Abstract
Background
The UK has poor lung cancer survival rates and high early mortality, compared to other countries. We aimed to identify factors associated with early death, and features of primary care that might contribute to late diagnosis.
Methods
All cases of lung cancer diagnosed between 2000 and 2013 were extracted from The Health Improvement Network database. Patients who died within 90 days of diagnosis were compared with those who survived longer. Standardised chest X-ray (CXR) and lung cancer rates were calculated for each practice.
Results
Of 20 142 people with lung cancer, those who died early consulted with primary care more frequently prediagnosis. Individual factors associated with early death were male sex (OR 1.17; 95% CI 1.10 to 1.24), current smoking (OR 1.43; 95% CI 1.28 to 1.61), increasing age (OR 1.80; 95% CI 1.62 to 1.99 for age ≥80 years compared to 65–69 years), social deprivation (OR 1.16; 95% CI 1.04 to 1.30 for Townsend quintile 5 vs 1) and rural versus urban residence (OR 1.22; 95% CI 1.06 to 1.41). CXR rates varied widely, and the odds of early death were highest in the practices which requested more CXRs. Lung cancer incidence at practice level did not affect early deaths.
Conclusions
Patients who die early from lung cancer are interacting with primary care prediagnosis, suggesting potentially missed opportunities to identify them earlier. A general increase in CXR requests may not improve survival; rather, a more timely and appropriate targeting of this investigation using risk assessment tools needs further assessment.
Keywords: Lung Cancer, Clinical Epidemiology
Key messages.
What is the key question?
What are the individual risk factors for early death from lung cancer in the UK, and are there any primary care features which may contribute to this?
What is the bottom line?
Thirty per cent of patients with lung cancer died within 90 days of diagnosis, with increasing age, male sex, socioeconomic deprivation, rural versus urban location and current smoking all independently associated with early death.
Why read on?
This is the first paper to look in detail at reasons for the excess early lung cancer mortality in the UK, highlighting that those with poorer prognosis are interacting with primary care before diagnosis and suggesting missed opportunities to identify them earlier.
Introduction
The UK has poor lung cancer survival rates compared to other countries,1–3 and this is partly explained by differences in early mortality.2 In the UK, patients normally consult their general practitioner (GP) prior to a secondary care referral, and so it is important to understand the factors in primary care that might lead to late diagnosis and consequent high early mortality from lung cancer.
We hypothesised that older, more socially deprived and isolated males, would be less likely to consult their GPs and, hence, would be at increased risk of early death from lung cancer. Additionally, we reasoned that people who were registered at practices in areas of high lung cancer incidence might be recognised earlier by their GPs, and that GPs who, in general, requested more chest X-rays (CXR), would identify and refer their patients earlier, with more favourable outcomes.
To address these questions, we used a large primary care dataset to identify patient and primary care features associated with death-certificate-only diagnoses and 30-day and 90-day mortality after a diagnosis of lung cancer.
Methods
Dataset
The Health Improvement Network (THIN) is a computerised, longitudinal, primary care dataset, which has been previously validated for lung cancer outcomes in addition to other major comorbid illnesses and exposures.4–11 All symptoms, medical diagnoses, prescriptions, investigations and results are entered into the computer system either during a consultation with a GP or following communication from secondary care.
Study population
Cases of lung cancer were extracted for the period January 2000–January 2013. To ensure that these were incident rather than prevalent, we excluded patients who registered less than 12 months prior to their diagnosis date.
Date of diagnosis and death were used to calculate the proportion of patients who died early (cases). This included people whose date of diagnosis and date of death were the same (‘death certificate only diagnoses’), and those who died within 30 or 90 days of diagnosis. Those who survived longer than 90 days were our comparator group.
Covariate descriptions
Smoking status was defined as current, ex, never or missing.12 Where there were multiple smoking records, the most recent record was used, but excluding the 6 months before the diagnosis date, to exclude changes related to symptomatic disease. The presence of comorbidities before lung cancer diagnosis was identified using Read codes and weighted to calculate a Charlson comorbidity index, using our established method.13–15
Townsend Index of deprivation, divided into quintiles, was used as a measure of socioeconomic status. Social isolation was defined using the household variable in THIN which identifies people living at the same address. Where only one person had a particular household identifier they were defined as living alone. Since individuals living in different flats or apartments within the same building may be given the same household identifier, if there were greater than 10 people with the same household identifier they were classified as unknown. We also identified urban versus rural residence, using a census-derived variable in the dataset, which classified areas into those with >10 000 inhabitants (urban), town and fringe, rural and unknown.
Primary care consultations and CXRs
We identified all records of consultations in the prediagnosis period, using codes which identified ‘face-to-face’ or telephone consultations. Multiple entries on the same date were counted as one consultation only. In previous work, we identified the period of 0–4 months from diagnosis date as the period during which GPs investigate people for lung cancer,12 so for our analysis we looked at the numbers of consultations in this time period. Patients were then grouped as low (0–5), medium (6–10), and high frequency consulters (>10). We identified all CXR requests for cases and controls in the 0–4-months prior to diagnosis. If there were multiple requests for the same person we counted only one. We combined this with consultation frequency to produce a composite variable indicating the number of times an individual had consulted in primary care (low, medium or high as described above) and also whether or not they had a CXR requested by their GP during the 0–4-month time period.
Practice level variables
We calculated annual standardised overall CXR requesting rates for each primary care practice between 1 January 2010 and 31 December 2012 as a marker of their general propensity to order CXRs. This time period was chosen as all practices included in the analysis were contributing and the level of data completeness was high. We standardised the rate of CXRs using the THIN population structure on 1 July 2010. We also calculated background lung cancer incidence rates as proxy markers of exposure/awareness using the same process.
Statistical methods
Statistical analyses were performed using Stata/MP, V.12 (StataCorp LP) software. Ethical approval for the study was provided by the Cegedim Strategic Data Medical Research Scientific Review Committee (Ref 13–015).
We calculated the proportion of people who died immediately (death-certificate-only diagnoses), within 30 days and within 90 days of their diagnosis within groups according to the demographic covariates, consultations and practice level variables described above. Kaplan–Meier survival curve was plotted for the whole population. Logistic regression was used to calculate ORs for death-certificate-only diagnosis, 0–30-day mortality (death certificate only +30) and 0–90-day mortality (death certificate only +90) within each of the variables described above and listed in table 1. We looked at each variable individually initially, then included those which showed an association in the univariate model (p<0.05) in the multivariate analysis. Likelihood ratio testing was used for all tests of significance.
