Abstract
Purpose
In the present study booster efficacies of Ag85 B, Bacillus Calmette-Guerin (BCG), and Ag85B peptides were evaluated using prime boost regimes in BALB/c mice.
Materials and Methods
Mice were primed with BCG vaccine and subsequently boosted with Ag85B, BCG and cocktail of Ag85B peptides.
Results
Based on analysis of immune response it was observed mice boosted with Ag85B peptides showed significant (p < 0.001) cytokines levels (interferon γ, interleukin 12) and BCG specific antibodies (anti-BCG and anti-purified protein derivative titre) compared to booster dose of BCG, Ag85B and BCG alone.
Conclusion
Our pilot results suggest that prime boost regimes with Ag85B peptides can boost waning BCG induced immunity and may improve immunogenicity of BCG vaccine. However, lot of work is further needed using experimental model of tuberculosis infection to justify the result.
Keywords: BCG vaccine, Tuberculosis, antigen 85B, antigen 85B peptide
Introduction
Bacillus Calmette-Guerin (BCG) is the only vaccine available for mass immunization against tuberculosis (TB) with more than three billion doses administered worldwide [1,2]. BCG vaccine generally promote high levels of immunity against the childhood manifestations of TB; however, field trials have shown that the incidence of TB increases at later stage after vaccination. This waning of BCG protection correlates with an increased number of TB cases in adolescence [3]. The reasons for the failure of BCG have been widely debated and have remained a topic of active research. During past 15-20 years, number of vaccine candidates have been evaluated with some advancing into clinical trials and preclinical development. Current failure of subunit vaccines based on antigen 85B (Ag85B) of Mycobacterium tuberculosis (MTB) to replace/boost BCG induced immunity [4] in adults suggests the need for identification of alternative approaches for effective vaccine development.
A primary hurdle in vaccine development is to develop a molecule which is effectively able to boost memory cells by undergoing rapid antigen processing by host's immune system. Therefore, an important issue for new vaccination strategies attempting to do better than standard intradermal BCG is the ability to induce long-term memory immunity that is protective against primary-disease progression and reactivation of latent disease [5]. Peptides of major immunogenic antigens of MTB can act as effective vaccine candidates as they can directly bind and interact with MHC molecules to activate T-cell response [6]. So far, only limited studies are available on use of MTB Ag85 peptides as vaccine candidates in animal models.
In our earlier studies, we have shown that homologous boosting with BCG increases humoral response (anti-BCG and total IgG titre) [7]. Objective of present study was to evaluate efficacy of three different molecules: BCG, Ag85B, and Ag85 peptide based on their ability to boost waning BCG induced immune response in mouse model. Main purpose was to study comparative booster efficacy of Ag85B and BCG, with that of monomeric peptide construct of Ag85B, for studying its vaccine efficacy.
Materials and Methods
Mice
Female BALB/c mice, 6-8-week-old, 25-30 g were purchased from Shree farms, Bhandhara, MS, India. All the mice were housed in Animal house facility developed by Central India Institute of Medical Sciences (CIIMS) at Nagpur Veterinary College, Nagpur. Prior to experiments, all mice were acclimatized for 15 days.
Vaccine and reagents
BCG vaccine (Moscow strain) was obtained from Serum Institute of India, Pune and stored at 4-8℃ before use. Mycobacterium bovis purified protein derivative (PPD) for anti-PPD assay was obtained from Indian Veterinary Research Institute (IVRI), Izatnagar, India. Freund's incomplete adjuvant was obtained from Genie, India.
Ag85 peptide designing
T-Cell epitopes for CD4 regions from reference Ag85B sequence were identified from available literature [8,9]. Peptides P1 (P25-P40) (LPGLVG LAGGAATAGA) and P2 (P91-P108) (NG WDINTPAFEW YYQSG), of Ag85B were designed by protocol described elsewhere [10].
