Abstract
Planar cell polarity (PCP) signaling is an evolutionarily conserved mechanism that coordinates polarized cell behavior to regulate tissue morphogenesis during vertebrate gastrulation, neurulation and organogenesis. In Xenopus and zebrafish, PCP signaling is activated by non-canonical Wnts such as Wnt11, and detailed understanding of Wnt11 expression has provided important clues on when, where and how PCP may be activated to regulate tissue morphogenesis. To explore the role of Wnt11 in mammalian development, we established a Wnt11 expression and lineage map with high spatial and temporal resolution by creating and analyzing a tamoxifen-inducible Wnt11-CreER BAC (bacterial artificial chromosome) transgenic mouse line. Our short- and long-term lineage tracing experiments indicated that Wnt11-CreER could faithfully recapitulate endogenous Wnt11 expression, and revealed for the first time that cells transiently expressing Wnt11 at early gastrulation were fated to become specifically the progenitors of the entire endoderm. During mid-gastrulation, Wnt11-CreER expressing cells also contribute extensively to the endothelium in both embryonic and extraembryonic compartments, and the endocardium in all chambers of the developing heart. In contrast, Wnt11-CreER expression in the myocardium starts from late-gastrulation, and occurs in three transient, sequential waves: first in the precursors of the left ventricular (LV) myocardium from E7.0 to 8.0; subsequently in the right ventricular (RV) myocardium from E8.0 to 9.0; and finally in the superior wall of the outflow tract (OFT) myocardium from E8.5 to 10.5. These results provide formal genetic proof that the majority of the endocardium and myocardium diverge by mid-gastrulation in the mouse, and suggest a tight spatial and temporal control of Wnt11 expression in the myocardial lineage to coordinate with myocardial differentiation in the first and second heart field progenitors to form the LV, RV and OFT. The insights gained from this study will also guide future investigations to decipher the role of non-canonical Wnt/ PCP signaling in endoderm development, vasculogenesis and heart formation.
Keywords: Wnt11, Planar cell polarity, Fate mapping, Gastrulation, Endoderm specification, Endothelial/ endocardial specification, Heart development
Introduction
In mice and humans there are 19 independent Wnt genes, encoding secreted glycoproteins that have diverse and critical roles during embryonic development, in adult tissue homoeostasis and in human diseases (Willert and Nusse, 2012). Wnt ligands can be broadly divided into two classes: canonical Wnts such as Wnt1 and Wnt3a, and non-canonical Wnts such as Wnt5a and Wnt11. Canonical Wnts bind to Frizzled (Fz) receptors and the Lrp5/6 family of co-receptors to activate cytoplasmic protein Dishevelled (Dsh/Dvl), which in turn stabilizes -catenin to activate gene transcription. Non-canonical Wnts, on the other hand, share certain components with the canonical Wnt pathway such as Fz and Dsh/Dvl, but signal through multiple -catenin independent branches that include the Wnt/Ca2+ and the planar cell polarity (PCP) pathways (Angers and Moon, 2009; MacDonald et al., 2009; van Amerongen and Nusse, 2009; Wallingford et al., 2000).
Of these non-canonical Wnt signaling branches, the PCP pathway has been studied most extensively in various vertebrate and invertebrate model organisms. Initially identified as one of the signaling mechanisms that coordinate cellular polarity in the plane of the epithelium, PCP signaling also regulates polarized cell behavior such as mediolateral (M-L) cell intercalation and directional migration during convergent extension (CE) tissue morphogenesis (Keller, 2002; Zallen, 2007). In addition to Fz and Dsh/Dvl, the PCP pathway requires a set of distinct “core” proteins such as the tetraspan membrane protein Van Gogh (Vang/Vangl) and the atypical cadherin Flamingo (Fmi). What functions downstream of these core proteins as PCP effectors remains elusive and is likely to be context- and tissue- dependent, and may include JNK, small GTPase Rho/Rac/Cdc42 and the formin protein Daam1 (Goodrich and Strutt, 2011; Habas et al., 2001; Tree et al., 2002; Wallingford, 2012).
In Xenopus and zebrafish, Wnt5a and Wnt11 have been identified as two primary ligands capable of activating non-canonical Wnt/ PCP signaling to regulate tissue morphogenesis during gastrulation (Heisenberg et al., 2000; Kilian et al., 2003; Tada and Smith, 2000; Walentek et al., 2013; Wallingford et al., 2001), although in other context they can also activate canonical Wnt signaling (Cha et al., 2008; Cha et al., 2009; Tao et al., 2005). During gastrulation, Wnt11 acts in both cell-autonomous and cell-non-autonomous fashion to regulate polarized cell intercalation and directional migration of mesodermal and endodermal cells (Heisenberg et al., 2000; Ulrich et al., 2003; Witzel et al., 2006). Consequently, perturbing Wnt11 expression results in failure of axial elongation and midline convergence of foregut endoderm in frog and zebrafish embryos (Heisenberg et al., 2000; Li et al., 2008; Matsui et al., 2005; Tada and Smith, 2000; Walentek et al., 2013).
In the mouse, PCP signaling has so far been implicated in a number of processes such as neurulation and cardiovascular and limb development (van Amerongen, 2012; Wang et al., 2012). PCP-mediated tissue morphogenesis is likely to have even broader impact on mammalian development and human diseases. Given the essential roles of Wnt5a/Wnt11 in initiating PCP signaling and the fact that they act in paracrine or autocrine fashion, determining their spatial and temporal expression pattern will shed light on where and when PCP signaling is potentially activated, and provide hints on what additional processes and tissues may require PCP function. Indeed, in situ hybridization studies of Wnt5a expression have led to novel models as to how PCP signaling could be operative in mammals to regulate heart and limb development (Gao et al., 2011; Gros et al., 2010; Sinha et al., 2012; Yamaguchi et al., 1999a).
In comparison, our understanding of Wnt11 in the mouse is more limited. In situ studies indicate that Wnt11 is expressed first in a scattered pattern around the primitive streak at early gastrulation, and later on in the developing heart, posterior trunk and urogenital system (Kispert et al., 1996). Wnt11-/- mouse embryos display no major gastrulation defects, but die in-utero or shortly after birth with multiple cardiac defects (Majumdar et al., 2003; Nagy et al., 2010; Zhou et al., 2007). Due to limited spatial and temporal resolution, the existing RNA in situ data do not provide sufficient information as to which cardiac lineage(s) express Wnt11 and the duration of Wnt11 expression in that lineage, and therefore the spatio-temporal requirement for Wnt11 in heart development remain to be elucidated. Moreover, in situ based expression studies often cannot inform us of the potential fate and lineage of the cells expressing the gene of interest, for instance the scattered Wnt11-expressing cells during early gastrulation in the mouse (Kispert et al., 1996).
To overcome the limitations of in situ hybridization, and to establish a Wnt11 expression map with high spatial and temporal resolution and lineage information, we generated a tamoxifen inducible Wnt11-CreER transgene using BAC (Bacterial Artificial Chromosome) recombineering technology. Because of their large size (150-300 kb), BAC transgenes have been shown to recapitulate endogenous gene expression patterns (Lee et al., 2001). CreER-T2 encodes a fusion protein between a Cre and a mutated estrogen receptor (ER) (Feil et al., 1997; Leone et al., 2003). CreER is normally sequestered in the cytoplasm by the ER domain. Exposure to tamoxifen leads to a temporary relief of this sequestration, allowing CreER to enter the nucleus and induce recombination. By crossing Wnt11-CreER with Rosa26 Cre reporters (R26R) (Madisen et al., 2010; Soriano, 1999) and administering tamoxifen during gestation, we can transiently induce Cre activation to permanently label Wnt11 expressing cells and their descendents. By analyses of embryos shortly after tamoxifen-induction, we can establish a high-resolution expression map of Wnt11-CreER and compare it with the existing in situ data. Alternatively, we can trace the fate of Wnt11-CreER expressing cells by collecting and analyzing embryos after a more extended period.
In the current study, we focused on using our Wnt11-CreER BAC transgene to perform detailed expression and lineage analyses during gastrulation and heart development. Our results uncovered strikingly specific and dynamic expression of Wnt11 in progenitors of the endodermal and endothelial lineages during early and mid- gastrulation. In the heart, we demonstrated that not only did the Wnt11-CreER expressing cells contribute to three major cardiac lineages (the endocardium, myocardium and epicardium), but they did so in a highly spatio-temporally controlled fashion. Collectively, our results provide novel and significant insights and open up multiple avenues to explore the involvement of Wnt11/PCP signaling during early endoderm development, vasculogenesis and heart formation.
Materials and Methods
Cloning and BAC recombineering to generate Wnt11-CreER BAC transgenic mice
To create an efficient system to target the tamoxifen-inducible CreER T2 into BACs, we cloned the CreER T2 fragment into the pIGCN21 vector, in front of an Frt-kanamycin (kan)-Frt cassette that contains an EM7 promoter to drive kan transcription in bacteria.
BAC clone RP23-122D14 was acquired from BACPAC Resource Center at Children’s Hospital Oakland Research Institute. This 196.9kb BAC contains the 19.6kb mouse Wnt11 locus as well as 102.7kb genomic sequence 5’ and 74.5kb genomic sequence 3’ of Wnt11 (Fig.1A). To target CreER T2 into the Wnt11 region, CreER T2- Frt-kan-Frt cassette was amplified using primers Wnt11 CreER F (GCGGTGGCCTGCAGGCGGCGGAGTTCGGTGCGGCTCCTGCAGGGTGCGACCCCCGGGAGCGCCG GGCGCGCGCGACGATGTCCAATTTACTGACCGTA) and Wnt11 CreER R (TCGCAGATTTTGGTGGGCTCACCCAACCTCTCCAGCTTCTCGCCCAATGGCCCATTGGAGTGAAAA CGGAGTCCTACTCTATTCCAGAAGTAGTGAGGA) to add 78 bp homology arms on each end. RP23-122D14 BAC DNA was purified and electroporated into EL250 cells, and standard BAC recombineering procedures (Lee et al., 2001) were carried out using the Wnt11 CreER T2-Frt-kan-Frt targeting cassette. LB agar plates containing 12.5 ug/ ml chloramphenicol and 25 ug/ ml kanamycin were used to select for successfully targeted clones. PCR and sequencing reactions were performed to confirm that CreER T2 was properly targeted into Wnt11 locus. Since EL250 cells also contain arabinose-inducible Flpe (Lee et al., 2001), the Frt flanked kan could also be deleted. The resulting Wnt11-CreER T2 BAC DNA was purified and used for pro-nuclear injection to create Wnt11-CreER transgenic founders.
Figure 1. Generation and characterization of Wnt11-CreER BAC transgene.
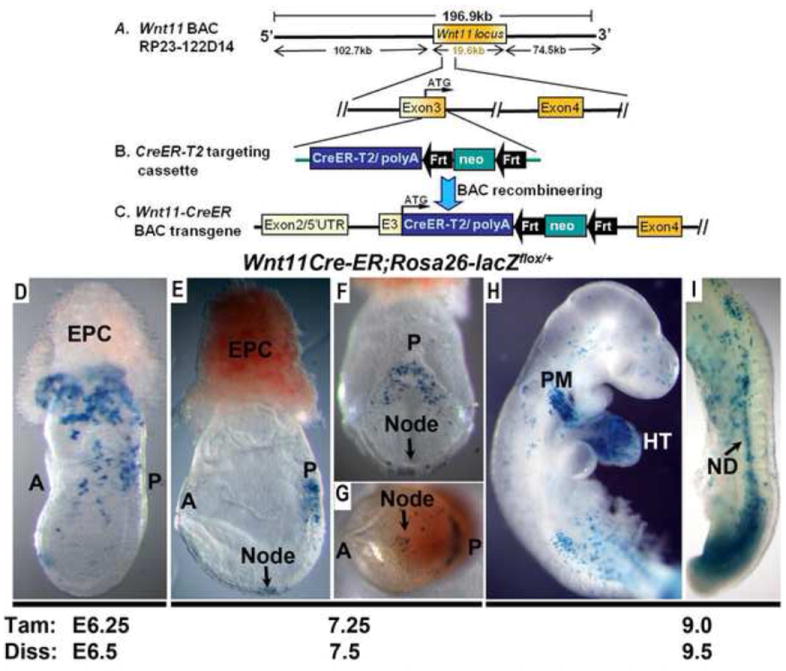
(A) Schematic diagram of BAC clone RP23-122D14 that contains the 19.6 kb mouse Wnt11 locus, and 102.7 kb and 74.5 kb genomic sequence flanking the 5’ and 3’ of Wnt11, respectively. (B, C) A CreER-T2 targeting cassette was used to partially replace the first coding exon (exon3) of Wnt11 by BAC recombineering to create the tamoxifen-inducible Wnt11-CreER BAC transgene. (D-H) Wnt11-CreER BAC transgenic mice were crossed with Cre reporter R26R-LacZ and the expression pattern of Wnt11-CreER was determined by X-gal staining to examine Cre-induced LacZ expression 6-12 hours after tamoxifen injection. (D) When tamoxifen was injected at E6.25 and X-gal staining was performed 6 hours later at E6.5, labeled cells were observed in the proximal extra-embryonic region near the ectoplacental cone, and in the posterior region of the embryo adjacent to the embryonic/extraembryonic border and the primitive streak. (E, F, G) Tamoxifen injection at E7.25 and X-gal staining at E7.5 resulted in labeled cells in the node (arrows in (E), (F) and (G)) and the posterior end of the embryo, surrounding the primitive streak and at the base of the allantois. (H, I) Tamoxifen administration at E9.0 and X-gal staining at E9.5 resulted in labeled cells in the posterior trunk/ tailbud region and the nephric duct (ND, arrow in H) and in the outflow tract and the second pharyngeal arch anteriorly (G). A (anterior); EPC (ectoplacental cone); HT (heart tube); ND (nephric duct (ND); P (posterior); PM (pharyngeal mesoderm).
Syntenic analysis of the vertebrate Wnt11 genes
Genetic elements surrounding the Wnt11 locus in mouse and its homologues in other vertebrate, namely human, chicken, zebrafish and Xenopus were identified using the UCSC genome browser, the zfin genome browser and the xenbase genome browser for each of the organisms, respectively. Conservation analysis across the Wnt11 locus spanning the Wnt11 containing BAC clone RP23-122D14 region was performed using the NCBI comparative genomic analysis tool on the dcode/ECR browser.
