Abstract
Purpose
Cultured tumor fragments from melanoma metastases have been used for years as a source of tumor-infiltrating lymphocytes (TIL) for adoptive cell therapy. The expansion of tumor-reactive CD8+ T cells with IL-2 in these early cultures is critical in generating clinically active TIL infusion products, with a population of activated 4-1BB CD8+ T cells recently found to constitute the majority of tumor-specific T cells.
Experimental Design
We used an agonistic anti-4-1BB antibody added during the initial tumor fragment cultures to provide in situ 4-1BB co-stimulation.
Results
We found that addition of an agonistic anti-4-1BB antibody could activate 4-1BB signaling within early cultured tumor fragments and accelerated the rate of memory CD8+ TIL outgrowth that were highly enriched for melanoma antigen specificity. This was associated with NFκB activation and the induction of T-cell survival and memory genes, as well as enhanced IL-2 responsiveness, in the CD8+ T cells in the fragments and emerging from the fragments. Early provision of 4-1BB co-stimulation also affected the dendritic cells (DC) by activating NFκB in DC and promoting their maturation inside the tumor fragments. Blocking HLA class I prevented the enhanced outgrowth of CD8+ T cells with anti-4-1BB, suggesting that an ongoing HLA class I-mediated antigen presentation in early tumor fragment cultures plays a role in mediating tumor-specific CD8+ TIL outgrowth.
Conclusions
Our results highlight a previously unrecognized concept in TIL adoptive cell therapy that the tumor microenvironment can be dynamically regulated in the initial tumor fragment cultures to regulate the types of T cells expanded and their functional characteristics.
Keywords: 4-1BB, co-stimulation, tumor-infiltrating lymphocyte, melanoma, immunotherapy
Introduction
Adoptive transfer using tumor-infiltrating lymphocytes (TIL) has emerged as a powerful immunotherapy against solid malignancies, especially metastatic melanoma [1, 2]. ACT involves the initial outgrowth of TILs using IL-2 from 4–6 mm2 cut fragments from metastatic melanoma surgical resections [1–3]. Initial tumor fragment cultures take about 4–5 weeks to yield enough TIL for secondary expansion. A non-myeloablative lymphodepleting chemotherapy regimen before adoptive transfer of autologous TIL has boosted clinical response rates to 45–50% [4, 5].
CD8+ TIL are critical in mediating tumor regression in a number of Phase II TIL trials. Current efforts are aimed to increase the expansion of CD8+ T cells from tumor tissue, and increase the anti-tumor activity of the final TIL infusion product to improve persistence after adoptive transfer [2, 3, 6–9]. Accelerating the rate of TIL expansion from the initial tumor fragment cultures has become priority [6, 10–12]. Metastatic melanomas contain a population of CD8+ T cells expressing immunomodulatory markers, such as PD-1 and 4-1BB, indicating a recent history of antigenic stimulation in the tumor microenvironment in vivo [13, 14]. CD8+ TIL expressing 4-1BB appear to represent the most highly enriched tumor-specific sub-population of T cells in melanoma [13]. Protocols are being developed to purify 4-1BB+ CD8+ T cells from melanoma tissues and expand these selected cells for infusion. Although this approach is promising, it has caveats, including the need to prepare single cell suspensions from tumor tissues, the small sizes of tumor tissue that can be available yielding few cells after enzymatic or mechanical disaggregation, and the possibility that not all tumor-specific CD8+ T cells may be in an activated (4-1BB+) state at the time the tumor is processed.
An alternative approach is to directly manipulate co-stimulatory pathways within the initial melanoma tumor fragment cultures. This approach capitalizes on the de novo expression of co-stimulatory molecules due to previous antigenic stimulation on resident CD8+ T cells which can accelerate the rate of TIL expansion out of the tumor fragments. Tumor fragments have been used for years to expand TIL by adding exogenous IL-2, but the inclusion of other immunomodulators in tumor fragment cultures to affect TIL expansion and phenotype has not been investigated.
In this study, we hypothesized that the activation of the 4-1BB co-stimulatory pathway in melanoma tumor fragments enhances CD8+ T-cell output, TIL tumor reactivity, and memory properties. This question is distinct from our previous studies, where the effects of 4-1BB agonists were tested at much later stages of ex vivo TIL expansion where 4-1BB co-stimulation improved output and function of T cells in the rapid expansion protocol (REP) and the survival of the post-REP TIL [15, 16]. We tested an agonistic anti-4-1BB antibody, added during the initiation of individual tumor fragment cultures (at the start of the whole TIL expansion process) and found that this increased the rate of CD8+ TIL expansion as well as the tumor reactivity of the expanded product. 4-1BB co-stimulation during these early tumor fragment cultures induced the expression of survival signaling pathways (NFκB) in CD8+ TIL, and the expression of anti-apoptotic and T-cell memory genes. We examined potential mechanisms of action and found that resident dendritic cells (DC) in the tumor fragments survive for considerable periods of time and express 4-1BB. These tumor fragment resident DC also activate NFκB, and up-regulate certain maturation markers in combination with 4-1BB agonism. We examined whether ongoing HLA class I antigen presentation occurs in the early tumor fragment cultures that may enhance the output of CD8+ TIL. Addition of a blocking anti-HLA class I antibody reduced the output of CD8+ TIL, suggesting that continual antigen presentation occurs ex vivo in these early tumor fragment cultures that was not considered before. Our results indicate that tumor fragments placed in culture to expand TIL for adoptive cell therapy are not static material, but small, dynamic tumor microenvironments that can be manipulated to alter the yield and phenotype of TIL being expanded for cell therapy as well as enrich for tumor reactivity and improved memory phenotype. The use of 4-1BB co-stimulation in this system can be the first of many ways to manipulate these ex vivo tumor microenvironments to develop protocols to expand optimally enhanced TIL for adoptive cell therapy.
Materials and Methods
Agonistic anti-4-1BB antibody
A fully-human IgG4 monoclonal agonistic anti-4-1BB antibody (mAb) (BMS 663513 Lot 6A20383/1187261) was provided by Bristol Myers Squibb. The anti-4-1BB antibody was added at day 0 of the fragment set up and each time the TIL were fed. Tumor fragments were cultured in the presence of 10 µg/ml anti-human PD-1 antibody (BMS 936558), anti-human PD-L1 antibody (cloneM1H1) (eBioscience, San Diego, CA), or 10 µg/ml anti-human CTLA-4 antibody (Ipilimumab, BMS) in some experiments.
TIL isolation and culture from melanoma tumors
Studies were performed under approved Institutional Review Board (IRB) laboratory protocols at MD Anderson Cancer Center (MDACC) and H. Lee Moffitt Cancer Center. Melanoma tumors were obtained from patients from a tumor bank under MDA lab protocol LAB06-0755. Melanoma tumors were surgically resected from stage IIIc-IV melanoma patients at MD Anderson Cancer Center and Moffitt Cancer Center as part of ongoing TIL adoptive cell therapy pre-clinical and clinical trials. The tumor was cut up into 4–6 mm2 fragments and the fragments placed in a 24 well plate with TIL culture media (TIL-CM) and 6,000 IU/ml Interleukin-2 (IL-2). The TIL-CM contained RPMI 1640 with Glutamax (Gibco/Invitrogen; Carlsbad, CA), 1× Pen-Strep (Gibco/ Invitrogen; Carlsbad, CA), 50 µm 2-mercaptoethanol (Gibco/ Invitrogen; Carlsbad, CA), 20 µg/ml Gentamicin (Gibco/ Invitrogen; Carlsbad, CA), and 1 mM pyruvate (Gibco/ Invitrogen; Carlsbad, CA). For our control cultures, the fragments were placed in 6,000IU/ml IL-2 with a fully-human IgG4 Isotype control (Eureka therapeutics ET904) in our experiments. The control cultures are referred to as ‘IL-2.’
Flow cytometry staining for NFκB, and IκBα
Tumors were cut up into multiple fragments, placed in TIL-CM, IL-2 ± anti-4-1BB antibody. After 1 week, the fragments and the cells that migrated out of the tumor fragments were harvested. The fragments were mechanically disaggregated and filtered. The fragments and the cells that migrated out of the fragment were washed with Facs Wash Buffer (FWB), and stained for CD3 (BD Biosciences) and CD8 (BD Pharmingen) on ice for 25 minutes. The cells were washed with FWB, and fixation buffer (BD Bioscience) was added to each tube. The tubes were placed at room temperature (RT) for 20 minutes. The cells were washed with FWB for 5 minutes at 1400 rpm, 4°C and Perm Buffer III (BD Bioscience) was added. The tubes were placed on ice for 30 minutes and washed in FWB for 5 minutes, 1400 rpm, 4°C and then stained using anti-NFκB (p65) Alexa 647 or anti-IκBα Alexa 647 (BD Pharmingen) and left in the dark at RT for 1 hour. The cells were washed with FWB for 5 minutes, 1400rpm, 4°C, then re-suspended in FWB and samples were subsequently acquired.