Experimental protocol
Mice were divided into four different experimental groups (n=10, per group) and immunized subcutaneously with 0.1 mL (105 colony-forming unit [CFU]) of BCG vaccine. A control group mice (n=10) were separately maintained and sham immunized with sterile saline. Twenty-eight days after primary immunization, mice in respective experimental groups were subcutaneously boosted twice in interval of four weeks with 102 CFU of BCG (group 2), Ag85B (25 µg/mL in adjuvant; group 3), and Ag85 peptides (20 µg/mL of P1/P2 cocktail in adjuvant; group 4). Twenty-eight days after last vaccination, blood samples were collected via retro-orbital route from groups of mice to estimate BCG specific antibodies (ant-BCG, anti-PPD). For analysis of cytokines, mice (n=6) were sacrificed on the same day after blood collection, spleens were isolated and homogenized. The homogenates were stimulated with 20 µg/mL PPD (span diagnostics) and cytokines were analyzed in stimulated homogenates after 8-12 hours.
Analysis of T cell and antibody response
Cytokines (interferon γ [IFN-γ], interleukin 12 [IL-12]) in spleen homogenates were measured using E Biosciences Kit (e-Biosciences, Inc., San Diego, CA, USA) according to standard sandwich enzyme-linked immunosorbent assay protocols. Anti-BCG titre was evaluated by protocol described earlier by Husain et al. [7]. For anti-PPD, the 96-well microtiter plates were coated with M. bovis PPD (5 ng) and incubated for 3 hours at 37℃. The rest of the procedure was similar as described for anti-BCG titre estimation.
Statistical analysis
Data are expressed as mean±standard deviation. Comparison of t-test was used for obtaining statistical significance. p<0.05 was considered statistically significant and p<0.001 was considered for highly significant values. Figures were plotted using MedCalc statistical software (Ostend, Belgium).
Ethics statement
The protocols for animal experiments were approved by Institutional Animal Ethics Committee of CIIMS, Nagpur and conform to the provisions of the Declaration of Helsinki (as revised in Tokyo 2004).
Results
Assessment of T-cell response
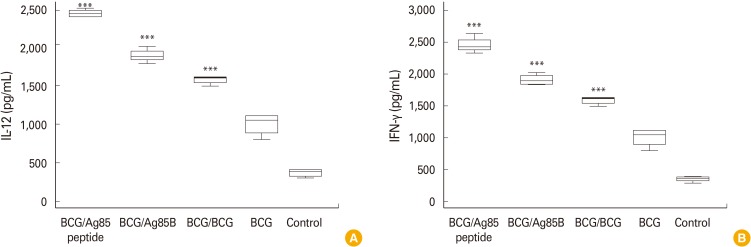
The present study was designed with an objective to evaluate boosting potential of three molecules namely BCG, Ag85B, and Ag85B peptides to boost BCG induced immune response. Fig. 1A and B shows IL-12 and IFN-γ levels in spleen organ homogenates of mice post booster dose with BCG, Ag85B, and Ag85B peptides. Results showed that groups of mice boosted with Ag85 peptide showed significant (p<0.001); IFN-γ and IL-12 levels compared to booster dose of Ag85B and BCG and control. Similarly, significant difference was observed between booster dose of BCG and Ag85B (p<0.001) to boost both cytokines; however, no significant difference between the same was observed to boost IL-12 levels. Mice boosted with all molecules showed significant levels of both cytokines (p<0.001) when compared with single dose of BCG and control.
Fig. 1.
Box whisker plots of IL-12 (A) and IFN-γ (B) levels in spleen homogenates of mice (n=6) after administration of booster doses with BCG, Ag85B, and Ag85 peptides. Twenty-eight days after last vaccination, mice from respective groups were sacrificed and their spleens were isolated, homogenized and stimulated with PPD. IL-12, interleukin 12; IFN-γ, interferon γ; BCG, Bacillus Calmette-Guerin; Ag85B, antigen 85B; PPD, purified protein derivative. Data was analyzed with the Student t-test (***p<0.001, BCG/BCG vs. BCG and control; ***p<0.001, Ag85B vs. BCG and control; ***p<0.001, peptide vs. all group). Error bars indicate the standard error of the mean; data are representative of 2 independent experiments.
