Abstract
Primary open angle glaucoma (POAG) is a degenerative disease commonly associated with aging and elevated intraocular pressure (IOP). Higher resistance to aqueous humor (AH) outflow through the trabecular meshwork (TM) generates the elevated IOP in POAG; unfortunately the underlying molecular mechanisms responsible for elevated resistance are unknown. It is widely accepted, however, that differences between normal and POAG TM tissues are presumably a consequence of cellular dysfunction. Here, we investigated the autophagic function and response to chronic oxidative stress in TM cells isolated from glaucomatous and age-matched donor eyes. Glaucomatous TM cells showed elevated senescence-associated-beta-galactosidase (SA-β-Gal) and cellular lipofuscin, together with decreased steady-state levels of LC3B-II, decreased levels of pRPS6K-T389 and reduced proteolysis of long-live proteins. Moreover, the glaucomatous cultures failed to activate autophagy when exposed to hyperoxic conditions. These results strongly suggest mTORdependent dysregulation of the autophagic pathway in cells isolated from the glaucomatous TM. Such dysregulated autophagic capacity can have a detrimental impact in outflow pathway tissue, i.e mechanotransduction, and thus represent an important factor contributing to the progression of the disease.
Keywords: trabecular meshwork, autophagy, glaucoma, mTOR, lysosomal proteolysis, oxidative stress
INTRODUCTION
Glaucoma is a group of human disorders characterized by a progressive loss of retinal ganglion cells and irreversible vision loss. There are multiple types of glaucoma, depending on the etiology. Primary open angle glaucoma (POAG), the most common form, is a disease often coincident with aging and elevated intraocular pressure (IOP) resulting from excessive resistance to aqueous humor (AH) drainage through the trabecular meshwork (TM), the primary outflow tract. Although the exact molecular mechanisms or regulatory pathways responsible for the resistance to outflow in POAG have still not been elucidated, it is widely accepted that differences between normal and POAG TM tissues are a consequence of cellular dysfunction [1].
Our laboratory previously reported increased number of cells positively stained for senescence-associated- β-galactosidase (SA-β-Gal) activity in the glaucomatous TM tissue compared to age-matched control tissue [2]. Senescence-associated-β-galactosidase is an activity of the lysosomal β-galactosidase detected at pH 6 (instead of pH 4–5) found in senescent cells [3]. The nature of this abnormal SA-β-Gal activity, which is not exclusive but can be observed in conditions others than senescence [4], i.e. starvation, remains unknown. Exposure of cultured TM cells to chronic oxidative stress led to increased SA-β-Gal activity, which correlated with increased lysosomal content [5]. This increase in SA-β-Gal activity could be partially blocked in the presence of 3-methyladenine (3-MA), an inhibitor of autophagosome formation, indicating that activation of macroautophagy is required for the occurrence of SA-β-Gal [6].
Autophagy, which means “self-eating”, is a general term that refers to the catabolic process in which cellular components are degraded by the machinery of the lysosomes. There are three different types of autophagy in mammalian cells based on the delivery route of the cytoplasmic material to the lysosomal lumen. Among them, macroautophagy (referred to here as autophagy) is the most extensively studied. This particular type of autophagy is characterized by the formation of a double membrane-bound organelle, the autophagosome, which engulfs the material targeted for degradation. Autophagosomes then fuse with lysosomes to form autolysosomes, in which the cytoplasmic cargos are degraded by resident hydrolases [7].
Autophagy has a greater variety of physiological and pathophysiological roles than initially thought, playing important housekeeping and quality control functions that contribute to health and longevity. Basal or constitutive autophagy is responsible for the routine turnover of old or damaged organelles, such as peroxisomes and mitochondria, as well as degradation of long-lived or aggregate-prone proteins that are too large to be degraded by the proteasome. Autophagy also acts as a survival mechanism under conditions of stress or environmental changes, such as nutrient starvation, oxidative stress, and mechanical stress. In addition to maintaining cellular homeostasis, autophagy plays a role in innate and adaptive immunity, programmed cell death, development, as well as prevention of cancer, neurodegeneration and aging [8]. Defects in autophagy have been linked to the progressive deterioration occurring during aging [9], and in an increasing number of diseases, including ocular diseases [10].
The TM is a dynamic tissue continuously exposed to different types of stresses, such as oxidative stress, mechanical stress, shear stress, and phagocytic stress [11]. Although short-term exposure to these stresses is expected to elicit adaptive responses, long-term exposure may lead to permanent alterations in the tissue physiology and contribute to the pathologic increase in aqueous humor outflow resistance frequently associated with glaucoma. Very recently, our laboratory has shown that activation of autophagy in TM cells is an early event occurring after application of sustained biaxial stretch, as well as in perfused eyes subjected elevated IOP [12]. In different studies, our laboratory demonstrated the induction of autophagy in TM cells subjected to chronic oxidative stress [5,6], a condition associated with the pathogenesis of glaucoma. In these studies, however, concomitant with the activation of the autophagic pathway, we observed reduced lysosomal acidification and impaired proteolytic maturation of lysosomal enzymes, resulting in decreased autophagic flux.
The importance of the autophagic lysosomal pathway in outflow pathway physiology is supported by the observation that elevated IOP and POAG are some of the ocular manifestations described in patients with lysosomal storage disorders [13,14]. Also, of extreme relevance is the fact that ASB10, a novel candidate POAG gene, has been recently reported to participate in ubiquitin-mediated degradation pathways [15]. Very interestingly, autophagy participates in the cellular degradation of myocilin, a gene linked to juvenile- and adult- open angle glaucoma [16]. Despite these data, and the observed role of autophagy in response to stress and adaptation in TM cells, the only example in the literature showing the status of the autophagic lysosomal pathway in glaucomatous TM cells is that inferred from the SA-β-Gal staining. Here, we systematically compared the autophagic function of TM cells isolated from glaucomatous and age-matched donor eyes.
