Abstract
Cell therapy using multipotent mesenchymal stromal cells (MSCs) is of high interest in various indications. As the pleiotropic effects mediated by MSCs rely mostly on their unique secretory profile, long-term persistence of ex-vivo-expanded cells in the recipient may not always be desirable. Irradiation is a routine procedure in transfusion medicine to prevent long-term persistence of nucleated cells and could therefore also be applied to MSCs. We have exposed human bone-marrow-derived MSCs to 30 or 60 Gy of γ-irradiation and assessed cell proliferation, clonogenicity, differentiation, cytokine levels in media supernatants, surface receptor profile, as well as expression of proto-oncogenes/cell cycle markers, self-renewal/stemness markers, and DNA damage/irradiation markers. Irradiated MSCs show a significant decrease in proliferation and colony-forming unit–fibroblasts. However, a subpopulation of surviving cells is able to differentiate, but is unable to form colonies after irradiation. Irradiated MSCs showed stable expression of CD73 and CD90 and absence of CD3, CD34, and CD45 during a 16-week follow-up period. We found increased vascular endothelial growth factor (VEGF) levels and a decrease of platelet-derived growth factor (PDGF)-AA and PDGF-AB/BB in culture media of nonirradiated cells. Irradiated MSCs showed an inverse pattern, that is, no increase of VEGF, and less consumption of PDGF-AA and PDGF-AB/BB. Interestingly, interleukin-6 (IL-6) levels increased during culture regardless of irradiation. Cells with lower sensitivity toward γ-irradiation showed positive β-galactosidase activity 10 days after irradiation. Gene expression of both irradiated and nonirradiated MSCs 13–16 weeks after irradiation with 60 Gy predominantly followed the same pattern; cell cycle regulators CDKN1A (p21) and CDKN2A (p16) were upregulated, indicating cell cycle arrest, whereas classical proto-oncogenes, respectively, and self-renewal/stemness markers MYC, TP53 (p53), and KLF4 were downregulated. In addition, DNA damage/irradiation markers ATM, ATR, BRCA1, CHEK1, CHEK2, MDC1, and TP53BP1 also mostly showed the same pattern of gene expression as high-dose γ-irradiation. In conclusion, we demonstrated the existence of an MSC subpopulation with remarkable resistance to high-dose γ-irradiation. Cells surviving irradiation retained their trilineage differentiation capacity and surface marker profile but changed their cytokine secretion profile and became prematurely senescent.
Introduction
Multipotent mesenchymal stromal cells (MSCs) are highly promising candidates for a diverse range of clinical applications in tissue regeneration, cell therapy, gene therapy, and immunomodulation. Their hallmark properties include plastic adherence; trilineage differentiation capacity into adipocytes, chondrocytes, and osteoblasts; as well as a surface phenotype defined by the absence of hematopoietic and antigen-presenting surface markers and the presence of MSC-characteristic antigens CD73, CD90, and CD105.1 Due to the absence of a unique surface marker suitable for their prospective isolation and the low frequency in human tissues, cells must be expanded in vitro to obtain sufficient cell numbers for clinical use. While the role of direct cell-to-cell interactions in mediating their pleiotropic functions is still unclear, their unique secretory profile appears to be sufficient for dampening proinflammatory immune responses and inducing lasting tissue regeneration and repair via a “touch-and-go” effect.2 Therefore, long-term persistence of ex-vivo-expanded cells in the recipient may not always be desirable. γ-Irradiation with 30 Gy is a routine procedure commonly employed by blood donor services to deplete blood products from nucleated cells and could be used to deliver a sublethal hit to MSCs that would be sufficient to abrogate their colony-forming ability and prevent their engraftment in vivo. Surprisingly, however, a small fraction of the irradiated cells were found to survive both 30 Gy as well as 60 Gy of irradiation, prompting us to further investigate the cell morphology, phenotype, and differentiation capacity to determine whether irradiation had affected or altered the cell biology in vitro. Cells may become senescent by a number of disparate mechanisms, including extracellular stresses, such as ionizing radiation or chemical agents. This effect has also been shown to be induced by ex vivo expansion.3 In addition, since most human cells do not express telomerase, the progressive shortening of chromosomal telomeres predetermines the number of possible cell divisions. Once a minimal threshold (“Hayflick”—limit) is passed, a persistent DNA damage signal is generated. Subsequently, the cell enters a permanent nondividing state, which entails an irreversible cell cycle arrest as well as concomitant alterations in cell morphology, gene expression, and cellular functions, termed replicative (or proliferative) cellular senescence. Regardless of the initiating event, once the cell has sensed a critical level of damage or dysfunction, the senescence program is activated. Intracellular responses to genotoxic stress rely on the dynamic orchestration of DNA-damage-induced complex regulatory pathways involving potent tumor suppressors, such as TP53 (p53) and/or CDKN2A (p16), in mammalian cells and constitute one of the most potent and pivotal antitumoral barriers. High levels of CDKN1A (p21) cause chronic DNA damage response signaling that maintains the senescence growth arrest.4 CDKN2A is also a cyclin-dependent kinase inhibitor that renders cell arrest permanent and therefore acts as a biomarker of aging and senescence.
