Abstract
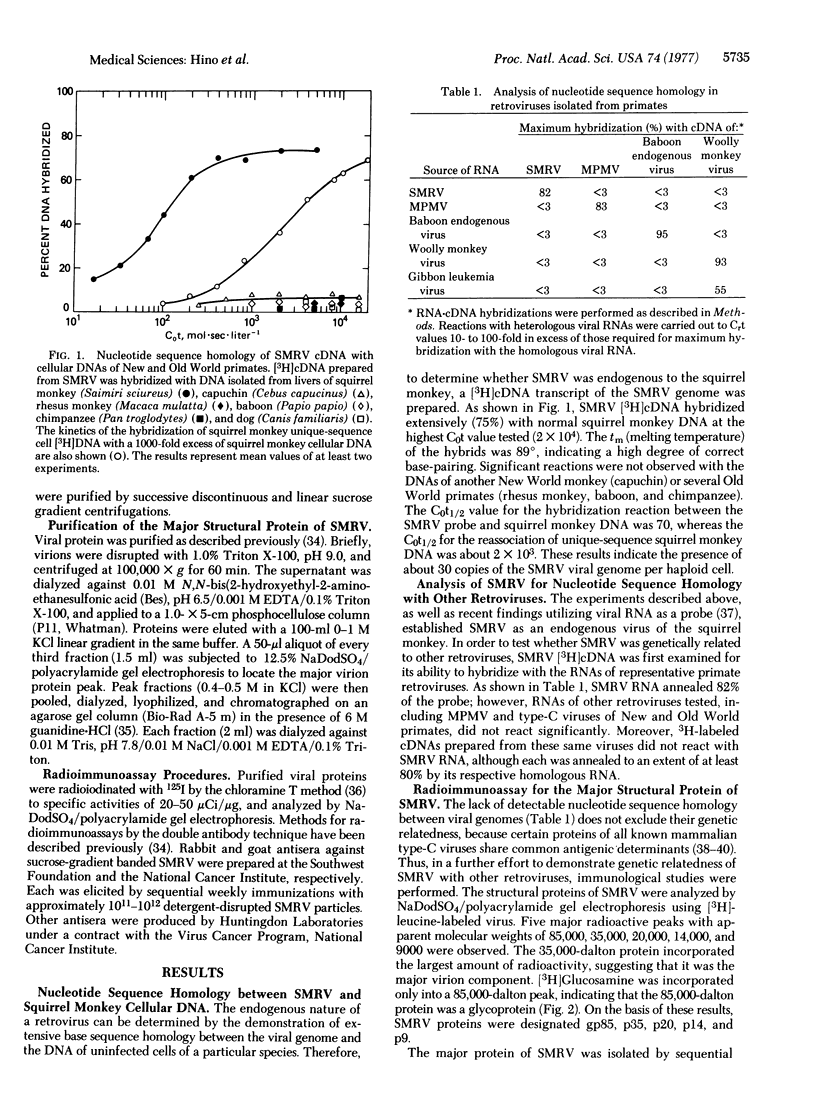
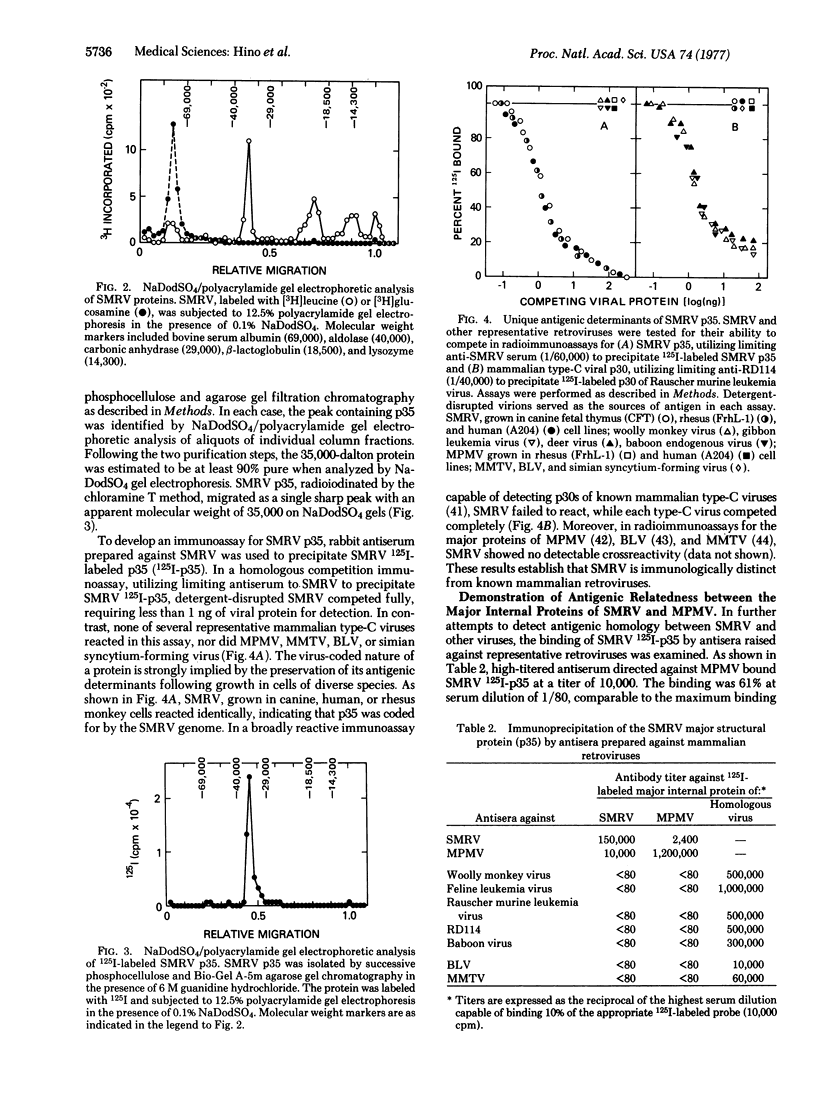
A reverse transcriptase-containing virus has recently been isolated from a squirrel monkey (Saimiri sciureus). Molecular hybridization studies demonstrate that the squirrel monkey retrovirus (SMRV) is endogenous to this New World primate, yet lacks detectable nucleotide sequence homology with cellular DNAs of representative Old World primates or with the genomes of previously isolated Old World primate retroviruses. The 35,000-dalton major structural protein (p35) of SMRV was purified and shown to possess antigenic determinants distinct from those of known retroviruses. While SMRV was found to lack antigenic determinants broadly shared among mammalian type-C viruses, immunologic crossreactivity was demonstrated between SMRV p35 and the major structural protein (p26) of Mason-Pfizer monkey virus, a prototype type-D retrovirus of Old World monkeys. These findings support the concept that SMRV and Mason-Pfizer monkey virus are evolutionarily related, and raise the possibility that a progenitor of type-D retroviruses became genetically associated with primates at a very early time in their evolution.
