biomarkers Core Facility (RS) at Columbia University, where sample custody was monitored using a secure web-based database that allowed a full inventory of stored samples and shipment records. Skin biopsy and blood in PAXgene tubes for RNA were added as additional biosamples. Biospecimens will be used to measure the OS biomarkers, urinary F2-isoprostanes and 8-oxodeoxyguanosine (18). They will also be used to analyze plasma lipid profiles (19, 20). The remaining biospecimens represent a resource for future studies.
Selected Exposure Analyses
We obtained data on exposures of interest and on potential confounding variables using the structured questionnaires described in eTables 1A and 1B. The exposures of interest included chemical and physical exposures associated with residence, workplace or hobbies; physical activity; and lifestyle factors, such as diet (see below), alcohol use, smoking, and psychological stress. Follow-up questionnaires queried about patients’ exposures since previous assessments.
Occupational Exposure Assessment
Occupational history was assessed using a semi-structured questionnaire developed by an industrial hygienist (LA). We focused in particular on occupational exposures previously thought to be associated with oxidative stress and ALS, notably, exposure to lead, pesticides, electromagnetic fields and military service. For subjects reporting an occupation thought to be associated with one of these four exposures, the questionnaire (1) identified specific agents to which they were most likely exposed and (2) assessed the extent and duration of exposures to those agents or factors. Interviewers followed a comprehensive and detailed protocol to ascertain characteristics of the work environment in which these exposures occurred, using visual aids (ventilation designs, personal protective equipment, etc.) to assist the proper identification of worksite-specific conditions and exposure-specific questionnaires (such as exposure to lead, agricultural chemical, electromagnetic fields, etc.) to estimate the extent and duration of exposure.
Assessment of Dietary Variables
To assess diet and specifically macro and micro nutrients associated with OS, we used a modification of the Block 2005 Food Frequency Questionnaire (FFQ).The modification entailed shortening the questionnaire so completion time would be approximately 15-20 minutes. Various brief versions of the original Block questionnaire have been validated in several studies (21-24). Our shortened FFQ assessed specific intake information, particularly anti-oxidants including multivitamins; antioxidant combination vitamins; vitamins A, C, and E; β-carotene; calcium; iron; zinc; and selenium. We asked the participants to self-administer the shortened Block FFQ at baseline and at the follow up visits (eTable 2). When subjects were unable to self-administer the FFQ, caregivers were asked to assist or an interviewer and subject completed the FFQ by phone. All completed FFQ forms were scanned by Nutritionquest® (formerly Block Dietary Data Systems), a company that designs nutrition questionnaires and provides dietary analysis services (25). Nutritionquest® converted FFQ data into average daily nutrient intake estimates using a standardized reference nutrient database, provided food-specific intake data as well as calculated macro- and micro-nutrient intake.
Assessment of Psychological Factors
Previous literature suggests that perceived stress may be associated with OS (26-28). We assessed perceived stress, depressive symptoms, and hopelessness, as well as factors which may mitigate the stress experience, including quality of life, hope for the future, and positive/negative mood using standardized measures. These measures were chosen because they 1) have been successfully used in the past with ALS patients (29-31), 2) have standardized norms, 3) are feasible for telephone administration, and 4) do not require extensive verbal communication. The content of the scales was also chosen to minimize patient fatigue and potential distress. Clinical psychologists at Columbia University (JR and MM) selected the instruments and were responsible for the assessment and interpretation of these psychological scales (see eTable 3).
Interview Training and Assisted Interview
The CUCC interviewers administered all interviews by telephone except for the self-administered dietary questionnaire. The interviewers were fully trained by the investigators at Columbia University, who were responsible for specific content areas. The interviews were in general limited to one hour, unless the patients wanted to finish during a single call. The interviewer monitored the patient's fatigue level and most interviews were divided into 2 occurrences and occasionally into more.
Depending on the patient's ability to verbally communicate, the spouse, a family member or friend was able to assist during the interview. We devised and validated the interview method by a study in which 18 non-dysarthric patients were interviewed by a standard telephone interview and then re-interviewed with the assistance of a family caregiver. The patients were not allowed to have any verbal or written communication with the family member but instead were asked to rely only on head nods, finger signals and other body language. We compared responses between the two methods on 98 selected key questionnaire items, including occupation, hobbies, physical activities, diet, and health issues, and used intraclass correlation statistics and kappa statistics when appropriate. Fifty items (51%) showed 100% agreement. Of the remainder, agreement was mostly in the moderate to excellent range, kappa = .5 to .9.
Data Management
All data were managed by the Data Coordinating Center (DCC), Mailman School of Public Health Biostatistics Department. Clinical assessments, interview questionnaire data, biosample collection status, and laboratory results were entered into structured web-based data entry screens and transmitted to a central relational database at the DCC. Security of web-based data transfer was established using Citrix authentication and encrypted transmission. DCC data management procedures have been certified by the Columbia University Information Security Office.
Statistical Analyses
We used t-tests and chi square statistics to compare eligible patients who were and were not enrolled. The overall study aims were to estimate associations between exposure to factors associated with OS and disease progression. For the overall study, we estimated the minimum detectable effect size for a sample of 350 patients, given a type 1 error of .01 (to be conservative due to the multiple comparisons) and power of 80%. This resulted in a minimum detectable correlation of .185 between exposure and outcome.
RESULTS
A total of 355 patients were eligible/enrolled (see below), and 477 potentially eligible/non-enrolled patients were reported by the sites in the screening logs. There were no differences between those enrolled and those not enrolled in demographics, disease duration, EEC diagnostic groups, clinical trial participation and use of alternative treatments. At the Columbia site only, enrolled patients were referred more by private neurologists and less by doctors other than neurologists compared to eligible non-enrolled patients (p = .006). However, there were no overall differences in the referral types among the two other groups. Regarding insurance coverage, enrolled patients were more likely to have private insurance and less likely to be covered by Medicare compared to the non-enrolled eligible group (p = .003) (Table 5). The most common reasons that potentially eligible patients did not enroll were 1) the study was not discussed with the patient (53%), 2) patients refused for other reasons (30%), 3) patients were overwhelmed with the diagnosis (8%), and 4) others (9% in total), including patients were too sick (3%), too busy (1.3%), off the originally-set monthly recruitment deadline (4.3%), and had issues with informed consent (0.4%).
Table 5.