Table 1.
Demographics of the lung cancer population
| Death certificate only | Died 1–30 days | Died 31–90 days | Comparator group | |
|---|---|---|---|---|
| 1071 (%) | 2036 (%) | 2976 (%) | 14 059 (%) | |
| Age at diagnosis (years) | ||||
| 30–39 | 1 (0.09) | 1 (0.10) | 4 (0.13) | 83 (0.59) |
| 40–44 | 8 (0.75) | 11 (0.54) | 16 (0.54) | 138 (0.98) |
| 45–49 | 15 (1.40) | 22 (1.08) | 41 (1.38) | 306 (2.18) |
| 50–54 | 33 (3.08) | 44 (2.16) | 77 (2.59) | 610 (4.34) |
| 55–59 | 45 (4.20) | 127 (6.24) | 196 (6.59) | 1194 (8.49) |
| 60–64 | 74 (6.91) | 197 (9.68) | 335 (11.26) | 1868 (13.29) |
| 65–69 | 114 (10.64) | 294 (14.44) | 416 (13.98) | 2202 (15.66) |
| 70–74 | 173 (16.15) | 312 (15.32) | 523 (17.57) | 2442 (17.37) |
| 75–79 | 228 (21.29) | 379 (18.61) | 612 (20.56) | 2410 (17.14) |
| >80 | 380 (35.48) | 648 (31.83) | 756 (25.40) | 1806 (19.96) |
| Sex | ||||
| Male | 634 (59.20) | 1174 (57.66) | 1777 (59.71) | 7883 (56.07) |
| Female | 437 (40.80) | 862 (42.34) | 1199 (40.29) | 6176 (43.93) |
| Townsend deprivation quintile | ||||
| 1 | 165 (15.41) | 356 (17.49) | 514 (17.27) | 2510 (17.85) |
| 2 | 190 (17.74) | 392 (19.25) | 550 (18.48) | 2695 (19.17) |
| 3 | 201 (18.77) | 428 (21.02) | 629 (21.14) | 1855 (20.31) |
| 4 | 269 (25.12) | 435 (21.37) | 659 (22.14) | 3088 (21.96) |
| 5 (most) deprived) | 224 (20.92) | 382 (18.76) | 548 (18.41) | 2453 (17.45) |
| Missing | 22 (2.05) | 43 (2.11) | 76 (2.55) | 458 (3.26) |
| Smoking status | ||||
| Never smoker | 158 (14.75) | 194 (9.53) | 226 (7.59) | 1356 (9.65) |
| Ex-smoker | 378 (35.29) | 783 (38.46) | 1330 (44.69) | 7007 (49.84) |
| Current smoker | 423 (39.50) | 874 (42.93) | 1272 (42.74) | 5149 (36.62) |
| Missing | 112 (10.46) | 185 (9.09) | 148 (4.97) | 547 (3.89) |
| Charlson score | ||||
| 0 | 415 (38.75) | 880 (43.22) | 1260 (42.34) | 6226 (44.28) |
| 1 | 264 (24.65) | 471 (23.13) | 701 (23.56) | 3435 (24.43) |
| 2–3 | 297 (27.73) | 488 (23.97) | 768 (25.81) | 3385 (24.08) |
| >4 | 95 (8.87) | 197 (9.68) | 247 (8.30) | 1013 (7.21) |
| Consultations and CXR* | ||||
| Low, no CXR | 396 (36.97) | 1032 (33.22) | 913 (30.68) | 3889 (27.66) |
| Medium, no CXR | 208 (19.42) | 513 (16.51) | 404 (13.58) | 1317 (9.37) |
| High, no CXR | 180 (16.81) | 478 (15.38) | 328 (11.02) | 1571 (11.17) |
| Low, CXR | 111 (10.36) | 486 (15.64) | 688 (23.12) | 4348 (30.93) |
| Medium, CXR | 98 (9.15) | 399 (12.84) | 454 (15.26) | 1937 (13.78) |
| High, CXR | 78 (7.28) | 199 (6.40) | 189 (6.35) | 997 (7.09) |
| Number in household | ||||
| Lives alone | 299 (27.92) | 609 (29.91) | 932 (31.32) | 4157 (29.57) |
| >1 person | 696 (64.99) | 1313 (64.49) | 1924 (64.65) | 9405 (66.90) |
| Unknown | 76 (7.10) | 114 (5.60) | 120 (4.03) | 497 (3.54) |
| Urban vs rural | ||||
| Urban | 750 (70.03) | 1437 (70.58) | 2035 (68.38) | 9468 (67.34) |
| Town and fringe | 147 (13.73) | 205 (10.07) | 314 (10.55) | 1411 (10.04) |
| Rural | 74 (6.91) | 116 (5.70) | 152 (5.11) | 684 (4.87) |
| Unknown | 100 (9.34) | 278 (13.65) | 475 (15.96) | 2496 (17.75) |
| Practice CXR quartile (per 100 population) | ||||
| 1–2.73 | 179 (16.71) | 398 (19.55) | 600 (20.16) | 3108 (22.11) |
| 2.74–3.84 | 144 (13.45) | 451 (22.15) | 661 (22.21) | 3201 (22.77) |
| 3.85–5.33 | 354 (33.05) | 527 (25.88) | 778 (26.14) | 3635 (25.86) |
| ≥5.34 | 394 (36.79) | 660 (32.42) | 937 (31.49) | 4115 (29.27) |
| Practice lung cancer quartile (per 100 000 population) | ||||
| <34.3 | 191 (17.83) | 397 (19.50) | 538 (18.08) | 2653 (18.86) |
| 34.3–49.56 | 262 (24.46) | 568 (27.90) | 774 (26.01) | 3699 (26.31) |
| 49.57–71.63 | 298 (27.82) | 570 (28.00) | 809 (27.18) | 3710 (26.39) |
| ≥71.64 | 320 (29.88) | 501 (24.60) | 855 (28.73) | 3998 (28.44) |
*0–4 months prediagnosis.
CXR, chest X-ray.
Since CXR and lung cancer incidence quartiles were grouped by practice, we explored the use of Huber's method of robust regression to allow for underestimates in the SE.16 There was minimal change in ORs and 95% CIs, so the results of the original multivariate analysis are presented.