Staining for Interferon-gamma and degranulation
Melanoma tumors were cut up into multiple fragments, and placed in culture with TIL media, IL-2, with or without anti-4-1BB antibody. After 3 weeks, the TIL were set up at a 1:1 ratio with HLA-matched melanoma tumor lines in a 96-well plate for 1 hour at 37°C. Golgi-Stop (BD Biosciences) was added to each well and the cultures were left at 37°C for a 5 hour period. During this time, CD107a antibody (BD Biosciences) was also added to each condition. The cells were harvested, washed with FWB and stained for CD3 and CD8 for 25 minutes at 4°C. The cells were washed with FWB, re-suspended in fixation buffer and incubated for 15 minutes at RT, then washed with FWB and re-suspended in Cytofix/Cytoperm buffer and incubated at room temperature for 20 minutes. The cells were washed with FWB and stained for IFN-gamma for 25 minutes in 4°C, washed and the samples were acquired.
Cytospin and immunocytochemistry staining for surface and nuclear markers
The tumor fragments were set up with or without anti-4-1BB antibody. After 1 week in culture, the cells were collected and washed in 1× D-PBS for 5 minutes at 1400 rpm then re-suspended in 1 ml 4% para-formaldehyde for 20 minutes at RT. The cells were washed in D-PBS and placed at 4°C, then cytospun onto glass slides at 750 rpm for 3 minutes. The samples for Ki67 (Dako) and NFκB (p65) (BD transduction laboratories) were placed in PBS, 0.2% Triton X-100 solution for 10 min at RT and washed in 1× D-PBS. Samples were placed in 3% H2O2/methanol for 10 minutes at RT. The samples were washed in D-PBS and 2.5% normal horse serum was added to all slides for 30 minutes in a humid chamber at RT followed by addition of the primary antibody at 4°C overnight. After, a biotinylated secondary antibody was added to the samples for 30 min at RT (Vectastain Universal Elite ABC Kit). The slides were then washed in D-PBS and peroxidase-conjugated avidin biotin complex ABC Reagent (Vectastain Universal Elite ABC Kit) was added for 30 min at RT. The samples were washed with D-PBS and 3,3-Diaminobenzidine (DAB) was added. The samples were counterstained and covered. Analysis was done using Leica Application Suite (LAS) V4.2 software (Leica Microsystems). For NFκB (p65) staining, analysis was also done using the LAS V4.2 software, and additional analysis was conducted using Vectra Intelligent Slide Analysis System (Vectra, Perkin Elmer) Nuance software 3.0.1.2, using composite coloring style “Fluorescence.”
Supplemental Materials and Methods
Fresh fragment flow cytometry staining
Melanoma fragments were mechanically disaggregated using glass slides, filtered, re-suspended in FWB, washed and stained for CD3, (BD Biosciences; Catalog 555342), CD8 (BD Pharmingen; Catalog 558207), 4-1BB (BioLegend; Catalog 309817; Lot B147320), and AmCyan Aqua live/dead dye (Molecular Probes by Life Technologies; Lot 1413034). One week after the fragments were set up with or without the anti-4-1BB antibody, the fragments were mechanically disaggregated, filtered, and washed with FWB for 5 minutes at 1400 rpm. The cells were stained for CD11c, CD80, CD86, 4-1BB, HLA-DR, (eBioscience, BD Biosciences, and BD Pharmingen) and AmCyan Aqua live/dead fixable dye for 25 minutes on ice. The cells were then washed in FWB for 5 minutes at 1400 rpm and fixed. The cells were then acquired using a BD FACScanto II flow cytometer machine.
TIL staining using flow cytometry after 3 weeks in culture
Fragments were set up with or without the addition of agonistic anti-4-1BB antibody. After 3 weeks, the TIL were harvested and stained for CD3, CD4, CD25, CD27, CD28, and CD8 (eBiosciences, BD Pharmingen, and BD Biosciences). T-regulatory cells (Tregs) were stained for CD4, CD25, and Foxp3 using a Treg staining kit from eBiosciences (La Jolla, CA). The cells were stained for 25 minutes on ice, then washed with FWB and BD Cytofix/Cytoperm Fixation and Permeabilization Solution (BD Biosciences Cat: 554722) (used for Granzyme B, bcl-2, and EOMES) or Perm Buffer III (BD Biosciences Cat: 558050) was used for bcl-6. The buffers were added to the samples for 20 minutes at RT in the dark. The cells were washed with 1× BD Perm/Wash Buffer for 5 minutes at 1400 rpm, followed by intracellular staining for Foxp3, Granzyme B, bcl-2, EOMES, and bcl-6 (eBiosciences, BD Biosciences) in 1× Fixation Buffer for 25 minutes on ice. The samples were washed with FWB for 5 minutes at 1400 rpm and fixed, followed by acquisition using a BD FACScanto II flow cytometer machine.
Measurement of tumor reactivity using IFN-gamma ELISA
After 3 weeks, the TIL were isolated, washed and co-cultured overnight in 96-well plates at a 1:1 ratio with tumor target cells from autologous or HLA-A-matched melanoma tumor cell lines. The supernatants were collected after 24 hr and IFN-gamma secretion was measured using a human IFN-gamma ELISA kit (Thermo Scientific KB132422). A 96-well ELISA plate reader (ELx808, Bio-Tek Instruments Inc., Houston, TX) was used to read the plate.
Statistical Analysis
Statistical analysis for comparison of the 2 groups was done using an unpaired, non-parametric, Mann-Whitney test to determine the statistical values for all the figures, with the test using biological relevance occurring when p< 0.05. Statistical analysis was done using Graph Pad Prism.
Results
4-1BB expression in melanoma tumor-infiltrating lymphocytes
We previously found that some CD3+CD8+ TIL expanded from tumor fragments expressed 4-1BB [16] and wanted to determine whether 4-1BB was expressed on the freshly isolated TIL from melanoma tumors used to derive tumor fragment cultures. Tumors were surgically resected from metastatic melanoma patients and single cell suspensions were prepared and stained for 4-1BB on the CD3+CD8+ subset. In 18 independent freshly isolated TIL samples, we found that 4-1BB was mainly expressed on CD8+ TIL (Supplemental Fig. S1A). We also looked at TIL emerging from tumor fragments 7 days after tumor fragment culture initiation from 7 patients and observed that a considerable frequency of TIL expressed 4-1BB (Supplemental Fig. S1B).
Agonistic anti-4-1BB antibody increases TIL expansion in vitro
The detection of 4-1BB+ CD8+ T cells in the isolated tumor fragments prompted us to ask whether co-stimulation of 4-1BB in these early cultures could affect the outcome of TIL outgrowth. We tested a co-stimulatory anti-4-1BB antibody from Bristol Myers Squibb (BMS) (through a Materials Transfer Agreement) we previously used in later stage TIL cultures that protected expanded CD8+ TIL from activation-induced cell death [15, 16]. Melanoma tumors were surgically resected from patients and cut into four 3–5mm2 fragments per condition and cultured over a 3 week period. We conducted dose titration experiments with the anti-4-1BB added on day 0 of culture to determine the optimal concentration of antibody that would increase the yield of CD8+ T cells from the fragments (Supplemental Fig. S2). As shown in Supplemental Fig. S2, represented in 3 different melanoma TIL patient samples, we found that 10 µg/ml anti-4-1BB antibody produced the most consistent result in terms of enhancing total TIL and CD8+ TIL outgrowth. In subsequent experiments, this dose was used each time the TIL were sub-cultured with fresh media and IL-2.