MATERIALS AND METHODS
Cell Culture
Primary cultures of human TM cells were prepared and maintained as previously described [17]. Briefly, the TM was dissected and digested with 2 mg/ml of collagenase for 1 h at 37°C. The digested tissue was placed in gelatin-coated 35 mm dishes and cultivated in low glucose Dulbecco's Modified Eagle Medium (DMEM) with l-glutamine and 110 mg/l sodium pyruvate, supplemented with 10% fetal bovine serum (FBS), 100 mM non-essential amino acids, 100 units/ml penicillin, 100 mg/ml streptomycin sulfate and 0.25 mg/ml amphotericin B; all the reagents were obtained from Invitrogen (Carlsbad, CA). Cells were maintained and propagated until passage three at 37 °C in a humidified air with 5% CO2 incubator. Primary cultures of human TM cells (hTM) were isolated from corneoscleral rims from non-glaucomatous donor eyes after corneal transplantation surgery at Duke University Eye Center. Primary cultures of glaucomatous TM cells (gTM) were prepared from cadaver eyes obtained less than 24 h post-mortem from donors with documented history of POAG [17]. Ages and glaucoma medications of eye donors are summarized in Table 1. The protocols involving the use of human tissue were consistent with the tenets of the Declaration of Helsinki. Cell lines were subcultivated 1:2 when confluent. Cells at passage four were used for all the experiments.
Table 1.
Summary of TM cells
| Cell Line | Donor Age (yr) |
Glaucoma Medications |
Glaucoma Surgery |
|---|---|---|---|
| hTM1 | 65 | None | No |
| hTM2 | 67 | None | No |
| hTM3 | 79 | None | No |
| gTM1 | 72 | Travatan | No |
| gTM2§ | 77 | Trusopt, Ocupress, Timoptic | No |
| gTM3§ | 81 | Timoptic, Xalatan | Argon laser |
[17]
Experimental Model of Chronic Oxidative Stress
Chronic oxidative stress was induced by subjecting the cells to normobaric hyperoxic conditions as described before [5]. Briefly, human TM cells were grown for two weeks at 40% O2 and 5% CO2 at confluency. Control cultures were grown under physiological oxygen conditions (5% O2, 5% CO2) in a triple-gas incubator (SANYO, MCO-5M).
Whole-cell Lysates Preparation
Cells were washed twice in PBS and lysated in RIPA buffer containing protease inhibitor cocktail (Thermo Scientific) and phosphatase inhibitor cocktail (Thermo Scientific). After sonication for one minute on ice, lysates were clarified by centrifugation. Cytoplasmic and nuclear lysates were obtained as follows. Cells were washed and incubated on ice for 15 minutes in Triton X-100 lysis buffer (50 mM Tris-HCl pH 7.5, 0.5% Triton X-100, 137.5 mM NaCl, 10% Glycerol, with protease and phosphatase inhibitors). Protein concentration was determined with a protein assay kit (Micro BCA, Thermo Scientific).
Western Blot Analysis
Protein samples (10 µg) were separated by 10% polyacrylamide SDS-PAGE gels (15% polyacrylamide for LC3 detection, 7% polyacrylamide for LAMP1 detection) and transferred to PVDF membranes (Bio-Rad). The membranes were blocked with 5% nonfat dry milk and incubated overnight with the specific primary antibodies (Table 2). The bands were detected by incubation with a secondary antibody conjugated to horseradish peroxidase and chemiluminescence substrate (ECL Plus, GE Healthcare). Blots were scanned and analyzed by densitometry using Image J.
Table 2.
List of primary antibodies used
| Target Protein | Company | Catalog Number | Dilution |
|---|---|---|---|
| Atg3 | Cell Signaling | 3415P | 1:1000 |
| Atg4 | Santa Cruz Biotechnology | SC-131433 | 1:1000 |
| Atg5 | Cell Signaling | 8540P | 1:1000 |
| Atg7 | Cell Signaling | 2631P | 1:1000 |
| Atg12 | Cell Signaling | 4180P | 1:1000 |
| Beclin | Cell Signaling | 3495P | 1:1000 |
| p62 | Sigma-Aldrich | P0067 | 1:1000 |
| LAMP1 | Abcam | Ab24170 | 1:1000 |
| CTSD | Santa Cruz Biotechnology | SC-6494 | 1:1000 |
| CTSB | Abcam | ab58802 | 1:1000 |
| LC3B | Cell Signaling | 3868S | 1:1000 |
| pRPS6KB(T421/S424) | Santa Cruz Biotechnology | SC-7984 | 1:1000 |
| pRPS6KB(T389) | Cell Signaling | 9205 | 1:1000 |
| RPS6KB | Santa Cruz Biotechnology | SC-230 | 1:1000 |
| Actin | Santa Cruz Biotechnology | sc-69879 | 1:2000 |
Measurement of Endogenous Cellular Lipofuscin Autofluorescence
Endogenous lipofuscin autofluorescence was detected under the FITC filter by fluorescence microscopy and quantified by flow cytometry (FACSCalibur; BD Biosciences). For this, cells were trypsinized, washed in PBS, and resuspended in 100 µL PBS. The fluorescence emitted by 10,000 cells in the emission wavelength of the lipofuscin (yellow-orange spectrum, FL-2 channel, 563–607 nm wavelength band) when excited with the 488 nm laser was recorded and analyzed (CellQuest software; BD Biosciences).
Quantification of Lysosomal Cellular Content
The lysosomal cellular content was evaluated using the lysosomotropic dye Lysotracker Red (LTR, Invitrogen) following the manufacturer’s instructions. Briefly, cells were incubated for 15 minutes at 37°C in fresh culture medium containing LTR (500 nM). Specific lysosomal labeling was confirmed by fluorescence microscopy. Cells were then washed, trypsinized and resuspended in 100 µL PBS. LTR fluorescence was quantified by flow cytometry. The fluorescence emitted by 10,000 cells in the FL-3 channel (red spectrum) when excited with the 488 nm laser was recorded and analyzed (CellQuest software; BD Biosciences).
Flow Cytometry Determination of SA-β-Gal Activity
Quantification of SA-β-Gal activity was performed by flow cytometry with the fluorogenic substrate C12FDG (Invitrogen). First, alkalinization of the lysosomal compartment was induced by treating cell monolayers with 300 µM chloroquine for 1 hour at 37°C under 5% CO2. The cells (1 × 106) were then trypsinized and incubated for 1 minute at 37°C in 50 µL of pre-warmed PBS containing C12FDG (33 µM). C12FDG uptake was stopped by adding 500 µL ice-cold PBS. The mean green fluorescence of 10,000 cells was recorded (FL-1 channel) 30 minutes later and quantified (CellQuest software; BD Biosciences). Nonstained control cells were included, to evaluate the baseline fluorescence. The laser intensity settings were adjusted to the lowest level so as not to detect autofluorescence.