The principal aim of this study was to investigate how high doses of γ-irradiation affect human bone marrow (BM)–derived MSCs and determine the optimal dose required for termination of MSC colony-forming ability. As a “positive” control, we chose irradiation with 60 Gy, a dose putatively sufficient for cell death. As expected, a large proportion of cells did not recover from exposure to ionizing radiation with 30 and 60 Gy. Remarkably, a radiation-resistant cell population survived this procedure and retained the ability to proliferate and to differentiate into adipocytes, osteoblasts, and chondrocytes, although at much reduced levels compared with nonirradiated controls. This prompted us to further characterize the cells to assess their prospective use in cell therapy by investigating their immunophenotype and cytokine and gene expression profile, and by karyotyping after culture in vitro. Following irradiation, we also observed senescence-type cell morphology in addition to expression of β-galactosidase, indicating premature cell senescence in surviving MSCs that was also confirmed by our analyses of cell cycle markers and cytokine profile of cell culture supernatants.
Materials and Methods
Isolation and expansion of human BM-derived MSCs
All MSCs used for the following experiments were primary MSCs, collected from BM aspirations (iliac crests) of volunteer healthy donors. Collection of this material has been approved by the Ethical Committee of University of Ulm after written informed consent of donors has been obtained according to the declaration of Helsinki. Isolation and cell culture were performed according to standardized GMP-compliant protocols designed for clinical-scale production of MSCs as described previously.5 Cell culture medium consisted of α-minimum essential medium (α-MEM; Lonza, Basel, Switzerland), supplemented with 10% of human platelet lysate (PL; IKT Ulm, Ulm, Germany) and 1 i.u. of Na-heparin (B. Braun, Melsungen, Germany or Ratiopharm, Neu-Ulm, Germany) per mL of medium.
Human dermal fibroblasts
Normal human dermal fibroblasts from adult skin (NHDF-Ad; Clonetics™) were purchased from Lonza at passage 2 and cultivated in α-MEM supplemented with 10% of human PL and 1 i.u. of Na-heparin.
Irradiation of MSCs
MSCs were plated on T-75 flasks (Nunc, Wiesbaden, Germany) at a seeding density of 20,000 or 5000 cells/cm2 and incubated for 24 h at 37°C and 5% CO2. Cell irradiation was performed using the ISO-classified (E.63545) BioBeam 8000 (Gamma-Service Medical GmbH, Leipzig, Germany) with a 137Cs source.
MSCs were γ-irradiated with a total dose of 30 or 60 Gy, respectively. Nonirradiated MSCs were used as a control for all experiments. Cells were harvested immediately following γ-irradiation and subcultivated in flasks or frozen for analysis.
For all analyses except cytokine quantification, cells were cultivated until reaching 70–80% confluence and then harvested and subcultivated. Partial medium exchange (50% of volume) was performed twice each week.
Cell proliferation and viability
Cell proliferation was assessed using the CyQUANT® Cell Proliferation Assay Kit (Invitrogen, Carlsbad, CA) according to manufacturer's instructions after 7 days of culture in the indicated conditions. Fluorescence was measured in relative fluorescence units using POLARstar Omega plate reader (BMG Labtech, Ortenberg, Germany) and background signal of lysis buffer was subtracted. Trypan blue staining and cell count in a Neubauer chamber was performed at 7, 14, 21, and 28 days after irradiation to determine the number of viable MSCs.
Modified colony-forming unit–fibroblast assay
Following irradiation, MSCs at passage 2 to passage 4 were seeded in six-well multiwell plates (Nunc) at different seeding densities of 9, 27, 54, or 162 cells/cm2. Cells were then cultivated at standard conditions in α-MEM (Lonza) supplemented with 10% of human PL for up to 28 days performing medium exchanges twice per week. Colonies were stained with Giemsa and counted after 8 and 28 days. This modification minimizes counting errors due to confluence and small, slow-proliferating colonies as well as low clonogenicity rates will be visible using this range of seeding densities and culture times. The experiment was performed three times using MSC preparations from different donors, each in triplicate wells.
Flow cytometric characterization and differentiation assays
Antibodies used for characterization of MSCs included CD3 (SK7), CD34 (8G12), CD45 (2D1), CD73 (AD2), CD90 (5E10), HLA-ABC (G46-2.6), and HLA-DR, DP, DQ (Tü39), all from BD Biosciences (Heidelberg, Germany), and CD105 (SN6) (AbD Serotec, Düsseldorf, Germany). All antibodies were used according to manufacturers' instructions. Relative fluorescence intensity of cells was acquired using an FACSAria with FACS DIVA Software (BD Immunocytometry Systems, Heidelberg, Germany).
Differentiation and senescence assays
About 2.25×104 MSCs/cm2, for adipogenic differentiation assay, and 5.00×103 MSCs/cm2, for chondrogenic and osteogenic differentiation assays, were seeded and differentiation was induced according to the manufacturers' instructions; adipogenic differentiation medium was purchased from Lonza and chondrogenic and osteogenic differentiation media were from Miltenyi Biotec (Bergisch-Gladbach, Germany). After differentiation, cells were fixed in 4% paraformaldehyde and osteogenic differentiation was detected by showing alkaline phosphatase activity. Adipogenic differentiation was monitored by staining with a saturated Oil Red O solution (counterstaining with Meyer's hematoxylin). Chondrogenic differentiation was performed by methylene blue staining. All reagents for staining were purchased from Sigma (Schnellendorf, Germany) except for methylene blue (Merck, Darmstadt, Germany). For senescence assays, about 2.50×102 MSCs/cm2 were seeded and X-gal staining for detection of senescence-associated β-galactosidase activity was performed. Differentiation experiments were performed three times using MSC preparations from different donors.
