Significance
Recent genetic studies suggest that variations in the gene encoding metabotropic glutamate receptor 3 (mGlu3) can influence aspects of cognitive function that involve the prefrontal cortex (PFC). Furthermore, mutations in this gene may predispose individuals to developing psychiatric disorders in which altered function of the PFC has been implicated. However, little is known about the precise roles of mGlu3 in regulating the function of the PFC. In the present study, we took advantage of newly identified molecular probes to show that mGlu3 can strongly influence synaptic plasticity within the PFC and that blockade of this receptor impairs specific learning abilities in mice. These results suggest that mGlu3 may be a therapeutic target for cognitive dysfunction in mental disorders.
Keywords: GRM3, medial prefrontal cortex, fear extinction, long-term depression, group II mGlu receptors
Abstract
Clinical studies have revealed that genetic variations in metabotropic glutamate receptor 3 (mGlu3) affect performance on cognitive tasks dependent upon the prefrontal cortex (PFC) and may be linked to psychiatric conditions such as schizophrenia, bipolar disorder, and addiction. We have performed a series of studies aimed at understanding how mGlu3 influences PFC function and cognitive behaviors. In the present study, we found that activation of mGlu3 can induce long-term depression in the mouse medial PFC (mPFC) in vitro. Furthermore, in vivo administration of a selective mGlu3 negative allosteric modulator impaired learning in the mPFC-dependent fear extinction task. The results of these studies implicate mGlu3 as a major regulator of PFC function and cognition. Additionally, potentiators of mGlu3 may be useful in alleviating prefrontal impairments associated with several CNS disorders.
Metabotropic glutamate receptor 3 (mGlu3) has become of increasing clinical interest due to its genetic association with psychiatric conditions. For example, several studies have identified single-nucleotide polymorphisms (SNPs) in GRM3, the human gene encoding mGlu3, that are associated with poor performance on cognitive tests that are dependent on function of the prefrontal cortex (PFC) and hippocampus (1, 2). Additionally, these SNPs have also been associated with variations in functional magnetic resonance imaging (fMRI) indexes of prefrontal cortical activity during working memory tasks (1, 3). Moreover, converging lines of evidence indicate that GRM3 represents a major locus associated with schizophrenia (1, 2, 4), bipolar disorder (5, 6), and substance abuse disorders (6–8). Because mGlu3 is densely expressed in PFC (9), a brain region implicated as a site of pathology in these disorders (10–12), this genetic evidence has led to an increased interest in determining the role of mGlu3 in regulating PFC function and behavior.
Previous studies have revealed that pharmacological activation of group II mGlu receptors (mGlu2 and mGlu3) results in long-term depression (LTD) of excitatory transmission in layer V of the rat medial prefrontal cortex (mPFC) (13–16). Although it is not known whether induction of LTD in the mPFC is mediated by mGlu2 or mGlu3, previous studies suggest that presynaptically localized mGlu2 is typically responsible for inhibition of synaptic transmission by group II mGlu receptor agonists at other synapses (17–23). However, evidence suggests that induction of LTD in the mPFC is dependent upon activation of a postsynaptic group II mGlu receptor (15, 16), suggesting that this response is mechanistically distinct from presynaptic effects of group II mGlu receptor agonists on transmission at other synapses. Unfortunately, a lack of pharmacological agents that can selectively antagonize mGlu3 or mGlu2 has impaired progress in this area. To allow us to begin studies aimed at understanding the role of mGlu3 in regulation of mPFC function, we developed a series of negative allosteric modulators (NAMs) that are highly selective for mGlu3 and are suitable for in vivo use (24). In addition, we now report characterization of a highly selective mGlu2 NAM. We used these compounds, along with mGlu2 and mGlu3 knockout (KO) mice, to evaluate the respective roles of mGlu2 and mGlu3 in acute regulation of synaptic transmission and induction of LTD in mPFC. Interestingly, we found that mGlu2 is involved in acute inhibition of synaptic transmission in the mPFC, but that induction of LTD at this synapse by group II mGlu receptor agonists is mediated exclusively by mGlu3. Furthermore, we found that mGlu3 NAMs impair extinction of conditioned fear, a behavioral task that is critically dependent upon the integrity of the mPFC. These data suggest that mGlu3 plays an essential role in the regulation of a specific form of synaptic plasticity in the mPFC that could be important for forms of cognitive function that require depression of excitatory inputs to mPFC and are thought to be disrupted in patients suffering from a range of CNS disorders.
Results
NMDA Receptor-Independent LTD with a Postsynaptic Component Is Induced by Pharmacological Activation of Group II mGlu Receptors in mPFC.
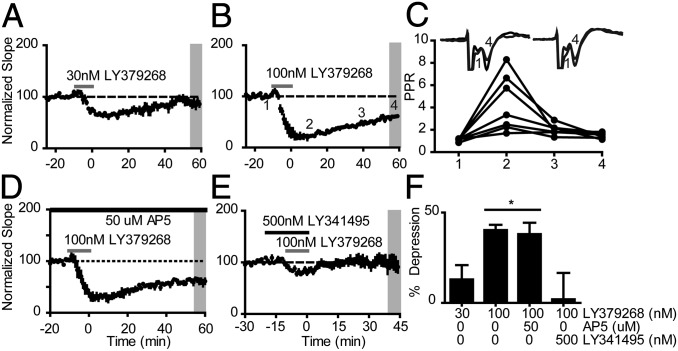
We recorded field excitatory postsynaptic potentials (fEPSPs) from layer V in response to stimulation of layer II/III in the prelimbic (PL) subregion of mPFC in ICR(CD1) mice. Application of the selective mGlu2/3 agonist LY379268 (30–100 nM for 10 min) produced a concentration-dependent, transient inhibition of the fEPSP slope (Fig. 1 A and B). Strong pharmacological activation of mGlu2/3 with 100 nM LY379268 produced LTD of fEPSPs up to 60 min after drug washout (40.3 ± 3.0% depression from baseline, n = 7; Fig. 1 B and F). Analysis of paired-pulse ratios (PPR) (25-ms interstimulus interval) throughout the experiment showed a phasic PPR increase corresponding to the peak of the transient inhibition, which then returned to baseline levels 60 min later when LTD was observed (Fig. 1C). This suggests that, although the initial fEPSP inhibition may have a presynaptic component, the observed LTD was not simply due to a long-term decrease in the neurotransmitter release probability. When experiments were performed in the presence of the N-methyl-d-aspartate (NMDA) receptor antagonist AP5 (50 μM), LTD was still observed (38.1 ± 6.3%, n = 5; Fig. 1 D and F). Conversely, when LY379268 was applied in the presence of group II antagonist LY341495 (500 nM), both the transient inhibition and induction of LTD were blocked (12.3 ± 4.6%, n = 6; Fig. 1 E and F), confirming this effect was solely due to actions at group II mGlu receptors. Thus, selective pharmacological activation of group II mGlu receptors produces an NMDA receptor-independent form of LTD of fEPSPs that is expressed postsynaptically.