Proteolysis Assay
Cells were labeled with 2 µCi/mL [3H]Leucine (MP Biomedicals) for 48 hours to maximize labeling of long-lived proteins. At the end of the labeling period, cells were washed five times with PBS and incubated with chase medium containing 50 times the molar concentration of leucine for two hours to chase-out short-lived proteins. Chase medium was then replaced, and cells were incubated with fresh chase medium for 16 hours. Culture media containing free [3H]Leucine released upon intracellular degradation was collected, clarified by centrifugation at 500 g × 5 min and precipitated overnight at 4° C in a final concentration of 10% trichloroacetic acid. Cells were washed and lysed in solubilization buffer (0.1 N NaOH, 0.1% sodium deoxycholate) at 37° C for 2 hours. Radioactivity was counted in the acid-soluble fraction and in the solubilized cells. Proteolysis was calculated by dividing the soluble radioactivity count per minute (cpm) by the total cellular radioactivity cpm.
Statistical Analyses
All experimental procedures were conducted with the three nonglaucomatous and glaucomatous primary cultures listed in Table 1. The percentage of increase of the experimental conditions compared with the control was calculated and averaged. Data are represented as the mean ± SD and were analyzed with unpaired or paired Student’s t-test. P < 5% was considered statistically significant.
RESULTS
SA-β-Gal, Lipofuscin, and Lysosomal Content in Glaucomatous and Control Cultured HTM Cells
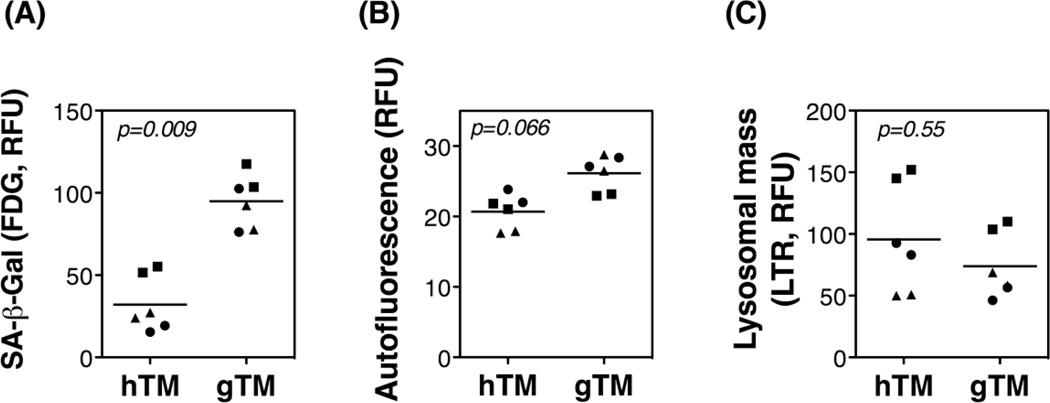
Primary gTM cells displayed a significant increase in SA-β-Gal activity compared to primary hTM cells (Figure 1A, 95.03 ± 7.89 vs. 32.14 ± 10.92 RFU, p=0.009). The levels of cellular autofluorescence, which represents lipofuscin or intralysosomal non degradable material, were also higher in glaucomatous cultures (Figure 1B, 26.15 ± 1.548 vs. 20.72 ± 1.522 RFU, p=0.066). Although not statistically significant, a trend towards diminished lysosomal mass was observed in glaucomatous cells compared to control cells (Figure 1C, 74.01 ± 16.84 vs. 95.62 ± 28.58 RFU, p=0.55).
Fig. 1.
SA-β-Gal, lipofuscin, and lysosomal content in normal and glaucomatous human TM cells: Primary cultures of TM cells isolated from three normal (hTM) and three glaucomatous (gTM) donor eye tissues were taken to passage four and then grown to confluence; SA-β-Gal activity (A), lipofuscin content (B), and lysosomal mass (C) were quantified by flow cytometry at day five post-confluence. Experiments were performed in duplicate, n=6, t-test.
Steady State Expression Levels of Autophagy and Lysosomal Proteins in Glaucomatous and Control Cultured TM Cells
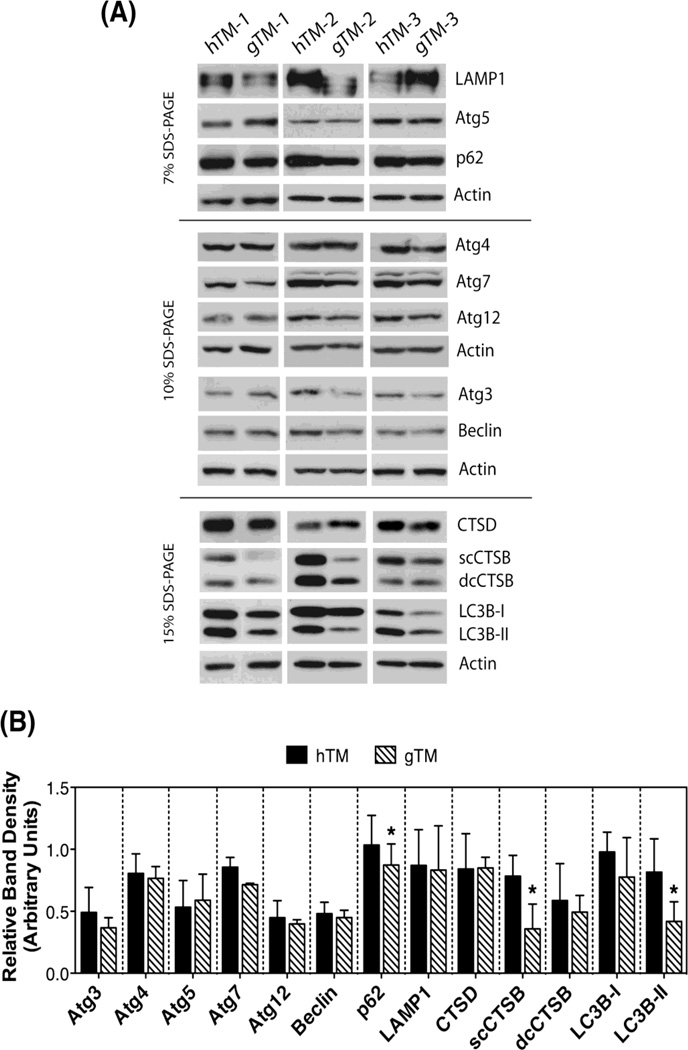
A connection between autophagy and SA-β-Gal activity has been suggested in several studies [4,18–22], including our previous work [5,6]. To assess a potential difference in the autophagy lysosomal pathway between gTM and hTM, we evaluated by Western-blot the steady state expression levels of a number of autophagy (Atg3, Atg4, Atg5, Atg7, Atg12, Beclin, LC3B) and lysosomal proteins (LAMP1, CTSD, CTSB), as well as that of p62, a scaffold protein that targets ubiquitinated proteins for autophagic degradation. Blots and densitometry quantification of the protein bands are shown in Figure 2A and 2B, respectively. Despite the expected variability among the samples, all gTM primary cultures demonstrated a significant decrease in p62 (0.873 ± 0.171 vs. 1.035 ± 0.238 AU, p=0.05), single chain CTSB (0.359 ± 0.199 vs. 0.783 ± 0.167 AU, p= 0.04), and LC3B-II (0.418 ± 0.358 vs. 0.816 ± 0.468 AU, p=0.032) protein levels. A decrease in Atg7 was also observed (0.686 ± 0.29 vs. 0.89 ± 0.37 AU, p=0.08). LAMP1, a lysosomal membrane protein, was found to be downregulated in two of the gTM cultures, but highly expressed in the third one.
