Abstract
Objective
The aim of this study was to investigate the role of pravastatin, as an adjunctive therapy, on inflammatory markers, lipid and glucose metabolism, psychopathology, and cognition in subjects with schizophrenia and schizoaffective disorder.
Methods
Schizophrenia or schizoaffective subjects (N=60) were randomized to receive either a 12-week supply of pravastatin 40 mg/day or placebo treatment. Anthropometric measures, lipids and glucose metabolism, inflammatory markers, psychopathology and cognitive performance were assessed at baseline, 6 weeks and 12 weeks.
Results
Pravastatin use was associated with a significant decrease in total cholesterol, low density lipoprotein (LDL) cholesterol and LDL particle number levels, but was not associated with any significant changes in cognition or psychopathology in the participants, except a significant decrease in the Positive and Negative Syndrome Scale (PANSS) positive symptoms score from baseline to week 6. However, this decrease failed to remain significant at 12 weeks. Interestingly, triglycerides, LDLCholesterol, Total cholesterol, LDL particle number, small LDL particle number, large very low density lipoprotein (VLDL) particle number and c-reactive protein (CRP) followed a similar pattern at 6 and 12 weeks as psychopathology.
Conclusions
These results suggest that a randomized trial with a larger sample size and a higher dosage of pravastatin, and would be helpful in further evaluating the anti-inflammatory properties of pravastatin, its association with improvements in cognitive symptoms, and its potential to reduce positive and negative symptoms associated with schizophrenia or schizoaffective disorders.
Keywords: statins, psychosis, inflammation, c-reactive protein, cognitive functioning, glucose metabolism
1. INTRODUCTION
Immune dysfunction and inflammation have been described in patients with schizophrenia (Miller et al., 2014; Najjar and Pearlman, 2014). Prenatal infections and maternal immune alterations have been implicated as significantly increasing the risk of schizophrenia in offspring and schizophrenia-related neurocognitive/neuroanatomical abnormalities (Brown et al., 2004; Gilmore et al., 2004; Meyer et al., 2009; Miller et al., 2013). Neuroinflammation can injure developing oligodendroglia, resulting in prominent white matter pathology and motor, cognitive, and behavioral impairment which are all associated with pre-psychosis and schizophrenia (Chew et al., 2013; Najjar and Pearlman, 2014). Elevated levels of cytokines have been described not only in schizophrenia patients but also in acutely relapsed patient with schizophrenia (Miller et al., 2011) and in unaffected first degrees relatives of patients with schizophrenia (Martinez-Gras et al., 2012). Previous research has also suggested a relationship between inflammation and acute coronary syndromes as well as metabolic syndrome (Libby et al., 2014; Maury and Brichard, 2010).
Pro-inflammatory cytokines such as tumor necrosis factor (TNF)-α, interleukin (IL)-1β and IL-6 are soluble polypeptide signaling proteins that play crucial roles in the early defense against infection and the initiation and/or progression of inflammation (Meyer et al., 2009). Cytokines, particularly interleukin-6 (IL-6), are the primary inducers of acute phase proteins, including C-reactive protein (CRP) which is an acute phase protein produced by hepatocytes whose serum concentration increases under acute and chronic inflammatory conditions (Agrawal, 2005; Black et al., 2004; Miller et al., 2014).
In the past decade, it has been clearly established that obesity, insulin resistance and type 2 diabetes are closely associated with chronic inflammation characterized by abnormal cytokine production; increased acute-phase reactants and other mediators; and activation of a network of inflammatory signaling pathways (Miller et al., 2011; Wellen and Hotamisligil, 2005).
Several studies have suggested that inflammatory and immunological processes are likely related to the manifestation of symptoms and treatment response of schizophrenia (McAllister et al., 1995; Zhang et al., 2004). Our group has reported that elevated levels of CRP are associated with marked negative symptoms and higher positive and negative syndrome scale scores (PANSS) in patients with schizophrenia (Fan et al., 2007). In a multi-center, cross-sectional study with a sample of 199 schizophrenia outpatients, we found that elevated white blood cell (WBC) counts are associated with worse clinical symptoms of schizophrenia as measured by the Brief Psychiatric Rating Scale (BPRS) total scores after controlling for potential confounding variables (Fan et al., 2010).
Inflammation is also associated with impaired cognition in schizophrenia. In a sample of 413 patients with schizophrenia, Dickerson et al. reported that those with CRP > 5 mg/mL had significantly lower cognitive scores than those with CRP < 5 mg/mL (Dickerson et al., 2007).
Some recent experimental studies have shown that besides their effects on the primary and secondary prevention of cardiovascular diseases, statins may also have beneficial anti-inflammatory effects through diverse mechanisms that have been recognized from in vivo and in vitro studies in experimental models (Forero-Pena and Gutierrez, 2013). Statins reduce atherogenesis and concomitantly the inflammatory state as reflected by the decreased serum levels of CRP. The mechanism by which statins lower CRP levels is still unknown (Pearson et al., 2009; Ridker et al., 2005a; Ridker et al., 2008).
Growing evidence from clinical studies with COX-2 inhibitors shows that anti-inflammatory drugs may have beneficial effects in schizophrenia, in particular in an early stage of the disorder (Muller et al., 2012). Drugs that possess anti-inflammatory effect might improve both psychiatric symptoms and metabolic disturbances in patients with schizophrenia.
Given the need for additional treatments, the present study was designed to determine if such a treatment warrants further investigation. We chose to use pravastatin, one of the most extensively studied statins in both primary and secondary prevention trials(del Sol and Nanayakkara, 2008), available in generic form, because it appears to have fewer side effects than other statins such as simvastatin (Kaesemeyer et al., 1999; Ridker et al., 2005a; Ridker et al., 2005b). Additionally, some research suggests that pravastatin can significantly lower CRP levels and IL-6 concentration, as well as significantly improve insulin resistance (Asanuma et al., 2008; Güçlü et al., 2004; Ridker et al., 1999).
We conducted a 12-week, randomized, double-blind, placebo-controlled pilot study of pravastatin 40 mg/day, as an adjunctive therapy in 60 schizophrenia subjects to examine pravastatin’s effects on inflammatory markers, lipid and glucose metabolism, psychopathology, cognition, and positive and negative symptom scales (PANSS).
2. METHODS
Subjects were recruited from the Freedom Trail Clinic at the Erich Lindemann Mental Health Center and procedues were performed at the Mallinckrodt General Clinical Research Center at the Massachusetts General Hospital (MGH), Boston. The study was approved by the institutional review boards of MGH and the Massachusetts Department of Mental Health. A total of 81 male and female outpatients between the ages of 18 and 68 years with the diagnosis of schizophrenia, any subtype, or schizoaffective disorder, any subtype, were screened for the study and 60 were randomized. After providing written informed consent, subjects underwent a diagnostic evaluation by a research psychiatrist using the Structured Clinical Interview for DSM-IV Axis I Disorders (SCID).
Subjects who were treated and compliant with their outpatient medications (any antipsychotic) were eligible for participation. Subjects were excluded on the basis of inability to provide informed consent, participation in other research studies, unstable psychiatric illness, current alcohol or substance abuse, current treatment with insulin, pregnancy, untreated thyroid disease, significant medical illness including severe cardiovascular, hepatic, or renal disease (serum creatinine > 1.5 mg/dL), anemia (hemoglobin < 11.0 mg/dL), history of severe head injury, or untreated muscle disease. Subjects treated with the following medications known to affect glucose tolerance were also excluded: anti-inflammatory drugs (including aspirin and ibuprofen), thiazide diuretics, agents that induce weight loss, or St John’s Wort. Similarly, subjects treated with colchicine, azole antifungals, macrolide antibiotics, HIV protease inhibitors, or a known hypersensitivity to pravastatin or to any of its components were excluded from the study. Baseline results of subjects with low density lipoprotein (LDL) Cholesterol >160 mg/dl with no co-morbidities, LDL >130 mg/dl for people with risk factors for cardiovascular disease, and LDL>100 mg/dl for people with diabetes mellitus were sent to the subjects’ primary care physician with their written consent. Subjects who did not received lipid lowering medication treatment within 6 weeks following the referral to the PCP were reassessed to determine eligibility for randomization. Study subjects who were eligible for randomization received a 12-week supply of pravastatin (40 mg/day) or placebo.