Fig. 1.
Group II mGlu LTD in mouse mPFC. (A and B) Average time course of fEPSP slopes recorded from layer V mPFC. Application of LY379268 at 30 nM (n = 6) and 100 nM (n = 7) transiently decreased the slope, but induced LTD only at 100 nM. (C) Paired-pulse ratio analysis for fEPSPs recorded from all slices in B. x-axis labels correspond to the time-points Inset in B. Insets show sample paired-pulse fEPSP traces from baseline (1) and 60 min after drug washout (4). (D) LTD induced by LY379268 was not altered by the NMDA receptor antagonist AP5 (n = 5). (E) LTD was blocked by a selective mGlu2/3 orthosteric antagonist LY341495 (n = 6). (F) Quantification of LTD measured 55–60 min after drug washout (average of shaded region in A, B, D, and E). * indicates P < 0.05 Tukey posttest vs. 30 nM and 500 nM LY341495. Data are expressed as mean ± SEM.
Induction of Group II mGlu LTD Requires Activation of mGlu3.
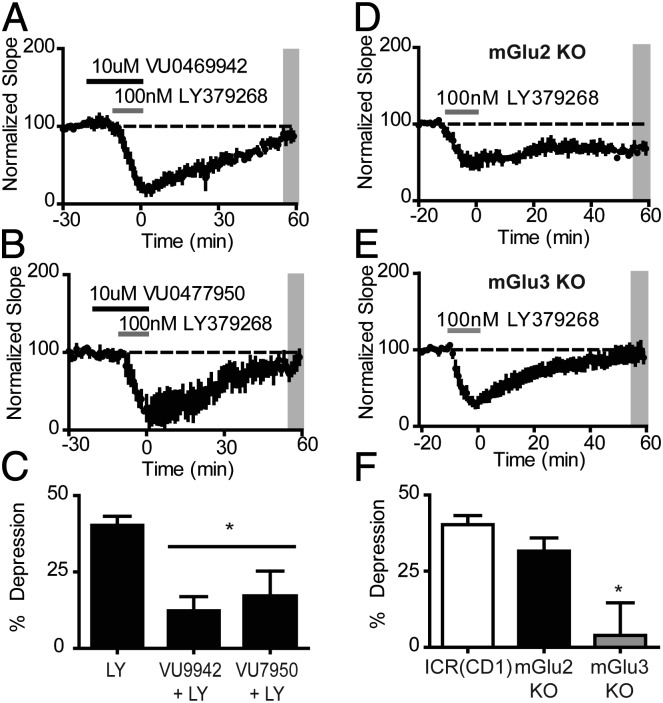
Next we sought to evaluate the contribution of the mGlu2 and mGlu3 subtypes to this form of LTD. We took advantage of two mGlu3-selective NAMs, VU0469942 and VU0477950, that we recently reported and characterized (24). When slices were pretreated with the mGlu3-selective NAM, VU0469942 (10 µM), the agonist LY379268 caused a large transient depression, but the slope returned to near baseline levels during the 60-min drug washout (12.3 ± 4.6%, n = 6, Fig. 2A). Thus, the mGlu3 NAM blocked the ability of LY379268 to induce LTD, but did not inhibit the acute inhibition of synaptic transmission. VU0477950 is a deuterated structural analog of VU0469942, which provides improved pharmacokinetics in terms of clearance relative to the nondeuterated compound (24). When experiments were repeated in the presence of VU0477950, a similar profile emerged (Fig. 2B). LY379268 caused a transient depression of the fEPSP slope, which then returned to baseline levels by the end of the experiment (16.9 ± 8.4%, n = 4). Compared with LY379268 alone (Fig. 2C), both VU0469942 and VU0477950 significantly decreased the magnitude of LTD measured 55–60 min after washout (P < 0.05; one-way ANOVA with a Tukey posttest).
Fig. 2.
mGlu3 activation is required for the induction of LTD by a group II agonist. (A and B) Preincubating slices with the mGlu3 NAMs VU0469942 (A, n = 6) and VU0477950 (B, n = 4) does not affect the transient inhibition of the fEPSP slope, but blocks LTD induced by LY379268. (C) Quantification of the effects of mGlu3 NAMs on LTD measured 55–60 min after drug washout (average of shaded region in A and B). * indicates P < 0.05 Tukey posttest vs. LY379268. (D and E) LY379268 induces LTD in mPFC slices from mGlu2 (n = 6) but not mGlu3 KO mice (n = 9). (F) Quantification of LTD in mGlu2 KO, mGlu3 KO, and the background strain ICR(CD1) (average of shaded region in D and E). * indicates P < 0.05 Tukey posttest vs. mGlu2 and ICR(CD1) mice. Data are expressed as mean ± SEM.
To further evaluate the role of the individual group II mGlu receptor subtypes, we compared LTD induced by LY379268 in mGlu2 and mGlu3 KO mice (Fig. 2 D and E). In mGlu2 KO mice agonist application induced a lasting depression of fEPSPs, indicative of LTD measured 60 min after drug washout (31.6 ± 4.3%, n = 5). In contrast, when LY379268 was applied to slices from mGlu3 KO mice, a transient depression was observed, but LTD was absent when assessed 60 min after washout of the agonist (4.0 ± 10.7%, n = 9). Compared with LTD measured in the ICR(CD1) background strain and mGlu2 KO mice (Fig. 2F), the magnitude of LTD was significantly smaller in mGlu3 KO mice (P < 0.05; Tukey posttest). Moreover, there was no difference between the magnitude of LTD observed in wild-type (WT) and mGlu2 KO mice (P > 0.05).