Cytokine analysis
Custom-designed MILLIPLEX® Human Cytokine/Chemokine 96-well Plate Assays (Cat. No. MPXCYTO-60K and No. HNDG3-36K; Millipore Corporation, Billerica, MA) and TGF-β1 Single Plex Kit (Cat. No. TGFB-64K-01) were used for the simultaneous quantification of human cytokines and chemokines of cell culture supernatants as per the manufacturer's specifications. At each time point of interest during MSC cultivation, one aliquot was stored at −80°C for subsequent simultaneous measurement via the MILLIPLEX Cytokine Assay on a Luminex LS-100 platform. Since freezing/thawing cycles affect absolute concentrations of cytokines (data not shown), samples were processed in a way that they did not differ in number of freezing/thawing cycles. To facilitate detection of less-pronounced alterations in the cytokine profile of irradiated cells compared with nonirradiated controls, cell culture medium was not replaced and cells were not subcultivated for the duration of these experiments.
Quantitative detection of mRNA transcripts by real-time reverse transcriptase polymerase chain reaction
Quantitative detection of mRNA transcripts was performed as described previously.6 Briefly, Qiagen® RNeasy® Mini Kit (Cat. No. 74104) in combination with Qiagen® RNase-Free DNase Set (Cat. No. 79254), for total RNA extraction, and Qiagen® Omniscript® RT Kit (Cat. No. 205111), for reverse transcription reaction, were used. About 3.5–5 ng of cDNA was used per real-time reverse transcriptase polymerase chain reaction (qPCR). qPCR was performed on an Applied Biosystems® 7500 Fast Real-Time PCR System with Applied Biosystems TaqMan® Fast Universal PCR Master Mix (2×), No AmpErase® UNG (Part No. 4352042), and the following TaqMan Gene Expression Assays from Applied Biosystems (Foster City, CA): ATM Hs01112355_g1, ATR Hs00992123_m1, BRCA1 Hs01556193_m1, CDC25A Hs00947994_m1, CDKN1A Hs00355782_m1, CDKN2A Hs00923894_m1, CHEK1 Hs00967506_m1, CHEK2 Hs00200485_m1, EEF1A1 Hs00265885_g1, KLF4 Hs00358836_m1, MDC1 Hs00206182_m1, MYC Hs00905030_m1, NANOG Hs02387400_g1 and Hs04260366_g1, POU5F1 Hs00999632_g1, REXO1 Hs00810654_m1, RPL13A Hs01926559_g1, SOX1 Hs01057642_s1, SOX2 Hs01053049_s1, TERC Hs03297287_s1, TERT Hs00972656_m1, TP53 Hs01034249_m1, and TP53BP1 Hs00996818_m1. All components were used according to the manufacturers' manuals. Data were analyzed with 7500 System SDS v2.0.4 and Microsoft Office Excel 2003/10 software. Three to eight replicates per experimental condition were analyzed. All qPCR data are presented as mean±standard deviation. For statistical analyses two-tailed Student's t-test was applied. p-Values <0.05 were considered as statistically significant (*) and p-values <0.01 were considered as statistical highly significant (**).
Karyotyping
For karyotyping, irradiated or nonirradiated cells were cultured at a density of 15,000 MSCs per T-75 flask (Nunc). Cells were treated with 750 mg/mL colchicin for at least 60 min. Routine analysis of G-banded chromosomes was performed according to standard procedures as described elsewhere.7 Karyotyping was performed by the Institute for Human Genetics, University of Ulm (Ulm, Germany). Band resolution was 300–400 per haploid chromosome set.
Statistics
Statistical analysis was performed using unpaired, two-sided Student's t-test. Threshold for highly significant difference was p<0.01 and for significant difference was p<0.05.
Results
Cell proliferation following γ-irradiation
In our experimental design, we originally aimed to determine the γ-radiation dose optimally suited to allow for a transient survival of MSCs in vivo. Thirty gray is the standard dose commonly used for γ-radiation of blood products to deplete transfusion products from immunocompetent cells. Radiation of MSCs with 60 Gy was performed to ensure that cells receive a “lethal hit” and subsequently undergo apoptosis rapidly. As expected, a large proportion of cells did not recover from exposure to ionizing radiation with 30 and 60 Gy as observed microscopically by the appearance of apoptotic bodies and loss of adherence. Surprisingly, we observed a cell population surviving this treatment. Therefore, we investigated the proliferative capacity and survival rates of cells irradiated with 30 or 60 Gy (Fig. 1). Nonirradiated MSCs served as control. MSCs irradiated with 30 and 60 Gy showed marked decline in proliferation compared with nonirradiated cells, respectively. However, a surviving cell population persisted and continued to proliferate up to 120 days after treatment (Fig. 1A, C).
FIG. 1.
Mesenchymal stromal cell (MSC) proliferation and viability after irradiation. MSCs were irradiated with either 30 or 60 Gy, harvested, and subsequently assessed for their proliferative capacity and viability. (A) Cumulative population doublings of irradiated and nonirradiated MSCs up to 120 days after irradiation with 30 or 60 Gy. (B) Cell viability of irradiated and nonirradiated adherent MSCs by trypan blue staining and cell counting. Panels (A) and (B) show raw data of population doublings of n=3 independent experiments. (C) Cell proliferation of irradiated MSCs relative to nonirradiated controls. Cells were seeded at either higher (20,000 cells/cm2) or lower density (5000 cells/cm2), irradiated with either 30 or 60 Gy, and cultivated up to 28 days. Shown are mean values of n=4 experiments. *p<0.05; **p<0.01.
