Abstract
AIM: To detect the expression of Fas ligand (FasL) in colon cancer tissues and cell lines and analyze the function of FasL-expressing colon cancer cells in inducing Fas-sensitive T lymphocyte apoptosis.
METHODS: Ninety surgically resected colon cancer tissues and 15 hepatic metastasis specimens were investigated by immunohistochemical method with normal colon mucosa and colon adenoma as control. The relationship between FasL expression and pathologic features was also analyzed. FasL expression of 4 colon cancer cell lines, SW620, Lovo, LS-174T and SW1116, were detected by Western blotting assay. The function of FasL expressed on colon cancer cells was determined by coculture assay with Jurkat T lymphocytes, the apoptotic rate of which was detected by flow cytometry assay.
RESULTS: Fifty-six (62.22%) cases of all the 90 colon cancer tissues and all (100%) the liver metastasis specimens expressed FasL, significantly higher than normal colon mucosa and colonic adenoma. Higher expression of FasL was found in more advanced stage of colon cancer and in cancer tissues with lymphatic or hepatic metastasis. All the colon cancer cell lines were found to express FasL. After coculture with the SW1116 cells for 24 h with an effector: target ratio 10:1, the rate of apoptosis of Jurkat cells rose from 1.9% to 21.0%.
CONCLUSION: The expression of FasL is upregulated in colon cancer and the functionally expressed FasL can induce apoptosis of Fas-expressing T lymphocytes.
Keywords: Fas ligand, Colon cancer, Immune privilege
INTRODUCTION
Immune escape of tumor cells and successful tumor outgrowth may be due to the inability of the immune system to react to the tumor. Antigenetically distinct cancer must overcome the host’s antitumor immunity to develop. More evidence points to the expression of Fas ligand (FasL) as a possible mediator of tumor immune privilege[1,2]. FasL and its receptor (Fas) are a set of regulatory components in the immune system. Activation of the Fas by FasL results in apoptosis of many cell types. FasL upregulation has been found in many types of malignancy, including melanoma[3], lung cancer[4], breast cancer[5,6], cholangiocarcinoma and ovarian cancer[7]. Furthermore, various cancer cell lines express FasL and kill lymphoid cells by Fas-mediated apoptosis in vitro[13]. Thus, the expression of FasL may play an important role in tumor cell immune escape.
The aim of this study was to determine whether FasL expression in human colon cancer cells was upregulated and associated with increased apoptosis of lymphocytes.
MATERIALS AND METHODS
Cell lines and tissues
Human colonic cancer cell lines SW620, Lovo, LS-174T, SW1116 and Jurkat human T leukemia cell line were maintained in RPMI1640 supplemented with 100 mL/L fetal bovine serum in a humidified 50 mL/L CO2 atmosphere. Ninety human tissues of colon cancer and 15 hepatic metastasis specimens were collected during surgical resections performed at Changzheng Hospital. None of the patients received chemo-or radiotherapy prior to resection. Normal colon mucosa obtained during operation and colon adenoma resected under colonoscope served as control.
Immunohistochemical detection of FasL protein
Paraffin-embedded tumor sections were deparaffinized in xylene and rehydrated prior to analysis. Slides were washed twice for 5 min in a wash buffer. Endogenous peroxidase was quenched with 30 mL/L hydrogen peroxide in methanol for 5 min. Slides were washed as before, blocked for 1 h in wash buffer containing 50 mL/L normal goat serum, then incubated for 1 h at 37 °C with rabbit anti-human FasL-specific IgG. Biotin-conjugated goat anti-rabbit second antibody was added and incubated for 20 min at 37 °C. Then SABC complex was added and the slides were stained with DAB solution. A known sample from a patient with breast cancer was used as a positive control, and a staining for negative control was performed as above, except for incubation with PBS instead of the primary antibody. Cells with brown granule staining of the membrane/cytoplasm and slides with more than 25% staining cells were considered positive.