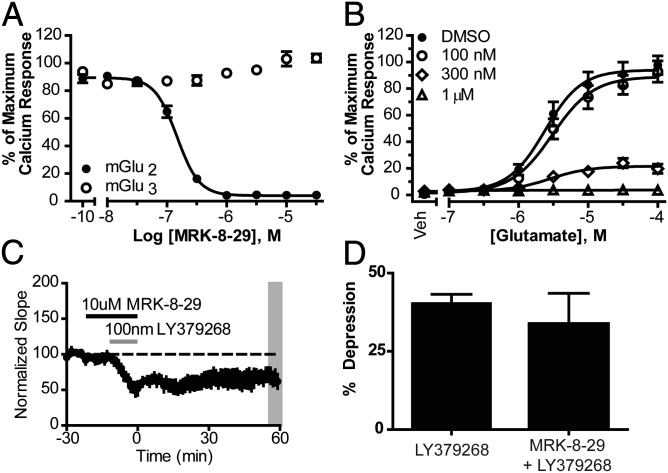
To further test the contribution of mGlu2 activation to this LTD response, we synthesized and characterized the reported mGlu2-selective NAM MRK-8-29 (25, 26). The synthesis of MRK-8-29 was completed in seven steps from commercially available 4,7-dichloroquinoline, using a modified version of the originally reported route (SI Materials and Methods). We then used a calcium-fluorescence assay to generate concentration-response curves for MRK-8-29 in stable cell lines expressing rat mGlu2 and rat mGlu3 receptors to assess the selectivity of the compound (SI Materials and Methods). Although MRK-8-29 did not alter the response of rat mGlu3 cells to an EC80 concentration of glutamate, it fully inhibited rat mGlu2 responses with an IC50 value of 146 nM (Fig. 3A). Furthermore, increasing concentrations of MRK-8-29 induced a progressive depression of the maximal efficacy of rat mGlu2 in response to glutamate, indicating an allosteric mechanism of inhibition (Fig. 3B). When LY379268 (100 nM) was applied to slices in the presence of MRK-8-29 (10 μM), there was a rapid and lasting depression of the fEPSP slope that was still evidenced 60 min after agonist washout, indicative of induction of LTD (33.7 ± 9.8%; n = 4; Fig. 3C). Compared with LY379268 alone, MRK-8-29 did not significantly affect the magnitude of LTD measured 60 min after agonist washout (Fig. 3D; P > 0.05; unpaired t test). Although the transient inhibition induced by LY379268 appeared to be reduced by MRK-8-29 relative to control and VU0469942 (Fig. S1A), this effect was not statistically significant (P > 0.05; one-way ANOVA). Likewise, the transient depression appeared to be attenuated in mGlu2 KO mice relative to WT and mGlu3 KO mice (Fig. S1B), and this effect also did not reach statistical significance (P > 0.05; one-way ANOVA). Taken together, these data indicate that whereas activation of mGlu2 plays an important role in the transient depression of synaptic transmission at this synapse, mGlu3 activation is required for the induction of group II mGlu LTD.
Fig. 3.
MRK-8-29 is a selective mGlu2 NAM. (A) Concentration-response curves for MRK-8-29 in the presence of an EC80 for glutamate at rat mGlu2 and rat mGlu3 receptors. (B) Progressive fold shift for MRK-8-29 at rat mGlu2 receptors. (C) Preincubation of slices with MRK-8-29 (n = 5) does not affect LTD induced by LY379268. (D) Quantification of the effects of MRK-8-29 on LTD at 55–60 min after drug washout (average of shaded region in C). Data are expressed as mean ± SEM.
Finally, to confirm that the actions of the mGlu3 NAM VU0469942 require activity at mGlu3, we evaluated the effects of this compound in slices prepared from mGlu2 and mGlu3 KO mice (Fig. S2 A–C). In control slices from both mGlu2 and mGlu3 KO mice, the agonist LY379268 induced a transient depression of fEPSPs (43.5 ± 9.3%, n = 6, and 66.4 ± 5.9%, n = 9, respectively). However, when LY379268 was applied in the presence of VU0469942, inhibition was almost completely attenuated in mGlu2 KO slices (11.3 ± 7.9%, n = 5), which express only mGlu3. In contrast, the mGlu3-selective NAM VU0469942 did not antagonize the effect of LY379268 in slices from mGlu3 KO mice (67.2 ± 1.9%, n = 3), in which case the agonist would be acting only on the mGlu2 subtype. These data confirm that the actions of VU0469942 require the expression of mGlu3 and are therefore mediated by selective inhibition of this receptor.
Postsynaptic mGlu3 Mediates Group II mGlu Agonist-Induced Ca2+ Elevations in Layer V Pyramidal Neurons in mPFC.
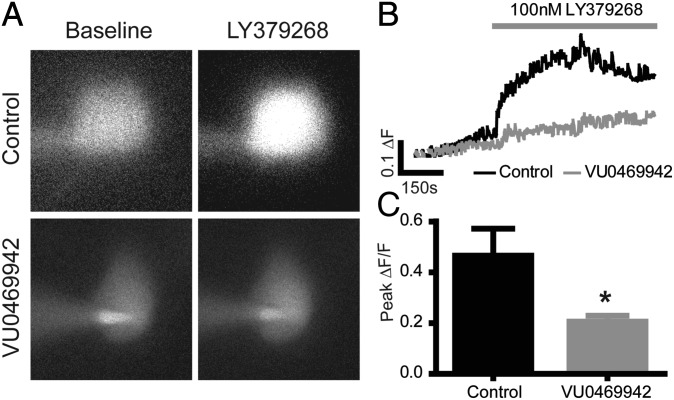
Previous reports and our analysis of PPRs suggest that group II LTD in mPFC is expressed postsynaptically. Furthermore, LTD is dependent upon intracellular Ca2+ mobilization induced by activation of group II mGlu receptors in layer V pyramidal cells (16). Based on our findings that LTD is dependent upon mGlu3 activation, we tested the hypothesis that Ca2+ signaling is downstream of mGlu3 by monitoring Ca2+ in individual layer V pyramidal neurons (Fig. 4 A–C). Cells were loaded with the Ca2+-sensitive dye, Fluo-4, through a patch pipette; experiments were performed in the presence of tetrodotoxin (TTX) (1 μM) to isolate postsynaptic receptor actions. In control experiments, when group II mGlu receptors were activated with LY379268 (100 nM for 10 min) there was an increase in fluorescence intensity relative to baseline (0.47 ± 0.11 ΔF/F peak), indicating an elevation in intracellular Ca2+. However, when these experiments were performed in the presence of the mGlu3 NAM, VU0469942 (10 μM), there was a significant reduction in the change in fluorescence (0.21 ± 0.024 ΔF/F peak; P < 0.05; unpaired t test). This is consistent with the hypothesis that group II agonists induce intracellular Ca2+ signals through activation of mGlu3 in layer V pyramidal neurons. Furthermore, these data suggest that postsynaptic mGlu3 is the critical site of action for induction of group II mGlu LTD.