Results
Descriptive statistics
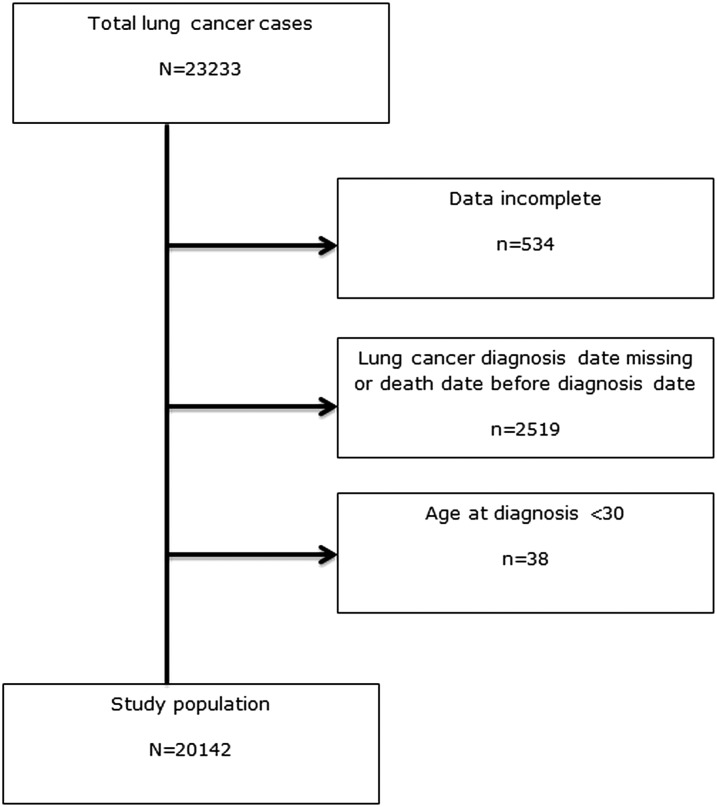
There were 29 073 patients with lung cancer in the initial THIN extract. We plotted the standardised CXR rates and those practices with rates of <1/100 population seemed to be distinct outliers, and so all patients registered with any of these 71 practices were excluded from further analysis as it seemed likely to us that GPs were not routinely coding the CXRs that they were requesting. This left 23 233 records. A further 3053 records with missing demographic data (year of birth, sex or invalid practice registration dates) or missing lung cancer diagnosis dates were also excluded. We also excluded 38 patients aged less than 30 years, as lung cancer is rare in this age group and may reflect a slightly different disease process. After these exclusions 20 142 cases of lung cancer from 444 general practices were used for the analysis (figure 1).
Figure 1.
Flowchart of inclusion and exclusion criteria for the health improvement network (THIN) dataset.
Five per cent (1071) of the cohort had death-certificate-only diagnosis, 10% (2036) died between 1 and 30 days of diagnosis, and a further 15% died between 31 and 90 days of diagnosis (table 1).
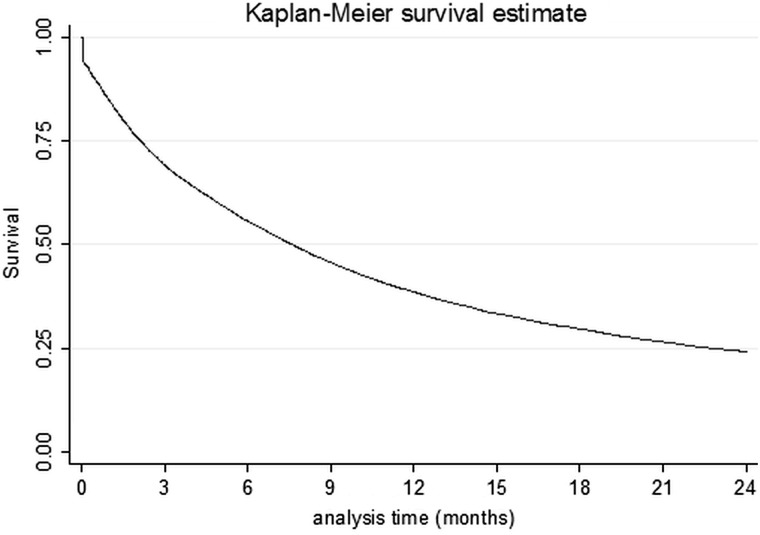
The Kaplan–Meier survival curve for the whole lung cancer population (truncated at 2 years postdiagnosis) is shown in figure 2.
Figure 2.
Kaplan–Meier plot for the whole study population.
Patient level variables
Overall, 57% (11 468) were male, with a median age at diagnosis of 72 years. Patients who were 80 years and over comprised 35% of the death-certificate-only group, 32% of the 1–30-day mortality group and 26% of the 31–90-day group. People who died early were more often current smokers (40% in the death-certificate-only group; 43% in both 1–30-day and 31–90-day mortality groups; and 36% in the control group) but less likely to be ex-smokers (35%, 38%, 45% and 50%, respectively). The death-certificate-only group had the highest proportion of never smokers (15%). More of those who died early were from the most socially deprived Townsend quintile; most marked in the death-certificate-only group (21% were in Townsend quintile 5 compared with 17% of controls) (table 1).
ORs followed similar trends across each of the three early death groups, so we elected to report results for the death-certificate-only +90 group for the remainder of this section. The univariate analysis for all groups is presented in table 2, with the multivariate analysis results for all groups presented in table 3.
Table 2.