2.1 Baseline evaluation
Baseline medical evaluation included weight, height, waist circumference, and vital signs with sitting and standing blood pressure, Abnormal Involuntary Movement Scale (AIMS), a physical examination and a nutritional assessment (including the evaluation of a food record, indirect calorimetry, and anthropometric measures). A 12-lead EKG was obtained and analyzed by the Department of Cardiology at the Massachusetts General Hospital.
Blood samples were obtained for fasting glucose and insulin levels, comprehensive metabolic assessment (kidney and liver function, electrolytes and acid/base disorders), lipid profile, complete blood count with differential, LDL particle size, CRP, IL-6, TNF-α, glycohemoglobin (HbA1c), and creatine kinase (CK). A urine drug screen and a urine pregnancy test were performed to determine if subjects met eligibility criteria.
2.2 Laboratory Assays
Laboratory assays were performed by the Chemistry Lab at the MGH and LabCorp. Serum insulin assays were performed using an Immulite Analyzer (Diagnostic Product Corporation, Los Angeles, CA) with an intra-assay coefficient of variation of 4.2–7.6%. Fasting plasma glucose was measured with a hexokinase reagent kit (A-gent glucose test, Abbott, South Pasadena, California). Glucose assays were run in duplicate, and the intra-assay coefficient of variation ranged from 2% to 3%. The plasma concentrations of IL-6 were measured by a commercially available enzyme-linked immunosorbent assay (R&D Systems, Minneapolis, Minn). Serum levels of CRP were measured via a high-sensitivity latex-enhanced immunonephelometric assay on a BN II analyzer (Dade Behring, Newark, Del), which allows precision in the very low ranges of CRP values (0.2 mg/L) compared to the regular CRP test. Fasting total plasma cholesterol and triglyceride levels were measured enzymatically (McNamara and Schaefer, 1987), and the high density lipoprotein (HDL) cholesterol fraction were measured after precipitation of low-density and very-low-density lipoproteins with dextran sulfate-magnesium (Warnick et al., 1982). LDL cholesterol values were estimated indirectly for participants with plasma triglyceride levels less than 400 mg/dL (when above this value LDL cholesterol was measured directly) (Friedewald et al., 1972).
Screening fasting lipid profiles were analyzed at the MGH Core Lab. LDL and HDL subclass particle concentrations and mean LDL and HDL particle diameters were measured with an automated Nuclear Magnetic Resonance (NMR) spectroscopic assay (Otvos, 2002). In brief, the particle concentrations of lipoprotein subclasses of different size were derived from the measured amplitudes of the distinct lipid methyl group NMR signals they emit. In the present study, the following lipoprotein subclasses were assessed: LDL particle, small LDL particle, large LDL particle, large HDL particle, and large VLDL particle.
Weighted-average LDL particle sizes in nanometers were calculated from the subclass levels (LipoScience, Raleigh, NC).
2.3 Psychopathology, Cognition and Assessment for Treatment Emergent Events
Positive and Negative Syndrome Scale (PANSS) (Kay et al., 1987), the Scale for the Assessment of Negative Symptoms (SANS) (Andreasen, 1989), Hamilton Rating Scale for Depression (HAM-D) (Hamilton, 1960), Quality of Life Questionnaire (QOL) (Heinrichs et al., 1984), and the Global Assessment Scale (GAS) (Endicott et al., 1976) were used to determine baseline and subsequent changes in psychopathology. The Measurement and Treatment Research to Improve Cognition in Schizophrenia (MATRICS) (Kern et al., 2008) was used to evaluate the level of cognitive functioning. Everyday functioning was assessed by the UCSD Performance-Based Skills Assessment-Brief (UPSA-B) (Patterson et al., 2001). Side effects were monitored at each visit using the Systematic Assessment for Treatment Emergent Events (SAFTEE-SI) (Levine and Schooler, 1986).
2.4 Statistical analysis
Statistical analyses were performed using the SAS version 9.2 statistical package (SAS Institute, Cary, NC). Differences between treatment groups with respect to baseline demographic variables were tested using two sample t-tests for continuous variables and Fisher’s exact test for categorical variables. The primary outcome measures in this study were change in LDL cholesterol, CRP, PANSS total score, and the NIMH MATRICS Neuropsychological Battery composite score over 12 weeks. Secondary outcome measures included the change over 12 weeks in inflammatory markers IL-6 and TNF-alpha, HDL and LDL particle size, fasting triglycerides, HOMA-IR, and anthropometric measures including body weight, BMI, and waist circumference. The effect of treatment on these outcome measures over 12 weeks were tested using analysis of covariance (ANCOVA) with change scores as the dependent variable and baseline score and treatment group as covariates. Additional ANCOVA models with six-week change score as the dependent variable and baseline score and treatment group as covariates were used to investigate whether treatment effects might be observable after just six weeks.
As a secondary analysis, the effect of treatment on the outcome measures LDL cholesterol, CRP, PANSS total score, and the NIMH MATRICS Neuropsychological Battery composite score were tested using repeated measures analysis of variance (ANOVA) models with baseline, week 6, and week 12 outcome measures as dependent variables, and treatment group, time, and a treatment by time interaction as possible covariates. If a model of linear change over time did not hold, a linear spline model with a knot at week 6 was used. For all models, likelihood ratio tests were used to test among nested covariance structures, and Akaine information criterion (AIC) was used to compare non-nested covariance structures.
Since previous studies have shown that statins may be more beneficial in subjects with CRP above 2 mg/L (Ridker et al., 2005a; Ridker et al., 2008; Ridker et al., 2001), we repeated the analysis in a subgroup of subjects on pravastatin with CRP > 2 mg/L and a subgroup of subjects on placebo with CRP > 2 mg/L. For all analyses, a p-value less than 0.05 (2-tailed) was used to indicate statistical significance.
3. RESULTS
3.1 Demographics
In total, 81 subjects were screened. Among them, 68 were enrolled and 60 were randomized to either pravastatin 40 mg/day or placebo treatment. A total of 49 subjects completed the study (N= 24 in the pravastatin group and N= 25 in the placebo group) (Figure 1).
Figure 1.
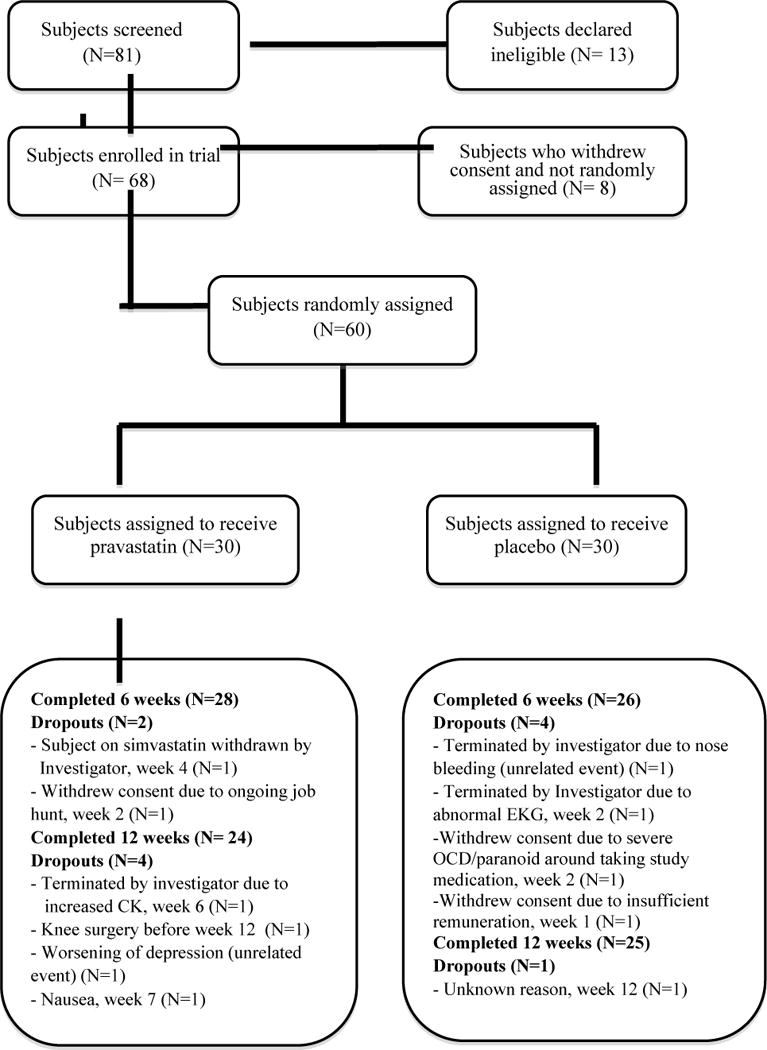
Subjects who entered and completed the study.