Mouse strains and genotyping
Wnt11-CreER BAC transgenic mice were genotyped by PCR using primers CreA1 (CCG GGC TGC CAC GAC CAA) and CreA2 (GGC GCG GCA ACA CCA TTT TT). Rosa26-tdTomato (B6;129S6-Gt(ROSA)26Sortm9(CAG-tdTomato)Hze/J) (R26R-tdTomato) and Rosa26-lacz (B6.129S4-Gt(ROSA)26Sortm1Sor/J) (R26R-lacZ) Cre reporter mice were obtained from the Jackson Laboratory and genotyped as described (Madisen et al., 2010; Soriano, 1999). All strains were maintained in a C57B6/ SJL/ FνB mixed background. Animal care and use was in accordance with NIH guidelines and was approved by the Animal Care and Use Committee of the University of Alabama at Birmingham.
Tamoxifen administration, embryo collection, imaging and X-gal staining
Wnt11-CreER mice were crossed with Rosa26-Cre reporter (R26R-lacz and R26R-tdTomato) mice to obtain Wnt11-CreER; R26R embryos. Pregnant dams were singly gavaged with tamoxifen (Sigma, T-5648, dissolved to 10mg/ml in corn oil) at 2-6mg/ 40g body weight. Embryos were retrieved 6 hours to 6 days post gavage and the yolk sac was retained for PCR genotyping. Embryos were temporarily fixed in 4% paraformaldehyde (PFA) at 4°C for 20 to 60 minutes depending on embryo stage and subsequently stored in PBS. X-gal staining was performed using as a standard protocol as previously described(Nagy et al., 2007). Bright-field and epi-fluorescent whole mount embryo images were captured using a Leica MZ16FA fluorescence stereomicroscope equipped with a multi-fluorescent filter set and a DFC490 CCD camera.
Immuno-staining and in situ hybridization
Fixed embryos/tissues were processed through sucrose gradients and embedded in OCT (Tissue-Tek/Sakura, Torrance, CA) for cryo-sectioning. Briefly, OCT embedded samples were cryo-sectioned at 10um-25um, air-dried at room temperature, fixed with 4%PFA, permeabilized with PbTX (0.1% Tween in PBS), incubated in blocking solution (PbTX+1%BSA) followed by incubation with primary antibodies overnight at 4°C. Sections were washed with PbTx the next day, incubated with appropriate secondary antibodies for 1hour at room temperature followed by washing with PbTx and mounting in Vectashield mounting medium containing DAPI (Vector Labs, Burlingame, CA). Primary antibodies used were mouse anti-MF20 (sarcomeric myosin heavy chain) (1:15, DSHB, Iowa) and anti-CD31(PECAM) (1:200, clone MEC 13.3, BD Biosciences, San Jose, CA). Alexa Fluor 647-conjugated donkey anti-mouse IgG (1:500, Invitrogen, Carlsbad, CA), Dylight-488 conjugated donkey anti-mouse IgG, FITC conjugated Donkey anti-rat IgG (1:400 and 1:200, Jackson ImmunoResearch, West Grove, PA) and HRP-conjugated goat anti-rat IgG (1:200, SCBT, Dallas, TX) were used as secondary antibodies. A tyramide signal amplification kit (PerkinElmer, Covina, CA, #NEL741E001KT) was used for anti-CD31 detection. All fluorescent confocal images were acquired with an Olympus FV1000 Laser Confocal Scanning microscope and were subsequently analyzed using the FV10-ASW software. Images were compiled and linearly adjusted for brightness, contrast and color balance using Adobe Photoshop CS5. Whole-mount in situ hybridization was carried out with a standard protocol (Wilkinson and Nieto, 1993) using a previously described Wnt11 anti-sense probe (Zhou et al., 2007). Fluorescent in situ hybridization on sections was carried out as previously described (Wang et al., 2002) with tyramide signal amplification kit.
Results
Generation of Wnt11-CreER BAC transgene
We identified a 197kb BAC clone (RP23-122D14) that contained the 19.6kb mouse Wnt11 locus as well as 102.7kb and 74.5kb genomic sequence 5’ and 3’ of Wnt11, respectively (Fig. 1A; Suppl. Fig. S1). To establish an efficient system to target the tamoxifen-inducible CreER T2 into the BAC, we first created a construct in which CreER T2 was linked to an Frt flanked kanamycin (kan) cassette (Fig. 1B). This cassette allows direct selection of kan resistant clones that have also co-incorporated CreER T2 through BAC recombineering, and can be deleted by induction of flpe activity in EL250 cells that harbor an arabinose-inducible flpe (Lee et al., 2001).
Using PCR, we added 78bp homology arms at each end of the CreER T2-Frt-kan-Frt cassette to target it into Wnt11 locus in the BAC. The inserted CreER T2 replaced part of the first coding exon of Wnt11, exon3, from the ATG start codon to the splicing donor sequence immediately behind exon3 (Fig. 1B). The resulting Wnt11-CreER BAC, therefore, would express CreER T2 under the control of Wnt11 promoter and regulatory sequences, but was expected not to produce Wnt11 protein due to the deletion of the start codon in exon3 (Fig.1C). Moreover, even if a truncated Wnt11 transcript could be made and a cryptic start codon could be used to initiate translation, we predict that no functional Wnt11 ligand can be made from Wnt11-CreER BAC transgene owing to the deletion of the first 83 codons in exon3, which encode the signal peptide required for Wnt11 secretion.
Establishment and characterization of a Wnt11-CreER transgenic line
We established one transgenic line from pronuclear injection of the Wnt11-CreER BAC DNA. To determine how faithfully the transgene could recapitulate endogenous Wnt11 expression, we first analyzed the spatial pattern of Wnt11-CreER expression at several embryonic stages. This was achieved by crossing Wnt11-CreER mice with Rosa26 Cre reporter (R26R-LacZ) mice to visualize Cre-activated reporter gene (LacZ) expression shortly (6 to 12 hours) after tamoxifen administration, which allowed transient nuclear translocation of Cre to induce homologous recombination and activate Cre reporter expression.
When we performed tamoxifen administration by oral gavage at E6.25, and dissected and stained embryos with X-gal 6 hours later (~E6.5), we observed LacZ-positive cells in the proximal extraembryonic region near the ectoplacental cone (EPC), and in the posterior region of the embryo proper, adjacent to the primitive streak at this stage (Fig.1D). This pattern closely resembled the reported endogenous Wnt11 gene expression at E6.5 revealed by in situ hybridization (Kispert et al., 1996). Tamoxifen induction at E7.25 and X-gal staining 6 hours later at E7.5 resulted in labeled cells in the node and the posterior end of the embryo, around the primitive streak and at the base of the allantois (Fig.1E-F). This pattern again mimics E7.5 Wnt11 in situ results (Kispert et al., 1996) (Suppl. Fig. S2). Lastly, when we injected tamoxifen at E9.0 and stained the embryos 12 hours later at E9.5, we observed labeled cells in the posterior trunk/ tailbud region and in the nephric duct (Fig.1I). Anteriorly, labeled cells are found primarily in the OFT and the second pharyngeal arch, where second heart field (SHF) progenitors are located (Fig.1H). The pattern of the labeled cells also closely matched Wnt11 in situ hybridization studies performed at E9.5 (Kispert et al., 1996; Zhou et al., 2007).
Collectively, our analyses of CreER induced LacZ expression shortly after tamoxifen induction indicate that the Wnt11-CreER BAC transgene can largely recapitulate endogenous Wnt11 expression during early mouse development.
Cells expressing Wnt11-CreER during gastrulation contribute to specific endodermal and mesodermal tissues in the embryonic and extra-embryonic compartments
Given that Wnt11-CreER expressing cells appeared to occupy distinct domains during gastrulation (Fig.1D-F), we sought first to determine which lineages they would contribute to by performing long term lineage tracing (2-6 days) after a single tamoxifen adminstration, either before gastrulation (E5.5 and earlier) or at the early (E6.0), mid (E6.5) or late (E7.5) streak stages. For the long term tracing experiments, we utilized the fluorescent Rosa26 Cre reporter R26R-tdTomato. R26R-tdTomato was constructed similarly as R26R-LacZ except that Cre-mediated recombination led to the permanent expression of tdTomato, a fast maturing, remarkably bright and photo-stable red fluorescent protein (Madisen et al., 2010; Shaner et al., 2007). These properties of tdTomato allowed direct and instantaneous visualization of labeled cells upon embryo dissection, and eliminated potential problems with LacZ staining in older embryos. Furthermore, co-immunofluorescent staining could be performed to determine the identity of the tdTomato positive cells.
Activation of Wnt11-CreER prior to gastrulation (E5.5 or earlier)
When pregnant dams were induced with tamoxifen at E5.5 or earlier and embryos were collected at E8.5 or later, only a few labeled cells were detected that were scattered around the embryos with no consistent pattern (data not shown). This result indicated that Wnt11-CreER was not expressed at significant levels prior to the onset of gastrulation in mice.
Activation of Wnt11-CreER at the early streak stage (E6.0)
Interestingly, when we administered a single dose of tamoxifen at E6.0, we found that Wnt11-CreER expressing cells contributed to specific lineages in both the extra-embryonic and embryonic regions. Most strikingly, when these E6.0 tamoxifen-induced embryos were harvested at E8.25 (Fig.2A & B – whole mount), the tdTomato labeled cells occupied almost the entire embryonic endoderm along the anterior-posterior axis (Fig.2F, G and J).
Figure 2. Cells expressing Wnt11-CreER at the onset of gastrulation contribute to the endodermal and endothelial lineages.
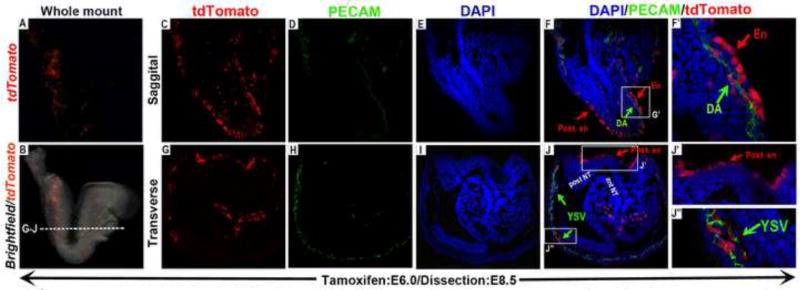
Wnt11-CreER mediated recombination and activation of tdTomato expression was induced by a single dose of tamoxifen administration at E6.0, and embryos were harvested and analyzed at E8.5. (A) Epi-fluorescence of tdTomato alone, and (B) merged with brightfield. Saggital (C-F’) and transverse (G-J”) sections of the embryos co-stained with DAPI (nucleus) revealed that tdTomato expressing cells (red) were present along the endoderm from anterior (F and magnified view in F’, red arrows) to the posterior (F, J and magnified view in J’). A small number of tdTomato expressing cells also co-expressed the endothelial marker PECAM (D, H, J and J”) and were present in the dorsal aorta in the embryo proper (green arrow in F; magnified in F’) and as a few clusters in the yolk sac vasculature (green arrow in J; magnified in J”). The dotted line in (B) indicates the plane of sectioning in G to J”. ant NT (anterior neural tube); DA (dorsal aorta); En (endoderm endoderm); Post. en (posterior endoderm); post NT (posterior neural tube); YSV (yolk sac vasculature).
Secondly, we found that in E6.0 tamoxifen-induced embryos harvested at E8.25, a small number of tdTomato labeled cells also co-expressed the endothelial marker PECAM (Platelet endothelial cell adhesion molecule) (green arrows in 2F, F’, J and J”). In the extra-embryonic region, these tdTomato/PECAM double positive cells were present as a few small clusters on the yolk sac (green arrows in Fig.2J &J”); whereas in the embryo proper, these cells were located along the dorsal aorta (green arrows in Fig.2F &F’).
Overall, these results indicated that at the onset of gastrulation, Wnt11-CreER was expressed in the progenitors of the entire embryonic endoderm and a small portion of the endothelial mesoderm that would give rise to the vasculature in both the embryonic and extra-embryonic compartments.
Activation of Wnt11-CreER at the mid streak stage (E6.5)
Embryos induced at E6.5 and harvested at E8.5 resulted in tdTomato labeling of a majority of endodermal cells along the entire A-P axis (Fig.3), similar to E6.0-tamoxifen induced embryos (Fig.2). Since the endoderm is known to give rise specifically to the epithelial lining of the respiratory and the digestive systems (Zorn and Wells, 2009), we also performed longer term tracing to determine the developmental potential and specificity of these tdTomato-labeled cells. When we retrieved E6.5-tamoxifen induced embryos at E12.5, we observed that tdTomato expressing cells colonized most of the endoderm-derived internal organs including the trachea, esophagus, lung, stomach and intestines (Fig.4E, E’, G, G’ and data not shown). Sectioning of these organs further confirmed that tdTomato labeled cells were present almost exclusively in the epithelial linings of these organs (Fig.4 F-F”, H-H”).
Figure 3. Cells expressing Wnt11-CreER at the mid streak stage contribute to the endodermal and mesodermal lineages.
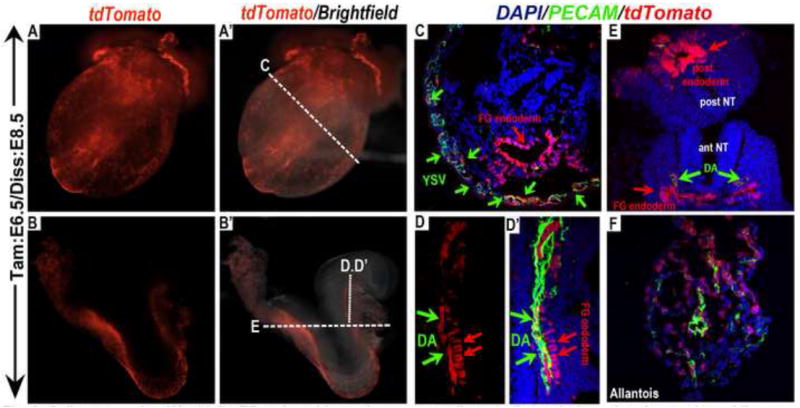
Wnt11-CreER mediated recombination and activation of tdTomato expression was induced by a single dose of tamoxifen administration at E6.5, and embryos were harvested and analyzed at E8.5. (A, B) Epi-fluorescence of tdTomato alone, and (A’, B’) merged with brightfield. (C-F) Embryo sections co-stained with PECAM (green) and DAPI (blue) revealed that tdTomato expressing cells occupied many more patches of the vasculature in the yolk sac (C) and the allantois (F) when compared to E6.0 tamoxifen induced embryos (Fig.2F). Increased contribution of tdTomato expressing cells to the endothelial lineage was also evident in the dorsal aorta (green arrows in (D) & (D’)). TdTomato expressing cells also occupied the entire endoderm, from the anterior to the posterior endoderm (red arrows in (C) &(E)). Dotted lines in (A’) & (B’) indicate the plane of sectioning in (C-E). ant NT (anterior neural tube); DA (dorsal aorta); FG (foregut); post NT (posterior neural tube); YSV (yolk sac vasculature).