Univariate ORs for death-certificate-only, death certificate +30-day mortality and Death certificate +90-day mortality groups
| OR | p Value (LRT) | OR | p Value (LRT) | OR | p Value (LRT) | |
|---|---|---|---|---|---|---|
| Death-certificate-only (95% CI) |
Death certificate +30 (95% CI) |
Death certificate +90 (95% CI) | ||||
| Age at diagnosis (years) | ||||||
| 30–39 | 0.23 (0.03 to 1.69) | 0.22 (0.07 to 0.70) | 0.23 (0.10 to 0.50) | |||
| 40–44 | 1.12 (0.54 to 2.34) | 0.79 (0.49 to 1.29) | 0.68 (0.46 to 0.99) | |||
| 45–49 | 0.95 (0.55 to 1.64) | 0.68 (0.48 to 0.98) | 0.68 (0.52 to 0.88) | |||
| 50–54 | 1.04 (0.70 to 1.56) | 0.72 (0.56 to 0.93) | 0.67 (0.56 to 0.82) | |||
| 55–59 | 0.73 (0.51 to 1.04) | 0.79 (0.66 to 0.96) | 0.82 (0.71 to 0.95) | |||
| 60–64 | 0.77 (0.57 to 1.03) | 0.79 (0.67 to 0.93) | 0.87 (0.77 to 0.98) | |||
| 65–69 | 1 | <0.001 | 1 | <0.001 | 1 | <0.001 |
| 70–74 | 1.37 (1.07 to 1.75) | 1.05 (0.91 to 1.21) | 1.10 (0.99 to 1.23) | |||
| 75–79 | 1.83 (1.45 to 2.30) | 1.29 (1.13 to 1.48) | 1.35 (1.22 to 1.50) | |||
| >80 | 2.62 (2.11 to 3.25) | 1.85 (1.63 to 2.10) | 1.70 (1.54 to 1.88) | |||
| Sex | ||||||
| Male | 1.14 (1.00 to 1.29) | 1.06 (0.98 to 1.15) | 1.12 (1.06 to 1.20) | |||
| Female | 1 | 0.046 | 1 | 0.124 | 1 | <0.001 |
| Townsend deprivation quintile | ||||||
| 1 | 1 | 0.001 | 1 | 0.003 | 1 | 0.001 |
| 2 | 1.07 (0.86 to 1.33) | 1.04 (0.92 to 1.18) | 1.02 (0.92 to 1.13) | |||
| 3 | 1.07 (0.87 to 1.32) | 1.05 (0.92 to 1.19) | 1.07 (0.97 to 1.18) | |||
| 4 | 1.33 (1.08 to 1.62) | 1.09 (0.96 to 1.23) | 1.07 (0.97 to 1.18) | |||
| 5 | 1.39 (1.13 to 1.71) | 1.17 (1.03 to 1.33) | 1.14 (1.03 to 1.26) | |||
| Missing | 0.73 (0.46 to 1.15) | 0.71 (0.54 to 0.93) | 0.75 (0.38 to 0.44) | |||
| Smoking status | ||||||
| Never smoker | 1 | <0.001 | 1 | <0.001 | 1 | <0.001 |
| Ex-smoker | 0.46 (0.38 to 0.56) | 0.63 (0.55 to 0.71) | 0.83 (0.75 to 0.93) | |||
| Current smoker | 0.71 (0.58 to 0.85) | 0.91 (0.80 to 1.03) | 1.17 (1.05 to 1.30) | |||
| Missing | 1.76 (1.35 to 2.28) | 1.92 (1.61 to 2.30) | 1.91 (1.63 to 2.24) | |||
| Charlson score | ||||||
| 0 | 1 | 0.001 | 1 | 0.001 | 1 | <0.001 |
| 1 | 1.15 (0.98 to 1.36) | 1.03 (0.93 to 1.13) | 1.02 (0.94 to 1.10) | |||
| 2–3 | 1.32 (1.13 to 1.54) | 1.09 (0.99 to 1.20) | 1.12 (1.04 to 1.21) | |||
| >4 | 1.41 (1.11 to 1.78) | 1.34 (1.16 to 1.54) | 1.30 (1.16 to 1.45) | |||
| Consultations and CXR* | ||||||
| Low, no CXR | 1 | <0.001 | 1 | <0.001 | 1 | <0.001 |
| Medium, no CXR | 1.41 (1.18 to 1.68) | 1.39 (1.23 to 1.56) | 1.39 (1.26 to 1.54) | |||
| High, no CXR | 1.13 (0.94 to 1.35) | 1.17 (1.04 to 1.32) | 1.03 (0.93 to 1.13) | |||
| Low, CXR | 0.28 (0.23 to 0.35) | 0.45 (0.40 to 0.50) | 0.54 (0.50 to 0.59) | |||
| Medium, CXR | 0.50 (0.40 to 0.63) | 0.78 (0.68 to 0.88) | 0.88 (0.80 to 0.97) | |||
| High, CXR | 0.82 (0.64 to 1.05) | 0.78 (0.66 to 0.92) | 0.78 (0.68 to 0.89) | |||
| Number in household | ||||||
| Lives alone | 1 | <0.001 | 1 | <0.001 | 1 | <0.001 |
| >1 person | 1.03 (0.89 to 1.18) | 0.99 (0.91 to 1.08) | 0.94 (0.88 to 1.01) | |||
| Unknown | 2.13 (1.63 to 2.78) | 1.73 (1.45 to 2.06) | 1.41 (1.21 to 1.64) | |||
| Urban vs rural | ||||||
| Urban | 1 | <0.001 | 1 | <0.001 | 1 | <0.001 |
| Town and fringe | 1.32 (1.09 to 1.58) | 1.07 (0.95 to 1.21) | 1.06 (0.96 to 1.17) | |||
| Rural | 1.37 (1.06 to 1.76) | 1.07 (1.01 to 1.41) | 1.12 (0.98 to 1.28) | |||
| Unknown | 0.51 (0.41 to 0.63) | 0.67 (0.60 to 0.75) | 0.77 (0.70 to 0.84) | |||
| Practice CXR quartile (per 100 population) | ||||||
| 1–2.73 | 1 | <0.001 | 1 | <0.001 | 1 | <0.001 |
| 2.74–3.84 | 0.78 (0.62 to 0.98) | 0.99 (0.88 to 1.12) | 1.04 (0.94 to 1.14) | |||
| 3.85–5.33 | 1.69 (1.40 to 2.04) | 1.28 (1.15 to 1.44) | 1.21 (1.01 to 1.32) | |||
| ≥5.34 | 1.66 (1.38 to 2.00) | 1.34 (1.20 to 1.50) | 1.28 (1.17 to 1.39) | |||
| Practice lung cancer quartile (per 100 000 population) | ||||||
| <34.3 | 1 | 0.338 | 1 | 0.101 | 1 | 0.299 |
| 34.3–49.56 | 0.98 (0.81 to 1.19) | 1.01 (0.90 to 1.13) | 1.02 (0.93 to 1.12) | |||
| 49.57–71.63 | 1.12 (0.92 to 1.35) | 1.04 (0.93 to 1.17) | 1.06 (0.97 to 1.17) | |||
| ≥71.64 | 1.11 (0.92 to 1.34) | 0.92 (0.82 to 1.03) | 0.99 (0.90 to 1.08) | |||
*0–4 months prediagnosis.