Fig. 2.
Expression levels of autophagy and lysosomal proteins in hTM and gTM. (A) Steady state expression levels of autophagy and lysosomal proteins in hTM and gTM cells at passage four grown to confluence for five days, evaluated by WB. (B) Normalized relative protein levels calculated from densitometric analysis of the blots. Data are the means ± SD, n=3, * p<0.05, t-test.
Mechanistic Target of Rapamycin (mTOR) Pathway
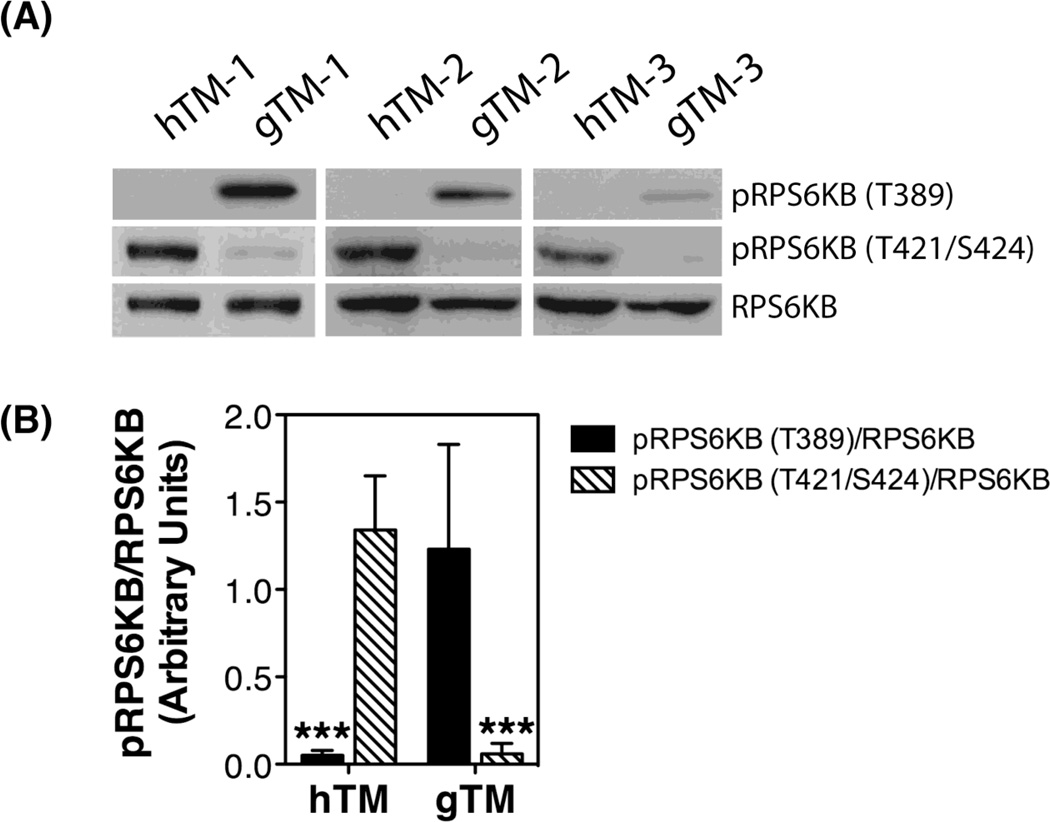
The kinase TOR is a critical regulator of autophagy induction, with activated mTOR suppressing, and negative regulation of mTOR promoting autophagy. We investigated the status of mTOR pathway by monitoring the phosphorylated levels of its downstream target p70S6 Kinase (RPS6KB/p70S6K). Two different rapamycin-sensitive phosphorylation sites were analyzed (the mTOR directly phosphorylated site Thr389, and the Thr421/Ser424 at the pseudosubstrate region). As observed in Figure 3A and quantified in Figure 3B, all gTM cultures showed a significant increase in the relative protein levels of pRPS6KB-T389 compared to hTM (1.23 ± 0.6 vs. 0.05 ± 0.3, p<0.0001). In contrast, the levels of pRPS6KB-T421/S424 were significantly reduced in the glaucomatous cells (0.06 ± 0.6 vs. 1.43 ± 0.31, p<0.0001)
Fig. 3.
mTOR pathway in hTM and gTM cells. (A) Protein levels of phosphorylated and non-phosphorylated downstream target of MTOR, RPS6KB. (B) Normalized relative protein levels of pRPS6KB calculated from densitometric analysis of the blots. Data are the means ± SD, n = 3, *** p< 0.001, t-test.