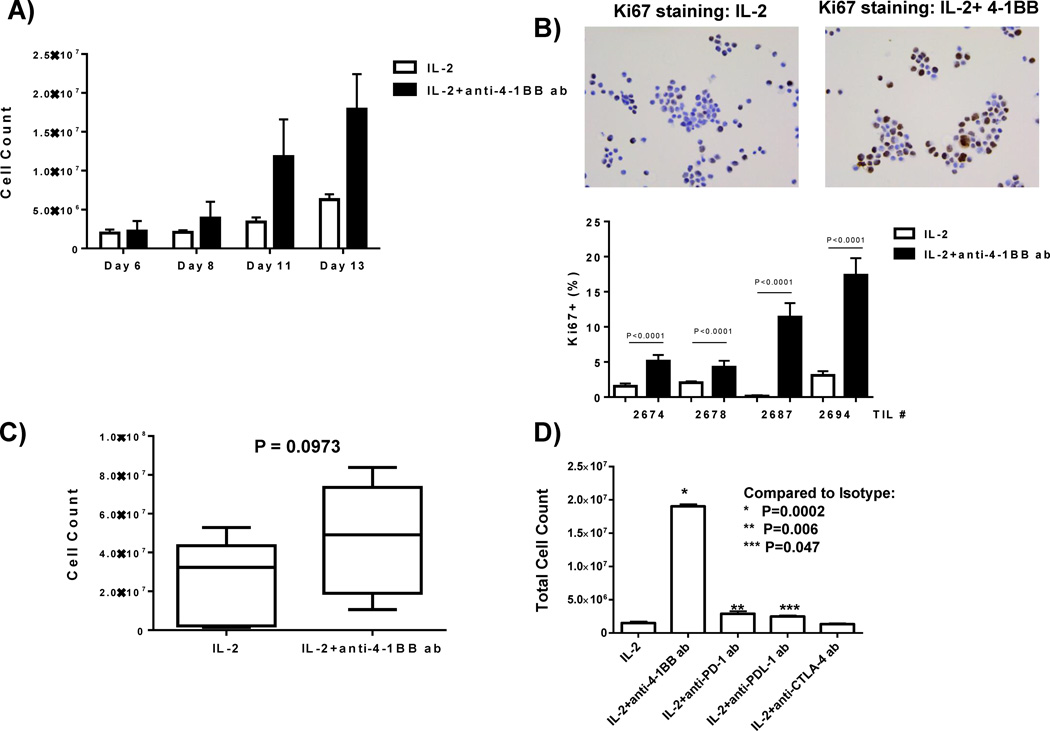
We tested how anti-4-1BB affects TIL yield and the rate of TIL outgrowth from the tumor fragments and found that addition of 4-1BB accelerated the rate of TIL outgrowth over the first two weeks of culture fragments from four representative melanoma TIL patients (Figure 1A). The error bars indicate the standard deviation cell counts from 4 independent melanoma patients. The accelerated rate of TIL outgrowth in the 4-1BB cultures was associated with a marked increase in proliferating cells emanating from the fragments during the first 7 days in culture, as shown by the increased nuclear Ki67 staining of isolated cells surrounding the tumor fragments after cytospin (Figure 1B; four independent experiments and melanoma TIL samples). We performed experiments on an additional seven melanoma patient samples, 4 fragments per melanoma patient samples (28 separate tumor fragments) and found that this enhanced outgrowth of TIL after 3 weeks was consistently observed in a larger sampling of patient tumors (Figure 1C). We determined how anti-4-1BB facilitates TIL outgrowth compared to other immunomodulatory (checkpoint) antibodies, anti-PD-1, anti-PD-L1, and anti-CTLA-4, currently being used in clinical trials and standard of care for melanoma (anti-CTLA-4). As shown in Figure 1D, anti-4-1BB antibody was superior in inducing TIL outgrowth from the tumor fragments compared to IL-2 alone or IL-2 plus these other checkpoint antibodies.
Figure 1. Anti-4-1BB agonistic antibody increases TIL expansion in vitro.
Melanoma tumors were surgically resected from patients. The tumors were then cut up into multiple fragments and placed in culture with IL-2 with or without the anti-4-1BB antibody. Cell counts were conducted from days 6–13 using a hemocytometer and Trypan Blue. At these early time points, the 4-1BB activated TIL grew faster compared to the IL-2 control, as demonstrated in 4 representative TIL patient samples each having multiple fragments in culture (A). When determining the proliferation of the TIL at early time points, we found an increased expression of Ki67 proliferation marker in the 4-1BB activated TIL as compared to the control as demonstrated using immunocytochemistry (B top) and further confirmed and quantified the immunocytochemistry in 4 independent TIL lines (representative of 16 fragments) (B bottom). The melanoma tumors were cut up into multiple fragments and placed in culture with IL-2 with or without the anti-4-1BB antibody over a 3 week period. Using a hemocytometer and Trypan Blue, we determined viable cell counts in the different conditions after the TIL were in culture for 3 weeks. We found that in 7 TIL samples (28 separate tumor fragments; 4 pooled fragments per patient sample), the TIL expanded with IL-2 and the anti-4-1BB antibody expanded better than the IL-2 control (C). After the fragments were in culture for 3 weeks with IL-2 alone or with IL-2 and different antibodies (anti-4-1BB, anti-PD-1, anti-PDL-1, anti-CTLA-4), cell counts were conducted using a hemocytometer after Trypan Blue staining. As demonstrated in 1 representative TIL sample (4 separate fragments), the TIL grown with the agonistic anti-4-1BB antibody exhibited the most TIL expansion as compared to the other antibodies (D). An unpaired, non-parametric, Mann-Whitney test was conducted to determine the statistical values for all the figures.
Addition of anti-4-1BB increased NFκB nuclear translocation in TIL
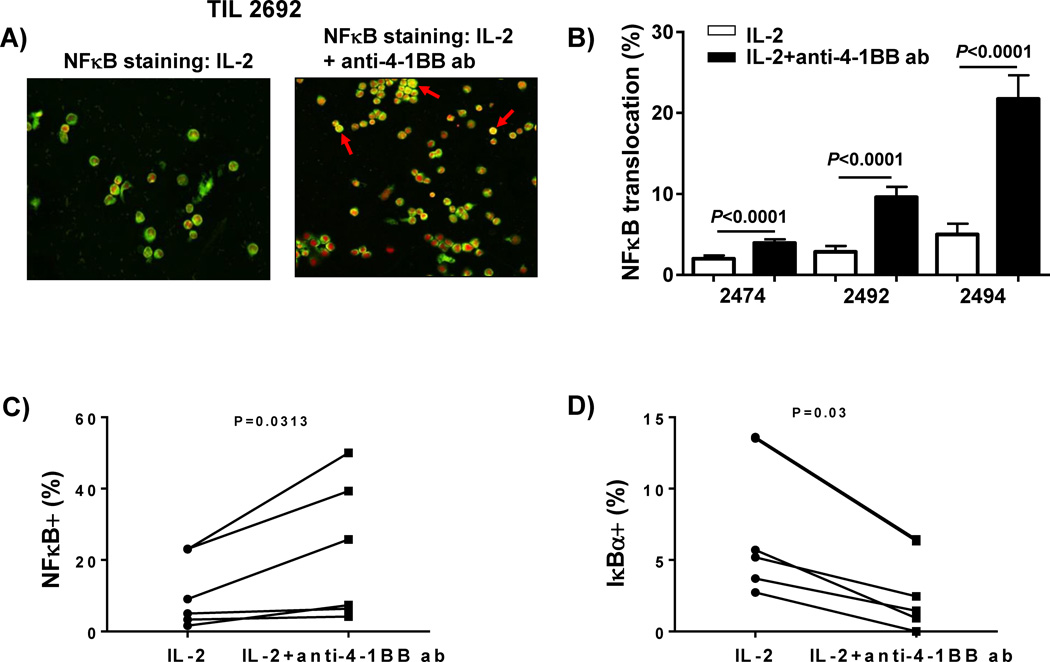
We determined the effect of 4-1BB co-stimulation on the TIL within the tumor microenvironment at the start of the tumor fragment cultures as well as on TIL that had migrated out from the fragments. One of the key signaling pathways activated by 4-1BB co-stimulation is the classical NFκB (p65/cREL) transcription factor pathway. 4-1BB co-stimulation results in TRAF recruitment that activates IKKαβ complexes. This results in degradation of IκBα and subsequent translocation of NFκB to the nucleus [17]. Melanoma tumor fragments (4 fragments per condition for each patient) were established in culture with or without addition of anti-41BB. After 1 week, the TIL that had migrated out of the tumor fragment supernatant were collected, cytospun onto glass slides, fixed and stained for NFκB (p65). We observed that the TIL grown with IL-2 and anti-4-1BB antibody had markedly more NFκB (green) translocation in the nucleus (red) as compared to the TIL grown in IL-2 (Figure 2A; arrows and yellow color indicate NFκB nuclear translocation). Staining for NFκB in TIL in cytospins from 3 different patient tumor samples found a consistent increase in translocated p65 in cultures treated with anti-4-1BB, after quantifying the staining using a digital imaging system (Figure 2B). We also isolated fragments 1 week after culture initiation and stained for NFκB (p65) and IκBα in the TIL using flow cytometry after mechanically disaggregating the isolated fragments into single cell suspensions. After gating on viable CD3+CD8+ T cells, we detected an increase in NFκB (p65) staining (Figure 2C) together with a decrease in IκBα expression (Figure 2D), indicative of NFκB activation, in the anti-4-1BB antibody group compared to the IL-2 alone control group, demonstrated in 6 independent melanoma patient samples. These results indicate that addition of agonist anti-4-1BB antibody to melanoma tumor fragments can activate NFκB in the T cells inside and outside the tumor fragments.