Full text
PDF


Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Aaronson S. A., Hartley J. W., Todaro G. J. Mouse leukemia virus: "spontaneous" release by mouse embryo cells after long-term in vitro cultivation. Proc Natl Acad Sci U S A. 1969 Sep;64(1):87–94. doi: 10.1073/pnas.64.1.87. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aaronson S. A., Stephenson J. R. Endogenous type-C RNA viruses of mammalian cells. Biochim Biophys Acta. 1976 Dec 23;458(4):323–354. doi: 10.1016/0304-419x(76)90006-8. [DOI] [PubMed] [Google Scholar]
- Aaronson S. A., Todaro G. J., Scolnick E. M. Induction of murine C-type viruses from clonal lines of virus-free BALB-3T3 cells. Science. 1971 Oct 8;174(4005):157–159. doi: 10.1126/science.174.4005.157. [DOI] [PubMed] [Google Scholar]
- Barbacid M., Stephenson J. R., Aaronson S. A. Evolutionary relationships between gag gene-coded proteins of murine and primate endogenous type C RNA viruses. Cell. 1977 Apr;10(4):641–648. doi: 10.1016/0092-8674(77)90097-6. [DOI] [PubMed] [Google Scholar]
- Barbacid M., Stephenson J. R., Aaronson S. A. Structural polypeptides of mammalian type C RNA viruses. Isolation and immunologic characterization of a low molecular weight polypeptide, p10. J Biol Chem. 1976 Aug 25;251(16):4859–4866. [PubMed] [Google Scholar]
- Benveniste R. E., Lieber M. M., Livingston D. M., Sherr C. J., Todaro G. J., Kalter S. S. Infectious C-type virus isolated from a baboon placenta. Nature. 1974 Mar 1;248(5443):17–20. doi: 10.1038/248017a0. [DOI] [PubMed] [Google Scholar]
- Benveniste R. E., Scolnick E. M. RNA in mammalian sarcoma virus transformed nonproducer cells homologous to murine leukemia virus RNA. Virology. 1973 Feb;51(2):370–382. doi: 10.1016/0042-6822(73)90436-4. [DOI] [PubMed] [Google Scholar]
- Benveniste R. E., Sherr C. J., Todaro G. J. Evolution of type C viral genes: origin of feline leukemia virus. Science. 1975 Nov 28;190(4217):886–888. doi: 10.1126/science.52892. [DOI] [PubMed] [Google Scholar]
- Benveniste R. E., Todaro G. J. Evolution of primate oncornaviruses: An endogenous virus from langurs (Presbytis spp.) with related virogene sequences in other Old World monkeys. Proc Natl Acad Sci U S A. 1977 Oct;74(10):4557–4561. doi: 10.1073/pnas.74.10.4557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Benveniste R. E., Todaro G. J. Evolution of type C viral genes: I. Nucleic acid from baboon type C virus as a measure of divergence among primate species. Proc Natl Acad Sci U S A. 1974 Nov;71(11):4513–4518. doi: 10.1073/pnas.71.11.4513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bittner J. J. THE MILK-INFLUENCE OF BREAST TUMORS IN MICE. Science. 1942 May 1;95(2470):462–463. doi: 10.1126/science.95.2470.462. [DOI] [PubMed] [Google Scholar]
- Chattopadhyay S. K., Lowy D. R., Teich N. M., Levine A. S., Rowe W. P. Evidence that the AKR murine-leukemia-virus genome is complete in DNA of the high-virus AKR mouse and incomplete in the DNA of the "virus-negative" NIH mouse. Proc Natl Acad Sci U S A. 1974 Jan;71(1):167–171. doi: 10.1073/pnas.71.1.167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chopra H. C., Mason M. M. A new virus in a spontaneous mammary tumor of a rhesus monkey. Cancer Res. 1970 Aug;30(8):2081–2086. [PubMed] [Google Scholar]
- Colcher D., Heberling R. L., Kalter S. S., Schlom J. Squirrel monkey retrovirus: an endogenous virus of a new world primate. J Virol. 1977 Aug;23(2):294–301. doi: 10.1128/jvi.23.2.294-301.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Colcher D., Teramoto Y. A., Schlom J. Interspecies radioimmunoassay for the major structural proteins of primate type-D retroviruses. Proc Natl Acad Sci U S A. 1977 Dec;74(12):5739–5743. doi: 10.1073/pnas.74.12.5739. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Devare S. G., Stephenson J. R., Sarma P. S., Aaronson S. A., Charder S. Bovine lymphosarcoma: development of a radioimmunologic technique for detection of the etiologic agent. Science. 1976 Dec 24;194(4272):1428–1430. doi: 10.1126/science.188129. [DOI] [PubMed] [Google Scholar]