Fig. 4.
Activation of postsynaptic mGlu3 increases intracellular Ca2+ in layer V pyramidal neurons. (A) Representative images demonstrating fluorescence levels during baseline (Left) and in the presence of LY379268 (Right). Top images are from a control experiment and Bottom images are from an experiment performed in the presence of the mGlu3 NAM VU0469942. (B) Time course of fluorescence measurements for the experiments shown in A. (C) Quantification and summary of Ca2+ imaging experiments (n = 7 per group). * indicates P < 0.05 unpaired t test vs. control. Data are expressed as mean ± SEM.
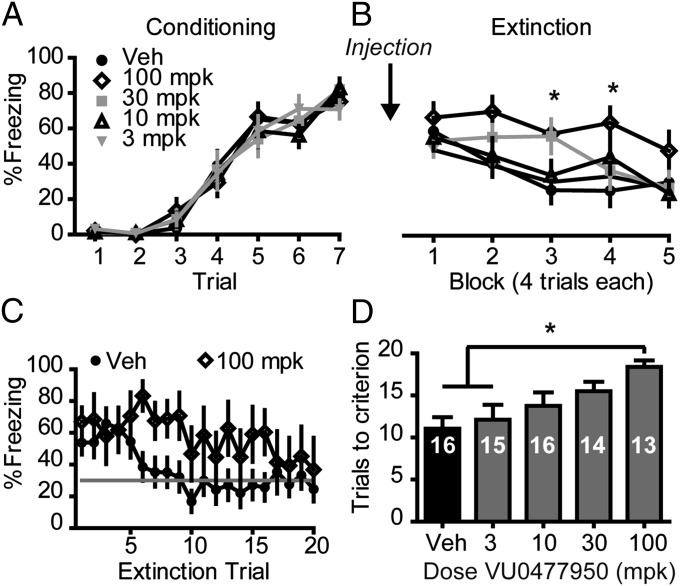
The mGlu3 NAM VU0477950 Impairs Fear Extinction Learning.
We next sought to investigate how mGlu3 is able to modulate fear extinction, a behavior that is dependent upon the integrity of the mPFC (27). On day 1, drug naive mice were conditioned by pairing a tone conditioned stimulus (CS) with a mild foot-shock unconditioned stimulus (US). After seven CS–US presentations, there was a significant increase in the amount of time spent freezing during the CS presentation across trials for all subjects (P < 0.0001), indicating all mice were conditioned to associate the tone with the foot shock (Fig. 5A). Twenty-four hours later, mice received an injection of vehicle or the mGlu3 NAM VU0477950 (3–100 mg/kg i.p.). Thirty minutes after injection, mice were placed in a new context and received 20 CS-alone presentations to evaluate initial cue memory and subsequent extinction learning (Fig. 5B). No effect of VU0477950 on initial cue memory (Fig. 4B and Fig. S3A) was observed as all mice had equivalent levels of freezing during the first block of CS-alone trials (P > 0.05). During subsequent CS presentations, vehicle-treated mice decreased freezing to an asymptotic level, a pattern of behavior consistent with extinction learning. However, in mice treated with the mGlu3 NAM, there was a dose-dependent impairment in extinction learning (P < 0.05, block × dose interaction). Specifically, mice treated with a 30-mg/kg or a 100-mg/kg dose maintained high levels of freezing through blocks 3 and 4, which reached significance in the 100-mg/kg group relative to vehicle-treated animals (Bonferroni posttests; P < 0.05). We quantified the impact of VU0477950 on extinction learning by analyzing the number of trials required to reach criterion. The learning criterion was established by examining the performance of vehicle-treated animals across all extinction trials and determining asymptotic performance (Fig. 5C). On average, vehicle-treated mice achieved criterion learning of 30% freezing in ∼11 trials (Fig. 5D). Consistent with analysis of freezing across blocks of trials, there was a dose-dependent increase in the number of trials required to reach criterion in mice treated with VU0477950. Moreover, mice treated with the 100-mg/kg dose of the compound showed a significant increase in the number of trials to reach criterion compared with mice in the vehicle and 3-mg/kg groups (P < 0.05; Tukey posttest). Interestingly, there appeared to be no impairment in memory retrieval for extinction in animals treated with 30 mg/kg or 100 mg/kg relative to vehicle-treated animals when measured 24 h later in a second extinction session (Fig. S3 B and C). Furthermore, increased levels of freezing could not simply be attributed to general motor suppression (Fig. S3D) or an anxiogenic effect induced by VU0477950 (Fig. S4 A–D), as demonstrated by no change in open field activity.
Fig. 5.
An mGlu3 NAM impairs fear extinction learning in mice. (A) On day 1, mice were fear conditioned with 7 paired CS–US trials. No drug was administered on the conditioning day. (B) On day 2, mice received an i.p. injection of vehicle or the mGlu3 NAM VU0477950 (3–100 mg/kg) and then were trained on fear extinction with 20 CS-alone trials. Data are presented as mean freezing across 4 consecutive trials. * indicates P < 0.05, 100-mg/kg Bonferroni posttest vs. vehicle-treated mice. (C) Extinction learning criterion was established by examining asymptotic learning (shaded horizontal line) across all 20 CS-alone trials in vehicle-treated animals. Freezing behavior of mice treated with 100 mg/kg is plotted for comparison. (D) Quantification of the learning impairment induced by VU0477950 as measured by trials to criterion. Numbers within the individual bars indicate number of mice within the respective group. * indicates P < 0.05 Tukey posttest vs. vehicle and 3-mg/kg–treated mice. Data are expressed as mean ± SEM.
Discussion
We have demonstrated that mGlu3 plays a critical role in the regulation of mPFC neuroplasticity and is required for a specific learned behavior that is dependent upon the integrity of this brain region. In agreement with reports from rat brain slices (13–16), we found that strong pharmacological activation of group II mGlu receptors results in LTD of fEPSPs recorded in layer V mPFC. The initial transient depression of the fEPSP slope was accompanied by a robust increase in the PPR, suggesting a presynaptic modulation of neurotransmitter release, which is a well-known function of group II mGlu receptors, and especially mGlu2 (28). One hour after the maximal transient depression, when we quantified LTD, the PPR had returned to baseline levels, suggesting the expression of lasting depression of synaptic transmission was likely mediated by a postsynaptic mechanism. This is consistent with previous studies showing postsynaptic actions of group II mGlu receptor agonists in mPFC pyramidal cells (15, 16) and with our finding that activation of mGlu3 induces calcium transients in these cells. Furthermore, under our experimental conditions this LTD does not require activation of NMDA receptors, as the magnitude was unaffected by an NMDA receptor antagonist.