Figure 2. Increased NFκB translocation in TIL expanded with the anti-4-1BB antibody.
Melanoma tumors were cut up into multiple fragments and placed in culture with IL-2 alone or IL-2 with or without an anti-4-1BB antibody. After 1 week, the cells were collected and cytospun onto a glass slide, followed by staining using Immunocytochemistry. Staining for NFκB (p65) revealed more translocation in the nucleus of TIL grown with IL-2 and the anti-4-1BB antibody compared to TIL grown with IL-2 alone. We used Vectra Intelligents slide analysis system (Nuance software) (A) to observe the translocation of NFκB (red is nucleus; green is NFκB; and yellow is overlay (translocation). The red arrow indicates one area where translocation occurred. Quantification of NFκB translocation in 10 different areas per sample is demonstrated in 3 independent TIL samples (12 fragments; 4 pooled fragments per patient sample) (B). In addition to immunocytochemistry, we also measured the percentage of NFκB and IκBα expression in the CD8+CD3+ TIL within the fragment after 1 week in culture using flow cytometry in 6 patient samples (24 representative fragments; 4 pooled fragments per patient) (C). The fragments were set up with IL-2 with or without the addition of anti-4-1BB antibody. After 1 week, the fragments were collected (4 fragments per patient sample) and mechanically disaggregated using glass slides. The cells were filtered and stained using flow cytometry. The TIL were gated on live, CD3+, CD8+ TIL and the percentage of NFκB (C) and IκBα (D) was measured. We found that the TIL cultured with IL-2 plus anti-4-1BB antibody exhibited an increase NFκB (C) with a paralleled decrease in IκBα (D) as demonstrated in 6 independent TIL samples (24 representative fragments). An unpaired, non-parametric, Mann-Whitney test was used to determine the statistical values for B–D.
Activating the 4-1BB pathway increases the CD8+ TIL expansion
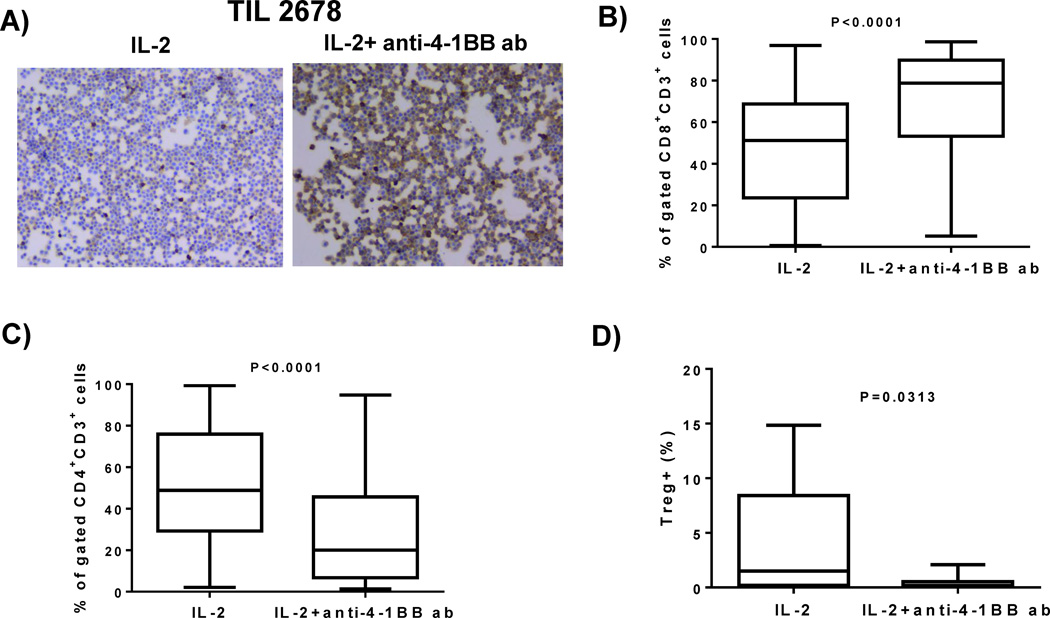
We determined whether the cells that were growing out of the fragment were enriched for CD8+ T cells and also measured the yield of CD4+ T cells and CD4+ CD25+Foxp3+ T-regulatory cells (Tregs). Melanoma fragments from patient tumors were set up with or without the addition of anti-4-1BB antibody. We conducted CD8 staining of cytospun cells that were isolated from outside the tumor fragments in early cultures (after 1 week). As shown in one representative patient sample, 4-1BB co-stimulation increased the number of CD8-stained cells versus IL-2 alone (Figure 3A). In 14 patient tumor samples (representing 56 independent tumor fragments), we found that addition of anti-4-1BB increased the percentage of CD8+ T cells expanded from the tumor fragments after 3 weeks (Figure 3B), while the percentage of CD4+ T cells decreased (Figure 3C). Staining for Tregs after 3 weeks in experiments on 7 patient tumor samples (representing 28 independent tumor fragments) found that 4-1BB co-stimulation in these early fragment cultures reduced their frequency (Figure 3D).
Figure 3. CD8+ TIL percentage is increased with the addition of anti-4-1BB antibody to TIL cultures.
The melanoma tumors were surgically resected from the patients, cut up into multiple fragments and placed in culture with IL-2 alone or with IL-2 and anti-4-1BB antibody. After 1 week in culture, we collected the cells, cytospun them onto a glass slide conducted Immunocytochemistry, and found that the TIL expanded with IL-2 and the anti-4-1BB antibody exhibited an increase in CD8+ cells, as shown in one representative TIL line (4 pooled fragments) (A). After the melanoma fragments were cultured with IL-2 alone or IL-2 and anti-4-1BB antibody for 3 weeks, the TIL were stained for the expression of CD3, CD8, and CD4 using flow cytometry. We observed that in 56 independent TIL lines grown with IL-2 and anti-4-1BB antibody exhibited an increase in the percentage of CD8+ TIL in the CD3 subset compared to the IL-2 control (B). When we examined the CD4+ expression in 56 independent TIL samples, we found that the TIL expanded with IL-2 alone exhibited an increase in CD4+, compared to TIL grown with IL-2 with an anti-4-1BB antibody (C). We went on to determine the percentage of CD3+CD4+CD25+FoxP3+ T regulatory cells (Tregs). Cells were stained for Tregs with anti-Foxp3, anti-CD4, and anti-CD25 using the Foxp3 staining kit from eBioscience. The control TIL (IL-2 alone) exhibited an increased percentage of Tregs than the TIL expanded in anti-4-1BB antibody (D) as demonstrated in 7 TIL samples (28 representative fragments). An unpaired, non-parametric, Mann-Whitney test was used to determine the statistical values for figures B–D.
Modulation of TIL effector phenotype by activating the 4-1BB pathway
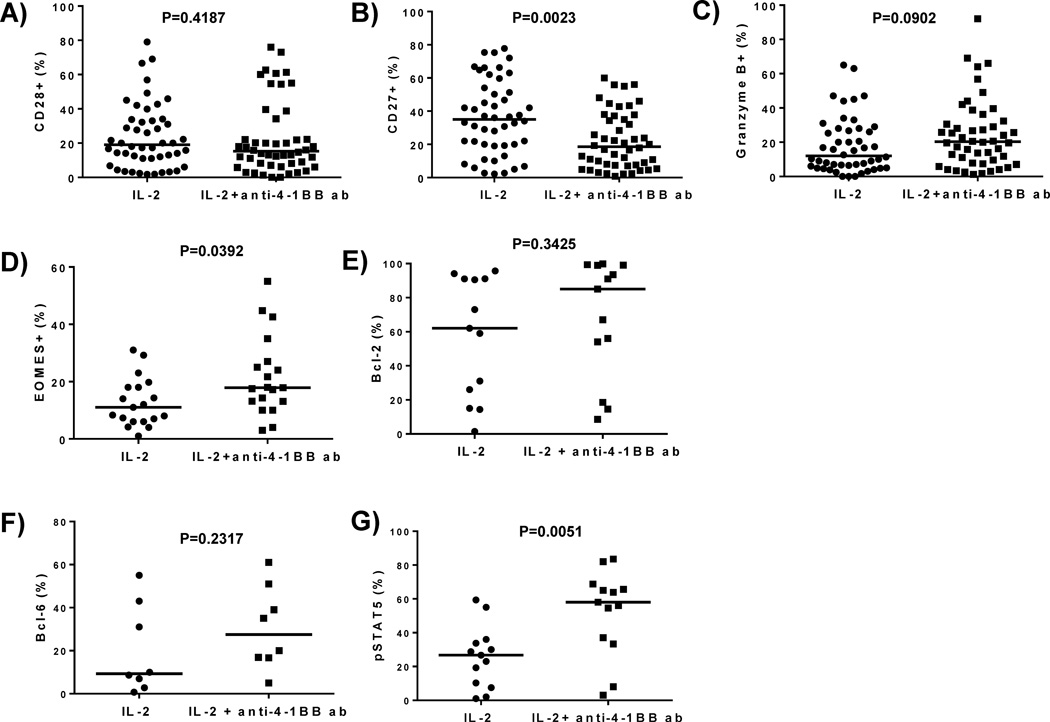
Melanoma tumor fragments were cultured with or without anti-4-1BB antibody. After 3 weeks, the TIL were stained using flow cytometry for effector-memory markers (CD28 and CD27). In 48 independent tumor fragment lines (representing 12 different melanoma patient samples), we found that CD28 expression was unaltered (Figure 4A), but CD27 expression decreased somewhat (Figure 4B). We tested whether TIL that grew out of tumor fragments over the 3-week period with anti-4-1BB co-stimulation could further expand after re-stimulation through the T-cell receptor (TCR) in a rapid expansion protocol (REP). In 6 independent patient melanoma samples, 4-1BB co-stimulation during the initial TIL outgrowth did not affect the fold-expansion of TIL after TCR re-stimulation in the REP (data not shown) or the phenotype of the post-REP cells in terms of CD8, CD28, and CD27 expression, (data not shown).