Differences in Insurance Coverage between Eligible and Enrolled Patients.
| Insurance type | Eligible and non-enrolled (n=477) | Eligible and enrolled (n=3541) |
|---|---|---|
| Private insurance2 | 51% | 64% |
| Medicare | 42% | 29% |
| Medicaid | 2% | 2% |
| Veterans Affairs | 1% | 1% |
| No insurance | 2% | 1% |
| Unknown | 2% | 3% |
Screening data was missing for one.
Private insurance includes Workers' compensation, Union, and individual insurances.
Over the three year recruitment period, we enrolled a total of 358 patients. Of these patients, three were disqualified as they were later found to not meet the inclusion criteria, resulting in a total of 355 patients. The overall enrollment status is summarized in Figure 2 and suggests a slower subject accrual period in the middle of the study. Site-specific enrollment is shown in Figure 3. The Columbia University Coordinating Center (CUCC) was the highest enrolling center and already, prior to any other center, enrolled 67 patients, who were investigated under the same protocol in an earlier MDA funded study (see above) and then subsequently enrolled 72 new patients for the ALS COSMOS study.
Figure 2.
The overall enrollment performance is demonstrated. The anticipated target was 350 patients (broken line). The Columbia University Coordinating Center enrolled 67 live patients from an earlier study, who agreed to participate in this study, and also began recruiting patients before other sites. Actual enrollment (solid line) showed a clear slowdown in the middle of the enrollment period, to 4.5 patients per month. Although the ceftriaxone clinical trial continued their enrollment throughout most of the COSMOS study, other clinical trials enrolled in the middle of the COSMOS study, perhaps one of the reasons why enrollment slowed down. TL: talampanel, NP: NP9001, DX: dexpramipexole, CK: CK2017357
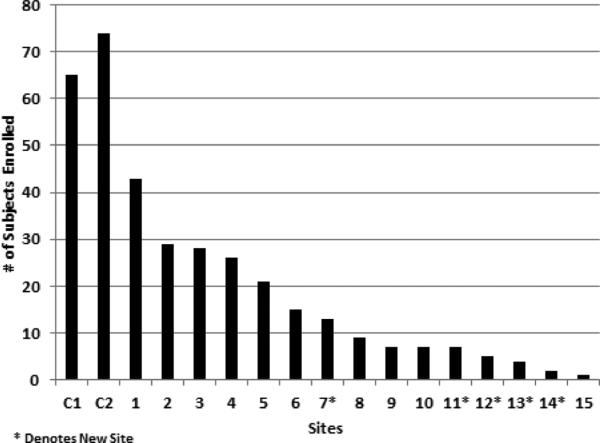
Figure 3.
The Columbia site had two patient populations composed of C1, an earlier MDA study (67 patients), and C2, usual recruitment based on the current NIH ALS COSMOS study.
The recruitment numbers markedly varied among study sites (Fig 3). The average number of all enrolled patients after the recruitment began was 0.5 per month and per active site. To further analyze the effects of imbalanced enrollment that may have skewed the data, we divided the sites into three groups based on average enrollment number per month: the CUCC site, which enrolled on average 2.0 patients per month after recruitment began at other sites, 72 patients in total; ‘high’ enroller group, which included 6 sites with an average > 0.5 patients per month, 160 patients in total; and ‘low’ enroller group, which included 9 sites with an average ≤ 0.5 patients per month, 56 patients in total.
The number of eligible/non-enrolled patients and the ratio of enrolled patients are described in Table 6. Sex, age, racial and ethnic background and educational attainment were not different among sites grouped according to enrollment (Fig 4 and Table 6). Disease characteristics are summarized in Table 7. Although no differences were found in disease characteristics, such as disease duration, ALSFRS-R, percentage of predicted FVC, and the body region of onset, there were significant differences in diagnostic certainty based on the EEC among the three enrollment groups with more definitive ALS category at the Columbia site.
Table 6.
Demographic Characteristics of Enrolled Patients by Sites Defined by Number of Enrolled Subjects.
| CUCC (n=139) | High Enroller (n=160) | Low Enroller (n=56) | Total (n=355) | |
|---|---|---|---|---|
| Eligible/non-enrolled patients | 249 | 110 | 118 | 477 |
| Percentage of enrolled patients among all eligible patients | 34% | 59% | 32% | 43% |
| Male:Female (%) | 58:42 | 61:39 | 61:39 | 60:40 |
| Age (mean±s.d.) | 62±10 | 60±11 | 61±9 | 61±10 (Range: 27 - 90) |
| Race | ||||
| White | 85% | 92% | 89% | 88% |
| African American | 6% | 4% | 6% | 6% |
| Asian | 4% | 1% | 0% | 2% |
| Others | 4% | 3% | 6% | 4% |
| Ethnicity Hispanic/Latino Educational | 5% | 2% | 6% | 4% |
| Less than high school | 3% | 5% | 0% | 3% |
| High school or GED | 23% | 19% | 23% | 21% |
| Some college, AA or technical college | 23% | 32% | 32% | 28% |
| 4-year college | 25% | 25% | 25% | 25% |
| Masters, doctorate or higher | 26% | 20% | 21% | 22% |
CUCC: Columbia University Coordinating Center. GED: general education development. AA: Associate of Art. There are no differences in the demographic features among the 3 enrolling groups of sites.
Figure 4.
Mean age for males was 59.7 ± 10.8 years (median=60.3 years) and 63.2 ± 9.2 years (median=64.3 years) for females.
Table 7.
Disease Characteristics of Enrolled Patients.
| CUCC (n=139) | High Enroller (n=160) | Low Enroller (n=56) | Total (n=355) | Comments | |
|---|---|---|---|---|---|
| Disease duration (months) | 11.6±4.8 | 11.5±4.4 | 11.3± 4.9 | 11.5±4.6 | Symptom onset to enrollment |
| ALSFRS-R | 36±6 | 36±7 | 37±6 | 36±7 | Range (7-47) |
| FVC% of expected | 79±22 | 82±22 | 75±23 | 80±23 | Rage (20-138) |
| Region of onset | B (31%) | B (28%) | B (39%) | B (31%) | |
| C (33%) | C (39%) | C (25%) | C (35%) | ||
| LS (35%) | LS (30%) | LS (30%) | LS (32%) | ||
| Others (1%) | Others (3%) | Others (5%) | Others (3%) | ||
| EEC Criteria | Chi-square | ||||
| Possible | 39%* | 20% | 20% | 27% | (P<0.001) |
| Probable | 47% | 37% | 34% | 40% | |
| Definite | 4%* | 40% | 45% | 27%* | |
| PMA | 9%* | 3% | 2% | 5% | |
| PUMND | 1% | 1% | 0% | 1% | |
CUCC: Columbia University Coordinating Center - high enroller site.
