Abstract
Purpose: The antiseptic qualities of aromatic and medicinal plants and their extracts have been recognized since antiquity, while attempts to characterize these properties in the laboratory date back the beginning of the XXth century. In the current study essential oils obtained from Pelargonium roseum (Geraniacea) were analyzed for their antibacterial and antifungal activities.
Methods: The antimicrobial activity of the Pelargonium essential oil was tested against Gram-negative bacteria (Pseudomonas aeruginosa, Proteus mirabilis, Escherichia coli), Gram-positive bacteria (Staphylococcus aureus, Enterococcus faecalis) and fungi (Candida albicans). Disc diffusion method was used to study antimicrobial activity.
Results: Inhibition zones showed that the studied essential oils were active against all of the studied bacteria. In the case of Candida albicans, the complete inhibition of the fungus’s development was observed. In the cases of Pseudomonas aeruginosa and Staphylococcus aureus we observed an inhibition comparable to that obtained by the use of an appropriate antimicrobial substance.
Conclusion: The volatile oils exhibited considerable inhibitory effects against all the organisms under test, in some cases comparable with those of the reference antibiotics. There were no considerable differences between the antimicrobial activities of the oil obtained by distillation and commercially available Pelargonium oils.
Keywords: Pelargonium roseum, Antimicrobial activity, Antifungal activity, Essential oils
Introduction
Pelargonium roseum (Pelargonium graveolens) is a species indigenous to Southern Africa (widespread especially in the Cape Town region) belonging to the Geraniaceae family, which was introduced in the Mediterranean part of Europe since the beginning of the eighteenth century.1
Pelargonium roseum has a woody, straight stem with branches; its leaves are usually alternate, palmately lobed or pinnate, often on long stalks, and sometimes with light or dark pattern; covered with short, rough hairs, which give the plant a strong, pleasant rose-like scent. The erect stems bear five-petaled flowers in umbel-like clusters; the flower has a single symmetry plane (zygomorphic), which distinguishes it from the Geranium flower, which has radial symmetry (actinomorphic) (Figure 1).1-3
Figure 1.
Pelargonium roseum
Other than being grown for their beauty as an ornamental plant, species of Pelargonium are important in the perfume industry and are cultivated and distilled for their scents. The distillates and absolutes (essential oils) extracted from Pelargonium species are frequently used in aromatherapy and massage therapy, and also sometimes as a supplement or to adulterate more expensive rose oils.4,5
In herbal medicine Pelargonium species has been used for intestinal problems, wounds and respiratory ailments; in recent years, respiratory/cold remedies containing Pelargonium oil have been sold in Europe and the United States. In several studies Pelargonium oil has been reported to help with hormonal balance, liver and kidney functions, to discharge toxins from the liver that may be impacting the body’s overall well-being; to support digestive, nervous and circulatory systems and to have antiseptic qualities.6-8
Essential oils derived from aromatic plants have demonstrated biological properties and can be used to prevent and treat human systemic diseases, including infectious diseases. Investigations into the antimicrobial activities, mode of action and potential uses of plant volatile oils have regained momentum, along with the revival in the use of traditional antibacterial approaches.5
The aim of this study was the assessment of the antibacterial and antifungal activity of Pelargonium roseum oil against different genera of bacteria and fungi, and compare these to the effect of certain frequently used antibacterial substances upon bacterial growth.
Materials and Methods
Cultivation of Pelargoonium roseum species was carried out in the herb garden of the University of Medicine and Pharmacy from Tîrgu Mureş, Romania, on a total area of 200 m2, over a period of three years. Harvesting of the aerial parts was performed in 3 stages; in July we harvested the ramifications of stalks with 3-5 leaves, in August the ramifications of well-developed stalks and the third harvest was made at the end of the vegetation period, in September. Throughout the cultivation period no herbicides were used, because we wanted to preserve unspoiled the qualities of the essential oils obtained subsequently.
Plant volatile oils are generally isolated from non-woody plant material by distillation methods, in this particular case the green material was the subject of steam hydrodistillation for a period 3 hours. Before fragmenting the green mass for distillation we removed the woody stems and the dried leaves. After hydrodistillation, the oil was dehydrated over anhydrous sodium sulphate in order to retain any residual traces of water, and kept in a cool and dark place before the analyses.