Figure 4. Phenotypic characterization of effector-memory markers in TIL isolated from tumor fragment cultures treated with or without-anti-4-1BB.
Fragments from melanoma tumors were set up in a 24 well plate with or without anti-4-1BB antibody. 3 weeks after the initial set-up, the TIL were harvested and flow cytometry was done to analyze effector-memory markers and cytolytic granule markers. When we measured the expression of CD28 in the CD3+ CD8+ subset in 48 independent TIL lines, we found no statistical significance in the expression of CD28 in the CD3+CD8+ subset in the different conditions (A). When we measured the expression of CD27 in the CD3+ CD8+ population in 48 independent TIL lines, we found that the CD27 expression in the CD3+ CD8+ subset was down-regulated in the TIL expanded with the anti-4-1BB antibody as compared to the TIL grown with IL-2 alone (B). When we measured the expression of the cytolytic granule marker Granzyme B in the CD3+ CD8+ subset in 48 independent TIL lines, we found the expression of Granzyme B was increased in the TIL expanded with IL-2 and anti-4-1BB antibody as compared to TIL expanded with IL-2 alone (C). When we measured the expression of Eomes using flow cytometry in 19 independent TIL, we found the TIL expanded with the anti-4-1BB antibody exhibited increased Eomes expression in the CD3+ CD8+ subset (D). When we determined the percentage of the anti-apoptotic molecule, bcl-2 in 13 independent TIL, we found that the TIL expanded with the anti-4-1BB antibody exhibited an increased bcl-2 percentage (E). We also determined the expression of bcl-6 (F) in 8 independent TIL lines and pSTAT5 in 13 independent TIL in the CD3+ CD8+ subset (G). We observed that bcl-6 and pSTAT5 percentage was increased when the TIL were expanded with IL-2 plus anti-4-1BB antibody for 3 weeks (F and G). Each TIL sample contained 4 pooled fragments. An unpaired, non-parametric, Mann-Whitney test was conducted to determine the statistical values for all the figures.
We investigated the effect of anti-4-1BB on the expression of molecules associated with effector (Granzyme B and Eomes), memory (bcl-6 and Eomes), and survival (bcl-2 and bcl-6) functions of Cytotoxic T Lymphocyte (CTL). Granzyme B is a cytolytic granule molecule in CD8+ T cells, while Eomes is a T-box transcription factor helping drive Granzyme B expression as well as being involved in memory T-cell maintenance [18] [19, 20]. Bcl-2 and bcl-6 are cell survival molecules in memory T cells [19–22]. We found an increased intracellular expression of the Granzyme B (Figure 4C) and Eomes (Figure 4D) in CD8+ TIL expanded from fragments with 4-1BB co-stimulation. We observed an increase in the mean fluorescence intensity (MFI) (data not shown) and percentage of Granzyme B in the CD8+ subset in 12 independent TIL samples (representative of 48 melanoma patient fragments) and an increase in Eomes in 19 independent melanoma patient samples. The MFI (data not shown) and percentage of bcl-2 and bcl-6 in the CD8+ subset was also increased (Figure 4E and F). Bcl-2 was increased in 13 independent TIL samples, while we observed an increase in 8 independent TIL samples. Each melanoma patient sample contained 4 fragments that were pooled together prior to staining.
Due to the accelerated outgrowth of CD8+ T cells from the tumor fragments after addition of anti-4-1BB, we also surmised that IL-2 signaling may be altered. Since there have been STAT5 binding sites identified in 4-1BB promoter region, and 4-1BB has its expression modulated by IL-2R signaling through activation of STAT5 [23], we specifically looked at CD25 (IL-2Rα) and pSTAT5. In 13 independent melanoma patients (representative of 52 independent fragments; 4 fragments per patient sample), TIL were collected after 3 weeks of expansion from tumor fragments and stained for pSTAT5. We measured a higher frequency of tyrosine-phosphorylated STAT5 (pSTAT5) expression in CD8+ TIL in the 4-1BB treatment group (Figure 4G).
Increased anti-tumor reactivity in TIL expanded with anti-4-1BB antibody
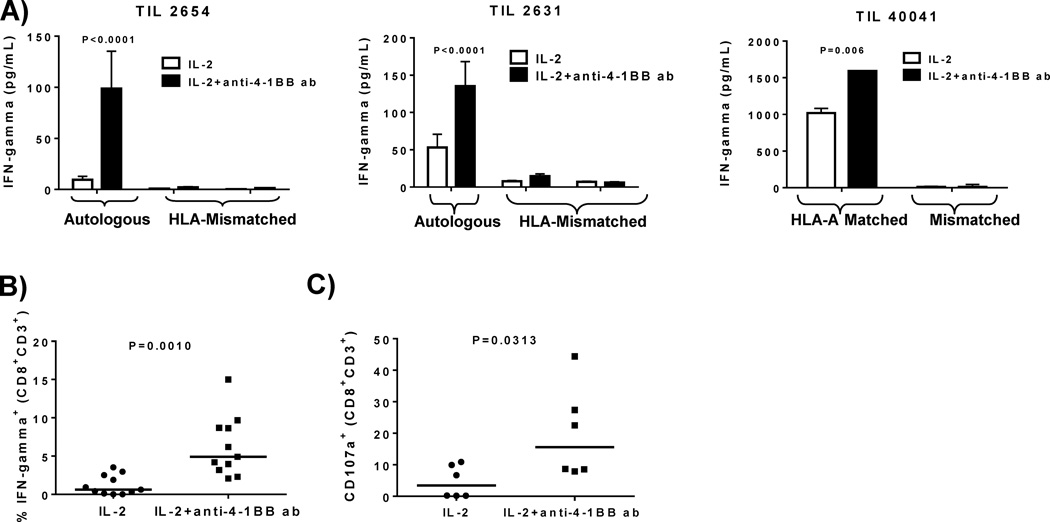
The induced expression of 4-1BB known to occur on antigen-activated CD8+ T cells together with our detection of a significant frequency of 4-1BB+ CD8+ TIL within tumors (Supplemental Fig. S1) suggests that addition of agonist anti-4-1BB may also increase the tumor reactivity of the TIL grown out from the treated fragments. We analyzed the anti-tumor reactivity of bulk TIL cultures grown out from tumor fragments after 3 weeks against autologous or HLA-A-matched allogeneic tumor lines by measuring the extent tumor-specific IFN-gamma release after a 24-hour tumor cell-TIL co-culture assay. We found that provision of 4-1BB co-stimulation to the tumor fragments yielded TIL with markedly higher anti-tumor reactivity after 3 weeks of culture (Figure 5A; data from three independent experiments and melanoma patient samples shown). We performed Intracellular Cytokine Staining (ICS) assays for IFN-gamma production on a single cell level using flow cytometry of TIL isolated after 3 weeks from fragment cultures from HLA-A2+ patients using HLA-A2+ melanoma tumor targets and found a significant increase in the frequency in IFN-gamma-positive CD8+ T cells in TIL isolated from fragment cultures treated with anti-4-1BB antibody (Figure 5B; data from 11 independent melanoma patient TIL shown, representative of 4 fragments pooled per patient sample (44 fragments total). We measured the frequency of CD107a release (a measure for antigen-specific T-cell degranulation) using flow cytometry. The TIL expanded with IL-2 and anti-4-1BB versus IL-2 alone also exhibited an increase in CD107a+ frequency after co-incubation with HLA-A-matched tumor targets in the CD3+CD8+ subset (Figure 5C; data from 6 melanoma patients, representative of 24 melanoma patient fragments, each patient sample containing 4 pooled fragments).