Figure 4. Contribution of E6.5 Wnt11-CreER expressing cells to the endothelium and endodermally derived internal organs in E12.5 embryos.
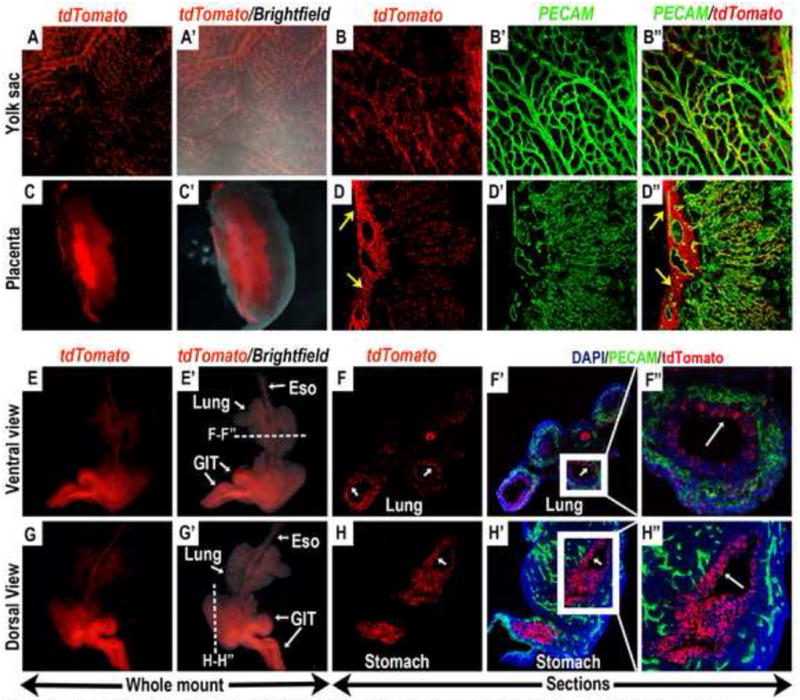
Wnt11-CreER mediated recombination and activation of tdTomato expression was induced by a single dose of tamoxifen administration at E6.5, and embryos were harvested and analyzed at E12.5. (A-D”) Whole mount epi-fluorescent analysis showed that tdTomato expressing cells were lining the yolk-sac vasculature (A and A’), and co-expressed the endothelial marker PECAM (B, B’ and B”). Bisection of the placenta revealed that widespread tdTomato expression could also be observed in the placental labyrinthe (C and C’). In the placenta, tdTomato expression was present mainly in the endothelial cells in the fetal vasculature (D’ and D”) as well as the supporting chorio-allanatoic mesenchyme (yellow arrows in D and D”) surrounding the larger placental vessels. (E-H”) In the embryo proper, tdTomato expressing cells contributed to the endodermally derived internal organs, namely the lungs and the gastro-intestinal tract. (E, E’, G, G’) whole mount epifluorescent analysis. Sectioning along the indicated planes in E’ and G’ (dashed white lines) revealed that the tdTomato expressing cells were present mostly in the epithelial lining of the lungs (white arrows in F and F’; magnified view in F”) and stomach (white arrow in H and H’; magnified view in H”). Eso: esophagus, GIT: gastro-intestinal tract.
Compared to the E6.0-tamoxifen induced embryos, the E6.5-tamoxifen induced Wnt11-CreER; R26R-tdTomato embryos harvested at E8.5 had greater number of tdTomato labeled cells contributing to the endothelial lineage. In the embryo proper, more tdTomato-expressing cells could be observed in the dorsal aortae (compare Fig.3D’ to Fig.2F’). This trend was even more evident in the extra-embryonic region, where tdTomato labeled cells occupied many more patches of the vasculature in the yolk sac (Fig.3A-C) and the allantois (Fig.3F). When these E6.5-tamoxifen induced embryos were harvested at E12.5, we found many tdTomato positive cells lining the blood vessels in the yolk sac (Fig.4A-B”) and co-expressing the endothelial marker PECAM. A large numbers of tdTomato-expressing cells could also be found in the allantois-derived tissues in the placenta, including the fetal vasculature in the labyrinth (Fig.4C-D”) and the supporting chorio-allanatoic mesenchyme (yellow arrows in Fig.4D, D”) at the base of the placenta.
Endocardium is a specialized layer of endothelial cells lining the myocardium of the heart. In E6.5 tamoxifen-induced Wnt11-CreER; R26R-tdTomato embryos harvested at E8.5, we found that tdTomato-expressing cells also contributed extensively to the endocardium (Fig.7A, B & B’). We will discuss this finding in more detail in the later sections where we examined the dynamic contribution of Wnt11-CreER cells to the heart.
Figure 7. Wnt11-CreER expression is initiated in the endocardial progenitors during gastrulation and maintained in the differentiated endocardium.
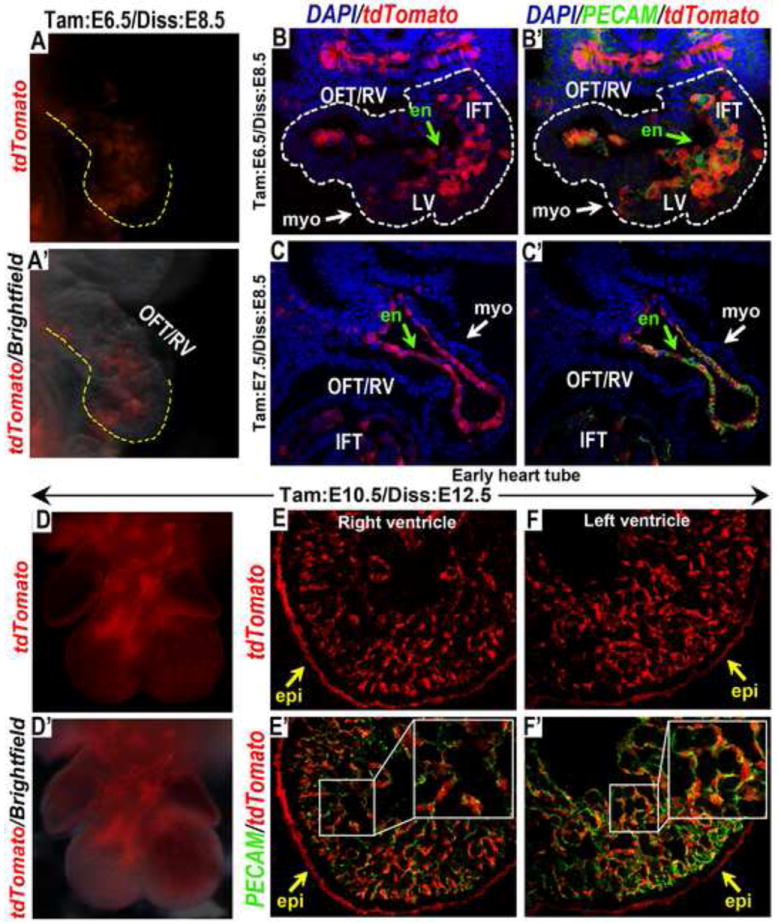
(A-B’) Activation of tdTomato expression by Wnt11-CreER was induced with tamoxifen at E6.5, and embryos were analyzed at E8.5. (A) Whole mount epi-fluorescent image of tdTomato alone, and (A’), merged with brightfield. (B, B’) Transverse sectioning and co-immunostaining with PECAM revealed that tdTomato expressing cells occupied almost the entire endocardium lining all chamber of the forming heart tube (outlined by dotted white line), including the inflow tract (IFT), the left ventricle and the right ventricle/OFT. By contrast, only few tdTomato expressing cells could be found in the LV myocardium (white arrows in B and B’). (C, C’) Tamoxifen induction at E7.5 revealed that tdTomato expressing cells contributed exclusive to the endocardium (green arrows in C & C’), but not the myocardium, in the OFT and RV. (D-F’) When tamoxifen induction was performed at E10.5 and the resulting embryos analyzed at E12.5, tdTomato expressing cells could still contribute to the entire endocardium of both the right and the left ventricles, indicating that Wnt11-CreER expression was maintained in the differentiated endocardium. In these embryos, tdTomato expressing cells also contributed to the epicardial layer around the ventricles (yellow arrows in E, E’, F & F’). en (endocardium); epi (epicardium); IFT (inflow tract); LV (left ventricle); myo (myocardium); OFT (outflow tract); RV (right ventricle).
Activation of Wnt11-CreER at the late streak stage (E7.5)
To assess the expression of the Wnt11-CreER transgene at the late primitive streak stage, we administered tamoxifen at E7.5 and retrieved embryos at E8.5-8.75. In these embryos, we continued to see tdTomato expressing cells in the hindgut endoderm (red arrows in Fig.5B & C”) and embryonic vasculature including the dorsal aortae (green arrows, Fig.5C”), the inter-somitic vessels (green arrows, Fig.5B) and the omphalomesenteric vein (lower green arrow in Fig. 5C”). When E7.5-induced embryos were harvested at E12.5, Wnt11-CreER lineages were observed in the yolk sac and allantoic vasculature, and lining the elaborate fetal vasculature in the yolk sac (Suppl. Fig.S3A&A’) and placenta (Suppl. Fig.S3B & B’).
Figure 5. Cells expressing Wnt11-CreER at late streak stage contribute to the posterior endoderm and distinct mesodermal tissues.

Wnt11-CreER mediated recombination and activation of tdTomato expression was induced by a single dose of tamoxifen administration at E7.5, and embryos were harvested and analyzed at E8.75. (A) Epi-fluorescence of tdTomato alone, and (A’) merged with brightfield. (B) Saggital section of the embryonic region (yellow box in (A’) revealed the contribution of tdTomato expressing cell to the endoderm (red arrow in B), the inter-somitic vessels (ISV, green arrows in B), the dorsal aorta (DA) and the posterior lateral plate mesoderm (tdTomato-positive cells between dorsal aorta and endoderm). (C-C”) Transverse sectioning of these embryos along the dotted line in (A’) and co-staining with nuclear marker DAPI (blue) and endothelial cell marker PECAM (green) showed that tdTomato expressing cells colonized the posterior lateral plate and intermediate mesoderm (yellow arrows in C”), the hindgut endoderm (red arrow in (C”)), the notochordal plate (white arrow in C”) and the endothelial lining of the dorsal aortae (paired green arrows in C”) and the omphalomesenteric vein (lower green arrow in C”). DA (dorsal aortae); HG (hindgut); ISV (inter-somitic vessels); LPM (lateral plate mesoderm); NP (notochordal plate); OV (omphalomesenteric vein).
Compared to earlier inductions, inductions at E7.5 resulted in additional tdTomato-labeling of distinct mesodermal tissues including the notochordal plate (white arrow in Fig.5C”), the posterior intermediate and lateral plate mesoderm (yellow arrows; Fig.5C-C”) and the myocardium of the left ventricle (LV, see later). This is consistent with in situ analyses demonstrating that endogenous Wnt11 is expressed strongly in the node, posterior trunk mesoderm and the cardiac crescent (Kispert et al., 1996) (Suppl. Fig.S2).
Wnt11-CreER expression terminates in the embryonic endoderm and extra-embryonic vasculature shortly after gastrulation
Our analyses indicated that cells expressing Wnt11-CreER during gastrulation (E6.0-7.5) contributed extensively to the endodermal lineage, but endogenous Wnt11 expression was not detected in the endoderm of E8.5-10.5 mouse embryos (Kispert et al., 1996). A possible explanation for this apparent discrepancy is that Wnt11 expression is only transiently activated in endodermal progenitors, but is terminated in their descendent shortly after gastrulation. To test this hypothesis, we administered tamoxifen at E8.5-9.0 and harvested Wnt11-CreER; R26R-tdTomato embryos between E10.5 and E12.5. Interestingly, we found that in these E8.5-9.0 tamoxifen induced embryos, no tdTomato expressing cells were found in the epithelial linings of the fore-, mid- and hindgut endoderm (white arrows, Fig.6E-H). In fact, when we carefully re-examined the embryos induced at E7.5, we found that although some tdTomato expressing cell could be found in the midgut (Fig.6B) and hindgut endoderm (Fig.5B, 5C’ and 6C), they were already absent from the foregut endoderm (Fig.6A & D).
Figure 6. Wnt11-CreER expression terminates in the embryonic endoderm and the extra-embryonic vasculature shortly after gastrulation.