Essential oil content varied between 0.082% and 0.16% being dependent on the proportion of leaves-stalks subject to distillation. The essential oil obtained after distillation was a transparent, slight light green liquid with a pleasant scent, reminiscent to the flagrance of roses. Three different samples of essential oils corresponding to three different years of cultivation were analyzed (S1, S2, S3). The analysis of antimicrobial and antifungal activity of the essential oil extracted from Pelargonium by hydrodistillation was compared with four commercial samples of essential oils with high purity degree received from different climatic zones (S4 - China, S5 - Reunion, S6 - France, S7 - Egypt), using microbiological methods.
The microorganisms used in order to test the antimicrobial activity of the essential oil were: Gram-negative bacteria (Pseudomonas aeruginosa ATCC 27853, Proteus mirabilis ATCC 29906, Escherichia coli ATCC 25992), Gram-positive bacteria (Staphylococcus aureus ATCC 25923, Enterococcus faecalis ATCC 19433), fungi (Candida albicans ATC 10231). Blood agar plates were used for the bacteria cultures while Sabouraud agar was used for the fungi culture. Seeding the culture medium under aseptic conditions was made with a sterilized platinum loop and was followed by incubation. Disc diffusion method was used to study antimicrobial activity, impregnating slices of filter paper with oil.
The paper slices impregnated with oil were placed in Petri dishes on an agar culture media incubated at 37°C for 24 hours, to allow diffusion of the sample into the agar. Following this procedure, zones of growth inhibition were measured. Six repetitions of the experiment were made in order to evaluate the antimicrobial and antifungal activity of the tested products.
The intensity of the antimicrobial effect was tested in comparison with reference antibacterial substances, used as microcapsules for antibiograms: fluoroquinolones (ciprofloxacin - CIP), sulfonamides (cotrimoxazol – sulfomethoxazol + trimetoprim - CTR), penicillins (ampicillin - AMP), cephalosporins (cefalotin - CFL, cefotaxim - CFT), aminoglycozides (gentamicin - GEN, amikacin - AMK).
Results and Discussion
One of our previous studies regarding the analysis of the essential oil by gas chromatography was published before; 61 components were detected, out of which 43 were identified. Citronellol (39.97-43.67 %) was found to be the main component of the essential oil, followed by geraniol (2.57–9.66 %), linalool (1.41-4.77 %) and their esters: citronellyl formate (11.23-13.55 %), geranyl formate (1.15-2.21 %), citronellyl butyrate (1.01-1.54 %), geranyl tiglate (1.22-1.75 %).9
Certain differences between the chemical compositions of different oils from different sources have been observed. It is interesting to mention that the main component S5 was not citronellol but geranyl (22.7 %). Significant concentrations of isomenthone (4.61-6.14 %), guaia-6, 9-diene (1.06-7.06 %) and gamma-eudesmol (3.28-5.62 %) were also detected.9
Other studies dealing with the chemical composition, antifungal and antioxidant activity of Pelargonium essential oil confirms that the main compounds are citronellol, geraniol, citronellyl formate and linalool.8,10
Table 1 summarizes the antibacterial activity of the studied volatile oils.
Table 1. Testing antimicrobial and antifungal activity of oils obtained from Pelargonium roseum in comparison with reference antibacterial substances.