This was also confirmed by quantification of viable, adherent, irradiated and nonirradiated cells via trypan blue exclusion (Fig. 1B). Figure 1A depicts the calculated number of cell doublings of the entire cell population. This number is the result of both cell proliferation and cell death. As most of the cells died following irradiation—either immediately or via a delayed effect—and only a small subset of the cell population continued to proliferate, the overall number of cell doublings remained at a constant level. We could not observe any statistically significant influence of cell seeding density on MSC proliferation (p>0.05) respective to their irradiation with 30 or 60 Gy (Fig. 1C), viability or phenotype. We also documented cell morphology after exposure to ionizing radiation microscopically (Supplementary Fig. S1; Supplementary Data are available online at www.liebertpub.com/tec). Nonirradiated control cells showed alterations in their distribution pattern and a senescence-characteristic cell shape8 starting after about 3–4 weeks of culture, which correlated with increased doubling times, suggesting that MSCs had entered replicative senescence. Cells that had survived irradiation treatment and remained plastic adherent showed a typical visible cell enlargement and altered growth pattern within the first 2 weeks after exposure to high doses of radiation. However, cells could be subcultivated and continued to proliferate for up to 16 weeks, although at very low to negative numbers of cumulative population doublings (Fig. 1A). Fibroblasts share many phenotypic and functional characteristics with MSCs as indicated by surface marker expression patterns, global gene expression, and immunosuppression in allogeneic transplantation.9 Therefore, we also investigated radiation resistance of normal human dermal fibroblasts from adult skin (NHDF-Ad). As illustrated in Supplementary Figure S2, we also observed a very similar proliferation profile for irradiated and nonirradiated NHDF-Ad, corroborating previous findings.10
Differentiation capacity and cellular senescence of irradiated cells
We were also interested whether MSCs retained their characteristic trilineage differentiation capacity following γ-irradiation with 30 and 60 Gy. To minimize an appearance of spontaneous MSC differentiation in vitro, MSCs were used at passages 0–3 for these experiments. Despite the slower proliferation and altered growth pattern of irradiated cells that prevented them from forming a confluent cell layer necessary for cell differentiation, we could document adipogenic, chondrogenic, and osteogenic differentiation potential in cell cultures of irradiated MSCs following an extended period of cultivation (Fig. 2), although at lower frequencies compared with nonirradiated MSCs. Cellular senescence was investigated via detecting lysosomal β-galactosidase activity at 3 and 10 days postirradiation with either 30 or 60 Gy. At day 3, neither irradiated nor control cells showed detectable β-galactosidase activity. However, at day 10, irradiated cells showed increased β-galactosidase activity in contrast to nonirradiated cells (Fig. 3). This observation suggests that γ-irradiation of MSCs with 30 and 60 Gy induced premature cellular senescence in surviving cells.
FIG. 2.
Differentiation capacity of irradiated MSCs. After exposure to either 30 or 60 Gy of radiation, MSCs were harvested and subsequently cultivated in differentiation-inducing or control media. Nonirradiated MSCs were used as control. Adipogenic differentiation was visualized by Oil Red O staining. Osteogenic differentiation was detected showing alkaline phosphatase activity. Chondrogenic differentiation was performed by methylene blue staining. Black scale bar indicates 100 μm for adipogenic and 250 μm for chondrogenic and osteogenic differentiation. Shown are representative images of one of the three differentiation assays. Color images available online at www.liebertpub.com/tec
FIG. 3.
MSCs enter cellular senescence after irradiation. After irradiation with 30 or 60 Gy, MSCs were harvested and cultivated at standard conditions for either 3 or 10 days. Senescence of cells was then determined via X-gal staining for detection of senescence-associated β-galactosidase activity (as indicated by arrows). Nonirradiated cells were used as control. Black scale bar indicates 600 μm. Shown are representative images of two independent experiments. Color images available online at www.liebertpub.com/tec
Clonogenicity of radiation-resistant cells
To investigate the colony-forming capacity of radiation-resistant cells, we performed colony-forming unit–fibroblast (CFU-F) assays following γ-irradiation with 30 or 60 Gy. While nonirradiated human MSCs formed colonies, irradiated MSCs showed significantly reduced clonogenicity after 8 days (Fig. 4 and Supplementary Table S1) for seeding densities higher than 9 MSCs/cm2. Only single colonies of small size were detectable at day 8 (Fig. 4) and day 14 (data not shown). A decrease in the number of colonies could be detected at day 28 after γ-irradiation (Supplementary Fig. S3). Cells that were able to form colonies are likely to be those surviving cells that we observed in our long-term proliferation and viability assays.
FIG. 4.
Colony-forming unit–fibroblast (CFU-F) assays of nonirradiated, 30-Gy-irradiated, and 60-Gy-irradiated MSCs after 8 days of culture. Representative scans of CFU-F in six-well tissue culture plates of one of the three MSC donors are shown. Seeding density of MSCs is indicated. A representative colony is shown for nonirradiated, 30-Gy–irradiated, and 60-Gy-irradiated MSCs. The table summarizes the results of assays from three donors. Color images available online at www.liebertpub.com/tec
Surface marker expression profile of irradiated MSCs
To better characterize the long-term and late effects of irradiation on MSC immunophenotype, we analyzed the cell surface marker expression profile of MSCs irradiated with 30 and 60 Gy by flow cytometry. Samples were analyzed before (pretreatment) and after exposure to 30 or 60 Gy radiation dose on days 3, 56, and 112. Nonirradiated cells were used as a control. Irradiated MSCs showed stable expression of hallmark antigens CD73, CD90, and CD105 as well as absence of CD3, CD34, and CD45 directly after treatment. In the following 16 weeks of cell culture, this surface marker profile of irradiated and nonirradiated MSCs showed mostly stable expression levels (Supplementary Fig. S4).
Cytokine profile analysis
Following these initial observations, we were interested in cytokine concentrations in culture media of MSCs exposed to high-dose radiation within the first 4 weeks after treatment (Fig. 5). The concentrations of the individual media components in these samples are a result of their concentrations in fresh medium, which is supplemented with human PL; their inherent stability in cell culture conditions; and their production and/or consumption by the cell population.