OCD, Obsessive Compulsive Disorder
CK, Creatine Kinase
EKG, Electrocardiogram
The baseline demographic characteristics of the study participants are summarized in Table 1.
Table 1.
Demographic characteristics of study participants
| Pravastatin | Placebo | |||||||
|---|---|---|---|---|---|---|---|---|
|
|
||||||||
| N | Mean | S.D. | N | Mean | S.D. | t | p-Value | |
|
|
||||||||
| Age | 30 | 42.57 | 11 | 30 | 44.53 | 12.55 | 0.65 | 0.47 |
| Age of disease onset | 29 | 21.1 | 7.37 | 30 | 22.03 | 6.74 | 0.62 | |
| Disease duration | 29 | 20.89 | 13.13 | 30 | 22.5 | 12.77 | 0.64 | |
| Smoking duration | 10 | 27.6 | 11.55 | 14 | 29.5 | 16.18 | 0.75 | |
| Pravastatin | Placebo | |||||
|---|---|---|---|---|---|---|
|
|
||||||
| N | % | N | % | p-Value | ||
|
|
||||||
| Gender | Female | 8 | 27 | 14 | 47 | 0.18 |
| Male | 22 | 73 | 16 | 53 | ||
| Race | Caucasian | 19 | 63 | 20 | 67 | 0.67 |
| African American | 4 | 13 | 5 | 17 | ||
| Asian | 4 | 13 | 1 | 3 | ||
| Hispanic | 1 | 3 | 3 | 10 | ||
| Native American | 1 | 3 | 0 | 0 | ||
| Other | 1 | 3 | 1 | 3 | ||
| Hispanic Ethnicity | Hispanic | 0 | 0 | 3 | 10 | 0.24 |
| Not Hispanic | 30 | 100 | 27 | 90 | ||
| Marital Status | Married | 1 | 3 | 1 | 3 | 1 |
| Not married | 29 | 97 | 29 | 97 | ||
| Smoking status | Smokers | 10 | 33 | 14 | 47 | 0.43 |
| Non smokers | 20 | 67 | 16 | 53 | ||
| Education | Middle school/some high school | 4 | 14 | 6 | 20 | 0.83 |
| High School diploma | 9 | 31 | 10 | 33 | ||
| Some college or higher | 16 | 55 | 14 | 47 | ||
| Diagnoses | Schizophrenia | 17 | 57 | 16 | 53 | 1 |
| Schizoaffective | 13 | 43 | 14 | 47 | ||
| Medications | Typical Antipsychotics** | 2 | 25 | 6 | 75 | 0.25 |
| Atypical antipsychotics | 23 | 49 | 24 | 51 | 1 | |
| Clozapine | 11 | 58 | 8 | 42 | 0.58 | |
| Olanzapine | 5 | 50 | 5 | 50 | 1 | |
| Aripiprazole | 7 | 54 | 6 | 46 | 1 | |
| Quetiapine | 3 | 60 | 2 | 40 | 1 | |
| Ziprasidone | 1 | 20 | 4 | 80 | 0.35 | |
| Paliperidone | 3 | 75 | 1 | 25 | 0.61 | |
| Risperidone | 6 | 67 | 3 | 33 | 0.47 | |
| Two Antipsychotics*** | 12 | 75 | 4 | 25 | 0.04 | |
P-value obtained using Fisher’s exact test
Loxapine, Thiothixene, Perphenazine, Chlorpromazine
Any combination, typical and atypical antipsychotics
There were no statistically significant differences between the placebo and pravastatin groups by age, gender, race, ethnicity, diagnosis or marital status at diagnosis.
3.2 Anthropometric measures
There were no statistically significant treatment effects in the ANCOVA models assessing change in body weight, Body Mass Index (BMI), or waist circumference from baseline to week 6 or from baseline to week 12 (Table 2).
Table 2.
Comparison of outcome measures between the pravastatin group and the placebo group
| Pravastatin | Placebo | Week 6- Baseline | Week 12- Baseline | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
|
||||||||||||||||
| Baseline (N=30) |
Six weeks (N=28) |
Twelve weeks (N=24) |
Baseline (N=30) |
Six weeks (N= 26) |
Twelve weeks (N=25) |
|||||||||||
|
|
||||||||||||||||
| Mean | SD | Mean | SD | Mean | SD | Mean | SD | Mean | SD | Mean | SD | t | P* | t | P* | |
| Body weight (lb) | 193.31 | 44.22 | 192.36 | 45.55 | 189.46 | 44.69 | 202.91 | 46.2 | 198.51 | 43.28 | 199.64 | 42.29 | 1.47 | 0.15 | −1.39 | 0.17 |
| BMI (kg/m2) | 29.7 | 6.19 | 29.51 | 6.34 | 29.27 | 6.65 | 33.04 | 8.4 | 32.36 | 7.92 | 32.89 | 7.74 | 1.04 | 0.3 | −1.24 | 0.22 |
| Waist Circumference (umbilicus, cm) | 105.15 | 15.24 | 103.15 | 15.65 | 104.18 | 16.1 | 109.94 | 19.01 | 104.57 | 28.14 | 104.23 | 28.31 | 0.76 | 0.45 | 0.7 | 0.48 |
| Fasting plasma glucose (mg/dl) | 99.26 | 17.37 | 96.57 | 12.38 | 103.16 | 19.29 | 99.5 | 14.55 | 99.42 | 15.16 | 101.2 | 15.89 | 1.04 | 0.3 | −0.63 | 0.53 |
| Fasting serum insulin (mIU/ml) | 9.03 | 6.74 | 9.58 | 7.95 | 12.83 | 14.39 | 11.99 | 9.85 | 9.49 | 7.16 | 12.35 | 12.31 | −0.89 | 0.38 | −0.49 | 0.63 |
| HOMA-IR | 2.26 | 1.8 | 2.33 | 1.88 | 3.78 | 5.64 | 3.15 | 3.08 | 2.49 | 2.23 | 3.3 | 3.78 | −0.47 | 0.64 | −0.59 | 0.56 |
| HbA1c (%) | 5.63 | 0.43 | 5.63 | 0.47 | 5.64 | 0.48 | 5.68 | 0.47 | 5.65 | 0.46 | 5.71 | 0.53 | −0.28 | 0.78 | 0.51 | 0.61 |
| LDL-C (mg/dl) | 106.22 | 28.23 | 79.57 | 28.09 | 85.13 | 25.38 | 104.03 | 22.81 | 107.46 | 24.54 | 104.21 | 19.34 | 4.63 | <.0001 | 3.94 | 0.0003 |
| HDL-C (mg/dl) | 48.1 | 12.57 | 47.32 | 10.66 | 47.91 | 12.04 | 45.36 | 10.07 | 45.65 | 10.6 | 47.3 | 10.59 | 0.93 | 0.36 | −0.11 | 0.91 |
| Triglycerides (mg/dl) | 174.4 | 158.32 | 127.32 | 75.31 | 134.33 | 92.47 | 145.46 | 104.24 | 145.73 | 91.69 | 135.39 | 81.44 | 1.84 | 0.07 | 1.05 | 0.3 |