- Drohan W., Colcher D., Schochetman G., Schlom J. Distribution of Mason-Pfizer virus-specific sequences in the DNA of primates. J Virol. 1977 Jul;23(1):36–43. doi: 10.1128/jvi.23.1.36-43.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fenner F. The classification and nomenclature of viruses. Summary of results of meetings of the International Committee on Taxonomy of Viruses in Madrid, September 1975. Virology. 1976 Jun;71(2):371–378. doi: 10.1016/0042-6822(76)90364-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fleissner E. Chromatographic separation and antigenic analysis of proteins of the oncornaviruses. I. Avian leukemia-sarcoma viruses. J Virol. 1971 Nov;8(5):778–785. doi: 10.1128/jvi.8.5.778-785.1971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- GREENWOOD F. C., HUNTER W. M., GLOVER J. S. THE PREPARATION OF I-131-LABELLED HUMAN GROWTH HORMONE OF HIGH SPECIFIC RADIOACTIVITY. Biochem J. 1963 Oct;89:114–123. doi: 10.1042/bj0890114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gelb L. D., Milstien J. B., Martin M. A., Aaronson S. A. Characterization of murine leukaemia virus-specific DNA present in normal mouse cells. Nat New Biol. 1973 Jul 18;244(133):76–79. doi: 10.1038/newbio244076a0. [DOI] [PubMed] [Google Scholar]
- Gilden R. V., Long C. W., Hanson M., Toni R., Charman H. P., Oroszlan S., Miller J. M., Van der Maaten M. J. Characteristics of the major internal protein and RNA-dependent DNA polymerase of bovine leukaemia virus. J Gen Virol. 1975 Dec;29(3):305–314. doi: 10.1099/0022-1317-29-3-305. [DOI] [PubMed] [Google Scholar]
- Gilden R. V., Oroszlan S., Huebner R. J. Coexistence of intraspecies and interspecies specific antigenic determinants on the major structural polypeptide of mammalian C-type viruses. Nat New Biol. 1971 May 26;231(21):107–108. doi: 10.1038/newbio231107a0. [DOI] [PubMed] [Google Scholar]
- Goldberg R. J., Scolnick E. M., Parks W. P., Yakovleva L. A., Lapin B. A. Isolation of a primate type-C virus from a lymphomatous baboon. Int J Cancer. 1974 Dec 15;14(6):722–730. doi: 10.1002/ijc.2910140605. [DOI] [PubMed] [Google Scholar]
- Heberling R. L., Barker S. T., Kalter S. S., Smith G. C., Helmke R. J. Oncornavirus: isolation from a squirrel monkey (Saimiri sciureus) lung culture. Science. 1977 Jan 21;195(4275):289–292. doi: 10.1126/science.63993. [DOI] [PubMed] [Google Scholar]
- Hino S., Stephenson J. R., Aaronson S. A. Radiommunoassays for the 70,000-molecular-weight glycoproteins of endogenous mouse type C viruses: viral antigen expression in normal mouse tissues and sera. J Virol. 1976 Jun;18(3):933–941. doi: 10.1128/jvi.18.3.933-941.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huebner R. J., Kelloff G. J., Sarma P. S., Lane W. T., Turner H. C., Gilden R. V., Oroszlan S., Meier H., Myers D. D., Peters R. L. Group-specific antigen expression during embryogenesis of the genome of the C-type RNA tumor virus: implications for ontogenesis and oncogenesis. Proc Natl Acad Sci U S A. 1970 Sep;67(1):366–376. doi: 10.1073/pnas.67.1.366. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jensen E. M., Zelljadt I., Chopra H. C., Mason M. M. Isolation and propagation of a virus from a spontaneous mammary carcinoma of a rhesus monkey. Cancer Res. 1970 Sep;30(9):2388–2393. [PubMed] [Google Scholar]