FIG. 5.
Cytokine analysis of cell culture supernatants after irradiation. MSCs were irradiated with either 30 or 60 Gy. Subsequently, cells were harvested and cultivated for up to 28 days in α-minimum essential medium supplemented with 10% of human platelet lysate. Samples of cell culture supernatants were obtained weekly and quantified for the indicated cytokines. Nonirradiated cells were used as control. Shown are mean and standard deviation (SD) values of n=3 experiments. *p<0.05.
We chose to investigate levels of platelet-derived growth factor (PDGF)-AA, PDGF-AB/BB, growth-regulated protein (GRO), interleukin-6 (IL-6), and vascular endothelial growth factor (VEGF) in the supernatants of irradiated or nonirradiated MSCs, as we could previously identify these factors as being essential for MSC proliferation11 and as relevant markers for further functional characterization of the cell population in vitro.
Levels of PDGF-AA and PDGF-AB/BB progressively declined during cell culture, and showed significantly more rapid decrease in nonirradiated MSC cultures compared with those irradiated with 30 or 60 Gy. Concentrations of GRO (CXCL1/2/3) also decreased markedly during the first 28 days of culture, irrespective of exposure to high doses of radiation. Significantly, irradiated MSC cultures did not show an increase in VEGF concentration as observed in nonirradiated control cultures. Moreover, IL-6 production by MSCs remained consistent in cells exposed to γ-radiation and nonirradiated controls. These results suggest that the high-dose radiation treatment markedly influences the cytokine profile of MSCs.
To verify that the exposure to ionizing radiation itself did not alter cytokine content of media samples, we also analyzed media samples that had been irradiated with either 30 or 60 Gy and incubated concomitantly with cell cultures at the same conditions. Moreover, γ-irradiation did not have any effect on the cytokine content of media samples as evidenced by analysis of PDGF-AA, PDGFAB/BB, GRO, intercellular adhesion molecule 1 (ICAM-1), sCD40L, and VEGF. IL-6 levels were below detection level in media samples (data not shown).
Cell cycle markers, stemness factors, and markers of DNA damage and irradiation
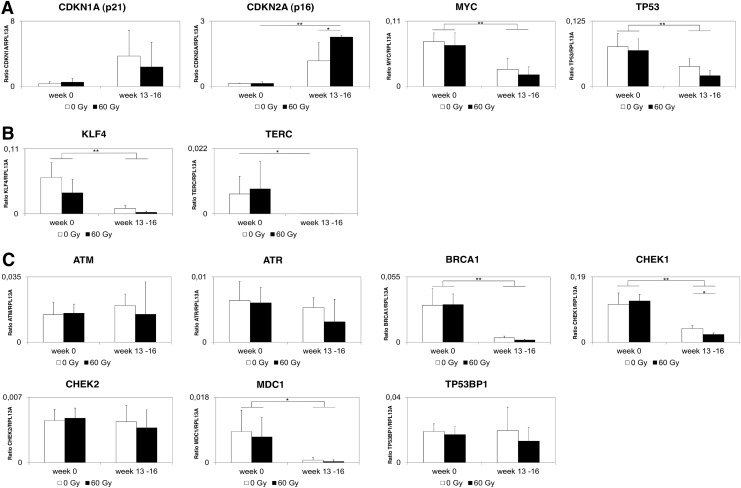
To investigate whether γ-irradiation induced cell transformation or senescence, we analyzed expression of key cell cycle regulators p16, p21, p53, and MYC, as well as telomerase components TERT and TERC by quantitative PCR in nonirradiated and 60-Gy γ-irradiated cells (Fig. 6A, B and Supplementary Fig. S5A, B). We found upregulation of p16 and p21 during 16 weeks of ex vivo cultivation of MSCs. While p16 and p21 increased in both nonirradiated and irradiated MSCs, the increase of p16 was markedly higher in cells exposed to 60 Gy of γ-irradiation compared with the nonirradiated control. Conversely, levels of MYC significantly decreased during the culture period in both nonirradiated and irradiated cells. The same significant decrease in expression of p53 was found in nonirradiated and irradiated MSCs. But, neither for MYC nor for p53 had we found a significant difference in expression between nonirradiated and 60-Gy γ-irradiated surviving cells after 13–16 weeks of culture.
FIG. 6.
Quantitative polymerase chain reaction analysis of (A) proto-oncogenes/cell cycle markers, (B) self-renewal/stemness markers, and (C) DNA damage/irradiation markers. TaqMan gene expression assays for CDC25A, NANOG, POU5F1 (OCT4), REXO1, SOX1, SOX2, and TERT gave no signals in any sample. MSCs were irradiated with 60 Gy and subsequently cultivated at standard conditions for up to 16 weeks. Nonirradiated cells (0 Gy) were used as control. Samples for total RNA isolation were taken directly after irradiation, respectively, nontreatment (week 0) and at week 13–16. Data are given as ratio versus reference gene RPL13A. Data versus a second reference gene are provided as Supplementary Figure 5. White bars: control (nonirradiated; 0 Gy); black bars: cells irradiated with 60 Gy. Shown are mean and SD values of n=3 experiments. *p<0.05; **p<0.01.
Moreover, we were unable to detect TERT in nonirradiated or irradiated MSCs at any time point. Finally, expression of TERC dramatically decreased in MSCs irradiated with 60 Gy as well as in the nonirradiated control after 13–16 weeks of culture, in some replicates below detection level (Fig. 6B and Supplementary Fig. S5B). The qPCR results presented in Figure 6 have been obtained by performing some of the experiments in parallel in the authors' different laboratories. All of the results obtained by the authors show reproducibility and are in accordance with each other.
