Abstract
Magnetic resonance imaging (MRI), multidetector computed tomography (MDCT), and positron emission tomography (PET) are complementary imaging modalities in the preoperative staging of patients with rectal cancer, and each offers their own individual strengths and weaknesses. MRI is the best available radiologic modality for the local staging of rectal cancers, and can play an important role in accurately distinguishing which patients should receive preoperative chemoradiation prior to total mesorectal excision. Alternatively, both MDCT and PET are considered primary modalities when performing preoperative distant staging, but are limited in their ability to locally stage rectal malignancies. This review details the role of each of these three modalities in rectal cancer staging, and how the three imaging modalities can be used in conjunction.
Keywords: Rectal cancer, staging, magnetic resonance imaging (MRI), computed tomography (CT), positron emission tomography (PET)
Introduction
Colorectal cancer is incredibly common, representing the 4th leading cause of cancer mortality and the 2nd most common malignancy worldwide, with nearly 1 million newly diagnosed colorectal cancers each year (1,2). Of all colorectal cancers, rectal cancer comprises over 1/3 of cases, with over 40% arising within 6 cm of the anal verge (1,3). While there is little doubt that colonoscopy and biopsy are, and will remain for the foreseeable future, the gold standard modalities for the initial diagnosis of rectal cancer, traditional radiologic imaging modalities are of vital importance with regard to the local staging of patients with a known diagnosis and the identification of distant metastatic disease (i.e., distant staging).
The importance of diagnostic imaging in accurate distant staging is beyond doubt, with multidetector computed tomography (MDCT), magnetic resonance imaging (MRI), and positron emission tomography (PET) all offering valuable means of identifying tumor spread to the liver, lungs, and distant lymph nodes; the three most common sites of distant metastatic disease (2,4). Traditionally, metastatic colorectal cancer at presentation has been treated solely with chemotherapy, although it is increasingly thought that this patient population might also benefit from local resection of their tumor, with associated increased quality of life measures and longer survival (even despite the presence of distant metastases), and in some cases, resection of metastases (particularly to the liver or lungs) may also be a feasible option. Accordingly, the identification of distant metastatic disease has a profound impact on the management algorithm employed for this group of patients, making accurate distant radiologic staging vital (4,5). However, local staging has become equally critical in patient management, particularly given the increasing incorporation of neoadjuvant chemoradiation into treatment protocols. More specifically, while the increasing adoption of total mesorectal excision (i.e., ‘en-bloc’ resection of the mesorectum) has significantly reduced the incidence of post-operative local recurrence within the surgical bed (once as high as 38%), locally advanced tumors are still far more likely to recur, and these locally advanced tumors are increasingly being treated with preoperative radiation and chemotherapy prior to total mesorectal excision, requiring radiology to be accurate in determining the local extension of tumors (T-stage), the relationship of a tumor to the mesorectal fascia, and the presence of suspicious locoregional lymph nodes (N-stage) (6). This review will describe the role of the three most important radiologic modalities in the local and distant staging of rectal cancer, namely MDCT, MRI, and PET or PET-CT, all of which serve complementary roles in the initial accurate staging of patients.
Magnetic resonance imaging (MRI)
Local staging
Technique
From a technique standpoint, while the protocols utilized in rectal MRI will vary slightly from institution to institution, high resolution T2 weighted images (with a slice thickness of 3 mm) with a small field of view (FOV) focusing on the rectum are the most critical to accurate diagnosis, as they provide the best means of evaluating the rectal wall and perirectal fat (allowing optimal discrimination of T2 from T3 tumors), and should be acquired in the axial, sagittal, and coronal planes. While the radiologist may choose to primarily focus on the axial images, the coronal and sagittal images become increasingly important when confronted by an infiltrative tumor involving larger portions of the rectum, or an excessively tortuous rectum. In addition, the coronal plane tends to be the most useful for establishing the relationship of a tumor with the internal and external anal sphincters, as tumoral involvement of the sphincter complex could potentially necessitate the performance of an abdominoperineal resection with en bloc resection of the sphincter complex. While the small FOV high-resolution T2 weighted images are the most important imaging sequences, most protocols will incorporate larger FOV T2 weighted images of the pelvis and pre- and post-gadolinium 3-dimensional fast spoiled gradient echo sequence (FSPGR) images to evaluate for the presence of pelvic lymphadenopathy (outside of the mesorectum) and to identify other salient pelvic abnormalities. Moreover, while the T2 weighted images are the most important to evaluate the tumor itself and its relationship with the rectal wall and mesorectal fat, the post-gadolinium images may be helpful in some select cases. In addition, diffusion weighting imaging (DWI) has increasingly been incorporated into these protocols, and can serve as a means for accentuating the primary tumor and locoregional lymph nodes. While DWI and post-gadolinium images are not absolutely critical for evaluation of the primary tumor, most rectal cancers will enhance avidly and demonstrate restricted diffusion (3,6).
Typically, the rectum will be ‘cleansed’ prior to the study using a standard preparation of sodium bisphosphonate or a sodium phosphate enema, in order to avoid fecal material interfering with study interpretation. Subsequently, many practices will administer a small volume (usually 60 cc) of a rectal contrast agent, which can either be ultrasound gel (a ‘positive’ contrast agent that is T2 hyperintense) or a mixture of barium sulfate and ferumoxsil (a ‘negative’ contrast agent that is T2 hypointense). These agents can help accentuate small or polyploid tumors that might be difficult to identify without adequate rectal distension, particularly in tumors that are higher in the rectum. Some, but not all, practices utilize a bowel paralytic such as glucagon, which can reduce artifacts related to bowel motion (3,6). The utilization of an endorectal coil has increasingly decreased, particularly as positioning of the coil can be problematic in higher rectal tumors, as well as those lesions that cause significant narrowing of the rectum, and moreover, it places limits on the field of view that may hinder complete assessment of a tumor’s involvement of the mesorectal fascia and slightly more distant mesorectal lymph nodes (7). While the use of an endorectal coil was originally advocated in the belief that it offered improved image quality and signal to noise ratio (SNR), there is very little evidence that the endorectal coil offers any substantial benefit over a standard phased array coil in terms of diagnostic quality.
T-stage and local tumor extension
A T1 tumor extends through the muscularis mucosa and into the submucosa, while a T2 tumor extends through the submucosa into the muscularis propria. In most cases, these two T-stages are treated equivalently, without the addition of preoperative chemotherapy or radiation, and distinguishing T1 and T2 tumors is not possible on MRI with a high degree of accuracy given that the submucosa and muscularis propria of the rectal wall cannot be consistently differentiated on MRI (8). However, T3 tumors (which extend beyond the muscularis propria) have been shown to have better outcomes (with a lesser risk of local recurrence) when treated with preoperative chemoradiation and these lesions can be distinguished from T1 and T2 tumors on MRI. T4 tumors are characterized by their spread into the visceral peritoneum, adjacent organs, or the levator musculature (3,6). On MRI, the three layers of the rectal wall are usually clearly discernible on T2-weighted images, with the mucosa and submucosa appearing relatively hyperintense, the muscularis appearing relatively hypointense in the middle of the wall, and a layer of hyperintense perirectal fat on the outside of the wall. Careful evaluation of the T2 hypointense muscularis throughout the areas abutting the rectal cancer is critical, and this thin hypointense line should be intact and clearly visible throughout the rectum for a tumor to be described as a T1 or T2 lesion. A tumor that has breached the T2 hypointense layer of the rectal wall (i.e., the muscularis is not clearly visualized adjacent to the tumor) can be considered to be at least a T3 tumor, necessitating preoperative chemoradiation (Figures 1,2,3,4) (3,6).