CXR, chest X-ray.
Table 3.
Multivariate ORs for death-certificate-only, death-certificate +30-day mortality and death certificate +90-day mortality groups
| OR | p Value (LRT) | OR | p Value (LRT) | OR | p Value (LRT) | |
|---|---|---|---|---|---|---|
| Death-certificate-only (95% CI) | Death certificate +30 (95% CI) | Death certificate +90 (95% CI) | ||||
| Age at diagnosis (years) | ||||||
| 30–39 | 0.23 (0.03 to 1.71) | 0.19 (0.06 to 0.61) | 0.20 (0.09 to 0.45) | |||
| 40–44 | 1.24 (0.59 to 2.61) | 0.74 (0.45 to 1.22) | 0.64 (0.43 to 0.94) | |||
| 45–49 | 0.96 (0.55 to 1.67) | 0.61 (0.43 to 0.88) | 0.62 (0.48 to 0.81) | |||
| 50–54 | 1.16 (0.77 to 1.73) | 0.69 (0.53 to 0.89) | 0.64 (0.53 to 0.78) | |||
| 55–59 | 0.78 (0.55 to 1.11) | 0.79 (0.65 to 0.95) | 0.81 (0.70 to 0.94) | |||
| 60–64 | 0.83 (0.62 to 1.12) | 0.81 (0.69 to 0.96) | 0.88 (0.78 to 1.00) | |||
| 65–69 | 1 | <0.001 | 1 | <0.001 | 1 | <0.001 |
| 70–74 | 1.38 (1.08 to 1.77) | 1.07 (0.93 to 1.24) | 1.13 (1.01 to 1.26) | |||
| 75–79 | 1.73 (1.37 to 2.19) | 1.30 (1.13 to 1.50) | 1.39 (1.25 to 1.55) | |||
| >80 | 2.28 (1.83 to 2.85) | 1.90 (1.66 to 2.16) | 1.80 (1.62 to 1.99) | |||
| Sex | ||||||
| Male | 1.21 (1.06 to 1.37) | 1.12 (1.04 to 1.22) | 1.17 (1.10 to 1.24) | |||
| Female | 1 | 0.004 | 1 | 0.005 | 1 | <0.001 |
| Townsend deprivation quintile | ||||||
| 1 | 1 | <0.001 | 1 | 0.005 | 1 | 0.017 |
| 2 | 1.06 (0.85 to 1.32) | 1.03 (0.90 to 1.18) | 1.00 (0.91 to 1.11) | |||
| 3 | 1.12 (0.90 to 1.39) | 1.08 (0.95 to 1.23) | 1.07 (0.96 to 1.18) | |||
| 4 | 1.44 (1.17 to 1.78) | 1.14 (1.00 to 1.30) | 1.07 (0.96 to 1.18) | |||
| 5 | 1.57 (1.25 to 1.97) | 1.28 (1.11 to 1.47) | 1.16 (1.04 to 1.30) | |||
| Missing | 1.35 (0.82 to 2.22) | 0.92 (0.68 to 1.25) | 0.86 (0.69 to 1.08) | |||
| Smoking status | ||||||
| Never smoker | 1 | <0.001 | 1 | <0.001 | 1 | <0.001 |
| Ex-smoker | 0.48 (0.39 to 0.58) | 0.66 (0.58 to 0.76) | 0.87 (0.78 to 0.97) | |||
| Current smoker | 0.80 (0.65 to 0.98) | 1.14 (1.00 to 1.31) | 1.43 (1.28 to 1.61) | |||
| Missing | 1.31 (1.00 to 1.71) | 1.85 (1.53 to 2.23) | 1.89 (1.60 to 2.23) | |||
| Charlson score | ||||||
| 0 | 1 | 0.125 | 1 | 0.168 | 1 | 0.234 |
| 1 | 1.17 (0.99 to 1.38) | 1.02 (0.92 to 1.13) | 0.96 (0.89 to 1.05) | |||
| 2–3 | 1.20 (1.02 to 1.41) | 1.03 (0.93 to 1.14) | 1.02 (0.94 to 1.11) | |||
| >4 | 1.10 (0.87 to 1.41) | 1.19 (1.02 to 1.38) | 1.09 (0.97 to 1.24) | |||
| Consultations and CXR* | ||||||
| Low, no CXR | 1 | <0.001 | 1 | <0.001 | 1 | <0.001 |
| Medium, no CXR | 1.43 (1.19 to 1.71) | 1.43 (1.27 to 1.62) | 1.43 (1.29 to 1.59) | |||
| High, no CXR | 1.05 (0.86 to 1.27) | 1.06 (0.94 to 1.21) | 0.96 (0.86 to 1.06) | |||
| Low, CXR | 0.28 (0.23 to 0.35) | 0.45 (0.40 to 0.51) | 0.54 (0.49 to 0.59) | |||
| Medium, CXR | 0.50 (0.40 to 0.63) | 0.80 (0.70 to 0.91) | 0.90 (0.82 to 1.00) | |||
| High, CXR | 0.88 (0.68 to 1.13) | 0.83 (0.70 to 0.98) | 0.81 (0.71 to 0.93) | |||
| Number in household | ||||||
| Lives alone | 1 | 0.025 | 1 | 0.019 | 1 | 0.204 |
| >1 person | 1.17 (1.01 to 1.36) | 0.035† | 1.09 (1.00 to 1.19) | 0.057† | 1.01 (0.94 to 1.08) | 0.912† |
| Unknown | 1.39 (1.05 to 1.84) | 1.28 (1.06 to 1.55) | 1.16 (0.98 to 1.36) | |||
| Urban vs rural | ||||||
| Urban | 1 | <0.001 | 1 | <0.001 | 1 | <0.001 |
| Town and fringe | 1.60 (1.32 to 1.95) | 1.13 (0.99 to 1.28) | 1.10 (0.99 to 1.22) | |||
| Rural | 1.69 (1.30 to 2.20) | 1.31 (1.10 to 1.56) | 1.22 (1.06 to 1.41) | |||
| Unknown | 0.50 (0.40 to 0.64) | 0.68 (0.60 to 0.78) | 0.79 (0.71 to 0.87) | |||
| Practice CXR quartile (per 100 population) | ||||||
| 1–2.73 | 1 | <0.001 | 1 | <0.001 | 1 | <0.001 |
| 2.74–3.84 | 0.71 (0.56 to 0.89) | 0.96 (0.84 to 1.09) | 1.03 (0.93 to 1.14) | |||
| 3.85–5.33 | 1.72 (1.42 to 2.09) | 1.38 (1.22 to 1.56) | 1.28 (1.16 to 1.40) | |||
| ≥5.34 | 1.78 (1.47 to 2.16) | 1.52 (1.35 to 1.71) | 1.41 (1.29 to 1.55) | |||
| Practice lung cancer quartile (per 100 000 population) | ||||||
| <34.3 | 1 | 0.522 | 1 | 0.009 | 1 | 0.064 |
| 34.3–49.56 | 0.92 (0.76 to 1.12) | 0.97 (0.86 to 1.10) | 0.98 (0.89 to 1.08) | |||
| 49.57–71.63 | 1.05 (0.86 to 1.28) | 1.02 (0.90 to 1.15) | 1.05 (0.95 to 1.15) | |||
| ≥71.64 | 1.01 (0.83 to 1.24) | 0.85 (0.75 to 0.97) | 0.93 (0.84 to 1.03) | |||
*0–4 months prediagnosis.