indicating the diagnostic categories leading to key differences in the diagnosis group. ALSFRS-R: ALS Functional Rating Scale-Revised. FVC: forced vital capacity. EEC: El Escorial Criteria. PMA: lower motor neuron type. PUMND: pure upper motor neuron disease
Discussion
The ALS COSMOS study was not a population-based epidemiological study and thus patients who had a very short course of the disease may have not been included. Further, although our study showed no enrollment selectivity by age, older patients may have not been referred to the ALS Clinic and may have been potentially excluded from center-based studies (32, 33). Our study was designed to investigate associations between oxidative stress (OS) and ALS disease progression in a multicenter design. Currently, we are investigating multiple risk factors in the life course environment, including environmental, occupational, lifestyle, dietary, and psychological factors (1). To find a cause and pathogenesis of ALS, more well-designed, in-depth epidemiologic studies are needed (34). Table 8 lists recent major epidemiologic studies, showing highly diverse studies focused on many different patient populations and objectives. Although population-based studies provide the best quality data for many questions, such studies are more difficult to conduct in the US than in European countries because of its distributed healthcare system (35). The National ALS Registry, which has been established in the US, aims to develop a population-based study in the future (36). Multicenter-based studies are clearly an alternative way to study a large number of ALS patients for epidemiologic investigations.
Table 8.
Recent Epidemiologic Studies in ALS.
| Studies (the first author) | Location of the study | ALS Patient # | Study Design | Study Objectives |
|---|---|---|---|---|
| Kamel 2002 (43) Chen 2007 (44) Fang 2009 (45) |
Boston, MA | 109 | Two center-based studies, CCS | Risk: self-reported occupational exposure to smoking, and head trauma. Lead exposure study |
| Sorenson 2002 (46) Mateen 2010 (47) |
Olmsted County, MN | 94 | PB | Incidence, long surviving cases |
| Horner 2003 (48) | USA | 107 | Military population | Incidence and risks |
| Weisskopf 2004 (49) Wang 2011 (50) Ascherio 2005 (51) Weisskopf 2005 (52) |
USA1 | 621 deaths or 832 patients | Cohort studies (see below) and Military service-subpopulation | Incidence; risks: cigarette smoking; Vitamin E; and military service |
| Abhinav 2007 (53) Johnston 2006 (54) |
South-East England | 138 | PB | Incidence and prevalence |
| Popat 2006 (55)& 2007 (56) | Northern California, USA | 111 (65 female) | Kaiser Permanente patient-based, CCS | Risks: reproductive factors and postmenopausal hormone (female only); NSAID effects |
| Forbes 2007 (57) | Scotland | 1222 | PB | Incidence and ascertainment of ALS |
| Chio 2009 (58) | Italy | 7 | Professional soccer players (n=7325) | Risks: soccer players |
| Atsuta 2009 (59) | Japan | 3428 (prevalent) | ALS Registration | Cross-sectional overview, Tracheostomy invasive ventilator |
| Zaldivar 2009 (60) | Cuba | 433 | Central Mortality Statistics | Mortality rate in different Hispanic populations |
| O'Toole 2008 (61) Yeo 2010 (62) Phukan 2011 (63) |
Ireland2 | 465 or 398 deaths | PB, Death certificate based and CCS | Incidence, survival, demographic and clinical features, co-morbid dementia occurrences |
| Donaghy 2009 (64) & 2010 (65) | Ireland3 | 109 | PB | Incidence |
| Hanby 2011 (66) | UK | 1502 | Center-based | ALS risks in siblings and offspring |
| Huisman 2011 (67) de Jong 2012 (68) |
Netherlands | 1128 (incident) and 833 (prevalent) | PB, PB and OOS | Incidence, prevalence, and disease characteristics, current smoking, alcohol consumption |
| Beghi 2007 (69) | Lombardi, Italy | 517 | PB | Incidence |
| Sabatelli 2012 (70) | Italy | 1757 | ITALSGEN4, PB, OOS | C9ORF72 mutations |
| Ohio 2012 (71) Ohio 2012 (72) Ohio 2010 (73) Ohio 2011 (74) |
Italy | 1332 | PARALS5, PB | Risks: genetic mutations Non-invasive ventilation & tracheostomy Phenotypic heterogeneity |
| Logroscino 2010 (75) | Ireland, UK, Italy | 1028 | PB | Incidence |
This is not an exhaustive list of epidemiologic studies and is limited to ‘classical’ ALS cases and studies published after 2000, cases studied involved were at least ~100. Endemic ALS cases are not included. A number of studies derived from one clearly identifiable study population were collapsed into one group. The number of patients varies depending on the study purpose (please refer to the references).
American Cancer Society's Cancer Prevention Study II (OPS II), Nurses' Health Study (NHS), Health Professionals Follow-up Study (HPFS), Multiethnic Cohort (MEO), NIH-American Association of Retired Persons Diet and Health Study (AARP-DH)
The Irish ALS Register
Registers of Northern Ireland (NI) and the Republic of Ireland (ROI)
Italian ALS Genetic Oonsortium (ITALSGEN)
Piemonte and Valle d'Aosta Register for ALS (PARALS).
Other abbreviations: OOS=case-control study; PB=population-based; NSAID=nonsteroidal anti-inflammatory drugs.
While an attractive and well-thought-out design is essential in highly innovative epidemiologic studies, effective patient recruitment is equally critical. Although our study was successful in reaching beyond its goal of the 350 patients required for statistical power, the study faced major challenges in enrollment. In fact, extensive recruitment efforts were made, including newsletters to patients and study sites and continuing encouragement to enrolling sites. Despite these efforts, it took three years to reach our target recruitment goal. To attain this goal, we modified the original protocol by extending the enrollment period (originally 2 years) and adding 5 additional clinical sites. After the first year, there was a substantial slowdown in enrollment, which coincided with large multicenter trials, as shown in Figure 2. Other factors that made recruitment a challenge include: 1) the design of the study, which requires many visits, and multiple outcome measures; these may have been perceived as burdensome not only by the patients but also by the site teams; 2) the observational nature of the study (as opposed to a clinical trial) possibly resulted in a perception of less potential benefits for patients and their clinicians. Prior analyses in clinical trials have not shown trial design factors to be significant determinants of enrollment (37). Furthermore, prior work suggests that “patient factors” and “doctor factors” are more important determinants of enrollment, at least in clinical trials (38). Patient factors include perceived fear of unexpected costs. This may relate to the one statistically significant difference in demographics between enrolled and potentially eligible, non-enrolled patients, i.e. insurance coverage, where subjects with privately purchased health insurance were more likely to enroll. Doctor factors may have contributed to our enrollment as 1) there was a large variation in the enrollment rate among sites, and 2) the main reason for eligible patients not enrolling was the fact that the study was not offered to them. Timely recruitment is crucial particularly in Federally-funded research. Our experience suggests a serious need to investigate and improve patient enrollment.