Figure 1.
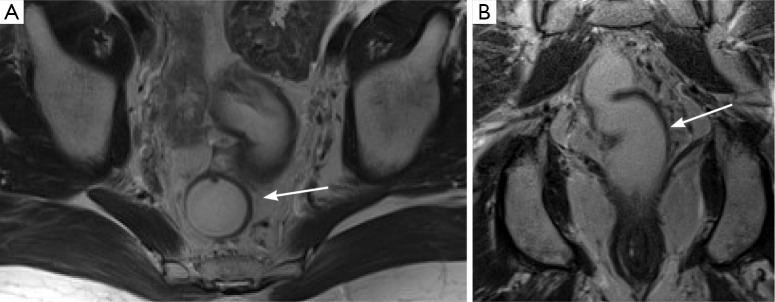
Normal appearance of the rectum on T2 weighted images. In both images, there is a clearly defined, T2 hypointense line (arrow) around the margins of the rectum, representing the intact muscularis propria.
Figure 2.
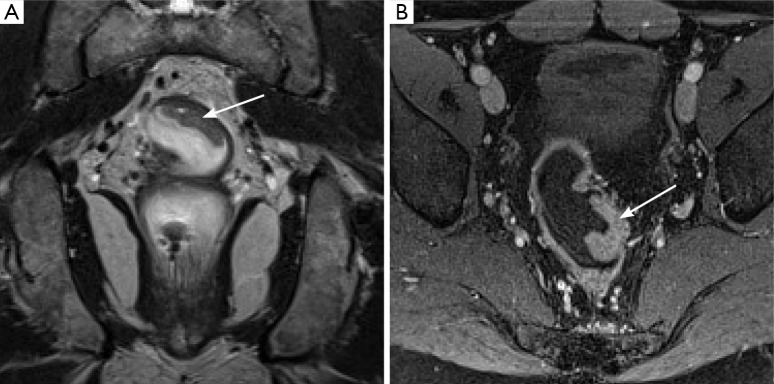
Example of a T2N0 rectal cancer. Coronal (A) T2 weighted image demonstrates a small polyploid mass (arrow) arising from the wall of the rectum. Importantly, the overlying hypointense line demarcating the muscularis propria remains intact, suggesting this is not a T3 lesion. Axial post-gadolinium image (B) nicely demarcates the mass (arrow), although evaluating extension through the muscularis is not possible on this sequence.
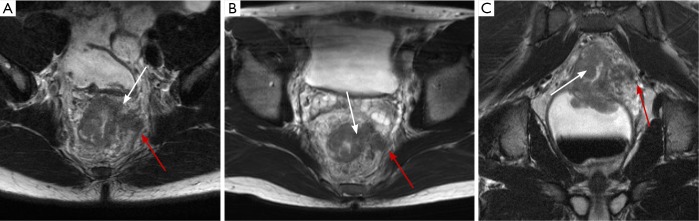
Figure 3.

Axial high-resolution T2 weighted image (A) demonstrates circumferential thickening (white arrow) around the entirety of the rectum, in keeping with the patient’s malignancy. In this case, the T2 hypointense muscularis is absent underlying the mass, suggesting this represents a T3 malignancy. Red arrow illustrates the intact mesorectal fascia or circumferential resection margin (CRM). Axial post-gadolinium axial image (B) demonstrates a heterogeneously enhancing malignant lymph node (arrow) in the 7 o’clock position.
Figure 4.

Axial (A) and coronal (B) T2 weighted images demonstrate a polyploid mass (arrow) arising from the right lateral aspect of the rectum, with complete loss of the underlying T2 hypointense muscularis (best visualized on the coronal image), in keeping with a T3 lesion. The mass (arrow) (C) demonstrates avid enhancement on the post-gadolinium image.
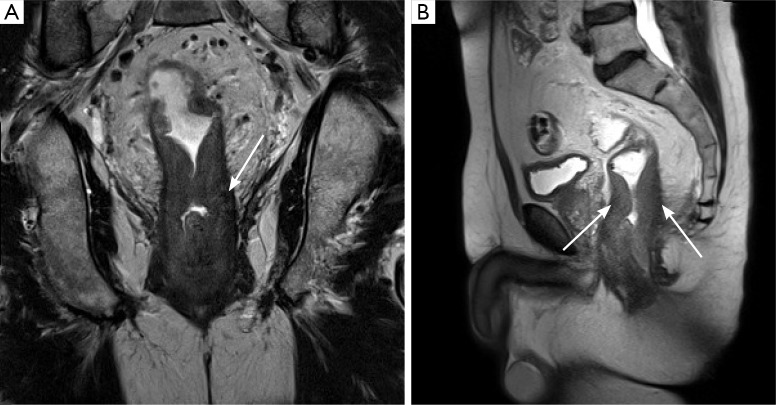
Once a tumor is characterized as either a T1/T2 or T3 lesion, the extent of involvement of the surrounding mesorectum and the adjacent pelvic structures can also have an important impact on patient prognosis. T3 tumors can be further subdivided into T3a (<5 mm extension beyond the muscularis) and T3b (>5 mm extension beyond the muscularis), and MRI has been shown to be relatively accurate in distinguishing these small differences in involvement. Such a distinction between T3a and T3b tumors may be of clinical importance, as >5 mm extension into the mesorectum has been found to be associated with a significantly lower 5-year survival rate (54% vs. 85%) (9). Just as important as the tumor’s T-stage, however, is the proximity of the tumor to the margins of the mesorectal fascia (also described as the ‘circumferential resection margin’ or ‘CRM’), as tumors that are 1 mm or less from the mesorectal fascia are at substantially higher risk of local recurrence (Figure 5) (8). A tumor’s relationship to the fascia is relatively easy to perceive on MRI, but is not usually possible to delineate with endoscopic ultrasound (EUS). Finally, particularly for advanced tumors, MRI offers an accurate means of assessing involvement of adjacent pelvic organs (including the prostate, seminal vesicles, uterus, vagina, etc.), the sacrum, the anal sphincters, the pelvic sidewalls, and adjacent vasculature (Figure 6) (3,6).
Figure 5.
Axial (A,B) and coronal (C) T2 weighted images demonstrate a rectal mass (white arrows) extending through the rectal wall at the 3 o’clock position into the mesorectal fat. In this case, the mass involves the CRM at this position (red arrow).
Figure 6.
T4 low rectal cancer (arrows) with involvement of both the internal and external sphincters illustrated on coronal (A) and sagittal (B) T2 weighted images.