Assessment of B-cell response
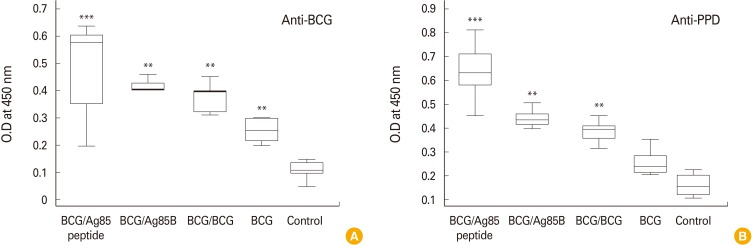
Humoral responses were also analyzed in our study with a view to get a correlation between T-cell and B-cell responses and also to find out ability of respective molecules to boost BCG specific IgG response. Fig. 2A and B shows anti-BCG and anti-PPD titres in serum of mice post-BCG vaccination and after booster doses with BCG, Ag85B, and Ag85B peptide boost. Mice boosted with Ag85B peptides showed significant anti BCG and anti-PPD response (p<0.001) as compared to booster dose with BCG and Ag85B. Booster doses with BCG and Ag85B also induced significant titres (p<0.05) compared with single BCG dose and control.
Fig. 2.
Box whisker plots of BCG specific antibodies anti-BCG (A) and anti-PPD (B) levels in serum of experimental mice (n=8) after administration of booster doses with BCG, Ag85B, and Ag85 peptide. Blood was collected 21 days after booster doses to study antimycobacterial cytokine reposne. BCG, Bacillus Calmette-Guerin; PPD, purified protein derivative; Ag85B, antigen 85B. Data were analyzed with the Student's t-test (**p<0.05, Ag85B and BCG/BCG vs. BCG and control; ***p<0.001, peptide vs. all group). Error bars indicate the standard error of the mean; data are representative of two independent experiments.
Discussion
Existing BCG vaccination protocol has failed to control tuberculosis infection. There is a need to have superior vaccination strategies to control TB infection especially in developing countries [11]. Previous studies have shown that repeat vaccination with BCG may be deleterious to protection against TB, and heterologous boost vaccines are likely to be used to enhance specific immunity primed by BCG [12] because BCG is not an effective booster vaccine itself. The prime-boost strategy using both a new prime and novel boost vaccines for TB is thus expected to enhance cellular immunity and produce the most effective vaccine regimen to protect against tuberculosis infection [13]. However, comparative studies on booster potential of different MTB antigens and peptides are needed to be carried out prior to moving on to protective studies. In the present study, we evaluated three different molecules based on their potential to boost BCG induced immune response, to find out which molecule can act as better candidate to be used in heterologous prime boost regimes. Based on results of our studies, it was found that mice boosted with peptides of Ag85B induced better cytokine and BCG specific antibody response compared to its boosting counterparts Ag85B and BCG itself. The results, although very preliminary, suggest important role of peptides to boost waning BCG immune response and their future role in development of TB vaccines.
Some of the researchers have worked and showed effectiveness of antigenic peptides as vaccine candidates when used alone or in heterologous prime boost regimes using in vitro [8,9] and animal models. Olsen et al. [14] in their studies showed efficient protection against MTB using 20-mer synthetic construct from ESAT-6 protein. Zugel et al. [15] evaluated immunogenicity of gp-96 associated peptides against intracellular bacteria like MTB and showed that it can act as potential vaccine candidate. Sugawara et al. [16] have used immunogenic synthetic Ag85A peptide as a booster together with recombinant BCG Tokyo (Ag85A) or Ag85A DNA. They have shown significant improvement in lung pathology and significant reduction in pulmonary CFU [16]. Gowthaman et al. [17] have shown that lipidated promiscuous peptide vaccines evokes better immune response and may be alternative to BCG in TB endemic regions. Peptides are comparatively smaller molecules and are supposed to be easily processed and presented and elicit immune response much quickly. The above hypothesis support our findings which demonstrate potential Ag85B peptides to efficiently boost memory cells primed by BCG and induced robust T-cell cytokine response. In our study, we also observed similar efficacy of booster doses with BCG and Ag85B to stimulate immune response, which was significant than single dose. Use of Ag85 in TB vaccine research has already been carried out extensively; in fact, vaccine construct of Ag85A has been recently evaluated in phase III clinical trials and was not found to confer any protection [4]. Similarly, there are dozens of vaccine candidates of other MTB antigens under clinical trials, which have somehow failed to surpass BCG in its immunogenicity. Under the present condition, our results suggest that low dose of BCG may be used as an alternative to subunit based vaccine to boost BCG induced immunity in TB endemic settings. However further evlaution is needed on this aspect to justify the results.