It is important to ensure that enrolled patients represent the eligible patient base in each study site. Other than the difference in insurance coverage, demographic, disease duration (symptom onset to the diagnosis), and disease characteristics were comparable for non-enrolled eligible and enrolled patient populations. Further, among enrolled patients, although the diagnostic certainty based on magnitude of enrollment, there were no differences in demographic and disease characteristics. Thus, it is fair to say that our study likely represents the patient population. For the factors that did vary as a function of enrollment status and site, we will determine whether these selection factors are potential confounders of the effects that emerge from the analyses that we will conduct at the completion of the study.
Supplementary Material
ACKNOWLEDGEMENT
We are deeply grateful for the patients and their families who enthusiastically participated in this labor intensive study. We would like to express our thanks to Dr. Annette Kirshner at NIEHS for her kind advice and strong support. NIH (NIEHS; R01ES016348) funded the entire study and MDA funded an earlier study, which was incorporated into the ALS COSMOS study. MDA Wings, Adams Foundation, and Ride for Life supported part of the study as well. Georgia Christodoulou, M.A. reviewed the manuscript.
Footnotes
ALS COSMOS Study Group: Site Investigators
Columbia University Coordinating Center, New York, NY: Hiroshi Mitsumoto, M.D., D.Sc., Jonathan Hupf, B.A., Jess Singleton, B.A., Christa Campanella, B.S., David Merle, BS, Tejal Shah, BS, Meredith Pasmantier Kim, B.A., Yei-Won Lee, B.A., Georgia Christodoulou, M.A., Kate Dalton, M.S., RD, Jessica Kidd, B.A., Erin Gilbert, B.A., Mary Kilty, MBA, MPH; Texas Neurology, P.A., Dallas, TX: Daragh Heitzman, M.D., FAAN, Wendy Rodriguez, NRCMA, Shari Hand, B.S., CCRC, Michelle Washington, NRCMA, Brent Spears, B.S., CCRC, Brandie Burson, NRCMA; Duke University, Durham, NC: Richard S. Bedlack, M.D., Ph.D., M.S., Karen Grace, RN, BSN, Candace Boyette, RN, FNP; California Pacific Medical Center, San Francisco, CA: Jonathan S. Katz, M.D., Robert G. Miller, M.D., Dallas Forshew, RN, BSN, Joni Beemsterboer, M.P.H., Will Harris, B.A., Shelley McCoy, B.S., Thaïs Zayas-Bazan, B.A., Chow Saephanh, B.A.; University of Kansas, Kansas City, KS: Richard J. Barohn, M.D., April L. McVey, M.D., Mazen M. Dimachkie, M.D., Mamatha Pasnoor, M.D., Yunxia Wang, M.D., Maureen Walsh, B.S., Laura Herbelin, B.S., CCRP, JoAnn Miller, B.S., Kristy Anderson; Mayo Clinic, Rochester, MN: Eric J. Sorenson, M.D., Sherry Klingerman, CCRP, Delana Weis, LPN, CCRP; University of California, Davis, Sacramento, CA: Björn Oskarsson, M.D., Nanette Joyce, D.O, M.A.S., Steffany Lim, B.S., CCRP, Michelle Cregan, CRA; University of Kentucky, Lexington, KY: Edward J. Kasarskis, M.D., Ph.D., Kathie Vanderpool, RN, B.S., Deborah Taylor, M.S., Samantha Thomas, M.S., Jason King, M.S., Robert Wells, CCRP; University of California, San Francisco, San Francisco, CA: Catherine Lomen-Hoerth, M.D., Ph.D., Jennifer Murphy, Ph.D. (UCSF, Cognitive Psychology), Y-Nhy Duong, B.A., Dennis Robins, B.A., Claudia Villerme, M.Ed.; University of Colorado, Aurora, CO: Yvonne D. Rollins, M.D., Ph.D., Steven P. Ringel, M.D., Dianna Quan, M.D., Elizabeth Whitethorn, B.S.; University of California, Irvine, Orange, CA: Tahseen Mozaffar, M.D., Ph.D., Annabel K. Wang, M.D., Veronica Martin, B.A., Brian Minton, B.S., B.A., Patricia Tully, FNP, Denise Davis; University of Nebraska Medical Center, Omaha, NE: J. Americo M. Fernandes Filho, M.D., Pariwat Thaisetthawatkul, M.D., Cindy Cowardin, RN, BSN, CCRP, Russell Herstein, B.A.; University of Iowa, Iowa City, IA: Andrea J. Swenson, M.D., Decontee Jimmeh-Fletcher, M.D., Heena Olalde, RN, MSN, Jeri Sieren, RN; University of Texas - Southwestern, Dallas, TX: Sharon P. Nations, M.D., Jeffrey Elliott, M.D., Jaya Trivedi, M.D., Nina Gorham, CCRP; SUNY - Upstate Medical University, Syracuse, NY: Jeremy M. Shefner, M.D., Ph.D., Mary Lou Watson, B.A., RRT, CCRP, Jennifer Moore, Katie Markis, B.A., CCRP, Megan Grosso, RPA-C, DPT, PT; Hospital for Special Care, New Britain, CT: Jinsy A. Andrews, M.D., M.S., Agnes Koczon-Jaremko, M.D., Janet Bowen, B.A., CRT.
Publication Committee: Björn Oskarsson, M.D., Eric J. Sorenson, M.D., and J. Americo M. Fernandes Filho, M.D.