Locoregional lymph node staging
While the superior soft tissue resolution of MRI does facilitate the identification of local lymph nodes (both in the mesorectum and the pelvis), the ability to discern a benign from a malignant lymph node is still partially based upon size criteria, inherently limiting sensitivity and specificity. The most commonly used size criteria, particularly in the mesorectum, is 5 mm, which provides a sensitivity of only 68% and a specificity of only 78%, as a sizeable number of ultimately metastatic nodes at histopathology measure under 5 mm in size. Morphologic data, including irregular lymph node margins and abnormal signal or enhancement may also be useful ancillary features. The presence of suspicious nodes is important for treatment planning, as mesorectal lymph nodes (which are typically resected with the surgical excision) close to the mesorectal fascia may necessitate wider surgical margins at that site, while lymph nodes outside of the mesorectum (which are not usually resected with the surgical specimen) may necessitate wider radiation, an extended surgical resection, or even upstaging to M1 disease (lymph nodes in the external iliac chains, obdurator chains, or the retroperitoneum) (3,6).
Accuracy of MRI for local staging
There is little doubt that MRI is an accurate modality for establishing the T-stage of a tumor and delineating its relationship with the mesorectal fascia (CRM). A meta-analysis by Al-Sukhni et al. in 2012 (10) encompassing 21 different studies found excellent sensitivities and specificities for establishing involvement of the CRM (up to 77% and 94% respectively), with a slightly lower performance for determining T-stage (87% and 75% respectively). The excellent performance of MRI in evaluating CRM involvement has been consistent across multiple studies in the literature, including a study by the MERCURY study group that found 92% specificity in predicting a negative surgical margin (11-13). However, as one would expect given the limitations of any anatomic imaging modality in evaluating lymph nodes, sensitivities and specificities for lymph node involvement in the study by Al-Sukhni et al. were only 77% and 71% respectively (10). While some had hoped that the inclusion of DWI into imaging protocols might help distinguish benign from malignant lymph nodes, this has not turned out to be the case: Metastatic lymph nodes do demonstrate lower mean ADC values, but ADC values have not proven particularly sensitive or specific for metastatic lymphadenopathy (14,15).
When compared to EUS, another modality commonly utilized for local staging, there is little doubt that EUS is superior in distinguishing T0, T1, and T2 tumors, a distinction that is not possible on MRI, and that may be clinically important in a small group of patients who might undergo local resection (T0 or T1 tumor) rather than total mesorectal excision (with a T2 tumor). In general, both modalities are probably relatively similar in their ability to distinguish T1 or T2 tumors from T3 tumors, and both modalities have similar limitations in distinguishing metastatic from benign lymph nodes in the mesorectum (although EUS can likely identify more lymph nodes than MRI given its spatial resolution). MRI can clearly better identify lymph nodes distant from the tumor (including the upper rectum), and the ability to evaluate CRM involvement is clearly an advantage of MRI (8).
Distant staging
In most cases, MDCT represents the best primary option for distant staging of rectal cancer, particularly given the propensity for tumors to metastasize to the lungs (where MRI is highly limited). Moreover, even with regards to evaluation of the liver (usually considered the greatest strength of MRI), in the vast majority of cases the routine preoperative addition of MRI to MDCT is likely to be of little benefit, as a study by Wiggans et al. found that the addition of MRI did not make a significant difference in patients with colorectal cancer to lesion detection, recurrence rates, or patient survival (16).
Nevertheless, the primary role of MRI in distant staging is as a trouble-shooting modality when confronted with an indeterminate lesion on MDCT, particularly in the liver. It is not at all uncommon to be confronted with a ‘too-small-to characterize’ lesion on MDCT measuring under 1 cm in size, which cannot be definitively characterized as either benign (i.e., cyst or hemangiomas) or malignant (i.e., metastasis) (17). Given the superior soft tissue resolution of MRI, as well as the ability to use several imaging sequences in conjunction to arrive at a diagnosis, the specificity of MRI for small liver lesions is superior to MDCT (18). In most cases, metastases will be T1 hypointense and T2 hyperintense (although lower in signal compared to cysts or hemangiomas) and will demonstrate peripheral enhancement. Moreover, the increasing utilization of diffusion weighted images in liver protocols offers another means of both identifying lesions which might not be conspicuous on either CT or standard MRI pulse sequences, as well as the risk stratification of liver lesions (as liver metastases will tend to have lower ADC values) (19,20).
Multidetector computed tomography (MDCT)
Technique
In cases with a known primary rectal malignancy, most institutions employ a single-phase technique, with the acquisition of venous phase images at roughly 60-70 s after the rapid injection of intravenous contrast (3-5 cc/s). In some instances, when seeking to better define subtle abnormal enhancement or delineate a subtle bowel lesion, the incorporation of arterial phase images may have some value in certain select cases (typically at 25-30 s after the injection of IV contrast). Particularly in those cases when the primary tumor has not yet been resected, and there is the intention to evaluate local tumor extension and mesorectal lymphadenopathy, neutral contrast agents (such as VoLumen) are utilized to distend the bowel without creating unnecessary streak or beam-hardening artifacts. Accordingly, positive oral contrast is usually avoided in these cases, as the dense contrast material may obscure subtle abnormalities in the adjacent bowel wall, and streak artifact may preclude accurate identification of small mesorectal lymph nodes (17,21). Given that MDCT is almost never utilized for local tumor staging, rectal contrast administration is not a common component of these imaging protocols, and no attempt is usually made to distend the rectum with contrast material.
The latest generation of MDCT scanners allows the acquisition of thin-section isotropic images, with identical resolutions in the x, y, and z-axes, at 0.5-0.75 mm collimation. These images are reconstructed to 3 mm for routine axial image review, as well as to 0.75 mm for the further generation of multiplanar reformations (coronal and sagittal) and 3-D images. The 3-D reconstructions typically include maximum intensity projection (MIP) images, which highlight the highest attenuation voxels in a dataset and project them into a 2-dimensional (2-D) representation, and volume rendered (VR) images, which utilize a complex computer algorithm to assign colors and transparencies to each voxel in a study based on its attenuation and relationship to other nearby voxels, thus creating a 3-D representation of the data set. These two 3-D techniques can be of great value in allowing the identification of lesions that might otherwise not be visualized on the standard 2-D images, as well as potentially highlight lesion features that might allow a more specific diagnosis (17,21-24).