| Total cholesterol (mg/dl) | 179.63 | 33.41 | 152.14 | 35.09 | 161.04 | 34.18 | 178.2 | 29.45 | 182.26 | 30.7 | 178.56 | 28.4 | 4.72 | <.0001 | 3.53 | 0.001 |
| LDL Particle number (nmol/l) | 1459.07 | 481.91 | 1221.43 | 483.79 | 1317.88 | 384.39 | 1444.17 | 435.99 | 1463.58 | 414.75 | 1543.22 | 604.72 | 2.83 | 0.01 | 3.39 | 0.002 |
| LDL Particle size (nm) | 20.9 | 0.64 | 20.87 | 0.48 | 20.18 | 3.08 | 20.95 | 0.63 | 21 | 0.56 | 21 | 0.55 | 1.03 | 0.31 | 1.22 | 0.23 |
| Small LDL particle Number (nmol/l) | 705.16 | 440.34 | 581.17 | 345.68 | 652.2 | 335.96 | 708.13 | 503.04 | 730.5 | 367.58 | 745.82 | 491.25 | 2.03 | 0.05 | 1.75 | 0.01 |
| Large HDL particle Number (μumol/l) | 4.44 | 4.09 | 4.53 | 3.27 | 4.52 | 3.33 | 4.36 | 3.15 | 4.25 | 3.17 | 4.52 | 2.96 | −0.05 | 0.96 | −2.1 | 0.04 |
| Large VLDL particle Number (mmol/l) | 6.66 | 9.4 | 4.25 | 4.84 | 4.94 | 6.58 | 5.75 | 8.39 | 5.45 | 6.89 | 5.24 | 6.96 | 1.35 | 0.18 | 0.99 | 0.33 |
| CRP (mg/L) | 1.8 | 2.02 | 1.58 | 1.81 | 2.71 | 5.51 | 4.69 | 11.52 | 3.13 | 6.9 | 4.03 | 7.96 | 0.39 | 0.7 | −0.07 | 0.94 |
| IL-6 | 3.15 | 2.78 | 2.92 | 2.25 | 2.93 | 1.84 | 3.06 | 2.68 | 3.02 | 2.67 | 3.18 | 2.91 | −0.2 | 0.84 | −0.17 | 0.87 |
| TNF-alpha | 2.29 | 0.95 | 2.73 | 2.51 | 2.46 | 1.32 | 2.16 | 1.46 | 2.11 | 1.46 | 2.04 | 1.252 | 0.17 | 0.87 | 1.65 | 0.11 |
Notes: 1) BMI, body mass index;2) HOMA-IR, homeostasis model of assessment of insulin resistance; 3) HbA1c, hemoglobin A1c; 4) CRP, Complement Reactive protein
P-values obtained from ANCOVA models using change scores as the dependent variable, and baseline score and treatment group as covariates
3.3 Lipids Metabolism and Inflammatory Markers
Pravastatin use was associated with a significantly greater decrease from baseline to week 6 and baseline to week 12 in LDL cholesterol (Week 6 – Baseline ANCOVA: p<.0001 and Week 12 – Baseline ANCOVA: p=0.0003), total cholesterol (Week 6 – Baseline ANCOVA: p<0001 and Week 12 – Baseline ANCOVA: p=0.001), and small LDL particle numbers (Week 6 – Baseline ANCOVA: p= 0.05 and Week 12 – Baseline ANCOVA: p=0.009). It was also associated with a significantly greater decrease from baseline to week 12 for LDL particle number (ANCOVA p=0.002). Moreover, pravastatin was associated with a significantly greater increase from baseline to week 12 in large HDL particle number (ANCOVA p= 0.04). Pravastatin use did not result in a significant change in CRP, LDL particle size, fasting triglycerides, Il-6, or TNF-α (Table 2).
Among the subgroup of subjects with CRP above 2 mg/L, pravastatin use was again associated with a significantly greater decrease in LDL cholesterol from baseline to both weeks 6 and 12 (Week 6 – Baseline ANCOVA: p=.02 and Week 12 – Baseline ANCOVA: p=0.013), and from baseline to week 12 for LDL particle number (ANCOVA p=0.002).
Similar results were obtained from the repeated measures ANOVA (Figure 2). For total cholesterol, the assumption of linearity over time did not hold (χ(2) = 16, p = 0.0003), so a linear spline model with a knot at week 6 was fit to the data. The interaction of treatment group and time between baseline and week 6 was statistically significant, and indicated a larger decrease in total cholesterol in the Pravastatin group during this time period (t(97) = −4.75, p < 0.0001). From week 6 to week 12 the interaction of treatment group and time was not statistically significant (t(97) = 0.73, p = 0.47). Similarly, for LDL cholesterol the assumption of linearity over time also did not hold (χ(2) = 13.9, p = 0.001), and a linear spline model with a knot at week 6 was again fit to the data. The interaction of treatment group and time between baseline and week 6 was again statistically significant, and indicated a larger decrease in LDL cholesterol in the Pravastatin group (t(97) = −4.79, p < 0.0001). From week 6 to week 12 the interaction was not statistically significant (t(97) = 0.48, p = 0.63). Since the assumption of linearity over time was satisfied for CRP (χ(2) = 4.3, p = 0.12), and the interaction between time and treatment group was not statistically significant (t(58) = 0.97, p = 0.34), a model including the main effects of time and treatment group were fit to the data but neither main effect was statistically significant (time t(58) = −0.39, p = 0.70; treatment t(58) = −0.94, p = 0.35). It is important to note that several patients had outlying CRP observations, but exclusion of those patients from the analysis did not substantively change any conclusions.
Figure 2.