Western blot assay
Protein extracted from the colon cancer cells was separated on 12% SDS-polyacrylamide gel and then transferred to nitrocellulose membranes with a semi-dry apparatus. After the membranes were transferred, they were blocked for 1 h with 50 g/L nonfat milk in PBS. Membranes were incubated with murine monoclonal antibody to human FasL (1:500 dilution, Transduction Lab) overnight at 4 °C, washed thrice for 5 min with PBS before addition of goat anti-mouse HRP-conjugated secondary antibody (1:1000 dilution, Gene Company) for 1 h at room temperature. Membranes were washed thrice for 5 min with TBS, and the peroxidase activity on the nitrocellulose sheet was visualized on an X-ray film with the ECL Western blotting detection system.
Flow cytometry assay
Jurkat cells and those cocultured with SW1116 cells were assayed for DNA content using the propidium iodide staining method. Briefly, cells (2×106) were collected, rinsed in PBS twice and then incubated in 50 mg/L propidium iodide at 4 °C for 30 min in the dark. Cell cycle distribution at different phases was analyzed with FACScan flow cytometry. More than 10000 events were acquired for analysis.
Statistical analysis
χ2 test was used to analyze the relationship between FasL expression and clinical pathologic features of colon cancer. P-values less than 0.05 were considered statistically significant.
RESULTS
Expression of FasL in colon cancer tissues
No FasL expression was detected in all the normal colon mucosae. Among the 20 colon adenomas, 5 (25.00%) were considered positive. FasL expression was detected in 56 (62.22%) of the 90 colon cancer tissues and all (100%) of the liver metastasis specimens, significantly higher than the control group (P<0.05). The rate of FasL expression was not correlated with macroscopic type, microscopic feature and invasion depth of colon cancer. Higher expression of FasL was found in more advanced stage of colon cancer (P<0.05) and in cancer tissues with lymphatic or hepatic metastasis (P<0.05) (Table 1 and Figure 1).
Table 1.
Relationship between FasL expression and clinicopathologic features of colon cancer.
| Group | Cases | Positive | Negative | χ2 value | |
| Macroscopic type | Polypoid | 26 | 15 | 11 | 0.349 |
| Ulcerative | 37 | 24 | 13 | ||
| Infiltrative | 27 | 17 | 10 | ||
| Microscopic feature | Well differentiated | 9 | 3 | 6 | 3.706 |
| Moderately differentiated | 46 | 30 | 16 | ||
| Poorly differentiated | 22 | 15 | 7 | ||
| Mucoid adenocarcinoma | 13 | 8 | 5 | ||
| Invasion depth | Whole layer | 47 | 34 | 13 | 4.492 |
| Muscular layer | 32 | 17 | 15 | ||
| Mucosa and submucosa | 11 | 5 | 6 | ||
| Lymphatic metastasis | Positive | 56 | 43 | 13 | 13.375a |
| Negative | 34 | 13 | 21 | ||
| Dukes’ stage | A | 11 | 5 | 6 | 18.187a |
| B | 23 | 8 | 15 | ||
| C | 31 | 20 | 11 | ||
| D | 25 | 23 | 2 | ||
| Liver metastasis | Positive | 23 | 22 | 1 | 14.689a |
| Negative | 67 | 34 | 33 |
P<0.05 vs others.
Figure 1.
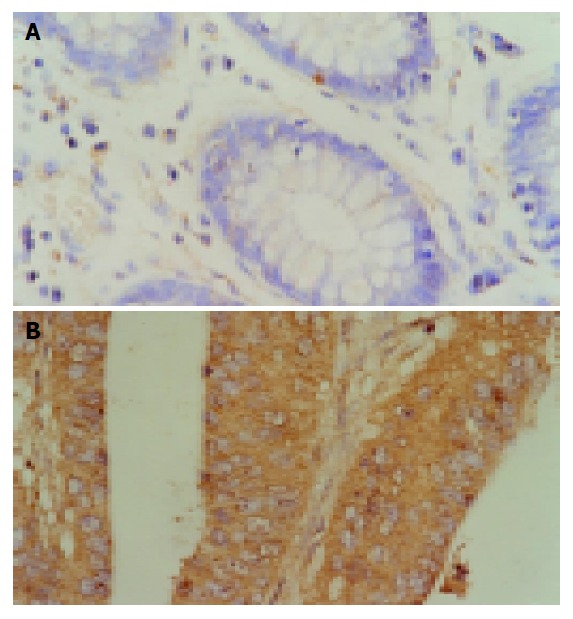
Expression of FasL in normal colon mucosae (A) and colon cancer (B).