Proteolysis of Long-lived and Total Protein Synthesis
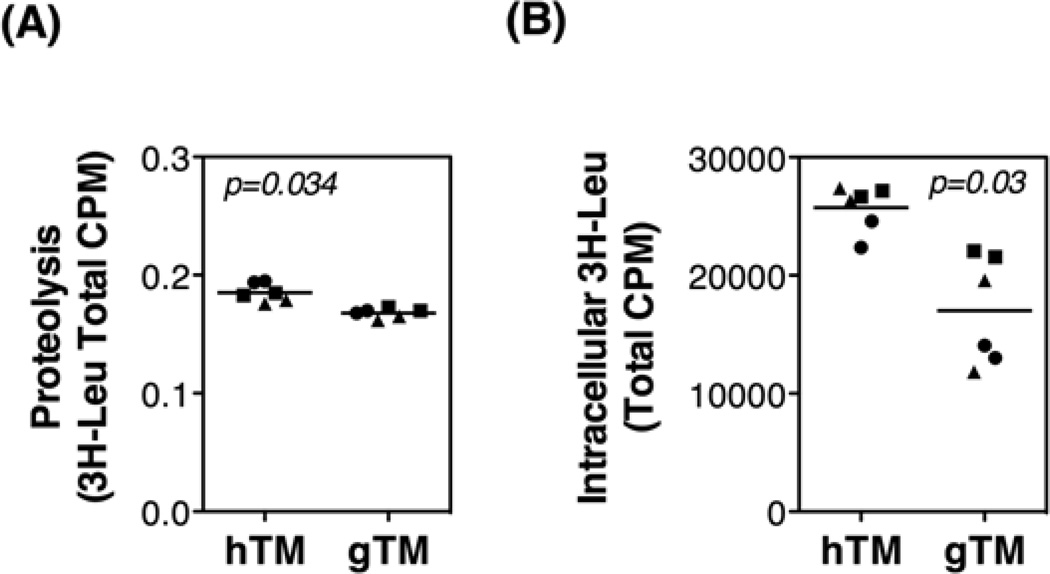
Proteolysis of long-lived proteins in confluent cultures of hTM cells and gTM cells was measured using a radiolabeled amino acid-based pulse chase assay. As shown in Figure 4A, gTM cultures showed a small but significant ~ 10% decrease in the autophagic degradation of long lived proteins, compared to control cultures (0.168 ± 0.002 vs 0.185 ± 0.005 cpm, p=0.034). Moreover, the amount of [3H]-Leu in the intracellular fraction, which correlates with total protein synthesis, was significantly reduced in gTM cells (Figure 4B, 17019 ± 2476 vs. 6420.7 ± 1280.5 cpm, p = 0.032).
Fig. 4.
Proteolysis of long-lived proteins and total protein synthesis in hTM and gTM cells. Primary cultures of hTM and gTM cells at passage four were grown to confluence for five days. (A) Cellular proteolysis as calculated by dividing the soluble radioactivity (cpm) found in the culture media by the total cellular radioactivity (cpm). (B) Total protein synthesis calculated by the amount of [3H]-Leu in the intracellular fraction. Experiments were performed in duplicate, n=3, t-test.
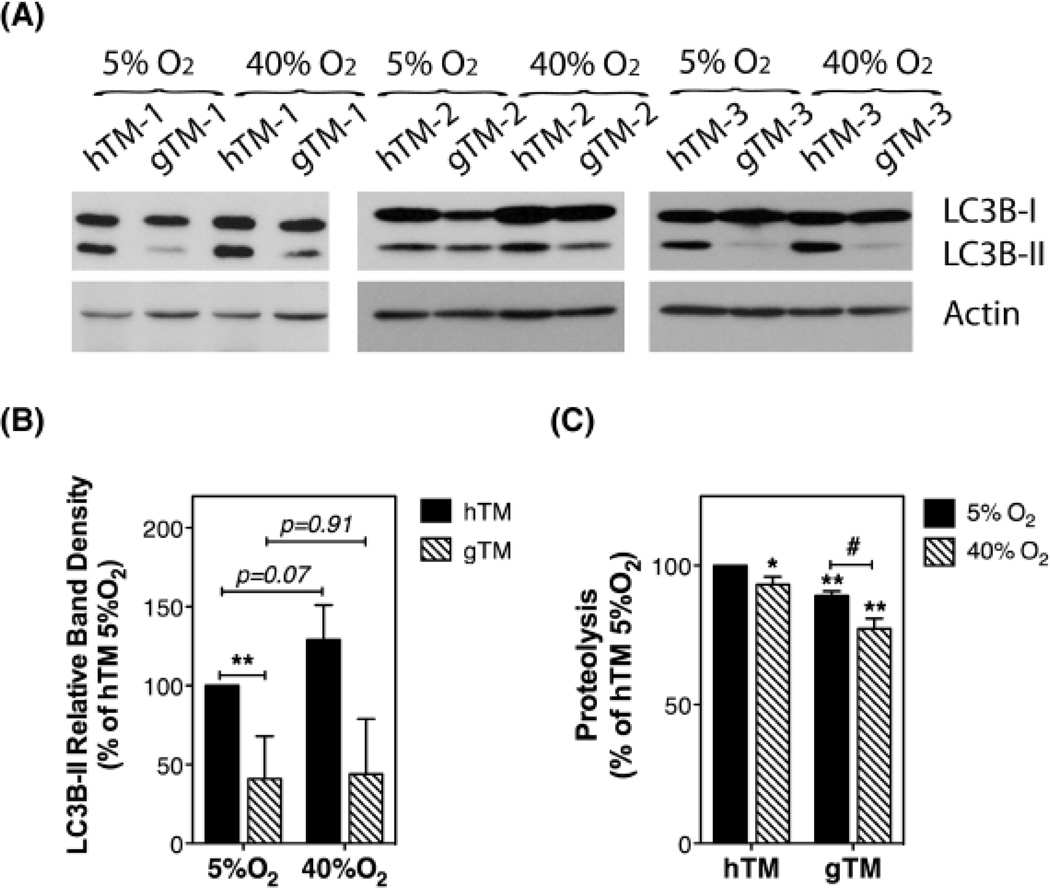
Autophagy and Oxidatively Stressed hTM and gTM Cells
Our laboratory previously reported the induction of autophagy in porcine TM cells exposed to chronic oxidative stress. Concomitant with the activation of the autophagic pathway, TM cells grown under oxidative stress conditions showed reduced autophagic flux and lysosomal proteolysis [5,6]. We wanted to investigate the autophagic response of gTM cells when subjected to chronic oxidative stress. For this, confluent cultures of hTM cells and gTM cells were grown for 10 days under hyperoxia (40% O2); controls were kept under physiological 5% O2 concentrations, an experimental model previously validated by our laboratory for application of chronic oxidative stress in post-mitotic TM cells [5]. LC3B-II levels and proteolysis of long-lived proteins were evaluated. While all hTM cultures demonstrated elevated LC3B-II levels when grown under 40% O2 conditions, a significant increase in LC3B-II in response to an oxidative environment was observed in just one out of the three gTM cultures tested (Figure 5A and B). In agreement with our previous study using porcine TM cells, proteolysis of long-lived proteins was significantly reduced in oxidatively stressed cultures when compared to non-stressed ones (Figure 5C). Such decrease in proteolysis with oxidative stress was more pronounced in gTM than in hTM cells.