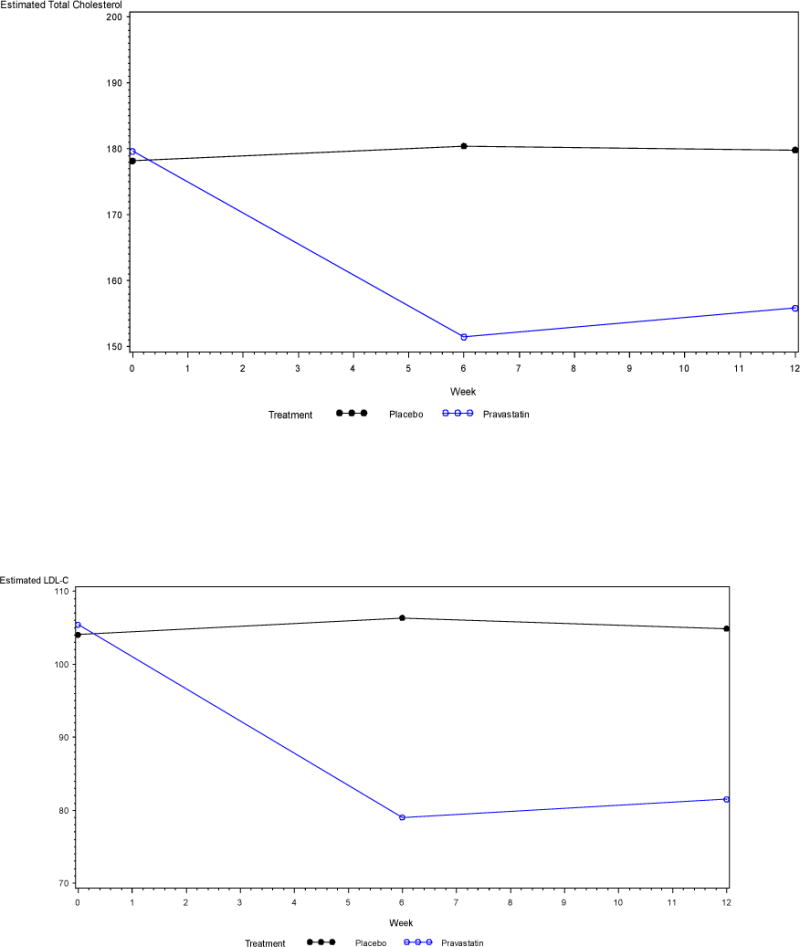
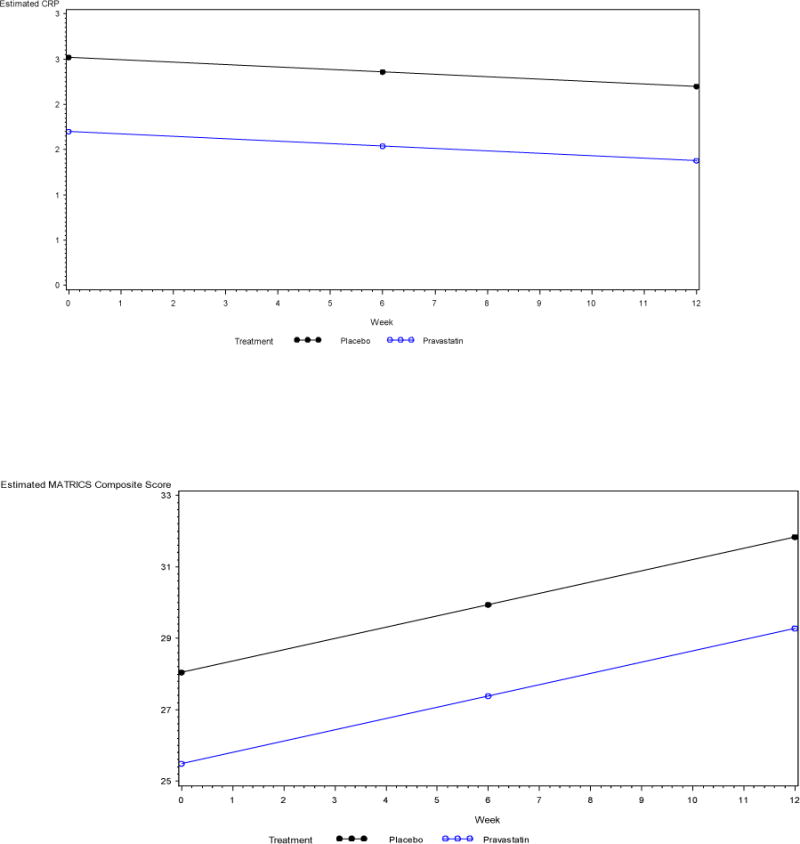
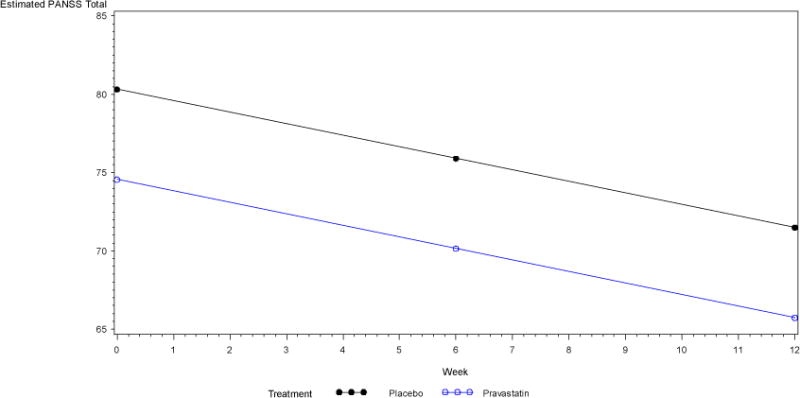
Estimated outcomes obtained from the repeated measures ANOVA
LDL-C, Low Density Lipoprotein Cholesterol
CRP, C-Reactive Protein
MATRICS, Measurement and Treatment Research to Improve Cognition in Schizophrenia
PANSS, Positive and Negative Syndrome Scale
3.4 Glucose metabolism
Pravastatin use was not significantly associated with change in glucose metabolism measurements such as fasting glucose, fasting insulin, HOMA-IR (homeostasis model of assessment of insulin resistance) and HbA1c (hemoglobin A1c) from baseline to either week 6 or week 12 (table 2). Similar results were obtained among the subgroup of subjects with CRP above 2 mg/L.
3.5 Psychopathology and cognitive performance
Pravastatin was not significantly associated with change in psychopathology or cognitive performance measurements from baseline to week 6 or week 12, except for a significant reduction in the PANSS Positive Symptoms Scale, which showed a statistically significant reduction of positive symptoms at 6 weeks from baseline (ANCOVA p=0.01) but failed to remain significant at 12 weeks from baseline (ANCOVA p=0.12) (Tables 3 and 4).
Table 3.
Comparison of cognitive performance between the pravastatin group and the placebo group
| Pravastatin | Placebo | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
|
||||||||||||||||
| Baseline (N=30) | Six weeks (N=28) | Twelve weeks (N=24) | Baseline (N=30) | Six weeks (N=26) | Twelve weeks (N=25) | |||||||||||
|
|
||||||||||||||||
| Mean | SD | Mean | SD | Mean | SD | Mean | SD | Mean | SD | Mean | SD | t | p* | t | p* | |
| Attention/vigilance | 32.12 | 12.66 | 35.25 | 12.47 | 34.04 | 11.69 | 37.33 | 12.77 | 37.72 | 13.92 | 38.04 | 15.19 | 0.79 | 0.43 | −0.26 | 0.8 |
| Working memory | 32.04 | 16.41 | 33.62 | 16.4 | 35.79 | 14.6 | 34.4 | 12.59 | 36.56 | 11.86 | 35.8 | 11.51 | −0.46 | 0.65 | 1.11 | 0.27 |
| Verbal Learning | 37.04 | 11.46 | 39.91 | 12.24 | 41.41 | 13.71 | 38.76 | 12.47 | 38.2 | 9.81 | 43.24 | 11.57 | 1.69 | 0.1 | −0.14 | 0.89 |
| Visual Learning | 35.5 | 15.26 | 37.16 | 15.42 | 38.33 | 19.13 | 33.48 | 11.71 | 35.16 | 13.96 | 37.68 | 10.86 | 0.21 | 0.83 | −0.3 | 0.76 |
| Problem solving | 39.87 | 9.25 | 42.41 | 9.35 | 42.45 | 11.03 | 42.6 | 9.02 | 44.84 | 9.69 | 46.16 | 11.17 | −0.17 | 0.87 | −0.57 | 0.57 |
| Social cognition | 36.95 | 9.41 | 37.08 | 13.86 | 36 | 11.64 | 34.96 | 15.08 | 35.6 | 15.13 | 35.76 | 13.68 | −0.17 | 0.86 | −0.6 | 0.55 |
| MATRICS composite Score | 25.33 | 16.98 | 28.87 | 18.79 | 29.37 | 18.09 | 28.75 | 12.31 | 29.8 | 13.44 | 32.04 | 13.11 | 0.72 | 0.47 | −0.11 | 0.91 |
P-values obtained from ANCOVA models using change scores as the dependent variable, and baseline score and treatment group as covariates
Table 4.