| Tested strains | Inhibition zones for volatile oils (mm) | Inhibition zones for antibacterial substances (mm) | ||||||||||||
| S1 | S2 | S3 | S4 | S5 | S6 | S7 | AMK | AMP | CFL | CFT | CIP | CTR | GEN | |
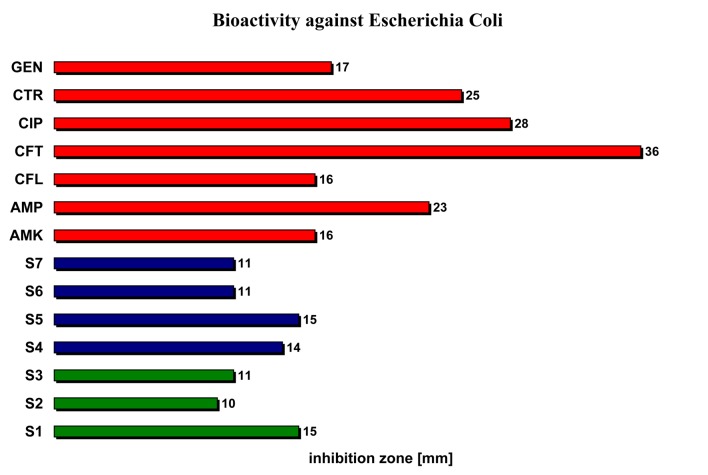
| Escherichia coli | 15 | 10 | 11 | 14 | 15 | 11 | 11 | 16 | 23 | 16 | 36 | 28 | 25 | 17 |
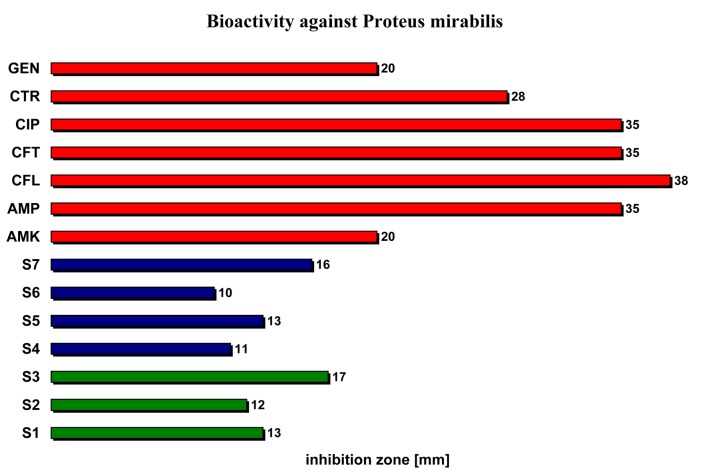
| Proteus mirabilis | 13 | 12 | 17 | 11 | 13 | 10 | 16 | 20 | 35 | 38 | 35 | 35 | 28 | 20 |
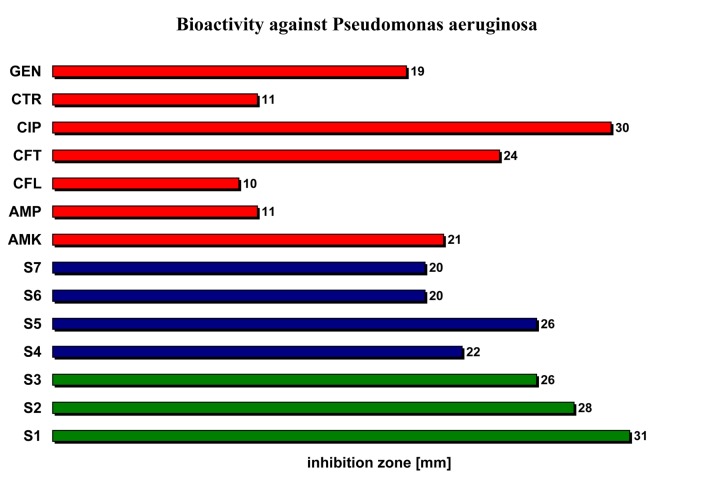
| Pseudomonas aeruginosa | 31 | 28 | 26 | 22 | 26 | 20 | 20 | 21 | 11 | 10 | 24 | 30 | 11 | 19 |
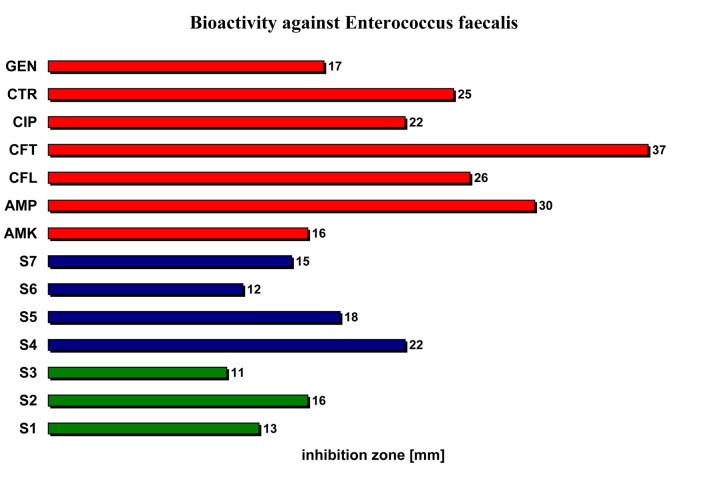
| Enterococcus faecalis | 13 | 16 | 11 | 22 | 18 | 12 | 15 | 16 | 30 | 26 | 37 | 22 | 25 | 17 |
| Staphylococcus aureus | 31 | 22 | 25 | 21 | 28 | 21 | 25 | 22 | 29 | 38 | 35 | 25 | 28 | 22 |
| Candida albicans | all | all | all | all | all | all | all | - | - | - | - | - | - | - |
Testing the activity of the studied volatile oils against Eserichia coli (Enterobacteriaceae) we noticed that their antibacterial activity is in averege two times lower compared with the reference antimicrobial substances (10-15 cm versus 16-36 cm), but a distinct small inhibition zone was identifiable (Figure 2).