Fig. 5.
Autophagy in oxidatively stressed hTM and gTM cells. Confluent cultures of hTM and gTM cells at passage four were grown for 10 days either at physiological 5% O2 concentrations or under hyperoxia (40% O2). (A) Protein expression levels of LC3B-I and LC3B-II. (B) Normalized relative protein levels calculated from densitometric analysis of the blots. (C) Cellular proteolysis as calculated by dividing the soluble radioactivity cpm found in the culture media by the total cellular radioactivity cpm. Data are the means ± SD, n=3, *,# p<0.05, ** p<0.01, t-test.
DISCUSSION
In this manuscript we have characterized for the first time the autophagic function of glaucomatous TM cells. Our data indicate dysregulation of the autophagic pathway and autophagic response to oxidative stress in TM cells isolated from the glaucomatous outflow pathway.
The study of autophagy in glaucomatous human cadaver tissue is subjected to important limitations and challenges. First, the supply of human cadaver eyes for research purposes has been and it is gradually declining over the last few years [23]; second, access to complete clinical histories can be challenging; third and most importantly, all glaucoma patients undergo therapy with glaucoma medications, usually a combination of different drugs, and/or might have undergone surgery, either laser or trabeculectomy. So far, there is no information of the effects of these therapeutic treatments in the autophagic lysosomal pathway, which might definitively affect the outcome. To overcome some of these limitations, we have opted for first characterizing the autophagic lysosomal function in glaucomatous TM cells as a starting point. We acknowledge that glaucomatous cells in vitro likely behave different than those in vivo conditions, but so do control cultures. The fact that results are observed across glaucomatous cell strains from 3 donors with glaucoma and 3 donors that were ostensibly non-glaucomatous, supports and strengths the significance of our results.
Very strikingly, we found that glaucomatous TM cells conserve in vitro the increase in SA-β-Gal activity we previously reported in situ in the glaucomatous outflow pathway [2]. Although the average age in the glaucoma group (76.67 ± 4.51 y.o) is slightly, but not significant, higher than in the control group (70.33 ± 7.57 y.o), we did not find a correlation with aging when looked at data individually, which might account for such a difference. SA-β-Gal activity is encoded by the lysosomal β-galactosidase [24], but the molecular mechanisms underlying its expression and its functional relevance in senescence and other non-senescent conditions (i.e low serum level or high confluency) are not fully understood. SA-β-Gal has been proposed to be a surrogate marker for the increase in lysosomal mass occurring during aging [25]. However, the lower levels of the lysosomal membrane protein LAMP1 found in two out of the three glaucomatous TM cultures, together with the observed trend towards decreased lysosomal mass in gTM cells, suggests that although both phenomena might occur simultaneously, they are not strictly dependent.
Impairment of autophagy has been shown to induce SA-β-Gal in primary human fibroblasts [19]. Very interestingly, all glaucomatous TM cells display lower steady-state levels of the autophagosome marker LC3B-II compared to their respective controls. Because of limited cell availability, we could not perform a proper flux assay to assess whether such decreased in the steady-state levels of LC3B-II was a result of impaired autophagic activation or reduced autophagic flux [26,27]. Intriguingly, our data pointed in both directions. On the one hand, along with the decreased in LC3B-II, glaucomatous TM cells demonstrated increased presence of intralysosomal non-degradable material, reduced proteolysis of long-lived proteins, and lower lysosomal content, altogether suggesting a decrease in autophagic flux. On the other hand, gTM cultures also showed significant lower amounts of p62. Protein levels of p62 have been used as a readout for autophagic degradation; however, as recently indicated in [27], there is not always a clear correlation between LC3B-II and p62.
The best well-known pathway regulating autophagy is mTOR signaling, which inversely regulates autophagic flux and protein synthesis [18,28,29]. mTOR activity is commonly monitored following the phosphorylation of its downstream substrate RPS6KB [27]. Very interestingly, gTM cells showed a dramatic increase in the levels of RPS6KB phosphorylated at Thr389 (pRPS6KB-T389), a direct mTOR phosphorylation site, indicating constitutive inhibition of autophagy in glaucomatous TM cells. Intriguingly, phosphorylation of RPS6KB at the other rapamycin-sensitive phosphorylation site examined, Thr421/Ser424, was significantly reduced in the glaucomatous cultures. Phosphorylation of these residues is believed to activate the kinase, raising the possibility that other signaling pathway participating in fully activation of RPS6KB are dysregulated in glaucoma. A major downside outcome of RPS6KB inhibition is diminished protein synthesis [30]. In agreement, gTM cells showed an almost 50% reduction in intracellular incorporation of 3H-Leu into nascent proteins.
The glaucomatous TM tissue is characterized by the increased expression of oxidative markers (oxidative DNA damage and peroxidized lipids) as well as diminished overall antioxidant potential [31,32,33]. Although the molecular mechanisms are not clear yet, oxidative stress is believed to play a major role in the pathogenesis of POAG [34,35], likely as a result of the particular higher sensitivity of the TM to oxidative radicals compared with other tissues in the anterior chamber of the eye [36]. In contrast to control cells, gTM cells failed to activate autophagy in response to chronic oxidative stress. Moreover, exposure to a hyperoxic environment further decreased lysosomal proteolysis in the glaucomatous cultures, supporting a key role of autophagy in the TM cellular response to oxidative stress.