Figure 5. Increased tumor specificity of TIL from tumor fragment cultures treated with IL-2 and anti-4-1BB.
After the fragments were set up with IL-2 alone or IL-2 and anti-4-1BB antibody and expanded over a 3 week period (pre-REP), the TIL were harvested and set up at a 1:1 ratio with autologous (A left and middle) or HLA-A matched (A right) melanoma tumor cells. Supernatants were collected after 24 hours and IFN-gamma secretion was measured using ELISA. We demonstrated in 3 independent TIL samples (4 pooled fragments per TIL sample) that IFN-gamma secretion was increased in the pre-REP TIL expanded with IL-2 and anti-4-1BB compared to TIL expanded with IL-2 alone (A). After 3 weeks in culture with or without the anti-4-1BB antibody, the TIL were set up at a 1:1 ratio with HLA-A matched tumor lines. Using flow cytometry, we measured the amount of IFN-gamma+ cells in the CD3+CD8+ subset (B). In 11 independent TIL patient samples (44 representative fragments; 4 pooled fragments per TIL sample), the TIL expanded with IL-2 and anti-4-1BB antibody exhibited an increase in IFN-gamma+ cells in the CD3+CD8+ subset (B). We also used flow cytometry to determine the amount of degranulation (CD107a) in the CD3+CD8+ subset after setting up a 1:1 ratio with TIL and HLA-A matched melanoma tumor lines (C). We found that in 6 independent TIL patient samples (24 representative fragments; 4 pooled fragments per TIL sample), the TIL grown with IL-2 and the anti-4-1BB antibody exhibited an increase in CD107a+ cells in the CD3+CD8+ subset (C). An unpaired, non-parametric, Mann-Whitney test was conducted to determine the statistical values for all the figures.
Anti-4-1BB addition modulates resident dendritic cells within melanoma tumor fragments
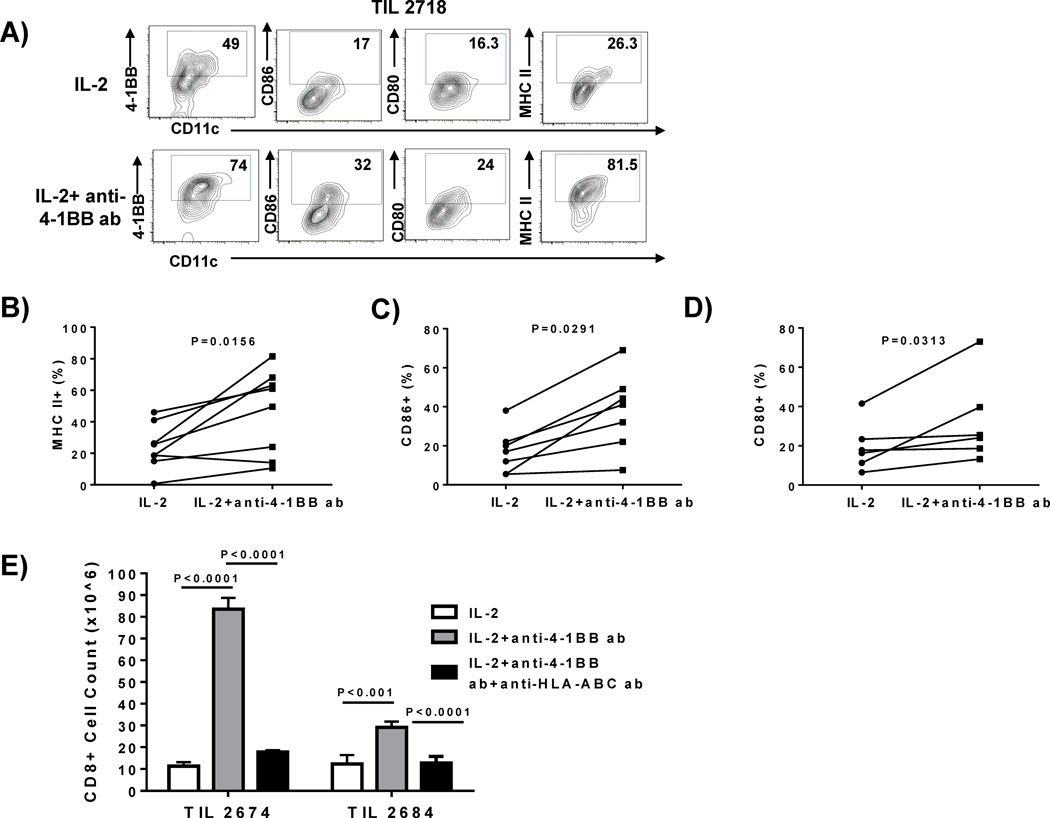
Our observation that agonist anti-4-1BB antibodies activate CD8+ T cells in the tumor microenvironment within the tumor fragments suggested that other tumor-resident leukocyte populations may be affected by 4-1BB co-stimulation given in this context. We performed flow cytometry analysis of cell suspensions obtained from tumor fragments that were in culture for 7 days to determine what other viable (surviving) resident cells were present and expressing 4-1BB that may be targeted by the added antibody. We stained for dendritic cells (DC), T cells, NK cells, and B cells. We found very few NK cells (CD3−CD56+) and B cells (CD3−CD20+) in the tumor fragments and these had no detectable 4-1BB expression (data not shown). CD8+ and CD4+ T cells were detected, with only the CD8+ cells expressing detectable 4-1BB. In the DC population (CD3−CD11c+ HLA class II+), we found a considerable frequency of viable 4-1BB+ DC persisting in these fragments. Addition of the agonistic anti-4-1BB during the tumor fragment culture led to activation of DC, manifested by a marked increase in CD86 and HLA class II expression in the gated live CD3−CD11c+ subset in one representative melanoma patient sample (4 fragments pooled) (Figure 6A). We measured the expression of markers on DC isolated from tumor fragment cultures from resected melanomas from 8 independent melanoma patient samples (representative of 32 fragments, 4 fragments pooled per patient sample) (A), 7 independent melanoma patient samples (representative of 28 fragments, 4 fragments pooled per patient sample), and 6 independent melanoma patient samples (representative of 24 fragments, 4 fragments pooled per patient sample). We found an increase in HLA (MHC) class II (Figure 6B), CD86 (Figure 6C), and CD80 (Figure 6D) on DC isolated from fragments on day 7 after addition of anti-4-1BB.
Figure 6. Addition of anti-4-1BB antibody to the melanoma fragments modulates resident dendritic cell phenotype.
The melanoma tumors were cut up into multiple fragments and placed in culture with IL-2 alone or IL-2 and anti-4-1BB antibody for 1 week. After 1 week in culture, the fragments were mechanically disaggregated, filtered, and stained for Dendritic Cells (DC) and their activation markers. In 1 representative TIL sample (4 pooled fragments), we found that the resident DCs within the melanoma fragment expressed 4-1BB (A) and the addition of IL-2 and the anti-4-1BB antibody increased CD86, CD80 and MHC II expression (A). When we observed the live, CD3−CD11c+ cells, we found that the DC had a more activated phenotype with increased MHC-II (B), CD86 (C) and CD80 (D) when anti-4-1BB antibody was added compared to the cultures grown in IL-2 alone. This was demonstrated in 8 independent TIL samples (32 representative fragments; 4 pooled fragments per TIL sample) for MHC II (B), 7 independent TIL samples (28 representative fragments) for CD86 expression (C), and 6 independent TIL samples (24 representative fragments) for CD80 expression (D). The melanoma fragments were cut up and set up with or without the addition of an anti-MHC-I (anti-HLA-ABC) antibody. After 3 hours, IL-2 ± anti-4-1BB antibody was added to the cultures and the cultures were expanded over a 3 week period. After 3 weeks, the cells were stained using flow cytometry and viable cell counts were conducted. We found that when we blocked MHC-I, there was a decrease in the expansion of CD8+ TIL, compared to the TIL expanded with IL-2 and anti-4-1BB antibody (E) as demonstrated in 2 independent TIL lines (8 representative fragments). An unpaired, non-parametric, Mann-Whitney test was used to determine the statistical values for figures B–E.