†Excluding unknown.
CXR, chest X-ray
Increasing age was strongly related to early death, with an adjusted OR of 1.80 (95% CI 1.62 to 1.99), in people aged ≥80 years compared with those aged 65–69 years. Ex-smokers were less likely to die early compared with never smokers; adjusted OR 0.87 (95% CI 0.78 to 0.97). Current smokers had increased mortality compared with never smokers (adjusted OR 1.43 (95% CI 1.28 to 1.61)), apart from a small reduction in early deaths in current smokers in the death-certificate-only group (adjusted OR 0.80 (95% CI 0.65 to 0.98)). Male sex conferred poorer outcomes; OR 1.17 (95% CI 1.10 to 1.24). Those with greatest levels of socioeconomic deprivation had increased odds of early death compared to more affluent subjects (adjusted OR 1.16 (95% CI 1.04 to 1.30) for Townsend quintile 5 compared with quintile 1). Rural versus urban location was also associated with poorer prognosis (adjusted OR 1.22 (95% CI 1.06 to 1.41)). We found no evidence of an independent association between early death and Charlson comorbidity index, or living alone versus in a shared dwelling.
GP consultations and individual CXR requests
Patients who died within 90 days of diagnosis consulted their GP more frequently than controls, with a median of five consultations (IQR 3–8) versus 4 (IQR 2–6). Those in the death-certificate-only group were least likely to have had a CXR performed in primary care (27%), compared with 52% of controls. In the multivariate analysis, the odds of early death were lower in people who had a CXR performed in primary care, regardless of the number of times they consulted their GP. Within this group of patients who had a CXR performed, the odds of early death increased as consulting frequency increased. Medium consulters who did not have a CXR performed had greatest overall odds of early death (table 3).
Practice level variables
Lung cancer rates
Background lung cancer rates varied greatly between regions, with a median UK standardised rate of 53/100 000 population (IQR 38/100 000–74/100 000). We found no association between background lung cancer rate at the practice with which the patient was registered and 90-day mortality, although there was a small reduction in 30-day mortality in the group registered at a practice which had the highest background lung cancer rate (OR 0.85 (95% CI 0.75 to 0.97) (table 3).
Practice CXR rates
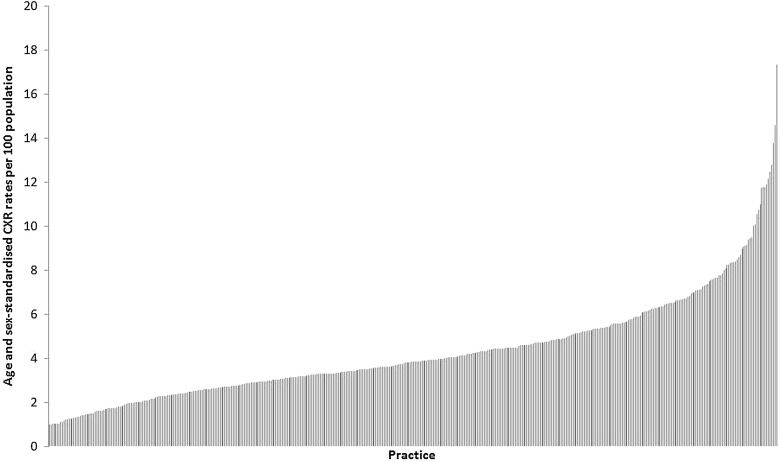
Age and sex-standardised CXR rates also differed widely between the practices (figure 3); with a median rate of 4 per 100 population per year (IQR 3/100–6/100 population). ORs for early death increased with increasing CXR quartile, even after adjustment for covariates including background lung cancer rate. People registered at a practice with a high propensity to investigate (CXR quartile 4) were 41% more likely to die early than those registered at the lowest quartile practices (death-certificate-only +90-day OR 1.41 (1.29 to 1.55)) (table 3).
Figure 3.
Age and sex-standardised chest X-ray rates per 100 population in the general practitioner practices studied.
Discussion
Our results show that 30% of patients with lung cancer died within 90 days of their diagnosis. Increasing age, male sex, socioeconomic deprivation, rural versus urban location and current smoking, were all strongly and independently associated with early death, although early death was less likely in ex-smokers compared with never smokers. Those who died early consulted their GP more frequently than those who did not, and were less likely to have had a CXR performed by primary care.
At a practice level, a higher CXR rate was not associated with a reduction in early deaths; in fact, patients seen at these practices were more likely to have died early than those seen at practices with lower CXR rates. Practices with higher background lung cancer rates do not seem to be any more likely to diagnose their patients at an early enough point in the disease process to impact on early mortality.
Strengths and weaknesses
This work is the first large study looking in detail at how events in primary care relate to early mortality in lung cancer. Previous work has confirmed that the validity of a lung cancer diagnosis in primary care databases is good and representative of National Cancer Registry data.11 17 The death-certificate-only group comprised 5.3% of the lung cancer population, which is broadly similar to death-certificate-only diagnosis figures from English Registry data,2 and lends weight to the validity of THIN as a reliable source of lung cancer cases.