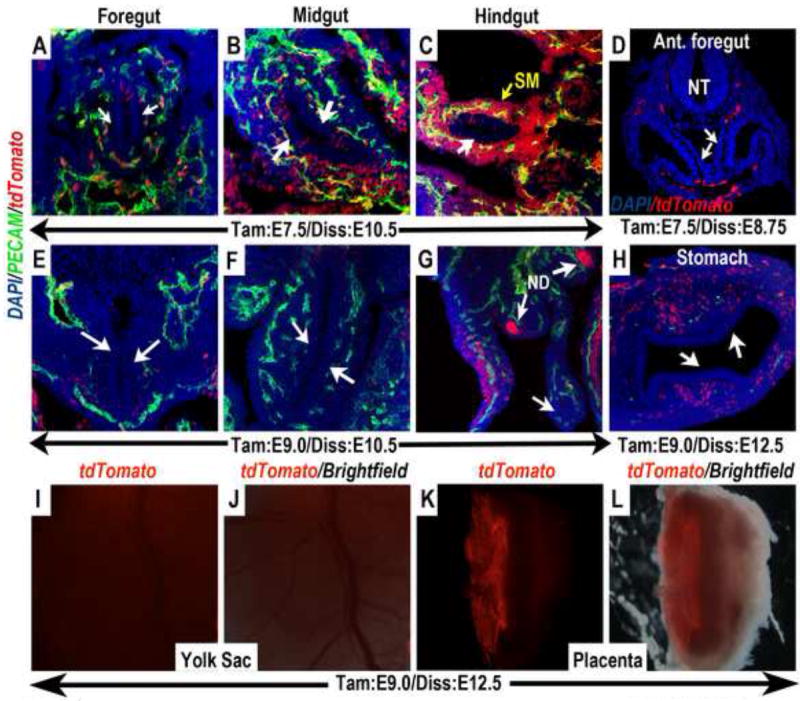
(A-D) In embryos in which a single dose of tamoxifen was administered at E7.5, the contribution of tdTomato expressing cells to the mid- (B) and hindgut (C) endoderm was reduced, and could no longer be found in the foregut endoderm (A & D). In contrast, abundant contribution of tdTomato expressing cells could be found in the splanchnic mesenchyme (SM) surrounding the hindgut (C) and various vasculature. (E-L) When tamoxifen was administered at E8.5-9.0, tdTomato expressing cells were completely absent from the foregut (white arrows in E and H), midgut (white arrows in F) and hindgut (white arrow in G) endoderm, but contributed specifically to the nephric duct (ND in G). The contribution of tdTomato expressing cells to the extra-embryonic vasculature in the yolk sac and placenta was also diminished. (I) Whole mount epi-fluorescence alone, and (J) merged image with brightfield revealed no tdTomato expressing cells in the yolk sac. (K and L) Bisection of the placenta revealed that tdTomato expressing cells also could no longer be found in the fetal vasculature (compare to Fig.4C-D”). The limited remaining tdTomato expression appeared to be in the extra-embryonic Reichert’s membrane. ND (nephric duct); NT (neural tube); SM (splanchnic mesenchyme).
Furthermore, we found that in E8.5-9.0 tamoxifen induced embryos, the contribution of tdTomato expressing cells to the extra-embryonic vasculature of the yolk sac and placenta was also diminished (Fig.6 E-H). These results together indicated that Wnt11-CreER was transiently expressed in the progenitors of the embryonic endoderm and extra-embryonic endothelium during gastrulation, but was turned off shortly afterward. This temporal analysis of Wnt11-CreER transgene during gastrulation also highlighted the significant advantage of CreER, which enabled us to determine not only the lineage contribution of Wnt11 expressing cells with high spatial resolution, but also the specific timing and duration of Wnt11 expression in each lineage.
Wnt11-CreER expressing cells contribute to multiple cardiac lineages in a spatio-temporally regulated manner
The most severe defect in Wnt11 mutant mice is malformation of the heart (Zhou et al., 2007), which can be further enhanced by simultaneous mutation of another non-canonical Wnt gene Wnt5a (Cohen et al., 2012). To more thoroughly understand how non-canonical Wnt signaling might promote cardiac development, we used Wnt11-CreER to investigate the specific spatial and temporal expression of Wnt11 in each lineage in the heart.
The Wnt11-CreER expression is initiated first in endocardial progenitors and maintained in the differentiated endocardium
The endocardium is a specialized layer of endothelial cells lining the myocardium. In Wnt11-CreER; R26R-tdTomato embryos induced at E6.5 and harvested at E8.5, tdTomato-labeled cells contributed not only to endothelial cells in the embryonic and extra-embryonic compartments (Fig.3), but also to the entire endocardium lining all chambers of the forming heart tube, including the inflow tract (IFT), the LV, the right ventricle (RV) and OFT (Fig.7A&A’; green arrows in Fig.7B & B’). In stark contrast, however, tdTomato-expressing cells did not contribute significantly to the myocardium in these embryos, and negligible numbers of tdTomato positive cells were found in the LV (Fig.7B, B’, white arrows). Therefore, the earliest onset of Wnt11 expression in cardiac progenitors appears to be within the progenitors of the endocardium during mid-gastrulation, and these Wnt11-expressing endocardial progenitors have already diverged from myocardial progenitors that will give rise to the first heart field (FHF) and SHF.
Unlike the transient expression of Wnt11-CreER in endothelial progenitors in the extra-embryonic compartment (Fig.6), Wnt11-CreER expression in the endocardium persisted for an extended period. When we performed tamoxifen inductions from E7.0 to 10.5, we continued to detect robust contribution of tdTomato labeled cells to the endocardium throughout the heart (Fig.7C-F’). This result suggests that Wnt11 is expressed not only in early endocardial progenitors at gastrulation, but also in the differentiated endocardium within the heart during organogenesis, and is consistent with expression of endogenous Wnt11 in the endocardium at E10.5 (Cohen et al., 2012) (Suppl. Fig.S4).
Sequential activation of Wnt11-CreER expression in the progenitors of the ventricular myocardium
Interestingly, when Wnt11-CreER; R26R-tdTomato embryos were induced with tamoxifen at different time points between E7.0 and 9.0 and harvested 1-4 days later, we observed a highly dynamic contribution of tdTomato-expressing cells to the ventricular myocardium. First, when tamoxifen was administered at E7.0-7.5, tdTomato-labeled cells contributed extensively to the myocardium in the LV and the inter-ventricular septum, but not to the myocardium of the RV, OFT or atrium (Fig.8A-D, Suppl. Fig.S5A & A’, Suppl. Fig.S6A & B). This pattern of contribution suggests that Wnt11 is expressed specifically in the myocardial progenitors of the FHF at E7.5, consistent with the in situ analysis showing that endogenous Wnt11 is expressed in the cardiac crescent (Kispert et al., 1996).
Figure 8. Sequential activation of dynamic Wnt11-CreER expression in the progenitors of the ventricular myocardium.
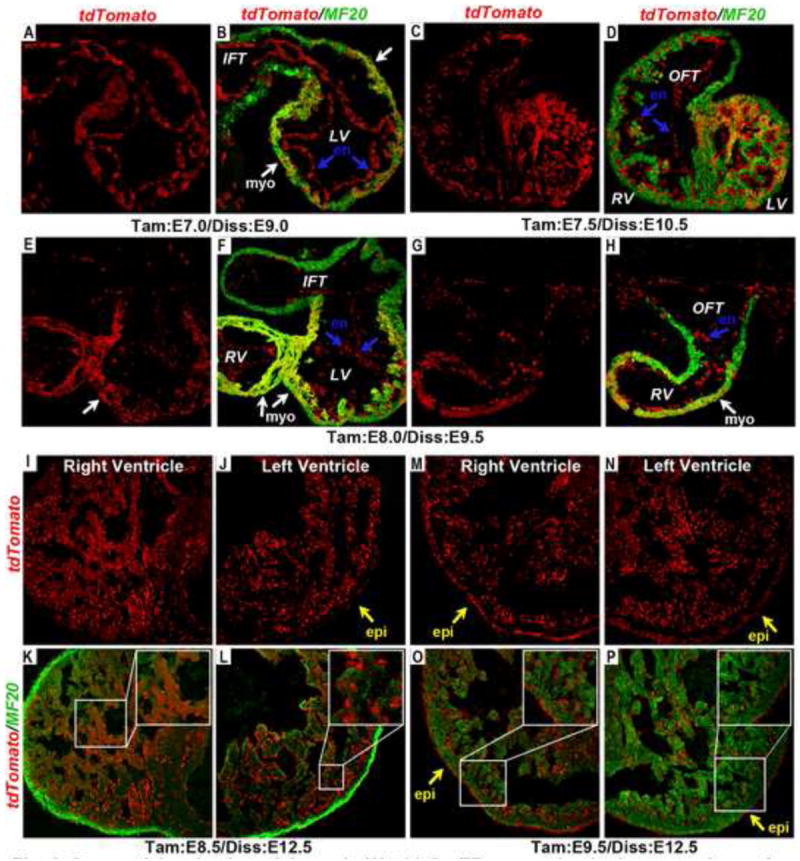
(A-D) When tamoxifen was administered at E7.0 (A, B) and 7.5 (C, D), tdTomato-labeled cells contributed extensively to the myocardium (MF20 positive, green) in the left ventricle and the inter-ventricular septum, but were absent from the myocardium of the right ventricle and OFT. (E-H) When tamoxifen was administered at E8.0, tdTomato-positive cells contributed to both the left and the right ventricular myocardium, but not to the atrial or OFT myocardium (F, H). (I-L) On the hand, when tamoxifen was administered at E8.5, tdTomato labeled cells contributed to the right ventricular myocardium (K), but their contribution to the left ventricular myocardium became diminished (L). (M-P) Finally, when tamoxifen was administered at E9.5, the contribution of tdTomato-positive cells to the left (P) and the right (O) ventricular myocardium was diminished. In all the stages analyzed above, tdTomato positive cells contributed extensively to the endocardium in all chambers. Additionally, sporadic tdTomato expressing cells were present in the epicardium of embryos that were tamoxifen-induced at E8.5 (yellow arrow in J). The entire epicardium became tdTomato positive when tamoxifen was administered at E9.5 (yellow arrows in M-P). Insets in K, L, O & P showed a magnified view of tdTomato expressing cells and MF20 positive myocardium. en (endocardium); epi (epicardium); IFT (Inflow tract); LV (left ventricle); myo (myocardium); OFT(outflow tract); RV (right ventricle).
Subsequently, when we administered tamoxifen at E8.0, we found that tdTomato-positive cells contributed to both the LV and the RV myocardium (Fig.8E & F), but not to the OFT myocardium (Fig.8 G & H). On the other hand, when tamoxifen was administered at E8.5, we found extensive contribution of tdTomato labeled cells to the RV and OFT myocardium (Fig.8I & K), but their contribution to the LV myocardium was diminished (Fig.8J & L, Suppl. Fig.S5B&B’).
Finally, when we performed tamoxifen induction at E9.5, we found that the contribution of tdTomato-positive cells to the RV myocardium was also diminished (Fig.8 M-P), and following inductions at E10.5, we could no longer find any tdTomato expressing cells in the myocardium of either ventricles (Fig.7 E-F’). Collectively, our analyses suggest Wnt11 is expressed in a highly dynamic and transient fashion, first in FHF precursors that give rise to the LV myocardium, and then in the SHF progenitors of the RV myocardium.
Spatially restricted contribution of Wnt11-CreER expressing cells to the OFT myocardium
When we administered tamoxifen at E8.5 and harvested embryos at E9.5, we found that in addition to the RV myocardium, tdTomato expressing cells were also present in the 2nd pharyngeal arch mesoderm known to harbor the SHF progenitors of the OFT myocardium (Kelly et al., 2001) (Fig. 9A and black arrows in Fig. 9B), and the contiguous superior wall of the OFT myocardium (Fig.9C). This spatially restricted contribution of Wnt11-CreER expressing cells to the superior wall of the OFT myocardium could be visualized also when we collected these E8.5-9.0 tamoxifen-induced embryos at E10.5. Sagittal and transverse sectioning and immunostaining with myocardial marker MF20 (an antibody against sarcomeric myosin heavy chain) in these embryos clearly indicated that tdTomato expressing cells were present only in the myocardium of the superior wall of the OFT (white arrow in Fig. 9D-I), but absent from the inferior ¼ region of the OFT myocardial wall (yellow arrow in Fig.9F-I).
Fig. 9. Spatially restricted contribution of Wnt11-CreER expressing cells to the OFT myocardium.
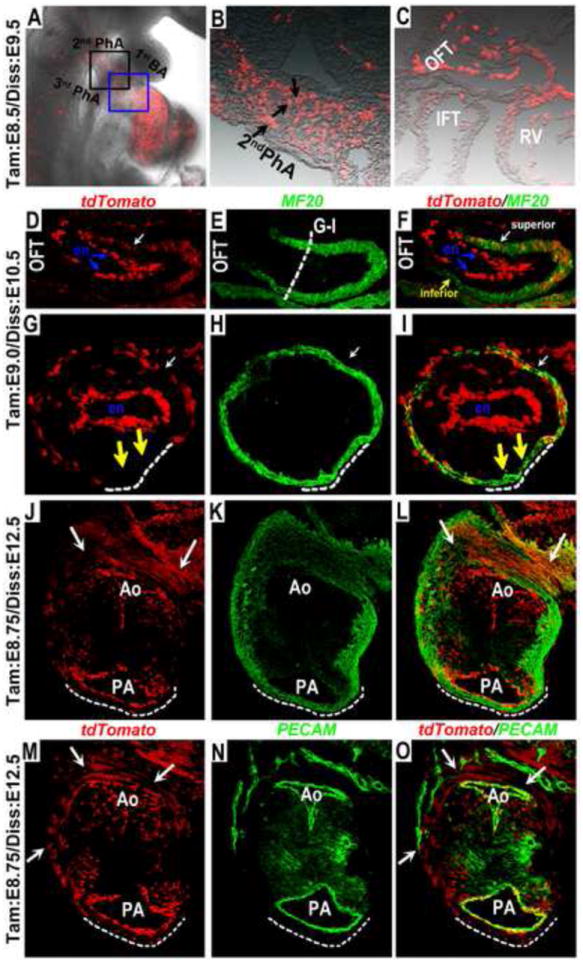
(A-C) Whole mount epi-fluorescent analysis showed that when tamoxifen was administered at E8.5, in the resulting E9.5 embryos tdTomato expressing cells were largely present in the mesoderm of the 2nd pharyngeal arch (black box in A and magnified view in B), the myocardium of the OFT superior wall and the right ventricle (blue box in A and magnified view in C), and the endocardium of the OFT, RV and IFT. (D-I) When embryos were harvested at E10.5 after tamoxifen induction at E9.0, tdTomato expressing cells were observed along the proximo-distal axis of the superior myocardial wall of the OFT (white arrow in D & F), which also co-expressed myocardial marker MF20. Transverse section of the OFT (G-I) indicated that the tdTomato expressing cells occupied ~3/4 of the superior and lateral OFT myocardial wall and were missing from the inferior OFT wall (white dotted line in G-I). When E9.0 tamoxifen induced embryos were harvested at E12.5, transverse sectioning and co-staining with MF-20 indicated that tdTomato labeled cells were present specifically in the sub-aortic myocardium (white arrows in J, L, M & O), but absent from sub-pulmonary myocardium (white dotted lines in J-O). In contrast, co-staining with PECAM indicated that tdTomato labeled cells contributed to the endocardium/endothelium in both the aorta and pulmonary artery. Ao (aorta), BA (branchial arch); en (endocadium); IFT (inflow tract); OFT (outflow tract); PA (pulmonary artery); PhA (pharyngeal arch); RV (right ventricle).