We also addressed the expression of other well-known self-renewal/stemness factors. For NANOG, POU5F1 (OCT4), REXO1, SOX1, and SOX2, no expression was found at any time point at any sample. KLF4 expression significantly decreased in both nonirradiated and irradiated MSCs during culture period, but it was still detectable (Fig. 6B and Supplementary Fig. S5B).
Additionally, we determined expression levels of prominent markers of DNA damage and irradiation (Fig. 6C and Supplementary Fig. S5C).12 For DNA damage sensors ATM and ATR, no significant change was found neither in nonirradiated nor in irradiated cells. Among DNA damage signal mediators, BRCA1 and MDC1 showed significant reduction in expression levels in both groups after 13–16 weeks of culture, whereas TP53BP1 expression did not change significantly. DNA damage transducers CHEK1 and CHEK2 showed heterogeneous behavior during culture period; while CHEK2 expression did not change significantly, expression of CHEK1 significantly decreased in both groups, but declined even more in 60-Gy γ-irradiated cells compared with nonirradiated cells. TaqMan gene expression assay for DNA damage effector CDC25A revealed no signal. Taken together, naturally aging nonirradiated MSCs and “lethal hit” γ-irradiated surviving counterparts showed remarkably similar expression behavior of determined target genes.
Finally, we performed karyotyping of irradiated and nonirradiated MSCs to investigate possible chromosomal aberrations as a result of either exposure to high doses of γ-irradiation and/or long-term cultivation of MSCs in vitro. Results from cytogenetic analysis are shown in Supplementary Table S2. Aneuploid karyotypes were detected only in two long-term cultures (in 2 out of 13 metaphases for 7554 and in 29 metaphases out of 31 for 7543), which might be related to senescence; karyotype analysis of the rest of cultures did not reveal any pathological findings during culture of nonirradiated MSCs up to 16 weeks. MSCs irradiated with 30 or 60 Gy could not be analyzed due to insufficient numbers of metaphases, further indicating that cells had prematurely entered replicative senescence. Further, we also analyzed normal human dermal fibroblasts from adult skin (NHDF-Ad) treated in the same fashion (Supplementary Table S3), but could also not perform karyotyping on irradiated samples due to an insufficient number of metaphases. Nonirradiated NHDF-Ad did not show pathological abnormalities in the chromosome sets during their cultivation period up to passage 12.
Discussion
Cell-based therapeutic agents are advancing as promising new therapeutic approaches in regenerative medicine. Clinically, MSCs may aid in the reconstitution of tissues as well as in the maintenance of tissue homeostasis. There is increasing evidence that at least some of the MSC-mediated effects observed are exerted through release of cytokines in a “touch-and-go effect,” which could make long-term persistence of these cells nonessential in defined indications.
Cell therapy using MSCs is of high interest in various indications due to their ease of isolation, high proliferative capacity, plasticity, and ability for self-renewal. This has also prompted some concern regarding their potential susceptibility for tumorigenicity. Djouad et al.13,14 showed that MSCs favor earlier onset and syngenic tumor growth in allogenic animals due to their immunosuppressive effect. So far, however, accumulated evidence shows that human MSCs themselves appear to be less prone to spontaneous transformation in vitro and rather likely to enter a stage of replicative senescence after 20–50 population doublings.15 Study of telomere length reveals rapid aging of human marrow stromal cells following in vitro expansion.16 In this state, cells remain alive and are metabolically active, but are unable to replicate. The original rationale for irradiating MSCs had been an apoptosis study in which irradiation of MSCs with 30 Gy was anticipated to be lethal to the entire cell population. Surprisingly, however, a small fraction of the irradiated cells had survived this procedure, prompting us to irradiate MSCs with the double dosage of 60 Gy. Again, we found that some cells even survived this treatment. We then further investigated the cell morphology, phenotype, and differentiation capacity to determine whether irradiation had affected or altered the cell biology in vitro. Importantly, we sought to find out whether those irradiated cells would show signs of malignant transformation, and thus gathering new insight into the susceptibility of MSCs to carcinogenesis. As we could observe, MSCs irradiated with 60 Gy do not show clonal outgrowth as CFU-F and at 30 Gy of γ-irradiation colony formation was massively impaired. A γ-irradiation dose-dependent reduction of CFU-F formation capacity has been described for adipose-tissue-derived MSCs recently.17 An occurrence of aneuploidy during ex vivo cultivation of MSCs was found to be unrelated to the culture process and donor dependent.15 Our results confirm the chromosomal stability of long-term-cultivated, BM-derived MSCs. Importantly, MSCs exposed to high doses of radiation failed to reach sufficient numbers of mitoses necessary for cytogenetic analysis, further suggesting that these cells had prematurely entered replicative senescence.
Senescent cells display phenotypic alterations and an irreversible cell cycle arrest in G1 phase. This arrest is mediated by cyclin-dependent kinase inhibitors p53 and p21 and/or p16 that induce cellular senescence. MYC, on the other hand, is a very strong proto-oncogene and it is very often found to be upregulated in many types of cancers.18 We found upregulation of p16 and p21 during ex vivo cultivation of MSCs in both nonirradiated and irradiated MSCs. However, the increase of p16 was markedly higher in cells exposed to 60 Gy of γ-irradiation compared with the nonirradiated controls, also indicating accelerated senescence in highly irradiated MSCs.