- Kawakami T. G., Huff S. D., Buckley P. M., Dungworth D. L., Synder S. P., Gilden R. V. C-type virus associated with gibbon lymphosarcoma. Nat New Biol. 1972 Feb 9;235(58):170–171. doi: 10.1038/newbio235170a0. [DOI] [PubMed] [Google Scholar]
- Krakower J. M., Barbacid M., Aaronson S. A. Radioimmunoassay for mammalian type C viral reverse transcriptase. J Virol. 1977 May;22(2):331–339. doi: 10.1128/jvi.22.2.331-339.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lasfargues E. Y., Kramarsky B., Sarkar N. H., Lasfargues J. C., Moore D. H. An established R3 mouse mammary tumor cell line; kinetics of mammary tumor virus (MTV) production. Proc Soc Exp Biol Med. 1972 Jan;139(1):242–247. doi: 10.3181/00379727-139-36119. [DOI] [PubMed] [Google Scholar]
- Leong J. A., Garapin A. C., Jackson N., Fanshier L., Levinson W., Bishop J. M. Virus-specific ribonucleic acid in cells producing rous sarcoma virus: detection and characterization. J Virol. 1972 Jun;9(6):891–902. doi: 10.1128/jvi.9.6.891-902.1972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lowy D. R., Rowe W. P., Teich N., Hartley J. W. Murine leukemia virus: high-frequency activation in vitro by 5-iododeoxyuridine and 5-bromodeoxyuridine. Science. 1971 Oct 8;174(4005):155–156. doi: 10.1126/science.174.4005.155. [DOI] [PubMed] [Google Scholar]
- Olson C., Miller L. D., Miller J. M., Hoss H. E. Transmission of lymphosarcoma from cattle to sheep. J Natl Cancer Inst. 1972 Nov;49(5):1463–1467. [PubMed] [Google Scholar]
- Parks W. P., Howk R. S., Scolnick E. M., Oroszlan S., Gilden R. V. Immunochemical characterization of two major polypeptides from murine mammary tumor virus. J Virol. 1974 Jun;13(6):1200–1210. doi: 10.1128/jvi.13.6.1200-1210.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parks W. P., Livingston D. M., Todaro G. J., Benveniste R. E., Scolnick E. M. Radioimmunoassay of mammalian type C viral proteins. 3. Detection of viral antigen in normal murine cells and tissues. J Exp Med. 1973 Mar 1;137(3):622–635. doi: 10.1084/jem.137.3.622. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parks W. P., Todaro G. J., Scolnick E. M., Aaronson S. A. RNA dependent DNA polymerase in primate syncytium-forming (foamy) viruses. Nature. 1971 Jan 22;229(5282):258–260. doi: 10.1038/229258a0. [DOI] [PubMed] [Google Scholar]
- Schincariol A. L., Joklik W. K. Early synthesis of virus-specific RNA and DNA in cells rapidly transformed with Rous sarcoma virus. Virology. 1973 Dec;56(2):532–548. doi: 10.1016/0042-6822(73)90056-1. [DOI] [PubMed] [Google Scholar]
- Stephenson J. R., Aaronson S. A. Endogenous C-type viral expression in primates. Nature. 1977 Mar 31;266(5601):469–472. doi: 10.1038/266469a0. [DOI] [PubMed] [Google Scholar]
- Stephenson J. R., Tronick S. R., Reynolds R. K., Aaronson S. A. Isolation and characterization of C-type viral gene products of virus-negative mouse cells. J Exp Med. 1974 Feb 1;139(2):427–438. doi: 10.1084/jem.139.2.427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Theilen G. H., Gould D., Fowler M., Dungworth D. L. C-type virus in tumor tissue of a woolly monkey (Lagothrix spp.) with fibrosarcoma. J Natl Cancer Inst. 1971 Oct;47(4):881–889. [PubMed] [Google Scholar]
- Tronick S. R., Golub M. M., Stephenson J. R., Aaronson S. A. Distribution and expression in mammals of genes ralated to an endogenous type C RNA virus of Odocoileus hemionus. J Virol. 1977 Jul;23(1):1–9. doi: 10.1128/jvi.23.1.1-9.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tronick S. R., Stephenson J. R., Aaronson S. A. Immunological properties of two polypeptides of Mason-Pfizer monkey virus. J Virol. 1974 Jul;14(1):125–132. doi: 10.1128/jvi.14.1.125-132.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]