Local staging
The MDCT appearance of rectal tumors can vary, including circumferential wall thickening, focal mural wall thickening, or a discrete polyploid mass (Figure 7). The conspicuity of these tumors can vary significantly depending on rectal distension, and the degree of enhancement can also vary widely. Even with the last generation of MDCT scanners, which have offered dramatic improvements in both spatial and temporal resolution compared to prior generations of technology, the layers of the rectal wall cannot be clearly differentiated in any phase of imaging (whether arterial, venous, or delayed). As such, like MRI, it is impossible to differentiate T0, T1, or T2 tumors. However, the mesorectal fat surrounding a tumor can be clearly visualized on CT, and in those cases where the tumor is seen to directly extend into the perirectal fat, a T3 tumor can be diagnosed. However, this is often confounded by the fact that perirectal fat stranding or induration secondary to rectal inflammation or peritumoral fibrosis cannot be definitively differentiated from tumor extension. Unfortunately, diagnosis of T4 tumors can be difficult in some cases as a result of MDCT’s general lack of soft tissue resolution in the pelvis, and it can be quite difficult in the more subtle cases to clearly delineate tumoral involvement of adjacent organs, the pelvic sidewalls, or the adjacent vasculature. Diagnosis in these cases is contingent on loss of fat planes between a tumor and the adjacent organ or structure (Figures 8,9).
Figure 7.
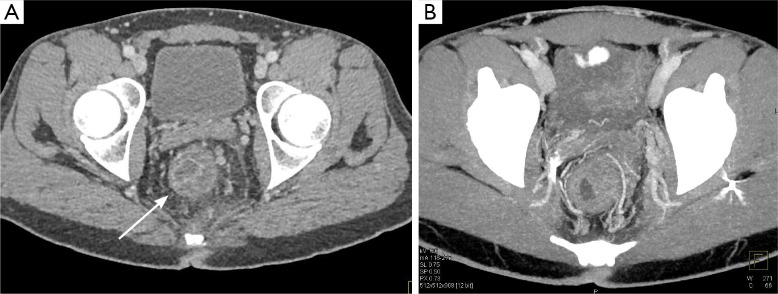
Rectal cancer on MDCT. Axial (A) contrast-enhanced and axial volume rendered (B) images demonstrate severe circumferential wall thickening of the rectum, with neovascularity nicely illustrated on the volume rendered 3-D image. While there is stranding and edema in the mesorectal fat, it is not possible to distinguish tumor invasion into the mesorectum from edema and inflammation. MDCT, multidetector computed tomography.
Figure 8.
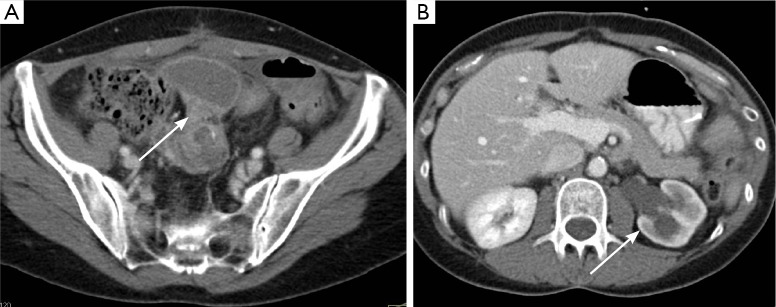
T4 rectal cancer on MDCT. In this case, a high rectal cancer (arrow in A) directly invades the bladder, resulting in severe left-sided hydronephrosis (arrow in B). The loss of fat plane between the bladder and rectum, as well as an appearance suggesting direct invasion, allow the diagnosis of a T4 tumor. MDCT, multidetector computed tomography.
Figure 9.
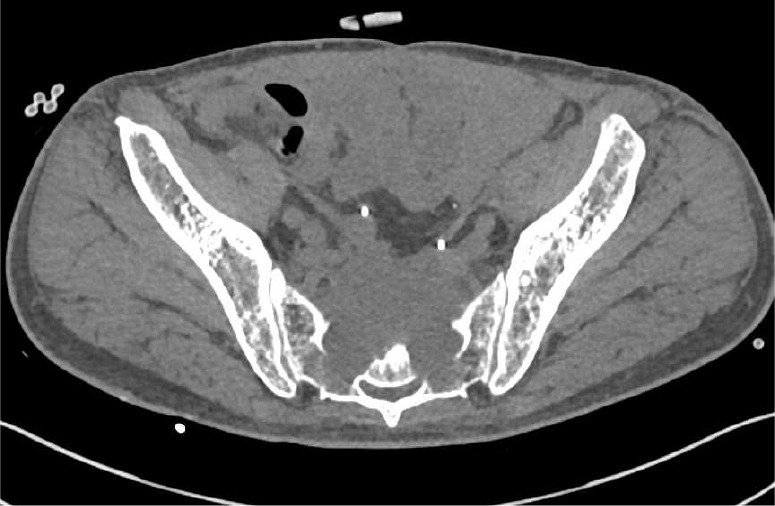
T4 rectal cancer with destruction of the sacrum on MDCT. A large bulky mass directly invades, and destroys, the adjacent sacrum. MDCT, multidetector computed tomography.
Unfortunately, despite multiple studies over the last 15 years seeking to establish MDCT as a tool for local rectal cancer staging, the results have been mixed (1). In a study by Juchems et al. in 2009 MDCT was unable to correctly differentiate lesions requiring neoadjuvant therapy from those lesions that could directly undergo surgical resection (25). Another study by Vliegen et al. in 2007 found that MDCT had a relatively poor accuracy in determining tumor involvement of the mesorectal fascia (26). However, in a study by Kanamoto et al. in 2007 the sensitivity/specificity for T1 and T2 tumors was 93.9%/94.3%, while the sensitivity/specificity for T3 tumors was 93.8%/94.3%, while another study by Taylor et al. in 2007 found that MDCT and MRI were relatively similar in their accuracies for CRM involvement (27,28). Overall, while individual studies dating back over several years have shown variable results, with some studies demonstrating T-staging and CRM involvement accuracies that are acceptable, a large meta-analysis by Kwok et al. examining close to 500 patients found that MDCT had a sensitivity of only 78% for extension of tumor through the rectal wall (with an accuracy of only 73%), as well as a sensitivity and specificity for mesorectal lymph node metastasis of only 52% and 78% respectively (29-31). Overall, there is little doubt that MDCT should not be utilized as a 1st line modality for the local staging of rectal cancer, particularly with regard to T-staging and assessment of the CRM (32). However, in those cases with clear tumor extension outside the rectum, the radiologist should not hesitate to make the diagnosis of a T3 or T4 tumor, even given the limitations of MDCT.
Distant staging
The American College of Radiology recommends that all patients with colorectal cancer undergo a preoperative staging MDCT not only because of its proven efficacy in the identification of metastatic disease, but also because of its ability to identify complications that might alter a patient’s management (perforation, obstruction, abscess, pulmonary embolus, etc.) (2).