References
- 1.D'Amico E, Factor-Litvak P, Santella RM, Mitsumoto H. Clinical perspective of oxidative stress in sproradic ALS. Free Rad Biol Med. 2013;65:509–527. doi: 10.1016/j.freeradbiomed.2013.06.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Andersen JK. Oxidative stress in neurodegeneration: cause or consequence? Nature medicine. 2004;10(Suppl):S18–25. doi: 10.1038/nrn1434. [DOI] [PubMed] [Google Scholar]
- 3.Beckman JSEA. Superoxide dismutase, oxidative stress, and ALS. In: Mitsumoto H, Perzedborski S, Gordon PH, editors. Amyotrophic Lateral Sclerosis. Taylor and Francis; New York: 2005. pp. 339–54. [Google Scholar]
- 4.Shaw PJ. Molecular and cellular pathways of neurodegeneration in motor neurone disease. J Neurol Neurosurg Psychiatry. 2005;76:1046–57. doi: 10.1136/jnnp.2004.048652. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Martin LJ, Liu Z, Chen K, Price AC, Pan Y, Swaby JA, et al. Motor neuron degeneration in amyotrophic lateral sclerosis mutant superoxide dismutase-1 transgenic mice: mechanisms of mitochondriopathy and cell death. J Comp Neurol. 2007;500:20–46. doi: 10.1002/cne.21160. [DOI] [PubMed] [Google Scholar]
- 6.Pasinelli P, Brown RH. Molecular biology of amyotrophic lateral sclerosis: insights from genetics. Nat Rev Neurosci. 2006;7:710–23. doi: 10.1038/nrn1971. [DOI] [PubMed] [Google Scholar]
- 7.Kaufmann P, Thompson JL, Levy G, Buchsbaum R, Shefner J, Krivickas LS, et al. Phase II trial of CoQ10 for ALS finds insufficient evidence to justify phase III. Annals of neurology. 2009;66:235–44. doi: 10.1002/ana.21743. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Gwinn KCR, Mitsumoto H, Bednarz K, Brown RH, Cudkowicz M, Gordon PH, et al. Amyotrophic Lateral Sclerosis: An Emerging Era of Collaborative Gene Discovery. PloSONE. 2007;2:e1234. doi: 10.1371/journal.pone.0001254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.World Federation NRGoAS [2013 July 3];A revised El Escorial World Federation of Neurology criteria for the diagnosis of amyotrophic lateral sclerosis. 2000 doi: 10.1080/146608200300079536. Available from: www.wfnals.org. [DOI] [PubMed]
- 10.de Carvalho M, Dengler R, Eisen A, England JD, Kaji R, Kimura J, Mills K, Mitsumoto H, Nodera H, Shefner J, Swash M. Electrodiagnostic criteria for diagnosis of ALS. J Clinical Neurophysiol. 2008:493–503. doi: 10.1016/j.clinph.2007.09.143. [DOI] [PubMed] [Google Scholar]
- 11.Cedarbaum J. ALS endpoints. Survival. Amyotroph Lateral Scler Other Motor Neuron Disord. 2004;5(Suppl 1):79–83. doi: 10.1080/17434470410020021. [DOI] [PubMed] [Google Scholar]
- 12.Gordon PH, Miller RG, Moore DH. Amyotrophic lateral sclerosis and other motor neuron disorders. ALSFRS-R. 2004;(Suppl 1):90–3. doi: 10.1080/17434470410019906. [DOI] [PubMed] [Google Scholar]
- 13.Gordon PH, Moore DH, Miller RG, Florence JM, Verheijde JL, Doorish C, et al. Efficacy of minocycline in patients with amyotrophic lateral sclerosis: a phase III randomised trial. Lancet Neurol. 2007;6:1045–53. doi: 10.1016/S1474-4422(07)70270-3. [DOI] [PubMed] [Google Scholar]
- 14.Miller R, Bradley W, Cudkowicz M, Hubble J, Meininger V, Mitsumoto H, et al. Phase II/III randomized trial of TCH346 in patients with ALS. Neurology. 2007;69:776–84. doi: 10.1212/01.wnl.0000269676.07319.09. [DOI] [PubMed] [Google Scholar]
- 15.Traynor BJ, Zhang H, Shefner JM, Schoenfeld D, Cudkowicz ME. Functional outcome measures as clinical trial endpoints in ALS. Neurology. 2004;63:1933–5. doi: 10.1212/01.wnl.0000144345.49510.4e. [DOI] [PubMed] [Google Scholar]
- 16.Woolley SC, York MK, Moore DH, Strutt AM, Murphy J, Schulz PE, et al. Detecting frontotemporal dysfunction in ALS: Utility of the ALS Cognitive Behavioral Screen (ALS-CBS (TM)). Amyotrophic Lateral Sclerosis. 2010;11:303–11. doi: 10.3109/17482961003727954. [DOI] [PubMed] [Google Scholar]
- 17.Rippon GA, Scarmeas N, Gordon PH, Murphy PL, Albert SM, Mitsumoto H, et al. An observational study of cognitive impairment in amyotrophic lateral sclerosis. Arch Neurol-Chicago. 2006;63:345–52. doi: 10.1001/archneur.63.3.345. [DOI] [PubMed] [Google Scholar]
- 18.Mitsumoto H, Santella RM, Liu X, Bogdanov M, Zipprich J, Wu HC, et al. Oxidative stress biomarkers in sporadic ALS. Amyotrophic lateral sclerosis : official publication of the World Federation of Neurology Research Group on Motor Neuron Diseases. 2008;9:177–83. doi: 10.1080/17482960801933942. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Dupuis L, Corcia P, Fergani A, De Aguilar JLG, Bonnefont-Rousselot D, Bittar R, et al. Dyslipidemia is a protective factor in amyotrophic lateral sclerosis. Neurology. 2008;70:1004–9. doi: 10.1212/01.wnl.0000285080.70324.27. [DOI] [PubMed] [Google Scholar]
- 20.Sutedja NA, van der Schouw YT, Fischer K, Sizoo EM, Huisman MHB, Veldink JH, et al. Beneficial vascular risk profile is associated with amyotrophic lateral sclerosis. J Neurol Neurosur Ps. 2011;82:638–42. doi: 10.1136/jnnp.2010.236752. [DOI] [PubMed] [Google Scholar]
- 21.Block G, Hartman AM, Naughton D. A reduced dietary questionnaire: development and validation. Epidemiology. 1990;1:58–64. doi: 10.1097/00001648-199001000-00013. [DOI] [PubMed] [Google Scholar]