The specific contribution of the Wnt11-CreER lineage to the superior wall of the OFT myocardium also prompted us to determine its long-term developmental potential. Previous lineage tracing studies, using enhancer trap transgene y96-myf5-nlacz-16 (y96-16) and a set of Hox Cre lines, indicated that cells initially occupying the inferior wall of the OFT myocardium gave rise specifically to the cardiomyocytes at the base of the pulmonary artery (Bajolle et al., 2006; Bertrand et al., 2011; Theveniau-Ruissy et al., 2008). In contrast, when we performed longer term tracing, we found that the Wnt11-CreER lineage, which initially occupied the superior wall of the OFT myocardium, contributed primarily to the cardiomyocytes around the base of the aorta (arrows in Fig.9J-0), but not the pulmonary artery (white dotted lines in Fig.9J-O).
Therefore, together with the previous literature (Bajolle et al., 2008; Bertrand et al., 2011; Rochais et al., 2009; Theveniau-Ruissy et al., 2008), our results indicate that the superior and inferior wall of the early OFT myocardium are molecularly distinct and pre-determined to contribute specifically to the sub-aortic and -pulmonary myocardium, respectively.
Wnt11-CreER lineages contribute to the epicardium
In addition to the myocardium and endocardium, the heart contains a third lineage know as the epicardium. The epicardium is derived from cells in the pro-epicardial organ situated over the septum transversum caudal to the heart tube. Pro-epicardial cells extend and attach onto the myocardial layer of the forming atria and ventricles from E9.5 onward and rapidly spread over the heart to form a contiguous epicardial layer covering the entire myocardium by E11.5 (Rodgers et al., 2008).
When tamoxifen was administered at E8.5 or earlier, we detected only few tdTomato-expressing cells in the epicardial layer (yellow arrow in Fig. 8F). However, when tamoxifen induction was performed at E9.5 (Fig.8I-P), E10.5 (Fig.7E, F, G&H) or later (Suppl. Fig.S7 and data not shown), the entire epicardial layer was consisted of tdTomato-expressing cells. Consistent with our Wnt11-CreER analysis in the epicardium, a previous report by Cohen et. al. (Cohen et al., 2012) and our in situ analyses (Suppl. Fig.S4) showed endogenous Wnt11 transcripts in the epicardium of the developing heart.
Collectively, as summarized in Table 1, our lineage analysis with the Wnt11-CreER BAC transgene provided a high-resolution expression and fate map of Wnt11 expressing cells in the heart. Whereas Wnt11-CreER expression in the endocardial and epicardial lineages is more persistent and present throughout all chambers of the heart, its expression in the myocardium is highly dynamic and present only transiently and successively in precursors of the LV, RV and OFT myocardium. Notably, the only regions where we detected no contribution of Wnt11-CreER expressing cells were the atrial myocardium (Suppl. Fig.S6) and the inferior wall of the OFT myocardium (Fig.9D-I).
Table1. Summary of the spatio-temporally specific contribution of the Wnt11-CreER expressing cells to the embryonic heart.
Cre activity was transiently induced in Wnt11-CreER; R26R-tdTomato embryos by a single tamoxifen administration at E6.5-10.5, and embryos were harvested and analyzed at E8.5-12.5 to determine the contribution of tdTomato labeled cell in the epicardium and the myocardium and endocardium in the left ventricle (LV), right ventricle (RV), outflow tract (OFT) and atrium (A).
| Tamoxifen administration (Embryonic Day-E) | Dissection (Embryonic Day –E) | Myocardium | Endocardium | Epicardium | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| LV | RV | OFT | A | LV | RV | OFT | A | |||
| 6.5 | 8.5 | - | - | - | - | ++ | ++ | ++ | ++ | - |
| 7.0 | 9.0 | ++ | - | - | - | +++ | +++ | +++ | ++ | - |
| 7.5 | 9.5/10.5 | +++ | - | - | - | +++ | +++ | +++ | ++ | - |
| 8.0 | 9.5 | ++ | ++ | +/- | - | +++ | +++ | +++ | ++ | - |
| 8.5 | 12.5/16.5 | + | +++ | ++ | - | +++ | +++ | +++ | ++ | + |
| 9.0 | 10.5/12.5 | +/- | ++ | +++ | - | +++ | +++ | +++ | ++ | ++ |
| 9.5 | 12.5 | - | + | ++ | - | +++ | +++ | +++ | ++ | +++ |
| 10.5 | 12.5 | - | - | + | - | +++ | +++ | +++ | ++ | +++ |
The (+) and (-) indicate the presence and absence of tdTomato expressing cell in each compartment, respectively, with (+++) indicating highest levels of tdTomato expressing cells present.
Discussion
In recent years, the PCP pathway has been recognized as a key signaling mechanism that regulates tissue morphogenesis in diverse animal species, and has garnered attention from investigators across different disciplines. Activation of PCP-mediated morphogenetic processes requires non-canonical Wnts as ligands, and Wnt11 has been identified as one of the primary PCP ligands during zebrafish and Xenopus gastrulation (Heisenberg et al., 2000; Li et al., 2008; Schambony and Wedlich, 2007; Tada and Smith, 2000; Walentek et al., 2013). The potential role of Wnt11 as a PCP ligand in mammals, however, has remained less clear because Wnt11 mutant mice do not display some of the characteristic phenotypes observed consequent to mutation of core PCP genes Vangl2, Dvl and Celsr. Phenotypic differences may be due to altered expression patterns during evolution, or redundancy of other non-canonical Wnts. Therefore, expression maps with high spatial and temporal resolution need to be established for mammalian non-canonical Wnt genes. Secondly, whereas PCP signaling is best known for regulating gastrulation in zebrafish and Xenopus, its function in mammals has only been found in neurulation and development of specific organs, and its involvement in mammalian gastrulation has not been examined extensively.
In the current study, we attempted to establish a framework to further explore the role of Wnt11 in in mammalian gastrulation, by generating both a high-resolution expression map of Wnt11 and a fate map of Wnt11 expressing cells. To this end, we created a tamoxifen-inducible Wnt11-CreER BAC transgene in the mouse. Analysis of this transgene over the course of gastrulation uncovered surprisingly dynamic and specific expression of Wnt11 in the progenitors of the endodermal, endothelial and cardiac lineages, which had not been recognized from previous in situ studies. These experiments indicate that at the early streak stage (E6.0), Wnt11-CreER expression is initiated primarily in the progenitors of the endoderm, expands progressively to the mesodermal progenitors of the endothelial/ endocardial lineage at the mid streak stage (E6.5), and finally to distinct mesoderm of the cardiac, notochord, posterior intermediate and lateral plate lineages by E7.5. The timing of Wnt11-CreER expression in the endoderm and mesoderm suggests that these lineages may sequentially activate Wnt11 expression as they transit the primitive streak during gastrulation, suggesting a potential role for Wnt11-initiated signaling in egression of progenitors during germ layer formation.
A Wnt11-CreER BAC transgene faithfully recapitulates endogenous Wnt11 expression
Due to their large size, BAC transgenes have been known to recapitulate endogenous gene expression patterns (Lee et al., 2001). The BAC clone we used to create Wnt11-CreER transgene contained 75-100 kb genomic sequence flanking each end of the mouse Wnt11 locus. Our analyses of CreER-induced reporter gene expression shortly after a pulse of tamoxifen revealed that Wnt11-CreER expression closely mimicked the reported endogenous Wnt11 expression from gastrulation to mid-gestation (Fig.1 and (Kispert et al., 1996). Our longer term tracing experiments further revealed expression of Wnt11-CreER in tissues such as the endocardium, epicardium, and nephric duct (Fig.6C, 7&8), in which expression of endogenous Wnt11 has also been reported (Cohen et al., 2012; Kispert et al., 1996). Finally, the same BAC clone (RP23-122D14) used in our study was also used to create a separate Wnt11-Cre BAC transgenic line (Wnt11-myrTagRFP-IRES-CE) as part of the GUDMAP Consortium (the GenitoUrinary Development Molecular Anatomy Project). Although the analyses of Wnt11-myrTagRFP-IRES-CE focuses on genito-urinary development, and its expression during early embryogenesis has not been reported (https://www.gudmap.org/Docs/Mouse_Strains/18_Wnt11_allele_characterisation.pdf), its ability to faithfully recapitulate endogenous Wnt11 expression in the developing mesonephros between E11.5-15.5 supports the idea that BAC clone RP23-122D14 contains most of the elements required to regulate Wnt11 expression. Therefore, we predict that the expression and lineage tracing analyses performed in gastrulation stage embryos with our Wnt11-CreER transgene reflect endogenous Wnt11 expression. However, since the Wnt11-CreER BAC transgene is a random insertion, it remains possible that some of its expression domains may not reflect those of endogenous Wnt11. Therefore observations and conclusions that we made in the current study can be further validated with the Wnt11-myrTagRFP-IRES-CE BAC transgene in the future.
Transient expression of Wnt11 in endodermal progenitors during early gastrulation
Coordinated morphogenetic movements during gastrulation result in the establishment of the three germ layers: the endoderm, mesoderm and ectoderm. At the onset of zebrafish gastrulation, Wnt11 is expressed in the epiblast around the germ ring and activates PCP signaling in ingressed/involuted mesendodermal cells to enable their directed migration (Ulrich et al., 2003). Together with its closely related homolog Wnt11-r (Wnt11-related) and Wnt4, Wnt11 also activates PCP signaling in endoderm precursors to promote their migration toward the midline and fusion of the foregut (Matsui et al., 2005).
Similarly to zebrafish, in Xenopus Wnt11 expression is initiated in the superficial layer of the involuting marginal zone (IMZ) at the onset of gastrulation, starting from the dorsal region and gradually spreading laterally to surround the entire blastopore (Walentek et al., 2013). Involution of this superficial IMZ layer gives rise to both endoderm and mesoderm (Fukuda and Kikuchi, 2005; Keller, 1975; Shook et al., 2004), suggesting that like zebrafish, Xenopus Wnt11 is also produced by endodermal and mesodermal progenitors to regulate their movement during gastrulation (Li et al., 2008; Schambony and Wedlich, 2007; Tada and Smith, 2000; Walentek et al., 2013).
In the mouse, in situ analysis indicated that at the onset of gastrulation (E6.0-6.5), Wnt11 is expressed in scattered cells in the posterior part of the embryo around the primitive streak (Kispert et al., 1996). The identity and fate of these cells, however, were unknown. In our short-term tracing experiments, we found that the Wnt11-CreER transgene faithfully recapitulated this scattered expression pattern around the primitive streak (Fig.1D). Most interestingly, our long-term tracing experiments revealed that these Wnt11-expressing cells were predominantly progenitors of the embryonic endoderm only (Fig.2, 3 and 4). Their scattered distribution around the primitive streak suggests that Wnt11 expression may be initiated either in the epiblast cells prior to traversing the organizer (primitive streak) as in zebrafish and Xenopus, or in the specified endodermal cells that have exited the primitive streak. More detailed expression and marker analyses will need to be performed in the future to address this question. In either case, the Wnt11-CreER transgene will be a useful tool for time-lapse imaging to track and investigate cellular behaviors that underlie the morphogenesis of endodermal lineage.
In Xenopus, Wnt11 is also expressed later in the foregut endoderm progenitors at the neurula stage, and this phase of Wnt11 expression is proposed to act through both the canonical Wnt pathway to suppress the foregut fate and the non-canonical pathway to promote foregut morphogenesis (Li et al., 2008). In contrast, our analyses with the Wnt11-CreER transgene suggest that Wnt11 expression in the mouse endodermal lineage is only transient, and is largely diminished in foregut endoderm progenitors by late gastrulation at E7.5. Whether the transient expression of Wnt11 contributes to mouse endoderm development by promoting its gastrulation movement, morphogenesis or specification needs to be determined in the future. Wnt11 null mice die between E12.5 and P2 and analysis of their endodermal development has not been reported (Majumdar et al., 2003; Nagy et al., 2010).
Early Wnt11-expressing mesodermal cells contribute to the endothelial lineage
Studies in Xenopus indicate that Wnt11 transcription is activated directly by Brachyury (Xbra; T in mammals), an evolutionarily conserved transcription factor that confers mesodermal identity to the epiblast. Consequently, Wnt11 and Xbra expression patterns were described to overlap almost completely in Xenopus (Tada and Smith, 2000). Intriguingly, although the Xbra ortholog T is highly expressed in the primitive streak during gastrulation in the mouse (Inman and Downs, 2006), in situ studies do not detect significant Wnt11 expression in the primitive streak (Kispert et al., 1996) (Suppl. Fig.S2). Our short term tracing experiments using Wnt11-CreER also suggest that Wnt11 expression is absent from the primitive streak (Fig.1D-F), and our long-term tracing experiments further reveal that throughout gastrulation Wnt11-CreER expressing cells do not contribute to the paraxial mesoderm, which T-CreER expressing cells give rise to (Anderson et al., 2013). Therefore, in contrast to Xenopus, mouse Wnt11 expression does not seem to be activated broadly by T.
Whereas there is only one Wnt11 in mammals, there are two Wnt11 genes in zebrafish, Xenopus and chick: Wnt11 and Wnt11-r. Based on sequence conservation and synteny, it has been proposed that the two Wnt11 genes arose from an ancient duplication, the true otholog of fish/ frog Wnt11 was lost in mammals, and the remaining mammalian Wnt11 is in fact the ortholog of Wnt11-r (Garriock et al., 2005; Garriock et al., 2007; Hardy et al., 2008). The lack of mouse Wnt11 expression in the primitive streak and its strong expression in the cardiac fields like Xenopus Wnt11-r strongly support this idea. On the other hand, the expression of mouse Wnt11 in the endoderm progenitors during early gastrulation appears to resemble that of Xenopus Wnt11 (Tada and Smith, 2000; Walentek et al., 2013), but differ from that of Xenopus Wnt11-r, which is not initiated until late gastrula (Garriock et al., 2005). Furthermore, Xenopus Wnt11-r is not expressed in the node, but both previous in situ studies and our analyses with Wnt11-CreER reveal specific expression of mouse Wnt11 in the node (Fig.1E&F), where T is expressed (Kispert et al., 1996; Wang et al., 2006; Yamaguchi et al., 1999b). Therefore, it is likely that in some contexts, mouse Wnt11 could still be transcriptionally activated by T, like Xenopus Wnt11. Collectively, these data imply that whereas Xenopus Wnt11 and 11-r have evolved distinct expression patterns over time, the sole Wnt11 gene in mammals has retained some features of both Wnt11 and Wnt11-r, likely resembling the ancestral Wnt11 prior to gene duplication.