The most common site of distant metastases for colorectal cancer patients as a whole is the liver. These metastatic lesions tend to be most conspicuous on venous phase images, and will typically appear as hypoenhancing solid nodules that are easily juxtaposed against the avidly enhancing surrounding liver parenchyma (Figure 10). In some cases, the arterial phase images may be of benefit, as small liver metastases may demonstrate a rim of surrounding hyperemia, prominent peripheral enhancement or a surrounding perfusion abnormality that might increase lesion conspicuity. There is a wealth of data in the literature supporting the efficacy of MDCT in identifying colorectal cancer liver metastases: The overall sensitivity of MDCT for liver metastases is very good, with sensitivities ranging from 77-94% (33-35). Particularly with larger lesions (i.e., lesions measuring over 1 cm), MDCT is relatively specific as well, as most lesions measuring over 1 cm in size can be reliably differentiated from benign liver lesions (such as cysts or hemangiomas). However, while MDCT is excellent in identifying larger metastases, it struggles with smaller lesions measuring under 1 cm in size, with reported sensitivities dropping to as low as 41.9% (18). The specificity of MDCT is also suboptimal for lesions under 1 cm, as it can be difficult to differentiate a tiny cyst or hemangioma from an early liver metastasis with confidence. Unfortunately, this can be quite problematic, as these small, nonspecific hypodensities measuring <1 cm (also known as ‘too small to characterize’ hypodensities) are very common, perhaps present in as many as 17% of all patients (36). Nevertheless, in the vast majority of cases, even in those patients with a known underlying malignancy, these small hypodensities in the liver are overwhelmingly likely to be benign (~90%), and can be safely followed over time. As a result, the relative lack of specificity of MDCT for smaller lesion is not clinically important in the vast majority of cases. It should be noted that many of these studies judging the efficacy of MDCT in identifying and characterizing liver metastases were performed on older generation scanners with inferior spatial and temporal resolutions to the last generation of technology. Accordingly, it is quite likely that these studies underestimate the efficacy of MDCT, which is likely to be substantially higher than the numbers reported in these studies.
Figure 10.
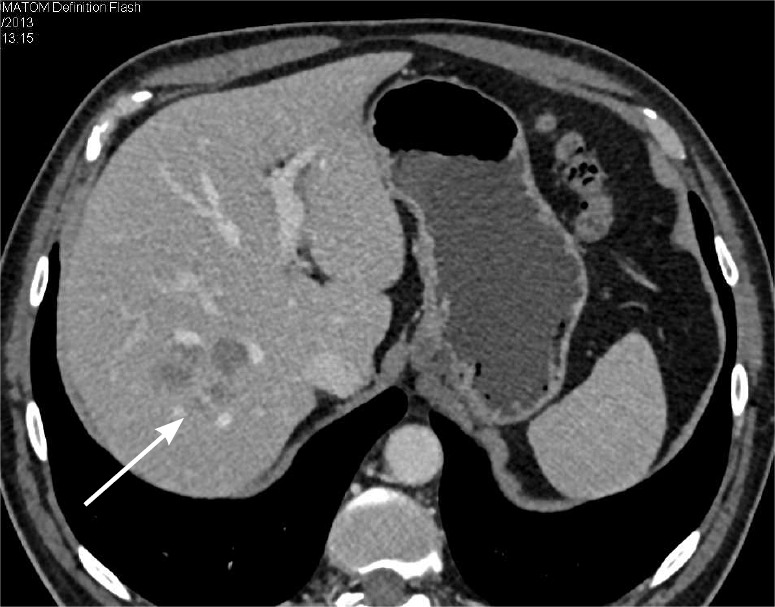
Typical MDCT appearance of colon cancer metastases to the liver. Axial contrast-enhanced MDCT image demonstrates small, ill-defined hypodense lesions (arrow) in the right hepatic lobe. MDCT, multidetector computed tomography.
Evaluation of lung metastases is also an important component of MDCT distant staging, and it is important that a chest CT be included when a patient undergoes their initial staging examination. In a study by Kirke et al., 17.9% of patients with rectal cancer had evidence of at least one pulmonary metastasis on MDCT, with an increasing risk of pulmonary metastasis with rising tumor grade (37). Just as importantly, rectal cancers seem more likely than other colon cancers to present with pulmonary metastases without liver metastases, likely reflecting the unique systemic venous drainage of the rectum compared to the remainder of the colon (2). Accordingly, the ACR guidelines recommend that a patient’s initial staging MDCT include images through the chest (2).
Unfortunately, as with MRI, MDCT has significant limitations in establishing a patient’s nodal status, largely because the diagnosis of a malignant lymph node is contingent on enlargement and size criteria. This is particularly a problem when evaluating mesorectal lymph nodes, where 95% of all malignant lymph nodes measure under 5 mm, and 50% of all malignant lymph nodes measure under 3 mm, making any size cut-off inaccurate (38). Although at least one study has suggested utilizing a size cut-off of 4.5 mm in the mesorectum, such a cut-off would clearly miss a sizeable number of positive lymph nodes (38). Not surprisingly, a study by Ju et al. found that MDCT had an accuracy of only 61.5% when evaluating perirectal lymph nodes (39).
Positron emission tomography (PET)
Technique
PET is a nuclear medicine examination utilizing 18F-fluoro-2-deoxy-D-glucose (FDG) as a primary tracer. This tracer, which acts glucose analog in the body, is transported into cells, phosphorylated, and subsequently accumulated, without entering the glycolytic cycle. Accordingly, given that many tumors demonstrate increased metabolism of glucose, FDG-PET utilizes the degree of FDG uptake as a surrogate measure of a tumor’s metabolic activity, and this uptake can be assessed both qualitatively (via visual examination of the degree of uptake of a tumor relative to other tissues) and quantitatively (via a SUV value). Not only is FDG taken up by tumors, but also there is also some degree of physiologic uptake by normal tissues and organs, including the bowel, renal collecting systems, muscle, fat, and brain. This places great importance on proper patient preparation prior to a study, as a patient’s blood glucose level, activity levels, ambient temperature, medications (particularly G-CSF), and food ingestion can all have a dramatic impact on the degree of uptake of FDG by not only by the tumor itself, but normal physiologic uptake as well. While PET was traditionally performed as a stand-alone examination, these studies are now almost always performed in conjunction with a CT (in dedicated PET-CT scanners), with acquisition of either a non-diagnostic non-contrast CT intended only for accurate localization of lesions or abnormalities seen on the PET portion of the study, or alternatively, a dedicated diagnostic quality intravenous contrast-enhanced CT meant to both serve both as a localizer for abnormalities on the PET, as well as a stand-alone diagnostic-quality MDCT examination (40,41).
Local staging
PET has a relatively low spatial resolution of only 5 mm, and as a result, is highly limited in its ability to locally stage tumors (Figure 11). Specifically, T-staging is not possible with PET-CT, as it has neither the anatomic detail (in terms of the layers of the rectal wall) or the spatial resolution to accurately judge the degree to which a tumor extends through the rectal wall (42). Moreover, PET is not particularly useful for evaluating locoregional lymph nodes in the mesorectum, as many of these perirectal or mesorectal lymph nodes measure 5 mm or less (below the resolution of PET), and moreover, ‘blooming’ (i.e., significant radiotracer uptake in a lesion artifactually appearing to extend into the adjacent soft tissues) from the primary lesion in the rectum can obscure uptake in small mesorectal lymph nodes (42). Nevertheless, while PET may not be of value in traditional TNM staging, it may have some value in terms of establishing a tumor’s ultimate prognosis based on examinations performed before and during a patient’s preoperative chemoradiation, although the data is certainly not conclusive. In a study by Lee et al., a formula utilizing the total lesion glycolysis (TLG) (a PET parameter) of the primary tumor was found to be predictive of a patient’s survival after neoadjuvant chemoradiation, a finding also confirmed elsewhere (43-47). Similarly, a meta-analysis by de Geus-Oei et al. suggested that PET-CT performed before and during a patient’s chemoradiation regimen was able to predict which patients would respond to the treatment (48). In addition, as some groups have begun to advocate for a “watch and wait” approach after chemoradiation for rectal cancer, choosing to defer surgery in those patients who have a clinical complete response (cCR) based on imaging, it is conceivable that pre- and post-therapy PET might offer a better correlation with “true pathologic response” compared to digital rectal examination, sigmoidoscopy, or other imaging studies (CT, MRI), although this will certainly require far more rigorous study if this treatment algorithm becomes more widely utilized (49).