Expression of FasL in colon cancer cell lines
Cell lysate of four cancer cell lines was analyzed using Western blot assay. All of them showed the FasL expression with different levels at 37 ku, and SW1116 exhibited the strongest signal (Figure 2).
Figure 2.
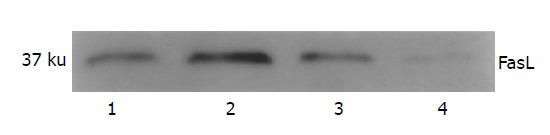
Western blotting result of the expression of FasL in colon cancer cell lines. Lane 1: SW620; lane 2: SW1116; lane 3: LS-174T; lane 4: Lovo.
Induction of lymphocyte apoptosis
Coculture test was performed between FasL-expressing SW1116 cells and Fas-sensitive Jurkat cells to determine whether FasL expressed in colonic cancer cell was functional. The apoptotic rate was 1.9% in Jurkat cells. After being cocultured with SW1116 cells for 24 h with an effector: target ratio 10:1, a marked increase in the hypodiploid DNA peak indicating the apoptosis of Jurkat cells was found. The rate of apoptosis rose to 21.0% (Figure 3).
Figure 3.
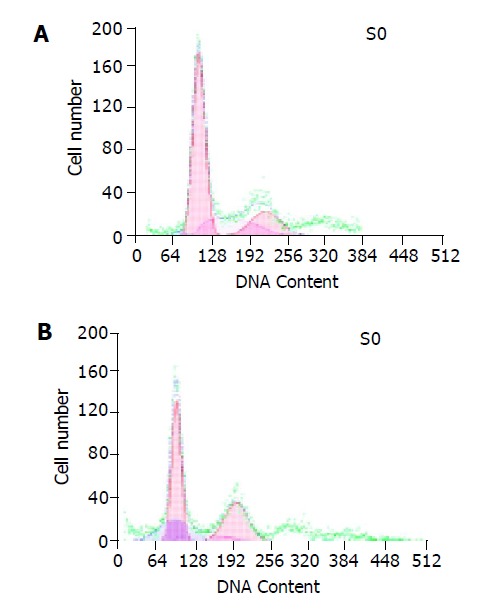
Apoptosis of Jurkat cells (A) and apoptosis of Jurkat cells after being cocultured with SW1116 cells (B).
DISCUSSION
FasL is a type-II transmembrane protein and belongs to the tumor necrosis factor family. Fas-FasL system has been implicated in the clonal depletion of autoreactive lymphocytes in peripheral lymphoid tissue and in the elimination of autoreactive lymphocyte population[8], thus contributing to homeostasis of the immune system. FasL expression can also induce potent inflammatory responses[9]. Its expression in normal tissue is restricted to T lymphocytes, macrophages, cornea, iris, ciliary bodies, retina, and Sertoli cells[10]. Through its ability to suppress both cellular and humoral immunity[11], FasL has been implicated in maintenance of the immune-privileged status in the eye[10] and testis[12].
The possibility of non-lymphoid tumors expressing FasL as a mediator of immune escape was initially raised by the finding that colon cancer cells express FasL to induce apoptosis of T lymphocytes and not of themselves[13]. The upregulation of FasL expression is found in gastrointestinal tract malignancy including esophageal cancer, stomach cancer, pancreatic cancer, hepatocellular carcinoma and colorectal cancer. Our result showed that most colon cancer specimens expressed FasL, a potent mediator of immunological privilege and immune response termination. The expression of FasL in colon cancer was significantly higher than normal colon mucosae and colonic adenoma, indicating that FasL expression may play an important role in tumorigenesis[14]. But the relationship between FasL expression and pathologic characteristics of colon cancer is still controversial[15,16]. In our group, FasL expression was significantly related with the Dukes’ stage, lymphatic and hepatic metastasis. Patients with more advanced colon cancer and those with lymphatic or hepatic metastasis have a more frequent FasL expression, indicating that FasL-positive colon cancer cells have the advantage of surviving from the immune clearance and progressing or metastasizing.