An unexpected finding from our studies with Wnt11-CreER is that during gastrulation, the first group of mesodermal cells expressing Wnt11 contributes specifically to the endothelial lineages in the embryonic and extra-embryonic compartments (Fig.2, 3 & 4). This finding is important because emerging evidence implicates a critical role for PCP/ non-canonical Wnt signaling in angiogenesis (Cirone et al., 2008; Descamps et al., 2012; Korn et al., 2014; Masckauchan et al., 2006; Stefater et al., 2011). Cell culture and zebrafish studies suggest that PCP signaling induced by non-canonical Wnts coordinates cellular polarity in endothelial cells to promote their migration, proliferation and survival during angiogenesis (Cirone et al., 2008; Masckauchan et al., 2006). Mouse genetic studies further reveal that endothelial cell-derived non-canonical Wnts act in an autocrine fashion to promote angiogenesis in postnatal retina and in tumors (Korn et al., 2014). Our analyses with Wnt11-CreER extend these findings by showing that Wnt11 is not only expressed in the endothelial lineage, but more importantly, its onset of expression occurs at early to mid-gastrulation, when endothelial progenitors first emerge from the posterior primitive streak. Therefore, our data raise the intriguing possibility that, in addition to angiogenesis, PCP/ non-canonical Wnt signaling may have earlier roles in endothelial progenitors prior to or during vasculogenesis, when formation of the primitive vascular plexus takes place. In support of this idea, mice lacking Fz2 and Fz7, two Fz receptors thought to signal through the PCP/ non-canonical Wnt pathway, fail to form an organized vascular plexus by E9.5, and die by E10.5 with the yolk sac engorged with blood (Yu et al., 2012). In addition to Wnt11, Wnt5a expression has also been reported in endothelial cells (Ishikawa et al., 2001) and Wnt11-/-; Wnt5a-/- mutants also die before E10.5 (Cohen et al., 2012). It will be interesting to determine in the future whether Wnt11-/-; Wnt5a-/- mutants display vascular abnormalities in addition to the reported heart defects.
Lastly, the onset of our Wnt11-CreER expression in the endothelial lineage appears to be earlier than the widely used Tie2-Cre (Kisanuki et al., 2001). With a single dose of tamoxifen administration at E6.5, we are able to detect extensive endothelial-labeling in the embryonic and extra-embryonic vasculature of Wnt11-CreER; R26-tdTomato embryos (Fig.3 and 4). Therefore, this Cre line can be a valuable tool for visualizing and investigating the early morphogenetic events associated with vasculogenesis, when groups of endothelial progenitor cells first differentiate and assemble into a network of small capillary vessels (Udan et al., 2013).
Wnt11 in heart development
The indispensable role of mammalian Wnt11 in heart development and function is clearly demonstrated by the severe heart defects in Wnt11-/- mutants (Cohen et al., 2012; Majumdar et al., 2003; Nagy et al., 2010; Zhou et al., 2007). Mouse and Xenopus studies have led to several models as to how Wnt11 functions in the heart, including establishing proper left-right asymmetry (Walentek et al., 2013), promoting myocardial specification by antagonizing canonical Wnt signaling (Cohen et al., 2012) or activating PCP signaling to increase cardiogenic gene expression (Afouda et al., 2008; Pandur et al., 2002), enhancing cell adhesion among ventricular cardiomyocytes (Garriock et al., 2005; Nagy et al., 2010), and activating Tgf expression to regulate OFT morphogenesis (Zhou et al., 2007). Our analyses with Wnt11-CreER revealed highly dynamic expression of Wnt11 in three major lineages of the heart, and highlight the challenges and potential approaches to further decipher the role of Wnt11 and non-canonical Wnt/ PCP signaling in mammalian heart formation.
Among the three cardiac lineages, our fate mapping analyses with Wnt11-CreER indicate that Wnt11 is expressed first in the progenitors of the endocardium. Extensive contribution of tdTomato-expressing cells to the endocardium was found when tamoxifen induction was performed at E6.5 (Fig.7), but their contribution to the myocardium did not occur until tamoxifen induction was performed at E7.0 and later (Fig.8). This result provides the genetic proof that in mouse, the majority of the endocardial and myocardial lineages have already diverged by mid-gastrulation, before they form the cardiac crescent. A similar conclusion was also reached in the chick by retroviral labeling of single cells (Wei and Mikawa, 2000). Moreover, in E6.5 tamoxifen-induced embryos the endothelium can also be labeled together with endocardium (Figs.3 and 4). Therefore, the endocardium and endothelium may arise either from a common pool of progenitors, as previously proposed (Milgrom-Hoffman et al., 2011), or from distinct epiblast precursors that happen to be specified and initiate Wnt11 expression at about the same time. Time lapse imaging with Wnt11-CreER embryos will help define the lineage relationship between the endothelium and endocardium in the future.
In contrast to its persistent expression in the endocardium throughout all chambers of the heart (Table 1), we found that Wnt11-CreER expression in the myocardium was highly dynamic and occurred in three waves: first in the precursors of the LV myocardium from E7.0 to 8.0; subsequently in the RV myocardium from E8.0 to 9.0; and finally in the OFT myocardium from E8.5 to 10.5. Its timing of expression coincides with the timing at which the progenitors in the FHF and SHF undergo myocardial differentiation to form the LV, RV and OFT. The expression pattern of mouse Wnt11 in the myocardium also resembles that of Xenopus Wnt11-r (Garriock et al., 2005), but unlike that of Xenopus Wnt11 which is expressed earlier in the dorsal marginal zone and promotes cardiac specification by activating Nkx2.5 and Gata-4/6 expression in the presumptive mesodermal progenitors (Pandur et al., 2002).
In Xenopus, the close timing between the onset of Wnt11-r expression and myocardial differentiation, together with the lack of myocardial specification defect in the Wnt11-r morphant, have led to the proposal that Wnt11-r may not function by inducing cardiac specification (Garriock et al., 2005). In contrast, mouse genetic studies have concluded that non-canonical Wnt signaling is required for myocardial differentiation, since Wnt11-/- mutants have smaller OFT and RV and Wnt11-/-; Wnt5a-/- mutants display severe loss of SHF progenitors and SHF-derived heart structures (Cohen et al., 2012). Given our finding that Wnt11 is expressed earlier in the foregut endoderm from E6.0 and the proximity between the endoderm and the SHF, it remains possible that endoderm-derived Wnt11 can act in a paracrine fashion to promote cardiac differentiation in the adjacent SHF progenitors in the mouse. Consistent with this idea, Xenopus Wnt11 is transcriptionally activated directly by Gata4/6 and mediates the cardiomyogenic effect of Gata4/6 (Afouda et al., 2008), and in the mouse Gata4/6 are expressed in the early endoderm (Morrisey et al., 1998; Rojas et al., 2009; Rojas et al., 2010). Therefore, in the future it will be important to investigate a potential Gata4/6→ Wnt11 pathway in the early endoderm for cardiac specification and differentiation in mouse.
The highly temporally specific and transient expression of Wnt11 within the mouse myocardium is also interesting, and may have additional implications for the potential functions of Wnt11 in the heart. For instance, Wnt11 may act in an autocrine fashion to enhance cell adhesion among cardiomyocytes as Xenopus Wnt11-r (Garriock et al., 2005), and we have found recently that as SHF progenitors enter the heart tube, they undergo a rapid up-regulation of cell cohesion (unpublished data, D.L. & J.W.). Alternatively, Wnt11 may activate PCP signaling to induce the morphogenetic events required for deploying myocardial progenitors from the SHF to the heart, as we have proposed previously for another non-canonical Wnt, Wnt5a (Sinha et al., 2012). Further manipulation of myocardial Wnt11 expression through both gain- and loss-of-function approaches will help elucidate its role in heart development and function.
Our analyses indicate that Wnt11 is also expressed in the epicardial lineage from E9.0 onwards. The functional importance of this expression is unclear. We note, however, that the ventricular myocardium in Wnt11-/- mutants displays reduced cell adhesion and aberrant cellular arrangement at E10.5 -12.5, when Wnt11 expression within the ventricular myocardium is already diminished (Fig.8, (Cohen et al., 2012; Nagy et al., 2010). Therefore, it is possible that Wnt11 secreted from the epicardium and/or endocardium can signal to the myocardium to regulate proper ventricular formation and function.
Finally, Wnt11 may also promote heart development by establishing proper left-right asymmetry. Core PCP genes in the mouse, including Dvl and Vangl, are required for posterior positioning of the nodal cilia to provide them with a posterior tilt (Hashimoto et al., 2010; Song et al., 2010). This posterior tilt allows rotational movement of the cilia to generate a leftward fluid in the node, thereby initiating left-right axis establishment and proper heart looping (Blum et al., 2009; Hirokawa et al., 2006). In Xenopus, Wnt11 is expressed by cells in the superficial mesoderm that involute to form the gastrocoel roof plate (GRP), the left-right organizer equivalent to the mouse node (Walentek et al., 2013). Blocking Wnt11 function in Xenopus perturbs GRP cilia polarization and GRP morphogenesis (Walentek et al., 2013). Therefore, given the strong and specific expression of Wnt11 in the mouse node (Kispert et al., 1996) (Fig.1, Suppl. Fig.S2), it may also play a role in nodal cilia polarization to regulate left-right asymmetry and heart looping in mammals.
In conclusion, our expression and lineage tracing studies with Wnt11-CreER have revealed unexpected early and specific expression of Wnt11 in endodermal and endothelial progenitors during mammalian gastrulation, and outlined highly dynamic and ordered expression of Wnt11 in various cardiac lineages during heart development. The insights gained from these findings will guide future investigations to further decipher the role of non-canonical Wnt/ PCP signaling in gastrulation and cardiovascular development.
Supplementary Material
Supplementary Figure S1. Synteny map of Wnt11 and the degree of sequence conservation of BAC clone RP23-122D14. (A) Synteny map for Wnt11 in human, chicken, Xenopus, zebrafish and mouse. (B) Schematic diagram of BAC clone RP23-122D14 that contains the mouse Wnt11 gene, and the alignment of the sequence in the BAC clone with that in the corresponding regions in Xenopus, zebrafish, chicken and human.
Supplementary Figure S2. Expression of Wnt11 in E7.5 mouse embryos. (A) Whole mount in situ hybridization showing that at E7.5, endogenous Wnt11 is expressed in the node (black arrow) and at the posterior end, around the base of the allantois. (B, C) Fluorescent in situ hybridization on transverse sections across the posterior region (red line in A) reveals that endogenous Wnt11 is not expressed within the primitive streak (ps, black and white arrows), but rather in the subjacent, presumptive mesoderm. (B) Fluorescent Wnt11 in situ signal (green) merged with brightfield view; (C) Wnt11 in situ signal merged with nuclei staining (DAPI, red). (D-F) Fluorescent in situ hybridization on sagittal sections across the distal region (red boxed area in A) indicates that endogenous Wnt11 is expressed in the node. (D) Fluorescent Wnt11 in situ signal alone, or merged with bridghtfield view (E) or nuclear staining (DAPI, F). A (anterior); EPC (ectoplacental cone); P (posterior); ps (primitive streak).
Supplementary Figure S3. Cells expressing Wnt11-CreER at E7.5 contributes to the extra-embryonic vasculature. When tamoxifen induction was performed at E7.5 and the embryos were recovered at E12.5, tdTomato expressing cells were found to line the vasculature in the yolk sac (tdTomato fluorescence alone (A) and merged with brightfied (A’)) and the placenta (tdTomato fluorescence alone (B) and merged with brightfield (B’)).
Supplementary Figure S4. Expression of Wnt11 in E10.5 mouse embryonic heart. (A-D) Fluorescent in situ hybridization on coronal sections of E10.5 mouse heart showing endogenous Wnt11 expression (A). Sections are co-stained with DAPI (B) and myocardial marker MF20 (C). (D) Merged view. (E-H) Enlarged view of the boxed area in (D) shows strongest expression of Wnt11 in the endocardium and epicardium (yellow arrows), but not in the myocardium. Endo (endocardium); Epi (epicardium).
Supplementary Figure S5. Dynamic contribution of Wnt11-CreER expressing cells to the ventricles. (A, A’) When tamoxifen was administered at E7.5, the resulting hearts displayed stronger tdTomato fluorescence in the left ventricle due to specific contribution of tdTomato cells to the left ventricular myocardium. (B, B’) Conversely, when tamoxifen was administered at E8.5, stronger tdTomato fluorescence was observed in the right ventricle due to specific contribution of tdTomato cells to the right ventricular myocardium.
Supplementary Figure S6. Wnt11-CreER lineage does not contribute to the inflow tract/atrial myocardium. When tamoxifen was administered at E7.0 (A, B) or E9.0 (C, D) and embryos were harvested at E9.0 and E10.5 respectively, no tdTomato expressing cells were observed in the MF20 positive myocardial layer in the inflow tract/atria, but could be found in the endocardial and epicardial layers throughout the heart. AV canal (atrioventricular canal); IFT (inflow tract); LV (left ventricle); OFT (outflow tract).
Supplementary Figure S7. Wnt11-CreER lineage contributes to the epicardium. (A, B) Sections of hearts from E18.5 Wnt11-CreER embryos that are induced with tamoxifen at E11.5. Co-staining with DAPI and epicardial marker WT1 reveal that the tdTomato-positive cells contribute specifically to the epicardium (white arrows in B). B is the enlarged view of the boxed area in A.
Highlights.
A Wnt11-CreER BAC transgene was created to recapitulate endogenous Wnt11 expression;
Lineage tracing revealed specific expression of Wnt11-CreER in endodermal and endothelial progenitors;
Wnt11-CreER expressing cells contribute to the three major cardiac lineages, the endocardium, myocardium and epicardium, in a spatio-temporally regulated manner;
Endocardium and myocardium are diverged by mid-gastrulation in the mouse.