Comparison of psychopathology (PANSS) between the pravastatin group and the placebo group
| Pravastatin | Placebo | Week 6- Baseline | Week 12- Baseline | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
|
||||||||||||||||
| Baseline (N=30) | Six weeks (N=28) | Twelve weeks (N=24) | Baseline (N=30) | Six weeks (N=26) | Twelve weeks (N=25) | |||||||||||
|
|
||||||||||||||||
| Mean | SD | Mean | SD | Mean | SD | Mean | SD | Mean | SD | Mean | SD | t | p* | t | p* | |
| PANSS Total | 75.16 | 21.87 | 66.64 | 16.67 | 65.54 | 18.65 | 79.93 | 18.53 | 73.88 | 17 | 72.44 | 16.88 | −1.18 | 0.24 | −1.25 | 0.22 |
| PANSS Positive Total | 17 | 6.83 | 13.82 | 5.17 | 14.12 | 5.54 | 20.58 | 7.4 | 18.88 | 7.13 | 18 | 6.28 | −2.59 | 0.01 | −1.61 | 0.12 |
| PANSS Negative Total | 20.53 | 6.43 | 19.67 | 5.91 | 19.54 | 6.85 | 20.34 | 4.79 | 19.07 | 5.31 | 19.92 | 6.1 | 0.97 | 0.34 | −0.4 | 0.69 |
| PANSS General Total | 37.63 | 11.89 | 33.14 | 8.69 | 31.87 | 9.31 | 39 | 9.85 | 35.92 | 9.86 | 34.52 | 9.13 | −0.92 | 0.36 | −1.17 | 0.25 |
P-values obtained from ANCOVA models using change scores as the dependent variable, and baseline score and treatment group as covariates
Note: PANSS, Positive and Negative Syndrome Scale
With regards to cognitive performance, the subgroup of subjects with CRP above 2 mg/L, pravastatin was associated with a significant improvement from baseline to week 6 for attention/vigilance tasks (ANCOVA p= 0.02) and working memory tasks (ANCOVA p= 0.03). Significant associations between pravastatin use and changes in psychopathology measurements were not observed within this subgroup.
The assumption of linearity over time was satisfied for the repeated measures ANOVA modeling MATRICS composite score (χ(2) = 1.6, p = 0.45). The interaction between treatment group and time was not statistically significant (t(47) = −0.39, p = 0.69), so a model including only the main effects of treatment group and time was fit. The main effect for time was statistically significant, but the main effect for treatment group was not (time t(47) = 4.66, p < 0.0001; treatment t(47) = −0.61, p = 0.54). For PANSS total, the assumption of linearity over time held (χ(2) = 4.5, p = 0.11), and the interaction between time and treatment group was not statistically significant (t(101) = −0.57, p = 0.57).
3.6 Adverse effects
The only noteworthy side effect, which occurred in 5 subjects in the pravastatin group compare to 1 subject in the placebo group was muscle soreness. Subjects receiving pravastatin also reported (vs. placebo) headache (6 vs. 5), and head cold (8 vs. 6).
4. DISCUSSION
This was a randomized, placebo-controlled pilot study of pravastatin 40 mg/day as an adjunctive therapy in 60 randomized subjects with schizophrenia or schizoaffective disorder to examine pravastatin’s effect on inflammatory markers, psychopathology, cognition, lipid and glucose measurements. To our knowledge, no previous studies have looked at how statins affect CRP levels in patients with schizophrenia or schizoaffective disorder.
Pravastatin significantly decreased total cholesterol, LDL cholesterol and LDL particle number levels in our study in both the main study cohort and the subgroup of subjects with CRP >2mg/L. It was not associated with significantly greater decreases in CRP levels or other inflammation markers. There was no difference in subjects with CRP > 2mg/L vs CRP <2 mg/L.
It is important to point out that one subject in the pravastatin group had a CRP level of 1.8 mg/L at baseline, 1.73 mg/L at 6 weeks and 26.98 mg/L at 12 weeks. According to the literature, CRP serum level may rapidly increase as a consequence of tissue injury or infection (bacterial infection and burn (>200 mg/L), viral infection (10–40 mg/L), trauma, necrosis of tissue, ageing, most forms of inflammation, parasitic invasion and malignant neoplasia) (Ansar and Ghosh, 2013). The medical records of this subject showed evidence of bronchitis, which could explain such an increase in the CRP level at week 12. Although it is not statistically significant when we exclude this subject in our data analysis, the results show a decrease in CRP value in the pravastatin group both at week 6 and week 12 (N=23, 1.91±2.12 mg/L at baseline; 1.65±1.97 mg/L at week 6; 1.66±1.95 mg/L at week 12). Pravastatin failed to show any significant changes in cognition and psychopathology in the participants, except a significant decrease in the PANSS positive symptoms score at 6 weeks compared to baseline. However, this decrease failed to remain significant at 12 weeks. Interestingly, we found that triglycerides, LDL-Cholesterol, Total cholesterol, LDL particle number, small LDL particle number, large VLDL particle number and CRP followed a similar pattern as psychopathology: values decreased at 6 weeks and then increased at 12 weeks although they did not return to the baseline value. It is possible that the dosage of pravastatin used in our study was too low to maintain the decreased 6 weeks value and to lead to a further decrease in lipid measurements, inflammation markers and psychopathology measurements at 12 weeks.
Since inflammation may represent a common pathophysiological process related to both schizophrenia psychopathology and metabolic disturbances, we hypothesized that the use of drugs that possess anti-inflammatory effect such as pravastatin could significantly decrease the levels of inflammatory markers, lipid measurements and glucose measurements, and that this could positively affect cognitive functioning, and negative and positive symptoms in subjects with schizophrenia and schizoaffective disorder.
Some of the previously mentioned studies showed that statin therapy is more beneficial in subjects with CRP levels above 2 mg/L(Ridker et al., 2001) (Ridker et al., 2005a) (Ridker et al., 2008). In light of these data we proposed to analyze a subgroup of subjects enrolled in our study that would present level of CRP >2 mg/L. Moreover, since systemic inflammation has multiple effects including impaired glucose metabolism we suggest studying in the subgroup subjects with high measurements of glucose metabolism impairment (i.e. HOMA-IR) to eventually investigate the relationship between changes in impaired glucose metabolism and psychopathology in schizophrenia. Focusing on restricting the analysis to a subgroup of subjects with higher baseline markers of inflammation and insulin resistance measurements permitted analysis of the effect of statins in decreasing the inflammatory markers, and characterization of the relationship between changes in inflammation and changes in cognitive function, and negative and positive symptoms in patients with schizophrenia and schizoaffective disorder.
The limitations of this study are lack of a healthy control group and a moderate sample size. The study is underpowered to accommodate multiple testing.
Future studies are needed using a larger sample size and a higher dosage of pravastatin to examine the long term effects of adjunctive statin therapy in subjects with schizophrenia or schizoaffective disorder on antipsychotics with baseline levels of CRP>2 mg/L. Future studies that compare pravastatin to other triglyceride-lowering agents like omega-3 fatty acids, topiramate and metformin could also be beneficial in order to further our understanding of the relationship between inflammatory markers, cognition and the psychopathology of schizophrenia.
5. CONCLUSIONS
Inflammation may represent a common pathophysiological process related to schizophrenia psychopathology, cognition and metabolic disturbances. Drugs that decrease inflammation might be beneficial in treating both psychiatric symptoms and medical comorbidity like diabetes, hyperlipidemia and obesity in patients with schizophrenia. Recent studies have demonstrated a beneficial role of statins in reducing inflammatory markers. However, it is still unclear whether statins may have clinical benefits that go beyond lowering cholesterol.
Further studies focused on the anti-inflammatory properties of statins may be beneficial to determine whether the use of statins can significantly improve cognitive symptoms and reduce positive and negative symptoms in patients with schizophrenia or schizoaffective disorders.
Acknowledgments
None
Funding
This work was supported by The Stanley Medical Research Institute
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
Conflicts of Interest
The Authors have declared that there are no conflicts of interest in relation to the subject of this study.
CONTRIBUTORS
Brenda Vincenzi, M.D. managed the analysis and interpretation of data and wrote the first draft of the manuscript
Shannon Stock, Ph.D. undertook the statistical analysis
Sarah M. Cleary, B.A., Claire E. Oppenheim, B.S. and Liana J. Petruzzi, B.A. managed the acquisition of data and the literature searches and analyses
Xiaoduo Fan, MD MPH, Paul M. Copeland, M.D., Oliver Freudenreich, M.D. and Corinne Cather, Ph.D. offered a critical revision of the manuscript for important intellectual content
David C. Henderson, M.D. and Christina P.C. Borba, Ph.D., MPH designed the study, wrote the protocol and contributed to the interpretation of data
All authors contributed to and have approved the final manuscript.