Human telomerase reverse transcriptase protein component TERT and RNA component TERC are catalytic subunits of the enzyme telomerase that maintains telomere ends by addition of telomere repeats and is also a known susceptibility gene for development of many cancers.19 As we were unable to detect TERT in nonirradiated or irradiated MSCs at any time point, this further supports our hypothesis that irradiation did not alter the telomere status of irradiated MSCs compared with nonirradiated controls.
In human fibroblasts, a rapid accumulation of characteristic foci of DNA damage response factors could be observed at the sites of DNA damage induced by ionizing radiation.10 While these foci disappeared within hours in cells that had received only 0.5 Gy, suggesting complete DNA repair, these foci persisted in cells irradiated with 10 Gy and correlated with the induction of cellular senescence. This suggests that high doses of γ-rays may generate a subset of DNA lesions that are not amenable to efficient DNA damage repair and consequently trigger cell cycle arrest.10 DNA damage repair becomes increasingly difficult during cell mitosis when the cell lacks the ability to repair radiation damage and is most sensitive. Hence, cell death is usually described to interfere with proliferation. In our study, we found that high-dose γ-radiation exerted a sustained inhibitory effect on MSC proliferation and markedly altered cell morphology to a senescence-characteristic pattern. The loss of absolute cell numbers as well as cumulative cell doubling numbers most likely result from a depletion of apoptotic MSCs combined with perturbation of reparative processes in surviving cells. We could confirm premature cellular senescence by detection of β-galactosidase activity at 10 days after irradiation, as well as increased expression of cell cycle regulators CDKN1A and CDKN2A at up to 16 weeks after irradiation, indicating cell cycle arrest.
Cells entering senescence concomitantly acquire a complex phenotype termed senescence-associated secretory phenotype that occurs only after establishment of persistent—but not transient—DNA damage response signaling.20 The expression of senescence-associated proteins alters the surrounding tissue microenvironment, reinforces the growth arrest, and allows damaged cells to communicate their compromised state to surrounding cells and induces their clearance by cells of the immune system.21 In our analysis of cytokine levels in cell culture supernatants of irradiated and nonirradiated MSCs, we found GRO and IL-6 levels in the media supernatants to be similar in the first 4 weeks of cell culture. However, levels of PDGF-AA, PDGF-BB, and VEGF showed distinct alterations compared with untreated controls. We also observed a distinct modulation of surface antigens due to the extended in vitro culture, but also some changes that were apparently caused by the radiation treatment. Therefore, given the importance of paracrine effects, exposure of MSCs to high doses of radiation may alter some of the characteristics of MSC activity in vivo. As MSCs are a highly heterogeneous population of plastic-adherent cells and our studies on MSC survival and the analyses of their phenotype and behavior following high-dose γ-radiation were performed in vitro, this study can only provide a more simplistic viewpoint and does not include the in vivo cell response and function in a tissue environment. In this regard, a recent observation revealed that γ-radiation causes an indefinite arrest of human fibroblasts in G1 phase unless the irradiated cells are subcultured. Moreover, the type of extracellular matrix used as a coating substrate to provide cell adhesion significantly affected the length of G1 arrest in human fibroblasts.22 Therefore, cellular senescence, as defined in vitro, may not represent cell fate in vivo.
There are certainly some limitations to this study. Major differences in the mechanistic responses to radiation injury exist that may also be tissue and cell-type specific. Work by Liu and Hornsby, for instance, showed that irradiation of human fibroblasts with a single dose of 44 Gy did not significantly increase the proportion of senescent cells.23 These variable parameters are likely to change the summative response to genotoxic stress and radiation damage.
These added levels of complexity necessitate further research to better understand the dynamic mechanisms contributing to organization and regulation of reparative processes following sublethal cellular damage in MSCs. Three questions are of particular interest: what is the identity of these irradiation-resistant cells? Is it, for example, a distinct subpopulation, or does the cell cycle stage during irradiation cause radiation resistance? and how is the short-term answer against high and low doses of γ-irradiation in radiation-resistant MSCs conceived? Understanding the fundamental mechanisms protecting MSCs from radiation is pivotal as it exemplifies the relationship between cellular senescence, tumorigenesis, and stemness and therefore certainly deserves further research.
In summary, the present study comprehensively documents long-term effects of γ-radiation with single doses of 30 and 60 Gy on MSC hallmark characteristics, cytokine profile, cell cycle regulators, stemness factors, and markers of DNA damage and irradiation. Our results corroborate the remarkable radiation resistance of MSCs and also provide further insight into the chromosomal stability of long-term-cultivated cells in vitro.
Supplementary Material
Acknowledgments
The authors appreciate the excellent technical assistance of Gisela Baur, Thomas Becker, Dorothee Erz, and Karin Fuchs. This project has received funding from the European Union's Seventh Framework Programme for research, technological development, and demonstration under grant agreement No. HEALTH-F5-2009-223236 (CASCADE) and HEALTH-2009-1.4.2-241879 (REBORNE) and BMBF-project GERONTOSYS-SyStaR. This publication forms part of the PhD theses of N.F. and A.E. at Ulm University.
Authors' Contributions
H.S., G.S.-S., A.E., and N.F. planned the project and developed the experimental settings; N.F., E.M.A., and G.S.-S. performed proliferation, CFU-F, differentiation, and vitality testings; N.F. and D.F. performed cytokine analyses; M.T.R. and N.F. analyzed MSCs by flow cytometry; A.L. and A.E. performed qPCR analyses; N.F., A.E., M.T.R., and H.S. wrote the article; and L.S. provided financial and administrative support.
Disclosure Statement
M.T.R., E.M.A., D.F., A.E., and H.S. are working for a nonprofit organization that is manufacturing platelet lysate.