Acknowledgments
This work was supported by NIH grant R01 HL109130 and American Heart Association Grants 0635262N and 11GRNT6980004 to JW and a pre-doctoral fellowship from AHA (12PRE12060081) to TS.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- Afouda BA, Martin J, Liu F, Ciau-Uitz A, Patient R, Hoppler S. GATA transcription factors integrate Wnt signalling during heart development. Development. 2008;135:3185–3190. doi: 10.1242/dev.026443. [DOI] [PubMed] [Google Scholar]
- Anderson MJ, Naiche LA, Wilson CP, Elder C, Swing DA, Lewandoski M. TCreERT2, a transgenic mouse line for temporal control of Cre-mediated recombination in lineages emerging from the primitive streak or tail bud. PloS one. 2013;8:e62479. doi: 10.1371/journal.pone.0062479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Angers S, Moon RT. Proximal events in Wnt signal transduction. Nat Rev Mol Cell Biol. 2009;10:468–477. doi: 10.1038/nrm2717. [DOI] [PubMed] [Google Scholar]
- Bajolle F, Zaffran S, Kelly RG, Hadchouel J, Bonnet D, Brown NA, Buckingham ME. Rotation of the myocardial wall of the outflow tract is implicated in the normal positioning of the great arteries. Circ Res. 2006;98:421–428. doi: 10.1161/01.RES.0000202800.85341.6e. [DOI] [PubMed] [Google Scholar]
- Bajolle F, Zaffran S, Meilhac SM, Dandonneau M, Chang T, Kelly RG, Buckingham ME. Myocardium at the base of the aorta and pulmonary trunk is prefigured in the outflow tract of the heart and in subdomains of the second heart field. Developmental biology. 2008;313:25–34. doi: 10.1016/j.ydbio.2007.09.023. [DOI] [PubMed] [Google Scholar]
- Bertrand N, Roux M, Ryckebusch L, Niederreither K, Dolle P, Moon A, Capecchi M, Zaffran S. Hox genes define distinct progenitor sub-domains within the second heart field. Developmental biology. 2011;353:266–274. doi: 10.1016/j.ydbio.2011.02.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blum M, Weber T, Beyer T, Vick P. Evolution of leftward flow. Seminars in cell & developmental biology. 2009;20:464–471. doi: 10.1016/j.semcdb.2008.11.005. [DOI] [PubMed] [Google Scholar]
- Cha SW, Tadjuidje E, Tao Q, Wylie C, Heasman J. Wnt5a and Wnt11 interact in a maternal Dkk1-regulated fashion to activate both canonical and non-canonical signaling in Xenopus axis formation. Development. 2008;135:3719–3729. doi: 10.1242/dev.029025. [DOI] [PubMed] [Google Scholar]
- Cha SW, Tadjuidje E, White J, Wells J, Mayhew C, Wylie C, Heasman J. Wnt11/5a complex formation caused by tyrosine sulfation increases canonical signaling activity. Current biology : CB. 2009;19:1573–1580. doi: 10.1016/j.cub.2009.07.062. [DOI] [PubMed] [Google Scholar]
- Cirone P, Lin S, Griesbach HL, Zhang Y, Slusarski DC, Crews CM. A role for planar cell polarity signaling in angiogenesis. Angiogenesis. 2008;11:347–360. doi: 10.1007/s10456-008-9116-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cohen ED, Miller MF, Wang Z, Moon RT, Morrisey EE. Wnt5a and Wnt11 are essential for second heart field progenitor development. Development. 2012;139:1931–1940. doi: 10.1242/dev.069377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Descamps B, Sewduth R, Ferreira Tojais N, Jaspard B, Reynaud A, Sohet F, Lacolley P, Allieres C, Lamaziere JM, Moreau C, Dufourcq P, Couffinhal T, Duplaa C. Frizzled 4 regulates arterial network organization through noncanonical Wnt/planar cell polarity signaling. Circ Res. 2012;110:47–58. doi: 10.1161/CIRCRESAHA.111.250936. [DOI] [PubMed] [Google Scholar]
- Feil R, Wagner J, Metzger D, Chambon P. Regulation of Cre recombinase activity by mutated estrogen receptor ligand-binding domains. Biochem Biophys Res Commun. 1997;237:752–757. doi: 10.1006/bbrc.1997.7124. [DOI] [PubMed] [Google Scholar]
- Fukuda K, Kikuchi Y. Endoderm development in vertebrates: fate mapping, induction and regional specification. Development, growth & differentiation. 2005;47:343–355. doi: 10.1111/j.1440-169X.2005.00815.x. [DOI] [PubMed] [Google Scholar]
- Gao B, Song H, Bishop K, Elliot G, Garrett L, English MA, Andre P, Robinson J, Sood R, Minami Y, Economides AN, Yang Y. Wnt signaling gradients establish planar cell polarity by inducing Vangl2 phosphorylation through Ror2. Developmental cell. 2011;20:163–176. doi: 10.1016/j.devcel.2011.01.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garriock RJ, D’Agostino SL, Pilcher KC, Krieg PA. Wnt11-R, a protein closely related to mammalian Wnt11, is required for heart morphogenesis in Xenopus. Developmental biology. 2005;279:179–192. doi: 10.1016/j.ydbio.2004.12.013. [DOI] [PubMed] [Google Scholar]
- Garriock RJ, Warkman AS, Meadows SM, D’Agostino S, Krieg PA. Census of vertebrate Wnt genes: isolation and developmental expression of Xenopus Wnt2, Wnt3, Wnt9a, Wnt9b, Wnt10a, and Wnt16. Developmental dynamics : an official publication of the American Association of Anatomists. 2007;236:1249–1258. doi: 10.1002/dvdy.21156. [DOI] [PubMed] [Google Scholar]
- Goodrich LV, Strutt D. Principles of planar polarity in animal development. Development. 2011;138:1877–1892. doi: 10.1242/dev.054080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gros J, Hu JK, Vinegoni C, Feruglio PF, Weissleder R, Tabin CJ. WNT5A/JNK and FGF/MAPK pathways regulate the cellular events shaping the vertebrate limb bud. Current biology : CB. 2010;20:1993–2002. doi: 10.1016/j.cub.2010.09.063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Habas R, Kato Y, He X. Wnt/Frizzled activation of Rho regulates vertebrate gastrulation and requires a novel Formin homology protein Daam1. Cell. 2001;107:843–854. doi: 10.1016/s0092-8674(01)00614-6. [DOI] [PubMed] [Google Scholar]
- Hardy KM, Garriock RJ, Yatskievych TA, D’Agostino SL, Antin PB, Krieg PA. Non-canonical Wnt signaling through Wnt5a/b and a novel Wnt11 gene, Wnt11b, regulates cell migration during avian gastrulation. Developmental biology. 2008;320:391–401. doi: 10.1016/j.ydbio.2008.05.546. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hashimoto M, Shinohara K, Wang J, Ikeuchi S, Yoshiba S, Meno C, Nonaka S, Takada S, Hatta K, Wynshaw-Boris A, Hamada H. Planar polarization of node cells determines the rotational axis of node cilia. Nature cell biology. 2010;12:170–176. doi: 10.1038/ncb2020. [DOI] [PubMed] [Google Scholar]
- Heisenberg CP, Tada M, Rauch GJ, Saude L, Concha ML, Geisler R, Stemple DL, Smith JC, Wilson SW. Silberblick/Wnt11 mediates convergent extension movements during zebrafish gastrulation. Nature. 2000;405:76–81. doi: 10.1038/35011068. [DOI] [PubMed] [Google Scholar]
- Hirokawa N, Tanaka Y, Okada Y, Takeda S. Nodal flow and the generation of left-right asymmetry. Cell. 2006;125:33–45. doi: 10.1016/j.cell.2006.03.002. [DOI] [PubMed] [Google Scholar]
- Inman KE, Downs KM. Localization of Brachyury (T) in embryonic and extraembryonic tissues during mouse gastrulation. Gene expression patterns : GEP. 2006;6:783–793. doi: 10.1016/j.modgep.2006.01.010. [DOI] [PubMed] [Google Scholar]
- Ishikawa T, Tamai Y, Zorn AM, Yoshida H, Seldin MF, Nishikawa S, Taketo MM. Mouse Wnt receptor gene Fzd5 is essential for yolk sac and placental angiogenesis. Development. 2001;128:25–33. doi: 10.1242/dev.128.1.25. [DOI] [PubMed] [Google Scholar]
- Keller R. Shaping the vertebrate body plan by polarized embryonic cell movements. Science. 2002;298:1950–1954. doi: 10.1126/science.1079478. [DOI] [PubMed] [Google Scholar]
- Keller RE. Vital dye mapping of the gastrula and neurula of Xenopus laevis I. Prospective areas and morphogenetic movements of the superficial layer. Developmental biology. 1975;42:222–241. doi: 10.1016/0012-1606(75)90331-0. [DOI] [PubMed] [Google Scholar]
- Kelly RG, Brown NA, Buckingham ME. The arterial pole of the mouse heart forms from Fgf10-expressing cells in pharyngeal mesoderm. Developmental cell. 2001;1:435–440. doi: 10.1016/s1534-5807(01)00040-5. [DOI] [PubMed] [Google Scholar]
- Kilian B, Mansukoski H, Barbosa FC, Ulrich F, Tada M, Heisenberg CP. The role of Ppt/Wnt5 in regulating cell shape and movement during zebrafish gastrulation. Mechanisms of development. 2003;120:467–476. doi: 10.1016/s0925-4773(03)00004-2. [DOI] [PubMed] [Google Scholar]
- Kisanuki YY, Hammer RE, Miyazaki J, Williams SC, Richardson JA, Yanagisawa M. Tie2-Cre transgenic mice: a new model for endothelial cell-lineage analysis in vivo. Developmental biology. 2001;230:230–242. doi: 10.1006/dbio.2000.0106. [DOI] [PubMed] [Google Scholar]
- Kispert A, Vainio S, Shen L, Rowitch DH, McMahon AP. Proteoglycans are required for maintenance of Wnt-11 expression in the ureter tips. Development. 1996;122:3627–3637. doi: 10.1242/dev.122.11.3627. [DOI] [PubMed] [Google Scholar]
- Korn C, Scholz B, Hu J, Srivastava K, Wojtarowicz J, Arnsperger T, Adams RH, Boutros M, Augustin HG, Augustin I. Endothelial cell-derived non-canonical Wnt ligands control vascular pruning in angiogenesis. Development. 2014;141:1757–1766. doi: 10.1242/dev.104422. [DOI] [PubMed] [Google Scholar]
- Lee EC, Yu D, Martinez de Velasco J, Tessarollo L, Swing DA, Court DL, Jenkins NA, Copeland NG. A highly efficient Escherichia coli-based chromosome engineering system adapted for recombinogenic targeting and subcloning of BAC DNA. Genomics. 2001;73:56–65. doi: 10.1006/geno.2000.6451. [DOI] [PubMed] [Google Scholar]
- Leone DP, Genoud S, Atanasoski S, Grausenburger R, Berger P, Metzger D, Macklin WB, Chambon P, Suter U. Tamoxifen-inducible glia-specific Cre mice for somatic mutagenesis in oligodendrocytes and Schwann cells. Mol Cell Neurosci. 2003;22:430–440. doi: 10.1016/s1044-7431(03)00029-0. [DOI] [PubMed] [Google Scholar]
- Li Y, Rankin SA, Sinner D, Kenny AP, Krieg PA, Zorn AM. Sfrp5 coordinates foregut specification and morphogenesis by antagonizing both canonical and noncanonical Wnt11 signaling. Genes & development. 2008;22:3050–3063. doi: 10.1101/gad.1687308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- MacDonald BT, Tamai K, He X. Wnt/beta-catenin signaling: components, mechanisms, and diseases. Developmental cell. 2009;17:9–26. doi: 10.1016/j.devcel.2009.06.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Madisen L, Zwingman TA, Sunkin SM, Oh SW, Zariwala HA, Gu H, Ng LL, Palmiter RD, Hawrylycz MJ, Jones AR, Lein ES, Zeng H. A robust and high-throughput Cre reporting and characterization system for the whole mouse brain. Nat Neurosci. 2010;13:133–140. doi: 10.1038/nn.2467. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Majumdar A, Vainio S, Kispert A, McMahon J, McMahon AP. Wnt11 and Ret/Gdnf pathways cooperate in regulating ureteric branching during metanephric kidney development. Development. 2003;130:3175–3185. doi: 10.1242/dev.00520. [DOI] [PubMed] [Google Scholar]
- Masckauchan TN, Agalliu D, Vorontchikhina M, Ahn A, Parmalee NL, Li CM, Khoo A, Tycko B, Brown AM, Kitajewski J. Wnt5a signaling induces proliferation and survival of endothelial cells in vitro and expression of MMP-1 and Tie-2. Molecular biology of the cell. 2006;17:5163–5172. doi: 10.1091/mbc.E06-04-0320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matsui T, Raya A, Kawakami Y, Callol-Massot C, Capdevila J, Rodriguez-Esteban C, Izpisua Belmonte JC. Noncanonical Wnt signaling regulates midline convergence of organ primordia during zebrafish development. Genes & development. 2005;19:164–175. doi: 10.1101/gad.1253605. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Milgrom-Hoffman M, Harrelson Z, Ferrara N, Zelzer E, Evans SM, Tzahor E. The heart endocardium is derived from vascular endothelial progenitors. Development. 2011;138:4777–4787. doi: 10.1242/dev.061192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morrisey EE, Tang Z, Sigrist K, Lu MM, Jiang F, Ip HS, Parmacek MS. GATA6 regulates HNF4 and is required for differentiation of visceral endoderm in the mouse embryo. Genes & development. 1998;12:3579–3590. doi: 10.1101/gad.12.22.3579. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nagy I, Railo A, Rapila R, Hast T, Sormunen R, Tavi P, Rasanen J, Vainio SJ. Wnt-11 signalling controls ventricular myocardium development by patterning N-cadherin and beta-catenin expression. Cardiovasc Res. 2010;85:100–109. doi: 10.1093/cvr/cvp254. [DOI] [PubMed] [Google Scholar]
- Nagy A, Gertsenstein M, Vintersten K, Behringer R. Staining Whole Mouse Embryos for {beta}-Galactosidase (lacZ) Activity. CSH Protoc. 2007;2007:pdb prot4725. doi: 10.1101/pdb.prot4725. [DOI] [PubMed] [Google Scholar]
- Pandur P, Lasche M, Eisenberg LM, Kuhl M. Wnt-11 activation of a non-canonical Wnt signalling pathway is required for cardiogenesis. Nature. 2002;418:636–641. doi: 10.1038/nature00921. [DOI] [PubMed] [Google Scholar]