Figure 11.
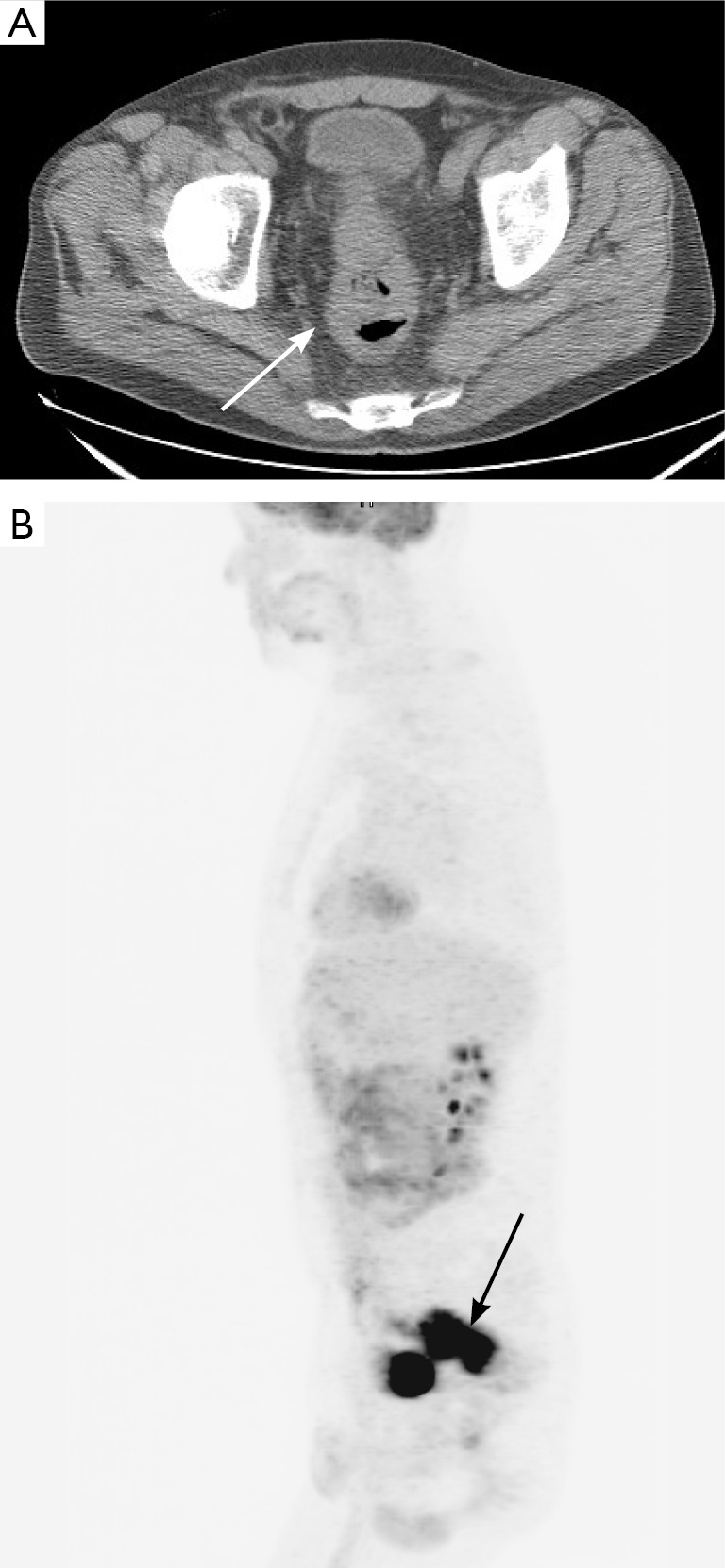
Axial non-contrast, non-diagnostic CT image (A) acquired as part of a PET-CT examination demonstrates severe mass-like thickening (arrow) of the rectum, corresponding to the patient’s known rectal cancer. PET image (B) demonstrates marked FDG uptake associated with the mass (arrow). Notably, the spatial resolution of PET does not allow local T staging of the lesion.
Distant staging
PET-CT serves as a very important modality in the distant staging of patients with colorectal cancer, potentially identifying 30% more distant metastases compared to MDCT (Figure 12) (42). In a study by Llamas-Elvira et al. PET showed an excellent diagnostic accuracy of 92% (as opposed to 87% for MDCT), changed the patient’s stage in 13.5% of cases, identified previously unknown disease in 19.2% of cases, changed the patient’s planned surgery in 11.5% of cases, and changed the patient’s therapy in 17.8% of cases (50). Another study by Abdel-Nabi et al. found PET-CT to be superior to MDCT in identifying liver metastases (51), while a study by Gearhart et al. found that PET-CT upstaged 50% of patients, downstaged 21% of patients, and changed the patient’s treatment plan in 27% of patients (52). This study noted that PET-CT was particularly likely to identify ‘discordant’ findings (i.e., findings not identified on MDCT) in patients with low rectal cancers due to the propensity of this group of lesions to metastasize to local lymph nodes in the pelvis (particularly nodes in the inguinal, femoral, or iliac chains), as PET-CT identified metastatic lymphadenopathy in 13.5% of patients in this study which were not diagnosed on MDCT (52).
Figure 12.
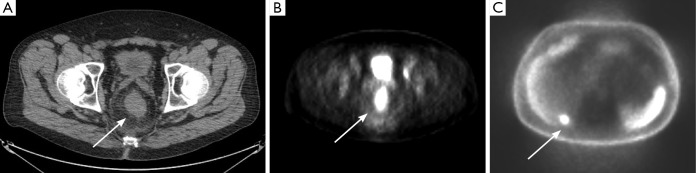
Axial non-contrast, non-diagnostic CT image (A) demonstrates mass-like thickening (arrow) of the rectum, corresponding to the patient’s known rectal cancer. PET image (B) at the same level demonstrates marked FDG uptake associated with the mass. PET image (C) though the liver demonstrates an occult metastasis (arrow), which was not identifiable on the patient’s formal contrast-enhanced MDCT. MDCT, multidetector computed tomography.