- 22.Cummings SR, Block G, McHenry K, Baron RB. Evaluation of two food frequency methods of measuring dietary calcium intake. Am J Epidemiol. 1987;126:796–802. doi: 10.1093/oxfordjournals.aje.a114716. [DOI] [PubMed] [Google Scholar]
- 23.Nieves JW, Golden AL, Siris E, Kelsey JL, Lindsay R. Teenage and current calcium intake are related to bone mineral density of the hip and forearm in women aged 30-39 years. Am J Epidemiol. 1995;141:342–51. doi: 10.1093/aje/141.4.342. [DOI] [PubMed] [Google Scholar]
- 24.Serdula M, Coates R, Byers T, Mokdad A, Jewell S, Chavez N, et al. Evaluation of a brief telephone questionnaire to estimate fruit and vegetable consumption in diverse study populations. Epidemiology. 1993;4:455–63. doi: 10.1097/00001648-199309000-00012. [DOI] [PubMed] [Google Scholar]
- 25. [2013 October 27];NutritionQuest. 2011 Available from: https://www.nutritionquest.com/
- 26.Epel ES, Blackburn EH, Lin J, Dhabhar FS, Adler NE, Morrow JD, et al. Accelerated telomere shortening in response to life stress. Proc Natl Acad Sci U S A. 2004;101:17312–5. doi: 10.1073/pnas.0407162101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Epel ES, Lin J, Wilhelm FH, Wolkowitz OM, Cawthon R, Adler NE, et al. Cell aging in relation to stress arousal and cardiovascular disease risk factors. Psychoneuroendocrinology. 2006;31:277–87. doi: 10.1016/j.psyneuen.2005.08.011. [DOI] [PubMed] [Google Scholar]
- 28.Sapolsky RM. Organismal stress and telomeric aging: an unexpected connection. Proc Natl Acad Sci U S A. 2004;101:17323–4. doi: 10.1073/pnas.0408041101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Albert SM, Rabkin JG, Del Bene ML, Tider T, O'Sullivan I, Rowland LP, et al. Wish to die in end-stage ALS. Neurology. 2005;65:68–74. doi: 10.1212/01.wnl.0000168161.54833.bb. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Rabkin JG, Wagner GJ, Del Bene M. Resilience and distress among amyotrophic lateral sclerosis patients and caregivers. Psychosom Med. 2000;62:271–9. doi: 10.1097/00006842-200003000-00020. [DOI] [PubMed] [Google Scholar]
- 31.Rabkin JG, Albert SM, Del Bene ML, O'Sullivan I, Tider T, Rowland LP, et al. Prevalence of depressive disorders and change over time in late-stage ALS. Neurology. 2005;65:62–7. doi: 10.1212/01.wnl.0000167187.14501.0c. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Lee JR, Annegers JF, Appel SH. Prognosis of amyotrophic lateral sclerosis and the effect of referral selection. J Neurol Sci. 1995;132:207–15. doi: 10.1016/0022-510x(95)00154-t. [DOI] [PubMed] [Google Scholar]
- 33.Logroscino G, Traynor BJ, Hardiman O, Chio A, Couratier P, Mitchell JD, et al. Descriptive epidemiology of amyotrophic lateral sclerosis: new evidence and unsolved issues. Journal of neurology, neurosurgery, and psychiatry. 2008;79:6–11. doi: 10.1136/jnnp.2006.104828. [DOI] [PubMed] [Google Scholar]
- 34.Chad D, Rowland L, Armon C, Bedlack R, Durham H, Factor-Litvak P. Peer Recommendations on How to Improve Clinical Research and Final Conference Wrap-up. Amyotroph Lateral Scler Frontotemporal Degener. 2013;14(Suppl.1):67–73. doi: 10.3109/21678421.2013.778605. [DOI] [PubMed] [Google Scholar]
- 35.Factor-Litvak P, Al-Chalabi A, Asherio A, Bradley W, Chío A, Garruto R, et al. Current Pathways for Epidemiological Research in Amyotrophic Lateral Sclerosis. Amyotroph Lateral Scler Frontotemporal Degener. 2013;14(Suppl. 1):33–43. doi: 10.3109/21678421.2013.778565. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Sherman A, Gubitz A, Al-Chalabi A, Bedlack R, Berry J, Conwit R. Infrastructure Resources for Clinical Research in Amyotrophic Lateral Sclerosis. Amyotroph Lateral Scler Frontotemporal Degener. 2013;14(Suppl. 1):53–61. doi: 10.3109/21678421.2013.779058. [DOI] [PubMed] [Google Scholar]
- 37.Bedlack RS, Pastula DM, Welsh E, Pulley D, Cudkowicz ME. Scrutinizing enrollment in ALS clinical trials: room for improvement? Amyotrophic lateral sclerosis : official publication of the World Federation of Neurology Research Group on Motor Neuron Diseases. 2008;9:257–65. doi: 10.1080/17482960802195913. [DOI] [PubMed] [Google Scholar]
- 38.Bedlack RS, Wicks P, Heywood J, Kasarskis E. Modifiable barriers to enrollment in American ALS research studies. Amyotrophic lateral sclerosis : official publication of the World Federation of Neurology Research Group on Motor Neuron Diseases. 2010;11:502–7. doi: 10.3109/17482968.2010.484494. [DOI] [PubMed] [Google Scholar]
- 39.Moser DJ, Schultz SK, Arndt S, Benjamin ML, Fleming FW, Brems CS, et al. Capacity to provide informed consent for participation in schizophrenia and HIV research. Am J Psychiatry. 2002;159(7):1201–7. doi: 10.1176/appi.ajp.159.7.1201. [DOI] [PubMed] [Google Scholar]
- 40.Abrahams S, Leigh PN, Harvey A, Vythelingum GN, Grise D, Goldstein LH. Verbal fluency and executive dysfunction in amyotrophic lateral sclerosis (ALS). Neuropsychologia. 2000;38:734–47. doi: 10.1016/s0028-3932(99)00146-3. [DOI] [PubMed] [Google Scholar]
- 41.Kertesz A, Nadkarni N, Davidson W, Thomas AW. The Frontal Behavioral Inventory in the differential diagnosis of frontotemporal dementia. J Int Neuropsychol Soc. 2000;6:460–8. doi: 10.1017/s1355617700644041. [DOI] [PubMed] [Google Scholar]
