Abstract
Catalytically active mouse beta-glucuronidase (beta-D-glucuronide glucuronosohydrolase, EC 3.2.1.31) is formed when Xenopus oocytes are injected with mouse RNA enriched for poly(A)-containing mRNA sequences. With the RNA from androgen-induced kidneys, the efficiency of translation is comparable to that of endogenous Xenopus messenger, and the fidelity of translation is high. Detection of glucuronidase messenger by formation of a catalytically active product is several orders of magnitude more sensitive than detection by incorporation of isotopically labeled amino acids. As well as providing a sensitive technique for examining the regulation of gene expression, the system makes available an opportunity to study the regulation of post-translational polypeptide processing of a lysosomal enzyme.
Full text
PDF
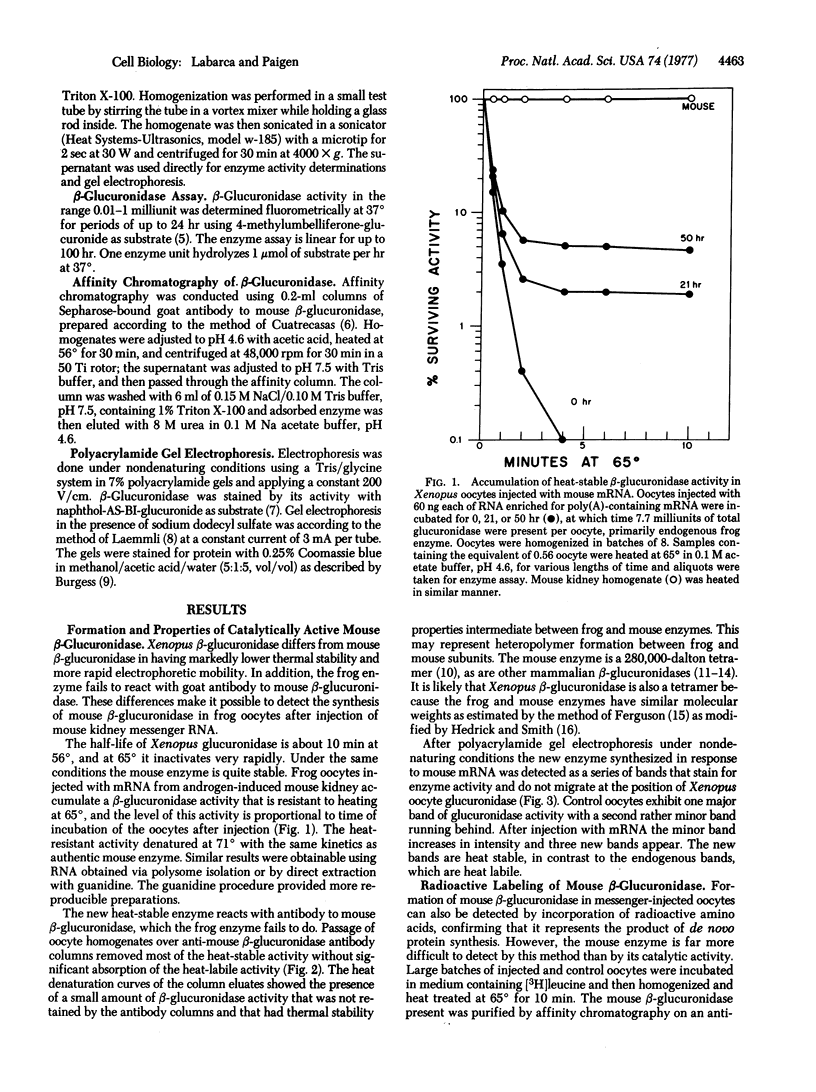
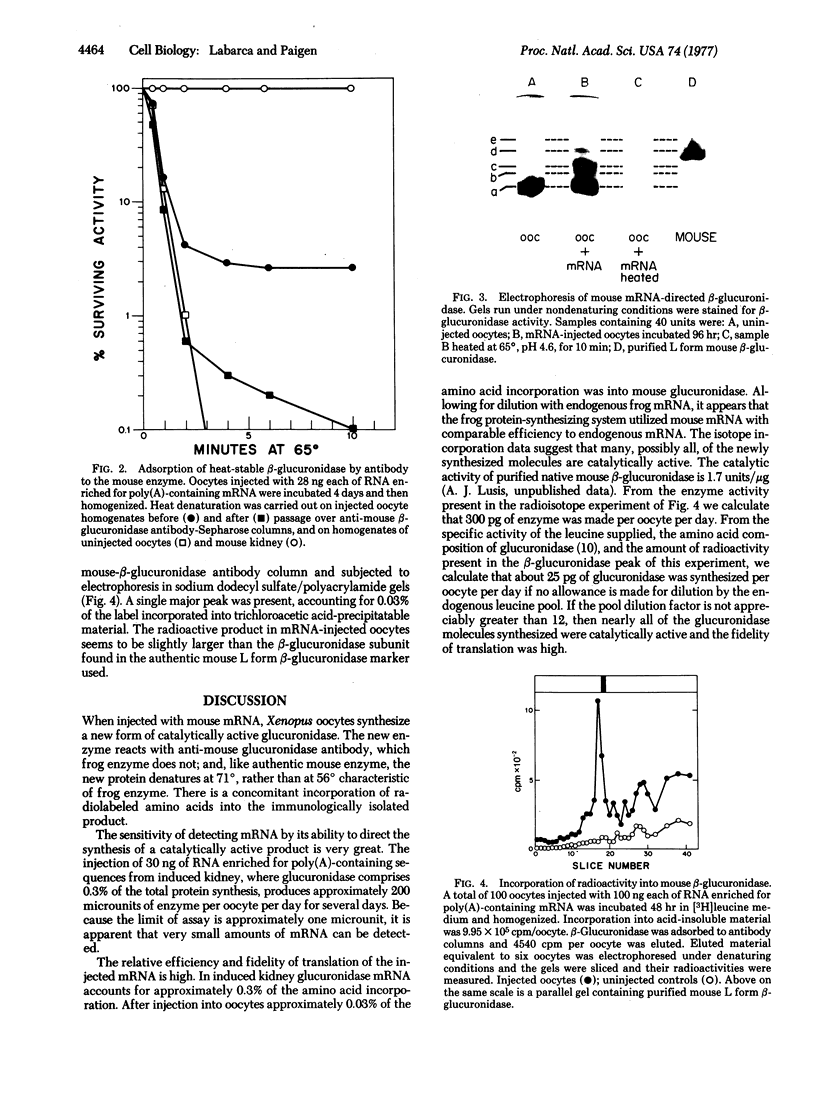

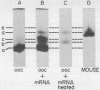
Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Burgess R. R. Separation and characterization of the subunits of ribonucleic acid polymerase. J Biol Chem. 1969 Nov 25;244(22):6168–6176. [PubMed] [Google Scholar]
- Cuatrecasas P. Protein purification by affinity chromatography. Derivatizations of agarose and polyacrylamide beads. J Biol Chem. 1970 Jun;245(12):3059–3065. [PubMed] [Google Scholar]
- Dean R. T. Rabbit beta-glucuronidase. Purification and properties, and the existence of multiple forms. Biochem J. 1974 Mar;138(3):395–405. doi: 10.1042/bj1380395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- FERGUSON K. A. STARCH-GEL ELECTROPHORESIS--APPLICATION TO THE CLASSIFICATION OF PITUITARY PROTEINS AND POLYPEPTIDES. Metabolism. 1964 Oct;13:SUPPL–SUPPL1002. doi: 10.1016/s0026-0495(64)80018-4. [DOI] [PubMed] [Google Scholar]
- Hastie N. D., Bishop J. O. The expression of three abundance classes of messenger RNA in mouse tissues. Cell. 1976 Dec;9(4 Pt 2):761–774. doi: 10.1016/0092-8674(76)90139-2. [DOI] [PubMed] [Google Scholar]