Another aspect of the study was that apart from evaluating T-cell response, we also focused on assessing B-cell response. Although it is well established fact about role of TH1 response in TB vaccines, especially cytokines like IFN-γ, IL-12, IL-17, while the role of humoral immunity in host defense against MTB is poorly understood. B Cells have been described as an important source of chemokines involved in granuloma development and consequently inhibit mycobacterial dissemination, resulting in recruitment of appropriate cells in the vicinity for the first few weeks after infection [18]. During the past few years, there are lots of published reports which suggest a key role of humoral response in TB immunology. In recent studies, it has been shown that antibody responses are essential to contain mycobacterial infection, and there is a synergy and mutual interdependence between cell mediated and humoral immunity [19]. Zuniga et al. [20], in their review article, have discussed briefly about different cellular and humoral mechanism evolved in control of tuberculosis infection and also suggested a possible role of B cells in modulating immune response in TB. Thus, focus on evaluation of molecules imparting efficient B-cell response apart from T-cell immunity is therefore needed so as they can be used as effective booster candidates. In our study, we found that peptide of Ag85B induced significant levels of BCG specific IgG response compared with Ag85B and BCG. However, this was just a pilot study which showed effectiveness of heterologous regime as a prophylactic measure using increase in immune response as correlate of immunogenicity. Evaluation of immunogenicity and protective efficacy of such peptides by subsequent challenge with mycobacteria and evaluation of CFU numbers in lungs will further help us in better understanding of use of such peptides in TB vaccination in future. Therefore, a lot of further work is need study the mechanism of action, use of conjugates along with MTB peptides, development of proper disease model and also assessment of mixed Th1/Th2 response that will allow optimization of such vaccination strategies, and ultimately the development of improved prime boost vaccination strategies against TB.
To conclude, peptides of Ag85B act as better booster candidate's compared to BCG and Ag85B and may be used to boost BCG induced immune response in future prime boost strategies. However, study requires further evaluation in animal studies using experimental model of TB infection to justify the present results.
Footnotes
No potential conflict of interest relevant to this article was reported.
A.A.H thanks Indian Council of Medical Research (ICMR), New Delhi for award of Senior Research Fellowship. All Authors would like to thank Central India Institute of Medical Sciences (CIIMS) for funding this study.
References
- 1.Gupta UD, Katoch VM. Animal models of tuberculosis for vaccine development. Indian J Med Res. 2009;129:11–18. [PubMed] [Google Scholar]
- 2.Gupta UD, Katoch VM, McMurray DN. Current status of TB vaccines. Vaccine. 2007;25:3742–3751. doi: 10.1016/j.vaccine.2007.01.112. [DOI] [PubMed] [Google Scholar]
- 3.Agger EM, Andersen P. A novel TB vaccine; towards a strategy based on our understanding of BCG failure. Vaccine. 2002;21:7–14. doi: 10.1016/s0264-410x(02)00447-4. [DOI] [PubMed] [Google Scholar]
- 4.Tameris MD, Hatherill M, Landry BS, et al. Safety and efficacy of MVA85A, a new tuberculosis vaccine, in infants previously vaccinated with BCG: a randomised, placebo-controlled phase 2b trial. Lancet. 2013;381:1021–1028. doi: 10.1016/S0140-6736(13)60177-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Skeiky YA, Sadoff JC. Advances in tuberculosis vaccine strategies. Nat Rev Microbiol. 2006;4:469–476. doi: 10.1038/nrmicro1419. [DOI] [PubMed] [Google Scholar]