The observed activation of tumor fragment-resident DC with anti-4-1BB suggested that an ongoing presentation of antigen occurs in early tumor fragment cultures that may help drive CD8+ TIL expansion. To test this, we added purified blocking anti-HLA-ABC antibody (W6/32) to the tumor fragment cultures on day 0 and harvested the TIL that had grown out of the fragments after 3 weeks. As shown in Figure 6E, in 2 representative melanoma TIL patient samples, we found that addition of anti-HLA-ABC antibody reversed the effect of anti-4-1BB on increasing the outgrowth of CD8+ T cells, as measured by the decreased CD8+ T-cell numbers.
Discussion
Adoptive cell therapy using TIL has shown considerable efficacy in mediating tumor regression [1–3]. Current response rates using a non-myeloablative chemotherapy pre-conditioning regimen of close to 40–50% have been observed at multiple centers [1, 3, 6, 24]. There is growing interest in making the TIL expansion process faster and more practical. CD8+ T cells have emerged to be a key subset in mediating clinical response [3]. We recently demonstrated that melanoma patients that responded to ACT using TIL had a significantly higher percentage of CD8+BTLA+ cells [3]. We have measured whether the addition of IL-2 and anti-4-1BB antibody added during the rapid expansion [15] augmented the levels of CD8+BTLA+ in the post-REP TIL, but did not detect a significant difference between the IL-2 alone condition versus IL-2 and anti-4-1BB condition. However, we have not measured whether the addition of IL-2 anti-4-1BB antibody added at the initial stages of melanoma TIL isolation and expansion augments the CD8+BTLA+ population.
A number of biological issues arise in making the TIL initial expansion process not only faster to reduce the wait times for patients, but also enrich tumor-reactive T cells, especially CD8+ that maintain effector-memory phenotypic properties for persistence after infusion. Although the culture of tumor fragments cut from resected metastatic lesions has been used as the most common approach for initial melanoma TIL expansion, there has been no attention paid to whether the microenvironment within these fragment cultures can be modulated to affect TIL expansion. Little is known about the dynamics of the early TIL expansion from tumor fragments and what processes take place within these small tumor pieces that can affect the yield and phenotype of the TIL. Given the emerging importance of tumor-reactive CD8+ TIL in adoptive cell therapy and the identification of early-intermediate T-cell activation markers on these cells marking tumor-specific TIL subsets, we surmised that modulation of T-cell co-stimulatory pathways on these activated TIL, such as 4-1BB, may have profound effects on the yield of TIL and their phenotypic and functional properties that may be beneficial for adoptive cell therapy approaches. 4-1BB is a marker of tumor-reactive CD8+ T cells [13] that is usually up-regulated 24-48 h after antigen exposure [15, 16, 25–27].
We investigated whether the provision of 4-1BB co-stimulation using agonist anti-4-1BB antibody at the initiation of TIL expansion can modulate the tumor microenvironment within early tumor fragment cultures. The addition of anti-4-1BB antibody increased the rate of TIL expansion from tumor fragments, and increased the yield of tumor-specific CD8+ T cells. Our data shows that blockade of HLA class I during early tumor fragment cultures prevents the increased outgrowth of CD8+T cells induced by anti-4-1BB. This suggests that ongoing antigen presentation by cells within the tumor fragments stimulates resident CD8+ T cells resulting in the up-regulation of 4-1BB that is further co-stimulated with the agonist antibody. This was supported by the detection of NFκB activation and increased Ki67 expression in CD8+ TIL isolated from the tumor fragments and from the surrounding culture environment after 4-1BB co-stimulation was provided. The 4-1BB antibody provides a potent signal enhancing CD8+ co-stimulation leading to increased cell division in these early cultures. 4-1BB ligation, or other modulatory antibodies targeting OX40 for example, may inhibit Tregs and further facilitate the expansion of CD8+ T cells in these early TIL cultures as well as alter the phenotype of CD4+ TIL, but this will need to be tested in future studies.
To dissect the effects of anti-4-1BB in the early tumor fragment cultures, we examined its effects on other leukocyte subsets. We focused on tumor fragment-resident DC, as these cells are critical antigen-presenting cells in the tumor microenvironment for the activation of resident CD8+ T cells. We found a sub-population of DC expressed 4-1BB and addition of the agonist anti-4-1BB increased the expression of DC maturation markers such as MHC class II, CD86, and CD80. Increased expression of these DC maturation markers was associated with the induction of NFκB activation in the DC induced by 4-1BB co-stimulation. Data from in vivo tumor model systems has also shown that DC are critical in driving localized T-cell activation and division in the tumor microenvironment [28–30].
Activating the 4-1BB co-stimulatory pathway in leukocytes within tumor fragments at the earliest stages of TIL expansion from metastatic melanomas can profoundly affect the outgrowth of tumor-reactive TIL associated with the modulation of tumor fragment-resident DC. This approach can be applied clinically to rapidly expand TIL enriched for tumor specificity due to the availability of clinical-grade human or humanized anti-4-1BB antibodies. Our work sets the stage for testing the effects of other immunomodulatory agents to manipulate the tumor microenvironment. This approach can be applied to improve the output of TIL from solid tumors, that have proven more difficult to expand tumor-reactive CD8+ T cells maintaining effector and memory properties. The advantage of our approach, is that is can feasibly also be used with small tumors or biopsies to enhance the outgrowth of tumor-specific CD8+ TIL without excessive up-front manipulation beyond cutting up the tumor into fragments for culture. This may be more amenable for clinical-grade TIL production and out-scaling TIL therapy.
Supplementary Material
Translational Relevance.
The success of adoptive T-cell therapy (ACT) using autologous tumor-infiltrating lymphocytes (TIL) depends on the expansion of effector-memory T cells (especially CD8+ cells) capable of recognizing tumor antigens. In this study we developed a novel approach at inducing a more rapid outgrowth of TIL from tumor fragments of melanoma metastases highly enriched for tumor specificity. We capitalized on the specific expression of the 4-1BB co-stimulatory molecule on a subset of tumor-resident CD8+ T cells enriched for tumor specificity. Using an agonistic GMP-grade anti-4-1BB antibody added directly to the initial tumor fragment cultures to provide in situ 4-1BB co-stimulation, we enhanced the rate of CD8+ T-cell outgrowth, NFκB survival gene activation, and tumor specificity of the expanded TIL. This method offers a practical way to improve the quality of TIL products for ACT by reducing the culture time needed, while ensuring high tumor-specificity and maintenance of effector-memory properties of the cells.
Acknowledgements
We are grateful to Bristol Myers Squibb (Drs. Marie Jure-Kunkel and Stacey Goldberg) for their generous gift of the agonistic anti-4-1BB antibody (BMS-663513). Author contributions: Conceived and designed experiments: LR, JAC, SPT, AAS, JW, PH. Performed the experiments: JAC, CK, JQC, CC, JR, SPT, AAS. Analyzed data: LR, JAC, SPT, AAS, JQC, JR. Wrote the paper: LR, JAC, SPT, AAS. JAC gratefully acknowledges support from the UT Health Innovation for Cancer Prevention Research Pre-Doctoral Fellowship, The University of Texas School of Public Health-Cancer Prevention and Research Institute of Texas grant #RP101503. We are also thankful to the TIL Lab (Chris Toth, Renjith Ramachandran, Seth Wardell, Audrey Gonzalez, and Orenthial Fulbright) at M.D. Anderson Cancer Center for the processing of melanoma tumors and initial culturing of TIL for our experiments. We are thankful to Dr. Vernon Sondak, Dr. Jonathan Zager, Dr. Ricardo Gonzalez, Christopher Puleo PA-C, Georgina Crago PA-C, and Carolyn Rich for assistance in acquiring surgically resected melanoma specimens at the H Lee Moffitt Cancer Center. We thank the Flow Cytometry Core Facility at the H. Lee Moffitt Cancer Center and Research Institute for its contribution to this work. This work was supported by a Developmental Research Program of the Melanoma SPORE (5 P50 CA093459-05-DRP21), a Team Science Award from the Melanoma Research Alliance (MRA), and the Dr. Miriam and Sheldon Adelson Medical Research Foundation (AMRF). This work was also supported in part by a NCI-K23 award (AAS 1 K23 CA178083-01).