Figure 2.
The bioactivity of the studied volatile oils relative to reference antibacterial substances against Eserichia coli.
Testing the activity of the studied volatile oils against Proteus mirabilis (Enterobacteriaceae) we noticed that their antibacterial activity is in averege two times lower compared with the reference antimicrobial substances (10-17 cm versus 20-38 cm) (Figure 3).
Figure 3.
The bioactivity of the studied volatile oils relative to reference antibacterial substances against Proteus mirabilis.
Testing the antibacterial activity of the studied volatile oils against Pseudomonas aeruginisa (Pseudomonacee) we noticed that their antibacterial activity is comparable with most sensitive reference antibacterial substance (ciprofloxacin - CIP) (31 cm for S1 versus 30 cm for CIP) (Figure 4)
Figure 4.
The bioactivity of the studied volatile oils relative to reference antibacterial substances against Pseudomonas aeruginisa.
Enterococcus faecalis (Enterococcacea) proved to be less sensitive to the studied volatile oils, their antibacterial activity being in averege lower compared with the reference antimicrobial substances (11-22 cm versus 16-37 cm) (Figure 5).
Figure 5.
The bioactivity of the studied volatile oils relative to reference antibacterial substances against Enterococcus faecalis.
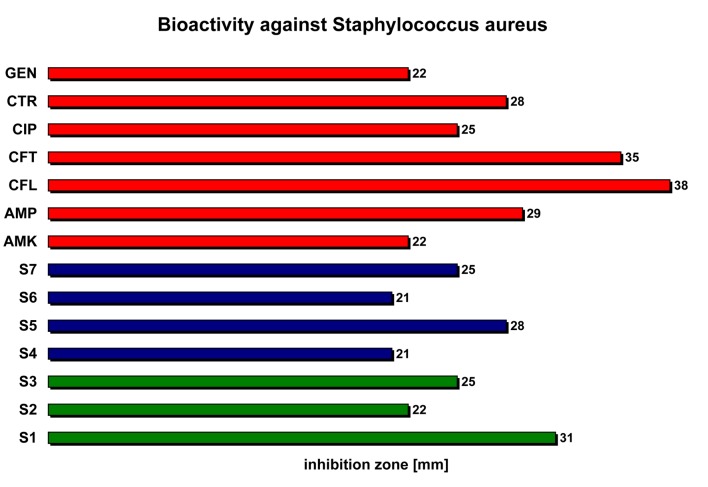
Staphylococcus aureus (Micrococcacea) proved o be extremly sensitive to the studied volatile oils, their antibacterial activity being comparable with the most sensitive reference antibiotic (cefalexin - CFL) (31 cm for S1 versus 38 cm for CFL) (Figure 6).
Figure 6.
The bioactivity of the studied volatile oils relative to reference antibacterial substances against Staphylococcus aureus.
No significant differences between the antibacterial activities of the cultivated and commercialy available oils were observed. The standard deviation values for the inhibition zones of the studied volatile oils were below ± 5%, while for the reference antibacterial substance the same values were below ± 3%.
The development of the fungi Candida albicans (Ascomycete) was completely inhibited by the volatile oils in 48 hours after inoculation.
The activity of the oils would be expected to relate to the respective composition of the plant volatile oils, the structural configuration of the constituent components of the volatile oils and their functional groups and possible synergistic interactions between components. Considering these aspects, we also tried to establish correlations between the chemical composition of the volatile oils and their bioactivity. Dorman and Deans, established a correlation of the antimicrobial activity of the compounds tested and their relative percentage composition in the plant volatile oils used in this study. The order of bioactivity depending on the diameter of the inhibition zone was: linalool (12.5 mmm) > geranyl acetate (9.4 mm) > nerol (8 mm) > geraniol (7 mm) > menthone (6,8 mm) > β-pinen (6.3 mm) > limonene (6.1 mm) > α-pinen (5.8 mm).5
We compared the percentage of these components in three of the active studied oil samples (S1, S5, S7) (Table 2).