All the cell lines of colon cancer detected in our experiment also expressed FasL and showed the potential of killing Jurkat T lymphocytes in coculture experiment, indicating that tumor-expressed FasL is functional. FasL-expressing cell lines of colon cancer can induce Fas-mediated apoptosis of Fas-sensitive lymphoid target cells in vitro, suggesting the Fas counterattack model[13] as a mechanism of immune escape in colon cancer. It shows that tumor cells may exploit an intrinsic cell death program of activated T cells that infiltrate them. Essentially the cancer cells counterattack the activated cytotoxic T cells with one of the T cell’ s own principal cytotoxic elements: FasL. This mechanism is analogous to the established model of FasL in mediating immune privilege. Tumor cells expressing FasL may induce apoptosis of Fas-sensitive antitumor immune effector cells[18,19] without provoking inflammation reaction[17,20], thus rendering the tumor immune privilege[1].
However, resistance to Fas-mediated apoptosis is a prerequisite for “counterattack model” of cancer cells, as autocrine tumor cell suicide or juxtacrine/paracrine cell death could occur. Tumor cells can either downregulate the Fas expression or have a disabled intracellular Fas apoptotic signal transduction pathway[22-24], thus rendering resistance to Fas-mediated apoptosis. Since colon cancer cells and tumor infiltrating lymphocytes can express both Fas and FasL, they can act either as effectors or as targets in Fas/FasL-mediated killing. The survival of tumor or host cells may depend on which cells can accomplish the task more efficiently. Cancer cells are resistant to Fas-mediated apoptosis, but can induce Fas-sensitive tumor infiltrating lymphocytes to apoptosis. That is the core of “counterattack model” which may explain the mechanism of tumor cells escaping from immune surveillance.
We also found that all the liver metastases of colon cancer expressed FasL, in accord with the results that FasL is more frequently expressed in liver metastases than in primary carcinomas[25]. This may indicate that FasL expressed on the surface of colon cancer cells has an additional role in malignancy, namely, FasL may be important in the colonization of colon cancer cells in the liver[26], a major target of metastasis. Metastasis cascade is a complex series of processes including angiogenesis, intravasation of tumor cells, transport by circulation, adhesive interaction with endothelial cells, extravasation and colonization of the target organ[27,28]. FasL expression in colon cancer cells may induce apoptosis of tumor-infiltrating lymphocytes but not themselves[21]. This can help tumor cells escape from immune supervision. In addition, FasL may enhance liver colonization competence through induction of apoptosis in Fas-expressing hepatocytes at the metastatic foci which destroy the normal liver tissue[21] and make enough space for the growth of metastatic tumor cells.
Footnotes
Supported by the National Natural Science Foundation of China, No. 39900143
Science Editor Wang XL Language Editor Elsevier HK
References
- 1.O'Connell J, Bennett MW, O'Sullivan GC, Collins JK, Shanahan F. The Fas counterattack: cancer as a site of immune privilege. Immunol Today. 1999;20:46–52. doi: 10.1016/s0167-5699(98)01382-6. [DOI] [PubMed] [Google Scholar]