Interestingly, a recent work by Pulliero et al. has reported a parallel increase in oxidative damage and increase LC3-II/LC3-I protein levels in TM tissue with aging [37]. These results do not contradict, rather complement our findings here. In cultured conditions, oxidative stress is known to induce autophagy in TM cells, as an early protective mechanism, followed by lysosomal basification and a decline in autophagic function [6]. Biphasic changes in autophagy are not unusual and are considered to contribute to disease progression in other tissues [38]. Similar to our findings here, the oxidatively stressed cultures also demonstrated increased SA-β-Gal, which seemed to be, at least, partly mediated by activation of autophagy and associated with decreased autophagic flux [6]. It is, therefore, possible that mechanisms related to those observed in oxidatively stressed TM cells lead to defective autophagic degradation in glaucoma disease.
In conclusion, the data presented here strongly support mTOR-dependent dysregulation of autophagy in glaucomatous TM cells. As discussed in [39], such dysregulated autophagic capacity can have a detrimental impact in outflow pathway tissue functionality,, i.e. its ability to cope and adapt to mechanical forces, including those triggered by high pressure [12], and represent an important factor contributing to the progressive failure of cellular TM function with age and contribute to the pathogenesis of ocular hypertension and POAG.
HIGHLIGHTS.
Glaucomatous TM cells display elevated SA-β-Gal activity and lipofuscin levels.
Autophagy is dysregulated in glaucomatous TM cells.
Glaucomatous TM cells fail to activate autophagy in response to chronic oxidative stress.
Potential role of impaired autophagy in the pathogenesis of POAG.
ACKNOWLEDGEMENTS
This work was supported by the National Institute of Health Grants R01EY020491 (Liton) and P30EY005722; the Brightfocus Foundation (Liton, G2012022), the Alcon Foundation (Liton, Young Investigator Grant), and by an unrestricted grant to Duke Eye Center from the Research to Prevent Blindness.
ABBREVIATIONS
- POAG
primary open angle glaucoma
- IOP
intraocular pressure
- AH
aqueous humor
- TM
trabecular meshwork
- SA-β-Gal
senescence-associated beta galactosidase
- 3-MA
3-methyladenine
- LTR
lysotracker red
- cpm
counts per minute
- Leu
leucine
- RFU
relative fluorescence units
- Atg
autophagy genes
- CTSB
cathepsin B
- CTSD
cathepsin D
- scCTSB
single chain cathepsin B
- mTOR
mechanistic target of rapamycin
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
REFERENCES
- 1.Stamer WD, Acott TS. Current understanding of conventional outflow dysfunction in glaucoma. Curr Opin Ophthalmol. 2012;23:135–143. doi: 10.1097/ICU.0b013e32834ff23e. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Liton PB, Challa P, Stinnett S, Luna C, Epstein DL, Gonzalez P. Cellular senescence in the glaucomatous outflow pathway. Exp Gerontol. 2005;40:745–748. doi: 10.1016/j.exger.2005.06.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Dimri GP, Lee X, Basile G, Acosta M, Scott G, Roskelley C, et al. A biomarker that identifies senescent human cells in culture and in aging skin in vivo. Proc Natl Acad Sci USA. 1995;92:9363–9367. doi: 10.1073/pnas.92.20.9363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Yang N-C, Hu M-L. The limitations and validities of senescence associated-beta-galactosidase activity as an aging marker for human foreskin fibroblast Hs68 cells. Exp Gerontol. 2005;40:813–819. doi: 10.1016/j.exger.2005.07.011. [DOI] [PubMed] [Google Scholar]
- 5.Liton PB, Lin Y, Luna C, Li G, Gonzalez P, Epstein DL. Cultured porcine trabecular meshwork cells display altered lysosomal function when subjected to chronic oxidative stress. Invest Ophthalmol Vis Sci. 2008;49:3961–3969. doi: 10.1167/iovs.08-1915. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Porter K, Nallathambi J, Lin Y, Liton PB. Lysosomal basification and decreased autophagic flux in oxidatively stressed trabecular meshwork cells: implications for glaucoma pathogenesis. Autophagy. 2013;9:581–594. doi: 10.4161/auto.23568. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Mizushima N. Autophagy: process and function. Genes Dev. 2007;21:2861–2873. doi: 10.1101/gad.1599207. [DOI] [PubMed] [Google Scholar]
- 8.Boya P, Reggiori F, Codogno P. Emerging regulation and functions of autophagy. Nat Cell Biol. 2013;15:713–720. doi: 10.1038/ncb2788. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Levine B, Kroemer G. Autophagy in the pathogenesis of disease. Cell. 2008;132:27–42. doi: 10.1016/j.cell.2007.12.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Frost LS, Mitchell CH, Boesze-Battaglia K. Autophagy in the eye: implications for ocular cell health. Exp Eye Res. 2014;124:56–66. doi: 10.1016/j.exer.2014.04.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Liton PB, Gonzalez P. Stress response of the trabecular meshwork. J Glaucoma. 2008;17:378–385. doi: 10.1097/IJG.0b013e31815f52a8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Porter KM, Jeyabalan N, Liton PB. MTOR-independent induction of autophagy in trabecular meshwork cells subjected to biaxial stretch. Biochim Biophys Acta. 2014;1843:1054–1062. doi: 10.1016/j.bbamcr.2014.02.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Spellacy E, Bankes JL, Crow J, Dourmashkin R, Shah D, Watts RW. Glaucoma in a case of Hurler disease. Br J Ophthalmol. 1980;64:773–778. doi: 10.1136/bjo.64.10.773. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Biswas J, Nandi K, Sridharan S, Ranjan P. Ocular manifestation of storage diseases. Curr Opin Ophthalmol. 2008;19:507–511. doi: 10.1097/ICU.0b013e32831215c3. [DOI] [PubMed] [Google Scholar]