- 42.Smith RA, Berg JE, Pope LE, Thisted RA. Measuring pseudobulbar affect in ALS. Amyotroph Lateral Sc. 2004;5:99–102. doi: 10.1080/17434470410020058. [DOI] [PubMed] [Google Scholar]
- 43.Kamel F, Umbach DM, Munsat TL, Shefner JM, Hu H, Sandler DP. Lead exposure and amyotrophic lateral sclerosis. Epidemiology. 2002;13:311–9. doi: 10.1097/00001648-200205000-00012. [DOI] [PubMed] [Google Scholar]
- 44.Chen HL, Richard M, Sandier DP, Umbach DM, Kamel F. Head injury and amyotrophic lateral sclerosis. American journal of epidemiology. 2007;166:810–6. doi: 10.1093/aje/kwm153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Fang F, Quinlan P, Ye W, Barber MK, Umbach DM, Sandler DP, et al. Workplace Exposures and the Risk of Amyotrophic Lateral Sclerosis. Environ Health Persp. 2009;117:1387–92. doi: 10.1289/ehp.0900580. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Sorenson EJ, Stalker AP, Kurland LT, Windebank AJ. Amyotrophic lateral sclerosis in Olmsted County, Minnesota, 1925 to 1998. Neurology. 2002;59:280–2. doi: 10.1212/wnl.59.2.280. [DOI] [PubMed] [Google Scholar]
- 47.Mateen FJ, Carone M, Sorenson EJ. Patients who survive 5 years or more with ALS in Olmsted County, 1925-2004. J Neurol Neurosur Ps. 2010;81:1144–6. doi: 10.1136/jnnp.2009.201251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Horner RD, Grambow SC, Coffman CJ, Lindquist JH, Oddone EZ, Allen KD, et al. Amyotrophic lateral sclerosis among 1991 Gulf War veterans: evidence for a time-limited outbreak. Neuroepidemiology. 2008;31:28–32. doi: 10.1159/000136648. [DOI] [PubMed] [Google Scholar]
- 49.Weisskopf MG, McCullough ML, Calle EE, Thun MJ, Cudkowicz M, Ascherio A. Prospective study of cigarette smoking and amyotrophic lateral sclerosis. Am J Epidemiol. 2004;160:26–33. doi: 10.1093/aje/kwh179. [DOI] [PubMed] [Google Scholar]
- 50.Wang H, O'Reilly EJ, Weisskopf MG, Logroscino G, McCullough ML, Thun MJ, et al. Smoking and risk of amyotrophic lateral sclerosis: a pooled analysis of 5 prospective cohorts. Arch Neurol. 2011;68:207–13. doi: 10.1001/archneurol.2010.367. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Ascherio A, Weisskopf MG, O'Reilly EJ, Jacobs EJ, McCullough ML, Calle EE, et al. Vitamin E intake and risk of amyotrophic lateral sclerosis. Annals of neurology. 2005;57:104–10. doi: 10.1002/ana.20316. [DOI] [PubMed] [Google Scholar]
- 52.Weisskopf MG, O'Reilly EJ, McCullough ML, Calle EE, Thun MJ, Cudkowicz M, et al. Prospective study of military service and mortality from ALS. Neurology. 2005;64:32–7. doi: 10.1212/01.WNL.0000148649.17706.D9. [DOI] [PubMed] [Google Scholar]
- 53.Abhinav K, Stanton B, Johnston C, Hardstaff J, Orrell RW, Howard R, et al. Amyotrophic lateral sclerosis in South-East England: a population-based study. The South-East England register for amyotrophic lateral sclerosis (SEALS Registry). Neuroepidemiology. 2007;29:44–8. doi: 10.1159/000108917. [DOI] [PubMed] [Google Scholar]
- 54.Johnston CA, Stanton BR, Turner MR, Gray R, Blunt AH, Butt D, et al. Amyotrophic lateral sclerosis in an urban setting: a population based study of inner city London. J Neurol. 2006;253:1642–3. doi: 10.1007/s00415-006-0195-y. [DOI] [PubMed] [Google Scholar]
- 55.Popat RA, Van Den Eeden SK, Tanner CM, Bernstein AL, Bloch DA, Leimpeter A, et al. Effect of reproductive factors and postmenopausal hormone use on the risk of amyotrophic lateral sclerosis. Neuroepidemiology. 2006;27:117–21. doi: 10.1159/000095550. [DOI] [PubMed] [Google Scholar]
- 56.Popat RA, Tanner CM, van den Eeden SK, Bernstein AL, Bloch DA, Leimpeter A, et al. Effect of non-steroidal anti-inflammatory medications on the risk of amyotrophic lateral sclerosis. Amyotrophic lateral sclerosis : official publication of the World Federation of Neurology Research Group on Motor Neuron Diseases. 2007;8:157–63. doi: 10.1080/17482960601179456. [DOI] [PubMed] [Google Scholar]
- 57.Forbes RB, Colville S, Parratt J, Swingler RJ. The incidence of motor nueron disease in Scotland. J Neurol. 2007;254:866–9. doi: 10.1007/s00415-006-0454-y. [DOI] [PubMed] [Google Scholar]
- 58.Chio A, Calvo A, Dossena M, Ghiglione P, Mutani R, Mora G. ALS in Italian professional soccer players: The risk is still present and could be soccer-specific. Amyotrophic Lateral Sclerosis. 2009;10:205–9. doi: 10.1080/17482960902721634. [DOI] [PubMed] [Google Scholar]
- 59.Atsuta N, Watanabe H, Ito M, Tanaka F, Tamakoshi A, Nakano I, et al. Age at onset influences on wide-ranged clinical features of sporadic amyotrophic lateral sclerosis. J Neurol Sci. 2009;276:163–9. doi: 10.1016/j.jns.2008.09.024. [DOI] [PubMed] [Google Scholar]
- 60.Zaldivar T, Gutierrez J, Lara G, Carbonara M, Logroscino G, Hardiman O. Reduced frequency of ALS in an ethnically mixed population. Neurology. 2009;72:1640–5. doi: 10.1212/WNL.0b013e3181a55f7b. [DOI] [PubMed] [Google Scholar]
- 61.O'Toole O, Traynor BJ, Brennan P, Sheehan C, Frost E, Corr B, et al. Epidemiology and clinical features of amyotrophic lateral sclerosis in Ireland between 1995 and 2004. J Neurol Neurosur Ps. 2008;79:30–2. doi: 10.1136/jnnp.2007.117788. [DOI] [PubMed] [Google Scholar]