Contributor Information
Brenda Vincenzi, Email: bvincenzi@partners.org, Schizophrenia Clinical and Research Program, Massachusetts General Hospital, Boston, Massachusetts, 25 Staniford Street, Boston, MA 02114
Shannon Stock, Email: sstock@holycross.edu, Department of Mathematics and Computer Science, College of the Holy Cross, Worcester, Massachusetts
Christina P.C. Borba, Email: cborba@partners.org, Schizophrenia Clinical and Research Program, Massachusetts General Hospital, Boston, Massachusetts, 25 Staniford Street, Boston, MA 02114; Harvard Medical School, Boston, Massachusetts.
Sarah M. Cleary, Email: sarah.m.cleary@gmail.com, Schizophrenia Clinical and Research Program, Massachusetts General Hospital, Boston, Massachusetts, 25 Staniford Street, Boston, MA 02114
Claire E. Oppenheim, Email: coppenheim@partners.org, Schizophrenia Clinical and Research Program, Massachusetts General Hospital, Boston, Massachusetts, 25 Staniford Street, Boston, MA 02114
Liana J. Petruzzi, Email: lpetruzzi@partners.org, Schizophrenia Clinical and Research Program, Massachusetts General Hospital, Boston, Massachusetts, 25 Staniford Street, Boston, MA 02114
Xiaoduo Fan, Email: xiaoduo.fan@umassmed.edu, University of Massachusetts Medical School UMass Memorial Medical Center, Worcester, Massachusetts, United States of America.
Paul M. Copeland, Email: pccopeland@partners.org, Harvard Medical School, Boston, Massachusetts Department of Medicine, Massachusetts General Hospital, Boston, Massachusetts.
Oliver Freudenreich, Email: Freudenreich.Oliver@partners.org, Schizophrenia Clinical and Research Program, Massachusetts General Hospital, Boston, Massachusetts, 25 Staniford Street, Boston, MA 02114 Harvard Medical School, Boston, Massachusetts.
Corinne Cather, Email: ccather@partners.org, Schizophrenia Clinical and Research Program, Massachusetts General Hospital, Boston, Massachusetts, 25 Staniford Street, Boston, MA 02114 Harvard Medical School, Boston, Massachusetts.
David C. Henderson, Email: dchenderson@partners.org, Schizophrenia Clinical and Research Program, Massachusetts General Hospital, Boston, Massachusetts, 25 Staniford Street, Boston, MA 02114 Harvard Medical School, Boston, Massachusetts.
References
- Agrawal A. CRP after 2004. Mol Immunol. 2005;42(8):927–930. doi: 10.1016/j.molimm.2004.09.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Andreasen NC. The Scale for the Assessment of Negative Symptoms (SANS): conceptual and theoretical foundations. Br J Psychiatry Suppl. 1989;(7):49–58. [PubMed] [Google Scholar]
- Ansar W, Ghosh S. C-reactive protein and the biology of disease. Immunol Res. 2013;56(1):131–142. doi: 10.1007/s12026-013-8384-0. [DOI] [PubMed] [Google Scholar]
- Asanuma Y, Oeser A, Stanley E, Bailey DG, Shintani A, Stein CM. Effects of C-reactive Protein and Homocysteine on Cytokine Production: Modulation by Pravastatin. Archives of Drug Information. 2008;1(1):14–22. doi: 10.1111/j.1753-5174.2007.00003.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Black S, Kushner I, Samols D. C-reactive Protein. J Biol Chem. 2004;279(47):48487–48490. doi: 10.1074/jbc.R400025200. [DOI] [PubMed] [Google Scholar]
- Brown AS, Hooton J, Schaefer CA, Zhang H, Petkova E, Babulas V, Perrin M, Gorman JM, Susser ES. Elevated maternal interleukin-8 levels and risk of schizophrenia in adult offspring. Am J Psychiatry. 2004;161(5):889–895. doi: 10.1176/appi.ajp.161.5.889. [DOI] [PubMed] [Google Scholar]
- Chew LJ, Fusar-Poli P, Schmitz T. Oligodendroglial alterations and the role of microglia in white matter injury: relevance to schizophrenia. Developmental neuroscience. 2013;35(2–3):102–129. doi: 10.1159/000346157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- del Sol AI, Nanayakkara PW. Pravastatin: an evidence-based statin? Expert opinion on drug metabolism & toxicology. 2008;4(6):821–825. doi: 10.1517/17425255.4.6.821. [DOI] [PubMed] [Google Scholar]
- Dickerson F, Stallings C, Origoni A, Boronow J, Yolken R. C-reactive protein is associated with the severity of cognitive impairment but not of psychiatric symptoms in individuals with schizophrenia. Schizophrenia research. 2007;93(1–3):261–265. doi: 10.1016/j.schres.2007.03.022. [DOI] [PubMed] [Google Scholar]
- Endicott J, Spitzer RL, Fleiss JL, Cohen J. The global assessment scale. A procedure for measuring overall severity of psychiatric disturbance. Arch Gen Psychiatry. 1976;33(6):766–771. doi: 10.1001/archpsyc.1976.01770060086012. [DOI] [PubMed] [Google Scholar]
- Fan X, Liu EY, Freudenreich O, Park JH, Liu D, Wang J, Yi Z, Goff D, Henderson DC. Higher white blood cell counts are associated with an increased risk for metabolic syndrome and more severe psychopathology in non-diabetic patients with schizophrenia. Schizophrenia research. 2010;118(1–3):211–217. doi: 10.1016/j.schres.2010.02.1028. [DOI] [PubMed] [Google Scholar]
- Fan X, Pristach C, Liu EY, Freudenreich O, Henderson DC, Goff DC. Elevated serum levels of C-reactive protein are associated with more severe psychopathology in a subgroup of patients with schizophrenia. Psychiatry research. 2007;149(1–3):267–271. doi: 10.1016/j.psychres.2006.07.011. [DOI] [PubMed] [Google Scholar]
- Forero-Pena DA, Gutierrez FR. Statins as modulators of regulatory T-cell biology. Mediators of inflammation. 2013;2013:167086. doi: 10.1155/2013/167086. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Friedewald WT, Levy RI, Fredrickson DS. Estimation of the concentration of lowdensity lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge. Clin Chem. 1972;18(6):499–502. [PubMed] [Google Scholar]
- Gilmore JH, Fredrik Jarskog L, Vadlamudi S, Lauder JM. Prenatal infection and risk for schizophrenia: IL-1beta, IL-6, and TNFalpha inhibit cortical neuron dendrite development. Neuropsychopharmacology. 2004;29(7):1221–1229. doi: 10.1038/sj.npp.1300446. [DOI] [PubMed] [Google Scholar]
- Güçlü F, Özmen B, Hekimsoy Z, Kirmaz C. Effects of a statin group drug, pravastatin, on the insulin resistance in patients with metabolic syndrome. Biomedicine & pharmacotherapy. 2004;58(10):614–618. doi: 10.1016/j.biopha.2004.09.005. [DOI] [PubMed] [Google Scholar]
- Hamilton M. A rating scale for depression. J Neurol Neurosurg Psychiatry. 1960;23:56–62. doi: 10.1136/jnnp.23.1.56. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heinrichs DW, Hanlon TE, Carpenter WT., Jr The Quality of Life Scale: an instrument for rating the schizophrenic deficit syndrome. Schizophrenia bulletin. 1984;10(3):388–398. doi: 10.1093/schbul/10.3.388. [DOI] [PubMed] [Google Scholar]