- Rochais F, Dandonneau M, Mesbah K, Jarry T, Mattei MG, Kelly RG. Hes1 is expressed in the second heart field and is required for outflow tract development. PloS one. 2009;4:e6267. doi: 10.1371/journal.pone.0006267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rodgers LS, Lalani S, Runyan RB, Camenisch TD. Differential growth and multicellular villi direct proepicardial translocation to the developing mouse heart. Developmental dynamics : an official publication of the American Association of Anatomists. 2008;237:145–152. doi: 10.1002/dvdy.21378. [DOI] [PubMed] [Google Scholar]
- Rojas A, Schachterle W, Xu SM, Black BL. An endoderm-specific transcriptional enhancer from the mouse Gata4 gene requires GATA and homeodomain protein-binding sites for function in vivo. Developmental dynamics : an official publication of the American Association of Anatomists. 2009;238:2588–2598. doi: 10.1002/dvdy.22091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rojas A, Schachterle W, Xu SM, Martin F, Black BL. Direct transcriptional regulation of Gata4 during early endoderm specification is controlled by FoxA2 binding to an intronic enhancer. Developmental biology. 2010;346:346–355. doi: 10.1016/j.ydbio.2010.07.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schambony A, Wedlich D. Wnt-5A/Ror2 regulate expression of XPAPC through an alternative noncanonical signaling pathway. Developmental cell. 2007;12:779–792. doi: 10.1016/j.devcel.2007.02.016. [DOI] [PubMed] [Google Scholar]
- Shaner NC, Patterson GH, Davidson MW. Advances in fluorescent protein technology. J Cell Sci. 2007;120:4247–4260. doi: 10.1242/jcs.005801. [DOI] [PubMed] [Google Scholar]
- Shook DR, Majer C, Keller R. Pattern and morphogenesis of presumptive superficial mesoderm in two closely related species, Xenopus laevis and Xenopus tropicalis. Developmental biology. 2004;270:163–185. doi: 10.1016/j.ydbio.2004.02.021. [DOI] [PubMed] [Google Scholar]
- Sinha T, Wang B, Evans S, Wynshaw-Boris A, Wang J. Disheveled mediated planar cell polarity signaling is required in the second heart field lineage for outflow tract morphogenesis. Developmental biology. 2012;370:135–144. doi: 10.1016/j.ydbio.2012.07.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Song H, Hu J, Chen W, Elliott G, Andre P, Gao B, Yang Y. Planar cell polarity breaks bilateral symmetry by controlling ciliary positioning. Nature. 2010;466:378–382. doi: 10.1038/nature09129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Soriano P. Generalized lacZ expression with the ROSA26 Cre reporter strain. Nat Genet. 1999;21:70–71. doi: 10.1038/5007. [DOI] [PubMed] [Google Scholar]
- Stefater JA, 3rd, Lewkowich I, Rao S, Mariggi G, Carpenter AC, Burr AR, Fan J, Ajima R, Molkentin JD, Williams BO, Wills-Karp M, Pollard JW, Yamaguchi T, Ferrara N, Gerhardt H, Lang RA. Regulation of angiogenesis by a non-canonical Wnt-Flt1 pathway in myeloid cells. Nature. 2011;474:511–515. doi: 10.1038/nature10085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tada M, Smith JC. Xwnt11 is a target of Xenopus Brachyury: regulation of gastrulation movements via Dishevelled, but not through the canonical Wnt pathway. Development. 2000;127:2227–2238. doi: 10.1242/dev.127.10.2227. [DOI] [PubMed] [Google Scholar]
- Tao Q, Yokota C, Puck H, Kofron M, Birsoy B, Yan D, Asashima M, Wylie CC, Lin X, Heasman J. Maternal wnt11 activates the canonical wnt signaling pathway required for axis formation in Xenopus embryos. Cell. 2005;120:857–871. doi: 10.1016/j.cell.2005.01.013. [DOI] [PubMed] [Google Scholar]
- Theveniau-Ruissy M, Dandonneau M, Mesbah K, Ghez O, Mattei MG, Miquerol L, Kelly RG. The del22q11.2 candidate gene Tbx1 controls regional outflow tract identity and coronary artery patterning. Circ Res. 2008;103:142–148. doi: 10.1161/CIRCRESAHA.108.172189. [DOI] [PubMed] [Google Scholar]
- Tree DR, Ma D, Axelrod JD. A three-tiered mechanism for regulation of planar cell polarity. Seminars in cell & developmental biology. 2002;13:217–224. doi: 10.1016/s1084-9521(02)00042-3. [DOI] [PubMed] [Google Scholar]
- Udan RS, Vadakkan TJ, Dickinson ME. Dynamic responses of endothelial cells to changes in blood flow during vascular remodeling of the mouse yolk sac. Development. 2013;140:4041–4050. doi: 10.1242/dev.096255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ulrich F, Concha ML, Heid PJ, Voss E, Witzel S, Roehl H, Tada M, Wilson SW, Adams RJ, Soll DR, Heisenberg CP. Slb/Wnt11 controls hypoblast cell migration and morphogenesis at the onset of zebrafish gastrulation. Development. 2003;130:5375–5384. doi: 10.1242/dev.00758. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van Amerongen R. Alternative Wnt pathways and receptors. Cold Spring Harbor perspectives in biology. 2012;4 doi: 10.1101/cshperspect.a007914. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van Amerongen R, Nusse R. Towards an integrated view of Wnt signaling in development. Development. 2009;136:3205–3214. doi: 10.1242/dev.033910. [DOI] [PubMed] [Google Scholar]
- Walentek P, Schneider I, Schweickert A, Blum M. Wnt11b is involved in cilia-mediated symmetry breakage during Xenopus left-right development. PloS one. 2013;8:e73646. doi: 10.1371/journal.pone.0073646. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wallingford JB. Planar cell polarity and the developmental control of cell behavior in vertebrate embryos. Annual review of cell and developmental biology. 2012;28:627–653. doi: 10.1146/annurev-cellbio-092910-154208. [DOI] [PubMed] [Google Scholar]
- Wallingford JB, Rowning BA, Vogeli KM, Rothbacher U, Fraser SE, Harland RM. Dishevelled controls cell polarity during Xenopus gastrulation. Nature. 2000;405:81–85. doi: 10.1038/35011077. [DOI] [PubMed] [Google Scholar]
- Wallingford JB, Vogeli KM, Harland RM. Regulation of convergent extension in Xenopus by Wnt5a and Frizzled-8 is independent of the canonical Wnt pathway. Int J Dev Biol. 2001;45:225–227. [PubMed] [Google Scholar]
- Wang J, Hamblet NS, Mark S, Dickinson ME, Brinkman BC, Segil N, Fraser SE, Chen P, Wallingford JB, Wynshaw-Boris A. Dishevelled genes mediate a conserved mammalian PCP pathway to regulate convergent extension during neurulation. Development. 2006;133:1767–1778. doi: 10.1242/dev.02347. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang J, Mager J, Schnedier E, Magnuson T. The mouse PcG gene eed is required for Hox gene repression and extraembryonic development. Mammalian genome : official journal of the International Mammalian Genome Society. 2002;13:493–503. doi: 10.1007/s00335-002-2182-7. [DOI] [PubMed] [Google Scholar]
- Wang J, Sinha T, Wynshaw-Boris A. Wnt signaling in mammalian development: lessons from mouse genetics. Cold Spring Harbor perspectives in biology. 2012;4 doi: 10.1101/cshperspect.a007963. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wei Y, Mikawa T. Fate diversity of primitive streak cells during heart field formation in ovo. Developmental dynamics : an official publication of the American Association of Anatomists. 2000;219:505–513. doi: 10.1002/1097-0177(2000)9999:9999<::AID-DVDY1076>3.0.CO;2-6. [DOI] [PubMed] [Google Scholar]
- Wilkinson DG, Nieto MA. Detection of messenger RNA by in situ hybridization to tissue sections and whole mounts. Methods Enzymol. 1993;225:361–373. doi: 10.1016/0076-6879(93)25025-w. [DOI] [PubMed] [Google Scholar]
- Willert K, Nusse R. Wnt proteins. Cold Spring Harbor perspectives in biology. 2012;4:a007864. doi: 10.1101/cshperspect.a007864. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Witzel S, Zimyanin V, Carreira-Barbosa F, Tada M, Heisenberg CP. Wnt11 controls cell contact persistence by local accumulation of Frizzled 7 at the plasma membrane. The Journal of cell biology. 2006;175:791–802. doi: 10.1083/jcb.200606017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamaguchi TP, Bradley A, McMahon AP, Jones S. A Wnt5a pathway underlies outgrowth of multiple structures in the vertebrate embryo. Development. 1999a;126:1211–1223. doi: 10.1242/dev.126.6.1211. [DOI] [PubMed] [Google Scholar]
- Yamaguchi TP, Takada S, Yoshikawa Y, Wu N, McMahon AP. T (Brachyury) is a direct target of Wnt3a during paraxial mesoderm specification. Genes & development. 1999b;13:3185–3190. doi: 10.1101/gad.13.24.3185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yu H, Ye X, Guo N, Nathans J. Frizzled 2 and frizzled 7 function redundantly in convergent extension and closure of the ventricular septum and palate: evidence for a network of interacting genes. Development. 2012;139:4383–4394. doi: 10.1242/dev.083352. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zallen JA. Planar polarity and tissue morphogenesis. Cell. 2007;129:1051–1063. doi: 10.1016/j.cell.2007.05.050. [DOI] [PubMed] [Google Scholar]
- Zhou W, Lin L, Majumdar A, Li X, Zhang X, Liu W, Etheridge L, Shi Y, Martin J, Van de Ven W, Kaartinen V, Wynshaw-Boris A, McMahon AP, Rosenfeld MG, Evans SM. Modulation of morphogenesis by noncanonical Wnt signaling requires ATF/CREB family-mediated transcriptional activation of TGFbeta2. Nat Genet. 2007;39:1225–1234. doi: 10.1038/ng2112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zorn AM, Wells JM. Vertebrate endoderm development and organ formation. Annual review of cell and developmental biology. 2009;25:221–251. doi: 10.1146/annurev.cellbio.042308.113344. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary Figure S1. Synteny map of Wnt11 and the degree of sequence conservation of BAC clone RP23-122D14. (A) Synteny map for Wnt11 in human, chicken, Xenopus, zebrafish and mouse. (B) Schematic diagram of BAC clone RP23-122D14 that contains the mouse Wnt11 gene, and the alignment of the sequence in the BAC clone with that in the corresponding regions in Xenopus, zebrafish, chicken and human.
Supplementary Figure S2. Expression of Wnt11 in E7.5 mouse embryos. (A) Whole mount in situ hybridization showing that at E7.5, endogenous Wnt11 is expressed in the node (black arrow) and at the posterior end, around the base of the allantois. (B, C) Fluorescent in situ hybridization on transverse sections across the posterior region (red line in A) reveals that endogenous Wnt11 is not expressed within the primitive streak (ps, black and white arrows), but rather in the subjacent, presumptive mesoderm. (B) Fluorescent Wnt11 in situ signal (green) merged with brightfield view; (C) Wnt11 in situ signal merged with nuclei staining (DAPI, red). (D-F) Fluorescent in situ hybridization on sagittal sections across the distal region (red boxed area in A) indicates that endogenous Wnt11 is expressed in the node. (D) Fluorescent Wnt11 in situ signal alone, or merged with bridghtfield view (E) or nuclear staining (DAPI, F). A (anterior); EPC (ectoplacental cone); P (posterior); ps (primitive streak).
Supplementary Figure S3. Cells expressing Wnt11-CreER at E7.5 contributes to the extra-embryonic vasculature. When tamoxifen induction was performed at E7.5 and the embryos were recovered at E12.5, tdTomato expressing cells were found to line the vasculature in the yolk sac (tdTomato fluorescence alone (A) and merged with brightfied (A’)) and the placenta (tdTomato fluorescence alone (B) and merged with brightfield (B’)).
Supplementary Figure S4. Expression of Wnt11 in E10.5 mouse embryonic heart. (A-D) Fluorescent in situ hybridization on coronal sections of E10.5 mouse heart showing endogenous Wnt11 expression (A). Sections are co-stained with DAPI (B) and myocardial marker MF20 (C). (D) Merged view. (E-H) Enlarged view of the boxed area in (D) shows strongest expression of Wnt11 in the endocardium and epicardium (yellow arrows), but not in the myocardium. Endo (endocardium); Epi (epicardium).
Supplementary Figure S5. Dynamic contribution of Wnt11-CreER expressing cells to the ventricles. (A, A’) When tamoxifen was administered at E7.5, the resulting hearts displayed stronger tdTomato fluorescence in the left ventricle due to specific contribution of tdTomato cells to the left ventricular myocardium. (B, B’) Conversely, when tamoxifen was administered at E8.5, stronger tdTomato fluorescence was observed in the right ventricle due to specific contribution of tdTomato cells to the right ventricular myocardium.
Supplementary Figure S6. Wnt11-CreER lineage does not contribute to the inflow tract/atrial myocardium. When tamoxifen was administered at E7.0 (A, B) or E9.0 (C, D) and embryos were harvested at E9.0 and E10.5 respectively, no tdTomato expressing cells were observed in the MF20 positive myocardial layer in the inflow tract/atria, but could be found in the endocardial and epicardial layers throughout the heart. AV canal (atrioventricular canal); IFT (inflow tract); LV (left ventricle); OFT (outflow tract).
Supplementary Figure S7. Wnt11-CreER lineage contributes to the epicardium. (A, B) Sections of hearts from E18.5 Wnt11-CreER embryos that are induced with tamoxifen at E11.5. Co-staining with DAPI and epicardial marker WT1 reveal that the tdTomato-positive cells contribute specifically to the epicardium (white arrows in B). B is the enlarged view of the boxed area in A.