Conclusions
MRI, MDCT, and PET are complementary imaging modalities in the preoperative staging of patients with rectal cancer, and each offers their own individual strengths and weaknesses. MRI is clearly the best available radiologic modality for the local staging of patients with rectal cancer, and has the potential to play an important role in accurately distinguishing which patients should receive preoperative chemoradiation prior to total mesorectal excision. Alternatively, while MDCT and PET are both quite limited in local staging, both should be considered primary modalities when performing preoperative distant staging. In particular, every patient with a newly diagnosed rectal cancer should undergo a preoperative staging MDCT which includes the chest, abdomen, and pelvis, as MDCT can not only accurately stage distant metastatic disease, but it can also identify acute complications which may change a patient’s treatment algorithm. Alternatively, PET may offer a valuable diagnostic adjunct for identifying distant metastatic disease, changing a patient’s management in a sizeable percentage of cases.
Acknowledgements
Disclosure: The authors declare no conflict of interest.
References
- 1.Samee A, Selvasekar CR. Current trends in staging rectal cancer. World J Gastroenterol 2011;17:828-34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Dewhurst C, Rosen MP, Blake MA, et al. ACR Appropriateness Criteria pretreatment staging of colorectal cancer. J Am Coll Radiol 2012;9:775-81. [DOI] [PubMed] [Google Scholar]
- 3.Dewhurst CE, Mortele KJ. Magnetic resonance imaging of rectal cancer. Radiol Clin North Am 2013;51:121-31. [DOI] [PubMed] [Google Scholar]
- 4.McKeown E, Nelson DW, Johnson EK, et al. Current approaches and challenges for monitoring treatment response in colon and rectal cancer. J Cancer 2014;5:31-43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Glimelius B, Tiret E, Cervantes A, et al. Rectal cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol 2013;24Suppl 6:vi81-8. [DOI] [PubMed] [Google Scholar]
- 6.Kaur H, Choi H, You YN, et al. MR imaging for preoperative evaluation of primary rectal cancer: practical considerations. Radiographics 2012;32:389-409. [DOI] [PubMed] [Google Scholar]
- 7.Gowdra Halappa V, Corona Villalobos CP, Bonekamp S, et al. Rectal imaging: part 1, High-resolution MRI of carcinoma of the rectum at 3 T. AJR Am J Roentgenol 2012;199:W35-42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Samdani T, Garcia-Aguilar J.Imaging in rectal cancer: magnetic resonance imaging versus endorectal ultrasonography. Surg Oncol Clin N Am 2014;23:59-77. [DOI] [PubMed] [Google Scholar]
- 9.Merkel S, Mansmann U, Siassi M, et al. The prognostic inhomogeneity in pT3 rectal carcinomas. Int J Colorectal Dis 2001;16:298-304. [DOI] [PubMed] [Google Scholar]
- 10.Al-Sukhni E, Milot L, Fruitman M, et al. Diagnostic accuracy of MRI for assessment of T category, lymph node metastases, and circumferential resection margin involvement in patients with rectal cancer: a systematic review and meta-analysis. Ann Surg Oncol 2012;19:2212-23. [DOI] [PubMed] [Google Scholar]
- 11.Videhult P, Smedh K, Lundin P, et al. Magnetic resonance imaging for preoperative staging of rectal cancer in clinical practice: high accuracy in predicting circumferential margin with clinical benefit. Colorectal Dis 2007;9:412-9. [DOI] [PubMed] [Google Scholar]
- 12.Purkayastha S, Tekkis PP, Athanasiou T, et al. Diagnostic precision of magnetic resonance imaging for preoperative prediction of the circumferential margin involvement in patients with rectal cancer. Colorectal Dis 2007;9:402-11. [DOI] [PubMed] [Google Scholar]
- 13.MERCURY Study Group . Diagnostic accuracy of preoperative magnetic resonance imaging in predicting curative resection of rectal cancer: prospective observational study. BMJ 2006;333:779. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Cho EY, Kim SH, Yoon JH, et al. Apparent diffusion coefficient for discriminating metastatic from non-metastatic lymph nodes in primary rectal cancer. Eur J Radiol 2013;82:e662-8. [DOI] [PubMed] [Google Scholar]
- 15.Heijnen LA, Lambregts DM, Mondal D, et al. Diffusion-weighted MR imaging in primary rectal cancer staging demonstrates but does not characterise lymph nodes. Eur Radiol 2013;23:3354-60. [DOI] [PubMed] [Google Scholar]
- 16.Wiggans MG, Shahtahmassebi G, Aroori S, et al. Assessment of the value of MRI scan in addition to CT in the pre-operative staging of colorectal liver metastases. J Gastrointest Cancer 2014;45:146-53. [DOI] [PubMed] [Google Scholar]
- 17.Raman SP, Horton KM, Fishman EK. Multimodality imaging of pancreatic cancer-computed tomography, magnetic resonance imaging, and positron emission tomography. Cancer J 2012;18:511-22. [DOI] [PubMed] [Google Scholar]
- 18.Berger-Kulemann V, Schima W, Baroud S, et al. Gadoxetic acid-enhanced 3.0 T MR imaging versus multidetector-row CT in the detection of colorectal metastases in fatty liver using intraoperative ultrasound and histopathology as a standard of reference. Eur J Surg Oncol 2012;38:670-6. [DOI] [PubMed] [Google Scholar]
- 19.Holzapfel K, Reiser-Erkan C, Fingerle AA, et al. Comparison of diffusion-weighted MR imaging and multidetector-row CT in the detection of liver metastases in patients operated for pancreatic cancer. Abdom Imaging 2011;36:179-84. [DOI] [PubMed] [Google Scholar]
- 20.Malayeri AA, El Khouli RH, Zaheer A, et al. Principles and applications of diffusion-weighted imaging in cancer detection, staging, and treatment follow-up. Radiographics 2011;31:1773-91. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Raman SP, Horton KM, Fishman EK. MDCT and CT angiography evaluation of rectal bleeding: the role of volume visualization. AJR Am J Roentgenol 2013;201:589-97. [DOI] [PubMed] [Google Scholar]
- 22.Raman SP, Horton KM, Fishman EK. Transitional cell carcinoma of the upper urinary tract: optimizing image interpretation with 3D reconstructions. Abdom Imaging 2012;37:1129-40. [DOI] [PubMed] [Google Scholar]
- 23.Raman SP, Horton KM, Fishman EK. MDCT evaluation of ureteral tumors: advantages of 3D reconstruction and volume visualization. AJR Am J Roentgenol 2013;201:1239-47. [DOI] [PubMed] [Google Scholar]