To delineate the roles the mGlu2 and mGlu3 receptor subtypes, we used newly identified mGlu2 or mGlu3 NAMs, as well as mGlu2 and mGlu3 receptor KO mice. Although the selective mGlu2 NAM MRK-8-29 slightly reduced the magnitude of the transient depression, it did not prevent the induction of LTD. In contrast, mGlu3 NAMs VU0469942 and VU0477950 completely blocked induction of LTD by the group II mGlu receptor agonist, but were without effect on the transient depression of fEPSPs. Similarly, LTD was observed in slices prepared from mGlu2, but not mGlu3, KO mice, whereas acute depression of synaptic transmission was intact in slices from mGlu3 KO mice. Taken together, these results provide strong evidence that activation of mGlu2 can induce transient depression of synaptic transmission in mPFC neurons, a response to mGlu2 activation that has been established at multiple other synapses (19–23). However, although mGlu2 can regulate transmission at this synapse, these data also reveal that activation of mGlu3 is required for the induction of LTD in mPFC pyramidal cells. In contrast to presynaptic effects of mGlu2 receptor activation at this and other synapses, our data suggest an important role for postsynaptically localized mGlu3 in induction of LTD. Also, similar to mGlu5-mediated LTD in the hippocampus, it is likely that maintenance of LTD at this synapse is mediated by postsynaptic mechanisms. Thus, in agreement with previous studies (16), we found that a selective mGlu2/3 agonist increases intracellular Ca2+ in layer V pyramidal cells and now show that this response is mediated by mGlu3. This effect is likely due to direct actions of the group II agonist on the postsynaptic neuron, as the experiments were performed in the presence of TTX. Although the exact mechanism by which mGlu3, a Gi/o coupled receptor, induces intracellular Ca2+ increases is unknown, similar effects of group II mGlu receptor agonists on intracellular Ca2+ are observed in hippocampal CA3 pyramidal cell and interneuron populations, and these responses are thought to be mediated by activation of mGlu3 (29). In addition, there are examples of other Gi/o coupled receptors inducing intracellular Ca2+ elevations (30). Overall, the results from our electrophysiology and imaging studies provide strong support for a critical role of postsynaptic mGlu3 signaling in the induction of LTD in mPFC.
The finding that activation of mGlu3 is required for induction of a form of synaptic plasticity in the mPFC is especially important in light of extensive studies demonstrating a central role of the mPFC in multiple domains of cognitive function and previous genetic studies implicating mGlu3 in aspects of cognitive function that require integrity of this cortical region. Based on this, it is possible that mGlu3-mediated LTD in the mPFC could be important for some aspects of mPFC-dependent cognition. Interestingly, previous studies suggest that intact functioning of the mPFC and especially regulation of excitatory inputs to the PFC from paralimbic regions (27, 31, 32) are central for fear extinction learning. Thus, our finding that the selective mGlu3 NAM VU0477950 induced a dose-dependent increase in the number of trials required to extinguish fear responses is consistent with a possible role of mGlu3 in this specific form of prefrontal cortical-dependent cognitive function. The highest dose of the mGlu3 NAM tested nearly doubled the number of trials needed to reach the extinction criterion. Furthermore, some animals failed to reach criterion after the maximum number of cues given, suggesting a major role for mGlu3 in the process of acquisition of extinction learning. Interestingly, there was no difference in the amount of freezing during the initial cue presentations, signifying that the mGlu3 NAM did not create a heightened fear state, which is corroborated by our data indicating that the mGlu3 NAM was not anxiogenic in an open-field assay. Furthermore, there was no difference in retrieval of the extinction memory assessed 24 h posttraining, despite the robust learning delay.
Importantly, the doses of VU0477950 used for these studies were based on extensive pharmacokinetic studies that revealed that these doses lead to free brain concentrations that are estimated to be in the range of those required to inhibit mGlu3 but well below concentrations tested for selectivity against other mGlu receptor subtypes. The relatively high doses required to achieve these concentrations in the CNS and for behavioral efficacy are related to the rapid clearance and high plasma protein binding of VU0477950, which limit the amount of free drug available to bind to the target (24). Estimates of unbound brain concentrations are based on precise measures of total brain concentrations, which are then corrected for in vitro measures of plasma and brain homogenate binding and do not provide a definitive measure of the actual concentration achieved at the receptor site. Thus, it is impossible to directly estimate the level of receptor occupancy achieved in the CNS with the doses used. However, it is also possible that in vivo efficacy requires high mGlu3 receptor occupancy, a property that has been reported for mGlu1 and mGlu5 NAMs (33, 34). In contrast, mGlu receptor positive allosteric modulators (PAMs) can produce full efficacy with relatively low occupancy in the CNS because of the contributions of both affinity and cooperativity to PAM potency at a receptor (35, 36). Unfortunately, there are currently no selective mGlu3 radioligands that would allow us to measure receptor occupancy of VU0477950. In the future, development of radiolabeled compounds that can be used with positron emission tomography (PET) and in vivo radioligand binding studies will be crucial for assessing the level of mGlu3 occupancy and blockade required for different behavioral responses to mGlu3 NAMs.
The fear extinction circuit is composed of reciprocal interactions between the mPFC subregions and the amygdala and hippocampus (27, 31, 32, 37). In vivo recordings have demonstrated that activity within the PL cortex, the region of the mPFC where we studied LTD, is correlated with freezing behavior during fear extinction (32, 38–40). During states of high fear and freezing, neurons within the PL will display robust firing in response to fear cues, such as a tone CS. Recent work has demonstrated that this pattern of activity likely reflects excitatory drive from the amygdala to the PL subregion of mPFC (31, 41). Moreover, PL neurons provide reinforcing feedback by sending robust projections to the amygdala, which may help drive freezing behavior. Furthermore, as the animals successfully extinguish fear responding, CS-induced firing of mPFC/PL neurons diminishes (38–40). Our data raise the intriguing possibility that this lasting depression of CS-induced firing in this mPFC subregion is an mGlu3-dependent process and could reflect a lasting depression of transmission from amygdala afferents. However, at present, it is not known whether the mGlu3-dependent LTD established here reflects depression of transmission at this specific synapse. Group II mGlu-mediated LTD of cortical inputs to the amygdala has been reported (42). Thus, it is also possible that the mGlu3 NAM could be acting within the amygdala to prolong freezing and impair extinction learning. However, activation of either mGlu2 or mGlu3 alone appears to be sufficient for the induction of LTD at this synapse (42); therefore selective attenuation of mGlu3 signaling is likely to have a minimal physiological effect. Future studies using complementary in vivo and in vitro methods making use of the newly available tools will help provide more mechanistic information about the role of mGlu3 in this form of mPFC-dependent cognitive function.