- 15.Keller KE, Yang Y-F, Sun YY, Sykes R, Acott TS, Wirtz MK. Ankyrin repeat and suppressor of cytokine signaling box containing protein-10 is associated with ubiquitin-mediated degradation pathways in trabecular meshwork cells. Mol Vis. 2013;19:1639–1655. [PMC free article] [PubMed] [Google Scholar]
- 16.Qiu Y, Shen X, Shyam R, Yue BYJT, Ying H. Cellular processing of myocilin. PLoS ONE. 2014;9 doi: 10.1371/journal.pone.0092845. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Stamer DW, Roberts BC, Epstein DL, Allingham RR. Isolation of primary open-angle glaucomatous trabecular meshwork cells from whole eye tissue. Curr Eye Res. 2000;20:347–350. [PubMed] [Google Scholar]
- 18.Narita M, Young ARJ, Arakawa S, Samarajiwa SA, Nakashima T, Yoshida S, et al. Spatial coupling of mTOR and autophagy augments secretory phenotypes. Science. 2011;332:966–970. doi: 10.1126/science.1205407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Kang HT, Lee KB, Kim SY, Choi HR, Park SC. Autophagy impairment induces premature senescence in primary human fibroblasts. PLoS ONE. 2011;6:e23367. doi: 10.1371/journal.pone.0023367. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Young AR, Narita M. Connecting autophagy to senescence in pathophysiology. Curr Opin Cell Biol. 2010;22:234–240. doi: 10.1016/j.ceb.2009.12.005. [DOI] [PubMed] [Google Scholar]
- 21.Gerland L-M, Peyrol S, Lallemand C, Branche R, Magaud J-P, Ffrench M. Association of increased autophagic inclusions labeled for beta-galactosidase with fibroblastic aging. Exp Gerontol. 2003;38:887–895. doi: 10.1016/s0531-5565(03)00132-3. [DOI] [PubMed] [Google Scholar]
- 22.Sasaki M, Miyakoshi M, Sato Y, Nakanuma Y. Autophagy mediates the process of cellular senescence characterizing bile duct damages in primary biliary cirrhosis. Lab Invest. 2010;90:835–843. doi: 10.1038/labinvest.2010.56. [DOI] [PubMed] [Google Scholar]
- 23.Curcio CA. Research Tissue Acquisition Working Group, Declining availability of human eye tissues for research. Invest Ophthalmol Vis Sci. 2006;47:2747–2749. doi: 10.1167/iovs.05-0978. [DOI] [PubMed] [Google Scholar]
- 24.Lee BY, Han JA, Im JS, Morrone A, Johung K, Goodwin EC, et al. Senescence-associated beta-galactosidase is lysosomal beta-galactosidase. Aging Cell. 2006;5:187–195. doi: 10.1111/j.1474-9726.2006.00199.x. [DOI] [PubMed] [Google Scholar]
- 25.Kurz DJ, Decary S, Hong Y, Erusalimsky JD. Senescence-associated (beta)-galactosidase reflects an increase in lysosomal mass during replicative ageing of human endothelial cells. J Cell Sci. 2000;113(Pt 20):3613–3622. doi: 10.1242/jcs.113.20.3613. [DOI] [PubMed] [Google Scholar]
- 26.Mizushima N, Yoshimori T. How to interpret LC3 immunoblotting. Autophagy. 2007;3:542–545. doi: 10.4161/auto.4600. [DOI] [PubMed] [Google Scholar]
- 27.Klionsky DJ, Abdalla FC, Abeliovich H, Abraham RT, Acevedo-Arozena A, Adeli K, et al. Guidelines for the use and interpretation of assays for monitoring autophagy. Autophagy. 2012;8:445–544. doi: 10.4161/auto.19496. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Poüs C, Codogno P. Lysosome positioning coordinates mTORC1 activity and autophagy. Nat Cell Biol. 2011;13:342–344. doi: 10.1038/ncb0411-342. [DOI] [PubMed] [Google Scholar]
- 29.Korolchuk VI, Saiki S, Lichtenberg M, Siddiqi FH, Roberts EA, Imarisio S, et al. Lysosomal positioning coordinates cellular nutrient responses. Nat Cell Biol. 2011;13:453–460. doi: 10.1038/ncb2204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Weichhart T. Mammalian target of rapamycin: a signaling kinase for every aspect of cellular life. Methods Mol Biol. 2012;821:1–14. doi: 10.1007/978-1-61779-430-8_1. [DOI] [PubMed] [Google Scholar]
- 31.Saccà SC, Pascotto A, Camicione P, Capris P, Izzotti A. Oxidative DNA damage in the human trabecular meshwork: clinical correlation in patients with primary open-angle glaucoma. Arch Ophthalmol. 2005;123:458–463. doi: 10.1001/archopht.123.4.458. [DOI] [PubMed] [Google Scholar]
- 32.Izzotti A, Saccà SC, Cartiglia C, De Flora S. Oxidative deoxyribonucleic acid damage in the eyes of glaucoma patients. Am J Med. 2003;114:638–646. doi: 10.1016/s0002-9343(03)00114-1. [DOI] [PubMed] [Google Scholar]
- 33.De La Paz MA, Epstein DL. Effect of age on superoxide dismutase activity of human trabecular meshwork. Invest Ophthalmol Vis Sci. 1996;37:1849–1853. [PubMed] [Google Scholar]
- 34.Zanon-Moreno V, Marco-Ventura P, Lleo-Perez A, Pons-Vazquez S, Garcia-Medina JJ, Vinuesa-Silva I, et al. Oxidative stress in primary open-angle glaucoma. J Glaucoma. 2008;17:263–268. doi: 10.1097/IJG.0b013e31815c3a7f. [DOI] [PubMed] [Google Scholar]
- 35.Kumar DM, Agarwal N. Oxidative stress in glaucoma: a burden of evidence. J Glaucoma. 2007;16:334–343. doi: 10.1097/01.ijg.0000243480.67532.1b. [DOI] [PubMed] [Google Scholar]
- 36.Izzotti A, Sacca S, Longobardi M, Cartiglia C. Sensitivity of ocular anterior-chamber tissues to oxidative damage and its relevance to glaucoma pathogenesis. Invest Ophthalmol Vis Sci. 2009 doi: 10.1167/iovs.09-3871. [DOI] [PubMed] [Google Scholar]
- 37.Pulliero A, Seydel A, Camoirano A, Saccà SC, Sandri M, Izzotti A. Oxidative damage and autophagy in the human trabecular meshwork as related with ageing. PLoS ONE. 2014;9:e98106. doi: 10.1371/journal.pone.0098106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Schneider JL, Cuervo AM. Autophagy and human disease: emerging themes. Curr. Opin. Genet. Dev. 2014;26C:16–23. doi: 10.1016/j.gde.2014.04.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Liton PB, Lin Y, Gonzalez P, Epstein DL. Potential role of lysosomal dysfunction in the pathogenesis of primary open angle glaucoma. Autophagy. 2009;5:122–124. doi: 10.4161/auto.5.1.7304. [DOI] [PMC free article] [PubMed] [Google Scholar]