- 2.O'Connell J, Bennett MW, O'Sullivan GC, Collins JK, Shanahan F. The Fas counterattack: a molecular mechanism of tumor immune privilege. Mol Med. 1997;3:294–300. [PMC free article] [PubMed] [Google Scholar]
- 3.Redondo P, Solano T, VAzquez B, Bauza A, Idoate M. Fas and Fas ligand: expression and soluble circulating levels in cutaneous malignant melanoma. Br J Dermatol. 2002;147:80–86. doi: 10.1046/j.1365-2133.2002.04745.x. [DOI] [PubMed] [Google Scholar]
- 4.Niehans GA, Brunner T, Frizelle SP, Liston JC, Salerno CT, Knapp DJ, Green DR, Kratzke RA. Human lung carcinomas express Fas ligand. Cancer Res. 1997;57:1007–1012. [PubMed] [Google Scholar]
- 5.Ben-Hur H, Mordechay E, Halperin R, Gurevich P, Zandbank J, Herper M, Zusman I. Apoptosis-related proteins (Fas, Fas ligand, bcl-2 and p53) in different types of human breast tumors. Oncol Rep. 2002;9:977–980. [PubMed] [Google Scholar]
- 6.Mottolese M, Buglioni S, Bracalenti C, Cardarelli MA, Ciabocco L, Giannarelli D, Botti C, Natali PG, Concetti A, Venanzi FM. Prognostic relevance of altered Fas (CD95)-system in human breast cancer. Int J Cancer. 2000;89:127–132. doi: 10.1002/(sici)1097-0215(20000320)89:2<127::aid-ijc5>3.0.co;2-4. [DOI] [PubMed] [Google Scholar]
- 7.Abrahams VM, Straszewski SL, Kamsteeg M, Hanczaruk B, Schwartz PE, Rutherford TJ, Mor G. Epithelial ovarian cancer cells secrete functional Fas ligand. Cancer Res. 2003;63:5573–5581. [PubMed] [Google Scholar]
- 8.Brunner T, Mogil RJ, LaFace D, Yoo NJ, Mahboubi A, Echeverri F, Martin SJ, Force WR, Lynch DH, Ware CF. Cell-autonomous Fas (CD95)/Fas-ligand interaction mediates activation-induced apoptosis in T-cell hybridomas. Nature. 1995;373:441–444. doi: 10.1038/373441a0. [DOI] [PubMed] [Google Scholar]
- 9.Chen JJ, Sun Y, Nabel GJ. Regulation of the proinflammatory effects of Fas ligand (CD95L) Science. 1998;282:1714–1717. doi: 10.1126/science.282.5394.1714. [DOI] [PubMed] [Google Scholar]
- 10.Griffith TS, Brunner T, Fletcher SM, Green DR, Ferguson TA. Fas ligand-induced apoptosis as a mechanism of immune privilege. Science. 1995;270:1189–1192. doi: 10.1126/science.270.5239.1189. [DOI] [PubMed] [Google Scholar]
- 11.Arai H, Chan SY, Bishop DK, Nabel GJ. Inhibition of the alloantibody response by CD95 ligand. Nat Med. 1997;3:843–848. doi: 10.1038/nm0897-843. [DOI] [PubMed] [Google Scholar]
- 12.Bellgrau D, Gold D, Selawry H, Moore J, Franzusoff A, Duke RC. A role for CD95 ligand in preventing graft rejection. Nature. 1995;377:630–632. doi: 10.1038/377630a0. [DOI] [PubMed] [Google Scholar]
- 13.O'Connell J, O'Sullivan GC, Collins JK, Shanahan F. The Fas counterattack: Fas-mediated T cell killing by colon cancer cells expressing Fas ligand. J Exp Med. 1996;184:1075–1082. doi: 10.1084/jem.184.3.1075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.O'Connell J, Bennett MW, O'Sullivan GC, Collins JK, Shanahan F. Resistance to Fas (APO-1/CD95)-mediated apoptosis and expression of Fas ligand in esophageal cancer: the Fas counterattack. Dis Esophagus. 1999;12:83–89. doi: 10.1046/j.1442-2050.1999.00033.x. [DOI] [PubMed] [Google Scholar]
- 15.O'Connell J, Bennett MW, O'Sullivan GC, Roche D, Kelly J, Collins JK, Shanahan F. Fas ligand expression in primary colon adenocarcinomas: evidence that the Fas counterattack is a prevalent mechanism of immune evasion in human colon cancer. J Pathol. 1998;186:240–246. doi: 10.1002/(SICI)1096-9896(199811)186:3<240::AID-PATH173>3.0.CO;2-L. [DOI] [PubMed] [Google Scholar]