- 62.Yeo L, Lynch C, Hardiman O. Validating population-based registers for ALS: how accurate is death certification? J Neurol. 2010;257:1235–9. doi: 10.1007/s00415-010-5494-7. [DOI] [PubMed] [Google Scholar]
- 63.Phukan J, Elamin M, Bede P, Jordan N, Gallagher L, Byrne S, et al. The syndrome of cognitive impairment in amyotrophic lateral sclerosis: a population-based study. J Neurol Neurosurg Psychiatry. 2012;83:102–8. doi: 10.1136/jnnp-2011-300188. [DOI] [PubMed] [Google Scholar]
- 64.Donaghy C, O'Toole O, Sheehan C, Kee F, Hardiman O, Patterson V. An all-Ireland epidemiological study of MND, 2004-2005. Eur J Neurol. 2009;16:148–53. doi: 10.1111/j.1468-1331.2008.02361.x. [DOI] [PubMed] [Google Scholar]
- 65.Donaghy C, Clarke J, Patterson C, Kee F, Hardiman O, Patterson V. The epidemiology of motor neuron disease in Northern Ireland using capture-recapture methodology. Amyotroph Lateral Sc. 2010;11:374–8. doi: 10.3109/17482960903329569. [DOI] [PubMed] [Google Scholar]
- 66.Hanby MF, Scott KM, Scotton W, Wijesekera L, Mole T, Ellis CE, et al. The risk to relatives of patients with sporadic amyotrophic lateral sclerosis. Brain. 2011;134(Pt 12):3454–7. doi: 10.1093/brain/awr248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Huisman MH, de Jong SW, van Doormaal PT, Weinreich SS, Schelhaas HJ, van der Kooi AJ, et al. Population based epidemiology of amyotrophic lateral sclerosis using capture-recapture methodology. J Neurol Neurosurg Psychiatry. 2011;82:1165–70. doi: 10.1136/jnnp.2011.244939. [DOI] [PubMed] [Google Scholar]
- 68.de Jong SW, Huisman MH, Sutedja NA, van der Kooi AJ, de Visser M, Schelhaas HJ, et al. Smoking, alcohol consumption, and the risk of amyotrophic lateral sclerosis: a population-based study. Am J Epidemiol. 2012;176:233–9. doi: 10.1093/aje/kws015. [DOI] [PubMed] [Google Scholar]
- 69.Beghi E, Millul A, Micheli A, Vitelli E, Logroscino G, Group S. Incidence of ALS in Lombardy, Italy. Neurology. 2007;68:141–5. doi: 10.1212/01.wnl.0000250339.14392.bb. [DOI] [PubMed] [Google Scholar]
- 70.Sabatelli M, Conforti FL, Zollino M, Mora G, Monsurro MR, Volanti P, et al. C9ORF72 hexanucleotide repeat expansions in the Italian sporadic ALS population. Neurobiol Aging. 2012;33:1848, e15–20. doi: 10.1016/j.neurobiolaging.2012.02.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Chio A, Calvo A, Mazzini L, Cantello R, Mora G, Moglia C, et al. Extensive genetics of ALS: a population-based study in Italy. Neurology. 2012;79:1983–9. doi: 10.1212/WNL.0b013e3182735d36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Chio A, Calvo A, Moglia C, Gamna F, Mattei A, Mazzini L, et al. Non-invasive ventilation in amyotrophic lateral sclerosis: a 10 year population based study. J Neurol Neurosurg Psychiatry. 2012;83:377–81. doi: 10.1136/jnnp-2011-300472. [DOI] [PubMed] [Google Scholar]
- 73.Chio A, Calvo A, Ghiglione P, Mazzini L, Mutani R, Mora G, et al. Tracheostomy in amyotrophic lateral sclerosis: a 10-year population-based study in Italy. J Neurol Neurosurg Psychiatry. 2010;81:1141–3. doi: 10.1136/jnnp.2009.175984. [DOI] [PubMed] [Google Scholar]
- 74.Chio A, Calvo A, Moglia C, Mazzini L, Mora G, group Ps. Phenotypic heterogeneity of amyotrophic lateral sclerosis: a population based study. J Neurol Neurosurg Psychiatry. 2011;82:740–6. doi: 10.1136/jnnp.2010.235952. [DOI] [PubMed] [Google Scholar]
- 75.Logroscino G, Traynor BJ, Hardiman O, Chio A, Mitchell D, Swingler RJ, et al. Incidence of amyotrophic lateral sclerosis in Europe. J Neurol Neurosurg Psychiatry. 2010;81:385–90. doi: 10.1136/jnnp.2009.183525. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Spitzer RL, Kroenke K, Williams JB. Validation and utility of a self-report version of PRIME-MD: the PHQ primary care study. Primary Care Evaluation of Mental Disorders. Patient Health Questionnaire. Jama. 1999;282:1737–44. doi: 10.1001/jama.282.18.1737. [DOI] [PubMed] [Google Scholar]
- 77.Rabkin JG, Struening EL. Live events, stress, and illness. Science. 1976;194:1013–20. doi: 10.1126/science.790570. [DOI] [PubMed] [Google Scholar]
- 78.Linn MW. A Global Assessment of Recent Stress (GARS) Scale. Int J Psychiatry Med. 1985;15:47–59. doi: 10.2190/xp8n-rp1w-ye2b-9q7v. [DOI] [PubMed] [Google Scholar]
- 79.Endicott J. Quality of Life Enjoyment and Satisfaction Questionnaire: A new measure. Psychopharmacol Bull. 1993:321–6. [PubMed] [Google Scholar]
- 80.Watson D, Clark LA, Tellegen A. Development and validation of brief measures of positive and negative affect: the PANAS scales. J Pers Soc Psychol. 1988;54:1063–70. doi: 10.1037//0022-3514.54.6.1063. [DOI] [PubMed] [Google Scholar]
- 81.Beck AT, Weissman A, Lester D, Trexler L. The measurement of pessimism: the hopelessness scale. J Consult Clin Psych. 1974;42:861–5. doi: 10.1037/h0037562. [DOI] [PubMed] [Google Scholar]
- 82.Manne SL, Taylor KL, Dougherty J, Kemeny N. Supportive and negative responses in the partner relationship: Their association with psychological adjustment among individuals with cancer. J Behav Med. 1997;20:101–25. doi: 10.1023/a:1025574626454. [DOI] [PubMed] [Google Scholar]
- 83.Gift AG. Visual Analog Scales - Measurement of Subjective Phenomena. Nurs Res. 1989;38:286–8. [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.