In summary, our data help define the role of mGlu3 in the regulation of prefrontal cortical function. Together with recent reports of working memory deficits in mGlu3 KO mice (43, 44), the effects of mGlu3 NAMs suggest that mGlu3 plays an important role in certain PFC-dependent behaviors. Additionally, allelic variations in GRM3, the human gene encoding mGlu3, have been reported to affect prefrontal activity and cognitive performance in healthy human subjects (45) and several studies have found associations between mutations in GRM3 and psychiatric disorders (1, 2, 4, 6–8). If these mutations are found to lead to a loss of mGlu3 function, this would suggest that selective positive allosteric modulators of mGlu3 may represent a novel therapeutic strategy for enhancing prefrontal function in patients.
Materials and Methods
Animals.
All animal studies were approved by the Vanderbilt University Medical Center Institutional Animal Care and Use Committee and were conducted in accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals (46). Male ICR (CD1) (Harlan Laboratories) mice were used in electrophysiology (4–8 wk old) and behavioral studies (6–7 wk old). Male mGlu2 and mGlu3 KO mice (6–12 wk old; gift from Eli Lilly and Company) were also used for electrophysiology studies.
Electrophysiology.
Coronal slices through the mPFC (300–400 μm) were prepared from ICR(CD1), mGlu2, and mGlu3 knockout mice with a vibrating microtome (VT1200s; Leica). After anesthesia with a mixture of ketamine and xylazine (100 mg/kg and 10 mg/kg i.p.), mice were perfused with a 4 °C sucrose-based cutting buffer containing 230 mM sucrose, 2.5 mM KCl, 10 mM MgSO4, 0.5 mM CaCl2, 1.25 mM NaH2PO4, 10 mM glucose, 26 mM NaHCO3, and 0.5 mM sodium ascorbate. Brain slices were then incubated at 32 °C for 12–15 min in an N-methyl-d-glucamine (NMDG)-based recovery solution as previously described (47) and then transferred to a holding chamber with artificial cerebrospinal fluid (aCSF) containing 126 mM NaCl, 2.5 mM KCl, 1 mM MgSO4, 2 mM CaCl2, 1.25 mM NaH2PO4, 10 mM glucose, 26 mM NaHCO3, 5 mM sodium ascorbate, and 12 mM N-acetylcystine. Recording aCSF was identical aside from the exclusion of sodium ascorbate and N-acetylcystine. fEPSPs were recorded from layer V of PL, using a pulled-glass pipette (3–5 MΩ), and evoked by electrical stimulation of layer II/III (0.05 Hz), using a concentric bipolar electrode. Three consecutive fEPSP slopes were averaged and then normalized to the mean baseline slope before drug application. LTD was measured as the average slope across the last 5 min of the recording session.
For calcium (Ca2+) imaging experiments, individual neurons in layer V of mPFC exhibiting properties of pyramidal cells were loaded with the indicator dye Fluo-4, Pentapotassium Salt, cell impermeant (Life Technologies), through a glass patch pipette as previously described (48). Detailed information for the Ca2+ imaging experiments can be found in SI Materials and Methods.
Behavioral Studies.
Behavioral studies were conducted on wild-type (WT) male ICR(CD1) mice. Before behavioral experiments, all animals were habituated to handling, transportation procedures, and injections for 2 consecutive days. Mice were fear conditioned with seven pairings of a tone CS (3.5 kHz, 80 dB, 30 s) with mild foot-shock US (2 s, 0.6 mA). Mice were returned to their home cages. Twenty-four hours after fear conditioning, CS retrieval and extinction learning were assessed with 20 CS-alone trials (5-s intertrial interval). To limit the effects of contextual conditioning, mice were fear conditioned in a round-walled, metal bar-floored chamber that was scented with 10% (vol/vol) vanilla extract odor and housed in a room with white ceiling lights. Extinction training occurred in a square-walled, solid-floored chamber that was scented with 10% (vol/vol) peppermint and housed in a room with red ceiling lights. Mice were dosed with vehicle or mGlu3 NAM VU0477950 (3–100 mg/kg) via i.p. injection 30 min before extinction training. Freezing behavior defined as the absence of movement other than respiration was used to measure fear and was quantified by computer video analysis software (Video Freeze; Med Associates). For locomotion experiments, mice were injected with vehicle or VU0477950 (100 mg/kg) i.p. and placed in an open field chamber equipped with infrared beams (Med Associates) to monitor motor activity for 1 h.
Please see SI Materials and Methods for information on drugs, statistical analysis, chemistry methodology, and details of the cell line Ca2+ mobilization assay.
Supplementary Material
Acknowledgments
This work was supported by a grant from the National Institute of Neurological Disease and Stroke (NS031373) (to P.J.C.), a postdoctoral fellowship from the Pharmaceutical Research and Manufacturers of America (PhRMA) Foundation (to A.G.W.), and a National Institutes of Health predoctoral Training Grant (T32 GM007628) (to C.J.W.).
Footnotes
Conflict of interest statement: P.J.C., C.W.L., C.M.N., and K.A.E. receive research support that includes salary support from Bristol-Myers Squibb and Astrazeneca. P.J.C., C.W.L., C.M.N., and K.A.E. are inventors on multiple composition of matter patents protecting allosteric modulators of G protein couple receptors (GPCRs).
*This Direct Submission article had a prearranged editor.
This article contains supporting information online at www.pnas.org/lookup/suppl/doi:10.1073/pnas.1416196112/-/DCSupplemental.