- 6.Allen PM, Strydom DJ, Unanue ER. Processing of lysozyme by macrophages: identification of the determinant recognized by two T-cell hybridomas. Proc Natl Acad Sci U S A. 1984;81:2489–2493. doi: 10.1073/pnas.81.8.2489. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Husain AA, Kashyap RS, Kalorey DR, et al. Effect of repeat dose of BCG vaccination on humoral response in mice model. Indian J Exp Biol. 2011;49:7–10. [PubMed] [Google Scholar]
- 8.Mustafa AS, Shaban FA, Abal AT, et al. Identification and HLA restriction of naturally derived Th1-cell epitopes from the secreted Mycobacterium tuberculosis antigen 85B recognized by antigen-specific human CD4(+) T-cell lines. Infect Immun. 2000;68:3933–3940. doi: 10.1128/iai.68.7.3933-3940.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Valle MT, Megiovanni AM, Merlo A, et al. Epitope focus, clonal composition and Th1 phenotype of the human CD4 response to the secretory mycobacterial antigen Ag85. Clin Exp Immunol. 2001;123:226–232. doi: 10.1046/j.1365-2249.2001.01450.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Kashyap RS, Shekhawat SD, Nayak AR, Purohit HJ, Taori GM, Daginawala HF. Diagnosis of tuberculosis infection based on synthetic peptides from Mycobacterium tuberculosis antigen 85 complex. Clin Neurol Neurosurg. 2013;115:678–683. doi: 10.1016/j.clineuro.2012.07.031. [DOI] [PubMed] [Google Scholar]
- 11.Barreto ML, Pereira SM, Ferreira AA. BCG vaccine: efficacy and indications for vaccination and revaccination. J Pediatr (Rio J) 2006;82(3 Suppl):S45–S54. doi: 10.2223/JPED.1499. [DOI] [PubMed] [Google Scholar]
- 12.McShane H, Hill A. Prime-boost immunisation strategies for tuberculosis. Microbes Infect. 2005;7:962–967. doi: 10.1016/j.micinf.2005.03.009. [DOI] [PubMed] [Google Scholar]
- 13.Martin C. The dream of a vaccine against tuberculosis; new vaccines improving or replacing BCG? Eur Respir J. 2005;26:162–167. doi: 10.1183/09031936.05.00109904. [DOI] [PubMed] [Google Scholar]
- 14.Olsen AW, Hansen PR, Holm A, Andersen P. Efficient protection against Mycobacterium tuberculosis by vaccination with a single subdominant epitope from the ESAT-6 antigen. Eur J Immunol. 2000;30:1724–1732. doi: 10.1002/1521-4141(200006)30:6<1724::AID-IMMU1724>3.0.CO;2-A. [DOI] [PubMed] [Google Scholar]
- 15.Zugel U, Sponaas AM, Neckermann J, Schoel B, Kaufmann SH. gp96-peptide vaccination of mice against intracellular bacteria. Infect Immun. 2001;69:4164–4167. doi: 10.1128/IAI.69.6.4164-4167.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Sugawara I, Udagawa T, Taniyama T. Protective efficacy of recombinant (Ag85A) BCG Tokyo with Ag85A peptide boosting against Mycobacterium tuberculosis-infected guinea pigs in comparison with that of DNA vaccine encoding Ag85A. Tuberculosis (Edinb) 2007;87:94–101. doi: 10.1016/j.tube.2006.05.001. [DOI] [PubMed] [Google Scholar]
- 17.Gowthaman U, Rai PK, Khan N, Jackson DC, Agrewala JN. Lipidated promiscuous peptides vaccine for tuberculosis-endemic regions. Trends Mol Med. 2012;18:607–614. doi: 10.1016/j.molmed.2012.07.008. [DOI] [PubMed] [Google Scholar]
- 18.Bosio CM, Gardner D, Elkins KL. Infection of B cell-deficient mice with CDC 1551, a clinical isolate of Mycobacterium tuberculosis: delay in dissemination and development of lung pathology. J Immunol. 2000;164:6417–6425. doi: 10.4049/jimmunol.164.12.6417. [DOI] [PubMed] [Google Scholar]
- 19.Abebe F, Bjune G. The protective role of antibody responses during Mycobacterium tuberculosis infection. Clin Exp Immunol. 2009;157:235–243. doi: 10.1111/j.1365-2249.2009.03967.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Zuniga J, Torres-Garcia D, Santos-Mendoza T, Rodriguez-Reyna TS, Granados J, Yunis EJ. Cellular and humoral mechanisms involved in the control of tuberculosis. Clin Dev Immunol. 2012;2012:193923. doi: 10.1155/2012/193923. [DOI] [PMC free article] [PubMed] [Google Scholar]