Data in THIN are prospectively recorded at the time of consultation in primary care which minimises reporting and recall bias, however, the information relies on accurate coding and timely data entry in primary care. To minimise errors related to this, we only used data entered by practices after the practice met the THIN data quality and completeness standard. This comprises a regular audit of the completeness of recording of data related to a group of key conditions and exposures against national figures.18
The principal limitation of this study is the lack of information regarding stage, performance status, histology and interventions in secondary care. We used early deaths as a marker of people in whom lung cancer was diagnosed late, however, some of these cases may have had early stage or completely asymptomatic disease and died from other comorbid conditions, although the relatively short interval between lung cancer diagnosis and death means that in the majority, underlying malignancy is likely to be the main cause. There are additional limitations with regard to variation in the quality and timeliness of CXR reporting that could have influenced the apparently higher proportion of early deaths in those practices with higher CXR rates.
Other work in the literature
The International Cancer Benchmarking Project (ICBP) produced early mortality figures for four cancer sites (including lung) for six countries.1 3 The 30-day mortality from lung cancer in the UK ranged from 21% to 26% and was substantially higher than in any of the other countries included (apart from Ontario in Canada at 24%).1 They also showed, as we have, that increasing age and male sex were associated with early death, and other work has confirmed this survival advantage in women.19 Additionally, they suggested that smoking habits may have a role to play, with the UK having a higher prevalence of current smokers than some of the other counties studied. 1 2 This is supported by our finding of a higher proportion of early deaths in current smokers compared with never or ex-smokers.
Current smoking at the time of diagnosis has been shown previously to predict poor prognosis. A study published by Tammemagi and colleagues reported an adjusted HR for death of 1.26 (95% CI 1.08 to 1.47) for current smokers versus ex or never smokers.20 Additionally work looking at health behaviours suggests that ex-smokers are more likely to see themselves as vulnerable to the adverse effects of smoking compared with current smokers,21 and are, perhaps, more likely to be health-aware leading to presentation earlier in the disease process. This may explain our finding that they are less likely than never smokers to die early.
Research from Leeds, England, looked at primary care CXR referral rates over a 2-year period and showed, in a much smaller sample, that there was marked variation in practice. They found no correlation between practice CXR rates and lung cancer survival (at 1 year).22
Interpretation of the results
Evidence presented in the National Cancer Intelligence Network (NCIN) ‘Routes to Diagnosis’ work suggests that 39% of lung cancer patients present via the emergency route, and that these people have a much lower 1 year survival.23 It is therefore not surprising that the proportion of patients who died early who had a CXR requested by primary care was lower than controls, and it seems likely that these people are diagnosed via an alternative route.
The National Institute for Health and Care Excellence (NICE) referral guidelines24 give clear advice on specific symptoms which warrant a CXR and urgent referral. However, many patients have a more non-specific presentation, which is often attributed to other comorbid conditions. This may explain the high numbers of consultations for some patients in the 4 months before diagnosis.
Higher CXR rates in general at practice level do not appear to translate to reduction in early deaths from lung cancer; in fact, people were more likely to die early from lung cancer if they were registered at a practice that performed more CXRs. This may be due in part to ascertainment bias, as performing more CXRs means you are more likely to detect underlying lung cancer in a terminally ill patient. This, in turn, highlights that the burden of undiagnosed lung cancer is unknown.
We do not wish to imply that the CXR is not a useful initial investigation in detection of symptomatic lung cancer. During the Mayo Lung Project, mass screening study using CXRs, 90% of peripheral cancers and 70% of central cancers were diagnosed after retrospectively reviewing films.25 Like any investigation, however, it needs to be used at an appropriate time, in a selected patient group.
Clinical relevance and conclusions
Patients who die early are interacting with primary care in the run-up to diagnosis, suggesting that there are opportunities to identify them earlier in the disease process. A general increase in CXRs at primary care level is of questionable clinical and cost effectiveness, and it is important to establish which interventions are likely to be useful at which point in the patient pathway. It has been argued that better access to CT scanning in primary care may be useful in reducing early deaths, however, it may be that more timely and appropriate targeting of the CXR remains the key. It seems clear that we need a greater focus on older and socially deprived groups. At an individual GP level lung cancer is rare; with each GP seeing on average one new case per annum.26 For this reason, we need to promote better use of risk assessment tools, 12 27–30 and use software prompts to help GPs to identify and investigate, in a timely manner, those at risk. This has potential to increase the proportion of patients who are diagnosed at an early stage and are, therefore, suitable for treatment with curative intent.
Footnotes
Contributors: EOD was involved in study design, performed the majority of data processing and analysis and drafted the article. RBH and DRB devised the original idea and RBH, DRB, TMM and SA contributed to the design of the study and analysis of the data. BI-O, HAP and JEG contributed to the initial stages of data organisation. All authors were involved in revising the article and approved the article before submission.
Funding: This work was supported by the Roy Castle Lung Cancer Foundation, grant number RB08F1.
Disclaimer: EOD and RH affirm that the manuscript is an honest, accurate and transparent account of the study being reported; that no important aspects of the study have been omitted; and that any discrepancies from the study as planned (and, if relevant, registered) have been explained.
Competing interests: RBH is British Lung Foundation–GlaxoSmithKline (GSK) professor of respiratory epidemiology, and he has received payment for an advisory meeting from GSK on idiopathic pulmonary fibrosis. DRB has received a lecture honorarium from Pierre Fabre and payment for an advisory meeting from Roche. He is coauthor of the British Thoracic Society Guidelines on the radical management of patients with lung cancer, and was clinical lead for the National Institute of Health Research guidelines on the diagnosis and treatment of lung cancer; there are no other relationships or activities that could appear to have influenced the submitted work.
Ethics approval: Ethical approval for the study was provided by the Cegedim Strategic Data Medical Research Scientific Review Committee (Ref 13-015).
Provenance and peer review: Not commissioned; externally peer reviewed.
Data sharing statement: Dr O'Dowd had full access to all the data and takes responsibility for the integrity and the accuracy of the data and analysis. There is no additional unpublished data from this study.