- 24.Raman SP, Horton KM, Fishman EK. Computed tomography of Crohn's disease: The role of three dimensional technique. World J Radiol 2013;5:193-201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Juchems MS, Ernst AS, Kornmann M, et al. Value of MDCT in preoperative local staging of rectal cancer for predicting the necessity for neoadjuvant radiochemotherapy. Rofo 2009;181:1168-74. [DOI] [PubMed] [Google Scholar]
- 26.Vliegen R, Dresen R, Beets G, et al. The accuracy of Multi-detector row CT for the assessment of tumor invasion of the mesorectal fascia in primary rectal cancer. Abdom Imaging 2008;33:604-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Kanamoto T, Matsuki M, Okuda J, et al. Preoperative evaluation of local invasion and metastatic lymph nodes of colorectal cancer and mesenteric vascular variations using multidetector-row computed tomography before laparoscopic surgery. J Comput Assist Tomogr 2007;31:831-9. [DOI] [PubMed] [Google Scholar]
- 28.Taylor A, Slater A, Mapstone N, et al. Staging rectal cancer: MRI compared to MDCT. Abdom Imaging 2007;32:323-7. [DOI] [PubMed] [Google Scholar]
- 29.Sinha R, Verma R, Rajesh A, et al. Diagnostic value of multidetector row CT in rectal cancer staging: comparison of multiplanar and axial images with histopathology. Clin Radiol 2006;61:924-31. [DOI] [PubMed] [Google Scholar]
- 30.Matsuoka H, Nakamura A, Masaki T, et al. Preoperative staging by multidetector-row computed tomography in patients with rectal carcinoma. Am J Surg 2002;184:131-5. [DOI] [PubMed] [Google Scholar]
- 31.Kwok H, Bissett IP, Hill GL. Preoperative staging of rectal cancer. Int J Colorectal Dis 2000;15:9-20. [DOI] [PubMed] [Google Scholar]
- 32.Beets-Tan RG, Beets GL, Borstlap AC, et al. Preoperative assessment of local tumor extent in advanced rectal cancer: CT or high-resolution MRI? Abdom Imaging 2000;25:533-41. [DOI] [PubMed] [Google Scholar]
- 33.Larsen LP, Rosenkilde M, Christensen H, et al. Can contrast-enhanced ultrasonography replace multidetector-computed tomography in the detection of liver metastases from colorectal cancer? Eur J Radiol 2009;69:308-13. [DOI] [PubMed] [Google Scholar]
- 34.Wicherts DA, de Haas RJ, van Kessel CS, et al. Incremental value of arterial and equilibrium phase compared to hepatic venous phase CT in the preoperative staging of colorectal liver metastases: an evaluation with different reference standards. Eur J Radiol 2011;77:305-11. [DOI] [PubMed] [Google Scholar]
- 35.Mainenti PP, Mancini M, Mainolfi C, et al. Detection of colo-rectal liver metastases: prospective comparison of contrast enhanced US, multidetector CT, PET/CT, and 1.5 Tesla MR with extracellular and reticulo-endothelial cell specific contrast agents. Abdom Imaging 2010;35:511-21. [DOI] [PubMed] [Google Scholar]
- 36.Jones EC, Chezmar JL, Nelson RC, et al. The frequency and significance of small (less than or equal to 15 mm) hepatic lesions detected by CT. AJR Am J Roentgenol 1992;158:535-9. [DOI] [PubMed] [Google Scholar]
- 37.Kirke R, Rajesh A, Verma R, et al. Rectal cancer: incidence of pulmonary metastases on thoracic CT and correlation with T staging. J Comput Assist Tomogr 2007;31:569-71. [DOI] [PubMed] [Google Scholar]
- 38.Perez RO, Pereira DD, Proscurshim I, et al. Lymph node size in rectal cancer following neoadjuvant chemoradiation--can we rely on radiologic nodal staging after chemoradiation? Dis Colon Rectum 2009;52:1278-84. [DOI] [PubMed] [Google Scholar]
- 39.Ju H, Xu D, Li D, et al. Comparison between endoluminal ultrasonography and spiral computerized tomography for the preoperative local staging of rectal carcinoma. Biosci Trends 2009;3:73-6. [PubMed] [Google Scholar]
- 40.Dibble EH, Karantanis D, Mercier G, et al. PET/CT of cancer patients: part 1, pancreatic neoplasms. AJR Am J Roentgenol 2012;199:952-67. [DOI] [PubMed] [Google Scholar]
- 41.Shrikhande SV, Barreto SG, Goel M, et al. Multimodality imaging of pancreatic ductal adenocarcinoma: a review of the literature. HPB (Oxford) 2012;14:658-68. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Grassetto G, Marzola MC, Minicozzi A, et al. F-18 FDG PET/CT in rectal carcinoma: where are we now? Clin Nucl Med 2011;36:884-8. [DOI] [PubMed] [Google Scholar]
- 43.Lee SJ, Kim JG, Lee SW, et al. Clinical implications of initial FDG-PET/CT in locally advanced rectal cancer treated with neoadjuvant chemoradiotherapy. Cancer Chemother Pharmacol 2013;71:1201-7. [DOI] [PubMed] [Google Scholar]
- 44.Gulec SA, Suthar RR, Barot TC, et al. The prognostic value of functional tumor volume and total lesion glycolysis in patients with colorectal cancer liver metastases undergoing 90Y selective internal radiation therapy plus chemotherapy. Eur J Nucl Med Mol Imaging 2011;38:1289-95. [DOI] [PubMed] [Google Scholar]
- 45.Grassetto G, Capirci C, Marzola MC, et al. Colorectal cancer: prognostic role of 18F-FDG-PET/CT. Abdom Imaging 2012;37:575-9. [DOI] [PubMed] [Google Scholar]
- 46.Murcia Duréndez MJ, Frutos Esteban L, Luján J, et al. The value of 18F-FDG PET/CT for assessing the response to neoadjuvant therapy in locally advanced rectal cancer. Eur J Nucl Med Mol Imaging 2013;40:91-7. [DOI] [PubMed] [Google Scholar]
- 47.Sun W, Xu J, Hu W, et al. The role of sequential 18(F) -FDG PET/CT in predicting tumour response after preoperative chemoradiation for rectal cancer. Colorectal Dis 2013;15:e231-8. [DOI] [PubMed] [Google Scholar]
- 48.de Geus-Oei LF, Vriens D, van Laarhoven HW, et al. Monitoring and predicting response to therapy with 18F-FDG PET in colorectal cancer: a systematic review. J Nucl Med 2009;50Suppl 1:43S-54S. [DOI] [PubMed] [Google Scholar]
- 49.Park IJ, Yu CS. Current issues in locally advanced colorectal cancer treated by preoperative chemoradiotherapy. World J Gastroenterol 2014;20:2023-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Llamas-Elvira JM, Rodríguez-Fernández A, Gutiérrez-Sáinz J, et al. Fluorine-18 fluorodeoxyglucose PET in the preoperative staging of colorectal cancer. Eur J Nucl Med Mol Imaging 2007;34:859-67. [DOI] [PubMed] [Google Scholar]
- 51.Abdel-Nabi H, Doerr RJ, Lamonica DM, et al. Staging of primary colorectal carcinomas with fluorine-18 fluorodeoxyglucose whole-body PET: correlation with histopathologic and CT findings. Radiology 1998;206:755-60. [DOI] [PubMed] [Google Scholar]
- 52.Gearhart SL, Frassica D, Rosen R, et al. Improved staging with pretreatment positron emission tomography/computed tomography in low rectal cancer. Ann Surg Oncol 2006;13:397-404. [DOI] [PubMed] [Google Scholar]