References
- 1.Egan MF, et al. Variation in GRM3 affects cognition, prefrontal glutamate, and risk for schizophrenia. Proc Natl Acad Sci USA. 2004;101(34):12604–12609. doi: 10.1073/pnas.0405077101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Harrison PJ, Lyon L, Sartorius LJ, Burnet PW, Lane TA. The group II metabotropic glutamate receptor 3 (mGluR3, mGlu3, GRM3): Expression, function and involvement in schizophrenia. J Psychopharmacol. 2008;22(3):308–322. doi: 10.1177/0269881108089818. [DOI] [PubMed] [Google Scholar]
- 3.Tan H-Y, et al. Epistasis between catechol-O-methyltransferase and type II metabotropic glutamate receptor 3 genes on working memory brain function. Proc Natl Acad Sci USA. 2007;104(30):12536–12541. doi: 10.1073/pnas.0610125104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Schizophrenia Working Group of the Psychiatric Genomics Consortium Biological insights from 108 schizophrenia-associated genetic loci. Nature. 2014;511(7510):421–427. doi: 10.1038/nature13595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Kandaswamy R, et al. Genetic association, mutation screening, and functional analysis of a Kozak sequence variant in the metabotropic glutamate receptor 3 gene in bipolar disorder. JAMA Psychiatry. 2013;70(6):591–598. doi: 10.1001/jamapsychiatry.2013.38. [DOI] [PubMed] [Google Scholar]
- 6.O’Brien NL, et al. 2014. The functional GRM3 Kozak sequence variant rs148754219 affects the risk of schizophrenia and alcohol dependence as well as bipolar disorder. Psychiatr Genet 24(6):277–278.
- 7.Enoch MA, et al. 2014. Expression of glutamatergic genes in healthy humans across 16 brain regions; altered expression in the hippocampus after chronic exposure to alcohol or cocaine. Genes Brain Behav 13(8):758–768.
- 8.Xia Y, et al. Association of single-nucleotide polymorphisms in a metabotropic glutamate receptor GRM3 gene subunit to alcohol-dependent male subjects. Alcohol Alcohol. 2014;49(3):256–260. doi: 10.1093/alcalc/agu004. [DOI] [PubMed] [Google Scholar]
- 9.Ghose S, et al. Metabotropic glutamate receptor 2 and 3 gene expression in the human prefrontal cortex and mesencephalon in schizophrenia. Int J Neurosci. 2008;118(11):1609–1627. doi: 10.1080/00207450802330702. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Lewis DA. Cortical circuit dysfunction and cognitive deficits in schizophrenia—implications for preemptive interventions. Eur J Neurosci. 2012;35(12):1871–1878. doi: 10.1111/j.1460-9568.2012.08156.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Noël X, Brevers D, Bechara A. A neurocognitive approach to understanding the neurobiology of addiction. Curr Opin Neurobiol. 2013;23(4):632–638. doi: 10.1016/j.conb.2013.01.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Price JL, Drevets WC. Neural circuits underlying the pathophysiology of mood disorders. Trends Cogn Sci. 2012;16(1):61–71. doi: 10.1016/j.tics.2011.12.011. [DOI] [PubMed] [Google Scholar]
- 13.Huang CC, Hsu KS. The role of NMDA receptors in regulating group II metabotropic glutamate receptor-mediated long-term depression in rat medial prefrontal cortex. Neuropharmacology. 2008;54(7):1071–1078. doi: 10.1016/j.neuropharm.2008.02.013. [DOI] [PubMed] [Google Scholar]
- 14.Huang CC, Yang PC, Lin HJ, Hsu KS. Repeated cocaine administration impairs group II metabotropic glutamate receptor-mediated long-term depression in rat medial prefrontal cortex. J Neurosci. 2007;27(11):2958–2968. doi: 10.1523/JNEUROSCI.4247-06.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Otani S, Auclair N, Desce JM, Roisin MP, Crépel F. Dopamine receptors and groups I and II mGluRs cooperate for long-term depression induction in rat prefrontal cortex through converging postsynaptic activation of MAP kinases. J Neurosci. 1999;19(22):9788–9802. doi: 10.1523/JNEUROSCI.19-22-09788.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Otani S, Daniel H, Takita M, Crépel F. Long-term depression induced by postsynaptic group II metabotropic glutamate receptors linked to phospholipase C and intracellular calcium rises in rat prefrontal cortex. J Neurosci. 2002;22(9):3434–3444. doi: 10.1523/JNEUROSCI.22-09-03434.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Benneyworth MA, et al. A selective positive allosteric modulator of metabotropic glutamate receptor subtype 2 blocks a hallucinogenic drug model of psychosis. Mol Pharmacol. 2007;72(2):477–484. doi: 10.1124/mol.107.035170. [DOI] [PubMed] [Google Scholar]
- 18.Galici R, et al. Biphenyl-indanone A, a positive allosteric modulator of the metabotropic glutamate receptor subtype 2, has antipsychotic- and anxiolytic-like effects in mice. J Pharmacol Exp Ther. 2006;318(1):173–185. doi: 10.1124/jpet.106.102046. [DOI] [PubMed] [Google Scholar]
- 19.Hermes ML, Renaud LP. Postsynaptic and presynaptic group II metabotropic glutamate receptor activation reduces neuronal excitability in rat midline paraventricular thalamic nucleus. J Pharmacol Exp Ther. 2011;336(3):840–849. doi: 10.1124/jpet.110.176149. [DOI] [PubMed] [Google Scholar]
- 20.Johnson KA, Niswender CM, Conn PJ, Xiang Z. Activation of group II metabotropic glutamate receptors induces long-term depression of excitatory synaptic transmission in the substantia nigra pars reticulata. Neurosci Lett. 2011;504(2):102–106. doi: 10.1016/j.neulet.2011.09.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Kew JN, Pflimlin MC, Kemp JA, Mutel V. Differential regulation of synaptic transmission by mGlu2 and mGlu3 at the perforant path inputs to the dentate gyrus and CA1 revealed in mGlu2 -/- mice. Neuropharmacology. 2002;43(2):215–221. doi: 10.1016/s0028-3908(02)00084-9. [DOI] [PubMed] [Google Scholar]
- 22.Poisik O, Marino M, Smith Y, Conn PJ. D1- and D2-like dopamine receptors regulate signaling properties of group I metabotropic glutamate receptors in the rat globus pallidus. Eur J Neurosci. 2007;26(4):852–862. doi: 10.1111/j.1460-9568.2007.05710.x. [DOI] [PubMed] [Google Scholar]
- 23.Yokoi M, et al. Impairment of hippocampal mossy fiber LTD in mice lacking mGluR2. Science. 1996;273(5275):645–647. doi: 10.1126/science.273.5275.645. [DOI] [PubMed] [Google Scholar]
- 24.Wenthur CJ, et al. Discovery of (R)-(2-fluoro-4-((-4-methoxyphenyl)ethynyl)phenyl) (3-hydroxypiperidin-1-yl)methanone (ML337), an mGlu3 selective and CNS penetrant negative allosteric modulator (NAM) J Med Chem. 2013;56(12):5208–5212. doi: 10.1021/jm400439t. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Celanire S, et al. Novel metabotropic glutamate receptor 2/3 antagonists and their therapeutic applications: A patent review (2005 - present) Expert Opin Ther Pat. 2014;0(0):1–22. doi: 10.1517/13543776.2014.983899. [DOI] [PubMed] [Google Scholar]
- 26. Merck Sharp & Dohme Corp. (2012) International Patent Application PCT/US2012/06202.