References
- 1.Rosenberg SA, Restifo NP, Yang JC, Morgan RA, Dudley ME. Adoptive cell transfer: a clinical path to effective cancer immunotherapy. Nat Rev Cancer. 2008;8(4):299–308. doi: 10.1038/nrc2355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Dudley ME. Adoptive cell therapy for patients with melanoma. J Cancer. 2011;2:360–362. doi: 10.7150/jca.2.360. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Radvanyi LG, Bernatchez C, Zhang M, Fox PS, Miller P, Chacon J, et al. Specific lymphocyte subsets predict response to adoptive cell therapy using expanded autologous tumor-infiltrating lymphocytes in metastatic melanoma patients. Clin Cancer Res. 18(24):6758–6770. doi: 10.1158/1078-0432.CCR-12-1177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Rosenberg SA, Packard BS, Aebersold PM, Solomon D, Topalian SL, Toy ST, et al. Use of tumor-infiltrating lymphocytes and interleukin-2 in the immunotherapy of patients with metastatic melanoma. A preliminary report. N Engl J Med. 1988;319(25):1676–1680. doi: 10.1056/NEJM198812223192527. [DOI] [PubMed] [Google Scholar]
- 5.Rosenberg SA, Spiess P, Lafreniere R. A new approach to the adoptive immunotherapy of cancer with tumor-infiltrating lymphocytes. Science. 1986;233(4770):1318–1321. doi: 10.1126/science.3489291. [DOI] [PubMed] [Google Scholar]
- 6.Besser MJ, Shapira-Frommer R, Treves AJ, Zippel D, Itzhaki O, Hershkovitz L, et al. Clinical responses in a phase II study using adoptive transfer of short-term cultured tumor infiltration lymphocytes in metastatic melanoma patients. Clin Cancer Res. 2010;16(9):2646–2655. doi: 10.1158/1078-0432.CCR-10-0041. [DOI] [PubMed] [Google Scholar]
- 7.Rosenberg SA, Dudley ME. Adoptive cell therapy for the treatment of patients with metastatic melanoma. Curr Opin Immunol. 2009;21(2):233–240. doi: 10.1016/j.coi.2009.03.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Prieto PA, Durflinger KH, Wunderlich JR, Rosenberg SA, Dudley ME. Enrichment of CD8+ cells from melanoma tumor-infiltrating lymphocyte cultures reveals tumor reactivity for use in adoptive cell therapy. J Immunother. 2010;33(5):547–556. doi: 10.1097/CJI.0b013e3181d367bd. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Valkenburg SA, Gras S, Guillonneau C, La Gruta NL, Thomas PG, Purcell AW, et al. Protective efficacy of cross-reactive CD8+ T cells recognising mutant viral epitopes depends on peptide-MHC-I structural interactions and T cell activation threshold. PLoS Pathog. 2010;6(8) doi: 10.1371/journal.ppat.1001039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Tran KQ, Zhou J, Durflinger KH, Langhan MM, Shelton TE, Wunderlich JR, et al. Minimally cultured tumor-infiltrating lymphocytes display optimal characteristics for adoptive cell therapy. J Immunother. 2008;31(8):742–751. doi: 10.1097/CJI.0b013e31818403d5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Besser MJ, Shapira-Frommer R, Treves AJ, Zippel D, Itzhaki O, Schallmach E, et al. Minimally cultured or selected autologous tumor-infiltrating lymphocytes after a lympho-depleting chemotherapy regimen in metastatic melanoma patients. J Immunother. 2009;32(4):415–423. doi: 10.1097/CJI.0b013e31819c8bda. [DOI] [PubMed] [Google Scholar]
- 12.Itzhaki O, Hovav E, Ziporen Y, Levy D, Kubi A, Zikich D, et al. Establishment and large-scale expansion of minimally cultured "young" tumor infiltrating lymphocytes for adoptive transfer therapy. J Immunother. 2011;34(2):212–220. doi: 10.1097/CJI.0b013e318209c94c. [DOI] [PubMed] [Google Scholar]
- 13.Ye Q, Song DG, Poussin M, Yamamoto T, Best A, Li C, et al. CD137 Accurately Identifies and Enriches for Naturally Occurring Tumor-Reactive T Cells in Tumor. Clin Cancer Res. 20(1):44–55. doi: 10.1158/1078-0432.CCR-13-0945. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Gros A, Robbins PF, Yao X, Li YF, Turcotte S, Tran E, et al. PD-1 identifies the patient-specific CD8(+) tumor-reactive repertoire infiltrating human tumors. J Clin Invest. 2014;124(5):2246–2259. doi: 10.1172/JCI73639. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Chacon JA, Wu RC, Sukhumalchandra P, Molldrem JJ, Sarnaik A, Pilon-Thomas S, et al. Co-stimulation through 4-1BB/CD137 improves the expansion and function of CD8(+) melanoma tumor-infiltrating lymphocytes for adoptive T-cell therapy. PLoS One. 8(4):e60031. doi: 10.1371/journal.pone.0060031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Hernandez-Chacon JA, Li Y, Wu RC, Bernatchez C, Wang Y, Weber JS, et al. Costimulation through the CD137/4-1BB pathway protects human melanoma tumor-infiltrating lymphocytes from activation-induced cell death and enhances antitumor effector function. J Immunother. 34(3):236–250. doi: 10.1097/CJI.0b013e318209e7ec. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Kim JO, Kim HW, Baek KM, Kang CY. NF-kappaB and AP-1 regulate activation-dependent CD137 (4-1BB) expression in T cells. FEBS Lett. 2003;541(1–3):163–170. doi: 10.1016/s0014-5793(03)00326-0. [DOI] [PubMed] [Google Scholar]
- 18.Pearce EL, Mullen AC, Martins GA, Krawczyk CM, Hutchins AS, Zediak VP, et al. Control of effector CD8+ T cell function by the transcription factor Eomesodermin. Science. 2003;302(5647):1041–1043. doi: 10.1126/science.1090148. [DOI] [PubMed] [Google Scholar]
- 19.Ichii H, Sakamoto A, Hatano M, Okada S, Toyama H, Taki S, et al. Role for Bcl-6 in the generation and maintenance of memory CD8+ T cells. Nat Immunol. 2002;3(6):558–563. doi: 10.1038/ni802. [DOI] [PubMed] [Google Scholar]
- 20.Ichii H, Sakamoto A, Kuroda Y, Tokuhisa T. Bcl6 acts as an amplifier for the generation and proliferative capacity of central memory CD8+ T cells. J Immunol. 2004;173(2):883–891. doi: 10.4049/jimmunol.173.2.883. [DOI] [PubMed] [Google Scholar]
- 21.Yoshida K, Sakamoto A, Yamashita K, Arguni E, Horigome S, Arima M, et al. Bcl6 controls granzyme B expression in effector CD8+ T cells. Eur J Immunol. 2006;36(12):3146–3156. doi: 10.1002/eji.200636165. [DOI] [PubMed] [Google Scholar]
- 22.Scheeren FA, Naspetti M, Diehl S, Schotte R, Nagasawa M, Wijnands E, et al. STAT5 regulates the self-renewal capacity and differentiation of human memory B cells and controls Bcl-6 expression. Nat Immunol. 2005;6(3):303–313. doi: 10.1038/ni1172. [DOI] [PubMed] [Google Scholar]
- 23.Verdeil G, Puthier D, Nguyen C, Schmitt-Verhulst AM, Auphan-Anezin N. STAT5-mediated signals sustain a TCR-initiated gene expression program toward differentiation of CD8 T cell effectors. J Immunol. 2006;176(8):4834–4842. doi: 10.4049/jimmunol.176.8.4834. [DOI] [PubMed] [Google Scholar]
- 24.Hershkovitz L, Schachter J, Treves AJ, Besser MJ. Focus on adoptive T cell transfer trials in melanoma. Clin Dev Immunol. 2010:260267. doi: 10.1155/2010/260267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Lynch DH. The promise of 4-1BB (CD137)-mediated immunomodulation and the immunotherapy of cancer. Immunol Rev. 2008;222:277–286. doi: 10.1111/j.1600-065X.2008.00621.x. [DOI] [PubMed] [Google Scholar]
- 26.Melero I, Hirschhorn-Cymerman D, Morales-Kastresana A, Sanmamed MF, Wolchok JD. Agonist antibodies to TNFR molecules that costimulate T and NK cells. Clin Cancer Res. 2013;19(5):1044–1053. doi: 10.1158/1078-0432.CCR-12-2065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Watts TH. TNF/TNFR family members in costimulation of T cell responses. Annu Rev Immunol. 2005;23:23–68. doi: 10.1146/annurev.immunol.23.021704.115839. [DOI] [PubMed] [Google Scholar]
- 28.Benencia F, Sprague L, McGinty J, Pate M, Muccioli M. Dendritic cells the tumor microenvironment and the challenges for an effective antitumor vaccination. J Biomed Biotechnol. 2012:425476. doi: 10.1155/2012/425476. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Engelhardt JJ, Boldajipour B, Beemiller P, Pandurangi P, Sorensen C, Werb Z, et al. Marginating dendritic cells of the tumor microenvironment cross-present tumor antigens and stably engage tumor-specific T cells. Cancer Cell. 21(3):402–417. doi: 10.1016/j.ccr.2012.01.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Ma Y, Shurin GV, Peiyuan Z, Shurin MR. Dendritic cells in the cancer microenvironment. J Cancer. 4(1):36–44. doi: 10.7150/jca.5046. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.