- 16.Zhu Q, Deng C. The role of Fas/Fas ligand in tumorgenesis, immune escape, and counterattack in colonic cancer. Zhonghua NeiKe ZaZhi. 2002;41:378–380. [PubMed] [Google Scholar]
- 17.Chen YL, Chen SH, Wang JY, Yang BC. Fas ligand on tumor cells mediates inactivation of neutrophils. J Immunol. 2003;171:1183–1191. doi: 10.4049/jimmunol.171.3.1183. [DOI] [PubMed] [Google Scholar]
- 18.Bennett MW, O'Connell J, O'Sullivan GC, Brady C, Roche D, Collins JK, Shanahan F. The Fas counterattack in vivo: apoptotic depletion of tumor-infiltrating lymphocytes associated with Fas ligand expression by human esophageal carcinoma. J Immunol. 1998;160:5669–5675. [PubMed] [Google Scholar]
- 19.Strand S, Hofmann WJ, Hug H, Müller M, Otto G, Strand D, Mariani SM, Stremmel W, Krammer PH, Galle PR. Lymphocyte apoptosis induced by CD95 (APO-1/Fas) ligand-expressing tumor cells--a mechanism of immune evasion? Nat Med. 1996;2:1361–1366. doi: 10.1038/nm1296-1361. [DOI] [PubMed] [Google Scholar]
- 20.Houston A, Bennett MW, O'Sullivan GC, Shanahan F, O'Connell J. Fas ligand mediates immune privilege and not inflammation in human colon cancer, irrespective of TGF-beta expression. Br J Cancer. 2003;89:1345–1351. doi: 10.1038/sj.bjc.6601240. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Yoong KF, Afford SC, Randhawa S, Hubscher SG, Adams DH. Fas/Fas ligand interaction in human colorectal hepatic metastases: A mechanism of hepatocyte destruction to facilitate local tumor invasion. Am J Pathol. 1999;154:693–703. doi: 10.1016/S0002-9440(10)65316-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Lee SH, Kim HS, Kim SY, Lee YS, Park WS, Kim SH, Lee JY, Yoo NJ. Increased expression of FLIP, an inhibitor of Fas-mediated apoptosis, in stomach cancer. APMIS. 2003;111:309–314. doi: 10.1034/j.1600-0463.2003.1110203.x. [DOI] [PubMed] [Google Scholar]
- 23.Bäumler C, Duan F, Onel K, Rapaport B, Jhanwar S, Offit K, Elkon KB. Differential recruitment of caspase 8 to cFlip confers sensitivity or resistance to Fas-mediated apoptosis in a subset of familial lymphoma patients. Leuk Res. 2003;27:841–851. doi: 10.1016/s0145-2126(03)00018-3. [DOI] [PubMed] [Google Scholar]
- 24.Snow AL, Chen LJ, Nepomuceno RR, Krams SM, Esquivel CO, Martinez OM. Resistance to Fas-mediated apoptosis in EBV-infected B cell lymphomas is due to defects in the proximal Fas signaling pathway. J Immunol. 2001;167:5404–5411. doi: 10.4049/jimmunol.167.9.5404. [DOI] [PubMed] [Google Scholar]
- 25.Mann B, Gratchev A, Böhm C, Hanski ML, Foss HD, Demel G, Trojanek B, Schmidt-Wolf I, Stein H, Riecken EO, et al. FasL is more frequently expressed in liver metastases of colorectal cancer than in matched primary carcinomas. Br J Cancer. 1999;79:1262–1269. doi: 10.1038/sj.bjc.6690202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Shiraki K, Tsuji N, Shioda T, Isselbacher KJ, Takahashi H. Expression of Fas ligand in liver metastases of human colonic adenocarcinomas. Proc Natl Acad Sci USA. 1997;94:6420–6425. doi: 10.1073/pnas.94.12.6420. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Hanahan D, Folkman J. Patterns and emerging mechanisms of the angiogenic switch during tumorigenesis. Cell. 1996;86:353–364. doi: 10.1016/s0092-8674(00)80108-7. [DOI] [PubMed] [Google Scholar]
- 28.Fidler IJ. Critical factors in the biology of human cancer metastasis: twenty-eighth G.H.A. Clowes memorial award lecture. Cancer Res. 1990;50:6130–6138. [PubMed] [Google Scholar]