- 27.Sotres-Bayon F, Quirk GJ. Prefrontal control of fear: More than just extinction. Curr Opin Neurobiol. 2010;20(2):231–235. doi: 10.1016/j.conb.2010.02.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Marek GJ. Metabotropic glutamate2/3 (mGlu2/3) receptors, schizophrenia and cognition. Eur J Pharmacol. 2010;639(1–3):81–90. doi: 10.1016/j.ejphar.2010.02.058. [DOI] [PubMed] [Google Scholar]
- 29.Ster J, et al. Enhancement of CA3 hippocampal network activity by activation of group II metabotropic glutamate receptors. Proc Natl Acad Sci USA. 2011;108(24):9993–9997. doi: 10.1073/pnas.1100548108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Murthy KS, Zhou H, Huang J, Pentyala SN. 2004. Activation of PLC-δ1 by Gi/o-coupled receptor agonists. Am J Physiol Cell Physiol 287(6):C1679–C1687.
- 31.Senn V, et al. Long-range connectivity defines behavioral specificity of amygdala neurons. Neuron. 2014;81(2):428–437. doi: 10.1016/j.neuron.2013.11.006. [DOI] [PubMed] [Google Scholar]
- 32.Sotres-Bayon F, Sierra-Mercado D, Pardilla-Delgado E, Quirk GJ. Gating of fear in prelimbic cortex by hippocampal and amygdala inputs. Neuron. 2012;76(4):804–812. doi: 10.1016/j.neuron.2012.09.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Michalon A, et al. Chronic pharmacological mGlu5 inhibition corrects fragile X in adult mice. Neuron. 2012;74(1):49–56. doi: 10.1016/j.neuron.2012.03.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Suzuki G, et al. Correlation of receptor occupancy of metabotropic glutamate receptor subtype 1 (mGluR1) in mouse brain with in vivo activity of allosteric mGluR1 antagonists. J Pharmacol Sci. 2009;110(3):315–325. doi: 10.1254/jphs.09011fp. [DOI] [PubMed] [Google Scholar]
- 35.Conn PJ, Lindsley CW, Meiler J, Niswender CM. Opportunities and challenges in the discovery of allosteric modulators of GPCRs for treating CNS disorders. Nat Rev Drug Discov. 2014;13(9):692–708. doi: 10.1038/nrd4308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Rook JM, et al. 2014. Relationship between in vivo receptor occupancy and efficacy of metabotropic glutamate receptor subtype 5 allosteric modulators with different in vitro binding profiles. Neuropsychopharmacology 40(3):755–765.
- 37.Amano T, Duvarci S, Popa D, Paré D. The fear circuit revisited: Contributions of the basal amygdala nuclei to conditioned fear. J Neurosci. 2011;31(43):15481–15489. doi: 10.1523/JNEUROSCI.3410-11.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Burgos-Robles A, Vidal-Gonzalez I, Quirk GJ. Sustained conditioned responses in prelimbic prefrontal neurons are correlated with fear expression and extinction failure. J Neurosci. 2009;29(26):8474–8482. doi: 10.1523/JNEUROSCI.0378-09.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Walker AG, Ummel JR, Rebec GV. Reduced expression of conditioned fear in the R6/2 mouse model of Huntington’s disease is related to abnormal activity in prelimbic cortex. Neurobiol Dis. 2011;43(2):379–387. doi: 10.1016/j.nbd.2011.04.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Wilber AA, et al. Chronic stress alters neural activity in medial prefrontal cortex during retrieval of extinction. Neuroscience. 2011;174:115–131. doi: 10.1016/j.neuroscience.2010.10.070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Pendyam S, et al. Fear signaling in the prelimbic-amygdala circuit: A computational modeling and recording study. J Neurophysiol. 2013;110(4):844–861. doi: 10.1152/jn.00961.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Lucas SJ, Bortolotto ZA, Collingridge GL, Lodge D. Selective activation of either mGlu2 or mGlu3 receptors can induce LTD in the amygdala. Neuropharmacology. 2013;66(0):196–201. doi: 10.1016/j.neuropharm.2012.04.006. [DOI] [PubMed] [Google Scholar]
- 43.Fujioka R, et al. Comprehensive behavioral study of mGluR3 knockout mice: Implication in schizophrenia related endophenotypes. Mol Brain. 2014;7:31. doi: 10.1186/1756-6606-7-31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Lainiola M, Procaccini C, Linden A-M. mGluR3 knockout mice show a working memory defect and an enhanced response to MK-801 in the T- and Y-maze cognitive tests. Behav Brain Res. 2014;266(0):94–103. doi: 10.1016/j.bbr.2014.03.008. [DOI] [PubMed] [Google Scholar]
- 45.Jablensky A, et al. Polymorphisms associated with normal memory variation also affect memory impairment in schizophrenia. Genes Brain Behav. 2011;10(4):410–417. doi: 10.1111/j.1601-183X.2011.00679.x. [DOI] [PubMed] [Google Scholar]
- 46. National Research Council Committee for the Update of the Guide for the Care and Use of Laboratory Animals (2001) Guide for the Care and Use of Laboratory Animals (National Academies Press, Washington, DC), 8th Ed.
- 47.Zhao S, et al. Cell type–specific channelrhodopsin-2 transgenic mice for optogenetic dissection of neural circuitry function. Nat Methods. 2011;8(9):745–752. doi: 10.1038/nmeth.1668. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Foster DJ, et al. M5 receptor activation produces opposing physiological outcomes in dopamine neurons depending on the receptor’s location. J Neurosci. 2014;34(9):3253–3262. doi: 10.1523/JNEUROSCI.4896-13.2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.