References
- 1.Dominici M., Le B.K., Mueller I., Slaper-Cortenbach I., Marini F., et al. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy 8,315, 2006 [DOI] [PubMed] [Google Scholar]
- 2.Singer N.G., and Caplan A.I.Mesenchymal stem cells: mechanisms of inflammation. Annu Rev Pathol 6,457, 2011 [DOI] [PubMed] [Google Scholar]
- 3.Digirolamo C.M., Stokes D., Colter D., Phinney D.G., Class R., et al. Propagation and senescence of human marrow stromal cells in culture: a simple colony-forming assay identifies samples with the greatest potential to propagate and differentiate. Br J Haematol 107,275, 1999 [DOI] [PubMed] [Google Scholar]
- 4.Coppe J.P., Rodier F., Patil C.K., Freund A., Desprez P.Y., et al. Tumor suppressor and aging biomarker p16(INK4a) induces cellular senescence without the associated inflammatory secretory phenotype. J Biol Chem 286,36396, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Fekete N., Rojewski M.T., Furst D., Kreja L., Ignatius A., et al. GMP-compliant isolation and large-scale expansion of bone marrow-derived MSC. PLoS One 7,e43255, 2012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Lotfi R., Wiegemann D.S., Asseck L., Erle A., Yildiz T., et al. Necrosis-associated factors (DAMPs) including S100A4 used to pulse dendritic cells (DCs) induce regulatory T cells. J Cell Sci Ther 3,134, 2012 [Google Scholar]
- 7.ISCB I.Consensus guidance for banking and supply of human embryonic stem cell lines for research purposes. Stem Cell Rev 5,301, 2009 [DOI] [PubMed] [Google Scholar]
- 8.Wagner W., Horn P., Castoldi M., Diehlmann A., Bork S., et al. Replicative senescence of mesenchymal stem cells: a continuous and organized process. PLoS One 3,e2213, 2008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.McGrail D.J., Ghosh D., Quach N.D., and Dawson M.R.Differential mechanical response of mesenchymal stem cells and fibroblasts to tumor-secreted soluble factors. PLoS One 7,e33248, 2012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Fumagalli M., and d'Adda di Fagagna F.SASPense and DDRama in cancer and ageing. Nat Cell Biol 11,921, 2009 [DOI] [PubMed] [Google Scholar]
- 11.Fekete N., Gadelorge M., Furst D., Maurer C., Dausend J., et al. Platelet lysate from whole blood-derived pooled platelet concentrates and apheresis-derived platelet concentrates for the isolation and expansion of human bone marrow mesenchymal stromal cells: production process, content and identification of active components. Cytotherapy 14,540, 2012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Sancar A., Lindsey-Boltz L.A., Unsal-Kacmaz K., and Linn S.Molecular mechanisms of mammalian DNA repair and the DNA damage checkpoints. Annu Rev Biochem 73,39, 2004 [DOI] [PubMed] [Google Scholar]
- 13.Djouad F., Bony C., Apparailly F., Louis-Plence P., Jorgensen C., et al. Earlier onset of syngeneic tumors in the presence of mesenchymal stem cells. Transplantation 82,1060, 2006 [DOI] [PubMed] [Google Scholar]
- 14.Djouad F., Plence P., Bony C., Tropel P., Apparailly F., et al. Immunosuppressive effect of mesenchymal stem cells favors tumor growth in allogeneic animals. Blood 102,3837, 2003 [DOI] [PubMed] [Google Scholar]
- 15.Tarte K., Gaillard J., Lataillade J.J., Fouillard L., Becker M., et al. Clinical-grade production of human mesenchymal stromal cells: occurrence of aneuploidy without transformation. Blood 115,1549, 2010 [DOI] [PubMed] [Google Scholar]
- 16.Baxter M.A., Wynn R.F., Jowitt S.N., Wraith J.E., Fairbairn L.J., et al. Study of telomere length reveals rapid aging of human marrow stromal cells following in vitro expansion. Stem Cells 22,675, 2004 [DOI] [PubMed] [Google Scholar]
- 17.Hou J., Han Z.P., Jing Y.Y., Yang X., Zhang S.S., et al. Autophagy prevents irradiation injury and maintains stemness through decreasing ROS generation in mesenchymal stem cells. Cell Death Dis 4,e844, 2013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Soucek L., Whitfield J., Martins C.P., Finch A.J., Murphy D.J., et al. Modelling Myc inhibition as a cancer therapy. Nature 455,679, 2008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Liu J.P., Chen W., Schwarer A.P., and Li H.Telomerase in cancer immunotherapy. Biochim Biophys Acta 1805,35, 2010 [DOI] [PubMed] [Google Scholar]
- 20.Rodier F., Coppe J.P., Patil C.K., Hoeijmakers W.A., Munoz D.P., et al. Persistent DNA damage signalling triggers senescence-associated inflammatory cytokine secretion. Nat Cell Biol 11,973, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Coppe J.P., Patil C.K., Rodier F., Krtolica A., Beausejour C.M., et al. A human-like senescence-associated secretory phenotype is conserved in mouse cells dependent on physiological oxygen. PLoS One 5,e9188, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Dimitrijevic-Bussod M., Balzaretti-Maggi V.S., and Gadbois D.M.Extracellular matrix and radiation G1 cell cycle arrest in human fibroblasts. Cancer Res 59,4843, 1999 [PubMed] [Google Scholar]
- 23.Liu D., and Hornsby P.J.Senescent human fibroblasts increase the early growth of xenograft tumors via matrix metalloproteinase secretion. Cancer Res 67,3117, 2007 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.