- Kaesemeyer WH, Caldwell RB, Huang J, Caldwell RW. Pravastatin sodium activates endothelial nitric oxide synthase independent of its cholesterol-lowering actions. J Am Coll Cardiol. 1999;33(1):234–241. doi: 10.1016/s0735-1097(98)00514-2. [DOI] [PubMed] [Google Scholar]
- Kay SR, Fiszbein A, Opler LA. The positive and negative syndrome scale (PANSS) for schizophrenia. Schizophrenia bulletin. 1987;13(2):261–276. doi: 10.1093/schbul/13.2.261. [DOI] [PubMed] [Google Scholar]
- Kern RS, Nuechterlein KH, Green MF, Baade LE, Fenton WS, Gold JM, Keefe RS, Mesholam-Gately R, Mintz J, Seidman LJ, Stover E, Marder SR. The MATRICS Consensus Cognitive Battery, part 2: co-norming and standardization. Am J Psychiatry. 2008;165(2):214–220. doi: 10.1176/appi.ajp.2007.07010043. [DOI] [PubMed] [Google Scholar]
- Levine J, Schooler NR. SAFTEE: a technique for the systematic assessment of side effects in clinical trials. Psychopharmacol Bull. 1986;22(2):343–381. [PubMed] [Google Scholar]
- Libby P, Tabas I, Fredman G, Fisher EA. Inflammation and its resolution as determinants of acute coronary syndromes. Circ Res. 2014;114(12):1867–1879. doi: 10.1161/CIRCRESAHA.114.302699. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martinez-Gras I, Garcia-Sanchez F, Guaza C, Rodriguez-Jimenez R, Andres-Esteban E, Palomo T, Rubio G, Borrell J. Altered immune function in unaffected first-degree biological relatives of schizophrenia patients. Psychiatry research. 2012;200(2–3):1022–1025. doi: 10.1016/j.psychres.2012.05.036. [DOI] [PubMed] [Google Scholar]
- Maury E, Brichard SM. Adipokine dysregulation, adipose tissue inflammation and metabolic syndrome. Mol Cell Endocrinol. 2010;314(1):1–16. doi: 10.1016/j.mce.2009.07.031. [DOI] [PubMed] [Google Scholar]
- McAllister CG, van Kammen DP, Rehn TJ, Miller AL, Gurklis J, Kelley ME, Yao J, Peters JL. Increases in CSF levels of interleukin-2 in schizophrenia: effects of recurrence of psychosis and medication status. Am J Psychiatry. 1995;152(9):1291–1297. doi: 10.1176/ajp.152.9.1291. [DOI] [PubMed] [Google Scholar]
- McNamara JR, Schaefer EJ. Automated enzymatic standardized lipid analyses for plasma and lipoprotein fractions. Clin Chim Acta. 1987;166(1):1–8. doi: 10.1016/0009-8981(87)90188-4. [DOI] [PubMed] [Google Scholar]
- Meyer U, Feldon J, Yee BK. A review of the fetal brain cytokine imbalance hypothesis of schizophrenia. Schizophrenia bulletin. 2009;35(5):959–972. doi: 10.1093/schbul/sbn022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miller BJ, Buckley P, Seabolt W, Mellor A, Kirkpatrick B. Meta-analysis of cytokine alterations in schizophrenia: clinical status and antipsychotic effects. Biological psychiatry. 2011;70(7):663–671. doi: 10.1016/j.biopsych.2011.04.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miller BJ, Culpepper N, Rapaport MH. C-reactive protein levels in schizophrenia: a review and meta-analysis. Clinical schizophrenia & related psychoses. 2014;7(4):223–230. doi: 10.3371/CSRP.MICU.020813. [DOI] [PubMed] [Google Scholar]
- Miller BJ, Culpepper N, Rapaport MH, Buckley P. Prenatal inflammation and neurodevelopment in schizophrenia: a review of human studies. Prog Neuropsychopharmacol Biol Psychiatry. 2013;42:92–100. doi: 10.1016/j.pnpbp.2012.03.010. [DOI] [PubMed] [Google Scholar]
- Muller N, Myint AM, Schwarz MJ. Immunological treatment options for schizophrenia. Current pharmaceutical biotechnology. 2012;13(8):1606–1613. doi: 10.2174/138920112800784826. [DOI] [PubMed] [Google Scholar]
- Najjar S, Pearlman DM. Neuroinflammation and white matter pathology in schizophrenia: systematic review. Schizophrenia research. 2014 doi: 10.1016/j.schres.2014.04.041. [DOI] [PubMed] [Google Scholar]
- Otvos JD. Measurement of lipoprotein subclass profiles by nuclear magnetic resonance spectroscopy. Clin Lab. 2002;48(3–4):171–180. [PubMed] [Google Scholar]
- Patterson TL, Goldman S, McKibbin CL, Hughs T, Jeste DV. UCSD Performance-Based Skills Assessment: development of a new measure of everyday functioning for severely mentally ill adults. Schizophrenia bulletin. 2001;27(2):235–245. doi: 10.1093/oxfordjournals.schbul.a006870. [DOI] [PubMed] [Google Scholar]
- Pearson TA, Ballantyne CM, Veltri E, Shah A, Bird S, Lin J, Rosenberg E, Tershakovec AM. Pooled analyses of effects on C-reactive protein and low density lipoprotein cholesterol in placebo-controlled trials of ezetimibe monotherapy or ezetimibe added to baseline statin therapy. Am J Cardiol. 2009;103(3):369–374. doi: 10.1016/j.amjcard.2008.09.090. [DOI] [PubMed] [Google Scholar]
- Ridker PM, Cannon CP, Morrow D, Rifai N, Rose LM, McCabe CH, Pfeffer MA, Braunwald E. C-reactive protein levels and outcomes after statin therapy. N Engl J Med. 2005a;352(1):20–28. doi: 10.1056/NEJMoa042378. [DOI] [PubMed] [Google Scholar]
- Ridker PM, Danielson E, Fonseca FA, Genest J, Gotto AM, Jr, Kastelein JJ, Koenig W, Libby P, Lorenzatti AJ, MacFadyen JG, Nordestgaard BG, Shepherd J, Willerson JT, Glynn RJ. Rosuvastatin to prevent vascular events in men and women with elevated C-reactive protein. N Engl J Med. 2008;359(21):2195–2207. doi: 10.1056/NEJMoa0807646. [DOI] [PubMed] [Google Scholar]
- Ridker PM, Morrow DA, Rose LM, Rifai N, Cannon CP, Braunwald E. Relative efficacy of atorvastatin 80 mg and pravastatin 40 mg in achieving the dual goals of low-density lipoprotein cholesterol <70 mg/dl and C-reactive protein <2 mg/l: an analysis of the PROVE-IT TIMI-22 trial. J Am Coll Cardiol. 2005b;45(10):1644–1648. doi: 10.1016/j.jacc.2005.02.080. [DOI] [PubMed] [Google Scholar]
- Ridker PM, Rifai N, Clearfield M, Downs JR, Weis SE, Miles JS, Gotto AM., Jr Measurement of C-reactive protein for the targeting of statin therapy in the primary prevention of acute coronary events. N Engl J Med. 2001;344(26):1959–1965. doi: 10.1056/NEJM200106283442601. [DOI] [PubMed] [Google Scholar]
- Ridker PM, Rifai N, Pfeffer MA, Sacks F, Braunwald E. Long-term effects of pravastatin on plasma concentration of C-reactive protein. Circulation. 1999;100(3):230–235. doi: 10.1161/01.cir.100.3.230. [DOI] [PubMed] [Google Scholar]
- Warnick GR, Benderson J, Albers JJ. Dextran sulfate-Mg2+ precipitation procedure for quantitation of high-density-lipoprotein cholesterol. Clin Chem. 1982;28(6):1379–1388. [PubMed] [Google Scholar]
- Wellen KE, Hotamisligil GS. Inflammation, stress, and diabetes. J Clin Invest. 2005;115(5):1111–1119. doi: 10.1172/JCI25102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang XY, Zhou DF, Cao LY, Zhang PY, Wu GY, Shen YC. Changes in serum interleukin-2, -6, and -8 levels before and during treatment with risperidone and haloperidol: relationship to outcome in schizophrenia. J Clin Psychiatry. 2004;65(7):940–947. doi: 10.4088/jcp.v65n0710. [DOI] [PubMed] [Google Scholar]