Table 2. Concentration of bioactive constituents from the studied volatile oils.
| Oil bioactive constituent | S1 (%) | S5 (%) | S7 (%) |
| linalool | 3.57 | 5.89 | 9.18 |
| geraniol acetate | - | 1.15 | 0.76 |
| geraniol | 7.43 | 17.34 | 9.52 |
| menthone | 0.47 | 3.55 | 1.17 |
| α-pinen | - | 0.69 | - |
| bioactivity | 1 | 2 | 3 |
Even if the sample S1 obtained through dystillation contains the lowest concentration of components considered to be bioactive, its antimicrobial activity is comparable with the commercialy available S5 and S7, probably because the commercialy available preparation are “enriched” with syntethic components, which significantly influence the oil propeties.4,5,7
Conclusion
The searching of new, natural, antimicrobial and antifungal sources, without negative impact on the environment, as alternatives to those obtained by chemical synthesis is a permanent necessity and also a challenge.
All the bacterial strains demonstrated some degree of sensitivity to the plant volatile oils tested, although differences in the growth of a number of bacteria is noticeable when specific volatile oils are used.
Further studies based on the dilution method is needed in order to establish the minimal concentration in which the studied essential oils has an inhibitory effect on Staphylococcus aureus, Pseudomonas aeruginisa or Candida albicans respectively.
The biological properties manifested by Pelargonium essential oil in this study substantiate its use in numerous health problems and medical conditions and validates its commercial exploitation in many industry branches.
However further studies must be performed to confirm the safety of these oils for use as antimicrobial agents and natural preservatives in different products.
Conflict of Interest
There was no conflict of interest in this study.
References
- 1.Pepeljnjak S, Kalodera Z, Zovko M. Investigation of antimicrobial activity of Pelargonium radula (Cav.) L'Herit. Acta Pharm. 2005;55(4):409–15. [PubMed] [Google Scholar]
- 2.Balchin-Lis M, Hart SL, Deans SG, Eaglesham E. Potential agrochemical and medicinal usage of essential oils of Pelargonium species. J Herbs Spices Med Plants. 1995;3:11–22. [Google Scholar]
- 3.Mahdieh M, Yazdani M, Mahdieh S. The high potential of Pelargonium roseum plant for phytoremediation of heavy metals. Environ Monit Assess. 2013;185(9):7877–81. doi: 10.1007/s10661-013-3141-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Dabiri M, Sefidkon F, Yousefi M, Bashiribod S. Volatile components of Pelargonium roseum R. Br. J Essent Oil Bear Pl. 2011;14(1):114–7. [Google Scholar]
- 5.Dorman HJ, Deans SG. Antimicrobial agents from plants: antibacterial activity of plant volatile oils. J Appl Microbiol. 2000;88(2):308–16. doi: 10.1046/j.1365-2672.2000.00969.x. [DOI] [PubMed] [Google Scholar]
- 6.Dorman HJD, Surai P, Deans SG. In vitro antioxidant activity of a number of plant essential oils and phytoconstituents. J Essent Oil Res. 2000;12:241–8. [Google Scholar]
- 7.Ben Hsouna A, Hamdi N. Phytochemical composition and antimicrobial activities of the essential oils and organic extracts from Pelargonium graveolens growing in Tunisia. Lipids Health Dis. 2012;11:167. doi: 10.1186/1476-511X-11-167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Dzamic AM, Sokovic MD, Ristici MS, Grujic SM, Mileski KS, Marin PD. Chemical composition, antifungal and antioxidant activity of Pelargonium graveolens essential oil. J Appl Pharm Sci. 2014;4(3):1–5. [Google Scholar]
- 9.Gâlea C, Csedö C, Máthè I. Analiza gaz-cromatografică a uleiurilor volatile de Pelargonium roseum. Revista de Medicină şi Farmacie. 2007;53:147–53. [Google Scholar]
- 10.Verma RS, Verma RK, Yadav AK, Chauban A. Changes in the essential oil composition of rose-scented geranium (Pelargonium graveolens L’Herit. ex Ait.) due to date of transplanting under hill conditions of Uttarakhand. Indian J Nat Prod Res. 2010;1(3):367–70. [Google Scholar]