References
- 1.Coleman MP, Forman D, Bryant H, et al. Cancer survival in Australia, Canada, Denmark, Norway, Sweden, and the UK, 1995–2007 (the International Cancer Benchmarking Partnership): an analysis of population-based cancer registry data. Lancet 2011;377:127–38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Holmberg L, Sandin F, Bray F, et al. National comparisons of lung cancer survival in England, Norway and Sweden 2001–2004: differences occur early in follow-up. Thorax 2010;65:436–41. [DOI] [PubMed] [Google Scholar]
- 3.Walters S, Maringe C, Coleman MP, et al. Lung cancer survival and stage at diagnosis in Australia, Canada, Denmark, Norway, Sweden and the UK: a population-based study, 2004–2007. Thorax 2013;68:551–64. [DOI] [PubMed] [Google Scholar]
- 4.Lo Re V, III, Haynes K, Forde KA, et al. Validity of The Health Improvement Network (THIN) for epidemiologic studies of hepatitis C virus infection. Pharmacoepidemiol Drug Saf 2009;18:807–14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Hall GC. Validation of death and suicide recording on the THIN UK primary care database. Pharmacoepidemiol Drug Saf 2009;18:120–31. [DOI] [PubMed] [Google Scholar]
- 6.Szatkowski L, Lewis S, McNeill A, et al. Can data from primary care medical records be used to monitor national smoking prevalence? J Epidemiol Community Health 2012;66:791–5. [DOI] [PubMed] [Google Scholar]
- 7.Ogdie A, Love T, Haynes K, et al. Validation of the health improvement network for the study of psoriatic arthritis. Pharmacoepidemiol Drug Saf 2013;22:62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Lewis JD, Schinnar R, Bilker WB, et al. Validation studies of the health improvement network (THIN) database for pharmacoepidemiology research. Pharmacoepidemiol Drug Saf 2007;16:393–401. [DOI] [PubMed] [Google Scholar]
- 9.Denburg MR, Haynes K, Shults J, et al. Validation of The Health Improvement Network (THIN) database for epidemiologic studies of chronic kidney disease. Pharmacoepidemiol Drug Saf 2011;20:1138–49. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Blak BT, Thompson M, Dattani H, et al. Generalisability of The Health Improvement Network (THIN) database: demographics, chronic disease prevalence and mortality rates. Inform Prim Care 2011;19:251–5. [DOI] [PubMed] [Google Scholar]
- 11.Iyen-Omofoman B, Hubbard RB, Smith CJP, et al. The distribution of lung cancer across sectors of society in the United Kingdom: a study using national primary care data. BMC Public Health 2011;11:857. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Iyen-Omofoman B, Tata LJ, Baldwin DR, et al. Using socio-demographic and early clinical features in general practice to identify people with lung cancer earlier. Thorax 2013;68:451–9. [DOI] [PubMed] [Google Scholar]
- 13.Charlson ME, Pompei P, Ales KL, et al. A new method of classifying prognostic co-morbidity in longitudinal-studies—development and validation. J Clin Epidemiol 1987;40:373–83. [DOI] [PubMed] [Google Scholar]
- 14.Powell HA, Tata LJ, Baldwin DR, et al. Early mortality after surgical resection for lung cancer: an analysis of the English National Lung cancer audit. Thorax 2013;68:826–34. [DOI] [PubMed] [Google Scholar]
- 15.Rich AL, Tata LJ, Free CM, et al. Inequalities in outcomes for non-small cell lung cancer: the influence of clinical characteristics and features of the local lung cancer service. Thorax 2011;66:1078–84. [DOI] [PubMed] [Google Scholar]
- 16.Huber PJ. A robust version of the probability ratio test. Ann Math Stat 1965;36:1753–8. [Google Scholar]
- 17.Dregan A, Moller H, Murray-Thomas T, et al. Validity of cancer diagnosis in a primary care database compared with linked cancer registrations in England. Population-based cohort study. Cancer Epidemiol 2012;36:425–9. [DOI] [PubMed] [Google Scholar]
- 18.Bourke A, Dattani H, Robinson M. Feasibility study and methodology to create a quality-evaluated database of primary care data. Inform Prim Care 2004;12:171–7. [DOI] [PubMed] [Google Scholar]
- 19.Thomas L, Doyle LA, Edelman MJ. Lung cancer in women—Emerging differences in epidemiology, biology, and therapy. Chest 2005;128:370–81. [DOI] [PubMed] [Google Scholar]
- 20.Tammemagi CM, Neslund-Dudas C, Simoff M, et al. Smoking and lung cancer survival—The role of comorbidity and treatment. Chest 2004;125:27–37. [DOI] [PubMed] [Google Scholar]
- 21.Weinberger M, Greene JY, Mamlin JJ, et al. Health beliefs and smoking-behavior. Am J Public Health 1981;71:1253–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Cheyne L, Milton R, Fear J, et al. Variability in gp referral rates for chest x-ray does not appear to affect stage or performance status of patients diagnosed with lung cancer. Thorax 2012;67:A90-A. [Google Scholar]
- 23.Elliss-Brookes L, McPhail S, Ives A, et al. Routes to diagnosis for cancer—determining the patient journey using multiple routine data sets. Br J Cancer 2012;107:1220–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Baldwin DR, White B, Schmidt-Hansen M, et al. Diagnosis and treatment of lung cancer: summary of updated NICE guidance. BMJ 2011;342:d2110. [DOI] [PubMed] [Google Scholar]
- 25.Muhm JR, Miller WE, Fontana RS, et al. Lung-cancer detected during a screening-program using 4-month chest radiographs. Radiology 1983;148:609–15. [DOI] [PubMed] [Google Scholar]
- 26.Hamilton W, Sharp D. Diagnosis of lung cancer in primary care: a structured review. Fam Pract 2004;21:605–11. [DOI] [PubMed] [Google Scholar]
- 27.Hamilton W, Peters TJ, Round A, et al. What are the clinical features of lung cancer before the diagnosis is made?—A population based case-control study. Thorax 2005;60:1059–65. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Bach PB, Kattan MW, Thornquist MD, et al. Variations in lung cancer risk among smokers. J Natl Cancer Inst 2003;95:470–8. [DOI] [PubMed] [Google Scholar]
- 29.Spitz MR, Etzel CJ, Dong Q, et al. An expanded risk prediction model for lung cancer. Cancer Prev Res 2008;1:250–4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Cassidy A, Myles JP, van Tongeren M, et al. The LLP risk model: an individual risk prediction model for lung cancer. Br J Cancer 2008;98:270–6. [DOI] [PMC free article] [PubMed] [Google Scholar]