- Hedrick J. L., Smith A. J. Size and charge isomer separation and estimation of molecular weights of proteins by disc gel electrophoresis. Arch Biochem Biophys. 1968 Jul;126(1):155–164. doi: 10.1016/0003-9861(68)90569-9. [DOI] [PubMed] [Google Scholar]
- Keller R. K., Touster O. Physical and chemical properties of beta-glucuronidase from the preputial gland of the female rat. J Biol Chem. 1975 Jun 25;250(12):4765–4769. [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Lusis A. J., Tomino S., Paigen K. Isolation, characterization, and radioimmunoassay of murine egasyn, a protein stabilizing glucuronidase membrane binding. J Biol Chem. 1976 Dec 25;251(24):7753–7760. [PubMed] [Google Scholar]
- MUSA B. U., DOE R. P., SEAL U. S. PURIFICATION AND PROPERTIES OF HUMAN LIVER BETA-GLUCURONIDASE. J Biol Chem. 1965 Jul;240:2811–2816. [PubMed] [Google Scholar]
- Paigen K., Swank R. T., Tomino S., Ganschow R. E. The molecular genetics of mammalian glucuronidase. J Cell Physiol. 1975 Apr;85(2 Pt 2 Suppl 1):379–392. doi: 10.1002/jcp.1040850406. [DOI] [PubMed] [Google Scholar]
- Plapp B. V., Cole R. D. Demonstration and partial characterization of multiple forms of bovine liver beta-glucuronidase. Biochemistry. 1967 Dec;6(12):3676–3681. doi: 10.1021/bi00864a009. [DOI] [PubMed] [Google Scholar]
- Swank R. T., Paigen K. Biochemical and genetic evidence for a macromolecular -glucuronidase complex in microsomal membranes. J Mol Biol. 1973 Jul 5;77(3):371–389. doi: 10.1016/0022-2836(73)90445-2. [DOI] [PubMed] [Google Scholar]
- Tominco S., Paigen K. Egasyn, a protein complexed with microsomal beta-glucuronidase. J Biol Chem. 1975 Feb 10;250(3):1146–1148. [PubMed] [Google Scholar]
- Tomino S., Paigen K., Tulsiani D. R., Touster O. Purification and chemical properities of mouse liver lysosomal (L form) beta-glucuronidase. J Biol Chem. 1975 Nov 10;250(21):8503–8509. [PubMed] [Google Scholar]